Abstract
Purpose
In the Earth’s crust, silicon (Si) is the most abundant element after oxygen, while, under salt stress, its role in the tolerance of aromatic and medicinal plants (AMPs) is not yet detailed. For this reason, in this study we evaluated the effect of exogenous Si on some tolerance-related parameters in salt-stressed fenugreek, as an important AMP.
Methods
3 mM of exogenous Si was applied to assess its impact on plant biomass and on some tolerance-related parameters in fenugreek (Trigonella foenum-graecum L.) grown under 150 mM NaCl stress.
Results
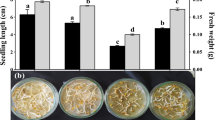
Results showed that salinity reduced growth parameters, relative water content, photosystem II efficiency, stomatal conductance and K+ and Ca2+ contents, while it increased the Na+ content, which could explain the obtained reduction in fenugreek growth and yield. However, Si supply reversed the depressive effects of salinity and improved fenugreek growth and yield. Adding exogenous Si also caused a significant reduction in Na+ content and increased K+ and Ca2+ concentrations. The content of malonyldialdehyd and hydrogen peroxide and the level of electrolyte leakage were significantly increased in salt-stressed fenugreek, while were significantly decreased after Si supplementation. The reduction in oxidative stress markers in Si-treated plants was correlated with a significant increase in both enzymatic and non-enzymatic antioxidant systems and an important accumulation of compatible solutes.
Conclusion
Therefore, exogenous Si was directly involved in the central defensive mechanisms to enhance salt tolerance of fenugreek, thus its application could be a promoting strategy to alleviate the damages of salinity on fenugreek growth and yield.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Data Availability
Not applicable.
References
Hu Y, Schmidhalter U (2004) Limitation of salt stress to plant growth. HOCK. E Plant Toxicol 4:205–238
Zhu JK (2003) Regulation of ion homeostasis under salt stress. Curr Opin Plant Biol 6:441–445. https://doi.org/10.1016/S1369-5266(03)00085-2
Said-Al Ahl HAH, Omer EA (2011) Medicinal and aromatic plants production under salt stress. A review. Herba Pol 72–87
Khoshsokhan F, Babalar M, Chaghazardi HR, Fatahi Moghadam MR (2012) Effect of salinity and drought stress on germination in thyme effect of salinity and drought stress on germination indices of two thymus species. Cercet Agron în Mold 1:27–35. https://doi.org/10.2478/v10298-012-0003-z
Sudhir P, Murthy SDS (2004) Effects of salt stress on basic processes of photosynthesis. Photosynth 42:481–486. https://doi.org/10.1007/S11099-005-0001-6
El Moukhtari A, Carol P, Mouradi M, et al (2021) Silicon improves physiological, biochemical, and morphological adaptations of alfalfa (Medicago sativa L.) during salinity stress. Symbiosis 85(3):305–324. https://doi.org/10.1007/S13199-021-00814-Z
Lamsaadi N, El Moukhtari A, Oubenali A, Farissi M (2022) Exogenous silicon improves salt tolerance of fenugreek (Trigonella foenum-graecum L.) during seed germination and early seedling stages. Biol 1:1–14. https://doi.org/10.1007/S11756-022-01035-5
Luo X, Dai Y, Zheng C et al (2021) The ABI4-RbohD/VTC2 regulatory module promotes reactive oxygen species (ROS) accumulation to decrease seed germination under salinity stress. New Phytol 229:950–962. https://doi.org/10.1111/NPH.16921
Chrysargyris A, Michailidi E, Tzortzakis N (2018) Physiological and biochemical responses of Lavandula angustifolia to salinity under mineral foliar application. Front Plant Sci 9:489. https://doi.org/10.3389/fpls.2018.00489
Ali M, Afzal S, Parveen A et al (2021) Silicon mediated improvement in the growth and ion homeostasis by decreasing Na+ uptake in maize (Zea mays L.) cultivars exposed to salinity stress. Plant Physiol Biochem 158:208–218. https://doi.org/10.1016/J.PLAPHY.2020.10.040
