Abstract
Mucosa-associated lymphoid tissue (MALT) lymphomas are often multifocal with patchy distribution that can be scattered throughout an involved organ. Previous studies have shown that each of these surrounding foci is often clonally related. Less is known about the clonal relationship of MALT lymphomas that involve multiple different organ systems. To investigate the clonal relationship of MALT lymphomas that involve multiple different organ systems, we retrospectively reviewed records of all marginal zone lymphoma from our database. Three cases were found to have multiple organ involvement and subjected to the analysis of the rearranged immunoglobulin variable regions using a polymerase chain reaction–based assay. Three cases of MALT lymphomas arising from multiple sites include liver and lung, gastric and lung, nasopharynx, and parotid. Our results demonstrate that two of these cases show a clonal relationship; however, one case appears clonally distinct and appears unrelated. With reviewing the literature of MALT lymphomas that involve multiple different organ systems, it overall supports that clearly these have the potential to disseminate to other extranodal locations; however, involvement of multiple different organ systems should not necessarily be considered definitive evidence for disseminated or advanced disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Extranodal marginal zone B-cell lymphoma of mucosa-associated lymphoid tissue (MALT), also known as MALT lymphoma, is a low-grade B-cell lymphoma that arises in extranodal sites. A wide range of extranodal sites can be involved, but typically, these tumors are associated with epithelial elements, e.g., stomach, lung, ocular adnexa, and salivary gland [1]. MALT lymphomas have been shown to represent approximately 7–8% of all non-Hodgkin lymphomas, with the most common site of involvement being the stomach, accounting for more than half of all cases [2, 3]. In general, chronic inflammation appears as a universal central component in the lymphomagenesis of MALT lymphomas, whether it be related to infectious, autoimmune, or some other unknown stimulus. [4,5,6].
Recurrent genetic abnormalities have been described in MALT lymphomas and vary based on the primary site of disease, but do show some preferential distribution, such as the t(11;18)(q21; q21) occurring mainly in pulmonary and gastric tumors [7]. Currently, there are no available unique immunohistochemical markers to identify organ-specific–derived MALT lymphomas. Molecular analysis of immunoglobulin heavy-chain and light-chain genes of MALT lymphoma feature clonally rearrangements and typically feature somatic mutation of their variable regions, consistent with derivation from post-germinal center memory B-cells [8].
MALT lymphomas can often show multifocal involvement of the same organ system, which has been best studied in the context of gastric MALT lymphoma where most cases appear to be clonally related [9,10,11,12,13]. MALT lymphomas have the potential to disseminate and in 17% of cases can involve lymph nodes and approximately 10% can have bone marrow involvement [14]. However, the clonal relationship of MALT lymphomas involving multiple different organ systems is incompletely understood. Herein, we evaluate and report three cases of MALT lymphomas at multiple sites including liver and lung, gastric and lung, nasopharynx, and parotid as well as review the literature relating to the clonal aspects of multi-sited MALT lymphomas.
Materials and methods
We retrospectively reviewed records of all marginal zone lymphoma at our institution from January 1991 to January 2021 from our database and collected data on the patients diagnosed with MALT lymphoma. Further inclusion criteria required a diagnosis of MALT lymphoma arising in more than one distinct separate organ system. The diagnosis of MALT lymphoma was made based on histology. The selected paired cases were reviewed independently with concordance by two experienced pathologists. For this study, bone marrow and regional lymph node involvement was not included as this was more strongly presumed to represent disease dissemination. Genomic DNA is extracted using Maxwell RSC DNA Purification Kits (Promega, USA). Immunoglobulin gene rearrangement assays were performed according to instructions of the Biomed-2 Polymerase Chain Reaction kit (Invivoscribe technologies, Inc., CA, USA) regarding immunoglobulin heavy chain gene (IGH) involving FR1, FR2, and FR3 as well as κ light chain gene (IGK) involving Vκ-Jκ and Kde (κ deleting element), followed by capillary electrophoresis.
Results
We identified 845 patients with MALT lymphoma who were diagnosed in our hospital system from 1991 to 2021. Of these, three patients were determined to have MALT lymphoma arising in two 2 different anatomic sites (Table 1).
Case 1 is an 89-year-old female with chronic dry cough. She denies fevers, chills, night sweats, pain, and unintentional weight loss. CT chest showed multiple large lung cystic abnormalities with thickened walls and multiple soft tissue nodular opacities. MRCP noted a segment 4A liver lesion measuring 2.2 cm.
