Abstract
The molecular detection of Norovirus GI and Norovirus GII in the Tunisian industrial wastewater treatment plant of Charguia I was conducted to test the effectiveness of secondary biological treatment using the activated sludge procedure and the UV-C254 tertiary treatment radiation using a UV disinfection prototype to upgrade the quality of the purified wastewater. A total of 140 sewage samples were collected from the two lines of sewage treatment procedures. Norovirus GI and Norovirus GII have been found and quantified using Real-Time Reverse Transcription Polymerase Chain Reaction (qRT-PCR) in 66.4 and 86.4% of the collected wastewater samples. The Norovirus GI and GII mean concentration values got in the treated effluents showed a significant decrease of Norovirus viral content rates detected from upstream to downstream of activated sludge procedures and at the output of UV-C254 treatment. These findings characterise and denote for the first time the effectiveness of biological and UV-C254 treatment for Norovirus GI and Norovirus GII removal in Tunis City, northern Tunisia. Also, these data underlined the fact that purified sewage makes up a route of gastroenteritis virus transmission and a cause of viral gastroenteritis associated with water-borne and food-borne outbreaks.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Wastewater carrying various microorganisms and chemical pollutants requires depollution and purification steps using different physical, physico-chemical and biological treatments (Sano et al. 2016). The purification or the depollution process should allow, at least the removal of a significant part of solid, dissolved and non-dissolved pollution (Sano et al. 2016; Dai et al. 2016). Based on pollution removal and the processes implemented, three major steps of treatments are defined in all wastewater treatment plants worldwide, the preliminary, the secondary and tertiary stages (Dai et al. 2016; Mir-Tutusaus et al. 2018; Turki et al. 2017). Sometimes tertiary treatments of disinfection (chlorination, ultraviolet radiation, ozonation) are necessary for wastewater treatment plants for effective pathogenic microorganism removal (Sano et al. 2016; Turki et al. 2017; Lizasoain et al. 2018). Among microorganisms likely to be found in wastewater, enteric viruses are at the top of the list for their high diversity and pathogenicity. Thus, in recent years, the enteric viruses were detected at high frequencies in the treated effluents in all Tunisian wastewater treatment plants. These findings showed high resistance of these viruses to different biological purification procedures (Sdiri-Loulizi et al. 2010; Ibrahim et al. 2015, 2016, 2017a, b, 2018, 2019). Low virological quality of the Tunisian purified sewage intended for reuse in agriculture, recycling and releases into natural receiving environments was described. This constitutes a major problem for public health, in the paediatric population. Therefore, the absence of a Tunisian national monitoring programme for the enteric virus circulation in wastewater increases the frequency of viral gastroenteritis related to water-borne and food-borne diseases (Sdiri-Loulizi et al. 2010; Ibrahim et al. 2015, 2016, 2017a, b, 2018, 2019).
Norovirus (NoV) genus was classified in the Caliciviridae family (https://www.ictvonline.org/virusTaxonomy.asp). In various regions of the world, NoV was detected with high frequencies in wastewater sampled in different purification plants. The surveillance of wastewater treatment plant establishes an appropriate strategy for the study of enteric virus circulation in their respective service zones, which support better understanding and knowledge of the molecular epidemiology of these viruses in the paediatric populations (Fioretti et al. 2016; Mabasa et al. 2018). The environmental studies, which revealed the high persistence of NoV GI and NoV GII in treated wastewater and their significant resistance to secondary biological and tertiary treatments, are very rare in Africa, including Tunisia, and only restricted to some countries of the world, such as Italy, Brazil, the United States, England and Uruguay (Lizasoain et al. 2018; La Rosa et al. 2010; Kitajima et al. 2014; Campos et al. 2016).
The main aim of this study was to improve the knowledge on NoV GI and NoV GII detection rates and contents in some Tunisian wastewater samples and to assess the effectiveness of wastewater biological treatment (activated sludge) and tertiary treatment (UV-C254) procedures at the scale of an urban industrial wastewater treatment plant.
Materials and Methods
The Industrial Urban Wastewater Treatment Plant and UV-C pilot reactor
Wastewater Treatment Plant (WWTP) The study has been conducted in an urban industrial wastewater treatment plant (WWTP) in the residential area of Charguia I, in the northeast suburbs of Tunis City, Tunisia. The flow of wastewater to be treated in this plant was around of 6 × 104 m3/day. The plant of Charguia I was drained by mixed industrial, urban, domestic, hospital and rainwater coming from different zones of the great Tunis.
