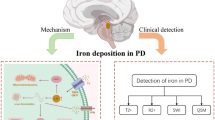
Abstract
The identification of sensitive and specific biomarkers for Parkinson’s disease (PD) poses an important clinical challenge. A potential biomarker for early diagnosis and disease monitoring of PD is region-specific iron. Iron accumulation in the substantia nigra pars compacta is considered a main characteristic of PD. However, questions remain, such as the relationship between nigral iron and clinical indices of PD (motor impairment or disease duration). Further, previous studies have suggested the influence of iron on other nuclei. Iron quantification using magnetic resonance imaging (MRI) allows for studies of the relationship between regional iron and clinical symptoms in vivo. Thus, in this review we discuss the following topics: the technological development of MRI in measuring brain iron, nigral iron as a potential marker for PD in both clinical and prodromal stages, other influences of regional iron on PD, and clinical translation and future perspectives.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Parkinson’s disease (PD) is one of the most common neurodegenerative diseases and has heterogeneous clinical symptoms [1, 2]. The clinical diagnosis of PD lacking objective evidence mainly depends on the occurrence of motor impairments, such as akinesia, rigidity, and resting tremor [2, 3]. Significant motor symptoms generally emerge only after 50%–70% of the dopaminergic neurons in the substantia nigra pars compacta (SNc) are irreversibly lost [4, 5], which indicates a pathologically advanced stage [6, 7]. In clinical practice, there are no sensitive and specific biomarkers for PD. Therefore, the rate of misdiagnosis is high, ranging from 12% to 74%, and the rates are markedly higher in patients with a shorter disease duration or without a clear response to dopaminergic medication [8]. Previous studies indicate little improvement in the diagnostic accuracy of PD over the past 20 years [8, 9], indicating an urgent need for a clinical biomarker to aid diagnosis.
Iron plays an essential role in physiological function during normal aging, including oxygen transport, DNA synthesis and repair, mitochondrial respiration, myelin synthesis, neurotransmitter synthesis and metabolism [10]. Dysfunction of iron homeostasis can contribute to the generation of free radicals, leading to the oxidation of proteins, lipids, lipoproteins, and other cellular components, which can result in neuronal death [10,11,12]. Pathological iron accumulation in the nigra has been reported in PD by previous histochemical studies [13,14,15]. A recent study also indicated that iron loading contributes to increasing the level of alpha-synuclein (the core pathology of PD) [16]. Therefore, nigral iron might be a potential marker for PD. However, these studies did not provide in vivo clinical verification or investigate the interaction between nigral iron and clinical symptoms.
To prompt clinical investigation, magnetic resonance imaging (MRI) has been developed to evaluate brain iron content in vivo and, with decades of progress, has reached a milestone in quantifying iron content. In this review, we discuss the technological development of MRI in assessing brain iron, nigral iron as a potential marker for PD in both clinical and prodromal stages, other influences of regional iron on PD symptoms, and clinical translation and future perspectives.
Technological Development of MRI in Measuring Brain Iron
Brain iron is mainly stored in the form of ferritin, which is reserved for the needs of the brain [17, 18]. In MRI, brain iron not only changes the relaxation behavior of tissue water surrounding ferritin, but also introduces susceptibility changes and microscopic field gradients. Iron levels in vivo have an effect on signal intensity, causing signal changes in T2 and T2* [19] and in phase values [20], enabling quantitative MRI iron mapping. However, these methods have limitations. First, both T2 (R2 = 1/T2) and T2* (R2* = 1/T2*) values are greatly influenced by the water content of diseased tissues, such as degenerating nuclei in PD. Second, the field-dependent R2 increase (FDRI) [21, 22], a difference in measures of brain R2 obtained with increasing magnetic field strength, is time-consuming and clinically challenging. Third, phase imaging is not easily reproducible because the signal phase is nonlocal and orientation-dependent [23, 24]. Recently, quantitative susceptibility mapping (QSM) gave rise to a major evolution in the quantification of iron content by converting phase shift to a localized magnetic susceptibility [23, 25, 26]. In past decades, field-dependent methods [21, 22], T2* mapping [19], T2′ mapping (R2′ = 1/T2′ = R2* − R2) [27, 28], phase imaging [29, 30] and QSM [31,32,33] for detecting iron content in healthy controls have been highly consistent with postmortem results. Therefore, MR iron mapping has promising potential for quantifying brain iron in vivo and for investigating the underlying pathological alterations in PD.
