Abstract
Prostate-specific membrane antigen (PSMA), also called glutamate carboxypeptidase II (GCP(II)), is a Zn-dependent metalloprotease that is known as a well prostate cancer indication and a potential targeting towards anti-cancer medicines and drug delivery. Because of its centrality in the diagnostics and treatment of prostate cancer, several types of inhibitors are designed with particular scaffolds. In this study, important groups of related inhibitors as well as reported experimental and computational studies are being reviewed, in which we examined three functional groups on each group of structures. The importance of computational biochemistry and the necessity of extensive research in this area on PSMA and its effective ligands are recommended.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Prostate-specific membrane antigen (PSMA), also known as glutamate carboxypeptidase II (GCPII), is a 750 amino acid transmembrane protein in the central nervous system, and as a surface membrane protein, it has a high degree of availability (Jones et al. 2020, Lutje et al. 2017). This type II transmembrane metallopeptidase catalyzes the configuration of N acetylaspartylglutamate (NAAG) to N acetylaspartate (NAA) and glutamate (Ferraris et al. 2012). It is really a Zn2+-dependent metalloprotease out from M28 peptidase group that is found on the cytoplasmic and apical surfaces of the prostate epithelium in benign prostatic cells. PSMA is shifted from the cytoplasm to the luminal surface of the prostatic channels when it undergoes malignant transformation, where that displays substrates with such a huge extracellular domain (Wright et al. 1995). It is also an important proposition for molecular imaging and targeted treatment utilizing highly specific radiolabeled PSMA ligands, in other words, inhibitors, owing to their strong and constant expression in PCa (Jones et al. 2020, Lutje et al. 2017).
The related ligands may now be synthesized using a variety of scaffold structures. Similar to glutamate, pentanedioic acid contains a zinc-binding group that almost always interacts with the catalytic zinc atom throughout the PSMA binding site and a substituent that generally resides either inside the S1 binding pocket or even inside of the protein that really extends to the substrate (Yang et al. 2016). Scaffolds consisting of several groups: phosphonates/phosphinates (Jackson et al. 2001), phosphoramidates (Maung et al. 2004), and ureas (Kozikowski et al. 2001) are efficient zinc-binding groups for binding related to GCPII. Furthermore, thiol (Majer et al. 2003) and hydroxamate (Stoermer et al. 2003) are known to be beneficial zinc-binding groups (Lutje et al. 2017). All the details related to three important general groups as effective inhibitors of PSMA, and related research studies will be discussed in this study.
GCPII structure
The ectodomain of GCPII is based on three interwoven domains (Davis et al. 2005). The extracellular part of GCPII folding into 3 different domains and residues arising from each of the three domains participate to substrate recognition by shaping the GCPII identification pocket (Fig. 1). The protease domain spans amino acids 57 to 116 and 352 to 590, and the apical domain protects the active site and forms the wide substrate binding tube with the protease domain. The GCPII structure contains two other inorganic ions in addition to two zinc ions: Cl− and Ca2+ (Hlouchová et al. 2012).
As well as the zinc ions, the active site of PSMA is containing a water molecule that has already been activated and interacts with the carbonyl oxygen of the inhibitors’ ureido motif (Pastorino et al. 2020). The binding site is required for PSMA’s hydrolytic activity, whereas inhibitors block the hydrolysis of enzyme and operate as an amide-bio isostere. In addition, the active site of the protein consists of two pockets: the S1′ pocket which is regarding the glutamate-sensing and the S1 pocket that is related to the non-pharmacophore site (Mesters et al. 2006; Bařinka et al. 2012) (Fig. 2). Furthermore, the “arginine patch,” which may transition among two different conformations and moreover specify the diameter of the S1 derivative pocket, is a key component of the S1 pocket (Barinka et al. 2002; Machulkin et al. 2016) (Fig. 2).
GCPII inhibitors are designed in a general way. The structure of the NAAG is used to design inhibitors of GCPII. A zinc-binding group (ZBG) in the GCPII inhibitors chelates the catalytic zinc ions in the active site. The glutamate moiety is linked to the zinc-binding group (ZBG) at position 2 (connector A). The residues at the S1′ site bind glutamate firmly. The hydrophilic subsite S1 binds an extra carboxyl group at the P1 position (linked through connector B). GCPII inhibitors are divided into three categories: phosphonate/phosphinate-based, urea-based, and thiol-based chemical structures
In addition, significant studies have been performed on mutated structures of GCPII. Mlochová et al. created and analyzed 12 GCPII mutants that targeted amino acids around substrate/inhibitor binding sites. The experimental findings, combined with molecular modeling, suggest that the amino acid residues delineating the S1′ pocket of enzyme, specifically Arg210, contribute primarily to the high affinity binding of GCPII substrates/inhibitors. However, the residues forming the S1 pocket may be more important for GCPII substrate specificity “fine-tuning” (Mlcochová et al. 2007). Klusák and colleagues also created a mutant of human GCPII (GCPII(E424A)) in which Glu424, a potential proton shuttle residue, is replaced with alanine to study peptide hydrolysis in greater detail. Considering N-Ac-Asp-Glu as a substrate, kinetic analysis of GCPII(E424A) demonstrated a complete loss of catalytic activity, implying that Glu424 is directly involved in peptide hydrolysis (Klusák et al. 2009). Furthermore, Barinka et al. in their research found that N-glycosylation is required for correct folding and subsequent secretion of human GCPII (Barinka et al. 2004). The predicted N-glycosylation sites are also critical for GCPII carboxypeptidase activity, according to the analysis. Researchers further reveal that an oligosaccharide moiety occupies all anticipated N-glycosylation sites and that glycosylation at sites other than the putative catalytic domain is crucial for GCPII’s NAAG-hydrolyzing activity, and it casts doubt on the validity of previously characterized structural models of GCPII (Barinka et al. 2004). Human glutamate carboxypeptidase II, on the other hand, operates as a folate hydrolase in the small intestine in addition to its well-known involvement in the central nervous system. A comparison of GCPII wild-type and His475Tyr variants’ thermal stabilities and folate-hydrolyzing activity was performed by Navrátil et al. (Navrátil et al. 2014). As more than just a conclusion, they mentioned that the crystallographic data show a lot about how polyglutamylated folates attach to GCPII, particularly the role of the arene binding site in recognizing the folic acid moiety. Furthermore, the structural and kinetic results imply that the wild-type and His475Tyr variants of GCPII are functionally equivalent (Navrátil et al. 2014). As a result, depending upon which region of the protein structure the mutation occurs in, it might impact the protein’s function and, as a result, modify the inhibitor’s action. In this study, we have tried to study different families of ligands and considering that the structure of the protein has no mutations.
GCPII inhibitors and ligands
Since inhibition of GCPII enzyme activity in the brain results in neuroprotection, selective GCPII inhibitors are becoming a significant field of GCPII-focused research. Furthermore, inhibitors can be employed as “homing devices” for GCPII-based prostate cancer imaging (Zhou et al. 2005; Rowe et al. 2016; Kiess et al. 2015). As a result, a large variety of GCPII inhibitors with various chemical scaffolds, which almost all of them originated from NAAG, have been reported during the last 20 years (Fig. 2).