Bashan Y, Holguin G, De-Bashan LE (2004) Azospirillum-plant relationships: Physiological, molecular, agricultural, and environmental advances (1997–2003). Can J Microbiol 50:521–577. https://doi.org/10.1139/w04-035
Benayad Z, Gómez-Cordovés C, Es-Safi NE (2014) Characterization of flavonoid glycosides from fenugreek (Trigonella foenum-graecum) crude seeds by HPLC–DAD–ESI/MS Analysis. Int J Mol Sci 15:20668–20685. https://doi.org/10.3390/IJMS151120668
Yadav R, Chowdhury P (2017) Screening the antioxidant activity of Trigonella foenum graecum seeds. Int J Pharm Res Appl 2:65–70
Jeni James J, Meena Devi V (2021) Efficacy of some traditional Indian spices for treating diabetes. Int J Res Sci Eng 9(3):46–53
Alrumaihi FA, Khan MA, Allemailem KS et al (2021) Methanolic fenugreek seed extract induces p53-dependent mitotic catastrophe in breast cancer cells, leading to apoptosis. J Inflamm Res 14:1511–1535. https://doi.org/10.2147/JIR.S300025
Singh B, Kaur R, Singh K (2008) Characterization of Rhizobium strain isolated from the roots of Trigonella foenumgraecum (fenugreek). African J Biotechnol 7:3671–3676
Tsegaye M, Assefa F, Zeleke J (2015) Symbiotic and phenotypic characterization of Rhizobium isolates nodulating fenugreek (Trigonellafoenum-graecum L.) from North and East Shewa. Ethiopia Int J Agron Agric Res 7:93–104
Nasseri M, Arouiee H, Kafi M, Neamati H (2012) Effect of silicon on growth and physiological parameters in fenugreek (Trigonella foenum-graceum L.) under salt stress. Int J Agric Crop Sci 4:1554–1558
Zaghdoudi M, Msilini N, Govindachary S et al (2011) Inhibition of photosystems I and II activities in salt stress-exposed fenugreek (Trigonella foenum graecum). J Photochem Photobiol B Biol 105:14–20. https://doi.org/10.1016/j.jphotobiol.2011.06.005
Rizwan M, Ali S, Ibrahim M et al (2015) Mechanisms of silicon-mediated alleviation of drought and salt stress in plants: A review. Environ Sci Pollut Res 22:15416–15431. https://doi.org/10.1007/S11356-015-5305-X
El Moukhtari A, Lamsaadi N, Oubenali A, et al (2021) Exogenous silicon application promotes tolerance of legumes and their N2 fixing symbiosis to salt stress. Silicon 1–18. https://doi.org/10.1007/S12633-021-01466-W
Fatemi H, Esmaiel Pour B, Rizwan M (2020) Isolation and characterization of lead (Pb) resistant microbes and their combined use with silicon nanoparticles improved the growth, photosynthesis and antioxidant capacity of coriander (Coriandrum sativum L.) under Pb stress. Environ Pollut 266:114982. https://doi.org/10.1016/J.ENVPOL.2020.114982
Shi Y, Zhang Y, Yao H et al (2014) Silicon improves seed germination and alleviates oxidative stress of bud seedlings in tomato under water deficit stress. Plant Physiol Biochem 78:27–36. https://doi.org/10.1016/J.PLAPHY.2014.02.009
El Moukhtari A, Lamsaadi N, Farssi O, et al (2022) Silicon- and phosphate-solubilizing Pseudomonas alkylphenolica PF9 alleviate low phosphorus availability stress in alfalfa (Medicago sativa L.). Front Agron 4. https://doi.org/10.3389/FAGRO.2022.823396/BIBTEX
Hoaglan D, Arnon D (1950) The water-culture method for growing plants without soil. Circ Calif Agric Exp Stn 347:1–32
Ghoulam C, Foursy A, Fares K (2002) Effects of salt stress on growth, inorganic ions and proline accumulation in relation to osmotic adjustment in five sugar beet cultivars. Environ Exp Bot 47:39–50. https://doi.org/10.1016/S0098-8472(01)00109-5
Arnon DI (1949) Copper enzymes in isolated chloroplasts. polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1–15. https://doi.org/10.1104/pp.24.1.1