Nigral Iron as a Potential Biomarker for PD (Clinical and Prodromal)
Iron-related oxidative damage in the SNc is a common explanation for the loss of dopaminergic neurons in PD. The nigral degeneration leads to the depletion of dopamine in the striatum, which is assumed to be correlated with disease severity. In efforts to obtain a relevant biomarker for clinical-stage PD patients, extensive MRI studies have been performed to investigate the nigral iron changes in PD and their correlation with disease severity. A majority of studies using MRI techniques [34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65] (Table 1) support the accumulation of nigral iron in PD, which agrees with the previous histochemical findings. More interestingly, many of the studies have also reported a significant correlation between nigral iron measured by MRI and part III of the Unified Parkinson’s Disease Rating Scale (UPDRS III score, motor impairment) or Hoehn–Yahr stages (clinical disease stage) [36, 40,41,42, 49, 50, 53,54,55,56,57,58,59,60]. Moreover, it has also been reported that nigral iron accumulation has a good discriminative capacity for distinguishing PD patients from controls [63, 64]. Therefore, the quantification of nigral iron content by MRI is a promising biomarker for PD.
Concerning its use as a biomarker, it is crucial to understand whether nigral iron content increases or remains stable throughout the duration of disease. However, published studies have reported conflicting findings [35, 41, 49, 50, 52, 54, 60, 61, 64, 65]. Except for a few studies reporting a positive correlation between nigral iron content and disease duration [50, 52, 54], most studies, including some longitudinal analyses, have failed to find such a correlation [35, 41, 49, 60, 61, 64, 65]. Various sample sizes and techniques used by the studies likely contribute to the discrepancies (Table 1). Further, no study has analyzed the interaction of disease duration and nigral iron independent of the motor impairment in PD. Though the majority of studies support the conclusion that iron is elevated at the time of diagnosis and remains constant during the course of the disease, further studies are needed to confirm the relationship without the influence of motor impairment.
Nigrosome 1, located in the caudal and mediolateral SNc, plays an early and large role in PD [4, 5]. Recently, Blazejewska et al. [66] reported the loss of dorsolateral nigral hyperintensity (DNH) in a T2* map in PD patients, and confirmed correspondingly low neuromelanin in this region. These findings indicate that regional iron accumulation in nigrosome 1 mirrors the loss of dopaminergic neurons, which suggests that evaluating the level of DNH might provide a biomarker for PD. Several studies have also confirmed its application to differentiate PD from normal aging with high sensitivity and specificity [67,68,69]. However, only 13 of 35 normal subjects showed bilateral or unilateral DNH in a study by He and colleagues [54]. Intriguingly, De Marzi et al. [70] have reported that, as a majority of patients with idiopathic rapid eye movement sleep behavior disorder (iRBD) go on to develop PD, the loss of DNH in iRBD patients was found to be similar to that in PD patients. This indicates that evaluating DNH could be used to classify early-stage PD, referred to as prodromal PD [71]. As PD is one of the most common neurodegenerative disorders, low DNH in normal individuals [54] may indicate prodromal PD or high-risk cases. Studies with larger samples and different clinical subtypes, especially longitudinal studies, are still needed to confirm the usefulness of measuring DNH levels for future clinical translation, especially in the identification of prodromal PD.