As previously stated, GCPII is thought to be a viable target for the therapy of a variety of neurological illnesses related to glutamatergic neurotransmission dysfunction. In animal models of several of these illnesses, GCPII inhibition has also been demonstrated to be neuroprotective. As a result, one of the most obvious goals of structural investigations of GCPII is to gain insight into the active site, enabling the development of more specific and powerful inhibitors with acceptable pharmacokinetic features.
Adaptable oxoanions like phosphate and sulfate, as well as divalent metal ion chelators like EDTA and EGTA, were the first chemicals to be found to inhibit GCPII (Robinson et al. 1987). Other zinc-binding groups were explored for their capacity to inhibit GCPII in addition to compounds with phosphonate or phosphinate groups as active site zinc ion chelators. The thiol-based inhibitor (2-MPPA) was discovered to be not only powerful but also the first orally accessible GCPII inhibitor (Majer et al. 2003). Compounds having a zinc-binding group of hydroxamate were also investigated; however, they were shown to be less effective GCPII inhibitors than those based on phosphonate or thiol (Stoermer et al. 2003). Aside from the inhibitors listed above, urea-based compounds have also been shown to effectively inhibit GCPII (Kozikowski et al. 2001, 2004). In this study, three different types of inhibitors were investigated. Researchers in the field of drug design will benefit from having a thorough understanding of the various classes of PSMA protein inhibitors. The goal of this study is to review current investigations of these three main categories of PSMA-related inhibitors, with the hope that the findings will provide new ideas to continue in the field of design and synthesis of effective multifunctional compounds in both imaging and therapy, and it was discovered that urea-based inhibitors are the most efficient, while phosphorus and thiol-based inhibitors are less effective.
The main ligand-binding cavity of PSMA is separated into three groups (related to the zinc-binding group called ZBG): phosphorus-based compounds, e.g., phosphonates and phosphinates, urea-based structures, and thiols (Fig. 2).
Phosphorus (and its related structures)-based GCPII inhibitors
Phosphorus-containing inhibitors were the first GCPII inhibitors to be discovered, and they were essential in gaining a better knowledge of GCPII’s physiological activities (Haas et al. 2010). The tetrahedral phosphorus group resembles the cleaved peptide bond’s (tetrahedral) transition state.
Phosphinate and phosphonates inhibitors
When in 1996, the phosphonate-based GCPII inhibitor 2-(phosphonomethyl)pentanedioic acid (2-PMPA) was developed, it quickly became a benchmark in terms of its performance and efficiency (Haas et al. 2010). With the exception of 2-PMPA and the other phosphonates, a variety of phosphinate PSMA inhibitors have been reported (Su et al. 1995). Nonetheless, due to a lack of oral bioavailability, they have never achieved their potential as therapeutic agents, and research has moved in a new path, particularly into urea-based inhibitors (Chang et al. 2005). As a result, the publication of orally accessible 2-PMPA prodrugs in 2016 came as a surprise (Kalariti et al. 2004). The early studies on phosphonate and phosphate inhibitors, including the effective PSMA inhibitor 2-PMPA with IC50 0.9 nM along with thiol-based PSMA inhibitors, were done at ZENECA and afterward Guilford Pharmaceuticals (Jackson et al. 1996; Majer et al. 2003).
Phosphoramidate inhibitors as radiopharmaceutical compounds
Berkman’s group then performed extensive studies with phosphoramidate inhibitors (IC50s 0.5–20 nM). The effectiveness of inhibitors is often reported by criteria called IC50 or Ki. IC50 is the half-maximal inhibitory concentration. This criterion, in reality, assesses a substance’s ability to impede a certain biological or metabolic activity. (Anderson et al. 2007; Liu et al. 2008b; Maung et al. 2004; Foss et al. 2012). Agents designed to target prostate-specific membrane antigen (PSMA) are a fast-developing category of radiopharmaceuticals for prostate cancer diagnostic imaging, according to Behr and co-workers (Behr et al. 2019).
CTT1057 is a potential new phosphoramidate PSMA-targeting 18F-labeled PET radiopharmaceutical with comparable biodistribution to urea-based PSMA-targeted therapies. Kopka and his colleagues claimed that the novel PSMA radioligands had quite a significant influence on the clinical management of the disease (Kopka et al. 2017). One of several problems as to inhibitors of PSMA in terms of providing therapeutic payloads, according to Choy et al., is their fast urine excretion. They used a 177Lu-labeled phosphoramidate-based PSMA inhibitor (CTT1298) (Choy et al. 2017). Keeping this research going, Huang and Heston provided the list of PSMA inhibitors with an average to low molecular weight and addressed a critical question in the study mentioned by Choy et al. on the efficacy of Lutecium-177 labeled phosphoramidate-based PSMA inhibitors (Huang and Heston 2017). The issue was whether adding an albumin-binding entity to low-molecular-weight medicines would improve the efficacy of PSMA targeted treatment. Further, to obtain improvement in tumor absorption and to increase PSMA targeted anti-tumor action, Choy and his colleagues utilized a tiny molecule reversibly linked to a bigger protein, albumin (Huang and Heston 2017).
Dannoon and colleagues tested a variety of synthetic phosphoramidate-based PSMA inhibitors with varying lipophilicity, as well as their fluorine-18 analogs, as PET imaging agents for prostate cancer (Dannoon et al. 2016). A highly precise and accurate molecular imaging agent or technique for classifying the patient with PCa was a major therapeutic need, according to Mease et al. (Mease et al. 2013). Nedrow-Byers et al. showed that copper-free link chemistry can easily build a PSMA-targeted SPECT agent (Nedrow-Byers et al. 2013). An irreversible phosphoramidate inhibitor, CTT-54, was also improved in one of Nedrow-Byers et al.’s studies to transport 99mTc-(CO)3-DTPA as a SPECT imaging payload to PSMA + cells in vivo and in vitro (Nedrow-Byers et al. 2012).
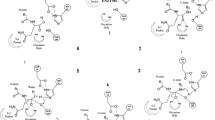
The chemical combination of Cy5.5 N-hydroxysuccinimide ester (Cy5.5-NHS) with a powerful PSMA inhibitor CTT-54.2 resulted in the development of a close fluorescent imaging probe (Cy5.5-CTT-54.2) in the research by Liu et al. (Liu et al. 2010). The goal of the study by Lapi et al. was to investigate phosphoramidates as a novel classification of effective PSMA inhibitors with better selectivity and approval characteristics (Lapi et al. 2009). Several findings that discovered which phosphoramidate peptidomimetic inhibitors of PSMA can be divided into three categories by Liu et al.: pseudoirreversible, moderately reversible, and rapidly reversible inhibitors (Liu et al. 2008a). According to the outcomes of such tests, the development of pseudo-irreversible PSMA inhibitors is likely to open up novel research and treatment possibilities for PCa and neurological diseases. Several examples of this type of inhibitor are shown in Fig. 3.
