Abstract
Background
Cardiac resynchronization therapy (CRT) patients with different pathophysiology may influence mechanical dyssynchrony and get different ventricular resynchronization and clinical outcomes.
Methods
Ninety-two dilated cardiomyopathy (DCM) and fifty ischemic cardiomyopathy (ICM) patients with gated single-photon emission computed tomography (SPECT) myocardial perfusion imaging (MPI) were included in this retrospective study. Patients were classified based on the concordance between the left ventricular (LV) lead and the latest contraction or relaxation position. If the LV lead was located on or adjacent to both the latest contraction and relaxation position, the patient was categorized into the both match group; if the LV lead was located on or adjacent to the latest contraction or relaxation position, the patient was classified into the one match group; if the LV lead was located on or adjacent to neither the latest contraction nor relaxation position, the patient was categorized to the neither group. CRT response was defined as \(\ge 5\%\) improvement of LV ejection fraction at the 6-month follow-up. Variables with P < .05 in the univariate analysis were included in the stepwise multivariate model.
Results
During the follow-up period, 58.7% (54 of 92) for DCM patients and 54% (27 of 50) for ICM patients were CRT responders. The univariate analysis and stepwise multivariate analysis showed that QRS duration, systolic phase bandwidth (PBW), diastolic PBW, diastolic phase histogram standard deviation (PSD), and left ventricular mechanical dyssynchrony (LVMD) concordance were independent predictors of CRT response in DCM patients; diabetes mellitus and left ventricular end-systolic volume were significantly associated with CRT response in ICM patients. The intra-group comparison revealed that the CRT response rate was significantly different in the both match group of DCM (N = 18, 94%) and ICM (N = 24, 62%) patients (P = .016). However, there was no significant difference between DCM and ICM in the one match and neither group. For the inter-group comparison, Kruskal-Wallis H-test revealed that CRT response was significantly different in all the groups of DCM patients (P < .001), but not in ICM patients (P = .383).
Conclusions
Compared with ICM patients, systolic PBW, diastolic PBW and PSD have better predictive and prognostic values for the CRT response in DCM patients. Placing the LV lead in or adjacent to the latest contraction and relaxation position can improve the clinical outcomes of DCM patients, but it does not apply to ICM patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
There are 30% to 40% of cardiac resynchronization therapy (CRT) recipients who do not benefit from CRT.1,2 LV mechanical dyssynchrony (LVMD) parameters measured by phase analysis from gated single-photon emission computed tomography (SPECT) myocardial perfusion imaging (MPI) provide repeatable and reproducible information about the presence of intraventricular synchronism.3,4 They have been found to be independent predictors for CRT patient selection5,6 and have been proven to have prognostic value.7,8 Moreover, the concordance of LV lead on or adjacent to the late contracting viable segments measured by gated SPECT MPI was associated with CRT response and heart failure rehospitalization and all-cause mortality.9
In dilated cardiomyopathy (DCM) patients, systolic and diastolic LVMD were independent predictors for CRT response; and pacing the LV lead in the segments with the latest contraction and relaxation would improve the CRT response rate.10 For ischemic cardiomyopathy (ICM) patients, the systolic phase bandwidth (PBW) as an LVMD parameter has been identified as an independent predictor of ventricular arrhythmia after CRT implantation.6 However, comparative studies on the predictive value of LVMD for CRT in HF patients with different pathophysiology are still limited. The purpose of this study was to compare the predictive and prognostic values of LVMD measured by gated SPECT MPI and the concordance of LV lead with the sites of the latest contraction or relaxation position in DCM and ICM patients.