Because the main degenerative alterations in PD are limited to the SNc, some of the above studies have tried to distinguish the SNc from the SN. Martin et al. [72] have suggested that because the SN pars reticulata (SNr), which has a low signal in T2 and phase imaging but a high signal in QSM, has a significantly higher iron content than the SNc; the SNc corresponds best to the high signal area that is interposed between the low signal structures of the SNr and the red nucleus (in T2 and phase imaging; opposite in QSM). Previous studies have confirmed the presence of iron accumulation in the SNc in PD patients [39, 41, 42, 52, 55,56,57,58,59, 61, 62]. Guan et al. [56] have further confirmed that PD patients with different motor subtypes also show iron accumulation in the SNc. However, identification of the SNc remains a challenge with the available imaging resolution. Thus, we hope that future studies using ultrahigh field MRI will contribute to its precise delineation [51].
Other researchers have explored the possible physiochemical underpinnings of nigral iron accumulation. Jin et al. [49] have suggested that nigral iron content is highly correlated with the level of serum ceruloplasmin (an important anti-oxidative protein), and shown that PD patients with a decreased level of serum ceruloplasmin have significantly higher iron content than PD patients with a normal level. Although it is unclear how serum cholesterol levels and uric acid interact with nigral iron content, Du et al. have demonstrated a significant negative correlation between nigral iron content and total cholesterol, and Kim and Lee have confirmed both low serum uric acid levels and increased nigral iron content in PD, but without a correlation between them [73, 74]. Serum samples can be easily obtained and have a high potential to provide necessary biological information. New studies, especially those focusing on the relationship between anti-oxidative materials and nigral iron accumulation, will deepen our understanding. Test–retest studies will be important for translating these findings into clinical applications for classifying at-risk individuals.
In brief, nigral degeneration is a core pathology of clinical-stage PD. Although a single biomarker cannot definitively identify PD, we believe that quantification of nigral iron or qualitative analysis of DNH will contribute to recognizing high-risk individuals and possibly aid diagnosis.
Other Influences of Regional Iron on PD
Subcortical nuclei, like the globus pallidus (GP), putamen, caudate nuclei (CN), red nuclei (RN), and dentate nuclei (DN), are also rich in iron [17]. However, the influences of iron in these nuclei are not well understood in PD. Previous MRI studies have found significant iron accumulation in the GP [37, 53, 55, 75], especially in late-stage PD patients [55]. Martin et al.[41] and Ye et al.[75] have reported that the iron content in the GP is significantly correlated with disease severity. In a cohort of PD patients with a long disease duration (>10 years), Graham et al. and Ryvlin et al. [34, 38] have found significantly decreased iron content in the putamen while Ye et al. [75] have reported significantly increased iron content in their PD patients. In addition, in a post-hoc correlation analysis, Bunzeck et al. and Guan et al. [56, 76] have reported that iron content in the CN is significantly negatively correlated with the severity of akinesia/rigidity in PD. Iron content in these nuclei plays an important role in maintaining physical function. Because of the degenerative changes in the basal ganglia in PD, the regional iron alterations may indicate abnormal metabolism. Sufficient evidence from basic research is still needed to explain these MRI findings.
It is thought that increased cerebellar function might contribute to the development of levodopa-induced dyskinesia in PD. Lewis et al. [50] have reported significant iron accumulation in the RN in patients with this form of PD, and suggested that increased RN iron might reflect structural changes associated with cerebello-thalamo-cortical compensation in PD. Guan et al. have also observed this phenomenon in late-stage PD patients [55], which likely reflects increased cerebellar compensation. The DN is another important region in the cerebellar circuit, and both He et al. and Guan et al. [56, 77] have reported significant iron accumulation in this region in tremor-dominant PD patients that correlates with tremor severity. Thus, iron content in the RN and DN possibly play roles in activating RN function (cerebellar compensation) and tremor generation, respectively.
Taken together, region-specific iron content shows complex regulation secondary to nigral degeneration in PD patients. Confirmation of these findings would greatly contribute to understanding the role of iron in PD and the search for promising biomarkers.