Urea-based GCPII inhibitors
The most utilized category of selective GCPII inhibitors is urea-based inhibitors, which were discovered throughout the twenty-first century (Kozikowski et al. 2001, 2004). The inhibitors typically require a glutamate residue that binds to the S1′ pocket of the enzyme, while the ureido group replicates the planar peptide bond of cleaved substrate (Ferraris et al. 2012; Chen et al. 2008). As a result, several other urea-based inhibitors (radionuclides, fluorophores, and poisons are all linked in some way) have been synthesized and effectively employed in prostate cancer experimental imaging and treatment (Zhou et al. 2005; Eder et al. 2012; Foss et al. 2005). DCIBzL, which has a phenyl ring that binds to the hydrophobic pocket at the S1 site, is a great example of this kind of molecule and one of the most powerful GCPII inhibitors (Chen et al. 2008).
Kozikowski et al. studied some simple substances to serve as powerful urea-based GCPII inhibitors (Kozikowski et al. 2004). Chandran et al. employed a combination of methods to coat a nanoparticle’s surface using a peptidomimetic inhibitor of PSMA depending on urea (Chandran et al. 2008). The extremely effective acyclic Ga (III) chelator N,N′-bis [2-hydroxy-5-(carboxyethyl)benzyl] ethylenediamine N,N′- diacetic acid (HBED-CC) was added as a lipophilic side chain in the hydrophilic pharmacophore and was observed to interact positively with the PSMA “active binding site” by Eder and co-workers (Eder et al. 2012). To improve binding characteristics and pharmacokinetics, Schäfer et al. used the 68 Ga chelator N, N′-bis[2-hydroxy-5(carboxyethyl)benzyl] ethylenediamine-N, N′-diacetic acid (HBED-CC) to dimerize the pharmacophore Glu-ureido-Lys (Schäfer et al. 2012). Scientists indicated that using a specially designed linker, the pharmacokinetics of tracers with the Glu-urea-based binding motif might be even better. Wüstemann et al. looked examined how the chelator moiety may affect pharmacokinetics, including tumor cell internalization (Wustemann et al. 2016). The findings suggest that drugs containing the chelator CHX-A”-DTPA uses a Glu-urea-based binding site in combination with hydrophobic linkers might be useful in the treatment of PCa. Zha et al. presented a new [68 Ga]-Glu-NH-CO–NH-Lys (Ahx)-linker-HBED-CC conjugate using a unique O-(carboxymethyl)-L-tyrosine as like the collection of linkers in order to build innovative agents with improved characteristics for PET imaging (Zha et al. 2018). The first findings strongly imply that [68 Ga] might be a good choice to detect PSMA expression in PCa using PET imaging. PSMA I&T, a theranostic tracer improved by Wirtz et al., was improved by altering the peptidic structure in an attempt to optimize PSMA binding and internalization in PSMA-expressing tumor cells (Wirtz et al. 2018). Giesel et al. also provided an intraindividual study of tracer-specific features of 18F-DCFPyL against 18F-PSMA-1007 in their clinical research (Giesel et al. 2018).
The production of isocyanate intermediates and urea linkages are the first two stages in the synthesis of urea-based PSMA inhibitors. According to Mosayebnia and colleagues, the isocyanate is produced in the liquid phase and subsequently interacts with the amine in the liquid phase or attached to the solid phase to establish the urea connection (Mosayebnia et al. 2018). Moreover, Mosayebnia et al. developed novel 99mTc-labeled peptides as a PSMA inhibitors for the finding of selective PCa inhibitors at a preliminary phase in another investigation (Mosayebnia et al. 2020). Glutamate-ureido inhibitors with labels seem to be a very often PSMA-targeting agents for nuclear medicine application fields, according to Pastorino et al. (Pastorino et al. 2020). Thus, recently, nuclear imaging tools and radiotherapeutics have been designed and tested, and more other most common PSMA-targeting drugs for nuclear medicine purposes are identified glutamate-ureido inhibitors.
In terms of clinical practice, PSMA is a possible candidate for both diagnostics and radioligand treatment (RLT) of prostate cancer, as Tateishi explains in a review study (Tateishi 2020). More research is needed to evaluate the diagnostic usefulness of PSMA-ligand PET for PCa (Felber et al. 2021). There are some examples of this kind of inhibitors in Fig. 4.
Thiol-based and other GCPII inhibitors
2-MPPA, a powerful thiol-based GCPII inhibitor
Thiol-based GCPII inhibitors are distinguished by their availability in the mouth (oral bioavailability). As a matter of fact, they were created in reaction to the phosphorus-based GCPII inhibitors’ poor pharmacokinetic profile and high polarity. In 2003, 2-(3-mercaptopropyl) pentanedioic acid (2-MPPA), a powerful thiol-based GCPII inhibitor, was discovered. 2-MPPA has been found to be orally accessible in rats (Majer et al. 2003) and, more significantly, effective in a variety of illnesses in association with models of animals, including neuropathic pain (Majer et al. 2003), diabetic neuropathy (Zhang et al. 2006), and related muscular dystrophy (Ghadge et al. 2003). 2-MPPA was tested in phase I clinical research (van der Post et al. 2005), but progress was suspended due to the animal toxicity. In general, compounds based on thiols are not good medicines since this group of inhibitors could be easily oxidized.
Hydroxamate compounds
Another potential zinc-binding group is hydroxamate compounds (Novakova et al. 2016b; Stoermer et al. 2003). Inhibitors based on hydroxamic acid with human GCPII nanomolar sensitivity have recently been found. They have a novel binding mechanism that contains a glutamates-like moiety which binds to the entering funnel rather than the S1′ pocket (Novakova et al. 2016b).
Stoermer et al. developed hydroxamic acids in a sequence as possible inhibitors of PSMA in a research investigation (Stoermer et al. 2003). They expanded their structure–activity relationship (SAR) analyses to include other ZBG and discovered that phosphinate and thiol-based PSMA inhibitors were effective in animal studies of a variety of neurological diseases. Then, he and other colleagues also developed a variety of thiol-based PSMA inhibitors using a scaffold of 3-(mercaptomethyl) benzoic acid or 2-(2-mercaptoethyl) benzoic acid in additional investigations. Majer et al. prepared a variety of 2-(thioalkyl)pentanedioic acids along with PSMA inhibitors (Stoermer et al. 2003). The number of methylene units between the thiol group and pentanedioic acid was discovered to affect the inhibitory efficacy of these thiol-based drugs towards PSMA. In previous investigations, Majer and co-workers developed and evaluated a variety of thiol-based inhibitors with a benzyl moiety for their ability to inhibit PSMA.
Takatsu et al. investigated the effects of 2-(3-mercaptopropyl) pentanedioic acid (2-MPPA), a novel PSMA inhibitor that may be taken orally, on impairments in prepulse inhibition (PPI) following injection of the N methyl-D-aspartate (NMDA) receptor antagonist dizocilpine (Takatsu et al. 2011). GCP II inhibition may be a therapy option that works for schizophrenia, according to their findings. Ferraris and colleagues developed-thiolactones as prodrugs from thiol-based PSMA inhibitors (Ferraris et al. 2014). The pharmacological of several radiopharmaceuticals utilized for the theranostic therapy of PCa was highlighted in research by Vahidfar et al. (Vahidfar et al. 2019). Some examples of this type of inhibitor are shown in Fig. 5.
