Abstract
The modification of silica-coated CoFe2O4 magnetic nanoparticles (CoFe2O4@SiO2) with chlorosulfonic acid, which can be utilized as an organic-inorganic hybrid heterogeneous catalyst, introduces an astonishing and efficient system for the synthesis and simplicity of the recovery of the catalyst. The applied CoFe2O4 magnetic nanoparticles are 22.98–45.30 nm measured that can be utilized as a catalyst for the preparation of 2-amino-4,6-diarylnicotinonitrile under microwave irradiation in solvent-free conditions by four component reaction of aromatic acetophenone, aldehydes analogues, malononitrile and ammonium acetate. The so synthesized magnetic nanocatalyst was characterized by X-ray powder diffraction, SEM, TGA and FT-IR techniques. This simple protocol suggests advantages such as shorter reaction times, high yield, catalyst recovery, achieving the high purity of products by simple recrystallization and facile work-up. Other noticeable characteristics contain the catalyst can be recovered at least five times without any clear decrease in its catalytic activity.

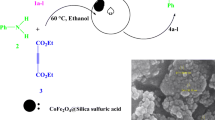
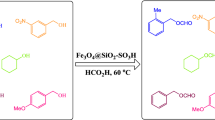
Synthesis of 2-amino-4,6-diarylnicotinonitrile in the presence of CoFe2O4@SiO2-SO3H as a reusable solid acid catalyst under microwave irradiation in solvent-free conditions.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Ahankar H, Ramazani A, Joo SW (2016) Magnetic nickel ferrite nanoparticles as an efficient catalyst for the preparation of polyhydroquinoline derivatives under microwave irradiation in solvent-free conditions. Res Chem Intermed 42(3):2487–2500
Kiasat AR, Davarpanah J (2015) Fe3O4@ Silica sulfuric acid core–shell composite as a novel nanomagnetic solid acid: synthesis, characterization and application as an efficient and reusable catalyst for one-pot synthesis of 3, 4-dihydropyrimidinones/thiones under solvent-free conditions. Res Chem Intermed 41(5):2991–3001
Koesnarpadi S, Santosa SJ, Siswanta D, Rusdiarso B (2015) Synthesis and characterizatation of magnetite nanoparticle coated humic acid (Fe3O4/HA). Procedia Environ Sci 30:103–108
Aghahosseini H, Ramazani A, Ślepokura K, Lis T (2018) The first protection-free synthesis of magnetic bifunctional l-proline as a highly active and versatile artificial enzyme: Synthesis of imidazole derivatives. J Colloid Interface Sci 511:222–232
Malekzadeh AM, Ramazani A, Rezaei SJT, Niknejad H (2017) Design and construction of multifunctional hyperbranched polymers coated magnetite nanoparticles for both targeting magnetic resonance imaging and cancer therapy. J Colloid Interface Sci 490:64–73
Dayyani N, Ramazani A, Khoee S, Shafiee A (2017) Synthesis and characterization of the first generation of polyamino-ester dendrimer-grafted magnetite nanoparticles from 3-aminopropyltriethoxysilane (APTES) via the convergent approach. Silicon:1–7
Tarasi R, Ramazani A, Ghorbanloo M, Khoobi M, Aghahosseini H, Joo SW, Shafiee A Synthesis of Magnetic Fe3O4@ polyethyleneimine. Mn (II) from Fe3O4,[3-(2, 3-Epoxypropoxy) propyl] trimethoxysilane, Polyethyleneimine and Mn (II) Acetate as a Novel Silicon-Containing Polymeric Organic-Inorganic Hybrid Nanomaterial and Its Catalytic Investigation Towards the Oxidation of Cyclohexene, Ethyl Benzene and Toluene in the Presence of H2O2 as an Oxidant. Silicon:1–9
Sheldon RA (2007) The E factor: fifteen years on. Green Chem 9(12):1273–1283
Rezaei A, Ramazani A, Gouranlou F, Woo Joo S (2017) Silica Nanoparticles/Nanosilica Sulfuric Acid as a Reusable Catalyst for Fast, Highly Efficient and Green Synthesis of 2-(Heteroaryl) acetamide Derivatives. Lett Org Chem 14(2):86–92
Ahankar H, Ramazani A, Ślepokura K, Lis T, Joo SW (2016) Synthesis of pyrrolidinone derivatives from aniline, an aldehyde and diethyl acetylenedicarboxylate in an ethanolic citric acid solution under ultrasound irradiation. Green Chem 18(12):3582–3593
Zhang F, Zhao Y, Sun L, Ding L, Gu Y, Gong P (2011) Synthesis and anti-tumor activity of 2-amino-3-cyano-6-(1H-indol-3-yl)-4-phenylpyridine derivatives in vitro. Eur J Med Chem 46(7):3149–3157
Bekhit AA, Baraka AM (2005) Novel milrinone analogs of pyridine-3-carbonitrile derivatives as promising cardiotonic agents. Eur J Org Chem 40(12):1405–1413
