Abstract
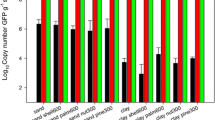
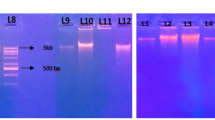
In this study, novel DNA extraction and purification methods were developed to obtain high-quantity and reliable quality DNA from the microbial community of agricultural yellow loess soil samples. The efficiencies of five different soil DNAextraction protocols were evaluated on the basis of DNA yield, quality and DNA shearing. Our suggested extraction method, which used CTAB, EDTA and cell membrane lytic enzymes in the extraction followed by DNA precipitation using isopropanol, yielded a maximum DNA content of 42.28 ± 5.59 µg/g soil. In addition, among the five different purification protocols, the acid-treated polyvinyl polypyrrolidone (PVPP) spin column purification method yielded high-quality DNA and recovered 91% of DNA from the crude DNA. Spectrophotometry revealed that the ultraviolet A 260/A 230 and A 260/A 280 absorbance ratios of the purified DNA were 1.82 ± 0.03 and 1.94 ± 0.05, respectively. PCR-based 16S rRNA amplification showed clear bands at ~1.5 kb with acid-treated PVPP–purified DNA templates. In conclusion, our suggested extraction and purification protocols can be used to recover high concentration, high purity, and high-molecular-weight DNA from clay and silica-rich agricultural soil samples.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Amsaleg, C.L.R., Garnier-Sillam, E., and Harry, M. 2001. Extraction and purification of microbial DNA from soil and sediment samples. Appl. Soil Ecol. 18, 47–60.
Arbeli, Z. and Fuentes, C.L. 2007. Improved purification and PCR amplification of DNA from environmental samples. FEMS Microbiol. Lett. 272, 269–275.
Bakken, L.R. and Lindahl, V. 1995. Recovery of bacterial cells from soil. Nucleic Acids in the Environ, pp. 9–27. Springer-Verlag, Berlin, Germany.
Bürgmann, H., Pesaro, M., Widmer, F., and Zeyer, J. 2001. A strategy for optimizing quality and quantity of DNA extracted from soil. J. Microbiol. Method. 45, 7–20.
Erb, R.W. and Wagner-Dobler, I. 1993. Detection of polychlorinated biphenyl genes in polluted sediments by direct DNA extraction and polymerase chain reaction. Appl. Environ. Microbiol. 59, 4065–4073.
Forsberg, K.J., Reyes, A., Wang, B., Selleck, E.M., Sommer, M.O.A., and Dantas, G. 2012. The shared antibiotic resistome of soil bacteria and human pathogens. Scienc. 337, 1107–1111.
Harry, M., Gambier, B., Bourezgui, Y., and Garnier-Sillam, E. 1999. Evaluation of purification procedures for DNA extracted from organic rich samples: interference with humic substances. Analysi. 27, 439–442.
Ikeda, S., Watanabe, K.N., Minamisawa, K., and Ytow, N. 2004. Evaluation of soil DNA from arable land in Japan using a modified direct-extraction method. Microbes Environ. 19, 301–309.
Islam, M.R., Sultana, T., Joe, M.M., Cho, J.C., and Sa, T. 2012. Comparisons of direct extraction methods of microbial DNA from different paddy soils. Saudi J. Biol. Sci. 19, 337–342.
Kathiravan, M.N., Karthick, R., and Muthukumar, K. 2011. Ex situ bioremediation of Cr (VI) contaminated soil by Bacillus sp.: Batch and continuous studies. Chem. Eng. J. 169, 107–115.
Kauffmann, I.M., Schmitt, J., and Schmid, R.D. 2004. DNA isolation from soil samples for cloning in different hosts. Appl. Microbiol. Biotechnol. 64, 665–670.
Kozdroj, J. and Van Elsas, J.D. 2000. Bacterial community DNA extracted from soils polluted with heavy metals. Polish J. Environ. Studie. 9, 403–407.
Krsek, M. and Wellington, E.M.H. 1999. Comparison of different methods for the isolation and purification of total community DNA from soil. J. Microbiol. Method. 39, 1–16.
Lakay, F.M., Botha, A., and Prior, B.A. 2007. Comparative analysis of environmental DNA extraction and purification methods from different humic acid-rich soils. Appl. Microbiol. 102, 265–273.