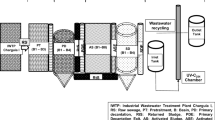
UV-C reactors: In this study, tertiary treatment is conducted using a classic UV-C monolamp cylindrical disinfection system, described earlier by Hassen et al. (2000). This photoreactor comprises only one low-pressure germicidal lamp with a length of 60 cm, a diameter of 2 cm, a wavelength of 253.7 nm and a power of 55 W. Finally, the recycling circuit is used for a second one passage of the wastewater through the irradiation chamber. The schematic design of the WWTP Charguia I and the UV-C disinfection monolamp reactor are shown in Fig. 1.
Sampling and Analysis Methods
The study has been carried out for 10 months of the present work, from June 2018 to April 2019, and 140 wastewater samples were collected at the arrival of WWTP Charguia (raw wastewater) and the output of each step of treatment. At least 7 wastewater samples were collected each month as follows: one sample at the input of WWTP or raw sewage (RS), one at the exit of primary decantation (PDE), one at the exit of activated sludge procedures (ASE), one at the exit of secondary decantation (SDE), sample at returned sludge (RtS), and two samples after the first and the second passage in UV-C pilot system (UVP1, UVP2), respectively. The physico-chemical analyses such as hydrogen potential (pH), temperature (Temp), electrical conductivity (EC), salinity (S), suspended solids (SS), chloride content (Cl), chemical oxygen demand (COD) and biological oxygen demand (BOD5) were determined for all the sewage samples collected at the input (RS) and the output (SDE) of the WWTP Charguia I according to the NF standard methods (AFNOR 1992). The bacteriological analyses applied to all wastewater samples (DPE, ASE, SDE, RtS, UVP1 and UVP2) were achieved by the enumeration of total coliforms (TC), faecal coliforms (FC), Escherichia coli (EC) and faecal streptococci (FS) by the most probable number technique (MPN) as recommended by Rodier (1978).
Virus isolation from wastewater: Viruses were isolated from 1 L of sewage, according to the US Environmental Protection Agency Protocol by using the beef extract and AlCl3 improved method (EPA 1992). NoV RNA extraction and the real-time RT-PCR were conducted as reported by Ibrahim et al. (2015). The NoVs GI and GII were detected and quantified using the QNIF2d4, COG2R4, JJV1NF1, JJV1R2/1 primers and QNIFS4, JJV1P2/1 and RING-1b1/3 probes. These primers and probes amplify the gene fragment coding for NoVs GI and GII capsid as recommended by Kageyama et al. (2004); Jothikumar et al. (2005); Da Silva et al. (2007) and Layman et al. (2009). NoVs GI and GII real-time RT-PCRs comprised the retro transcription step at 50 °C for 5 min and the polymerase activation step at 95 °C for 20 s, followed by 40 cycles of DNA amplification, which each cycle includes a DNA denaturation at 95 °C for 3 s, hybridisation, an elongation and an optical FAM reading of the primers at 60 °C for 30 s. The NoVs GI and GII concentrations were achieved using a standard ten dilution series (100 to 1010). Quantification efficiency using Real-Time Reverse Transcription Polymerase Chain Reaction (RT-PCR) of all samples negative and positive controls, was about 98% in each reaction. All NoV result contents were expressed as genome copies per µL (gc/µL).
Statistical Analysis
Main statistical analysis was performed based on the One-way ANOVA test using SPSS software (SPSS for Windows, version 22, Chicago, IL, USA). The NoV GI and GII mean content values were compared by the least significant difference, according to the Student–Newman–Keuls test.
Results
Physico-Chemical and Microbiological Analysis
The results of physical and chemical parameters showed a substantial average decrease of the mean values of the SS, the COD and the BOD5 from upstream to downstream of the activated sludge procedure (Table 1).
The obtained average value of the different bacteriological parameters in the wastewater and sludge samples collected from the different basins of activated sludge procedures and after two-successive passages by tertiary treatment using UV-C254 during the sampling period and analysed are presented in Table 1. These results showed a substantial reduction in the faecal bacteria concentrations, such as the TC, FS, FC and EC from the first to the last basins of the biological wastewater treatment procedure (Table 1). However, the obtained data showed a significant decrease in the numbers of these faecal bacteria at the output of two-successive passages through UV-C254. Abatement rates of these bacteria between the two steps of the purified sewage using biological and tertiary treatment are similar; they were about 99.9% or 3 log units (Table 1).