Methods
Patient Population
CRT recipients were consecutively enrolled in a retrospective database at the First Affiliated Nanjing Medical University Hospital from May 2009 to August 2020. Study subjects selected retrospectively had DCM: a presence of LV dilation and LV systolic dysfunction in the absence of other etiological factors that might cause LV dysfunction by echocardiography according to the recent criteria or CAD that causes global systolic dysfunction;11 or ICM: epicardial coronary artery stenosis greater than 50% or previous history of coronary revascularization or myocardial infarction.12 A total of 92 DCM and 50 ICM patients who met the above criteria were included in the study, as shown in Figure 1. All patients met standard indications for CRT at the time of implantation: LVEF < 35%, QRS duration \(\ge \) 120 milliseconds with sinus rhythm, New York Heart Association (NYHA) functional class greater or equal to II, and optimal medical therapy for at least 3 months before CRT implantation. Exclusion criteria were as follows: atrial fibrillation, right bundle branch block, pregnancy or breastfeeding, and those being upgraded from right ventricular pacing. This study was approved by the Institutional Ethical Committee of the First Affiliated Hospital of Nanjing Medical University.
Echocardiography
Echocardiography was performed at baseline before CRT implantation and at the 6-month CRT clinical follow-up. LV function was assessed twice by two experienced ultrasound experts, who were blinded to the clinical data before and 6 months after CRT implantation, and the mean value was used as the final record. LVEF was calculated using the 2-dimensional modified biplane Simpson method. Echocardiographic response to CRT was defined as an increase in LVEF by 5% or more.
SPECT MPI Assessment
Gated SPECT MPI was performed around 60 minutes after injection using 20-30mCi of 99mTc-sestamibi. All the images were acquired in a dual-headed camera (CardioMD, Philips Medical Systems) with a standard protocol with 20% energy window around 140 KeV, 180° orbit, 32 steps with 25 seconds per step, 8-bin gating, and 64 planar projections per gate. Image reconstruction and reorientation were performed by Emory Reconstruction Toolbox (ERToolbox; Atlanta, GA) using the OSEM method with 3 iterations and 10 subsets and filtered by a Butterworth low-pass filter with an order of 10 subsets and a cutoff frequency of 0.3 cycles/cm.
The resulting short-axis images were sent to an interactive tool for automatized accessing LV contour parameters by an automatic myocardial sampling algorithm that searched the maximal count circumferential profiles in each cardiac frame. Furthermore, the onset of mechanical contraction and relaxation throughout the cardiac cycle were obtained by multi-harmonic Fourier approximations.13 Then, the LVMD was represented by phase distribution of systolic and diastolic dyssynchrony for the entire left ventricle, and quantitative parameters of LVMD were calculated as phase standard deviation (PSD) and phase bandwidth (PBW).13,14
CRT Implantation and LV Lead Position
The right atrial and ventricular leads were positioned under fluoroscopic guidance by a transvenous approach. The LV lead location was determined by coronary venous angiography cine images in the left anterior oblique (LAO) and right anterior oblique (RAO), and then correlated to the 13-segment polar map of the systolic and diastolic dyssynchrony.12,15 LV lead located on or adjacent segment of the latest contraction or relaxation segment was classified as being concordant to systolic phase or diastolic phase (one match), respectively, as depicted in two ICM examples in Figure 2.
Illustrative examples of systolic match and diastolic match. Polar maps of patient 1 with ICM showing the LV lead located on the latest contraction segment (green in A), not on or adjacent to the latest relaxation segment (red box in B). This patient is classified as systolic match. The LV lead of Patient 2 is located on the latest relaxation segment (green box in D) and not on or adjacent to the latest contraction segment (red box in C)
Statistical Analysis
The differences between the DCM and ICM were compared by the unpaired t-test for continuous variables, expressed as mean ± standard deviation, and Chi-square test for categorical variables expressed in number and percentage. The systolic and diastolic LVMD within all patients, DCM patients, and ICM patients were compared by paired t-test. Univariate binary logistic regression analysis was performed on all clinically relevant variables to estimate potential predictors for CRT response. Due to the collinearity between LVMD parameters, and in order to avoid model overfitting, they were entered one by one with those selected variables that were found significant in the univariate analysis in a stepwise fashion into the multivariate logistic regression to obtain the optimal models. Kruskal-Wallis H-test was be used to analyze the difference of CRT response rate among three groups in DCM and ICM patients, respectively. Differences in survival over time were analyzed by the log-rank Kaplan-Meier survival analysis. P < .05 was considered to be statistically significant. Statistical analysis was performed by the Python Statsmodels package16 and IBM SPSS Statistics software version 26 (SPSS Inc, Chicago, Illinois).