D’souza MR, Devaraj V (2013) Role of calcium in increasing tolerance of Hyacinth bean to salinity. J Appl Biol Biotechnol 1:20. https://doi.org/10.7324/JABB.2013.1302
Mouradi M, Bouizgaren A, Farissi M et al (2016) Seed osmopriming improves plant growth, nodulation, chlorophyll fluorescence and nutrient uptake in alfalfa (Medicago sativa L.) – rhizobia symbiosis under drought stress. Sci Hortic 213:232–242. https://doi.org/10.1016/j.scienta.2016.11.002
Mahmood S, Daur I, Al-Solaimani SG et al (2016) Plant growth promoting rhizobacteria and silicon synergistically enhance salinity tolerance of mung bean. Front Plant Sci 7:876. https://doi.org/10.3389/FPLS.2016.00876
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Brennan T, Frenkel C (1977) Involvement of hydrogen peroxide in the regulation of senescence in pear1. Plant Physiol 59:411–416. https://doi.org/10.1104/pp.59.3.411
Hori K, Wada A, Shibuta T (1997) Changes in phenoloxidase activities of the galls on leaves of Ulmus davidana formed by Tetraneura fusiformis (Homoptera: Eriosomatidae). Appl Entomol Zool 32:365–371. https://doi.org/10.1303/aez.32.365
Beyer WF, Fridovich I (1987) Assaying for superoxide dismutase activity: Some large consequences of minor changes in conditions. Anal Biochem 161:559–566. https://doi.org/10.1016/0003-2697(87)90489-1
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16:144–158
Chang CC, Yang MH, Wen HM, Chern JC (2002) Estimation of total flavonoid content in propolis by two complementary colometric methods. J Food Drug Anal 10:178–182. https://doi.org/10.38212/2224-6614.2748
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Grieve CM, Grattan SR (1983) Rapid assay for determination of water-soluble quaternary ammonium compounds. Plant Soil 70:303–307. https://doi.org/10.1007/BF02374789
Dubois M, Gilles KA, Hamilton JK et al (2002) colorimetric method for determination of sugars and related substances. Microb Cell Fact 28:350–356. https://doi.org/10.1021/AC60111A017
Oukaltouma K, El Moukhtari A, Lahrizi Y et al (2021) Phosphorus deficiency enhances water deficit impact on some morphological and physiological traits in four faba bean (Vicia faba L.) varieties. Ital J Agron 16:1–13. https://doi.org/10.4081/IJA.2020.1662
Latef Aaa, Abu Alhmad MF, Hammad SA (2017) Foliar application of fresh moringa leaf extract overcomes salt stress in fenugreek (Trigonella foenum-graecum) plants. Egypt J BotVol 57(1):157–179. https://journals.ekb.eg/https://doi.org/10.21608/ejbo.2017.317.1011
Bayat H, Alirezaie M, Neamati H, Abdollahi SA (2013) Effect of silicon on growth and ornamental traits of salt stressed calendula (Calendula officinalis L.). J Ornam Plant 3:207–214
Kafi M, Rahimi Z (2011) Effect of salinity and silicon on root characteristics, growth, water status, proline content and ion accumulation of purslane. Soil Sci Plant Nutr 57:341–347. https://doi.org/10.1080/00380768.2011.567398
Robatjazi R, Roshandel P, Hooshmand D (2020) Benefits of silicon nutrition on growth, physiological and phytochemical attributes of basil upon salinity stress. Int J Hortic Sci Technol 7:37–50. https://doi.org/10.22059/ijhst.2020.288551.318
Gengmao Z, Shihui L, Xing S et al (2015) The role of silicon in physiology of the medicinal plant (Lonicera japonica L.) under salt stress. Sci Rep 5:1–11. https://doi.org/10.1038/srep12696
Ahanger MA, Aziz U, Alsahli AA et al (2019) Influence of exogenous salicylic acid and nitric oxide on growth, photosynthesis, and ascorbate-glutathione cycle in salt stressed Vigna angularis. Biomolecules 10:1–16. https://doi.org/10.3390/biom10010042
Mousavi SA, Roosta HR, Esmaeilizadeh M, Eshghi S (2022) Alleviating the adverse effects of salinity and alkalinity stresses on some physiological traits by selenium and silicon foliar applications on cucumber (Cucumis sativus L.) plants. J Plant Nutr 1–18. https://doi.org/10.1080/01904167.2022.2043370