Clinical Translation and Future Perspectives
To improve the current diagnostic accuracy of PD, a biomarker with high sensitivity and specificity would be of great use. Reviewing the decades of effort toward this goal indicates that identification of a single biomarker is unlikely, whereas the use of multiple biomarkers may be beneficial. Iron-related biomarkers offer useful information about the disease mechanism and clinical diagnosis as the improved techniques (such as QSM) and increased field strength provide more precise measurements than previously available with high resolution and signal-to-noise ratios. Novel statistical methods, including voxel-wise analysis throughout the brain [78], would also contribute to a better understanding of the iron abnormalities in PD and provide potentially sensitive biomarkers. Of course, to determine their potential for clinical translation, test-retest reproducibility, the ability to monitor disease progression without age bias, and compensatory mechanisms or treatments still need to be investigated with larger cohorts or exclusive subtypes [79]. Once prodromal PD was defined [71], the first investigation of a nigral iron abnormality in iRBD patients (high-risk prodromal PD symptom) was reported [70]. Future efforts should aim to deepen understanding of the roles of regional iron in prodromal symptoms and also verify the candidate biomarkers (to be used in combination with other biomarkers) longitudinally. The studies reviewed here indicate strong support for the clinical translation of iron-related biomarkers for future early diagnosis of PD and disease monitoring.
References
Jankovic J, McDermott M, Carter J, Gauthier S, Goetz C, Golbe L, et al. Variable expression of Parkinson’s disease: a base-line analysis of the DATATOP cohort. The Parkinson Study Group. Neurology 1990, 40: 1529–1534.
Lees AJ, Hardy J, Revesz T. Parkinson’s disease. Lancet 2009, 373: 2055–2066.
Hughes AJ, Daniel SE, Kilford L, Lees AJ. Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: A clinico-pathological study of 100 cases. J Neurol Neurosurg Psychiatry 1992, 55: 181–184.
Damier P, Hirsch EC, Agid Y, Graybiel AM. The substantia nigra of the human brain. II. Patterns of loss of dopamine-containing neurons in Parkinson’s disease. Brain 1999, 122: 1437–1448.
Fearnley JM, Lees AJ. Ageing and Parkinson’s disease: substantia nigra regional selectivity. Brain 1991, 114: 2283–2301.
Braak H, Ghebremedhin E, Rub U, Bratzke H, Del TK. Stages in the development of parkinson’s disease-related pathology. Cell Tissue Res 2004, 318: 121–134.
Braak H, Del TK, Rub U, de Vos RA, Jansen SE, Braak E. Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging 2003, 24: 197–211.
Adler CH, Beach TG, Hentz JG, Shill HA, Caviness JN, Driver-Dunckley E, et al. Low clinical diagnostic accuracy of early vs advanced parkinson disease: clinicopathologic study. Neurology 2014, 83: 406–412.
Rajput AH, Rozdilsky B, Rajput A. Accuracy of clinical diagnosis in parkinsonism–a prospective study. Can J Neurol Sci 1991, 18: 275–278.
Ward RJ, Zucca FA, Duyn JH, Crichton RR, Zecca L. The role of iron in brain ageing and neurodegenerative disorders. Lancet Neurol 2014, 13: 1045–1060.
Ward RJ, Dexter DT, Crichton RR. Neurodegenerative diseases and therapeutic strategies using iron chelators. J Trace Elem Med Biol 2015, 31: 267–273.
Gaasch JA, Lockman PR, Geldenhuys WJ, Allen DD, Van der Schyf CJ. Brain iron toxicity: differential responses of astrocytes, neurons, and endothelial cells. Neurochem Res 2007, 32: 1196–1208.
Sofic E, Riederer P, Heinsen H, Beckmann H, Reynolds GP, Hebenstreit G, et al. Increased iron (III) and total iron content in post mortem substantia nigra of Parkinsonian brain. J Neural Transm 1988, 74: 199–205.
Dexter DT, Wells FR, Agid F, Agid Y, Lees AJ, Jenner P, et al. Increased nigral iron content in postmortem parkinsonian brain. Lancet 1987, 2: 1219–1220.