Case 2 is a 63-year-old female with history of HIV and hepatitis C. She noted an enlargement of the right parotid gland which fluctuated in size over the subsequent months. She reported pain in the right face and ear and had numbness of the right side of the face. CT scan demonstrated a large lobulated mass involving the superficial and deep lobes of the right parotid gland, measuring 4.6 × 3.2 cm, and bulky heterogeneous soft tissue in the nasopharynx, right greater than left extending to the choana.
Case 3 is a 50-year-old male with iron deficiency anemia, gait disturbance, intermittent blurry vision, fatigue, and weight loss. No lymphadenopathy and hepatosplenomegaly was noted. Chest X-ray revealed a 2.5-cm cavitary lesion in right upper lobe with bilateral multifocal cavitating opacities. CT of abdomen showed marked circumferential mural thickening of the gastric body and antrum.
In summary, the specimen pairs we identified include liver and lung (case 1), parotid and nasopharynx (case 2), and stomach and lung (case 3). Biopsies of the different sites were performed simultaneously for two patients (case 1 and case 3) and approximately 3 months apart for the remaining patient (case 2). All cases were assumed to have synchronous disease by initial computed tomography scan. No lymphadenopathy or further evidence of metastatic involvement was noted for all cases. No bone marrow biopsy was performed. Two patients (case 1 and case 3) were treated with rituximab and bendamustine and responded well with no clinical evidence for persistent disease. The remaining patient (case 2) received radiation therapy and is considered clinically in complete remission.
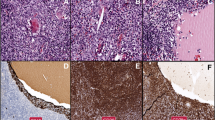
The histologic findings from the three patients appear similar, meeting established criteria for MALT lymphomas [1]. The tumors showed a dense monomorphic lymphocytic infiltrate featuring irregular/angulated nuclei and scant to moderate amounts of pale cytoplasm and inconspicuous nuclei. Scattered large cells were present, but no evidence for large cell lymphoma (Fig. 1). Immunohistochemical staining for all cases revealed neoplastic cells exhibiting CD20-expression consistent with B-lineage without significant CD5 or CD10. Immunoglobulin light chain restriction was clearly identified in case 1. CD43 appeared positive in case 3 and negative in case 1 and case 2.
Morphologic features and immunohistochemistry from representative cases (Case 1). HE staining (× 100) showing effacement of the normal architecture by a dense monomorphic lymphocytic infiltrate featuring irregular/angulated nuclei and scant to moderate amounts of pale cytoplasm and inconspicuous nuclei in liver (a) and lung (c). Neoplastic cells demonstrates strong immunohistochemical positivity for CD20 in liver (b) and lung (d)
Monoclonal rearrangement of the immunoglobulin gene was detected in all 3 tumor pairs. Criterion for establishment for a monoclonal gene rearrangement was determined if a prominent peak is noted on a background of polyclonal peaks in at least one PCR tube. We considered patients to have different clones if clonality analysis detected a different prominent clone peak when compared to the lymphoma arising in another site. The electropherogram peaks of these tumor pairs were compared and show that case 2 (parotid and nasopharynx) and case 3 (stomach and lung) appear clonally related, while case 1 (lung and liver) reveals distinct clonal peaks (Fig. 2).
Discussion
Mucosa-associated lymphoid tissue (MALT) lymphomas represent a diverse group of lymphoid neoplasms of B-cell origin, occurring typically in adult patients and usually with indolent clinical behavior, with 10-year overall survival rates greater than 80% [15]. Clear adverse prognostic features are limited, as even bone marrow involvement may not necessarily confer a significantly worse prognosis. However, transformation to diffuse large B-cell lymphoma (< 10% of cases) has been shown to significantly lower overall survival, approximately 50% at 5 years (16). Multifocal involvement for many lymphomas at diagnosis is often considered to represent disseminated disease; however, clear established guidelines about the evaluation and significance for multi-sited MALT lymphoma are lacking.
After reviewing the relevant literature, we found 20 cases reported of MALT lymphomas involving multiple different organ systems [10, 11, 17,18,19,20,21,22,23], summarized in Table 1, with a median age of 62 (33–89 years) with males and females being affected equally. Of those cases, 55% (11/20) appear to be clonally related with the most frequently involved organ system being the stomach (38%; 15/40), followed closely by lung (30%; 12/40) and stomach-lung being the most commonly encountered pair of involved organ systems (50%; 10/20). The proportion of lung MALT lymphomas in our review (12/40; 30%) is notably higher than expected based on the reported literature of 9% of MALT lymphomas (1). These 12 cases involving the lung, which included 10 stomach-lung, 1 parotid-lung, and 1 liver-lung case, half of these appear to be clonally related (6/12; 50%), which does not appear to differ significantly from the overall clonal relationship of all the organ systems we reviewed. A synchronous presentation appears to be more common (13/19; 68%), as opposed to metachronous disease (32%; 6/19). Of the synchronous cases, most appeared to be clonally related (62%; 8/13), with a metachronous presentation less likely to exhibit a clonal relationship (33%; 2/6). Of the data available, the median time for a metachronous clinical presentation between two MALT lymphomas was 3.9 years (4 months–18 years).