Virological Results
Norovirus GI and Norovirus GII Detection Rates
The results of the NoV GI and NoV GII detection showed that 66.4 and 86.4% of wastewater sampled at the industrial WWTP Charguia I and after the two-successive tertiary treatment passages by UV-C254 are positive for the NoV GI (93/140) and NoV GII (121/140), respectively. The NoV GI distribution in the five successive different basins of the activated sludge procedure (RS to SDE) is used to determine the frequencies of these viruses in each type of pond: 85% (n = 17/20) in RS, 90% (n = 18/20) in PDE, 60% (n = 12/20) in ASE, 55% (n = 11/20) in RtS and 70% (n = 14/20) in SDE (Fig. 2). The NoV GII frequencies are distributed as follows: 100% (n = 20/20), 95% (n = 19/20), 95% (n = 19/20), 80% (n = 16/20), 90% (n = 18/20) at the output of every basin type of RS, PDE, ASE, Rets and SDE (Fig. 2). At the output of the first passage (UVP1) of UV-C254 treatment, 55 (n = 11/20) and 80% (n = 16/20) of the collected wastewater samples are contaminated by NoV GI and NoV GII (Fig. 2). At the output of the second passage of UV-C254 treatment (UVP2), NoV GI and NoV GII are detected in 50 (n = 10/20) and 65% (n = 13/20) of the collected sewage samples, respectively (Fig. 2).
Norovirus GI and Norovirus GII Abatement
The concentration of the NoV GI and NoV GII is determined in all wastewater samples positive for these viruses by the real-time RT-PCR technique. The results showed that the mean values of the concentration of the NoV GI and NoV GII (genome copies/μL or gc/µL) in the first two basins (RS and PDE) are much higher than those recorded in the last three basins (ASE, RtS and SDE) at the scale of the activated sludge treatment procedure (Tables 2 and 3). NoV GI and NoV GII are detected in both the basins RS and PDE with high frequencies (85% of NoV GI and 100% of NoV GII in RS, 90% of NoV GI and 95% of NoV GII in PDE) and with important mean values of concentration (917 gc/µL of NoV GI and 74 gc/µL of NoV GII in RS; 483 gc/µL of NoV GI and 50 gc/µL of NoV GII in PDE) (Tables 2 and 3). Similarly, in the other three basins of the activated sludge procedure (ASE, RtS, SDE) and at the exit of the two-tertiary successive passages (UVP1 and UVP2), the NoV GI and NoV GII are also encountered with high frequencies. NoV GI frequencies are of the order of 60; 55; 70; 55 and 50% in RS, PDE, ASE, RtS, SDE, and at the exit of tertiary treatment UVP1 and UVP2. The NoV GII frequencies were around 95, 80, 90, 80 and 65% in RS, PDE, ASE, RtS, SDE, and at the exit of the tertiary treatment UVP1 and UVP2. However, the NoV GI and NoV GII mean concentration values got in the last three maturation ponds of the activated sludge procedure, ASE, RtS and SDE, appeared moderate to low as compared to those recorded in the first two basins (RS and PDE) (Tables 2 and 3). The mean concentration values registered at the scale of the activated sludge procedure showed a significant decrease of the NoV GI and NoV GII viral contents from the first to the last basins (RS—SDE) of the five successive and different ponds (P < 0.05) (Tables 2 and 3). A substantial reduction in the NoV GI and NoV GII average content values are also observed at the exit of two-successive UV-C254 passages as compared to those recorded at the output of the secondary decantation basins (SDE) (Tables 2 and 3). Distribution of the NoV GI and the NoV GII average concentration values registered at the exit of two-successive UV-C254 passages are reported in Tables 2 and 3.
Monthly Repartition
The NoV GI and NoV GII are encountered continuously throughout the sampling period, from June 2018 to April 2019, with elevated frequencies during winter, spring, summer and autumn. NoV GI frequencies were close to 100, 93 and 86% in September, January and March, respectively (Fig. 3). Similarly, NoV GII frequencies were close to 91, 100, 91 and 86% in July, September, February and April, respectively (Fig. 3). However, the high NoV GI and NoV GII concentration mean values are detected both in the cold seasons of winter and spring. The average NoV-GI-contents are in the order of 623, 990 and 820 gc/μL in January, in February and in March; respectively (Table 2). Besides, the NoV GII average contents are of the order of 35, 51 and 53 gc/µL in February, March and April, respectively (Table 3).
In the two basins of the activated sludge procedure (RS and PDE), NoV GI and NoV GII are found along the period of study with important average concentration values and a net peak during the winter and spring seasons (Tables 2 and 3). However, NoV GI and NoV GII are presented in the last three basins of the biological treatment procedure (AS, RtS and SDE) with lower mean concentration values as compared to those registered in the first two basins (RS and PDE) and with a notable peak during the winter and the spring (Tables 2 and 3).
At the output of the tertiary treatment, NoV GI and NoV GII appeared to exist also during the sampling period with a peak during the winter, with lower frequencies and average concentration values than those recorded at the exit of the last basins of the activated sludge procedure (SDE) (Tables 2 and 3).