Murata T, Shimada M, Sakakibara S, Yoshino T, Kadono H, Masuda T, Shimazaki M, Shintani T, Fuchikami K, Sakai K (2003) Discovery of novel and selective IKK-β serine-threonine protein kinase inhibitors. Part 1 Bioorg Med Chem Lett 13(5):913–918
Mantri M, de Graaf O, van Veldhoven J, Göblyös A, von Frijtag Drabbe Künzel JK, Mulder-Krieger T, Link R, de Vries H, Beukers MW, Brussee J (2008) 2-Amino-6-furan-2-yl-4-substituted nicotinonitriles as A2A adenosine receptor antagonists. J Med Chem 51(15):4449–4455
Deng J, Sanchez T, Al-Mawsawi LQ, Dayam R, Yunes RA, Garofalo A, Bolger MB, Neamati N (2007) Discovery of structurally diverse HIV-1 integrase inhibitors based on a chalcone pharmacophore. Bioorg Med Chem Lett 15(14):4985–5002
Shishoo C, Devani M, Bhadti V, Ananthan S, Ullas G (1983) Reaction of nitriles under acidic conditions: a novel, direct formation of condensed 4-chloropyrimidines. Tetrahedron Lett 24(42):4611–4612
Kambe S, Saito K, Sakurai A, Midorikawa H (1980, 1980) A simple method for the preparation of 2-amino-4-aryl-3-cyanopyridines by the condensation of malononitrile with aromatic aldehydes and alkyl ketones in the presence of ammonium acetate. Synthesis (05):366–368
Maiti S, Biswas S, Jana U (2010) Iron (III)-catalyzed four-component coupling reaction of 1, 3-dicarbonyl compounds, amines, aldehydes, and nitroalkanes: a simple and direct synthesis of functionalized pyrroles. J Med Chem 75(5):1674–1683
Gu Y (2012) Multicomponent reactions in unconventional solvents: state of the art. Green Chem 14(8):2091–2128
Dömling A, Ugi I (2000) Multicomponent reactions with isocyanides. Angew Chem Int Ed 39(18):3168–3210
Gers CF, Nordmann J, Kumru C, Frank W, Müller TJ (2014) Solvatochromic fluorescent 2-substituted 3-ethynyl quinoxalines: Four-component synthesis, photophysical properties, and electronic structure. J org chem 79(8):3296–3310
Hosseinzadeh Z, Ramazani A, Ahankar H, Ślepokura K, Lis T (2018) Sulfonic acid-functionalized silica-coated magnetic nanoparticles as a reusable catalyst for the preparation of pyrrolidinone derivatives in a green solvent conditions. SILICON Accepted for publication. https://doi.org/10.1007/s12633-017-9737-4
Shah HC, Shah VH, Desai ND (2009) A novel strategy for the synthesis of 2-amino-4, 6-diarylnicotinonitrile. Arkivoc 2:76–87
CrysAlisPro in KM4-CCD software; Agilent Technologies: (2012). Yarnton, U.K.,
Sheldrick GM (2015) Acta Crystallogr Sect. C. Cryst, Struct. Commun 71:3–8
Brandenburg K (2014) DIAMOND Version 3.2k, Crystal Impact GbR. Bonn, Germany,
Kiasat AR, Davarpanah J (2013) Fe3O4@ silica sulfuric acid nanoparticles: An efficient reusable nanomagnetic catalyst as potent solid acid for one-pot solvent-free synthesis of indazolo [2, 1-b] phthalazine-triones and pyrazolo [1, 2-b] phthalazine-diones. J Mol Catal A Chem 373:46–54
Naeimi H, Nazifi ZS (2013) A highly efficient nano-Fe3O4 encapsulated-silica particles bearing sulfonic acid groups as a solid acid catalyst for synthesis of 1, 8-dioxo-octahydroxanthene derivatives. J Nanopart Res 15(11):2026
Nemati F, Heravi MM, Rad RS (2012) Nano-Fe3O4 encapsulated-silica particles bearing Sulfonic acid groups as a magnetically separable catalyst for highly efficient knoevenagel condensation and michael addition reactions of aromatic aldehydes with 1, 3-cyclic diketones. Chin J Catal 33(11):1825–1831
Sadri F, Ramazani A, Massoudi A, Khoobi M, Azizkhani V, Tarasi R, Dolatyari L, Min B-K (2014) Magnetic CoFe2O4 nanoparticles as an efficient catalyst for the oxidation of alcohols to carbonyl compounds in the presence of oxone as an oxidant. Bull Kor Chem Soc 35(7):2029
Acknowledgements
This work was supported by the “University of Zanjan”.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hosseinzadeh, Z., Ramazani, A., Ahankar, H. et al. Synthesis of 2-amino-4,6-diarylnicotinonitrile in the presence of CoFe2O4@SiO2-SO3H as a reusable solid acid nanocatalyst under microwave irradiation in solvent-freeconditions. Silicon 11, 2169–2176 (2019). https://doi.org/10.1007/s12633-018-0034-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-018-0034-7