Abstract
Worldwide, coastal wetlands are threatened by disrupted hydrology, urbanization, and sea-level rise. In southwest Florida, coastal wetlands include tidal creeks and coastal ponds, which are the primary habitats used by juvenile Tarpon, Megalops atlanticus, an important sport fish. Coastal ponds can occur near uplands and are ephemerally connected to the open estuary, creating conditions of variable dissolved oxygen and salinity. Juveniles can tolerate wide-ranging abiotic conditions, but little is known about how they egress from their remote nursery habitats, which often requires them to cross > 1 km of mangrove forest to reach the open estuary. The objective of this study was to (1) compare Tarpon body condition among ponds close to the open estuary versus those ponds farther away on the Cape Haze peninsula of Charlotte Harbor, Florida, and (2) using acoustic telemetry determine what factors contribute to Tarpon emigration from the ponds to open estuarine waters. We tested the hypothesis that distinct groups of Tarpon occur in isolated ponds, leading to variation in fish length and body condition, and that opportunities for emigration from these ponds hinge on high water events. No pond stood out as having Tarpon of low body condition. Factors contributing to increased probabilities of Tarpon emigration were low barometric pressure, high-water level, and Tarpon body length. Tarpon emigrated from ponds near tidal creeks during summer king tides, while tropical cyclone conditions were needed to allow for movement from ponds farther in the landscape. The juvenile Tarpon were later detected at the mouths of large rivers 30 km up-estuary. The characterizations of water levels and event criteria needed for successful Tarpon nurseries should aid in habitat conservation and the creation of Tarpon nursery habitat in restoration designs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Coastal wetland ecosystems, composed of estuaries, salt marshes, mangrove forests, seagrass meadows, and flood plain swamps, are among the most productive and most threatened ecosystems on the planet (Bassett et al. 2013). In the USA, the densities of human populations are highest within 80 km of the coast, leading to drastic alterations to the land-sea interface (Rappaport and Sachs 2003). The intense use of the coastal zone has led to water control efforts, impoundments, dredging, and loss of habitats to urban development (Dahl 2006; Brockmeyer et al. 2022; Bice et al. 2023). These habitat impacts can affect the connectivity and quality of juvenile fish habitat, including for those species that are valuable to coastal economies.
Intertidal mangrove habitats are used as nurseries that estuarine and reef fishes require during their early life history (Peters et al. 1998; Adams et al. 2006). Nurseries have been described as habitats that have high juvenile fish density, promote growth and survivorship, and ultimately allow for residency or emigration depending on life history (Beck et al. 2001). They contribute a greater than average number of individuals to the adult population on a per unit area basis than most other habitats (Dahlgren et al. 2006). The definition of nursery has been expanded such that mangroves and other habitat types should not be considered individual units but as a complex and connected landscape (Nagelkerken et al. 2015; Litvin et al. 2018). At low latitudes, the importance of mangroves in supporting fish nurseries is well-known (Lindall et al. 1973), yet there is a general lack of data on the spatial coverage of such habitats and the environmental conditions needed to support them (Wilson et al. 2019).
To meet the contemporary definition of nursery habitat, it is important to show that juvenile fish can transition between habitat types and recruit to the adult population (Beck et al. 2001; Gillanders 2005; Barbour et al. 2014). Compared to other estuarine-dependent species, juvenile Tarpon Megalops atlanticus are thought to be at greater risk of becoming trapped in coastal nurseries, which can be disconnected from the estuary during much of the year (Wilson et al. 2019). When juveniles have grown and are ready to leave the nursery habitat, they may need to travel long distances over structurally complex habitat to reach open estuarine waters (Mol et al. 2000; Adams and Murchie 2015). As such, it is important to understand the emigration dynamics of juvenile Tarpon to help conserve this recreational fishery found throughout the North Atlantic Ocean, Gulf of Mexico, and Caribbean Sea (Adams et al. 2014). In southwest Florida, for example, Tarpon are among the most sought-after sport fish and are targeted by recreational anglers as both juveniles and adults, generating an overall economic impact of 108 million dollars annually (Fedler 2011).
Juvenile Tarpon rely exclusively on coastal wetlands, which consist of a mosaic of pond and creek habitats, often with fringing vegetation, a mix of water depths, variable freshwater inflow, and restricted connection to the estuary that inhibits access by predatory fishes (Cyr 1991; Adams and Murchie 2015). The earliest life stages occupy estuarine impoundments, mangrove-lined ponds, and salt marsh pools (Robins 1977; Mace et al. 2018; Cianciotto et al. 2019), which maintain connection with the ocean for only brief periods of high water (Wade 1962). These stagnant pools and ponds experience highly variable salinities, periods of extremely shallow water, high hydrogen sulfide concentrations, and anoxia (Wade 1962; Robins 1977; Cyr 1991). Tarpon are adapted to such low oxygen habitats because they can gulp air into a highly vascularized physostomous swim bladder to extract atmospheric oxygen (Seymour et al. 2008). The tidally restricted habitats, both natural (e.g., ponds, creeks, marsh pools) and human-made (e.g., impoundments, mosquito ditches), offer favorable conditions for Tarpon during their early life stages (Mace et al. 2018).
In coastal southwest Florida, the predominant natural habitat consists of wetlands and associated water bodies, such as tidal creeks and coastal ponds. Coastal ponds often form at the origin of tidal creeks on the fringe of terrestrial upland habitats and are disconnected from the adjacent estuarine waters except for periods of connectivity during flooding events (Fig. 1). Evidence suggests Tarpon gain access to these habitats during early life stages (larval or post larval) at small sizes (Mace et al. 2018), while emigration occurs at relatively large sizes (300–400 mm standard length [SL]) after 1 year of growth (Breder 1944; Cyr 1991; Crabtree et al. 1997). Landscape habitat heterogeneity creates segregated groups of juveniles that could differ in length and body condition. Environmental factors that could differ across the coastal wetland landscape, and potentially influence length and body condition, include increased competition from high densities of fish (Wilson et al. 2019; Matthews and Marsh-Matthews 2003), reduced prey availability (Jones 1986), increased predation risk (Halpin 2000), or some combination of these. If habitat conditions impede growth, Tarpon may elect to exit the nursery at smaller sizes, which could increase vulnerability to predation (Sogard and Able 1994).
To emigrate from their juvenile habitat in coastal ponds, Tarpon undergo an ontogenetic shift in habitat use that involves navigation through flooded coastal wetlands consisting of mangrove and saltmarsh habitat. These movements can traverse kilometers of red mangrove Rhizophora mangle prop roots and black mangrove Avicennia germinans pneumatophores, woody debris, salt panne, and salt marsh vegetation before reaching a tidal creek. Emigration presumably becomes more challenging for Tarpon that remain in ponds for extended durations (> 1 year) and reach larger sizes. It is possible that juveniles could become trapped at some locations. Thus, it is important to conduct emigration studies across multiple years so it can be determined whether a location contributes juvenile Tarpon to the open estuary population every year, once every few years, or not at all. Given the relatively large size that Tarpon reach in nursery habitats (Cyr 1991; Crabtree et al. 1997) and the landscape they must navigate to reach a tidal creek, successful emigration likely occurs during high-water events.
The objective of this study was to (1) compare Tarpon body condition among ponds close to the open estuary versus those ponds farther away on the Cape Haze peninsula of Charlotte Harbor, Florida, and (2) using acoustic telemetry determine what factors contribute to Tarpon emigration from the ponds to open estuarine waters. We tested the hypothesis that distinct groups of Tarpon occur in isolated ponds, leading to variation in fish length and body condition. In addition, we sought to provide evidence of emigration from purported nursery habitat to the estuarine population by tracking the movement of juvenile Tarpon across a habitat mosaic from the juvenile habitat located in coastal ponds to the open estuary. We hypothesize that a combination of environmental and biological variables interacting simultaneously allows for habitat connectivity and provides the cues necessary for emigration. This work will justify conservation of remnant Tarpon nursery habitat by identifying which coastal ponds are most likely to contribute juvenile Tarpon to the adult population. Further, results can be used to characterize future restoration work (e.g., determination of elevations, flooding frequencies, landscape resistance between ponds and open estuary) to maximize the impact of resiliency planning.