Discussion
The wastewater treatment performed in wastewater treatment plants leads to a significant variable reduction in the enteric virus and the faecal bacterial content. Different experimental procedures have been adopted in previous studies to assess the load in enteric viruses in a raw effluent and the rate of elimination of these microbes in the purification systems (Ibrahim et al. 2015, 2016, 2017a, b, 2018, 2019). In this study, we focus our research on the viral removal effectiveness of NoV, taken as a virus model at a scale of the Tunisian industrial WWTP of Charguia I and by using a monolamp disinfection UV-C254 system. Thus, testing the virological quality of the purified wastewater, which is intended for agricultural reuse, recycling, and releases into natural environments represents an important hazard, a reason for public health concern. Therefore, there is a need to prevent water-borne and related food-borne viral, gastroenteritis dissemination (Qiu et al. 2015). All the average value of the main physico-chemical parameters found at the output of the activated sludge procedures tested (SDE) appeared lower than those set by the Tunisian Standard of treated wastewater discharges (ST-106 02) (COD = 77 ± 14 < to 90 mg O2/L; BOD5 = 18 ± 10 < to 30 mg O2/L; SS = 17 ± 11 < 30 mg/L). However, the average content of the faecal bacteria at the output of the activated sludge procedures (SDE) appeared much higher than those recommended by the Tunisian Standard (ST 106–02), with faecal coliforms = (16 ± 10) x 104 > to 2 × 103 MPN/100 mL, and faecal Streptococci = (6 ± 5) x 105 > to 103 MPN/100 mL. Thus, these results showed an excellent physico-chemical and a poor bacteriological wastewater quality of the purified effluents by activated sludge procedures. These results were comparable to those described in other studies showing the effectiveness of activated sludge procedures for the physico-chemical pollution removal and the low performance of this biological treatment for the bacteriological abatement (1.5–2 U logs) (Qiu et al. 2015; Campos et al. 2016; Dai et al. 2016; La Rosa et al. 2010; Sano et al. 2016). Earlier Tunisian environmental studies conducted in the same area and using a rotating biodisk procedure have reported similar results, and opposite results are found in all these previous studies in natural oxidising ponds (Ibrahim et al. 2015, 2016, 2017a, b, 2018, 2019). Also, all these last studies of Ibrahim et al. revealed a bad physico-chemical quality and a good bacteriological one of the purified effluents by natural oxidising pond procedure. In contrast, in all these last studies done, satisfactory physico-chemical purification and a huge deficiency were observed concerning the bacterial abatement of a rotating biodisks procedure.
The results gained by tertiary UV-C254 system disinfection revealed a significant improvement of the microbiological quality of the treated wastewater, with an average microbial residual much lower than the ones fixed by the Tunisian wastewater discharge standard (ST-106 02) (faecal coliforms = [3 ± 1] × 10–1 < to 2 × 103 MPN/100 mL and faecal Streptococci = [3 ± 1] × 10–1 < to 103 MPN/100 mL). Since we registered a good bacteriological quality of the treated wastewater after the UV-C254 disinfection system that could allow recycling operations, agriculture reuses and safe releases into natural aquatic environments.
Also, virological results showed that NoV GI and NoV GII are detected in 66.4, 86.4 and 63.6%, respectively, of the collected wastewater samples during the ten months of study. The first environmental studies conducted by Ibrahim et al. (2015, 2019) reported the NoV GI and NoV GII detection in 1 and 65% of the wastewater sampled at the scale of the pilot WWTP El Menzeh I, in the residential area of El Menzeh I of the Tunis City. The top detection rate of around 66.4% registered for NoV GI could reveal the emergence of gastroenteritis viruses in wastewater that might circulate in this area of study.
NoV GI and NoV GII frequencies recorded in this study are higher than those recorded in the earlier Tunisian environmental and clinical studies in the Monastir region, Sahel of Tunisia (Sdiri-Loulizi et al. 2008, 2010; Hassine-Zaafrane et al. 2014). Therefore, the top NoV rates confirmed these viruses are recognised as emerging pathogenic contaminants in several natural environments, and they establish an actual public health risk associated with water-borne and food-borne diseases (Teixeira et al. 2017; Kitajima et al. 2018).
Likewise, acquired significant frequencies of NoV GI (66.4%) and NoV GII (86.4%) in this work were analogous to those described in several previous and recent environmental studies conducted in polluted sewage sampled from various wastewater treatment plants worldwide. NoV is detected with high rates in wastewater samples in Venezuela (75%), Singapore (100%), in Southern Arizona (75%), in France (98%) in China (100%), in Uruguay (72%), in Italy (90.2%), in Japan (66.6%) and South Africa (72.2%) (Aw and Gin 2010; Prévost et al. 2015; Zhou et al. 2016; Victoria et al. 2016; Kazama et al. 2017; Suffredini et al. 2018; Mabasa et al. 2018). But two other environmental reports revealed that gastroenteritis viruses were detected with low to moderate frequencies in wastewater samples in Greece (6.3%), in Norway (26%) and Brazil (33.9%) (Myrmel et al. 2015; Kitajima et al. 2018).
