Abstract
Land cover conversion intensively occurred in the Tibetan Plateau of China during the past decades. However, responses of soil properties and soil microbial activities to land cover conversion under different land cover types have not been fully understood. The objective was to assess the effects of land cover conversion on soil C and N stocks and soil microbial properties of topsoil of an alpine meadow in the Tibetan Plateau. Soil cores of surface soil (0–20 cm) were collected from three adjacent land cover types: native alpine meadow, artificial grassland and mound-shaped denuded land. Soil microbial metabolic diversity was measured using Biolog EcoPlates. Significant losses of SOC and TN storages were subject to land cover conversion in topsoil, decreasing 14.5 and 10.9 % of alpine meadow to artificial grassland and 52.9 and 51.7 % to denuded land. Alpine meadow soils had the highest soil microbial biomass and microbial metabolic activity of three land cover types. Microbial biomass and microbial metabolic activity decreased markedly after alpine meadow conversion to denuded land. Land use conversion from original alpine meadow to artificial grassland and denuded land caused decline in soil organic carbon density, total nitrogen density and soil microbial activity. Soil carbon emissions from alpine meadow conversion were released to the atmosphere and could influence climatic change.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Land use and land cover changes are considered as the main components and a primary cause of global environmental changes (Turner et al. 1995). Changes in land use lead to changes in land cover, which affect the provision of ecosystem services and wildlife habitat (Lawler et al. 2014). Comprehensive knowledge of these dynamics may be useful to reconstruct past land cover changes and also predict future changes (Hietel et al. 2004). Specifically, conversion of native vegetation to other ecosystems is widely reported as an important disturbance, which may result in changes in soil fertility and plant growth. Different soil status can alter the structure and dynamics of soil microbial communities (Ding et al. 2013). Furthermore, such changes can lead to a shift of the soil–atmosphere carbon balance leading to further climatic change (Fernandez and Kennedy 2015). Together, land cover change can affect soil physical, chemical and biological properties (Braimoh and Vlek 2004; Ross et al. 1999; Zeng et al. 2009; Zhao et al. 2013).
The Qinghai–Tibetan Plateau (QTP) ecosystem is more fragile and sensitive to global climate change and anthropogenic disturbance than are other ecosystems because of its high elevation (Wang et al. 2007; Yang et al. 2006). Alpine meadows, the representative vegetation of Tibetan Plateau, cover approximately 6.37 × 105 km2 (Ni 2002), comprising 15.3 % of the cold region (Yang et al. 2013). The alpine meadow ecosystem is of special interest, because its soils bear a larger organic carbon pool than grassland soils in other regions of China or tropical savannah soils (Genxu et al. 2008). Colder soils have been shown to carry higher organic C loads than warmer soils (Mcdaniel and Munn 1985). However, there had been dramatic changes of vegetation cover in alpine meadows due to overgrazing, human activities and rodent damage (Zhou et al. 2005). The changes of land cover not only alter ecosystem communities, but also alter the nutrient of the soil and the activities of soil microorganism (Wang et al. 2006a; Zhang et al. 2013). Hence, ecosystem alteration causes changes in C and N cycling by altering plant production, rates of soil organic matter accumulation and decomposition, and the subsequent C and N storage in soils (Wang et al. 2009). Meanwhile, the alpine grassland ecosystem bearing large soil C stocks can became a source of atmospheric CO2 if grassland soils become degraded. Huge amounts of C have been emitted from the grassland soils of the plateau due to grassland degradation, which strengthens the trend of global warming (Wang et al. 2002).
Previous studies have reported differences in SOC or TN stocks among typical vegetation types in the northeastern margin of the QTP (Liu et al. 2012). The northeastern margin of the QTP is mainly influenced by the Asian monsoon system and the geomorphological situation is consistent with mountain topography. Unique alternating mountain basin landforms in the area form a series of relative isolated and delimited land use units. With economic development and population growth, the alpine meadow had suffered severe degradation due to natural and anthropogenic activities (Harris 2010). Wang et al. (2003) found that organic matter content and the total N content of the alpine meadow soil decreased by 14,890 and 5505 kg/hm2, respectively, as the vegetation coverage reduced from 90 % to less than 30 %. Meanwhile, Li et al. (2009) showed that mound making by zokors had negative impact on properties and organic matter content of the topsoil at an alpine grassland site of the QTP. On the other hand, Wang et al. (2006b) found that rehabilitation of degraded grassland ecosystems promoted above-ground biomass, particularly grass biomass, and ground cover, which could increase soil organic C and N stocks. The potential for soil carbon and nitrogen storage in alpine meadow soils after land cover change did not appear to have been widely investigated.