Methods
Study Area and Coastal Pond Designations
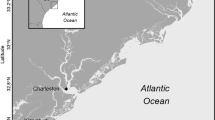
Charlotte Harbor is a 700-km2 shallow (generally < 4 m) coastal plain estuary on the southwest coast of Florida (Hammett 1990). The regional climate is subtropical, with mean seasonal water temperatures ranging from 12 to 36 °C and infrequent air temperatures below freezing (Poulakis et al. 2003). Annual rainfall averages 134 cm with as much as 70% of it typically occurring during summer months (July–August). Tropical cyclones have considerable effects on the estuarine system (Taylor 1974; Stevens et al. 2006). Tidal range is typically < 0.7 m (NOAAa).
Charlotte Harbor remains one of the least altered estuaries in peninsular Florida; most of the 250 km2 of mangrove forests and salt marshes are protected by public ownership (e.g., State Parks, Aquatic Preserves) or by regulation, with about one-third of the tidally affected area composed of wetlands (Pierce et al. 2004; Idelberger et al. 2011). Although the immediate natural areas surrounding Charlotte Harbor have been placed under protection, the population within the watershed has doubled since the 1990s (CHNEP 2023). Increases in population growth and land use have contributed to increased nitrogen loading and residual zones of hypoxia, anoxia, algal blooms, shoreline hardening, channelization, water withdrawal from tributaries and aquifers, manipulation of water flow regimes, increased nutrient input, and seagrass loss in the watershed (CHNEP 2000; Turner et al. 2006).
Coastal pond study sites were within the Charlotte Harbor Buffer Preserve State Park on the Cape Haze Peninsula (Fig. 2). The 183 km2 Charlotte Harbor Buffer Preserve State Park is Florida’s third largest state park and protects more than 160 km of coastline (FDEP 2023). This study was conducted in six remote mangrove dominated ponds fringed by primarily red mangrove, white mangrove Laguncalaria racemosa, and black mangrove. Surrounding habitats consist of continuations of mangrove forests and salt pannes made up of herbaceous halophytes, including salt grass Distichlis spicata, saltwort Batis maritima, and glasswort Salicornia spp.
Location of study area in Charlotte Harbor, Florida (USA), including the locations of Vemco acoustic receivers (blue dots) and HOBO water-level logging equipment (pink dots). Blue squares indicate ditched habitats and yellow squares are marsh habitats. Pond designations are adjacent to squares indicated by P, M, and D for proximal, mesial, and distal, respectively
Of the six ponds, three had ephemeral connections with the estuary, while three had consistent connections to the estuary through derelict mosquito ditches. Pond size varied from 5586 to 13,298 m2 and average depths ranged from 0.5 to 1.2 m with soft mud substrate. Salinity was reported using the Practical Salinity Scale. Profiles taken during companion fish sampling ranged from oligohaline (salinity of 5) to hypersaline (salinity of 70) and water temperatures ranged from 17 to 37 °C. Ponds were separated from the connecting creek systems over distances of 0.4–1.3 km through dense mangrove forests and salt pannes. Estimated distances were measured from pond to the receiving tidal creek, along the wetland contours, using Google Earth Pro (version 7.3.6.9345, 2023) satellite imagery. Ponds were given one of three distinct designations based on their position within the landscape. Pond connections were defined in relation to the distance from a tidal creek: proximal < 0.7 km, mesial 0.7 to 1 km, and distal 1.1 to 1.3 km. Following this scheme, ponds representing natural coastal wetland ponds with ephemeral connections are referred to as proximal marsh, mesial marsh, and distal marsh. The three ponds sampled with consistent connections provided by mosquito ditches are named proximal ditched, mesial ditched, and distal ditched. Construction and presence of mosquito ditching was confirmed with historic aerial photography (University of Florida Digital Collections 2023) and field verification.
Field Sampling
Field sampling was conducted from March 2019 to May 2022 and consisted of 11 sampling events targeting young-of-the-year (YOY) and age 1 fish. Fish were captured during daylight hours using a center bag seine (40 m; 25-mm stretch nylon mesh) and hook-and-line angling at each pond. The seine was set from a raft in a semicircle shape, extending 5 m from the shoreline or in open water targeting schools of rolling fish. Surface and bottom water parameters were measured at each sampling site using a handheld water quality instrument (Xylem, Inc., Yellow Springs, OH, USA) and included temperature (°C), salinity, specific conductivity (mS), dissolved oxygen (mg L−1), and pH.
Body Condition and Tracking Cohorts
Because of the remote locations of study sites and the limited amount of equipment feasible for transport by crew members, body height (mm) measurements in relation to SL (mm) was chosen as the best option for obtaining estimates of body condition. Body height is a common condition factor used to estimate body condition of fish (Jones et al. 1999; Poléo et al. 1995; Fernandes et al. 2020). To assess for differences in body condition among ponds, body height was measured from the dorsal side, anterior to the dorsal fin, to the ventral side of the fish in addition to measuring fish SL.
Tarpon were measured for body condition comparisons starting in the second year of the study, from May 2020 to May 2022. Following Ogle (2013), a two-parameter power function with a multiplicative error term was used to model the length-height relationship. The model used was Hi-aLbieϵi, where a and b are constants and ϵi is the multiplicative error term for the ith fish. This length-height model was then transformed to a linear model by taking the natural logarithms of both sides: log(Hi) = log(a) + blog(Li) + ϵi.
Body condition in the proximal, mesial, and distal ponds was analyzed using generalized linear models with R version 4.03.2020. To account for natural changes in body dimensions as fish age, we test for variations along multiple size specific regressions. The natural log-transformed lengths 4.6 (100 mm SL), 5.0 (150 mm), 5.5 (245 mm), and 6 (400 mm) were used to represent sizes present among ponds based on the mean size of 5.14 (170 mm) among all data. An analysis of covariance (ANCOVA) was used with the response variable of body height (mm) and independent variables of SL (mm) and pond type (proximal, mesial, distal) to determine if body condition differed. Marginal mean effects were computed and contrasted using the R package ‘emmeans’ (Lenth 2018). To test for variation in body condition among ponds, 95% confidence intervals associated with the post hoc, pairwise contrasts were examined based on marginal means. The p-values were adjusted for multiple comparisons using the Tukey method, and an alpha level of 0.05 was used for identifying statistically significant among-group differences. Age estimates were used to help identify cohorts, that is, which fish in a pond were YOY recruits and which fish were recruited in a previous year and had not emigrated. Scales from select individuals were collected from below the midpoint of the dorsal fin to obtain age estimates using methods described by Elmo et al. (2021). Scales provide a non-lethal option for estimation of juvenile Tarpon age and were found to accurately discriminate between age 0 and age 1 (Elmo et al. 2021). Three scales were removed from each fish prior to release, cleaned, and stored in individual bags until age estimation, which was completed by two readers using a Olympus SZX7 zoom stereo microscope, Olympus SC50 camera, and Olympus Cellsens software (Olympus Corporation, Tokyo, Japan). Age estimation of juveniles above 1 year was not reliable because the deposition of the annulus became too compact within the scale. The size at which 50% of the sampled population reached its first year of growth was used to determine the approximate size of age 1 fish. This size estimate at 1 year of growth was added to the length frequency plots to assist in tracking cohorts.
Emigration
Tarpon were tagged with acoustic transmitters during the spring of 2019, 2020, and 2021. Upon capture, Tarpon were held in a floating net pen until tagging. To minimize tag burden and associated negative effects (e.g., growth, survival, condition), fish ~ 250 mm SL or larger were selected for tagging, which occurred within 5 min of capture. Tagging consisted of a surgical procedure to insert an acoustic transmitter into the abdominal cavity of the fish. Tags used were Innovasea V9, 69 kHz (Innovasea Inc., Halifax, NS, Canada) except for one tag, which was a V13 implanted in a larger fish. A 12-mm half-duplex (HDX) passive integrated transponder (PIT) tag 2.12-mm diameter, 0.1 g in air (Oregon RFID, Portland, Oregon, USA), was used to identify any previously acoustic-tagged fish. The Innovasea V9 acoustic transmitter has an estimated battery life of 1.5 years (503 days). To implant tags, one to two scales were removed anterior to the vent and a 10-mm incision was made off center of the ventral midline using a scalpel. Fish were calmed by turning them ventral side up and covering their eyes. The incision was closed with a single Polysorb™ suture. Fish remained in water for the entirety of the tagging process, which was completed in < 5 min for each fish. Fish health was briefly assessed post-surgery for signs of physiological stress and fish were held until equilibrium was reached, at which time they were released at the capture location.