While no clear genetic predisposition for the development of MALT lymphomas has been established, these cases of MALT lymphomas involving multiple different organ systems with or without a clonal relationship might be of interest for future studies. Lymphatic and/or venous dissemination might explain some of the multi-sited disease; however, it remains a possibility of some unknown risk factor for developing MALT lymphomas involving multiple organ systems in some of these patients. In addition, despite that MALT lymphomas of different anatomic locations share many clinicopathological characteristics, there remains considerable variations in etiology as well as immunoglobulin gene usage and molecular landscape [5, 24,25,26,27,28]. In the future, this growing knowledge of the molecular aspects of site-specific disease raises an increased importance on accurate staging and clearly establishing the extent and clonal relationship of these neoplasms.
References
Campo E, Harris NL, Pileri SA et al (2017) WHO Classification of Tumors of Haematopoietic and Lymphoid Tissues. IARC WHO Classification of Tum
Ranaldi R, Goteri G, Baccarini MG et al (2002) A clinicopatho- logical study of 152 surgically treated primary gastric lymphomas with survival analysis of 109 high grade tumors. J Clin Pathol 55:346–351. https://doi.org/10.1136/jcp.55.5.346
A clinical evaluation of the international lymphoma study group classification of non-hodgkin’s lymphoma (1997) The non-hodgkin’s lymphoma classification project. Blood 89(11):3909–3918. https://doi.org/10.1182/blood.V89.11.3909
Kalpadakis C, Pangalis GA, Vassilakopoulos TP et al (2014) Clinical aspects of malt lymphomas. Curr Hematol Malig Rep 9(3):262–272. https://doi.org/10.1007/s11899-014-0218-1
Thieblemont C, Bertoni F, Copie-Bergman C, Ferreri AJM, Ponzoni M (2014) Chronic inflammation and extra-nodal marginal-zone lymphomas of MALT-type. Semin Cancer Biol 24:33–42. https://doi.org/10.1016/j.semcancer.2013.11.005
Du M, Diss TC, Xu C, Peng H, Isaacson PG, Pan L (1996) Ongoing mutation in MALT lymphoma immunoglobulin gene suggests that antigen stimulation plays a role in the clonal expansion. Leukemia 10(7):1190–1197
Ye H, Liu H, Attygalle A et al (2003) Variable frequencies of t(11;18)(Q21;q21) in MALT lymphomas of different sites: significant association with CagA strains of H pylori in gastric MALT lymphoma. Blood 102(3):1012–1018. https://doi.org/10.1182/blood-2002-11-3502
Qin Y, Greiner A, Trunk MJ, Schmausser B, Ott MM, Müller-Hermelink HK (1995) Somatic hypermutation in low-grade mucosa-associated lymphoid tissue-type B-cell lymphoma. Blood 86(9):3528–3534. https://doi.org/10.1182/blood.V86.9.3528.bloodjournal8693528
Du MQ, Xu CF, Diss TC et al (1996) Intestinal dissemination of gastric mucosa-associated lymphoid tissue lymphoma. Blood 88:4445–4451. https://doi.org/10.1182/blood.V88.12.4445.bloodjournal88124445
Kawamata N, Miki T, Fukuda T et al (1995) Determination of a common clonal origin of gastric and pulmonary mucosa associated lymphoid tissue lymphomas presenting five years apart. Intern Med 34:220–223. https://doi.org/10.2169/internalmedicine.34.220
Alpen B, Wundisch T, Dierlamm J et al (2004) Clonal relationship in multifocal non-Hodgkin’s lymphoma of mucosa-associated lymphoid tissue (MALT). Ann Hematol 83:124–126. https://doi.org/10.1007/s00277-003-0763-5
McCormick C, Philp E, Mansi J et al (1994) Clonal analysis of three morphologically distinct lymphomas occurring in the same patient. J Clin Pathol 47:1038–1042. https://doi.org/10.1136/jcp.47.11.1038
Du MQ, Diss TC, Dogan A et al (2000) Clone-specific PCR reveals wide dissemination of gastric MALT lymphoma to the gastric mucosa. J Pathol 192:488–493. https://doi.org/10.1002/1096-9896(2000)9999:9999%3c::AID-PATH727%3e3.0.CO;2-J
Montalbaan C, Castrillo JM, Abraira V et al (1995) Gastric B-cell mucosa-associated lymphoid tissue (MALT) lymphoma. Clinicopathological study and evaluation of the prognostic factors in 143 patients. Ann Oncol 6:355–362. https://doi.org/10.1093/oxfordjournals.annonc.a059184