Gastroenteritis virus frequencies found in this study showed that the NoV GII (86.4%) is the most and frequently Caliciviruses detected in the Tunisian sewage samples, followed by NoV GI (66.4%). These data are under other previous clinical and Tunisian environmental studies showing the dominance of NoV GII than NoV GI (Sdiri-Loulizi et al. 2010; Ibrahim et al. 2015; Dai et al. 2016). The dominance of NoV GII in Tunisian effluent from various wastewater treatment plants could be explained by these viruses being more prevalent in the Tunisian paediatric population. Similarly, these data follow those recounted in earlier environmental reports describing the NoV GII predominance in influents and effluents sampled at the scale of dissimilar wastewater treatment plants in different regions in the world, such as Venezuela, Norway, France, China and South Africa (Aw and Gin 2010; Kokkinos et al. 2011; Sima et al. 2011; Prévost et al. 2015; Teixeira et al. 2017; Mabasa et al. 2018).
In opposite for the obtained results on NoV GI and GII detection rates, the NoV GI and NoV GII quantification showed that the NoV GI average content recorded in wastewater is greater than the one registered for NoV GII. The NoV GI and NoV GII presented the mean contents of 9.17 × 102 and 0.74 × 102 gc/µL in influent and 5.9 × 101, 0.5 × 101 gc/µL in the effluent of the industrial wastewater treatment plant of Charguia I. The significant NoV GI mean content (9.17 × 102 gc/µL) and the detection rate (66.4%) recorded in this study confirmed the increase of water-borne epidemic associated with viral gastroenteritis since these viruses are more likely to generate and induce water-borne outbreaks than NoV GII (Prévost et al. 2015; Matthews et al. 2012). Therefore, the data achieved in this study determine that wastewater surveillance makes up a novel strategy for water-borne outbreaks associated with viral gastroenteritis prevention in regions that lack NoV outbreak monitoring, such as Tunisia.
The achieved virological detection rate and the quantitative results of the present study allowed to mention the moderate reduction of the NoV GI and NoV GII in the treated effluents by activated sludge and UV-C254. The NoV GI and NoV GII abatement rates were on an average of 93 and 77%; respectively. Based on the average content across the steps of recommended biological wastewater treatment procedures, the NoV GII removal rate was greater than of the NoV GI by the activated sludge process. The NoV GI and NoV GII abatement rates of 65% and 55% appeared furthermost secured by UV-C254 irradiation than by the activated sludge procedure. This result constitutes the first Tunisian documentation showing the effectiveness of the biological and tertiary wastewater treatment using activated sludge procedures and a UV-C254 disinfection reactor for the two Caliciviruses NoV GI and NoV GII removal. These last results were confirmed earlier in two similar studies carried out on wastewater procedure of natural oxidation ponds and rotating biological disks for NoV GII and rotavirus A removal (Ibrahim et al. 2015, 2016). However, these results are dissimilar to those described in two previous environmental studies, which revealed the failure of activated sludge procedure in the NoV GI, NoV GII and RVA removal in two wastewater treatment plants in the Monastir region, Sahel area of Tunisia by Sdiri-Loulizi et al. (2010) and Hassine-Zaafrane et al. (2015). Similarly, two previous studies in Southern Arizona showed that NoV GII and NoV GI are eliminated with important log reduction ranging from 1.7 to up 2 logs 10 by activated sludge and trickling filters (Kitajima et al. 2014; Schmitz et al. 2016).
The NoV GI and NoV GII distribution during the ten months of the study showed a moderate rate detection of these Caliciviruses throughout the sampling period in all seasons. However, seasonal detection of NoV GI and NoV GII in wastewater is well distributed along all the months of the study, with a distinct peak during the cold and rainy months corresponding to winter and spring seasons in Tunisia. Similarly, these data are in agreement with those described in the two earlier studies conducted by Sdiri-Loulizi et al. (2010) and Dai et al. (2016) indicating the most prevalence of these viruses in winter.
Conclusions
This study confirms the frequent occurrence of NoV Caliciviruses in raw and treated wastewater in industrial wastewater of Charguia I in Tunis City, Tunisia. Also, this study confirmed the moderate effectiveness of the activated sludge procedure for removing faecal bacteria, NoV GI and NoV GII removal, revealing a poor bacteriological and virological quality of the treated wastewater. However, excellent bacteriological quality and improvement of the virological quality of the treated wastewater intended for recycling, agriculture, reuse and discharges into natural receiving environments were obtained by UV-C254 irradiation. The tertiary treatment by ultraviolet irradiation tested or other comparable processes are crucial and appeared necessary in all wastewater treatment plants to upgrade the microbiological quality of the effluents.