Alamri S, Hu Y, Mukherjee S et al (2020) Silicon-induced postponement of leaf senescence is accompanied by modulation of antioxidative defense and ion homeostasis in mustard (Brassica juncea) seedlings exposed to salinity and drought stress. Plant Physiol Biochem 157:47–59. https://doi.org/10.1016/J.PLAPHY.2020.09.038
Yang Z, Li JL, Liu LN et al (2020) Photosynthetic regulation under salt stress and salt-tolerance mechanism of sweet sorghum. Front Plant Sci 10:1722. https://doi.org/10.3389/FPLS.2019.01722/BIBTEX
Farissi M, Mouradi M, Farssi O et al (2018) Variation in leaf gas exchange, chlorophyll fluorescence and membrane potential root cortex cells of two alfalfa populations under salinity: Assessment of antioxidant potential role in salt tolerance. Arch Biol Sci 70:413–423. https://doi.org/10.2298/ABS171019001F
Ganieva RA, Allahverdiyev SR, Guseinova NB, Kavakli HI (1998) Effect of salt stress and synthetic hormone polystimuline K on the photosynthetic activity of cotton (Gossypium hirsutum). Tr J Bot 22:217–221
Shekari F, Abbasi A, Mustafavi SH (2017) Effect of silicon and selenium on enzymatic changes and productivity of dill in saline condition. J Saudi Soc Agric Sci 16:367–374. https://doi.org/10.1016/j.jssas.2015.11.006
Shen Z, Pu X, Wang S et al (2022) Silicon improves ion homeostasis and growth of liquorice under salt stress by reducing plant Na+ uptake. Sci Reports 12(1):1–13. https://doi.org/10.1038/s41598-022-09061-8
Hajiboland R, Cherghvareh L, Dashtebani F (2017) Effects of silicon supplementation on wheat plants under salt stress. J Plant Process Funct 5:1–11
Fahimi J, Bouzoubaâ Z, Achemchem F et al (2017) Effect of silicon on improving salinity tolerance of Taliouine Crocus sativus L. Acta Hort 1184:219–227
Abbas T, Balal RM, Shahid MA et al (2015) Silicon-induced alleviation of NaCl toxicity in okra (Abelmoschus esculentus) is associated with enhanced photosynthesis, osmoprotectants and antioxidant metabolism. Acta Physiol Plant 37:1–15. https://doi.org/10.1007/S11738-014-1768-5/FIGURES/8
Gou T, Chen X, Han R et al (2020) Silicon can improve seed germination and ameliorate oxidative damage of bud seedlings in cucumber under salt stress. Acta Physiol Plant 42:1–11. https://doi.org/10.1007/s11738-019-3007-6
Bertolino LT, Caine RS, Gray JE (2019) Impact of stomatal density and morphology on water-use efficiency in a changing world. Front Plant Sci 10:1–11. https://doi.org/10.3389/FPLS.2019.00225/BIBTEX
Siddiqui MH, Al-Whaibi MH, Faisal M, Al Sahli AA (2014) Nano-silicon dioxide mitigates the adverse effects of salt stress on Cucurbita pepo L. Environ Toxicol Chem 33:2429–2437. https://doi.org/10.1002/ETC.2697
Avestan S, Ghasemnezhad M, Esfahani M, Byrt CS (2019) Application of nano-silicon dioxide improves salt stress tolerance in strawberry plants. Agron 9:1–17. https://doi.org/10.3390/AGRONOMY9050246
Rohanipoor A, Norouzi M, Moezzi A, Hassibi P (2013) Effect of silicon on some physiological properties of maize (Zea mays) under salt stress. J Biol Environ Sci 71–79
Esmaeili S, Salehi H, Eshghi S (2015) Silicon ameliorates the adverse effects of salinity on turfgrass growth and development. J Plant Nutr 38:1885–1901. https://doi.org/10.1080/01904167.2015.1069332
Epstein E (1994) The anomaly of silicon in plant biology. Proc Natl Acad Sci USA 91:11–17. https://doi.org/10.1073/PNAS.91.1.11
Feng Ma J (2004) Role of silicon in enhancing the resistance of plants to biotic and abiotic stresses. Soil Sci Plant Nutr 50:11–18. https://doi.org/10.1080/00380768.2004.10408447
Wang L, Ning C, Pan T, Cai K (2022) role of silica nanoparticles in abiotic and biotic stress tolerance in plants: A review. Int J Mol Sci 23:2–16. https://doi.org/10.3390/IJMS23041947