Soil microorganisms control ecosystem processes such as C storage and nutrient recycling (Talbot et al. 2014). They play key roles in ecosystem functioning and in the sustainability of soil health. Microbial-based indicators, such as microbial biomass and community structure, can be useful ecological indicators of stress caused by anthropogenic activities (Wardle 1992). While the fluctuations of the soil microbial biomass pools are thought to be important drivers in affecting soil carbon and nutrient cycling, community-level physiological profiling can effectively distinguish spatial and temporal changes in microbial communities (Garland and Mills 1991). Changes in microbial communities often precede changes in the health and viability of the whole environment. Although the Biolog assay has been used to assess the metabolic characteristics of microbial communities (Zak et al. 1994), it has several limitations (Nannipieri et al. 2003). For example, it cannot reflect the potential metabolic diversity in situ and detects only culturable bacteria. However, it is useful for studying the functional diversity of soil bacterial communities in different habitats (Jiang et al. 2010).
In this study, land cover mosaics of alpine meadow, artificial grassland and denuded land were characteristic of the alpine areas on the Tibetan Plateau in Qinghai Province, northwest China. Soil characteristics and microbial properties were compared among three land cover types in an alpine meadow on the Tibetan Plateau. The specific objectives within these three land cover types were: (1) to compare soil carbon and nitrogen stocks of topsoil and (2) to evaluate the variations of soil microbial properties in an alpine meadow. The results provided key data for the biogeochemical characteristics in the protection of alpine grassland ecosystems of the Qinghai–Tibetan Plateau, China.
Materials and methods
Site characteristics
The study was conducted at the Qilian Alpine Ecology and Hydrology Research Station, Chinese Academy of Sciences (Fig. 1). The study area was located at latitude 38°15′N and longitude 99°52′E in the northeast part of the Qinghai–Tibetan Plateau. The average altitude was about 3000 m above sea level. The annual air temperature was approximately 0.2 °C, with the highest mean monthly temperature reaching 19.0 °C in July and the lowest reaching −18.4 °C in January. The mean annual precipitation was 495.1 mm, with 80–93 % of the precipitation occurring from June to October. Native vegetation was mainly alpine Kobresia meadow. The soil type in the study area was mainly classified into mattic cryic cambisoils (Chinese soil taxonomy: alpine meadow soil; FAO: cambisoils). The most significant characteristic was that there were mattic epipedon in alpine meadow soil.
Experimental design, soil sampling and laboratory analysis
Three land cover types, alpine meadow, artificial grassland and denuded land, were selected for this study. Three plots (30 × 40 m) were selected for each land cover type. The plots with different land cover types were located as close as possible to each other, in plates with comparable topography and soil and vegetable types (which appeared to be present before land cover change). The topography of the study area was undulating with flat to almost flat zones. Due to alteration of original thick sod layer of depth 0–20 cm during land cover conversion, the study about the effect of soil properties and soil microbial activity was carried out at 0–20 cm depth.
-
1.
The native alpine Kobresia meadow was undisturbed as control and contained approximately 20 species that together made up 80 % of the total vegetation cover. The dominant species were Kobresia capillifolia and Kobresia humilis, and the main accompanying species were Festuca ovina, Stipa capillata, Potentilla fragarioides, Elymus nutans, Poa annua, Leontopodium nanum, Gentiana lawrencei, Achnatherum splendens, Saussurea superba, and Carex sp. The soil surface was well developed with a mixture of root mass and the root systems were densely distributed with rich organic matter.
-
2.
The mound-shaped denuded land was heavily degraded grassland with little or no vegetation. This was representative of secondary bare land or heavily damaged turf landscape that occurs after degradation of the original vegetation (Zhou et al. 2005). The severely degraded grassland was characterized by low vegetation cover and the loss of the upper soil horizons. The mounds were mainly made of Myospalax fontanierii and Marmota himalayana. They made mounds by digging, wriggling and mixing soil while foraging the plants. All selected mounds were intact since their origin and had a size of about 0.16 m2. On each replicate, five soil slices of mound surface (10 × 5 cm in area and 20 cm in depth) were randomly collected and mixed.
-
3.
The artificial grassland was a field of oat (Avena sativa), which had been converted from alpine meadow 6 years before. The oat stubble coverage was about 25 %. Crops were routinely planted in mid-May and harvested in late September; thus land was utilized 4 months per year. In this area, oats were cultivated as forage and hay crop without mechanization or pesticide treatments. To establish artificial grassland, farmers sow oat seeded with a manual dibble and did not apply fertilizers. After harvest, the oat stubble was left in the field and plowed into the soil as manure.
Soil samples were collected from the three land cover types in January 2013, when the oat crop had been harvested and rodents had the slowest digging activities. In each plot, the litter layer was removed and then five soil cores (3.8 cm diameter × 20 cm long) were randomly collected from the 0–20 cm layer and mixed to obtain a homogenous sample. A portion of each soil sample was air dried for analyses of soil properties. The remaining portion of each sample was stored at 4 °C until soil microbial biomass and Biolog analyses.