References
AFNOR (1992). Mise en œuvre des dispositifs d’assainissement autonome. Normalisation française, DTU, France64: 1
Aw, T. G., & Gin, K. Y. (2010). Environmental surveillance and molecular characterisation of human enteric viruses in tropical urban wastewater. Journal of Applied Microbiology, 109(2), 716–730. https://doi.org/10.1111/j.1365-2672.2010.04701.x.
Campos, C. J. A., Avant, J., Lowther, J., Till, D., & Lees, D. N. (2016). Human norovirus in untreated sewage and effluents from primary, secondary and tertiary treatment processes. Water Research, 103, 224–232. https://doi.org/10.1016/j.watres.2016.07.045.
Da Silva, A. K., Le Saux, J. C., Parnaudeau, S., Pommepuy, M., Elimelech, M., & Le Guyader, F. S. (2007). Evaluation of removal of noroviruses during wastewater treatment, using real-time reverse transcription-PCR: Different behaviours of genogroups I and II. Applied and Environmental Microbiology, 73, 7891–7897. https://doi.org/10.1128/AEM.01428-07.
Dai, H., Chen, W., & Lu, X. (2016). Applying multi-aim optimisation method for the activated sludge process: A review. Water Science Technology, 73(2), 223–235. https://doi.org/10.2166/wst.2015.489.
EPA. (1992). Environmental protection agency: Standard for the disposal of sewage sludge. Federal Register: Part, 503, 9387–9404.
Fioretti, J. M., Rocha, M. S., Fumian, T. M., Ginuino, A., da Silva, T. P., de Assis, M. R., et al. (2016). The occurrence of human Sapoviruses in wastewater and stool samples in Rio de Janeiro, Brazil. Journal of Applied Microbiology, 121(3), 855–862. https://doi.org/10.1111/jam.13205.
Hassen, A., Mahrouk, M., Ouzari, H., Cherif, M., Boudabous, A., & Damelincourt, J. J. (2000). UV disinfection of treated wastewater in a large-scale pilot plant and inactivation of selected bacteria in a laboratory UV device. Bioresource Technology, 74(2), 141–150. https://doi.org/10.1016/S0960-8524(99)00179-0.
Hassine-Zaafrane, M., Kaplon, J., Ben Salem, I., Sdiri-Loulizi, K., Sakly, N., Pothier, P., et al. (2015). Detection and genotyping of group A rotaviruses isolated from sewage samples in Monastir, Tunisia between April 2007 and April 2010. Journal of Applied Microbiology, 119(5), 1443–1453. https://doi.org/10.1111/jam.12920.
Hassine-Zaafrane, M., Sdiri-Loulizi, K., Kaplon, J., Ben Salem, I., Pothier, P., Aouni, M., et al. (2014). Molecular detection of human Noroviruses in influent and effluent samples from two biological sewage treatment plants near Monastir, Tunisia. Food Environmental Virology, 6, 125–131. https://doi.org/10.1007/s12560-014-9147-3.
Ibrahim, C., Cherif, N., Hammami, S., Pothier, P., & Hassen, A. (2015). Quantification and molecular characterisation of Norovirus after two wastewater treatment procedures. Water Air Soil Pollution, 226, 187–193. https://doi.org/10.1007/s11270-015-2402-x.
Ibrahim, C., Chérif, N., Hammami, S., Pothier, P., & Hassen, A. (2016). Quantification and genotyping of Rotavirus A within two wastewater treatment processes. Clean: Soil, Air, Water, 44(4), 393–401. https://doi.org/10.1002/clen.201400588.
Ibrahim, C., Hammami, S., Mejri, S., Mehri, I., Pothier, P., & Hassen, A. (2017a). Detection of Aichi virus genotype B in two lines of wastewater treatment processes. Microbial Pathogenesis, 109, 305–312. https://doi.org/10.1016/j.micpath.2017.06.001.
Ibrahim, C., Mehri, I., Hammami, S., Mejri, S., Hassen, A., & Pierre, P. (2017b). Removal of human astroviruses from hospital wastewater by two biological treatment methods: Natural oxidising lagoons and rotating biodisks. Desalination Water Treatment, 89, 287–296. https://doi.org/10.5004/dwt.201.721.356.
Ibrahim, C., Hassen, A., Pothier, P., Mejri, S., & Hammami, S. (2018). Molecular detection and genotypic characterisation of enteric adenoviruses in hospital wastewater. Environmental Science Pollution Research, 25, 1–11. https://doi.org/10.1007/s11356-018-1399-2.
Ibrahim, C., Hammami, S., Chérif, N., Mejri, S., Pierre, P., & Hassen, A. (2019). Detection of sapoviruses in two biological lines of Tunisian hospital wastewater treatment. International Journal of Environmental Health Research, 26, 1–14. https://doi.org/10.1080/09603123.2018.1546835.
