Abstract
Bioproduction of short chain carboxylic acids (SCCAs) and medium chain carboxylic acids (MCCAs) has emerged as an alternative strategy to upgrade low-value organic waste and reduce fossil fuels requirement. Using synthesis gas (syngas) to generate SCCAs and MCCAs by microbial communities would be an option to address part of the current energy challenge. Syngas fermentation offers a pathway for the sustainable synthesis of fuels and chemicals with advantages over catalytic syngas conversion. In the same way, chain elongation is an anaerobic microbial process driven by ethanol, carbohydrates, and SCCAs (e.g., acetate, lactate) to obtain high-value MCCAs (e.g., caproate, valerate). Because these technologies use organic wastes as feedstocks, mixed microbial communities are often considered biocatalysts. However, the management of microbial communities is the biggest bottleneck for efficient and simultaneous production of SCCAs and MCCAs. Understanding and steering these microbiomes is critical to optimize bioprocess performance. Therefore, this review discusses the metabolic pathways of both syngas fermentation and chain elongation. Also, to examine the overall performance of microbial communities involved in syngas fermentation and chain elongation, the influence of reactor parameters on the growth and metabolic activity of the key microorganisms is presented. The experimental strategies for simultaneous syngas fermentation and chain elongation processes are also presented and discussed. Finally, the use of multi-omics to better understand both syngas fermentation and chain elongation processes is discussed to steer these bioproduction processes towards full-scale applications.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Use of oil and other fossil resources for transportation and service chemicals is deeply engrained today. However, their handling is unsustainable as the nature of fossil fuels relies on finite reserves, and herein they have negative environmental impacts, such as the release of carbon dioxide (CO2) and other pollutants (e.g., sulfur and nitrogen oxides) during combustion [1]. Thus, we need alternative processes to produce energy and chemicals. Examples of cleaner and effective processes are those based on biotechnological applications such as anaerobic digestion, dark fermentation, bioethanol, and biodiesel production. Nonetheless, the transition to a sustainable energy supply is not straightforward and will take considerable time. Thus, short-term solutions can lessen the environmental impact of fossil fuels.
Waste biomass valorization is being developed to produce high-value fuels or chemicals to reduce fossil fuels global consumption. Anaerobic digestion and dark fermentation are well-documented technologies for valorizing biowastes through fermentation of easily degradable carbohydrate-rich substrates, such as corn starch and sugar cane [2]. Alternatively, fermentable sugars can be obtained through acid or enzymatic hydrolysis of insoluble cellulosic biomass [3]. However, most biomass sources like straw or bagasse contain a great amount of material that cannot be efficiently converted by fermentative microorganisms. A feasible alternative is to gasify the organic biomass and use the synthesis gas (or syngas) as a feedstock to produce ethanol, acetate, and other valuable compounds.
Commonly, in biomass gasification, the lignocellulosic structure of biomass is thermally cracked into carbon monoxide (CO), hydrogen (H2), and carbon dioxide (CO2) as the main constituents of syngas and minor amounts of methane (CH4), water steam, and trace gases [4,5,6]. The most common process to produce syngas is coal gasification, which uses steam and oxygen (from air) at high temperatures, a process that produces large amounts of CO2. One more environmentally friendly way to create syngas, called methane dry reforming, involves getting two potent greenhouse gases to react, CH4 (e.g., from natural gas) and CO2. But that process is not widely used at industrial scales, partly because it requires temperatures of at least 700 °C to initiate the chemical reaction [7].
Traditionally, the conversion of syngas to organic molecules is executed by the Fischer-Tropsch metal catalysts synthesis. However, this process is highly exothermic, low energy efficient, and presents internal diffusion limitations [8]. Alternatively, syngas fermentation is a potential microbial pathway in which anaerobic microorganisms mediate the biocatalytic conversion of syngas components to various useful biochemicals and biofuels (e.g., H2, ethanol, acetic acid). Through metabolic pathways, such as Wood-Ljungdahl or carboxydrotrophic acetogenesis, anaerobic bacteria utilize the CO and/or CO2 as the carbon source and H2 as the energy source [9]. Microbial communities capable of performing syngas fermentation can adapt to different proportions of the gas mixture and operate at mesophilic temperatures, and overall, this process could be a carbon-neutral process. Bacteria belonging to the genera Clostridium, Acetobacterium, Rhodospirillum, Rubrivivax, and Citrobacter have been widely reported as efficient syngas fermenters [10, 11]. However, inefficient gas-liquid mass transfer of the gases due to their low solubility, the low growth rate of fermenters, and the toxicity of CO are the most common challenges in syngas fermentation [12].