Sofic E, Paulus W, Jellinger K, Riederer P, Youdim MB. Selective increase of iron in substantia nigra zona compacta of parkinsonian brains. J Neurochem 1991, 56: 978–982.
Wan W, Jin L, Wang Z, Wang L, Fei G, Ye F, et al. Iron deposition leads to neuronal alpha-synuclein pathology by inducing autophagy dysfunction. Front Neurol 2017, 8: 1.
Hallgren B, Sourander P. The effect of age on the non-haemin iron in the human brain. J Neurochem 1958, 3: 41–51.
Morris CM, Candy JM, Oakley AE, Bloxham CA, Edwardson JA. Histochemical distribution of non-haem iron in the human brain. Acta Anat (Basel) 1992, 144: 235–257.
Langkammer C, Krebs N, Goessler W, Scheurer E, Ebner F, Yen K, et al. Quantitative MR imaging of brain iron: a postmortem validation study. Radiology 2010, 257: 455–462.
Haacke EM, Xu Y, Cheng YC, Reichenbach JR. Susceptibility weighted imaging (SWI). Magn Reson Med 2004, 52: 612–618.
Bartzokis G, Mintz J, Sultzer D, Marx P, Herzberg JS, Phelan CK, et al. In vivo MR evaluation of age-related increases in brain iron. AJNR Am J Neuroradiol 1994, 15: 1129–1138.
Bartzokis G, Beckson M, Hance DB, Marx P, Foster JA, Marder SR. MR Evaluation of age-related increase of brain iron in young adult and older normal males. Magn Reson Imaging 1997, 15: 29–35.
Liu C, Li W, Tong KA, Yeom KW, Kuzminski S. Susceptibility-weighted imaging and quantitative susceptibility mapping in the brain. J Magn Reson Imaging 2015, 42: 23–41.
Yablonskiy DA, Haacke EM. Theory of NMR signal behavior in magnetically inhomogeneous tissues: the static dephasing regime. Magn Reson Med 1994, 32: 749–763.
Li W, Wu B, Liu C. Quantitative susceptibility mapping of human brain reflects spatial variation in tissue composition. Neuroimage 2011, 55: 1645–1656.
Liu T, Liu J, de Rochefort L, Spincemaille P, Khalidov I, Ledoux JR, et al. Morphology enabled dipole inversion (MEDI) from a single-angle acquisition: comparison with COSMOS in human brain imaging. Magn Reson Med 2011, 66: 777–783.
Qin Y, Zhu W, Zhan C, Zhao L, Wang J, Tian Q, et al. Investigation on positive correlation of increased brain iron deposition with cognitive impairment in Alzheimer disease by using quantitative MR R2’ mapping. J Huazhong Univ Sci Technolog Med Sci 2011, 31: 578–585.
Gelman N, Gorell JM, Barker PB, Savage RM, Spickler EM, Windham JP, et al. MR imaging of human brain at 3.0 T: preliminary report on transverse relaxation rates and relation to estimated iron content. Radiology 1999, 210: 759–767.
Zhu WZ, Zhong WD, Wang W, Zhan CJ, Wang CY, Qi JP, et al. Quantitative MR phase-corrected imaging to investigate increased brain iron deposition of patients with Alzheimer disease. Radiology 2009, 253: 497–504.
Xu X, Wang Q, Zhang M. Age, gender, and hemispheric differences in iron deposition in the human brain: an in vivo MRI study. Neuroimage 2008, 40: 35–42.
Langkammer C, Schweser F, Krebs N, Deistung A, Goessler W, Scheurer E, et al. Quantitative susceptibility mapping (QSM) as a means to measure brain iron? a post mortem validation study. Neuroimage 2012, 62: 1593–1599.
Li W, Wu B, Batrachenko A, Bancroft-Wu V, Morey RA, Shashi V, et al. Differential developmental trajectories of magnetic susceptibility in human brain gray and white matter over the lifespan. Hum Brain Mapp 2014, 35: 2698–2713.