Studies in the field of computational biochemistry
Wu et al. developed a brief digital collection, which was displayed the inhibitory efficacy versus PSMA to find the best pharmacophores from a phosphoramidate peptidomimetic inhibitor of PSMA. Computational docking was utilized to suggest that PSMA active site has a pharmacophore description, based on information on enzyme inhibition and the recombinant a new X-ray crystal structure of the protein (Wu et al. 2007). In the other case, Wu et al. developed and tested a small analog library for inhibitory efficacy towards PSMA in order to find the pharmacophore of a phosphoramidate peptidomimetic inhibitor of PSMA. The lead inhibitor’s structure is based on N-acyl derivatives and includes a phosphoramidate group that has some important interactions with the active site of the PSMA, containing two zinc atoms. Docking studies were employed to suggest a pharmacophore model (Wu et al. 2007). Also, Wu et al. created six glutamate-containing phosphoramidate derivatives of different hydroxysteroids. Individual compounds in the collection had inhibitory potencies equivalent to a simple phenyl alkyl analog. Molecular docking was utilized in this research to get the binding energy (Wu et al. 2008).
Phosphoramidate peptidomimetic inhibitors of PSMA may be divided into three categories, according to Liu et al.’s findings: pseudoirreversible, moderately reversible, and quickly reversible inhibitors (Liu et al. 2008a). The development of pseudoirreversible PSMA inhibitors is likely to open up novel research and treatment possibilities for PCa patients. RNA aptamers, according to Rockey et al., are a new class of medicines with a huge future for prostate cancer diagnostics and therapy. They employed a “rational truncation” method guided by RNA structure determination and protein/RNA docking algorithms (Rockey et al. 2011). Novakova et al. conducted a detailed structural and computational analysis aiming at determining the role of the effector function in PSMA binding and affinity (Novakova et al. 2016a). They achieved this by determining the crystal structures of human GCPII in combination with a variety of phosphoramidate-based inhibitors. As a result, their findings indicate that phosphoramidates had better binding affinities than matching phosphonates.
Naushad et al. used molecular visualization software to develop models of different versions applying the crystal structure of PSMA as a pattern to establish a possible future inhibitor, that all eight prevalent genetic variations have been reported to be effective (Naushad et al. 2016). Pandit et al. used computational methods to discover active sites and interactions of urea-based PSMA inhibitors with the protein by altering the core structure of the ligand (Pandit et al. 2018). A novel PSMA inhibitor was also created to confirm the in silico study, and they were able to effectively test the three-dimensional quantitative SAR (3D-QSAR) and molecular docking-based development of the PSMA inhibitors. Sharma and Baruah summarized the most frequently reported dysregulated miRNAs in PCa from the literature and reviewed the already available evidence in a review (Sharma and Baruah 2019). Differentially expressed genes (DEGs) in prostate cancer were discovered using a combined bioinformatics technique (Baruah and Sharma 2019).
Ivanenkov et al. developed and synthesized a PSMA-specific small-molecule carrier loaded with Doxorubicin for a preliminary biological assessment (Dox) (Ivanenkov et al. 2019). A 3D molecular docking research was also carried out to clarify the exact principle and mechanism of binding and to improve the target affinity by further optimizing the linker region. Glu-urea-Lys-based PSMA-targeting conjugates with paclitaxel were developed, according to the study by Machulkin et al. (Machulkin et al. 2019). A number of novel PSMA-targeting conjugates containing paclitaxel have been developed and produced. Finally, 3D-molecular docking research was carried out, too. Abdullahi et al. conducted in silico modeling investigations on some unique inhibitors of prostate cancer (PC3) cell lines employing C14-urea-tetrandrine components (Abdullahi et al. 2020). They used the DFT and QSAR models to optimize each structure. Their findings following computational studies might contribute to the development and production of novel C14-urea-tetrandrines with improved inhibitory properties against the PC3 prostate cell line. In silico docking studies were used to evaluate a collection of peptides holding such a well Glu-Urea-Lys pharmacophore and PSMA inhibitor using only new non-urea functional groups for accurate PCa identification at a preliminary phase by Mosayebnia and colleagues (Mosayebnia et al. 2020). LLE (Liquid–liquid extraction) in flow-based 45Ti purification, using computer-aided design, and the manufacturing of a salan-natTi/45Ti-chelidamic acid (CA)-PSMA ligand comprising the Glu-urea-Lys pharmacophore were presented by Pedersen et al. (Søborg Pedersen et al. 2020). In our recent study, new compounds as urea-based inhibitors were proposed by using the CADD method including molecular dynamic simulation and docking study (Nikfarjam et al. 2021).
Over the last few years, GCPII crystal structures have been explored in a variety of methods utilizing a range of ligands, including GCPII inhibitors based on phosphorus and urea. These discoveries give insight on the structural features of each of GCPII’s key binding sites, as well as the potential for developing novel inhibitors. In Table 1, GCPII-related structures in RCSB database are provided.
Conclusion
PSMA is the most attractive proposition for researchers looking to use nuclear medicine techniques to detect and treat individuals with PCa. Reviewing the studies performed on each group of important PSMA inhibitors, it was observed that the research process is being carried out with considerable speed and accuracy by experts and researchers. Considering that the protein considered in these studies is a metalloprotein and due to the range of appropriate computational tools and packages for the study of metalloproteins, it is suggested that researchers in computational biochemistry further study each group of inhibitors that are introduced in this review. Researchers will also be able to improve the process of developing and conducting research to find suitable inhibitors by using drug design and drug delivery methods in computational chemistry and computational biochemistry. In addition, the reported findings in the literature review reveal a variety of inhibitor binding mechanisms inside the non-prime site(s) of GCPII, which might be used to develop new GCPII-specific drugs. In addition, by using the integration of quantum mechanics and molecular dynamics techniques, the mechanism of action of each group of inhibitors is studied as much as possible. Then, using new programming methods in the field of drug design such as machine learning (ML), the need for biochemical calculations along with laboratory and clinical research can be addressed.