Interestingly, the acetate and ethanol obtained from the syngas fermentation can be further transformed by microorganisms in a process known as microbial chain elongation (CE), obtaining high value biochemicals classified in short chain carboxylic acids (SCCAs, C1 to C4), such as propionate (C3) and butyrate (C4), and medium chain carboxylic acids (MCCAs, C5 to C12), such as valerate (C5) and caproate (C6). Carboxylate CE occurs via cyclical pathways such as the reverse β-oxidation and analogous, for example, the Wood-Ljungdahl pathway coupled to acetyl-CoA reduction [13,14,15]. Among the microorganisms capable to elongate carboxylates are bacteria of the genera Clostridium, Veillonella, Eubacterium, Megasphaera, Rhodospirillum, and Caproiciproducens [16]. The production of MCCAs by single strain cultures has been intensively investigated with relevant results [15, 17, 18]. Nonetheless, mixed-culture fermentation could be more suitable to produce MCCAs from organic waste due to its resilience. However, the MCCA yields could be negatively affected by the competitive bioreactions existing in mixed-cultures, such as the acetoclastic methanogenesis or propionic production [19]. Therefore, understanding the cooperative and competitive relationships among key functional microorganisms is of great significance to further increase the production rate of MCCAs.
Although ethanol and acetate production from syngas is a feasible process, inducing CE in the same consortium can increase the value of the process. This is because MCCAs have a higher market value than other short-chain compounds. Therefore, this review is focused on key microorganisms to better understand syngas fermentation, CE, and the relationship of both processes. This review aims to (1) review the metabolic pathway of both syngas fermentation and CE; (2) summarize representative microbial communities for both syngas fermentation and CE; (3) analyze the simultaneous syngas fermentation and CE; and (4) discuss the use of advanced molecular tools to better understand both syngas fermentation and CE processes.
Basics of Syngas Fermentation
Syngas Generation
Obtaining syngas from biomass involves a series of thermal and chemical processes to convert the organic matter in biomass into a mixture of H2 and CO, known as syngas. The primary steps in this process typically include biomass feedstock preparation, drying, pyrolysis, and gasification. First, the biomass feedstock, which can range from wood chips and agricultural residues to municipal solid waste, is collected and prepared by removing impurities and reducing its moisture content [7, 20]. Then, the prepared biomass is subjected to drying to reduce further moisture levels, which is essential for efficient conversion. Next, pyrolysis is employed to heat the biomass in the absence of oxygen, causing it to release volatile components in the form of gases, liquids, and char. Finally, gasification takes place, where the remaining char reacts with a controlled amount of oxygen or steam to generate syngas (containing H2, CO, and CO2, principally), which can be utilized for various applications, including power generation, fuel production, and chemical synthesis [21]. The composition and quality of the syngas can be adjusted by optimizing the gasification process parameters and feedstock selection.
Syngas Fermentation
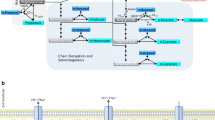
The main syngas fermenting microorganisms are chemolithoautotrophic bacteria that under strict anaerobic conditions convert carbon-based gas streams to SSCAs (acetate, principally) and alcohols [22]. These microorganisms are divided in two groups based on their metabolism. The first is the acetogenic bacteria, which require H2 and CO2 for acetate generation through the Wood-Ljungdahl pathway [23]. The second is carboxydotrophic microorganisms, which utilize the CO to produce CO2 and H2 by carboxydotrophic hydrogenesis, or by acetate generation through carboxydotrophic acetogenesis (Fig. 1) [4].
In the Wood-Ljungdahl pathway, microorganisms use CO and/or CO2 as carbon source and H2 as energy source, while the carboxydotrophic microorganisms utilize only CO as carbon and energy source. Then, in the acetogenesis stage, the intermediate acetyl-CoA is produced through reduction reactions. Finally, during solventogenesis, products such as ethanol (Eqs. (1) and (2)) or acetate (Eqs. (3) and (4)) are generated [26]. Since ethanol production generates less energy than acetate, microorganisms use solventogenesis as a passive step [6].
Microorganisms Performing Syngas Fermentation
Acetogenesis is a process conducted by various microorganisms, including bacteria, archaea, and even eukaryotes. Nevertheless, acetogenic bacteria have been documented as the predominant group during syngas fermentation, owing to their ability to adapt their metabolism based on environmental conditions and substrate availability [25]. Overall, the source of acetogenic bacteria can be obtained from mixed cultures or single strain cultures [27,28,29].