Analyses of soil properties
Soil moisture content (SMC) was determined by drying the soils at 105 °C for 24 h. Soil pH was measured in a soil:water mixture (1:2.5 w/v) using a PB-21 pH detector (Sartorius, Göttingen, Germany). Soil electrical conductivity (EC) was measured in a soil:water mixture (1:5 w/v) using a DDSJ-308A electrical conductivity analyzer (Shanghai Rex Instrument Co. Ltd., Shanghai, China). Soil bulk density (BD) was measured for the soil cores using a cutting ring (volume, 100 cm3); this allowed us to estimate the density of soil organic carbon and total nitrogen at each site. Soil total organic C (SOC) was determined by the dichromate oxidation method. Total nitrogen (TN) was determined by the semimicro Kjeldahl method. To measure total phosphorus (TP), the soil was digested with HF–HClO4 and then TP was determined by the molybdenum blue colorimetric method. Available phosphorus (AP) was measured after extraction by the molybdenum antimony resistance colorimetric method (Lu 1999). To measure NH4+-N and NO3−-N, 10 g dry weight of soil samples was suspended in 50 ml of 2 mol/L KCL solution. After shaking at room temperature for 1 h and subsequent standing for 30 min, the supernatant was filtered through a filter paper. NH4+-N and NO3−-N were analyzed by a continuous flow analyzer (SKALAR, Dutch).
Determinations of SOC and TN density
The gravel content (>2 mm) of alpine meadow was approximately 10 % (C AM = 0.10), of artificial grassland approximately 5 % (C AR = 0.05) and of mound-shaped denuded land approximately 15 % (C DL = 0.15). The topsoil thickness was 20 cm. SOCD and TND were calculated density for topsoil profile (T = 20) using the following equations (Yang et al. 2008):
where SOCD and TND are SOC density (kg/m2) and TN density (kg/m2), respectively; and T, BD, SOC and C are soil thickness (cm), bulk density (g/cm3), soil organic carbon (g/kg) and volume percentage of the >2 mm fraction, respectively. TN density of topsoil was described analogously.
Analysis of microbial biomass C and N
Soil microbial biomass C and N (SMBC/SMBN) were determined using the chloroform fumigation extraction method (Brookes et al. 1985; Vance et al. 1987). A K EC of 0.45 (Vance et al. 1987) and a K EN of 0.54 (Brookes et al. 1985) were used to account for the extraction efficiency of C and N, respectively. The values were calculated using the following equations:
where E C and E N are the differences between the C and N concentrations in fumigated and non-fumigated extracts and \(K_{{{\text{EC}} }} = 0. 4 5 {\text{ and }}K_{{\text{EN}} }= 0. 5 4.\)
Soil microbial metabolic diversity
Biolog-ECO microplates (BIOLOG Inc., Hayward, CA, USA) were used to determine the metabolic diversity of microbial communities following a procedure adapted from Garland and Mills (1991). The 31 substrates represented six classes of organic compounds: amides/amines, amino acids, carbohydrates, carboxylic acids, polymers and miscellaneous substrates (Insam 1997).
Briefly, fresh soil samples (10 g fresh weight) were serially diluted to a 10−3 suspension in buffer (0.85 % NaCl; w/v) before inoculation onto Biolog plates (150 μL soil suspension per well). The plates were incubated at 25 °C in the dark and scanned every 12 h at 590 nm for 8 days using an Emax precision microplate reader (BIOLOG Inc.). The data were analyzed by Microlog Release 4.2 software (ML3420, Microlog, USA). The absorbance values recorded at 96 h were used to calculate the diversity indices and in the principal component analysis (PCA). In this study, the average well color development (AWCD) was calculated as follows:
where C i refers to the 31 absorbance values of the different C sources and R is the absorbance value of the control well. The absorbance values recorded at 96 h were used to calculate the Shannon–Wiener diversity index (Zak et al. 1994) as follows:
Statistical analysis
All statistical analyses were carried out with SPSS 11.5 software for Windows. All data were shown as mean values ± standard error. One-way analysis of variance (one-way ANOVA) was used to calculate the statistical significance. The least significant difference (LSD) test was performed for post hoc multiple comparisons if the data were homogeneous, or otherwise Tamhane’s T2 test was performed. A Pearson’s correlation analysis was carried out among variables using the bivariate correlations procedure. Principal component analyses (PCA) were conducted on the BIOLOG data to investigate the differences in soil microbial metabolic diversity of three land cover types. Differences were considered to be significant if P was <0.05.