References
Abdullahi M, Uzairu A, Shallangwa GA, Mamza P, Arthur DE, Ibrahim MT (2020) In-silico modelling studies on some C14-urea-tetrandrine derivatives as potent anti-cancer agents against prostate (PC3) cell line. J King Saud Univ Sci 32:770–779. https://doi.org/10.1016/j.jksus.2019.01.008
Anderson MO, Wu LY, Santiago NM, Moser JM, Rowley JA, Bolstad ES, Berkman CE (2007) Substrate specificity of prostate-specific membrane antigen. Bioorg Med Chem 15:6678–6686. https://doi.org/10.1016/j.bmc.2007.08.006
Barinka C, Byun Y, Dusich CL, Banerjee SR, Chen Y, Castanares M, Kozikowski AP, Mease RC, Pomper MG, Lubkowski J (2008b) Interactions between human glutamate carboxypeptidase II and urea-based inhibitors: structural characterization. J Med Chem 51:7737–7743. https://doi.org/10.1021/jm800765e
Barinka C, Hlouchova K, Rovenska M, Majer P, Dauter M, Hin N, Ko Y-S, Tsukamoto T, Slusher BS, Konvalinka J, Lubkowski J (2008b) Structural basis of interactions between human glutamate carboxypeptidase II and its substrate analogs. J Mol Biol 376:1438–1450. https://doi.org/10.1016/j.jmb.2007.12.066
Barinka C, Rinnová M, Sácha P, Rojas C, Majer P, Slusher BS, Konvalinka J (2002) Substrate specificity, inhibition and enzymological analysis of recombinant human glutamate carboxypeptidase II. J Neurochem 80:477–87. https://doi.org/10.1046/j.0022-3042.2001.00715.x
Bařinka C, Rojas C, Slusher B, Pomper M (2012) Glutamate carboxypeptidase II in diagnosis and treatment of neurologic disorders and prostate cancer. Curr Med Chem 19:856–870. https://doi.org/10.2174/092986712799034888
Bařinka C, Rovenská M, Mlčochová P, Hlouchová K, Plechanovová A, Majer P, Tsukamoto T, Slusher BS, Konvalinka J, Lubkowski J (2007) Structural Insight into the pharmacophore pocket of human glutamate carboxypeptidase II. J Med Chem 50:3267–3273. https://doi.org/10.1021/jm070133w
Barinka C, Sácha P, Sklenár J, Man P, Bezouska K, Slusher BS, Konvalinka J (2004) Identification of the N-glycosylation sites on glutamate carboxypeptidase II necessary for proteolytic activity. Protein Sci 13:1627–1635. https://doi.org/10.1110/ps.04622104
Baruah MM, Sharma N (2019) In silico identification of key genes and signaling pathways targeted by a panel of signature microRNAs in prostate cancer. Med Oncol 36:43. https://doi.org/10.1007/s12032-019-1268-y
Behr SC, Aggarwal R, Vanbrocklin HF, Flavell RR, Gao K, Small EJ, Blecha J, Jivan S, Hope TA, Simko JP, Kurhanewicz J, Noworolski SM, Korn NJ, de los Santos R, Cooperberg MR, Carroll PR, Nguyen HG, Greene KL, Langton-Webster B, Berkman CE, Seo Y, (2019) Phase I study of CTT1057, an (18)F-labeled imaging agent with phosphoramidate core targeting prostate-specific membrane antigen in prostate cancer. J Nucl Med 60:910–916. https://doi.org/10.2967/jnumed.118.220715
Chandran SS, Banerjee SR, Mease RC, Pomper MG, Denmeade SR (2008) Characterization of a targeted nanoparticle functionalized with a urea-based inhibitor of prostate-specific membrane antigen (PSMA). Cancer Biol Ther 7:974–982. https://doi.org/10.4161/cbt.7.6.5968
Chang HJ, Yoo BC, Lim SB, Jeong SY, Kim WH, Park JG (2005) Metabotropic glutamate receptor 4 expression in colorectal carcinoma and its prognostic significance. Clin Cancer Res 11:3288–3295. https://doi.org/10.1158/1078-0432.CCR-04-1912
Chen Y, Foss CA, Byun Y, Nimmagadda S, Pullambhatla M, Fox JJ, Castanares M, Lupold SE, Babich JW, Mease RC, Pomper MG (2008) Radiohalogenated prostate-specific membrane antigen (PSMA)-based ureas as imaging agents for prostate cancer. J Med Chem 51:7933–7943. https://doi.org/10.1021/jm801055h
Choy CJ, Ling X, Geruntho JJ, Beyer SK, Latoche JD, Langton-Webster B, Anderson CJ, Berkman CE (2017) (177)Lu-labeled phosphoramidate-based PSMA inhibitors: the effect of an albumin binder on biodistribution and therapeutic efficacy in prostate tumor-bearing mice. Theranostics 7:1928–1939. https://doi.org/10.7150/thno.18719
Dannoon S, Ganguly T, Cahaya H, Geruntho JJ, Galliher MS, Beyer SK, Choy CJ, Hopkins MR, Regan M, Blecha JE, Skultetyova L, Drake CR, Jivan S, Barinka C, Jones EF, Berkman CE, Vanbrocklin HF (2016) Structure–activity relationship of 18F-labeled phosphoramidate peptidomimetic prostate-specific membrane antigen (PSMA)-targeted inhibitor analogues for PET imaging of prostate cancer. J Med Chem 59:5684–5694. https://doi.org/10.1021/acs.jmedchem.5b01850
Davis MI, Bennett MJ, Thomas LM, Bjorkman PJ (2005) Crystal structure of prostate-specific membrane antigen, a tumor marker and peptidase. Proc Natl Acad Sci 102:5981. https://doi.org/10.1073/pnas.0502101102
Eder M, Schäfer M, Bauder-Wust U, Hull WE, Wängler C, Mier W, Haberkorn U, Eisenhut M (2012) 68Ga-complex lipophilicity and the targeting property of a urea-based PSMA inhibitor for PET imaging. Bioconjug Chem 23:688–97. https://doi.org/10.1021/bc200279b
Felber VB, Valentin MA, Wester H-J (2021) Design of PSMA ligands with modifications at the inhibitor part: an approach to reduce the salivary gland uptake of radiolabeled PSMA inhibitors? EJNMMI Radiophar Chem 6:10. https://doi.org/10.1186/s41181-021-00124-1
Ferraris DV, Majer P, Ni C, Slusher CE, Rais R, Wu Y, Wozniak KM, Alt J, Rojas C, Slusher BS, Tsukamoto T (2014) δ-Thiolactones as prodrugs of thiol-based glutamate carboxypeptidase II (GCPII) inhibitors. J Med Chem 57:243–247. https://doi.org/10.1021/jm401703a
Ferraris DV, Shukla K, Tsukamoto T (2012) Structure-activity relationships of glutamate carboxypeptidase II (GCPII) inhibitors. Curr Med Chem 19:1282–1294. https://doi.org/10.2174/092986712799462658
Foss CA, Mease RC, Cho SY, Kim HJ, Pomper MG (2012) GCPII imaging and cancer. Curr Med Chem 19:1346–1359. https://doi.org/10.2174/092986712799462612
Foss CA, Mease RC, Fan H, Wang Y, Ravert HT, Dannals RF, Olszewski RT, Heston WD, Kozikowski AP, Pomper MG (2005) Radiolabeled small-molecule ligands for prostate-specific membrane antigen: in vivo imaging in experimental models of prostate cancer. Clin Cancer Res 11:4022–4028. https://doi.org/10.1158/1078-0432.CCR-04-2690