Thieblemont C, Berger F, Dumontet C et al (2000) Mucosa-associated lymphoid tissue lymphoma is a disseminated disease in one third of 158 patients analyzed. Blood 95(3):802–806. https://doi.org/10.1182/blood.V95.3.802.003k19_802_806
Cogliatti SB, Schmid U, Schumacher U et al (1991) Primary B-cell gastric lymphoma: a clinicopathological study of 145 patients. Gastroenterology 101(5):1159–1170. https://doi.org/10.1016/0016-5085(91)90063-q
Konoplev S, Lin P, Qiu X, Medeiros LJ, Yin C (2010) Clonal relationship of extranodal marginal zone lymphomas of mucosa-associated lymphoid tissue involving different sites. Am J Clin Pathol 134(1):112–118. https://doi.org/10.1309/AJCP0HT6ZGSZKNFT
Stephen MR, Farquharson MA, Sharp RA, Jackson R (1998) Sequential malt lymphomas of the stomach, small intestine, and Gallbladder. J Clin Pathol 51(1):77–79. https://doi.org/10.1136/jcp.51.1.77
Yoshida-Sakai N, Oe K, Kimura Y et al (2022) Coexistence of gastric and pulmonary MALT lymphoma and a clonally related, disseminated large B cell lymphoma with conspicuous angiotropism. Ann Hematol 101(6):1383–1385. https://doi.org/10.1007/s00277-022-04752-8
Wang Y, Dong S, Jiang Z, Li A (2013) A case report of concurrent gastric and pulmonary mucosa-associated lymphoid tissue lymphomas and review of the literature on clonality analysis. Ann Hematol 92(12):1707–1709. https://doi.org/10.1007/s00277-013-1749-6
Iwano M, Okazaki K, Uchida K et al (2004) Characteristics of gastric B-cell lymphoma of mucosa-associated lymphoid tissue type involving multiple organs. J Gastroenterol 39(8):739–746. https://doi.org/10.1007/s00535-004-1382-1
Oh S, Kim N, Oh DH et al (2015) Concurrent gastric and pulmonary mucosa-associated lymphoid tissue lymphomas with pre-existing intrinsic chronic inflammation: a case report and a review of the literature. Gut Liver 9(3):424–429. https://doi.org/10.5009/gnl14072
Condom M, Climent F, Fernández D et al (2021) Clonal relationship in multisited mucosa-associated lymphoid tissue lymphomas: a single-centre experience. Br J Haematol 192(6):1020–1025. https://doi.org/10.1111/bjh.16717
Kalpadakis C, Pangalis GA, Vassilakopoulos TP et al (2014) Clinical aspects of malt lymphomas. Curr Hematol Malig Rep 9(3):262–272. https://doi.org/10.1007/s11899-014-0218-1
Moody S, Thompson JS, Chuang SS et al (2018) Novel GPR34 and CCR6 mutation and distinct genetic profiles in MALT lymphomas of different sites. Haematologica 103(8):1329–1336. https://doi.org/10.3324/haematol.2018.191601
Remstein ED, Dogan A, Einerson RR et al (2006) The incidence and anatomic site specificity of chromosomal translocations in primary extranodal marginal zone B-cell lymphoma of mucosa-associated lymphoid tissue (MALT lymphoma) in North America. Am J Surg Pathol 30(12):1546–1553. https://doi.org/10.1097/01.pas.0000213275.60962.2a
Streubel B, Simonitsch-Klupp I, Müllauer L et al (2004) Variable frequencies of MALT lymphoma-associated genetic aberrations in MALT lymphomas of different sites. Leukemia 18(10):1722–1726. https://doi.org/10.1038/sj.leu.2403501
Cascione L, Rinaldi A, Bruscaggin A et al (2019) Novel insights into the genetics and epigenetics of MALT lymphoma unveiled by next generation sequencing analyses. Haematologica 104(12):e558–e561. https://doi.org/10.3324/haematol.2018.214957
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Given the retrospective nature and absence of protected health information, IRB approval was not sought.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, K., Ward, N. Clonal relationship of extranodal marginal zone lymphomas of mucosa-associated lymphoid tissue (MALT lymphoma) involving multiple organ systems with review of the literature. J Hematopathol 15, 215–220 (2022). https://doi.org/10.1007/s12308-022-00516-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12308-022-00516-5