Results and discussion
Soil characteristics
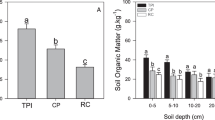
The physical and chemical properties of the tested soils in the top 20 cm were listed in Table 1. Artificial grassland had much lower soil pH (P < 0.05), while denuded land had higher values compared to the reference alpine meadow. However, no significant differences in soil pH were found between denuded land and alpine meadow. Soil pH values of >7 indicated alkaline conditions at most sampling sites in the study area. Lower soil moisture content was found for denuded land than for alpine meadow and artificial grassland (P < 0.01) due to less coverage and higher evaporation. Soil electrical conductivities were greater in artificial grassland than in alpine meadow and denuded land, whereas they did not differ between artificial grassland and denuded land (P ≥ 0.05). Generally, the average soil electrical conductivities were much greater in artificial grassland and denuded land than in alpine meadow as a result of greater evaporation rates from more bare soil surfaces, which facilitated the upward flow of soluble salts from deep layers with groundwater, thus increasing soil electrical conductivity. Average soil bulk density was greater in alpine meadow than in artificial grassland and denuded land (P < 0.05). SOC in different land cover types also followed the order: alpine meadow > artificial grassland > denuded land, even though there were no significant differences in SOC between alpine meadow and artificial grassland.
Soil characteristics provided a medium for root development and moisture and nutrient for plant growth. Alteration of original alpine meadow to the other land cover types significantly changed vegetation composition and soil status. Especially in the cold season, it was not covered with vegetation, which left the soil bare and prone to erosion (Wu et al. 2010). Conservation of the original alpine grassland brought about loss in SOC in Northern China (Wu and Tiessen 2002). In this study, the average SOC in artificial grassland and denuded land decreased 9.54 and 30.20 %, respectively, compared to alpine meadow plots. In general, perennial plants (Kobresia capillifolia and Kobresia humilis) stored more C in soil than annuals (Avena sativa), because the annual cycle of cultivation did little to maintain storage because perennial crops were in the ground longer and intercepted more of the sun’s radiation than annuals (Whitmore et al. 2014). On the other hand, the constant additions of below-ground biomass by perennials roots was also attributed to the SOC accumulation in alpine meadow plots. Comparatively, foraging and mounding creating by rodent animals significantly reduced the below-ground plant biomass (Li et al. 2009). Therefore, more carbon would be released into the atmosphere in denuded land plots than the other land cover types (Wang et al. 2010).
Abundance of nitrogen and phosphorus
For three land cover types, soil total nitrogen in the alpine meadow was higher than in the artificial grassland and denuded land. Soil total nitrogen contents in alpine meadow and denuded land soil showed significant differences (P < 0.05) (Table 2). Only subtle changes in soil total phosphorus contents were observed among the three land cover types. Soil NH4+ showed similar tendencies. However, artificial grassland soil contained higher soil NO3− and available phosphorus than the other land cover types. This could be explained by the fact that soil inorganic nutrients were released back into soil due to soil microbial decomposition of oat residues.
Inorganic N (NO3− + NH4+) concentration ranged from 19.94 to 45.11 mg N kg/soil. There was a trend toward higher inorganic nutrient concentrations in plots with more existent dead plant roots, though no significant differences were observed in soil TN between alpine meadow and artificial grassland plots. Thus, short-term relatively small amendment applications could alter soil biological processes and nutrient cycling (Ninh et al. 2015).
Carbon and nitrogen stocks
Variations in C and N stocks exhibited similar tendencies with C and N contents (Fig. 2). As values of bulk density were distinct at 0–20 cm depth across land cover types (Table 1), gaps of C and N contents in topsoil were narrowed as compared to C and N stocks. SOC and TN stocks presented declines when original alpine meadow changes. The SOCD and TND in artificial grassland soils were characterized by a slight drop within a 14.5 and a 10.9 % decrease, respectively, as compared to original alpine meadow soils. Meanwhile, a great decrement in SOCD and TND was observed in denuded land soils as compared to alpine meadow soils, within a 52.9 and a 51.7 % decrease, respectively. The data indicated a greater decline in SOCD and TND of denuded land spots.
The amount of C retained by soils was influenced greatly by the rate of input of organic matter and its decomposition by biological or chemical means (Whitmore et al. 2014). Losses of SOC and TN storage were subjected to land cover conversion in topsoil, decreasing 14.5 and 10.9 % of alpine meadow to artificial grassland and 52.9 and 51.7 % to denuded land. Guo and Gifford (2002) showed that a change from perennial grassland to arable land was likely to lead to a significant loss in soil C. The development of artificial grasslands in place of current perennial plants might lead to loss in soil C, which was consistent with the results of Qin et al. (2014). The activities of rodents accelerated erosion and the degradation rate by loosening the Kobresia sod and killing its roots (Zhou et al. 2005). This result coincided with the finding that highlighted mound making had negative impact on soil properties and organic matter content of the topsoil (Li et al. 2009).