Meena M, Divyanshu K, Kumar S et al (2019) Regulation of L-proline biosynthesis, signal transduction, transport, accumulation and its vital role in plants during variable environmental conditions. Heliyon 5:1–20. https://doi.org/10.1016/j.heliyon.2019.e02952
El Moukhtari A, Cabassa-Hourton C, Farissi M, Savouré A (2020) How does proline treatment promote salt stress tolerance during crop plant development? Front Plant Sci 11:1127. https://doi.org/10.3389/FPLS.2020.01127
Suprasanna P, Nikalje GC, Rai AN (2016) Osmolyte accumulation and implications in plant abiotic stress tolerance. Osmolytes Plants Acclim to Chang Environ Emerg Omi Technol 1–12. https://doi.org/10.1007/978-81-322-2616-1_1
Mousavi SA, Roosta HR, Esmaeilizadeh M, Eshghi S (2022) Alleviating the adverse effects of salinity and alkalinity stresses on some physiological traits by selenium and silicon foliar applications on cucumber (Cucumis sativus L.) plants. J Plant Nutr 1–18 https://doi.org/10.1080/01904167.2022.2043370
Zhu Y, Jiang X, Zhang J et al (2020) Silicon confers cucumber resistance to salinity stress through regulation of proline and cytokinins. Plant Physiol Biochem 156:209–220. https://doi.org/10.1016/J.PLAPHY.2020.09.014
Slimen IB, Najar T, Ghram A et al (2014) Reactive oxygen species, heat stress and oxidative-induced mitochondrial damage. A review Int J Hyperth 30:513–523. https://doi.org/10.3109/02656736.2014.971446
Al-aghabary K, Zhu Z, Shi Q (2007) Influence of silicon supply on chlorophyll content, chlorophyll fluorescence, and antioxidative enzyme activities in tomato plants under salt stress. J Plant Nutr 27:2101–2115. https://doi.org/10.1081/PLN-200034641
Aghaei K, Komatsu S (2013) Crop and medicinal plants proteomics in response to salt stress. Front Plant Sci 4:8. https://doi.org/10.3389/fpls.2013.00008
Kim YH, Khan AL, Waqas M et al (2014) Silicon application to rice root zone influenced the phytohormonal and antioxidant responses under salinity stress. J Plant Growth Regul 33:137–149. https://doi.org/10.1007/S00344-013-9356-2/FIGURES/5
Zhang W, Xie Z, Wang L et al (2017) Silicon alleviates salt and drought stress of Glycyrrhiza uralensis seedling by altering antioxidant metabolism and osmotic adjustment. J Plant Res 130:611–624. https://doi.org/10.1007/s10265-017-0927-3
Acknowledgements
The authors are grateful to all those who participated in the elaboration of this study. We thank all the partners involved in ANPMA-CNRST-USMS Project. We also thank the administrative and technical staff of the Polydisciplinary Faculty of Beni-Mellal for their support.
Funding
This work was supported by the National Agency of Medicinal and Aromatic Plants (ANPMA-Morocco), National Center of Scientific Research (CNRST-Morocco) and Sultan Moulay Slimane University (USMS-Morocco), convention number:348/20.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception. Methodology and data analysis were performed by Lamsaadi Nadia; El Moukhtari Ahmed and Ziati Irouane. Validation; Farissi Mohamed. The first draft of the manuscript was written by Lamsaadi Nadia. Revision and comment of the manuscript; Mouradi Mohammed, El Hassni Majida, Ghoulam Cherki and Farissi Mohamed. Supervision and funding acquisition; Farissi Mohamed. All authors read and approved the submitted version of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lamsaadi, N., El Moukhtari, A., Irouane, Z. et al. Beneficial role of exogenous silicon on yield, antioxidant systems, osmoregulation and oxidative stress in fenugreek (Trigonella foenum-graecum L.) under salinity stress. Silicon 15, 547–561 (2023). https://doi.org/10.1007/s12633-022-02034-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-022-02034-6