Single Strain Cultures
In single strain cultures syngas fermentation, the medium is sterilized before inoculation with one type of microorganism. The most commonly studied microorganisms in single strain cultures syngas fermentations are bacteria belonging to genera, such as Clostridium, Alkalibaculum, and Butyribacterium [27]. The obtained products depend on the selected bacteria and experimental conditions; nevertheless, some commonly reported products from single strain culture fermentation include ethanol, butanol, hexanol, acetic acid, butyric acid, and hexanoic acid [25].
In this sense, one of the primary acetogens commonly documented was Clostridium ljungdahlii, an anaerobic strain isolated from an enrichment inoculated with chicken yard waste at an initial pH of 5.0 and incubated at 37 °C under an atmosphere of synthesis gas (CO, H2, CO2, CH4, 73:15:10:2) [30]. This bacterium demonstrates the capacity to convert syngas and/or simple carbohydrates into ethanol through the Wood-Ljungdahl pathway [31]. From the same genus, C. autoethanogenum also can transform syngas into ethanol and acetate. For example, Xu et al. [32] conducted batch experiments to investigate the bioconversion of CO and syngas by C. autoethanogenum. Their study reported a maximum ethanol concentration of 75 mM when 100% CO was used as the substrate. In contrast, when biomass-generated syngas was employed (with a composition of 36.2% CO, 23.0% H2, 15.4% CO2, and 11.3% N2), the ethanol production was only 5.4 mM. This lower ethanol yield may be attributed to the presence of O2 and C2H2. Other strains that can metabolize CO to ethanol, butanol, or even hexanol are Butyribacterium methylotrophicum and Clostridium carboxidivorans [25]. Overall, in single culture strains, the productivity of the culture depends on the type of microorganism, gas mixture, medium composition, and fermentation conditions (reactor type, temperature, pH, pressure) [27]. However, most of the metabolites produced by single strain cultures contain only two carbons, and they are characterized by low cellular density, limited substrate utilization, susceptibility to contamination, lack of robustness, high operational costs, and strict growth conditions [6].
Mixed Cultures
The inoculum for mixed cultures can originate from wastewater anaerobic sludge or similar [22, 33]. Compared with single strain cultures, mixed cultures are robust systems, capable of metabolizing different syngas mixtures, and more tolerant to environmental stress and changes [4, 6, 26, 34]. Table 1 includes studies of mixed cultures for syngas fermentation. Considering that H2 and CO2 present low water solubility (Henry’s constants of 7.7 × 10–6 and 3.4 × 10–4 mol/m3 Pa, respectively), mass transfer represents the main bottleneck in syngas fermentation [40]. Additionally, autotrophic bacteria are slow growers with duplication times of 15 to 21 h in the case of H2/CO2/CO fermentation by Clostridium genus [20]. However, using biofilms for syngas fermentation overcomes the problems mentioned before. For instance, the fixed biomass decouples the hydraulic retention time from the cellular retention time, allowing slow-growing bacteria to develop [24]. Moreover, using a biofilm attached to a hollow fiber membrane enhances the delivery of syngas to the microbiome, and thus improves fermentation performance [6, 40].
In 2018, Liu et al. [41] investigated the effects of pH and temperature on the conversion efficiency, product, and microbial community composition during batch syngas fermentation. The study reached acetate and ethanol concentrations up to 56.6 mM and 67.4 mM, both at pH 9 under mesophilic conditions with a high relative abundance of the genera Clostridium and Acetobacterium, and reporting other several acetogens. Alternatively, the use of acetogens-enriched sludge as inoculum improved the substrate utilization rate and acetate percentage in SCCAs [33]. In such batch experiments, the acetate yield increased by 1.77 times with CO2 sparge in the headspace of the reactor; also, the genera Clostridium, Eubacterium, Methylophilus, and Nannocystis were detected in the culture. Moreover, the addition of sulfate in syngas mixed cultures was explored in batch experiments. In presence of 0.9 g sulfate/L, the maximum acetate concentration achieved was 75 mM at a H2 feeding of 21.4 mM/d. In such study, Acetobacterium and Desulfovibrio were the dominant microorganisms in the consortia, suggesting co-metabolism between acetogens and sulfate-reducing bacteria [34]. Although mixed cultures have shown promising results, the product yield and specificity compared to single strain cultures remain relatively low [27]. Therefore, it is imperative to dedicate research efforts to selecting, managing, and controlling of mixed cultures to improve syngas fermentation.