Results
A total of 142 patients (DCM, 92; ICM, 50) who underwent SPECT MPI before CRT implantation were included in this study. The baseline characteristics of the included patients are shown in Table 1. For all patients, the age was \(64.6\pm 14.5\) years, and 71.1% (N = 101) patients were male. The baseline QRS duration (\(157.9 \pm 23.3\)), medical therapy records, and LV functions were also shown in the baseline table. The differences between systolic and diastolic LVMD in all patients, DCM patients, and ICM patients were significant (all P < .001).
In the univariate analysis for DCM patients, QRS duration (95%CI 1.0-1.05, P = .014), NT-proBNP (95%CI 0.0-0.61, P = .026), non-sustained ventricular tachycardia (NS-VT) (95%CI 0.1-0.64, P = .004), Scar burden (95%CI 0.93-1.0, P = .029), all LVMD parameters (systolic PSD, 95%CI 0.96-1.0, P = .017; systolic PBW, 95%CI 0.99-1.0, P = .009; diastolic PSD, 95%CI 0.95-0.99, P = .003; diastolic PBW, 95%CI 0.99-1.0, P = .003), LVMD concordance (95%CI 0.13-0.5, P < .001), and LV lead in scarred myocardium (95%CI 1.09-8.18, P = .033) were statistically significant predictors of CRT response. However, for ICM patients, only diabetes mellitus (DM) (95%CI 0.05-0.62, P = .007), QRS duration (95%CI 1.0-1.07, P = .044), NS-VT (95%CI 0.08-0.94, P = .039), LVEDV (95%CI 0.98-1.0, P = .009), LVESV (95%CI 0.98-1.0, P = .009) were statistically significant predictors of CRT response, as shown in Table 2.
In the multivariate models for DCM patients, QRS duration, NT-proBNP, 3 LVMD parameters (systolic PBW: 95% CI 0.98-1.00, P = .041; diastolic PSD: 95% CI 0.94-1.00, P = .041; diastolic PBW: 95% CI 0.98-1.00, P = .028) and LVMD concordance (P < .003 for all) were significantly independent predictors of CRT response. For ICM patients, DM and LVESV were significant independent predictors of CRT response; however, all LVMD parameters and LVMD concordance were not significant. The results of the multivariate analysis are shown in Tables 3, 4, 5 and 6.
Patients were divided into three groups based on the latest contraction or relaxation segment at the LV lead location: patients whose LV lead were concordant or adjacent to the latest contraction and relaxation segment (both match: DCM, N = 18; ICM, N = 24), and patients whose LV lead were concordant or adjacent to the latest contraction or relaxation segment (one match: DCM, N = 57; ICM, N = 22), and patients whose LV lead were neither concordant nor adjacent to the latest contraction or relaxation segment (neither: DCM, N = 17; ICM, N = 4). The intra-group comparison revealed that the CRT response rate of DCM patients (94%, N = 18) was much higher than ICM patients (62%, N = 24) in both match group (P = .016). There was no significant difference in the one match group (P = .363) and neither group (P = .521) between DCM and ICM patients, as shown in Figure 3. For the inter-group comparison, Kruskal-Wallis H-test revealed that CRT response was significantly different in the three groups of DCM patients (P < .001), but not in ICM patients (P = .383).
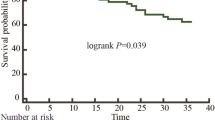
During the mean follow-up time of \(39\pm 24\) months (IQR 19-55), 10 (10.87%) DCM patients and 9 (18%) ICM patients died. Kaplan-Meier survival curves showed significantly longer survival in DCM patients with the concordance between LV lead with the latest contraction and relaxation position (P = .050), as shown in Figure 4. However, there is no significant difference of survival time in ICM patients based on the concordance between LV lead and the latest contraction or relaxation position, as shown in Figure 5.