Acoustically tagged individuals were continuously monitored by eight autonomous stationary acoustic receivers (V2RW receivers, Innovasea Inc., Halifax, NS, Canada; Fig. 2) between March 2019 and December 2022. One receiver was deployed in each pond, except the distal ditched pond, to detect fish presence before emigration. The distal ditched pond did not have an acoustic receiver because of its low densities of Tarpon and the requirement for fish to transition through the mesial ditched and proximal ditched ponds before reaching the creek array. Three receivers were placed throughout the connecting creek corridor to detect fish as they emigrated from the ponds into the Charlotte Harbor estuarine system. All receivers were placed along mangrove shorelines and secured using steel poles or PVC housing. Unique detections for individual fish were associated with a date and time to quantify fish movement in relation to a suite of environmental and physical characteristics. Supplemental telemetry data was contributed by a separate Florida Fish and Wildlife Conservation Commission (FWC) study with an acoustic array of 23 receivers monitoring the mouths of the Peace (n = 19) and Myakka rivers (n = 4) in upper Charlotte Harbor (Scharer et al. 2017).
Data Acquisition: Emigration Response Variables
To assess the factors driving Tarpon emigration from these habitats, variables related to suspected emigration cues were collected. Variables consisted of data that have been reported to elicit a movement response in fishes, including barometric pressure (Heupel et al. 2003; Udyawer et al. 2013), water level fluctuations (Bailey and Secor 2016; Rehage et al. 2022), water temperature (Bailey and Secor 2016; Mace et al. 2018), age and growth (Cyr. 1991; Crabtree et al. 1997), and pond connection type and distance relative to the estuary (Sheaves and Johnston 2008).
Daily minimum and maximum barometric pressure (mm Hg) data were collected to represent the presence of storm activity. Data were queried for the daily minimum and maximum barometric pressure to account for daily fluctuations.
Local hydrologic data, hereafter referred to as local water level, were derived from pond-specific water level loggers (HOBO U20L-01 Data Loggers, Onset Brands, Bourne, MA, USA) and included the daily minimum and maximum water levels and water temperatures. Data loggers were affixed to steel poles with PVC housings to restrict movement and minimize biofouling. Housings were secured 20 mm above the sediment to prevent sedimentation from occurring around the logger while minimizing the risk of air exposure during low-water periods. Loggers were programmed to measure water temperature (°C) and pressure (PSI) every hour. Minimum and maximum water temperatures from each day were used for analysis. Water level measurements were corrected for the distance of the logger above the sediment using field measured reference water heights (mm). Loggers deployed in air (mounted to tree trunks in the pond vicinity) were used to detect local barometric pressure changes which, when combined with water level logger data, were used to compensate for any atmospheric changes in the study area. The field-measured reference water levels combined with local barometric pressure data were used to derive precise pond water level data using HOBOware Pro software (version 3.7.20). Missing data due to logger malfunctions were substituted with data from multivariate imputation via chained equations using the MICE package in R (van Buuren and Groothuis-Oudshoorn 2011). The MICE procedure fills missing data by running a series of regressions with each variable of missing data being modeled conditionally upon the other variables present in the dataset (Azur et al. 2011).
Regional water level data consisted of the minimum and maximum daily water levels (m) queried from the National Oceanic and Atmospheric Administration Tides & Currents hydrologic station (NOAAb) in the Caloosahatchee River. The Caloosahatchee River tidal gauge is the closest water level gauge to the study area, approximately 40 km to the southeast. This gauge provided water level data, hereafter referred to as regional water level, that are applicable to the region’s water level. To account for fluctuations over a single day, the minimum and maximum daily values were included for both the local and regional water level data.
To better understand the role of fish length in relation to emigration success, SL (mm) was estimated for the time of emigration using a model of growth described by Graham et al. (2021), which was estimated at 1.56 mm day−1 for juvenile Tarpon in the northern Gulf of Mexico. Estimated standard length at time of emigration was determined by multiplying the number of days post tagging by 1.56 and adding the standard length recorded at time of tagging for each tagged Tarpon. Juveniles have been documented transitioning from these habitats at about 400 mm SL, which coincides with the end of 1 year of growth (Cyr 1991).
Connection type and distance between the pond and the estuary were evaluated for their influence on emigration. Pond distance to the estuary was measured using Google Earth satellite imagery. Ponds were grouped in two ways: (1) distance to estuary (proximal < 0.7 km; mesial 0.7 to 1 km; distal 1.0 to 1.3 km) and (2) presence of mosquito ditch connections (ditched or marsh). These groupings were based on their potential influence on emigration (Sheaves and Johnston 2008). In particular, greater distance to estuary is expected to reduce the frequency and extent of flood water inundation, and mosquito ditching increases the frequency of connectivity between ponds and the estuary. We assumed that pond distance and connectivity play a role in emigration opportunities.
Statistical Modeling: Identifying Cues for Habitat Shifts
A mixed effects logistic regression (mixed effects generalized linear regression models or GLMMs) was used to determine the environmental and biological variables that act as cues for prompting the emigration response in juvenile Tarpon. To fit the models, continuous telemetry data were summarized into daily observations represented by a binary (i.e., 0, 1) variable that was 1 on the day fish were detected within the acoustic array associated with the connecting creek system (indicating successful emigration into the estuary) and 0 otherwise. Predictor variables used to model emigration events were daily summaries of the biological (i.e., predicted SL) and environmental variables (e.g., minimum and maximum daily values) described above.
The best approximating logistic regression models relating environmental conditions to emigration success were identified using Akaike’s information criterion (AIC; Akaike 1973) with a small sample bias adjustment (AICc; Hurvich and Tsai 1989). Akaike’s information criterion is an entropy-based measure used to compare candidate models, with better predicting models producing lower AIC values (Burnham and Anderson 2002). To facilitate model comparisons, we calculated Akaike weights, which range from zero to one, where better-supported models have higher weights (Burnham and Anderson 2002). For each set of candidate models (described below), we based all inferences on the best approximating model (i.e., the model with the lowest AICc score and highest Akaike weight). Pearson correlations (ρ) were calculated for all pairwise combinations of continuous predictor variables because highly correlated variables could not be included in the same model. Here, correlated variables were identified as those with Pearson correlation coefficients >|0.6|. Once the pairs of highly correlated variables were identified, a model selection exercise was conducted for each set of variables to determine which of the predictors would be included in the final set of models. The exercise involved fitting two logistic regression models, where each model had only one of the correlated variables, comparing their relative fit using AICc, and selecting the predictor variable from the model with the lowest AICc score.
For model fitting, a candidate set of logistic regression models was developed. Tarpon that were never detected in the surrounding creek system (i.e., those that never emigrated) were excluded from the logistic regression analyses, as we were primarily focused on identifying the biological and environmental drivers of successful emigration. The analyses were conducted at two levels of organization. A local analysis was conducted for fish that successfully emigrated and had all environmental variable data present at the time of emigration. Water level loggers deployed in ponds malfunctioned during the time of emigration for some Tarpon; therefore, these fish were excluded from the local analysis but included in the regional analysis. The regional analysis consisted of all tagged fish that emigrated during the study period and used data from the NOAA tide gauge.
Results
Length, Age, and Condition
Over 1000 juvenile Tarpon were collected in four different coastal ponds: proximal marsh (n = 376; mean ± SD = 171 ± 68 mm SL), proximal ditched (n = 412; mean ± SD = 215 ± 54 mm SL), mesial marsh (n = 56; mean ± SD = 243 ± 107 mm SL), distal marsh (n = 176; mean ± SD = 315 ± 172 mm SL), and distal ditched (n = 1; SL = 358 mm SL).
Scale samples from 25 fish were retained for age estimates (mean = 280 mm SL; range = 179–388 mm SL). Approximately half (n = 13) were determined to be age 1 or greater (hereafter referred to as age 1 +) and 12 were determined to be YOY. Of the age 1 + fish, seven were > 300 mm SL. Thus, the length threshold of fish greater than 300 mm SL was chosen to represent fish that are estimated to be age 1 + and was added to length-frequency plots to help track the fate of Tarpon cohorts in each pond per year (Fig. 3).
In 2019, nearly all juveniles in the proximal marsh and proximal ditched ponds were estimated to be YOY (i.e., new recruits). The mesial pond had a third or fewer fish than in the other ponds, and all but three individuals were estimated to be YOY. In the distal marsh pond, most juveniles (91% of measured lengths) were estimated as age 1 + , suggesting these fish had remained in the pond after entering in a previous year.