Soil microbial biomass C and N
Although microbial biomass represented <5 % of SOM, the contribution of microbial-derived compounds to SOM was potentially much greater (Grandy et al. 2009). SMBC and SMBN differed among land cover types (Fig. 3). SMBC values in the alpine meadow were higher than in denuded land (P < 0.05), whereas they did not differ between alpine meadow and artificial grassland (P ≥ 0.05). SMBN in the alpine meadow was significantly higher than in the artificial grassland and denuded land (P < 0.05). A series of bivariate correlations analyses showed that soil MBC and MBN were strongly related to soil bulk density and soil organic carbon (Table 3). Microbial biomass was a sensitive indicator of soil disturbances caused by land cover changes (Melo et al. 2012). Many factors were considered to elucidate the effects of vegetation types on soil microbial biomass (Wardle 1992). The quantity and quality of substrates via leaf litter inputs and root exudations influenced soil carbon and nutrient availability. Soil carbon and nutrient availability were important drivers that affected soil microbial biomass (Wardle 1992), as SMBC was proved to have positive correlations with soil organic carbon and total nitrogen (Table 3). Correspondingly, higher SMBC and SMBN in the original alpine meadow than in the artificial grassland and denuded land were mainly attributed to higher soil organic carbon and total nitrogen. Furthermore, soil moisture might play an important role in influencing microbial biomass, as soil moisture was positively correlated with microbial biomass C. Less soil moisture in the denuded land than the other land cover types could suppress the growth and activity of soil microbial communities. Soil microorganisms could adopt a survival strategy for coping with harsh environmental conditions, such as sporulation and cell shrinkage (Jones and Lennon 2010). This result provided evidence for a shift of microbial biomass among three land cover types.
Community-level physiological profiles (CLPP) analysis
The AWCD values obtained from Biolog Ecoplates could be used to reflect the microbial metabolic activity (Zheng et al. 2005). The highest AWCD values were obtained from alpine meadow soils and the lowest from denuded land soils. All of the AWCD values were low after 24 h incubation, indicating that the carbon sources were not fully used during that period. However, the AWCD values for alpine meadow soil increased rapidly after 24 h, while those for artificial grassland and denuded land soils increased more slowly (Fig. 4). The three land cover types could be ranked in terms of their Shannon–Wiener diversity index (H) values as follows: denuded land < artificial grassland < alpine meadow.
The results of the PCA of Biolog data are shown in Table 4 and Fig. 5. The first principal component (PC 1) explained 33.68 % of the variation and the second (PC 2) explained 20.19 % of the variation (eigenvalues 10.44 and 6.26, respectively). Axis 1 and 2 explained 53.87 % of the variation in soil microbial community metabolic diversity. For three land cover types, the PC 2 could not clearly separate the denuded land soil samples from the alpine meadow soil samples. However, PC 1 clearly divided the substrate utilization patterns of the microbial community between alpine meadow and artificial grassland. The variability in PC1 was explained by the different use of carbohydrates, carboxylic acids, amino acids, miscellaneous substrates and polymers (β-methyl-d-glucoside, d-xylose, 2-hydroxy benzoic acid, α-cyclodextrin, N-acetyl-d-glucosamine, γ-hydroxybutyric acid, l-threonine, glycogen, glycyl-l-glutamic acid, d-cellobiose, glucose-1-phosphate, α-ketobutyric acid, α-d-lactose, d-malic acid). The variability in PC 2 was also explained by different use of carbohydrates, carboxylic acids, amino acids, polymers and miscellaneous substrates (d-galactonic acid γ-lactone, d-galacturonic acid, l-asparagine, Tween 80, d-mannitol, 4-hydroxy benzoic acid, l-serine and d-Glucosaminic acid) (loading values of principal components >0.600) (Table 4). The use of carbohydrates by microorganisms was significantly greater in the alpine meadow than in artificial grassland and denuded land (P < 0.05) (Table 5).
The differences in the carbon source metabolism activities of soil microorganisms under different land cover types were mainly caused by soil properties (Chakraborty et al. 2011). As denuded land soils had a limited nutrient content due to less organic matter input, an increase in soil fertility could promote the soil microbial community metabolism activities (Islam et al. 2011). A large amount of SOC could provide C resources to microbes and promoted C utilization (Zhao et al. 2013). Alpine meadow soil had dense root systems, while artificial grassland and denuded land soil had loose and had sparse root systems. The root distribution and its activities might accelerate soil nutrient translation and promoted the soil metabolic activity of soil microbial communities. For example, retaining crop residues in the field and changed in soil organic matter could also affect the metabolic diversity of soil microbial communities (Bending et al. 2002). The spatial heterogeneity of soil factors such as C/N ratio and pH could affect soil microbial functional diversity (Zhang et al. 2013). In the present study, soil electrical conductivity might play an important role in influencing microbial metabolic activity, as soil electrical conductivity was positively correlated with microbial metabolic activity. Salinity (soil electrical conductivity) inhibited microbial activities by affecting the availability of water or the physiological and metabolic processes of the microbial cells (Jannike et al. 2006). Alteration of the original alpine meadow to the other land cover types could reduce the resistance of microbial communities to environmental stress or disturbances (Degens et al. 2001). These results demonstrated that soil microbial community activities in soil exhibited obvious differences among the three land cover types. However, the CLPP values represented only a subpopulation of culturable aerobic microorganisms (Chakraborty et al. 2011). Future research is required to evaluate the structure of microbial communities using genetic analyses.