Discussion
The main finding of the present study was that systolic PBW, diastolic PSD and PBW were strong predictors of CRT response only in DCM patients. Furthermore, Kaplan-Meier analysis showed that the concordance of LV lead to the latest contraction and relaxation position were independent predictors of death from any cause and had significantly longer survival than LV lead only located in one latest position or none in DCM patients. Whether it is DCM or ICM, it is necessary to avoid placing the LV lead in a non-latest contraction or relaxation position whenever possible.
Predictive Value of LVMD for CRT Patient Selection
Research on selecting appropriate patients for CRT with LVMD measured by gated SPECT MPI has been widely studied. In a study of 42 CRT patients, the receiver operating characteristic curve analysis showed that the optimal cutoff value of PSD and PBW were \(43^\circ \) (sensitivity and specificity of 70%) and \(135^\circ \) (sensitivity and specificity of 74%), respectively.5 In a study with 324 consecutive patients with non-ICM CRT patients, it was demonstrated that systolic PSD, adjust to age, hypertension, diabetes, aspirin, beta-blockers, diuretics, QRS, and EF, was an independent predictor of all-cause mortality (HR 1.97, 95% CI 1.06-3.66, P = .033).17 However, in a multi-center VISION-CRT clinical trial (N = 195), it was found that the systolic LVMD did not have a predictive value for CRT response, but they did not discuss it based on different pathology.18 Peix et al19 further analyzed part of the data from this clinical trial and found that CRT recipients with more dyssynchrony at baseline had significant improvement in non-ischemic patients with non-compaction myocardium, whose PSD was reduced from \(89.5\pm 14.2^\circ \) to \(63.7\pm 20.5^\circ \) (P = .028).
For DCM patients, Henneman et al5 demonstrated that baseline systolic LVMD could be used to predict CRT response by the cutoff value of 43° for PSD with 74% sensitivity and specificity and 135° for PBW with 70% sensitivity and specificity. Wang et al10 found that both systolic and diastolic LVMD had predictive value for CRT patient selection in 84 DCM patients (systolic PSD: 95% CI 0.92-1.00, P = .043; systolic PBW: 95% CI 0.99-1.00, P = .038; diastolic PSD: 95% CI 0.94-1.00, P = .032; diastolic PBW: 95% CI 0.99-1.00, P = .024). Similar results were found in our study that systolic PBW (95% CI 0.98-1.00, P = .041), diastolic PBW (95% CI 0.98-1.00, P = .028), and diastolic PSD (95% CI 0.94-1.00, P = .041) were independent significant predictors for CRT patient selection, except for systolic PSD (95% CI 0.94-1.00, P = .063), but its P value is still very close to .05.
For ICM patients, the presence of transmural scar tissue, which may affect the measurement of LVMD,20 often resulted in non-response to CRT.21 However, few studies have been done on the predictive value of LVMD for CRT in ICM patients. A study found that the difference between stress LVMD and rest LVMD was an independent predictor instead of rest LVMD for all-cause mortality in ICM patients;22 however, not all CRT patients receive stress gated SPECT MPI. Our study demonstrated that both systolic and diastolic LVMD were not independent predictive factors for CRT response in ICM patients. This might be due to the presence of hibernating myocardium or severely scarred and dysfunctional myocardium, which requires further evaluation.22
LVMD to Guide CRT Lead Placement
The optimal LV lead position has been suggested to be the latest or adjacent to the latest segment mechanical activation.9 In a study with 90 CRT patients, the patients with a concordant LV lead position (the LV lead placed in the site of the latest mechanical activation measured by SPECT MPI) had significant improvement in LV volumes and LV systolic function than the patients with a discordant LV lead position (79% vs. 26%, P < .01).23
For DCM patients, the CRT response could be increased when the LV lead is placed in the latest contraction and relaxation segment,10 which was proved in our study among 92 DCM patients. A study with 64 CRT patients found that systolic PSD and PBW were significant factors to differentiate wide QRS duration (\(\ge 150\) ms) with narrow QRS duration (120-150 ms) among 47 DCM patients, and there were no similar results in ICM patients (N = 17).24 In our study, both systolic and diastolic LVMD concordance between the latest activation segments and LV lead position were not independent predictive factors for CRT response in ICM patients. This finding was not surprising because the latest mechanical activation might be affected by the scar location due to the delayed electrical activation/conduction that might interfere with myocardial scar.24 Furthermore, It showed a weak predictive value for CRT response in ICM patients by the concordance of the LV lead with the latest contraction or relaxation position, which was totally different compared with DCM.