Ghadge GD, Slusher BS, Bodner A, Canto MD, Wozniak K, Thomas AG, Rojas C, Tsukamoto T, Majer P, Miller RJ, Monti AL, Roos RP (2003) Glutamate carboxypeptidase II inhibition protects motor neurons from death in familial amyotrophic lateral sclerosis models. Proc Natl Acad Sci U S A 100:9554–9559. https://doi.org/10.1073/pnas.1530168100
Giesel FL, Will L, Lawal I, Lengana T, Kratochwil C, Vorster M, Neels O, Reyneke F, Haberkon U, Kopka K, Sathekge M (2018) Intraindividual comparison of (18)F-PSMA-1007 and (18)F-DCFPyL PET/CT in the prospective evaluation of patients with newly diagnosed prostate carcinoma: a pilot study. J Nucl Med 59:1076–1080. https://doi.org/10.2967/jnumed.117.204669
Haas HS, Linecker A, Pfragner R, Sadjak A (2010) Peripheral glutamate signaling in head and neck areas. Head Neck 32:1554–1572. https://doi.org/10.1002/hed.21438
Hlouchová K, Bařinka C, Konvalinka J (2012) Chapter 3 Glutamate carboxypeptidase II as a therapeutic target. Proteinases as Drug Targets. Royal Soc Chem. https://doi.org/10.1039/9781849733151-00062
Huang SS, Heston WDW (2017) Should low molecular weight PSMA targeted ligands get bigger and use albumin ligands for PSMA targeting? Theranostics 7:1940–1941. https://doi.org/10.7150/thno.20284
Ivanenkov YA, Machulkin AE, Garanina AS, Skvortsov DA, Uspenskaya AA, Deyneka EV, Trofimenko AV, Beloglazkina EK, Zyk NV, Koteliansky VE, Bezrukov DS, Aladinskaya AV, Vorobyeva NS, Puchinina MM, Riabykh GK, Sofronova AA, Malyshev AS, Majouga AG (2019) Synthesis and biological evaluation of Doxorubicin-containing conjugate targeting PSMA. Bioorg Med Chem Lett 29:1246–1255. https://doi.org/10.1016/j.bmcl.2019.01.040
Jackson PF, Cole DC, Slusher BS, Stetz SL, Ross LE, Donzanti BA, Trainor DA (1996) Design, synthesis, and biological activity of a potent inhibitor of the neuropeptidase N-acetylated alpha-linked acidic dipeptidase. J Med Chem 39:619–622. https://doi.org/10.1021/jm950801q
Jackson PF, Tays KL, Maclin KM, Ko Y-S, Li W, Vitharana D, Tsukamoto T, Stoermer D, Lu X-CM, Wozniak K, Slusher BS (2001) Design and pharmacological activity of phosphinic acid based NAALADase inhibitors. J Med Chem 44:4170–4175. https://doi.org/10.1021/jm0001774
Jones W, Griffiths K, Barata PC, Paller CJ (2020) PSMA theranostics: review of the current status of PSMA-targeted imaging and radioligand therapy. Cancers (Basel) 12. https://doi.org/10.3390/cancers12061367
Kalariti N, Lembessis P, Koutsilieris M (2004) Characterization of the glutametergic system in MG-63 osteoblast-like osteosarcoma cells. Anticancer Res 24:3923–3929. https://ar.iiarjournals.org/content/24/6/3923.short/. Accessed Oct 2004
Kiess AP, Banerjee SR, Mease RC, Rowe SP, Rao A, Foss CA, Chen Y, Yang X, Cho SY, Nimmagadda S, Pomper MG (2015) Prostate-specific membrane antigen as a target for cancer imaging and therapy. Q J Nucl Med Mol Imaging 59:241–268. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4859214/. Accessed July 2015
Klusák V, Bařinka C, Plechanovová A, Mlčochová P, Konvalinka J, Rulíšek L, Lubkowski J (2009) Reaction mechanism of glutamate carboxypeptidase II revealed by mutagenesis, X-ray crystallography, and computational methods. Biochemistry 48:4126–4138. https://doi.org/10.1021/bi900220s
Kopka K, Benešová M, Bařinka C, Haberkorn U, Babich J (2017) Glu-ureido-based inhibitors of prostate-specific membrane antigen: lessons learned during the development of a novel class of low-molecular-weight theranostic radiotracers. J Nucl Med 58:17s–26s. https://doi.org/10.2967/jnumed.116.186775
Kozikowski AP, Nan F, Conti P, Zhang J, Ramadan E, Bzdega T, Wroblewska B, Neale JH, Pshenichkin S, Wroblewski JT (2001) Design of remarkably simple, yet potent urea-based inhibitors of glutamate carboxypeptidase II (NAALADase). J Med Chem 44:298–301. https://doi.org/10.1021/jm000406m
Kozikowski AP, Zhang J, Nan F, Petukhov PA, Grajkowska E, Wroblewski JT, Yamamoto T, Bzdega T, Wroblewska B, Neale JH (2004) Synthesis of urea-based inhibitors as active site probes of glutamate carboxypeptidase II: efficacy as analgesic agents. J Med Chem 47:1729–1738. https://doi.org/10.1021/jm0306226
Lapi SE, Wahnishe H, Pham D, Wu LY, Nedrow-Byers JR, Liu T, Vejdani K, Vanbrocklin HF, Berkman CE, Jones EF (2009) Assessment of an 18F-labeled phosphoramidate peptidomimetic as a new prostate-specific membrane antigen-targeted imaging agent for prostate cancer. J Nucl Med 50:2042–2048. https://doi.org/10.2967/jnumed.109.066589
Liu T, Toriyabe Y, Kazak M, Berkman CE (2008a) Pseudoirreversible inhibition of prostate-specific membrane antigen by phosphoramidate peptidomimetics. Biochemistry 47:12658–12660. https://doi.org/10.1021/bi801883v
Liu T, Wu LY, Hopkins MR, Choi JK, Berkman CE (2010) A targeted low molecular weight near-infrared fluorescent probe for prostate cancer. Bioorg Med Chem Lett 20:7124–7126. https://doi.org/10.1016/j.bmcl.2010.09.057
Liu T, Wu LY, Kazak M, Berkman CE (2008b) Cell-surface labeling and internalization by a fluorescent inhibitor of prostate-specific membrane antigen. Prostate 68:955–964. https://doi.org/10.1002/pros.20753
Lütje S, Slavik R, Fendler W, Herrmann K, Eiber M (2017) PSMA ligands in prostate cancer—probe optimization and theranostic applications. Methods 130:42–50. https://doi.org/10.1016/j.ymeth.2017.06.026
Machulkin AE, Ivanenkov YA, Aladinskaya AV, Veselov MS, Aladinskiy VA, Beloglazkina EK, Koteliansky VE, Shakhbazyan AG, Sandulenko YB, Majouga AG (2016) Small-molecule PSMA ligands. Current state SAR and Perspectives. J Drug Target 24:679–693. https://doi.org/10.3109/1061186X.2016.1154564
Machulkin AE, Skvortsov DA, Ivanenkov YA, Ber AP, Kavalchuk MV, Aladinskaya AV, Uspenskaya AA, Shafikov RR, Plotnikova EA, Yakubovskaya RI, Nimenko EA, Zyk NU, Beloglazkina EK, Zyk NV, Koteliansky VE, Majouga AG (2019) Synthesis and biological evaluation of PSMA-targeting paclitaxel conjugates. Bioorg Med Chem Lett 29:2229–2235. https://doi.org/10.1016/j.bmcl.2019.06.035