Conclusions
This paired study for adjacent fields with three land cover types revealed specific differences in soil properties and soil microbial activity. Land cover conversion from native alpine meadow to artificial grassland and denuded land resulted in visible SOC and TN losses in topsoil. Alpine meadow soil had the highest soil microbial biomass and microbial metabolic activity of the three land cover types. The destruction of soil properties, losses of nutrients and rooting layer, owing rodents damaging, could cause long-term damage of land productivity. This study provided implications for assessing effects of land cover conversion on soil C and N stocks and soil microbial properties, and highlighted the importance of protecting alpine meadow ecosystem.
References
Bending GD, Turner MK, Jones JE (2002) Interactions between crop residue and soil organic matter quality and the functional diversity of soil microbial communities. Soil Biol Biochem 34:1073–1082. doi:10.1016/S0038-0717(02)00040-8
Braimoh AK, Vlek PLG (2004) The impact of land-cover change on soil properties in northern Ghana. Land Degrad Dev 15:65–74. doi:10.1002/Ldr.590
Brookes PC, Landman A, Pruden G, Jenkinson DS (1985) Chloroform fumigation and the release of soil-nitrogen—a rapid direct extraction method to measure microbial biomass nitrogen in soil. Soil Biol Biochem 17:837–842. doi:10.1016/0038-0717(85)90144-0
Chakraborty A, Chakrabarti K, Chakraborty A, Ghosh S (2011) Effect of long-term fertilizers and manure application on microbial biomass and microbial activity of a tropical agricultural soil. Biol Fertil Soils 47:227–233. doi:10.1007/s00374-010-0509-1
Degens BP, Schipper LA, Sparling GP, Duncan LC (2001) Is the microbial community in a soil with reduced catabolic diversity less resistant to stress or disturbance? Soil Biol Biochem 33:1143–1153. doi:10.1016/S0038-0717(01)00018-9
Ding GC, Piceno YM, Heuer H, Weinert N, Dohrmann AB, Carrillo A, Andersen GL, Castellanos T, Tebbe CC, Smalla K (2013) Changes of soil bacterial diversity as a consequence of agricultural land use in a semi-arid ecosystem. PLoS One 8:e59497. doi:10.1371/journal.pone.0059497
Fernandez CW, Kennedy PG (2015) Moving beyond the black-box: fungal traits, community structure, and carbon sequestration in forest soils. New Phytol 205:1378–1380. doi:10.1111/Nph.13289
Garland JL, Mills AL (1991) Classification and characterization of heterotrophic microbial communities on the basis of patterns of community-level sole-carbon-source utilization. Appl Environ Microbiol 57:2351–2359
Genxu W, Yuanshou L, Yibo W, Qingbo W (2008) Effects of permafrost thawing on vegetation and soil carbon pool losses on the Qinghai–Tibetan Plateau, China. Geoderma 143:143–152. doi:10.1016/j.geoderma.2007.10.023
Grandy AS, Strickland MS, Lauber CL, Bradford MA, Fierer N (2009) The influence of microbial communities, management, and soil texture on soil organic matter chemistry. Geoderma 150:278–286. doi:10.1016/j.geoderma.2009.02.007
Guo LB, Gifford RM (2002) Soil carbon stocks and land use change: a meta analysis. Glob Change Biol 8:345–360. doi:10.1046/j.1354-1013.2002.00486.x
Harris RB (2010) Rangeland degradation on the Qinghai–Tibetanan plateau: a review of the evidence of its magnitude and causes. J Arid Environ 74:1–12. doi:10.1016/j.jaridenv.2009.06.014
Hietel E, Waldhardt R, Otte A (2004) Analysing land-cover changes in relation to environmental variables in Hesse, Germany. Landscape Ecol 19:473–489. doi:10.1023/B:Land.0000036138.82213.80
Insam H (1997) A new set of substrates proposed for community characterization in environmental samples. Springer, USA
Islam MR, Chauhan PS, Kim Y, Kim M, Sa T (2011) Community level functional diversity and enzyme activities in paddy soils under different long-term fertilizer management practices. Biol Fertil Soils 47:599–604. doi:10.1007/s00374-010-0524-2
Jannike W, Florian W, Georg JR (2006) Impact of salinity on soil microbial communities and the decomposition of maize in acidic soils. Geoderma 137:100–108. doi:10.1016/j.geoderma.2006.08.001