In 2020, more age 1 + Tarpon were recorded in the ponds compared to the previous year. In the proximal marsh pond, nearly all were YOY (87%). However, in both the proximal ditched and distal marsh pond, 43% were age 1 + , suggesting some fish remained in the pond from a previous year.
In 2021, which followed an active tropical storm season, the numbers of Tarpon measured in the ponds were much higher than in previous years. Of the Tarpon measured, 97% were YOY. This was the case for most ponds, including the distal marsh, which had a substantial number of age 1 + fish measured in the previous 2 years.
Tarpon body condition was assessed for 137 (proximal marsh), 188 (proximal ditched), 11 (mesial marsh), and 60 (distal marsh) individuals ranging from 91 to 436 mm SL (mean ± SD = 178.7 ± 56.3). Body condition did not significantly vary among ponds for all size-class comparisons except for those at 245 mm SL (natural log 5.5) in the proximal and distal ponds (Table 1, Fig. 4). The Tarpon at 245 mm SL (natural log 5.5) had better body condition in the proximal pond than those in the distal pond (p = 0.02).
Emigration
A total of 53 Tarpon were tagged with acoustic transmitters, ranging from 232 to 850 mm SL; 17 were tagged in the proximal marsh pond, 10 in the proximal ditched pond, 10 in the mesial marsh pond, 15 in the distal marsh pond, and 1 in the distal ditched pond. There was a low tagging effort in the distal ditched pond because of the low presence of Tarpon there. This pond was the smallest and shallowest. Survivorship following tagging was confirmed using a directional hydrophone; only one fish was confirmed to have died due to tagging stress.
Of the 53 tagged Tarpon, 27 were tracked until emigration occurred and 19 were tracked in the ponds until end of battery life. Combined, this represents an 87% apparent survival in these ponds. No fish were detected moving back to the ponds once detected in the creek.
The local and regional analyses provided similar results, with both revealing the importance of high-water-level events and barometric pressure, the latter of which serves as a proxy for weather events that promote high water (e.g., passage of cold fronts, storms). In the local analysis (n = 17 fish included), maximum daily water level in each pond, regional maximum daily water level, estimated fish length, pond designation and connection type, and the minimum daily water temperature had the greatest effect on emigration (Table 2). In the regional analysis (n = 27 fish included), the global model best explained emigration and included maximum regional daily water level, minimum daily barometric pressure, estimated fish length, and pond designation and connection type (Table 3).
In 2019, 12 acoustic transmitters were deployed, including five in the proximal marsh pond and seven in the distal marsh pond (Fig. 5). Four Tarpon were detected emigrating from the proximal marsh pond to the estuary. Emigration occurred during two events when four (80%) fish emigrated during a king tide (an abnormally high tide that can lead to coastal flooding; Roman-Rivera and Ellis 2018). All six fish tagged with V9 transmitters in the distal marsh pond remained through the following wet season until battery life was reached. The single fish tagged with a V13 transmitter is not displayed but was detected for more than 2 years within distal marsh pond.
Data showing movements of Tarpon tagged in 2019, excluding the single fish tagged with a V13 acoustic transmitter in the distal marsh pond. All Tarpon tagged in the proximal marsh pond emigrated between 31 July 2019 and 02 August 2019. Tarpon ID number 4 was detected traveling to the mouth of the Peace River in Upper Charlotte Harbor. Fish tagged in the distal marsh pond remained until the battery life expired and their fate was unknown
In 2020, 20 acoustic transmitters were deployed, including five in the proximal marsh, five in the proximal ditched, four in the mesial marsh, five in the distal marsh, and one in the distal ditched pond (which was first detected in mesial ditched pond) (Fig. 6). One fish was suspected to have suffered mortality during tagging in the proximal ditched pond on 26 May 2020. One fish transitioned from the distal marsh pond into the downstream mesial marsh pond where it remained until battery life was reached. Fifteen of 19 successfully tagged (79%) fish were detected in the surrounding creek system indicating successful emigration. All study ponds with tagged fish experienced successful emigration in 2020, with emigration occurring during king tides and tropical cyclones from April through November 2020. Six fish emigrated during three separate king tide events. Two fish emigrated from the proximal marsh pond during Tropical Storm Cristobal (landfall 07 June 2020). Three fish from the mesial marsh pond, two from the proximal marsh, and one from the proximal ditched emigrated during Tropical Storm Sally (13 September 2020). An additional fish, tagged in the distal marsh pond, was suspected of also emigrating during this storm but was not detected in the creek (our criteria to establish successful emigration) and thus could not be counted in emigration estimates. During Hurricane Eta (10 November 2020), two fish emigrated from the distal marsh pond.
Data showing movements of Tarpon tagged in 2020. Sixteen of the Tarpon emigrated during various high-water events. Tarpon ID 1, 2, 3, 5, 7, and 8 emigrated during king-tide events. Tarpon 6 and 10 emigrated during Tropical Storm Cristobal on 29 May 2020. Tarpon 4, 9, 11, 12, and 13 emigrated during flooding caused by Tropical Storm Sally on 07 September 2020. Tarpon ID 15 is also suspected to have emigrated during the high water caused by Tropical Storm Sally but was not detected by the acoustic array entering the surrounding creek system. Tarpon 16 and 17 emigrated during Hurricane Eta on 10 November 2020. Tarpon 18 transitioned from the distal marsh pond to the mesial marsh pond during high-water conditions from Hurricane Eta
In 2021, 21 tags were deployed, including seven in proximal marsh pond, five in proximal ditched, six in mesial marsh, and three in the distal marsh pond (Fig. 7). Seven (33%) fish moved from the ponds into the creek system, indicating successful emigration into Charlotte Harbor. Five fish emigrated during king-tide events from the proximal marsh pond. Two fish emigrated from the proximal marsh pond in response to the flooding caused by Tropical Storm Fred (21 August 2021). No fish were confirmed to have emigrated from mesial marsh and distal marsh ponds in 2021; however, two fish were suspected of emigrating from the mesial marsh pond during Hurricane Ian (30 September 2022). These two fish were absent from pond receivers following Hurricane Ian but were not detected by the creek array (our criteria to establish successful emigration) and thus could not be counted in emigration estimates. The three fish tagged within the distal marsh pond suffered mortality during a period of severe drought from May to June 2021. A total of 25 dead, non-tagged Tarpon were recovered from the same pond during this period.
Data showing the movements of Tarpon tagged in 2021. Seven of the Tarpon emigrated during various high-water events. Tarpon 6 and 7 emigrated during flooding caused by Tropical Storm Fred on 16 August 2021. Tarpon 8, 9, 10, 11, and 12 emigrated during king-tide events on 09 October 2021 and 13 October 2021. Battery life expired before a large-scale flooding event, Hurricane Ian, occurred on 30 September 2022. Tarpon 15 and 17 were suspected to have emigrated but were not detected in the surrounding creek array. A drought from May to June 2021 caused severe evaporation in the distal marsh pond, leading to the mortality of all tagged Tarpon
While distance traveled from pond to creek system varied between the proximal, mesial, and distal designated ponds, average travel times to reach the estuary were similar; the average time elapsed between the last detection in any pond and the first detection in a creek was 3 h 42 min. Tarpon emigrated from the proximal ponds in as little as 1 h 13 min and as long as 12 h 38 min (average = 3 h 38 min). Average travel time for Tarpon to reach the creek from the mesial ponds was 4 h 22 min and ranged from 3 h 55 min to 4 h 51 min. The two fish that completed emigration from the distal marsh pond did so in 1 h 59 min and 4 h 11 min, respectively.
Post emigration, Tarpon were detected 45 km up Charlotte Harbor into major river systems. A total of 9 of the 26 successful emigrants (35%) were detected by an acoustic array located at the lower reaches of the Peace and Myakka rivers, the two major rivers that feed into northern Charlotte Harbor. One Tarpon emigrated from the distal ditched pond and completed the 45-km route from pond to creek to river in 23 h, while another emigrated from the proximal ditched pond and reached the river in 98 days.
Discussion
Understanding how early-life-stage Tarpon use ephemerally connected ponds is an important step toward conservation of juvenile Tarpon habitat. In this study, we verified that these habitats are functioning Tarpon nurseries and gained a better understanding of the environmental and physiological variables that trigger juvenile Tarpon emigration. Because the tidal creek is open to the estuary, we suggest that movements from coastal ponds to tidal creek represent the contribution of the nursery habitat to the estuarine subadult population of Tarpon. We identified the interaction between the intrinsic (e.g., position of pond within the landscape, fish size and age) and extrinsic (e.g., tropical storms, king tides) factors that affect emigration and the complexity at which they must coincide to allow for this ontogenetic shift in Tarpon life history.