LVMD in Different Pathophysiology of Heart Failure
Compared with DCM patients, poor predictive performance in ICM patients is due to the global scar burden, multiple scar segments, and regional ischemia, which may affect the remodeling response to biventricular pacing.12 The contractility of myocardial scar tissue is impaired. Due to its electrophysiological inertia, it destroys the depolarizing waves from the adjacent myocardium, thereby prolonging the activation time of the ventricles.12 In addition, the presence of scar tissue means that the availability of recruitable contractile cardiomyocytes is reduced to bolster myocardial pump and LV hemodynamics.12 Therefore, placing the LV lead on or adjacent to a scar or ischemia may have a poor effect. These results indicate that routine ischemia assessment before CRT device implantation may help identify CRT responders and help guide the placement of LV lead.
Study Limitations
The main study limitation was the small number of retrospective patients, which limited the statistical analysis and the generalizability of our findings. Using two different imaging modalities to identify the latest contraction or relaxation segments by SPECT MPI and the location of LV lead by coronary venography limited the granularity that can describe the location and consistency of LV lead. However, this definition method has gained wide acceptance.12,25,26
New Knowledge Gained
The role of systolic and diastolic LVMD for CRT patient selection and guide LV lead placement is still unclear. This study demonstrates that systolic PBW, diastolic PBW and PSD, concordance between LV lead with the latest contraction or relaxation segment were independent predictive variables for CRT patient selection in DCM patients. In particular, in our sample, the same conclusion was not found in ICM patients.
Conclusions
Compared with ICM patients, systolic PBW, diastolic PBW and PSD have better predictive and prognostic values for the CRT response in DCM patients. Placing the LV lead on or adjacent to the latest contraction and relaxation position can improve the clinical outcomes of DCM patients, but it does not apply to ICM patients.
Change history
19 October 2021
A Correction to this paper has been published: https://doi.org/10.1007/s12350-021-02834-0
Abbreviations
- CRT:
-
Cardiac resynchronization therapy
- DCM:
-
Dilated cardiomyopathy
- ICM:
-
Ischemic cardiomyopathy
- LV:
-
Left ventricular
- LVEDV:
-
Left ventricular end-diastolic volume
- LVEF:
-
Left ventricular ejection fraction
- LVESV:
-
Left ventricular end-systolic volume
- LVMD:
-
Left ventricular mechanical dyssynchrony
- MPI:
-
Myocardial perfusion imaging
- PBW:
-
Phase bandwidth
- PSD:
-
Phase histogram standard deviation
References
Fornwalt BK, Sprague WW, Bedell P, Suever JD, Gerritse B, Merlino JD, et al. Agreement is poor among current criteria used to define response to cardiac resynchronization therapy. Circulation 2010;121:1985-91.
Zhou W, Garcia EV. Nuclear image-guided approaches for cardiac resynchronization therapy (CRT). Curr Cardiol Rep 2016;18:7.
Trimble MA, Velazquez EJ, Adams GL, Honeycutt EF, Pagnanelli RA, Barnhart HX, et al. Repeatability and reproducibility of phase analysis of gated single-photon emission computed tomography myocardial perfusion imaging used to quantify cardiac dyssynchrony. Nucl Med Commun 2008;29:374-81.
Lin X, Xu H, Zhao X, Folks RD, Garcia EV, Soman P, et al. Repeatability of left ventricular dyssynchrony and function parameters in serial gated myocardial perfusion SPECT studies. J Nucl Cardiol 2010;17:811-6.
Henneman MM, Chen J, Dibbets-Schneider P, Stokkel MP, Bleeker GB, Ypenburg C, et al. Can LV dyssynchrony as assessed with phase analysis on gated myocardial perfusion SPECT preferably predict response to CRT? J Nucl Med 2007;48:1104-11.