Majer P, Jackson PF, Delahanty G, Grella BS, Ko Y-S, Li W, Liu Q, Maclin KM, Poláková J, Shaffer KA, Stoermer D, Vitharana D, Wang EY, Zakrzewski A, Rojas C, Slusher BS, Wozniak KM, Burak E, Limsakun T, Tsukamoto T (2003) Synthesis and biological evaluation of thiol-based inhibitors of glutamate carboxypeptidase II: discovery of an orally active GCP II inhibitor. J Med Chem 46:1989–1996. https://doi.org/10.1021/jm020515w
Maung J, Mallari JP, Girtsman TA, Wu LY, Rowley JA, Santiago NM, Brunelle AN, Berkman CE (2004) Probing for a hydrophobic a binding register in prostate-specific membrane antigen with phenylalkylphosphonamidates. Bioorg Med Chem 12:4969–4979. https://doi.org/10.1016/j.bmc.2004.06.031
Mease RC, Foss CA, Pomper MG (2013) PET imaging in prostate cancer: focus on prostate-specific membrane antigen. Curr Top Med Chem 13:951–962. https://doi.org/10.2174/1568026611313080008
Mesters JR, Barinka C, Li W, Tsukamoto T, Majer P, Slusher BS, Konvalinka J, Hilgenfeld R (2006) Structure of glutamate carboxypeptidase II, a drug target in neuronal damage and prostate cancer. Embo j 25:1375–1384. https://doi.org/10.1038/sj.emboj.7600969
Mesters JR, Henning K, Hilgenfeld R (2007) Human glutamate carboxypeptidase II inhibition: structures of GCPII in complex with two potent inhibitors, quisqualate and 2-PMPA. Acta Crystallogr D Biol Crystallogr 63:508–513. https://doi.org/10.1107/S090744490700902X
Mlcochová P, Plechanovová A, Barinka C, Mahadevan D, Saldanha JW, Rulísek L, Konvalinka J (2007) Mapping of the active site of glutamate carboxypeptidase II by site-directed mutagenesis. Febs J 274:4731–41. https://doi.org/10.1111/j.1742-4658.2007.06021.x
Mosayebnia M, Hajimahdi Z, Beiki D, Rezaeianpour M, Hajiramezanali M, Geramifar P, Sabzevari O, Amini M, Hatamabadi D, Shahhosseini S (2020) Design, synthesis, radiolabeling and biological evaluation of new urea-based peptides targeting prostate specific membrane antigen. Bioorganic Chemistry 99:103743. https://doi.org/10.1016/j.bioorg.2020.103743
Mosayebnia M, Rezaeianpour S, Rikhtechi P, Hajimahdi Z, Beiki D, Kobarfard F, Sabzevari O, Amini M, Abdi K, Shahhosseini S (2018) Novel and efficient method for solid phase synthesis of urea-containing peptides targeting prostate specific membrane antigen (PSMA) in comparison with current methods. Iran J Pharm Res 17:917–926. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6094430/. Accessed May 2018
Naushad SM, Janaki Ramaiah M, Stanley BA, Prasanna Lakshmi S, Vishnu Priya J, Hussain T, Alrokayan SA, Kutala VK (2016) In silico approaches to identify the potential inhibitors of glutamate carboxypeptidase II (GCPII) for neuroprotection. J Theor Biol 406:137–142. https://doi.org/10.1016/j.jtbi.2016.07.016
Navrátil M, Ptáček J, Šácha P, Starková J, Lubkowski J, Bařinka C, Konvalinka J (2014) Structural and biochemical characterization of the folyl-poly-γ-l-glutamate hydrolyzing activity of human glutamate carboxypeptidase II. Febs j 281:3228–42. https://doi.org/10.1111/febs.12857
Nedrow-Byers JR, Jabbes M, Jewett C, Ganguly T, He H, Liu T, Benny P, Bryan JN, Berkman CE (2012) A phosphoramidate-based prostate-specific membrane antigen-targeted SPECT agent. Prostate 72:904–912. https://doi.org/10.1002/pros.21493
Nedrow-Byers JR, Moore AL, Ganguly T, Hopkins MR, Fulton MD, Benny PD, Berkman CE (2013) PSMA-targeted SPECT agents: mode of binding effect on in vitro performance. Prostate 73:355–362. https://doi.org/10.1002/pros.22575
Nikfarjam Z, Bavi O, Amini SK (2021) Potential effective inhibitory compounds against Prostate Specific Membrane Antigen (PSMA): a molecular docking and molecular dynamics study. Arch Biochem Biophys 699:108747. https://doi.org/10.1016/j.abb.2020.108747
Novakova Z, Cerny J, Choy CJ, Nedrow JR, Choi JK, Lubkowski J, Berkman CE, Barinka C (2016a) Design of composite inhibitors targeting glutamate carboxypeptidase II: the importance of effector functionalities. FEBS J 283:130–143. https://doi.org/10.1111/febs.13557
Novakova Z, Wozniak K, Jancarik A, Rais R, Wu Y, Pavlicek J, Ferraris D, Havlinova B, Ptacek J, Vavra J, Hin N, Rojas C, Majer P, Slusher BS, Tsukamoto T, Barinka C (2016b) Unprecedented binding mode of hydroxamate-based inhibitors of glutamate carboxypeptidase II: structural characterization and biological activity. J Med Chem 59:4539–4550. https://doi.org/10.1021/acs.jmedchem.5b01806
Pandit A, Sengupta S, Krishnan MA, Reddy RB, Sharma R, Venkatesh C (2018) First report on 3D-QSAR and molecular dynamics based docking studies of GCPII inhibitors for targeted drug delivery applications. J Mol Struct 1159:179–192. https://doi.org/10.1016/j.molstruc.2018.01.059
Pastorino S, Riondato M, Uccelli L, Giovacchini G, Giovannini E, Duce V, Ciarmiello A (2020) Toward the discovery and development of PSMA targeted inhibitors for nuclear medicine applications. Curr Radiopharm 13:63–79. https://doi.org/10.2174/1874471012666190729151540
Plechanovová A, Byun Y, Alquicer G, Škultetyová LU, Mlčochová P, Němcová A, Kim H-J, Navrátil M, Mease R, Lubkowski J, Pomper M, Konvalinka J, Rulíšek L, Bařinka C (2011) Novel substrate-based inhibitors of human glutamate carboxypeptidase II with enhanced lipophilicity. J Med Chem 54:7535–7546. https://doi.org/10.1021/jm200807m
Robinson MB, Blakely RD, Couto R, Coyle JT (1987) Hydrolysis of the brain dipeptide N-acetyl-L-aspartyl-L-glutamate. Identification and characterization of a novel N-acetylated alpha-linked acidic dipeptidase activity from rat brain. J Biol Chem 262:14498–14506. https://doi.org/10.1016/S0021-9258(18)47823-4
Rockey WM, Hernandez FJ, Huang SY, Cao S, Howell CA, Thomas GS, Liu XY, Lapteva N, Spencer DM, McNamara JO, Zou X, Chen SJ, Giangrande PH (2011) Rational truncation of an RNA aptamer to prostate-specific membrane antigen using computational structural modeling. Nucleic Acid Ther 21:299–314. https://doi.org/10.1089/nat.2011.0313
Rowe SP, Gorin MA, Allaf ME, Pienta KJ, Tran PT, Pomper MG, Ross AE, Cho SY (2016) PET imaging of prostate-specific membrane antigen in prostate cancer: current state of the art and future challenges. Prostate Cancer Prostatic Dis 19:223–230. https://doi.org/10.1038/pcan.2016.13