Jiang W, Wang JJ, Tang JS, Hou F, Lu YT (2010) Soil bacterial functional diversity as influenced by cadmium, phenanthrene and degrade bacteria application. Environ Earth Sci 59:1717–1722. doi:10.1007/s12665-009-0153-y
Jones SE, Lennon JT (2010) Dormancy contributes to the maintenance of microbial diversity. Proc Natl Acad Sci 107:5881–5886. doi:10.1073/pnas.0912765107
Lawler JJ, Lewis DJ, Nelson E, Plantinga AJ, Polasky S, Withey JC, Helmers DP, Martinuzzi S, Pennington D, Radeloff VC (2014) Projected land-use change impacts on ecosystem services in the United States. Proc Natl Acad Sci USA 111:7492–7497. doi:10.1073/pnas.1405557111
Li XG, Zhang ML, Li ZT, Shi XM, Ma Q, Long RJ (2009) Dynamics of soil properties and organic carbon pool in topsoil of zokor-made mounds at an alpine site of the Qinghai–Tibetanan Plateau. Biol Fertil Soils 45:865–872. doi:10.1007/s00374-009-0398-3
Liu W, Chen S, Qin X, Baumann F, Scholten T, Zhou Z, Sun W, Zhang T, Ren J, Qin D (2012) Storage, patterns, and control of soil organic carbon and nitrogen in the northeastern margin of the Qinghai–Tibetanan Plateau. Environ Res Lett 7:035401. doi:10.1088/1748-9326/7/3/035401
Lu RK (1999) Analytical methods for soils and agricultural chemistry. China Agricultural Science and Technology Press, Beijing, pp 107–240
Mcdaniel PA, Munn LC (1985) Effect of temperature on organic carbon-texture relationships in mollisols and aridisols. Soil Sci Soc Am J 49:1486–1489. doi:10.2136/sssaj1985.03615995004900060031x
Melo VS, Desjardins T, Silva ML, Santos ER, Sarrazin M, Santos MMLS (2012) Consequences of forest conversion to pasture and fallow on soil microbial biomass and activity in the eastern Amazon. Soil Use Manag 28:530–535. doi:10.1111/Sum.12003
Nannipieri P, Ascher J, Ceccherini MT, Landi L, Pietramellara G, Renella G (2003) Microbial diversity and soil functions. Eur J Soil Sci 54:655–670. doi:10.1046/j.1351-0754.2003.0556.x
Ni J (2002) Carbon storage in grasslands of China. J Arid Environ 50:205–218. doi:10.1006/jare.2201.0902
Ninh HT, Grandy AS, Wickings K, Snapp SS, Kirk W, Hao J (2015) Organic amendment effects on potato productivity and quality are related to soil microbial activity. Plant Soil 386:223–236. doi:10.1007/s11104-014-2223-5
Qin Y, Yi S, Ren S, Li N, Chen J (2014) Responses of typical grasslands in a semi-arid basin on the Qinghai–Tibetanan Plateau to climate change and disturbances. Environ Earth Sci 71:1421–1431. doi:10.1007/s12665-013-2547-0
Ross DJ, Tate KR, Scott NA, Feltham CW (1999) Land-use change: effects on soil carbon, nitrogen and phosphorus pools and fluxes in three adjacent ecosystems. Soil Biol Biochem 31:803–813. doi:10.1016/S0038-0717(98)00180-1
Talbot JM, Bruns TD, Taylor JW, Smith DP, Branco S, Glassman SI, Erlandson S, Vilgalys R, Liao HL, Smith ME, Peay KG (2014) Endemism and functional convergence across the North American soil mycobiome. Proc Natl Acad Sci USA 111:6341–6346. doi:10.1073/pnas.1402584111
Turner BL, Skole D, Sanderson S, Fischer G, Fresco L, Leemans R (1995) Land-use and land-cover change. Global Change Report, Sweden
Vance ED, Brookes PC, Jenkinson DS (1987) An extraction method for measuring soil microbial biomass-C. Soil Biol Biochem 19:703–707. doi:10.1016/0038-0717(87)90052-6
Wang GX, Qian J, Cheng GD, Lai YM (2002) Soil organic carbon pool of grassland soils on the Qinghai–Tibetanan Plateau and its global implication. Sci Total Environ 291:207–217. doi:10.1016/S0048-9697(01)01100-7
Wang GX, Cheng GD, Shen YP, Qian J (2003) Influence of land cover changes on the physical and chemical properties of alpine meadow soil. Chin Sci Bull 48:118–124. doi:10.1360/03tb9023
Wang G, Wang Y, Kubota J (2006a) Land-cover changes and its impacts on ecological variables in the headwaters area of the Yangtze River, China. Environ Monit Assess 120:361–385. doi:10.1007/s10661-005-9068-6
Wang WY, Wang QJ, Wang HC (2006b) The effect of land management on plant community composition, species diversity, and productivity of alpine Kobersia steppe meadow. Ecol Res 21:181–187. doi:10.1007/s11284-005-0108-z
Wang G, Wang Y, Li Y, Cheng H (2007) Influences of alpine ecosystem responses to climatic change on soil properties on the Qinghai–Tibetan Plateau, China. Catena 70:506–514. doi:10.1016/j.catena.2007.01.001