LaMontagne, M.G., Michel, F.C.Jr., Holden, P.A., and Reddy, C.A. 2002. Evaluation of extraction and purification methods for obtaining PCR-amplifiable DNA from compost for microbial community analysis. J. Microbiol. Method. 49, 255–264.
Liesack, W., Weyland, H., and Stackebrandt, E. 1991. Potential risks of gene amplification by PCR as determined by 16S rDNA analysis of a mixed-culture of strict barophilic bacteria. Microb. Ecol. 21, 191–198.
McGarvey, K.M., Queitsch, K., and Fields, S. 2012. Wide variation in antibiotic resistance proteins identified by functional metagenomic screening of a soil DNA library. Appl. Environ. Microbiol. 78, 1708–1714.
Miller, D.N., Bryant, J.E., Madsen, E.L., and Ghiorse, W.C. 1999. Evaluation and optimization of DNA extraction and purification procedures for soil and sediment samples. Appl. Environ. Microbiol. 65, 4715–4724.
More, M.I., Herrick, J.B., Silva, M.C., Ghiorse, W.C., and Madsen, E.L. 1994. Quantitative cell lysis of indigenous microorganisms and rapid extraction of microbial DNA from sediment. Appl. Environ. Microbiol. 60, 1572–1580.
Orsini, M. and Romano-Spica, V. 2001. A microwave-based method for nucleic acid isolation from environmental samples. Lett. Appl. Microbiol. 33, 17–20.
Porteous, L.A., Armstrong, J.L., Seidler, R.J., and Watrud, L.S. 1994. An effective method to extract DNA from environmental samples for polymerase chain reaction amplification and DNA finger print analysis. Curr. Microbiol. 29, 301–307.
Romanowski, G., Lorenz, M.G., and Wackernagel, W. 1993. Use of polymerase chain reaction and electroporation of Escherichia coli to monitor the persistence of extracellular plasmid DNA introduced into natural soils. Appl. Environ. Microbiol. 59, 3438–3446.
Sagar, K., Singh, S.P., Goutam, K.K., and Konwar, B.K. 2014. Assessment of five soil DNA extraction methods and a rapid laboratory-developed method for quality soil DNA extraction for 16S rDNA-based amplification and library construction. J. Microbiol. Method. 97, 68–73.
Sambrook, J., Fritsch, E.F., and Maniatis, T. 1987. Molecular cloning-a laboratory manual. Cold Spring Harbor, USA, Cold Spring Harbor Laboratory Press, London.
Selvin, J., Kennedy, J., Lejon, D.P.H., Kiran, G.S., and Dobson, D.W. 2012. Isolation identification and biochemical characterization of a novel halo-tolerant lipase from the metagenome of the marine sponge Haliclona simulans. Microb. Cell Fact. 11, 72.
Smith, G.B. and Tiedje, J.M. 1992. Isolation and characterization of a nitrite reductase gene and its use as a probe for denitrifying bacteria. Appl. Environ. Microbiol. 58, 376–384.
Tsai, Y.L. and Olson, B.H. 1990. Effects of Hg2+, CH3-Hg+, and temperature on the expression of mercury resistance genes in environmental bacteria. Appl. Environ. Microbiol. 56, 3266–3272.
Wilkinson, D.E., Jeanicke, T., and Cowan, D.A. 2002. Efficient molecular cloning of environmental DNA from geothermal sediments. Biotechnol. Lett. 24, 155–161.
Yeates, C., Gillings, M.R., Davison, A.D., Altavilla, N., and Veal, D.A. 1997. PCR amplification of crude microbial DNA extracted from soil. Lett. Appl. Microbiol. 25, 303–307.
Yeh, Y.F., Chang, S.C., Kuo, H.W., Tong, C.G., Yu, S.M., and Ho, T.H. 2013. A metagenomic approach for the identification and cloning of an endoglucanase from rice straw compost. Gen. 519, 360–366.
Young, C.C., Burghoff, R.L., Keim, L.G., Minak-Bernero, V., Lute, J.R., and Hinton, S.M. 1993. Polyvinylpyrrolidone-agarose gel electrophoresis purification of polymerase chain reaction-amplifiable DNA from soils. Appl. Environ. Microbiol. 59, 1972–1974.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kathiravan, M.N., Gim, G.H., Ryu, J. et al. Enhanced method for microbial community DNA extraction and purification from agricultural yellow loess soil. J Microbiol. 53, 767–775 (2015). https://doi.org/10.1007/s12275-015-5454-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-015-5454-0