Survival
Tarpon survived in the coastal ponds as evidenced by their presence in sampling events after their initial YOY recruitment period in summer and by length-frequency plots that showed the juveniles reached larger sizes representative of age 1 + . Tagged Tarpon also exhibited high survivorship in the coastal ponds. For example, we determined that emigration occurred for 27 (51%) of the tagged fish, and another 19 (36%) were found to survive in the juvenile habitat before battery life was reached (approximately 1.5 years), yielding an estimated survivorship of 87%. The high survivorship in the coastal ponds is likely due to the absence of large predatory fish. During 3 years of sampling coastal ponds with a 40-m seine, the only large-bodied fish present that could reasonably predate on juvenile Tarpon were larger Tarpon or large juvenile Snook (FWC, unpubl. data). Other large-bodied fish (> 150 mm SL) collected in abundance (> 10 individuals) were mostly herbivorous (e.g., mullets, cichlids). A low abundance of aquatic predators in coastal wetland nurseries was also found using 40-m seines in nearby Tampa Bay (Schulz et al. 2020). One source of predation not assessed is avian wildlife. Osprey Pandion haliaetus and bald eagles Haliaeetus leucocephalus have been observed feeding juvenile Snook to their nesting young on live cameras streaming online (personal observation), and Tarpon remains have been found at the base of trees near study ponds, suggesting predation from raptors (personal observation).
In some coastal ponds, rainfall events may be important to Tarpon survival. Ponds with limited connections to the estuary are susceptible to extreme variations in physical conditions (Heath et al. 1993) and can support only species adapted to low dissolved oxygen such as Tarpon (Geiger et al. 2000) and resident marsh species (e.g., mosquitofish, sailfin molly; Chervinski 1983; Nordlie 2006). In the absence of rainfall, dry-down events are possible that increase mortality through poor water conditions and heightened exposure to predation by avian predators such as Osprey and Great Blue Heron Ardea herodias (Beebe 1927; Chaverri 1994; Mol et al. 2000). In this study, drought conditions were observed in the distal marsh pond in 2021, leading to evaporation of > 95% of water and water temperatures of 36.6 C° and salinity of 67.5. This event resulted in a large-scale die-off of fish species that are known to tolerate high water temperatures and hypoxic conditions, including Florida gar Lepisosteus platyrhincus (Frick et al. 2007), blue tilapia Oreochromis aureus (Magid and Babiker 1975), and Mayan cichlid Cichlasoma urophthalmus (Schofield et al. 2007; Schofield et al. 2009). As conditions deteriorated, the pond was reduced to a 2-m diameter puddle and Tarpon were the only fish present. Eventually, the dewatering event led to mortality of 25 Tarpon, including three fish that were tagged as a part of this study. Water level was monitored daily and the remaining Tarpon were observed alive until rain events brought water to sufficient levels.
Length, Age, and Condition
Assessment of the body condition and size distribution among ponds supports the premise that juvenile Tarpon grow and emigrate from these habitats. Although differences in Tarpon body condition could occur among ponds because of a lack of mixing, it appears that the coastal ponds studied here provided adequate resources for the developing Tarpon. Acoustic telemetry confirmed that distinct groups of Tarpon do occur in isolated ponds until there is an opportunity for movement. A comparison of Tarpon body condition among coastal wetland ponds at different positions in the landscape (i.e., close to the open estuary versus far away) yielded little differences. No pond stood out as having Tarpon of low body condition.
The differences in pond proximity to the adjacent estuary did not affect Tarpon body condition but did influence emigration, which in turn affected the size and age classes present. For example, nearly all the juvenile Tarpon tagged in the proximal marsh pond survived and ultimately emigrated each year as evidenced by detection on acoustic receivers in the receiving tidal creek. As a result, very few age-1 sized fish were caught in the proximal marsh pond. The substantial carryover of Tarpon into a second year (a YOY cohort becoming age 1) coincided with years when acoustically tagged fish did not emigrate from the mesial marsh and distal marsh pond (i.e., 2019). For emigration to occur from these ponds, tropical cyclone–induced flooding is needed to allow for juvenile Tarpon to cross the > 1 km of mangrove forest to reach a tidal creek. Such flooding did occur during late 2020 (Hurricane Eta), which allowed tagged Tarpon to emigrate from the most distal pond. Successful emigration from the distal pond in 2020 is also reflected in the length-frequency plots; age-1 + Tarpon were absent in 2021, indicating a lack of carryover from the previous year. Tropical cyclone flooding also appears to be an important driver of YOY recruitment into coastal ponds. For example, the passage of Hurricane Eta allowed for substantial entry of YOY into most ponds as evidenced by high numbers of YOY in 2021.
Emigration
The primary drivers of habitat connectivity in these types of systems (e.g., coastal ponds in the Southeastern U.S., billabongs in Australia) include floods contributed by tropical cyclone activity, local rainfall, and tidal activity (Sheaves 2005; Sheaves and Johnston 2008). For example, a total of 12 Tarpon emigrated during the flood conditions produced by four distinct tropical cyclone events, accounting for 44% of all fish emigrating during this study. Declining barometric pressure and water temperature, likely associated with tropical cyclones and flooding events, were shown to increase the probability of emigration. Hurricane-strength storms have a return period of 7–8 years in southwest Florida (Neumann 1991), although the frequency of these high-intensity storms is increasing (Elsner et al. 2008). If flooding events do not occur in great enough frequency to allow for Tarpon movement across the landscape or if they are prevented by stormwater infrastructure (e.g., weirs), the risks of mortality will likely increase because of drought or poor water quality conditions (Doudoroff 1957; Mckeown 1984), especially for fish in distal ponds.
Ontogenetic movement between habitats is a risky endeavor, and there likely exists a tradeoff between the risk of movement and the gain of emigration (Sheaves 2005), which helps explain why Tarpon size was important to emigration success. Statistical models showed that smaller fish were less likely to emigrate, despite periods of high water. If ample prey are present, reducing competition for resources, small juvenile Tarpon (< 300 mm SL) may choose to remain within the nursery for an extended period, forgoing emigration opportunities until they reach a larger size. Larger Tarpon, however, may require a greater volume of flood water to allow for locomotion across the coastal landscape. During emigration, there is a risk of being trapped while in transit by the falling tide and flood conditions (Gibson 2003) and that risk increases with increased size. The difficulties of Tarpon emigration are evident by the range of transit times (1–12 h) documented from coastal ponds to the receiving tidal creek. Tarpon may need to search for the correct corridors of movement created by flooding to reach the open estuary.
The interconnectedness of tidal creeks and their chains of coastal ponds are subject to anthropogenic effects such as disrupted hydrology and mosquito ditching, potentially leading to habitat degradation. Mosquito control ditches can make year-round connections from the estuary to coastal ponds possible (Smith et al. 2007) and our study showed an increase in opportunities for emigration. Tarpon in the ditched ponds emigrated as soon as 1 day post tagging. Whether this increased connectivity is a benefit or drawback to juvenile Tarpon is still undetermined. Without the mosquito ditching, Tarpon of the same size emigrated from the proximal marsh pond every year at 1–2 years of age. However, the proximal ditched Tarpon tagged in 2021 remained for a following wet season (> 2 years of age) and did not emigrate during the life of the acoustic transmitters even though opportunities for emigration (e.g., flooding events) occurred. Tarpon tagged in ditched ponds may not have to rely on small windows of connection with the estuary for emigration because ditches provided connection throughout a normal tidal cycle. In addition, water level and prey sources could be more suitable for Tarpon to remain within the ditched ponds during periods of drought than the naturally connected coastal ponds.
Coastal wetland ponds function as one of the primary nursery habitats for Tarpon in the first year of life. As described by Breder (1944), Tarpon under 600-mm total length (TL) are known to inhabit small bodies of water that are frequently landlocked, while juveniles larger than 600-mm TL are generally found well up major rivers. This transition from primary nursery within the coastal ponds into the secondary nursery of major rivers was evident from the acoustic telemetry detections in this study, with approximately 35% of the emigrated fish moving into the lower reaches of the Peace and Myakka River systems. The lower rivers likely provide refuge from a suite of predators, as it has been shown that there are one-third fewer predators in the lower Peace River estuary than in high salinity waters near the passes (Stevens et al. 2013). The timing of Tarpon movement into the rivers also suggests that a source of prey may be important. For example, on 21 September 2020, the Peace River surpassed flood stage (3.5 m; USGS), which coincided with the first river detection from Tarpon during that year on 14 September 2020. Increased freshwater river discharge may promote movement to rivers as access to flood-plain-derived resources, such as prey and habitat, becomes available during episodic and seasonal flooding (Blewett et al. 2017).