Wang W, Wang QJ, Lu ZY (2009) Soil organic carbon and nitrogen content of density fractions and effect of meadow degradation to soil carbon and nitrogen of fractions in alpine Kobresia meadow. Sci China Ser D 52:660–668. doi:10.1007/s11430-009-0056-5
Wang J, Wang G, Hu H, Wu Q (2010) The influence of degradation of the swamp and alpine meadows on CH4 and CO2 fluxes on the Qinghai–Tibetanan Plateau. Environ Earth Sci 60:537–548. doi:10.1007/s12665-009-0193-3
Wardle DA (1992) A comparative-assessment of factors which influence microbial biomass carbon and nitrogen levels in soil. Biol Rev 67:321–358. doi:10.1111/j.1469-185X.1992.tb00728.x
Whitmore AP, Kirk GJD, Rawlins BG (2014) Technologies for increasing carbon storage in soil to mitigate climate change. Soil Use Manag. doi:10.1111/sum.12115
Wu RG, Tiessen H (2002) Effect of land use on soil degradation in alpine grassland soil, China. Soil Sci Soc Am J 66:1648–1655. doi:10.2136/sssaj2002.1648
Wu GL, Liu ZH, Zhang L, Hu TM, Chen JM (2010) Effects of artificial grassland establishment on soil nutrients and carbon properties in a black-soil-type degraded grassland. Plant Soil 333:469–479. doi:10.1007/s11104-010-0363-9
Yang H, Lu Q, Wu B, Yang H, Zhang J, Lin Y (2006) Vegetation diversity and its application in sandy desert revegetation on Tibetan Plateau. J Arid Environ 65:619–631. doi:10.1016/j.jaridenv.2005.08.010
Yang YH, Fang JY, Tang YH, Ji CJ, Zheng CY, He JS, Zhu BA (2008) Storage, patterns and controls of soil organic carbon in the Tibetan grasslands. Glob Change Biol 14:1592–1599. doi:10.1111/j.1365-2486.2008.01591.x
Yang Y, Chen R, Han C, Qing W (2013) Measurement and estimation of the summertime daily evapotranspiration on alpine meadow in the Qilian Mountains, northwest China. Environ Earth Sci 68:2253–2261. doi:10.1007/s12665-012-1907-5
Zak JC, Willig MR, Moorhead DL, Wildman HG (1994) Functional diversity of microbial communities - a quantitative approach. Soil Biol Biochem 26:1101–1108. doi:10.1016/0038-0717(94)90131-7
Zeng DH, Hu YL, Chang SX, Fan ZP (2009) Land cover change effects on soil chemical and biological properties after planting Mongolian pine (Pinus sylvestris var. mongolica) in sandy lands in Keerqin, northeastern China. Plant Soil 317:121–133. doi:10.1007/s11104-008-9793-z
Zhang XF, Zhao L, Xu SJ Jr, Liu YZ, Liu HY, Cheng GD (2013) Soil moisture effect on bacterial and fungal community in Beilu River (Tibetan Plateau) permafrost soils with different vegetation types. J Appl Microbiol 114:1054–1065. doi:10.1111/jam.12106
Zhao D, Li F, Yang Q, Wang R, Song Y, Tao Y (2013) The influence of different types of urban land use on soil microbial biomass and functional diversity in Beijing, China. Soil Use Manag 29:230–239. doi:10.1111/Sum.12034
Zheng H, Ouyang ZY, Wang XK, Fang ZG, Zhao TQ, Miao H (2005) Effects of regenerating forest cover on soil microbial communities: a case study in hilly red soil region, Southern China. For Ecol Manage 217:244–254. doi:10.1016/j.foreco.2005.06.005
Zhou H, Zhao X, Tang Y, Gu S, Zhou L (2005) Alpine grassland degradation and its control in the source region of the Yangtze and Yellow Rivers, China. Grassl Sci 51:191–203. doi:10.1111/j.1744-697X.2005.00028.x
Acknowledgments
This research was financially supported by projects of the National Basic Research Program of China (2013CBA01086) and National Natural Science Foundation of China (Grant No. 41222001 and 91025011). We thank our colleagues at the Qilian Alpine Ecology and Hydrology Research Station for their valuable assistance with the fieldwork. We also greatly appreciate the responsible editor and the two anonymous reviewers whose comments dramatically improved this paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhu, P., Chen, R., Song, Y. et al. Effects of land cover conversion on soil properties and soil microbial activity in an alpine meadow on the Tibetan Plateau. Environ Earth Sci 74, 4523–4533 (2015). https://doi.org/10.1007/s12665-015-4509-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12665-015-4509-1