Basics of Production of Short and Medium Chain Organic Acids
In the anaerobic digestion processes, mixed microbial communities can turn organic matter into H2, CO2, and SCCAs. The SCCAs are carboxylates of 1 to 4 carbon atoms (C1–C4) with relativity low market value. Moreover, the SCCAs are completely miscible in the liquid phase of fermentation, making the extraction processes expensive and complex [42]. SCCAs have also been used as a substrate for methane generation in the same fermenter of anaerobic digestion; nevertheless, methane has low added value [43].
Recently, CE has emerged as a technology capable of extending SCCAs carbon structure to MCCAs under anaerobic conditions [44]. Furthermore, CE is an efficient fermentation with MCCAs selectivity above 80%. MCCAs are less soluble than their precursors because they contained from 5 to 12 carbon atoms in their structure. Thus, MCCAs high-energy content and simpler extraction make them cost-effective bioproducts that are employed in fragrances, rubbers, pharmaceuticals, food additives, and antimicrobials [45, 46]. Additionally, among MCCAs generated by CE, caproate (C6) presents a higher efficiency of conversion and an economic value of 2000 to 3000 USD/ton [47].
Two circular reactions are responsible for the microbial CE: the fatty acid biosynthesis and the reverse β-oxidation pathway (Fig. 2). The reverse β-oxidation is the most widely studied pathway for CE, where the overall process adds an acetyl-CoA molecule to a SCCA electron acceptor as shown in Fig. 2. The first required environmental condition is the presence of energy-rich reduced compounds (e.g., ethanol, methanol, lactate, H2) that oxidizes to acetyl-CoA by NAD+ and the reduction of ferredoxin by NADH [50]. Furthermore, and before the fatty acid biosynthesis pathway starts, the acetyl-CoA converts to malonyl-ACP by spending 1 ATP molecule and forming malonyl-ACP. Once the fatty acid biosynthesis cycle begins, the process is like the reverse β-oxidation and is catalyzed by a series of analogous reductases (Fig. 2).
In both cases, SCCAs are reduced by an electron donor, commonly ethanol or even H2, and two carbons are added to the SCCAs per cycle [48]. For instance, acetate (C2) elongates to butyrate (C4) and then to caproate (C6), while propionate (C3) elongates to valerate (C5) and to heptoic acid (C7) [13]. Generally, a high H2 partial pressure or a reductive environment is required to prevent oxidation of the generated MCCAs [43]. The additional step before fatty acid biosynthesis is longer and less efficient than the reverse β-oxidation pathway [48]. However, CE carried out by a mixed microbial community made fatty acid biosynthesis more active than reverse β-oxidation [48].
Microorganisms Performing Chain Elongation
CE can occur by single strain cultures, co-culture, or mixed culture, where wild-type strains from the genera Clostridium, Caproiciproducens, Megasphaera, Eubacterium, and Ruminococcaceae have been isolated and characterized [49]. Specifically, Clostridium kluyveri is the most accepted model microorganism that carries out CE from ethanol and acetate through the reverse β-oxidation pathway [51]. Furthermore, Eubacterium pyruvativorans is an analogous model whose metabolic strategy resembles C. kluyveri [52].
Single Strain Cultures
San-Valero et al. [53] reported the influence of electron acceptors on caproate production by C. kluyveri, obtaining a significant increase in caproate concentration when adding a mixture of acetate/butyrate and ethanol as the electron donor. Similarly, a high concentration of caproate (C6) has been achieved by the strain CPB6 of the family Ruminococcaceae with lactate as an electron donor [54]. In pure anaerobic culture, Megasphaera elsdenii produced a mixture of carboxylates from C2 to C6, using carbohydrates as substrate and lactate or butyrate as electron acceptors. Additionally, it has been reported that M. elsdenii can produce until C7-C8 MCCAs in presence of fructose and C2-C4 SCCAs [55]. Strains for the genus Caproiciproducens have been isolated from mixed cultures of CE and can convert different types of saccharides to acetate, butyrate, caproate, and H2/CO2 [56, 57].
Strain Co-cultures
Strain co-cultures are designed to take advantage of the interactions between two strains without other microorganisms that could negatively impact the performance of the process. In 2016, Diender et al. [58] reported the establishment of C. autoethanogenum and C. kluyveri co-culture, capable of converting CO and syngas to a mixture of butyrate (8.5 mM/d) and caproate (2.6 mM/d) and their respective alcohols [58]. In the same study, the addition of acetate stimulated the production rates. Similarly, Fernández-Blanco et al. [15] studied the optimal operational conditions and the MCCAs and alcohols resulting from a co-culture of C. aceticum and C. kluyveri in fed syngas bioreactors with ethanol as an exogenous electron agent. This work showed maximum concentrations of butyrate and caproate of 80 and 70.6 mM, respectively, while considerable amounts of butanol were produced from the SCCAs reduction by C. aceticum.