Management Implications
The greatest threat to Florida’s Tarpon fishery is not from overfishing, since it is largely a catch-and-release practice, but rather from changes in the coastal and estuarine habitat (Wade 1973). Early life stages of Tarpon face the greatest threat from anthropogenic impacts; risk of mortality is high during their initial recruitment to nursery habitat and as the juveniles reach the adult population (Cyr 1991). These nursery habitats are located at the interface between coastal development and the open estuary. The initial risk of Tarpon being unable to access suitable nursery habitat is of great concern for the recruitment of Tarpon into the fishery (Wilson et al. 2019). If they can find suitable nursery habitat, the relatively large size at which Tarpon emigrate should lead to high survivability for the juvenile fish once they enter the estuary. A large size would leave them at risk for only the largest apex predators such as sharks and the Atlantic bottle-nosed dolphin Tursiops truncatus (Babcock 1951). Identifying and conserving the nursery habitats that remain should be a priority to sustain a future for the Tarpon fishery in Florida.
Opportunities to protect Tarpon nurseries may occur during land-use planning and the creation of stormwater infrastructure if the locations of the nurseries are known and the will exists to change the status quo to account for Tarpon life history. Coastal development can alter or destroy wetlands, which eliminates habitat for Tarpon recruiting to nursery habitats. Even where coastal wetlands still exist, the creation of stormwater drainage networks is a common practice in developed areas to mitigate the effects of flooding from rain and coastal flooding (tidal, storm surge), although sea-level rise threatens to reduce the efficacy of such infrastructure (Rosenzweig et al. 2018). Poorly placed weirs or stormwater ponds can block routes of Tarpon emigration and disrupt the hydrological connectivity of Tarpon nurseries, leading to ponds that dry up or become overly hypersaline during the dry season. However, stormwater infrastructure, if designed properly, could be of use to both human and Tarpon populations. Strategies to improve designs to account for both residential needs and ecosystem functions include landscape-scale planning to incorporate proper surface storage of flood waters, low impact development to manage stormwater flow, and possibly the managed retreat or buyout of vulnerable coastal areas (Wilby and Keenan 2012; Rosenzweig et al. 2018; Rogers et al. 2020; Gold et al. 2022). Emphasis must be placed on habitats located farther upland that function as Tarpon nurseries and might mitigate the effects of sea level rise while maintaining the right wetland to estuary connectivity. Therefore, the conservation of coastal ponds not only consists of the habitat proper but also the protection of hydrological connectivity of both the upstream freshwater resources and the downstream connectivity to the estuary.
Data Availability
All datasets generated or analyzed in the current study are available from the Florida Fish and Wildlife Conservation Commission, Fisheries-Independent Monitoring program upon request.
References
Adams, A.J., R.K. Wolfe, W.E. Pine, and B.L. Thornton. 2006. Efficacy of PIT tags and an autonomous antenna system to study the juvenile life stage of an estuarine-dependent fish. Estuaries and Coasts 29: 311–317.
Adams, A.J., A.Z. Horodysky, R.S. McBride, K. Guindon, J. Shenker, T.C. MacDonald, H.D. Harwell, R. Ward, and K. Carpenter. 2014. Global conservation status and research needs for tarpons (Megalopidae), ladyfishes (Elopidae) and bonefishes (Albulidae). Fish and Fisheries 15 (2): 280–311.
Adams, A. J. and K. J. Murchie. 2015. Recreational fisheries as conservation tools for mangrove habitats. In: K. J. Murchie and P. Daneshgar (eds) Mangroves as fish habitat. Pages 43–56 in American Fisheries Society, Symposium 83. Bethesda, MD.
Akaike, H., 1973. Information theory and an extension of the maximum likelihood principle. In: Petrov, B. and Csaki, F. (eds) Pages 267–281in Proceedings of the 2nd International Symposium on Information Theory, Akademiai Kiado, Budapest.
Azur, M.J., E.A. Stuart, C. Frangakis, and P.J. Leaf. 2011. Multiple imputation by chained equations: What is it and how does it work? International Journal of Methods in Psychiatric Research 20 (1): 40–49.
Babcock, L.L. 1951. The tarpon: A description of the fish with some hints on its capture. Reprint: Hassel Street Press 2021.
Bailey, H., and D.H. Secor. 2016. Coastal evacuations by fish during extreme weather events. Scientific Reports 6 (1): 30280.
Barbour, A.B., A.J. Adams, and K. Lorenzen. 2014. Size-based, seasonal, and multidirectional movements of an estuarine fish species in a habitat mosaic. Marine Ecology Progress Series 507: 263–276.
Basset, A., E. Barbone, M. Elliott, B.L. Li, S.E. Jorgensen, P. Lucena-Moya, I. Pardo, and D. Mouillot. 2013. A unifying approach to understanding transitional waters: Fundamental properties emerging from ecotone ecosystems. Estuarine, Coastal and Shelf Science 132: 5–16.
Beck, M.W., K.L. Heck, K.W. Able, D.L. Childers, D.B. Eggleston, B.M. Gillanders, B. Halpern, C.G. Hays, K. Hoshino, T.J. Minello, R.J. Orth, P.F. Sheridan, and M.P. Weinstein. 2001. The identification, conservation, and management of estuarine and marine nurseries for fish and invertebrates. BioScience 51: 633–641.
Beebe, W. 1927. A tarpon nursery in Haiti. Bulletin of the New York Zoological Society 30: 41–145.
Bice, C.M., J. Huisman, M.E. Kimball, M. Mallen-Cooper, B.P. Zampatti, and B.M. Gillanders. 2023. Tidal barriers and fish–impacts and remediation in the face of increasing demand for freshwater and climate change. Estuarine, Coastal and Shelf Science 289: 108376.
Blewett, D.A., R.A. Hensley, and P.W. Stevens. 2006. Feeding habits of common snook, Centropomus undecimalis, in Charlotte Harbor. Florida. Gulf and Caribbean Research 18 (1): 1–14.
Blewett, D.A., P.W. Stevens, and J. Carter. 2017. Ecological effects of river flooding on abundance and body condition of a large, euryhaline fish. Marine Ecology Progress Series 563: 211–218.
Breder, C.M., Jr. 1944. Material for study of the life history of Tarpon atlanticus. Zoologica 29: 217–252.
Brockmeyer, R.E., M. Donnelly, J.R. Rey, and D.B. Carlson. 2022. Manipulating, managing and rehabilitating mangrove-dominated wetlands along Florida’s east coast (USA): Balancing mosquito control and ecological values. Wetlands Ecology and Management 30: 987–1005.
Burnham, K. P., and D. R. Anderson, 2002. Model Selection and Multimodel Inference. A Practical Information-Theoretic Approach. Springer, New York.
Chaverri, D.C. 1993. Aspectos biométricos de una población de sábalo, Megalops atlanticus (Pisces: Megalopidae). Revista De Biología Tropical 41 (S1): 13–18.
Chaverri, D.C. 1994. Ecología básica y alimentación del sábalo Megalops atlanticus (Pisces: Megalopidae). Revista De Biología Tropical 42: 225–232.
Chervinski, J. 1983. Salinity tolerance of the mosquito fish, Gambusia affinis (Baird and Girard). Journal of Fish Biology 22 (1): 9–11.
CHNEP. 2000. Long term monitoring strategy and gaps analysis. Charlotte Harbor National Estuary Program, North Fort Myers, FL.
CHNEP. 2023. Coastal and Heartland National Estuary Partnership Wateratlas. https://chnep.wateratlas.usf.edu/watershed/geography.asp?wshedid=2&wbodyatlas=watershed. Accessed 11 Aug 2023.
Cianciotto, A.C., J.M. Shenker, A.J. Adams, J.J. Rennert, and D. Heuberger. 2019. Modifying mosquito impoundment management to enhance nursery habitat value for juvenile common snook (Centropomus undecimalis) and Atlantic tarpon (Megalops atlanticus). Environmental Biology of Fishes 102: 403–416.