Wu B, Li W, Guidon A, Liu C. Whole brain susceptibility mapping using compressed sensing. Magn Reson Med 2012, 67: 137–147.
Ryvlin P, Broussolle E, Piollet H, Viallet F, Khalfallah Y, Chazot G. Magnetic resonance imaging evidence of decreased putamenal iron content in idiopathic Parkinson’s disease. Arch Neurol 1995, 52: 583–588.
Antonini A, Leenders KL, Meier D, Oertel WH, Boesiger P, Anliker M. T2 relaxation time in patients with Parkinson’s disease. Neurology 1993, 43: 697–700.
Gorell JM, Ordidge RJ, Brown GG, Deniau JC, Buderer NM, Helpern JA. Increased iron-related MRI contrast in the substantia nigra in Parkinson’s disease. Neurology 1995, 45: 1138–1143.
Kosta P, Argyropoulou MI, Markoula S, Konitsiotis S. MRI evaluation of the basal ganglia size and iron content in patients with Parkinson’s disease. J Neurol 2006, 253: 26–32.
Graham JM, Paley MN, Grunewald RA, Hoggard N, Griffiths PD. Brain iron deposition in Parkinson’s disease imaged using the PRIME magnetic resonance sequence. Brain 2000, 123 Pt 12: 2423–2431.
Michaeli S, Oz G, Sorce DJ, Garwood M, Ugurbil K, Majestic S, et al. Assessment of brain iron and neuronal integrity in patients with Parkinson’s disease using novel mri contrasts. Mov Disord 2007, 22: 334–340.
Zhang W, Sun SG, Jiang YH, Qiao X, Sun X, Wu Y. Determination of brain iron content in patients with Parkinson’s disease using magnetic susceptibility imaging. Neurosci Bull 2009, 25: 353–360.
Martin WR, Wieler M, Gee M. Midbrain iron content in early Parkinson disease: a potential biomarker of disease status. Neurology 2008, 70: 1411–1417.
Nestrasil I, Michaeli S, Liimatainen T, Rydeen CE, Kotz CM, Nixon JP, et al. T1rho and T2rho MRI in the evaluation of Parkinson’s disease. J Neurol 2010, 257: 964–968.
Gupta D, Saini J, Kesavadas C, Sarma PS, Kishore A. Utility of susceptibility-weighted MRI in differentiating Parkinson’s disease and atypical Parkinsonism. Neuroradiology 2010, 52: 1087–1094.
Baudrexel S, Nurnberger L, Rub U, Seifried C, Klein JC, Deller T, et al. Quantitative mapping of T1 and T2* discloses nigral and brainstem pathology in early Parkinson’s disease. Neuroimage 2010, 51: 512–520.
Du G, Lewis MM, Sen S, Wang J, Shaffer ML, Styner M, et al. Imaging nigral pathology and clinical progression in Parkinson’s disease. Mov Disord 2012, 27: 1636–1643.
Du G, Lewis MM, Styner M, Shaffer ML, Sen S, Yang QX, et al. Combined R2* and diffusion tensor imaging changes in the substantia nigra in Parkinson’s disease. Mov Disord 2011, 26: 1627–1632.
Peran P, Cherubini A, Assogna F, Piras F, Quattrocchi C, Peppe A, et al. Magnetic resonance imaging markers of Parkinson’s disease nigrostriatal signature. Brain 2010, 133: 3423–3433.
Wang Y, Butros SR, Shuai X, Dai Y, Chen C, Liu M, et al. Different iron-deposition patterns of multiple system atrophy with predominant Parkinsonism and idiopathetic Parkinson diseases demonstrated by phase-corrected susceptibility-weighted imaging. AJNR Am J Neuroradiol 2012, 33: 266–273.
Jin L, Wang J, Zhao L, Jin H, Fei G, Zhang Y, et al. Decreased serum ceruloplasmin levels characteristically aggravate nigral iron deposition in Parkinson’s disease. Brain 2011, 134: 50–58.