Tsai SC, Chang YC, Chiang KF, Lin WY, Huang JL, Hung GU, et al. LV dyssynchrony is helpful in predicting ventricular arrhythmia in ischemic cardiomyopathy after cardiac resynchronization therapy a preliminary study. Medicine (United States) 2016;95:e2840.
Pazhenkottil AP, Buechel RR, Husmann L, Nkoulou RN, Wolfrum M, Ghadri JR, et al. Long-term prognostic value of left ventricular dyssynchrony assessment by phase analysis from myocardial perfusion imaging. Heart 2011;97:33-7.
Wang L, Yang MF, Cai M, Zhao SH, He ZX, Wang YT. Prognostic significance of left ventricular dyssynchrony by phase analysis of gated spect in medically treated patients with dilated cardiomyopathy. Clin Nucl Med 2013;38:510-5.
Zhang X, Qian Z, Tang H, Hua W, Su Y, Xu G, et al. A new method to recommend left ventricular lead positions for improved CRT volumetric response and long-term prognosis. J Nucl Cardiol 2019;28:672-84.
Wang C, Shi J, Ge J, Tang H, He Z, Liu Y, et al. Left ventricular systolic and diastolic dyssynchrony to improve cardiac resynchronization therapy response in heart failure patients with dilated cardiomyopathy. J Nucl Cardiol 2020. https://doi.org/10.1007/s12350-020-02132-1.
Elliott P, Andersson B, Arbustini E, Bilinska Z, Cecchi F, Charron P, et al. Classification of the cardiomyopathies: A position statement from the european society of cardiology working group on myocardial and pericardial diseases. Eur Heart J 2008;29:270-6.
Bose A, Kandala J, Upadhyay GA, Riedl L, Ahmado I, Padmanabhan R, et al. Impact of myocardial viability and left ventricular lead location on clinical outcome in cardiac resynchronization therapy recipients with ischemic cardiomyopathy. J Cardiovasc Electrophysiol 2014;25:507-13.
Chen J, Kalogeropoulos AP, Verdes L, Butler J, Garcia EV. Left-ventricular systolic and diastolic dyssynchrony as assessed by multi-harmonic phase analysis of gated SPECT myocardial perfusion imaging in patients with end-stage renal disease and normal LVEF. J Nucl Cardiol 2011;18:299-308.
Chen J, Garcia EV, Folks RD, Cooke CD, Faber TL, Tauxe EL, et al. Onset of left ventricular mechanical contraction as determined by phase analysis of ECG-gated myocardial perfusion SPECT imaging: Development of a diagnostic tool for assessment of cardiac mechanical dyssynchrony. J Nucl Cardiol 2005;12:687-95.
Singh JP, Klein HU, Huang DT, Reek S, Kuniss M, Quesada A, et al. Left ventricular lead position and clinical outcome in the multicenter automatic defibrillator implantation trial–cardiac resynchronization therapy (MADIT-CRT) trial. Circulation 2011. https://doi.org/10.1161/CIRCULATIONAHA.110.000646.
Seabold S, Perktold J. Statsmodels: Econometric and statistical modeling with Python. In: Proceedings of the 9th Python in Science Conference; 2010. p. 92-96.
Goldberg AS, Alraies MC, Cerqueira MD, Jaber WA, Aljaroudi WA. Prognostic value of left ventricular mechanical dyssynchrony by phase analysis in patients with non-ischemic cardiomyopathy with ejection fraction 35-50% and QRS < 150 ms. J Nucl Cardiol 2014;21:57-66.
Peix A, Karthikeyan G, Massardo T, Kalaivani M, Patel C, Pabon LM, et al. Value of intraventricular dyssynchrony assessment by gated-SPECT myocardial perfusion imaging in the management of heart failure patients undergoing cardiac resynchronization therapy (VISION-CRT). J Nucl Cardiol 2019. https://doi.org/10.1007/s12350-018-01589-5.
Peix A, Padrón K, Cabrera L, Castañeda O, Milán D, Castro J, et al. Intraventricular synchronism assessment by gated - SPECT myocardial perfusion imaging in cardiac resynchronization therapy. Does cardiomyopathy type influence results? EJNMMI Res 2020. https://doi.org/10.1186/s13550-020-00703-4.