University of Florida. 2023. Digital Collections, George A. Smathers Libraries. https://ufdc.ufl.edu/. Accessed 10 Feb 2023.
Crabtree, R.E., E.C. Cyr, C.D. Chaverri, W.O. McLarney, and J.M. Dean. 1997. Reproduction of tarpon, Megalops atlanticus, from Florida and Costa Rican waters and notes on their age and growth. Bulletin of Marine Science 61 (2): 271–285.
Cyr, E. C. 1991. Aspects of the life history of the tarpon, Megalops atlanticus, from south Florida. Ph.D. Dissertation, University of South Carolina, Columbia. 138 p.
Dahl, T. E. 2006. Florida’s wetlands: An update on status and trends, 1985 to 1996. U.S. Fish and Wildlife Service, Washington, D.C.
Dahlgren, C.P., G.T. Kellison, A.J. Adams, B.M. Gillanders, M.S. Kendall, C.A. Layman, J.A. Ley, I. Nagelkerken, and J.E. Serafy. 2006. Marine nurseries and effective juvenile habitats: Concepts and applications. Marine Ecology Progress Series 312: 291–295.
Doudoroff, P. 1957. Water quality requirements of fishes and effects of toxic substances. In: Brown, M. E. (ed) Pages 415–417 in The Physiology of Fishes, Vol. II, Behavior. Academic Press, London.
Elmo, G.M., D.P. Crane, M.E. Kimball, K.L. Williams, and P.W. Stevens. 2021. Validity of daily and annual age estimation and back-calculation methods for early life stages of a subtropical-tropical species, the tarpon (Megalops atlanticus). Fisheries Research 243: 106057.
Elsner, J.B., J.P. Kossin, and T.H. Jagger. 2008. The increasing intensity of the strongest tropical cyclones. Nature 455: 92–95.
FDEP. 2023. Florida State Parks. https://www.floridastateparks.org/parks-and-trails/charlotte-harbor-preserve-state-park. Accessed 10 Feb 2023.
Fedler, T., 2011. The economic impact of recreational tarpon fishing in the Caloosahatchee River and Charlotte Harbor region of Florida. The Everglades Foundation, Palmetto Bay, FL, p. 26.
Fernandes, A.F., E.M. Turra, É.R. de Alvarenga, T.L. Passafaro, F.B. Lopes, G.F. Alves, V. Singh, and G.J. Rosa. 2020. Deep Learning image segmentation for extraction of fish body measurements and prediction of body weight and carcass traits in Nile tilapia. Computers and Electronics in Agriculture 170: 105274.
Frick, N.T., S.J. Bystriansky, and S.J. Ballantyne. 2007. The metabolic organization of a primitive air-breathing fish, the Florida gar (Lepisosteus platyrhincus). Journal of Experimental Zoology Part A: Ecological Genetics and Physiology 307 (1): 7–17.
Geiger, S.P., J.J. Torres, and R.E. Crabtree. 2000. Air breathing and gill ventilation frequencies in juvenile tarpon, Megalops atlanticus: Responses to changes in dissolved oxygen, temperature, hydrogen sulfide, and pH. Environmental Biology of Fishes 59: 181–190.
Gibson, R. N. 2003. Go with the flow: Tidal migration in marine animals. In: Migrations and Dispersal of Marine Organisms. Pages 153–161 in Proceedings of the 37th European Marine Biology Symposium, Reykjavík, Iceland, 5–9 August 2002. Springer, Netherlands.
Gillanders, B.M. 2005. Using elemental chemistry of fish otoliths to determine connectivity between estuarine and coastal habitats. Estuarine, Coastal and Shelf Science 64 (1): 47–57.
Gold, A.C., C.M. Brown, S.P. Thompson, and S.P. and M. F. Piehler. 2022. Inundation of stormwater infrastructure is common and increases risk of flooding in coastal urban areas along the US Atlantic coast. Earth’s Future 10 (3): e2021002139.
Graham, P.M., J.S. Franks, E.J. Anderson, R.T. Leaf, and J.D. Tilley. 2021. Age and growth of early-life-stage Atlantic tarpon (Megalops atlanticus) from the northcentral Gulf of Mexico. Journal of Fish Biology 99 (4): 1190–1200.
Halpin, P.M. 2000. Habitat use by an intertidal salt-marsh fish: Trade-offs between predation and growth. Marine Ecology Progress Series 198: 203–214.
Hammett, K. M. 1990. Land use, water use, streamflow characteristics, and water-quality characteristics of the Charlotte Harbor inflow area, Florida. Hydrologic assessment of the Charlotte Harbor estuarine system (USA). Water Supply Paper 2359. U.S. Geological Survey, Washington, DC. https://doi.org/10.3133/wsp2359A
Heath, A.G., B.J. Turner, and W.P. Davis. 1993. Temperature preferences and tolerances of three fish species inhabiting hyperthermal ponds on mangrove islands. Hydrobiologia 259: 47–55.
Heupel, M.R., C.A. Simpfendorfer, and R.E. Hueter. 2003. Running before the storm: Blacktip sharks respond to falling barometric pressure associated with Tropical Storm Gabrielle. Journal of Fish Biology 63 (5): 1357–1363.
Hurvich, C.M., and C.L. Tsai. 1989. Regression and time series model selection in small samples. Biometrika 76: 297–307.
Idelberger, C.F., C.J. Stafford, and S.E. Erickson. 2011. Distribution and abundance of introduced fishes in Florida’s Charlotte Harbor estuary. Gulf and Caribbean Research 23 (1): 13–22.
Jones, G.P. 1986. Food availability affects growth in a coral reef fish. Oecologia 70: 136–139.
Jones, R.E., R.J. Petrell, and D. Pauly. 1999. Using modified length–weight relationships to assess the condition of fish. Aquacultural Engineering 20 (4): 261–276.
Lenth, R., H. Singmann, J. Love, P. Buerkner, and M. Herve. 2018. emmeans: Estimated marginal means, aka least-squares means (R package, Version 1.4) [Computer software].
Lindall, W. N., J. R. Hall, W. A. Fable, and L. A. Collins. 1973. A survey of fishes and commercial invertebrates of the shore and estuarine zone between Cape Romano and Cape Sable, Florida. Marine Fisheries Service, U.S. Department of Commerce, Springfield, VA. (PB-235 215 NTIS)
Litvin, S.Y., M.P. Weinstein, M. Sheaves, and I. Nagelkerken. 2018. What makes nearshore habitats nurseries for nekton? An emerging view of the nursery role hypothesis. Estuaries and Coasts 41: 1539–1550.
Mace, M.M., M.E. Kimball, and E.R. Haffey. 2018. Recruitment and habitat use of early life stage tarpon (Megalops atlanticus) in South Carolina estuaries. Estuaries and Coasts 41: 841–854.
Magid, A.M., and M.M. Babiker. 1975. Oxygen consumption and respiratory behavior of three Nile fishes. Hydrobiologia 46: 359–367.
Matthews, W.J., and E. Marsh-Matthews. 2003. Effects of drought on fish across axes of space, time and ecological complexity. Freshwater Biology 48 (7): 1232–1253.
McKeown, B.A. 1984. Fish migration. London: Croom Helm.
Mol, J.H., D. Resida, J.S. Ramlal, and C.R. Becker. 2000. Effects of El Nino-related drought on freshwater and brackish-water fishes in Suriname, South America. Environmental Biology of Fishes 59: 429–440.
Nagelkerken, I., M. Sheaves, R. Baker, and R.M. Connolly. 2015. The seascape nursery: A novel spatial approach to identify and manage nurseries for coastal marine fauna. Fish and Fisheries 16 (2): 362–371.
Neumann, C. J., 1991. The National Hurricane Center Risk Analysis Program (HURISK). NOAA Technical Memorandum NWS NHC 38, National Oceanic and Atmospheric Administration (NOAA), Washington, DC.
NOAA Center for Operational Oceanographic Products and Services, 2023b. Tides and Currents. https://tidesandcurrents.noaa.gov/waterlevels.html?id=8725520. Accessed 10 Feb 2023.
NOAA Center for Operational Oceanographic Products and Services, 2023a. https://tidesandcurrents.noaa.gov/. Accessed 10 Feb 2023.
Nordlie, F.G. 2006. Physicochemical environments and tolerances of cyprinodontoid fishes found in estuaries and salt marshes of eastern North America. Reviews in Fish Biology and Fisheries 16: 51–106.