The International Committee on Taxonomy of viruses. (2020). Retrieved July, 2019 from http://www.ictvonline.org/virusTaxonomy.asp.
Jothikumar, N., Lowther, J. A., Henshilwood, K., Lees, D. N., Hill, V. R., & Vinjé, J. (2005). Rapid and sensitive detection of noroviruses by using TaqMan based one-step reverse transcription-PCR assays and application to naturally contaminated shellfish samples. Applied and Environmental Microbiology, 71(4), 1870–1875. https://doi.org/10.1128/AEM.71.4.1870-1875.2005.
Kageyama, T., Shinohara, M., Uchida, K., Fukushi, S., Hoshino, F. B., Kojima, S., et al. (2004). Coexistence of multiple genotypes, including newly identified genotypes, in outbreaks of gastroenteritis due to norovirus in Japan. Journal of Clinical Microbiology, 42, 2988–2995. https://doi.org/10.1128/JCM.42.7.2988-2995.2004.
Kazama, S., Miura, T., Masago, Y., Konta, Y., Tohma, K., Manaka, T., et al. (2017). Environmental surveillance of norovirus genogroups I and II for sensitive detection of epidemic variants. Applied and Environmental Microbiology, 83(9), e03406–e3416. https://doi.org/10.1128/AEM.03406-16.
Kitajima, M., Iker, B. C., Pepper, I. L., & Gerba, C. P. (2014). Relative abundance and treatment reduction of viruses during wastewater treatment process identification of potential viral indicators. Science Total Environment, 488–489, 290–296. https://doi.org/10.1016/j.scitotenv.2014.04.087.
Kitajima, M., Rachmadi, A. T., Iker, B. C., Haramoto, E., & Gerba, C. P. (2018). Temporal variations in genotype distribution of human sapoviruses and aichi virus 1 in wastewater in Southern Arizona, United States. Journal of Applied Microbiology, 124(5), 1324–1332. https://doi.org/10.1111/jam.13712.
Kokkinos, P., Ziros, P., Meri, D., Filippidou, S., Kolla, S., Galanis, A., et al. (2011). An additional/alternative approach for virological surveillance in Greece? International Journal of Environmental Research and Public Health, 8(6), 1914–1922. https://doi.org/10.3390/ijerph8061914.
La Rosa, G., Pourshaban, M., Iaconelli, M., & Muscillo, M. (2010). Quantitative real-time of enteric viruses in influent and effluent samples from wastewater treatment plants in Italy. Annali dell' Istituto Superiore di Sanita, 46(3), 266–273. https://doi.org/10.4415/ANN_10_03_07.
Layman, W. H., Walsh, J. F., Kotch, J. B., Weber, D. J., Gunn, E., & Vinjé, J. (2009). A prospective study of etiologic agents of acute gastroenteritis outbreaks in child-care centres. Journal de Pediatria, 154(2), 253–257. https://doi.org/10.1016/j.jpeds.2008.07.057.
Lizasoain, A., Tort, L. F. L., García, M., Gillman, L., Alberti, A., Leite, J. P. G., et al. (2018). Human enteric viruses in a wastewater treatment plant: evaluation of activated sludge combined with UV disinfection process reveals different removal performances for viruses with different features. Letters in Applied Microbiology, 66(3), 215–221. https://doi.org/10.1111/lam.12839.
Mabasa, V. V., Meno, K. D., Taylor, M. B., & Mans, J. (2018). Environmental surveillance for noroviruses in selected South African wastewaters 2015–2016: Emergence of the novel GII.17. Food and Environmental Virology, 10(1), 16–28. https://doi.org/10.1007/s12560-017-9316-2.
Matthews, J. E., Dickey, B. W., Miller, R. D., Felzer, J. R., Dawson, B. P., Lee, A. S., et al. (2012). Epidemiology of published norovirus outbreaks: A review of risk factors associated with attack rates and genogroup. Epidemiology and Infection, 140, 1161–1172. https://doi.org/10.1017/S0950268812000234.
Mir-Tutusaus, J. A., Baccar, R., Caminal, G., & Sarrà, M. (2018). Can white-rot fungi be a real wastewater treatment alternative for organic micropollutants removal? A review. Water Research, 138, 137–151. https://doi.org/10.1016/j.watres.2018.02.056.
Myrmel, M., Lange, H., & Rimstad, E. (2015). A 1-year quantitative survey of noro-, adeno-, human boca—, and hepatitis E viruses in raw and secondarily treated sewage from two plants in Norway. Food and Environmental Virology, 7(3), 213–223. https://doi.org/10.1007/s12560-015-9200-x.