Schäfer M, Bauder-Wust U, Leotta K, Zoller F, Mier W, Haberkorn U, Eisenhut M, Eder M (2012) A dimerized urea-based inhibitor of the prostate-specific membrane antigen for 68Ga-PET imaging of prostate cancer. EJNMMI Res 2:23. https://doi.org/10.1186/2191-219X-2-23
Sharma N, Baruah MM (2019) The microRNA signatures: aberrantly expressed miRNAs in prostate cancer. Clin Transl Oncol 21:126–144. https://doi.org/10.1007/s12094-018-1910-8
Søborg Pedersen K, Baun C, Michaelsen Nielsen K, Thisgaard H, Ingemann Jensen A, Zhuravlev F (2020) Design, synthesis, computational, and preclinical evaluation of (nat)Ti/(45)Ti-labeled urea-based glutamate PSMA ligand. Molecules 25. https://doi.org/10.3390/molecules25051104
Stoermer D, Liu Q, Hall MR, Flanary JM, Thomas AG, Rojas C, Slusher BS, Tsukamoto T (2003) Synthesis and biological evaluation of hydroxamate-based inhibitors of glutamate carboxypeptidase II. Bioorg Med Chem Lett 13:2097–2100. https://doi.org/10.1016/S0960-894X(03)00407-4
Su SL, Huang IP, Fair WR, Powell CT, Heston WD (1995) Alternatively spliced variants of prostate-specific membrane antigen RNA: ratio of expression as a potential measurement of progression. Cancer Res 55:1441–1443. https://cancerres.aacrjournals.org/content/55/7/1441.short/. Accessed Apr 1995
Takatsu Y, Fujita Y, Tsukamoto T, Slusher BS, Hashimoto K (2011) Orally active glutamate carboxypeptidase II inhibitor 2-MPPA attenuates dizocilpine-induced prepulse inhibition deficits in mice. Brain Res 1371:82–86. https://doi.org/10.1016/j.brainres.2010.11.048
Tateishi U (2020) Prostate-specific membrane antigen (PSMA)-ligand positron emission tomography and radioligand therapy (RLT) of prostate cancer. Jpn J Clin Oncol 50:349–356. https://doi.org/10.1093/jjco/hyaa004
Tykvart J, Šácha P, Bařinka C, Knedlík T, Starková J, Lubkowski J, Konvalinka J (2012) Efficient and versatile one-step affinity purification of in vivo biotinylated proteins: expression, characterization and structure analysis of recombinant human glutamate carboxypeptidase II. Protein Expr Purif 82:106–115. https://doi.org/10.1016/j.pep.2011.11.016
Vahidfar N, Fallahpoor M, Farzanehfar S, Divband G, Ahmadzadehfar H (2019) Historical review of pharmacological development and dosimetry of PSMA-based theranostics for prostate cancer. J Radioanal Nucl Chem 322:237–248. https://doi.org/10.1007/s10967-019-06800-6
van der Post JP, de Visser SJ, de Kam ML, Woelfler M, Hilt DC, Vornov J, Burak ES, Bortey E, Slusher BS, Limsakun T, Cohen AF, van Gerven JM (2005) The central nervous system effects, pharmacokinetics and safety of the NAALADase-inhibitor GPI 5693. Br J Clin Pharmacol 60:128–136. https://doi.org/10.1111/j.1365-2125.2005.02396.x
Wang H, Byun Y, Barinka C, Pullambhatla M, Bhang H-EC, Fox JJ, Lubkowski J, Mease RC, Pomper MG (2010) Bioisosterism of urea-based GCPII inhibitors: synthesis and structure–activity relationship studies. Bioorg Med Chem Lett 20:392–397. https://doi.org/10.1016/j.bmcl.2009.10.061
Wirtz M, Schmidt A, Schottelius M, Robu S, Gunther T, Schwaiger M, Wester HJ (2018) Synthesis and in vitro and in vivo evaluation of urea-based PSMA inhibitors with increased lipophilicity. EJNMMI Res 8:84. https://doi.org/10.1186/s13550-018-0440-2
Wright GL Jr, Haley C, Beckett ML, Schellhammer PF (1995) Expression of prostate-specific membrane antigen in normal, benign, and malignant prostate tissues. Urol Oncol 1:18–28. https://doi.org/10.1016/1078-1439(95)00002-Y
Wu LY, Anderson MO, Toriyabe Y, Maung J, Campbell TY, Tajon C, Kazak M, Moser J, Berkman CE (2007) The molecular pruning of a phosphoramidate peptidomimetic inhibitor of prostate-specific membrane antigen. Bioorg Med Chem 15:7434–7443. https://doi.org/10.1016/j.bmc.2007.07.028
Wu LY, Do JC, Kazak M, Page H, Toriyabe Y, Anderson MO, Berkman CE (2008) Phosphoramidate derivatives of hydroxysteroids as inhibitors of prostate-specific membrane antigen. Bioorg Med Chem Lett 18:281–284. https://doi.org/10.1016/j.bmcl.2007.10.096
Wustemann T, Bauder-Wust U, Schäfer M, Eder M, Benesova M, Leotta K, Kratochwil C, Haberkorn U, Kopka K, Mier W (2016) Design of internalizing PSMA-specific Glu-ureido-based radiotherapeuticals. Theranostics 6:1085–95. https://doi.org/10.7150/thno.13448
Yang X, Mease RC, Pullambhatla M, Lisok A, Chen Y, Foss CA, Wang Y, Shallal H, Edelman H, Hoye AT, Attardo G, Nimmagadda S, Pomper MG (2016) [18F]Fluorobenzoyllysinepentanedioic acid carbamates: new scaffolds for positron emission tomography (PET) imaging of prostate-specific membrane antigen (PSMA). J Med Chem 59:206–218. https://doi.org/10.1021/acs.jmedchem.5b01268
Zha Z, Ploessl K, Choi SR, Wu Z, Zhu L, Kung HF (2018) Synthesis and evaluation of a novel urea-based (68)Ga-complex for imaging PSMA binding in tumor. Nucl Med Biol 59:36–47. https://doi.org/10.1016/j.nucmedbio.2017.12.007
Zhang AX, Murelli RP, Barinka C, Michel J, Cocleaza A, Jorgensen WL, Lubkowski J, Spiegel DA (2010) A remote arene-binding site on prostate specific membrane antigen revealed by antibody-recruiting small molecules. J Am Chem Soc 132:12711–12716. https://doi.org/10.1021/ja104591m
Zhang W, Murakawa Y, Wozniak KM, Slusher B, Sima AA (2006) The preventive and therapeutic effects of GCPII (NAALADase) inhibition on painful and sensory diabetic neuropathy. J Neurol Sci 247:217–223. https://doi.org/10.1016/j.jns.2006.05.052
Zhou J, Neale JH, Pomper MG, Kozikowski AP (2005) NAAG peptidase inhibitors and their potential for diagnosis and therapy. Nat Rev Drug Discov 4:1015–1026. https://doi.org/10.1038/nrd1903
Author information
Authors and Affiliations
Contributions
ZN: literature search and data analysis, writing—original draft, visualization. FZ: literature search and data analysis. AN: supervision, editing. OB: conceptualization, supervision, writing—review and editing.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nikfarjam, Z., Zargari, F., Nowroozi, A. et al. Metamorphosis of prostate specific membrane antigen (PSMA) inhibitors. Biophys Rev 14, 303–315 (2022). https://doi.org/10.1007/s12551-021-00919-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12551-021-00919-1