Lewis MM, Du G, Kidacki M, Patel N, Shaffer ML, Mailman RB, et al. Higher iron in the red nucleus marks Parkinson’s dyskinesia. Neurobiol Aging 2013, 34: 1497–1503.
Lotfipour AK, Wharton S, Schwarz ST, Gontu V, Schafer A, Peters AM, et al. High resolution magnetic susceptibility mapping of the substantia nigra in Parkinson’s disease. J Magn Reson Imaging 2012, 35: 48–55.
Du G, Liu T, Lewis MM, Kong L, Wang Y, Connor J, et al. Quantitative susceptibility mapping of the midbrain in Parkinson’s disease. Mov Disord 2016, 31: 317–324.
Langkammer C, Pirpamer L, Seiler S, Deistung A, Schweser F, Franthal S, et al. Quantitative susceptibility mapping in Parkinson’s disease. PLOS One 2016, 11: e162460.
He N, Ling H, Ding B, Huang J, Zhang Y, Zhang Z, et al. Region-specific disturbed iron distribution in early idiopathic Parkinson’s disease measured by quantitative susceptibility mapping. Hum Brain Mapp 2015, 36: 4407–4420.
Guan X, Xuan M, Gu Q, Huang P, Liu C, Wang N, et al. Regionally progressive accumulation of iron in Parkinson’s disease as measured by quantitative susceptibility mapping. Nmr Biomed 2017, 30(4). doi: 10.1002/nbm.3489.
Guan X, Xuan M, Gu Q, Xu X, Huang P, Wang N, et al. Influence of regional iron on the motor impairments of Parkinson’s disease: a quantitative susceptibility mapping study. J Magn Reson Imaging 2017, 45: 1335–1342.
Wieler M, Gee M, Martin WR. Longitudinal midbrain changes in early Parkinson’s disease: iron content estimated from R*/MRI. Parkinsonism Relat Disord 2014, 21: 179–183.
Wang C, Fan G, Xu K, Wang S. Quantitative assessment of iron deposition in the midbrain using 3D-enhanced T2 star weighted angiography (ESWAN): a preliminary cross-sectional study of 20 Parkinson’s disease patients. Magn Reson Imaging 2013, 31: 1068–1073.
Ulla M, Bonny JM, Ouchchane L, Rieu I, Claise B, Durif F. Is R2* a new MRI biomarker for the progression of Parkinson’s disease? A longitudinal follow-up. PLOS One 2013, 8: e57904.
Wallis LI, Paley MN, Graham JM, Grunewald RA, Wignall EL, Joy HM, et al. MRI assessment of basal ganglia iron deposition in Parkinson’s disease. J Magn Reson Imaging 2008, 28: 1061–1067.
Rossi ME, Ruottinen H, Saunamaki T, Elovaara I, Dastidar P. Imaging brain iron and diffusion patterns: a follow-up study of Parkinson’s disease in the initial stages. Aca Radiol 2014, 21: 64–71.
Rossi M, Ruottinen H, Soimakallio S, Elovaara I, Dastidar P. Clinical MRI for iron detection in Parkinson’s disease. Clin Imaging 2013, 37: 631–636.
Murakami Y, Kakeda S, Watanabe K, Ueda I, Ogasawara A, Moriya J, et al. Usefulness of quantitative susceptibility mapping for the diagnosis of Parkinson disease. AJNR Am J Neuroradiol 2015, 36: 1102–1108.
Barbosa JH, Santos AC, Tumas V, Liu M, Zheng W, Haacke EM, et al. Quantifying brain iron deposition in patients with Parkinson’s disease using quantitative susceptibility mapping, R2 and R2. Magn Reson Imaging 2015, 33: 559–565.
Zhang J, Zhang Y, Wang J, Cai P, Luo C, Qian Z, et al. Characterizing iron deposition in Parkinson’s disease using susceptibility-weighted imaging: an in vivo MR study. Brain Res 2010, 1330: 124–130.