Prévost, B., Lucas, F. S., Goncalves, A., Richard, F., Moulin, L., & Wurtzer, S. (2015). A large-scale survey of enteric viruses in the river and wastewater underlines the health status of the local population. Environment International, 79, 42–50. https://doi.org/10.1016/j.envint.2015.03.004.
Qiu, Y., Lee, B. E., Neumann, N., Ashbolt, N., Craik, S., Maal-Bared, R., et al. (2015). Assessment of human virus removal during municipal wastewater treatment in Edmonton, Canada. Journal of Applied Microbiology, 119(6), 1729–1739. https://doi.org/10.1111/jam.12971.
Rodier, J. (1978). L’analyse de l’eau: Eaux Naturelles, Eaux résiduaires, Eaux de Mer (6th ed.). Paris: Dunod.
Sima, L. C., Schaefffer, J., Le Saux, J. C., Parnaudeau, S., Elimelech, M., & Le Guyader, F. S. (2011). Calicivirus removal in a membrane bioreactor wastewater treatment plant. Applied and Environmental Microbiology, 77(15), 5170–5177. https://doi.org/10.1128/AEM.00583-11.
Sano, D., Amarasiri, M., Hata, A., Watanabe, T., & Katayama, H. (2016). Risk management of infectious viral diseases in wastewater reclamation and reuse: Review. Environment International, 91, 220–229. https://doi.org/10.1016/j.envint.2016.03.001.
Schmitz, B. W., Kitajima, M., Campillo, M. E., Gerba, C. P., & Pepper, I. L. (2016). Virus reduction during advanced bardenpho and conventional wastewater treatment processes. Environmental Science Technology, 50(17), 9524–9532. https://doi.org/10.1021/acs.est.6b01384.
Sdiri-Loulizi, K., Gharbi-Khélifi, H., de Rougemont, A., Chouchane, S., Sakly, N., Ambert-Balay, K., et al. (2008). Acute infantile gastroenteritis associated with human enteric viruses in Tunisia. Journal of Clinical Microbiology, 46(4), 1349–1355. https://doi.org/10.1128/JCM.02438-07.
Sdiri-Loulizi, K., Hassine, M., Aouni, Z., Gharbi-Khelifi, H., Chouchane, S., Sakly, N., et al. (2010). Detection and molecular characterisation of enteric viruses in environmental samples in Monastir, Tunisia, between January 2003 and April 2007. Journal of Applied Microbiology, 109, 1093–1104. https://doi.org/10.1111/j.1365-2672.2010.04772.x.
Suffredini, E., Iaconelli, M., Equestre, M., Valdazo-González, B., Ciccaglione, A. R., Marcantonio, C., et al. (2018). Correction to: Genetic diversity among genogroup II noroviruses and Progressive emergence of GII.17 in wastewaters in Italy (2011–2016) revealed by next-Generation and Sanger sequencing. Food and Environmental Virology, 10(2), 222–223. https://doi.org/10.1007/s12560-017-9328-y.
Teixeira, D. M., Spada, P. K., Morais, L. L., Fumian, T. M., Lima, I. C., Oliveira, D. S., et al. (2017). Norovirus genogroups I and II in environmental water samples from Belém City, Northern Brazil. Journal of Water and Health, 15(1), 163–174. https://doi.org/10.2166/wh.2016.275.
Turki, Y., Mehri, I., Lajnef, R., Rejab, A. B., Khessairi, A., Cherif, H., et al. (2017). Biofilms in bioremediation and wastewater treatment: Characterisation of bacterial community structure and diversity during seasons in municipal wastewater treatment process. Environmental Science and Pollution Research, 24(4), 3519–3530. https://doi.org/10.1007/s11356-016-8090-2.
Victoria, M., Tort, L. F., Lizasoain, A., García, M., Castells, M., Berois, M., et al. (2016). Norovirus molecular detection in Uruguayan sewage samples reveals high genetic diversity and GII.4 variant replacements a long time. Journal of Applied Microbiology, 120(5), 1427–1435. https://doi.org/10.1111/jam.13058.
Zhou, N., Lin, X., Wang, S., Tao, Z., Xiong, P., Wang, H., et al. (2016). Molecular epidemiology of GI and GII noroviruses in sewage: 1-year surveillance in eastern China. Journal of Applied Microbiology, 121(4), 1172–1179. https://doi.org/10.1111/jam.13218.
Acknowledgements
This study received the support of the CERTE (Techno Park of Borj-Cédria, Tunisia) and the National Reference Centre of Enteric Viruses (Dijon, France).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ibrahim, C., Hammami, S., Khelifi, N. et al. The Effectiveness of Activated Sludge Procedure and UV-C254 in Norovirus Inactivation in a Tunisian Industrial Wastewater Treatment Plant. Food Environ Virol 12, 250–259 (2020). https://doi.org/10.1007/s12560-020-09434-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12560-020-09434-0