Ogle, D. 2013. fishR Vignette: Length-weight relationships. Fisheries Research 99: 244–247.
Peters, K.M., R.E. Matheson Jr., and R.G. Taylor. 1998. Reproduction and early life history of common snook, Centropomus undecimalis (Bloch). Florida. Bulletin of Marine Science 61 (2): 509–529.
Pierce, R.H., D.L. Wetzel, and E.D. Estevez. 2004. Charlotte Harbor initiative: Assessing the ecological health of Southwest Florida’s Charlotte Harbor estuary. Ecotoxicology 13: 275–284.
Poléo, A.B., S.A. Osxnevad, K. Össtbye, E. Heibo, R.A. Andersen, and L.A. Vøllestad. 1995. Body morphology of crucian carp Carassius carassius in lakes with or without piscivorous fish. Ecography 18 (3): 225–229.
Poulakis, G.R., D.A. Blewett, and M.E. Mitchell. 2003. The effects of season and proximity to fringing mangroves on seagrass-associated fish communities in Charlotte Harbor, Floridar. Gulf of Mexico Science 21 (2): 171–184.
Rappaport, J., and J.D. Sachs. 2003. The United States as a coastal nation. Journal of Economic Growth 8: 5–46.
Rehage, J.S., R.E. Boucek, R.O. Santos, J.A. Massie, N.M. Viadero, and R.J. Rezek. 2022. Untangling flow-ecology relationships: Effects of seasonal stage variation on common snook aggregation and movement rates in the everglades. Estuaries and Coasts 45 (7): 2059–2069.
Rickards, W.L. 1968. Ecology and growth of juvenile tarpon, Megalops atlanticus, in a Georgia salt marsh. Bulletin of Marine Science 18 (1): 220–239.
Robins, C.R. 1977. The tarpon—Unusual biology and man’s impact determine its future. Marine Recreational Fisheries 2: 105–112.
Rogers, B.C., N. Bertram, B. Gersonius, A. Gunn, R. Löwe, C. Murphy, R. Pasman, M. Radhakrishnan, C. Urich, T.H.F. Wong, and K. Arnbjerg-Nielsen. 2020. An interdisciplinary and catchment approach to enhancing urban flood resilience: A Melbourne case. Philosophical Transactions of the Royal Society A 378 (2168): 20190201.
Román-Rivera, M.A., and J.T. Ellis. 2018. The king tide conundrum. Journal of Coastal Research 34 (4): 76–771.
Rosenzweig, B.R., L. McPhillips, H. Chang, C. Cheng, C. Welty, M. Matsler, D. Iwaniec, and C.I. Davidson. 2018. Pluvial flood risk and opportunities for resilience. Wiley Interdisciplinary Reviews: Water 5 (6): e1302.
Scharer, R.M., P.W. Stevens, C.P. Shea, and G.R. Poulakis. 2017. All nurseries are not created equal: Large-scale habitat use patterns in two smalltooth sawfish nurseries. Endangered Species Research 34: 473–492.
Schofield, P.J., W.F. Loftus, and M.E. Brown. 2007. Hypoxia tolerance of two centrarchid sunfishes and an introduced cichlid from karstic Everglades wetlands of southern Florida, USA. Journal of Fish Biology 71: 87–99.
Schofield, P.J., W.F. Loftus, and J.A. Fontaine. 2009. Salinity effects on behavioural response to hypoxia in the non-native Mayan cichlid Cichlasoma urophthalmus from Florida Everglades wetlands. Journal of Fish Biology 74 (6): 1245–1258.
Schulz, K., P.W. Stevens, J.E. Hill, A.A. Trotter, J.L. Ritch, K.L. Williams, J.T. Patterson, and Q.M. Tuckett. 2020. Coastal wetland restoration improves habitat for juvenile sportfish in Tampa Bay, Florida, USA. Restoration Ecology 28 (5): 1283–1295.
Seymour, R.S., N.C. Wegner, and J.B. Graham. 2008. Body size and the air-breathing organ of the Atlantic tarpon Megalops atlanticus. Comparative Biochemistry and Physiology Part a: Molecular & Integrative Physiology 150 (3): 282–287.
Sheaves, M. 2005. Nature and consequences of biological connectivity in mangrove systems. Marine Ecology Progress Series 302: 293–305.
Sheaves, M., and R. Johnston. 2008. Influence of marine and freshwater connectivity on the dynamics of subtropical estuarine wetland fish metapopulations. Marine Ecology Progress Series 357: 225–243.
Shenker, J.M., E. Cowie-Mojica, R.E. Crabtree, H.M. Patterson, C. Stevens, and K. Yakubik. 2002. Recruitment of Tarpon (Megalops atlanticus) leptocephali into the Indian River Lagoon, Florida. Contributions in Marine Science 35: 55–69.
Smith, T.J., III., G. Tiling, and P.S. Leasure. 2007. Restoring coastal wetlands that were ditched for mosquito control: A preliminary assessment of hydro-leveling as a restoration technique. Journal of Coastal Conservation 11 (1): 67–74.
Sogard, S.M., and K.W. Able. 1994. Diel variation in immigration of fishes and decapod crustaceans to artificial seagrass habitat. Estuaries 17: 622–630.
Stevens, P.W., S.L. Fox, and C.L. Montague. 2006. The interplay between mangroves and saltmarshes at the transition between temperate and subtropical climate in Florida. Wetlands Ecology and Management 14: 435–444.
Stevens, P.W., M.F.D. Greenwood, and D.A. Blewett. 2013. Fish assemblages in the oligohaline stretch of a southwest Florida river during periods of extreme freshwater inflow variation. Transactions of the American Fisheries Society 142: 1644–1658.
Taylor, J.L. 1974. The Charlotte Harbor estuarine system. Florida Scientist 37: 205–216.
Turner, R.E., N.N. Rabalais, B. Fry, N. Atilla, C.S. Milan, J.M. Lee, C. Normandeau, T.A. Oswald, E.M. Swenson, and D.A. Tomasko. 2006. Paleo-indicators and water quality change in the Charlotte Harbor Estuary (Florida). Limnology and Oceanography 51: 518–533.
Udyawer, V., A. Chin, D.M. Knip, C.A. Simpfendorfer, M.R. Heupel, and M.R. 2013. Variable response of coastal sharks to severe tropical storms: Environmental cues and changes in space use. Marine Ecology Progress Series 480: 171–183.
Univeristy of Florida. George A. Smathers Libraries. 2023. https://ufdc.ufl.edu/collections/aerials.
USGS. Peace River at SR 70 at Arcadia, FL. 2023. https://waterdata.usgs.gov/monitoring-location/02296750/#parameterCode=00065&period=P7D. Accessed 10 Feb 2023.
Van Buuren, S., and K. Groothuis-Oudshoorn. 2011. mice: Multivariate imputation by chained equations in R. Journal of Statistical Software 45: 1–67.
Wade, R.A. 1962. The biology of the tarpon, Megalops atlanticus, and the ox-eye, Megalops cyprinoides, with emphasis on larval development. Bulletin of Marine Science 12 (4): 545–622.
Wade, R. A., and C. R. Robins. 1973. The tarpon and its sport fishery: A review of our knowledge and outline of the problem. International Game Fish Conference 15: 16–27)
Wilby, R.L., and R. Keenan. 2012. Adapting to flood risk under climate change. Progress in Physical Geography 36 (3): 348–378.
Wilson, J.K., A.J. Adams, and R.N. Ahrens. 2019. Atlantic tarpon (Megalops atlanticus) nursery habitats: Evaluation of habitat quality and broad-scale habitat identification. Environmental Biology of Fishes 102 (2): 383–402.
Acknowledgements
Field sampling and data analysis was supported by Charlotte County RESTORE Act Direct Component award 4RDCGR040058-01-02 and with funds collected from the State of Florida Saltwater Fishing License sales, U.S. Department of the Interior, U.S. Fish and Wildlife Service, and Federal Aid for Sport Fish Restoration to the Florida Fish and Wildlife Commission. We thank the staff of the Charlotte Harbor Field Laboratory for their dedicated sampling. Richard Paperno and Alexis Trotter provided comments on early versions of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Pedro Morais
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bunting, M.S., Stevens, P.W., Blewett, D.A. et al. Emigration of Juvenile Tarpon Megalops atlanticus from Ephemerally Connected Coastal Ponds. Estuaries and Coasts (2024). https://doi.org/10.1007/s12237-024-01424-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12237-024-01424-w