Blazejewska AI, Schwarz ST, Pitiot A, Stephenson MC, Lowe J, Bajaj N, et al. Visualization of Nigrosome 1 and its Loss in PD: pathoanatomical correlation and in vivo 7 T MRI. Neurology 2013, 81: 534–540.
Bae YJ, Kim JM, Kim E, Lee KM, Kang SY, Park HS, et al. Loss of nigral hyperintensity on 3 Tesla MRI of Parkinsonism: comparison with (123) I-FP-CIT SPECT. Mov Disord 2016, 31: 684–692.
Reiter E, Mueller C, Pinter B, Krismer F, Scherfler C, Esterhammer R, et al. Dorsolateral nigral hyperintensity on 3.0T susceptibility-weighted imaging in neurodegenerative Parkinsonism. Mov Disord 2015, 30: 1068–1076.
Schwarz ST, Afzal M, Morgan PS, Bajaj N, Gowland PA, Auer DP. The ‘Swallow Tail’ appearance of the healthy nigrosome - a new accurate test of Parkinson’s disease: a case-control and retrospective cross-sectional MRI study at 3T. PLoS One 2014, 9: e93814.
De Marzi R, Seppi K, Hogl B, Muller C, Scherfler C, Stefani A, et al. Loss of dorsolateral nigral hyperintensity on 3.0 tesla susceptibility-weighted imaging in idiopathic rapid eye movement sleep behavior disorder. Ann Neurol 2016, 79: 1026–1030.
Berg D, Postuma RB, Adler CH, Bloem BR, Chan P, Dubois B, et al. MDS research criteria for prodromal Parkinson’s disease. Mov Disord 2015, 30: 1600–1611.
Martin WR. Quantitative estimation of regional brain iron with magnetic resonance imaging. Parkinsonism Relat Disord 2009, 15 Suppl 3: S215–S218.
Kim TH, Lee JH. Serum uric acid and nigral iron deposition in Parkinson’s disease: a pilot study. PLoS One 2014, 9: e112512.
Du G, Lewis MM, Shaffer ML, Chen H, Yang QX, Mailman RB, et al. Serum cholesterol and nigrostriatal R2* values in Parkinson’s disease. PLoS One 2012, 7: e35397.
Ye FQ, Allen PS, Martin WR. Basal ganglia iron content in Parkinson’s disease measured with magnetic resonance. Mov Disord 1996, 11: 243–249.
Bunzeck N, Singh-Curry V, Eckart C, Weiskopf N, Perry RJ, Bain PG, et al. Motor phenotype and magnetic resonance measures of basal ganglia iron levels in Parkinson’s disease. Parkinsonism Relat Disord 2013, 19: 1136–1142.
He N, Huang P, Ling H, Langley J, Liu C, Ding B, et al. Dentate nucleus iron deposition is a potential biomarker for tremor-dominant Parkinson’s disease. NMR Biomed 2017, 30(4). doi: 10.1002/nbm.3554.
Acosta-Cabronero J, Cardenas-Blanco A, Betts MJ, Butryn M, Valdes-Herrera JP, Galazky I, et al. The whole-brain pattern of magnetic susceptibility perturbations in Parkinson’s disease. Brain 2017, 140: 118–131.
Wang J, Hoekstra JG, Zuo C, Cook TJ, Zhang J. Biomarkers of Parkinson’s disease: current status and future perspectives. Drug Discov Today 2013, 18: 155–162.
Acknowledgements
This review was supported by the 13th Five-year Plan for the National Key Research and Development Program of China (2016YFC1306600), the Fundamental Research Funds for the Central Universities of China (2017XZZX001-01), the 12th Five-year Plan for the National Science and Technology Supporting Program of China (2012BAI10B04), and the National Natural Science Foundation of China (81571654, 81371519, and 81301190).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guan, X., Xu, X. & Zhang, M. Region-Specific Iron Measured by MRI as a Biomarker for Parkinson’s Disease. Neurosci. Bull. 33, 561–567 (2017). https://doi.org/10.1007/s12264-017-0138-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12264-017-0138-x