Ludwig DR, Friehling M, Schelbert EB, Schwartzman D. Impact of scar on SPECT assay of left ventricular contraction dyssynchrony. Eur J Nucl Med Mol Imaging 2014;41:529-35.
Yokoshiki H, Mitsuyama H, Watanabe M, Mitsuhashi T, Shimizu A. Cardiac resynchronization therapy in ischemic and non-ischemic cardiomyopathy. J Arrhythmia 2017;33:410-6.
Aljaroudi W, Alraies MC, Menon V, Jaber WA. Predictors and incremental prognostic value of left ventricular mechanical dyssynchrony response during stress-gated positron emission tomography in patients with ischemic cardiomyopathy. J Nucl Cardiol 2012. https://doi.org/10.1007/s12350-012-9592-z.
Boogers MJ, Chen J, Van Bommel RJ, Borleffs CJW, Dibbets-Schneider P, Van Der Hiel B, et al. Optimal left ventricular lead position assessed with phase analysis on gated myocardial perfusion SPECT. Eur J Nucl Med Mol Imaging 2011;38:230-8.
Lin X, Xu H, Zhao X, Chen J. Sites of latest mechanical activation as assessed by SPECT myocardial perfusion imaging in ischemic and dilated cardiomyopathy patients with LBBB. Eur J Nucl Med Mol Imaging 2014;41:1232-9.
Ypenburg C, Schalij MJ, Bleeker GB, Steendijk P, Boersma E, Dibbets-Schneider P, et al. Impact of viability and scar tissue on response to cardiac resynchronization therapy in ischaemic heart failure patients. Eur Heart J 2007;28:33-41.
Adelstein EC, Tanaka H, Soman P, Miske G, Haberman SC, Saba SF, et al. Impact of scar burden by single-photon emission computed tomography myocardial perfusion imaging on patient outcomes following cardiac resynchronization therapy. Eur Heart J 2011;32:93-103.
Disclosure
All authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Contributions
ZH—Contribution: Conception and design; analysis and interpretation of data; drafting of the manuscript and revising it critically for important intellectual content. DL—Contribution: Revising it critically for important intellectual content. CC—Contribution: Revising it critically for important intellectual content. H-YQ—Contribution: Revising it critically for important intellectual content. ZZ—Contribution: Revising it critically for important intellectual content. XH—Contribution: Revising it critically for important intellectual content. JZ—Contribution: Revising it critically for important intellectual content. M-LC—Contribution: Revising it critically for important intellectual content. CW—Contribution: Acquisition, analysis, and interpretation of data; revising the manuscript critically for important intellectual content. WZ—Contribution: Conception and design; analysis and interpretation of data; revising the manuscript critically for important intellectual content; and final approval of the manuscript submitted.
Corresponding authors
Ethics declarations
Ethical approval
The study was approved by the scientific councils of the participating county scientific councils and complied with the Declaration of Helsinki. Written informed consent was obtained from all participants, and patient anonymity was maintained during data analysis.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The authors of this article have provided a PowerPoint file, available for download at SpringerLink, which summarises the contents of the paper and is free for re-use at meetings and presentations. Search for the article DOI on SpringerLink.com.
The authors have also provided an audio summary of the article, which is available to download as ESM, or to listen to via the JNC/ASNC Podcast.
All editorial decisions for this article, including selection of reviewers and the final decision, were made by guest editor Saurabh Malhotra, MD, MPH.
Funding
This research was supported by a grant from the American Heart Association (Project Number: 17AIREA33700016, PI: Weihua Zhou) and a new faculty grant from Michigan Technological University Institute of Computing and Cybersystems (PI: Weihua Zhou), and grants from the National Nature Science Foundation of China (81900295) and the Natural Science Foundation of Jiangsu Province (BK 20191071).
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
He, Z., Li, D., Cui, C. et al. Predictive values of left ventricular mechanical dyssynchrony for CRT response in heart failure patients with different pathophysiology. J. Nucl. Cardiol. 29, 2637–2648 (2022). https://doi.org/10.1007/s12350-021-02796-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-021-02796-3