Mixed Cultures
Mixed culture fermentation offers an advantage in CE because its high microbial diversity improves the process stability and resilience under changing conditions. Also, the process does not require sterile conditions, making it suitable for organic waste or gas utilization as substrates [51]. Table 2 comprises the main reports related to CE by mixed cultures.
In 2015, Weimer et al. [66] employed a ruminal mixed culture that was bioaugmented with a rumen-derived strain of C. kluyveri for cellulosic biomass fermentation. In presence of ethanol as an electron donor, the major generated MCCAs included valerate (C5) and caproate (C6) over a 48–72-h time period in batch reactors, with a caproate concentration of 52.5 mM. In the same way, Leng et al. [62] used a mixed microbial community in a semi-continuous reactor fed with a stoichiometric ratio of 4:3:1 of glycerol-ethanol-acetate for CE. The study reached a caproate production of 2.95 mM C/d where the fermentation and CE were mainly facilitated by a microbial community of Eubacterium limosum, C. kluyveri, and Massilibacterium senegalese [62]. For the full conversion of butyrate from food waste fermentation, the liquid phase of a mixed culture reactor was recycled as previously reported [44]. This work reported the enhanced production by 4.1 times of MCCAs by elongating butyrate to caproate and valerate to heptanoate, with a high abundance of the strain Clostridium sensu stricto 12.
Mixed cultures can include syngas fermenting bacteria, which use CO2 and CO as a carbon source and H2 as an electron donor for acetate generation. Then, the acetate is used as an electron acceptor by the CE microorganisms [49]. The first study that demonstrated the in situ production of MCCAs from H2 and CO2 in a hollow-fiber membrane biofilm reactor by mixed microbial culture achieved a caproate production rate of 31.4 mM C/d [36]. In the same report, the microbial community of the biofilm was dominated by C. ljungdahlii and C. kluyveri. Furthermore, Jourdin et al. [60] showed a biofilm-driven production of acetate, butyrate, and caproate from CO2 and carbon-felt electrodes as the electron donor, with a caproate productivity of 8.2 mM/d. Recently, a membrane biofilm reactor with an inorganic carbon source and H2 fed through hollow membranes was employed and achieved up to C8 MCCAs production (28.1 mM C/m2-d). The biofilm microbial community presented the well-known acetogen Acetobacterium and phylotypes related to CE microorganisms such as Bacteroidales, Rhodocyclaceae, Alcaligenaceae, Thermoanaerobacteriales, and Erysipelotrichaceae [40].
Syngas Fermentation to Chain Elongation: Perspectives from the Microbial Ecology of Mixed Communities
Syngas fermentation and CE presents a promising platform for biotechnological CO2 and CO fixation and fermentation of rich-organic substrates, respectively. Therefore, the synergy of combining syngas fermentation and CE has been a topic in previous studies [5, 13]. The first approach is a two-stage process (Fig. 3A), where syngas fermentation is the first stage and the second stage is a CE reactor fed with the effluent from the first reactor, preferably with high acetate and ethanol concentrations. The first reactor depends on carboxydotrophic and acetogenic bacteria, and the second reactor requires bacteria able to use ethanol and acetate. CE studies have used this strategy in which a single strain is used for the syngas fermentation stage (e.g., C. carboxidivorans, C. ljungdahlii, C. autoethanogenum), and C. kluyveri or a mixed culture is employed for the CE stage [67, 68].
Different strategies of syngas aided chain elongation (CE). A Two-stage process, B simultaneous syngas fermentation and CE, C syngas fermentation and CE aided to anaerobic digestion, and D dark fermentation aided to simultaneous syngas fermentation and CE. Short (SCCAs) and medium chain (MCCAs) carboxylic acids
Another approach consisted in the simultaneous combination of both processes when only syngas is fed (Fig. 3B). For example, Diender et al. [58] used a stable co-culture of C. autoethanogenum and C. kluyveri in culture bottles to convert syngas and CO ultimately to SCCAs and MCCAs. A similar synergy between a co-culture of C. aceticum and C. kluyveri was reported in a continuous bubbling reactor, where up to 79.4 mM butyrate and 68.9 mM caproate were obtained from a syngas mixture with 30% CO, 5% CO2, 15% H2, and 50% N2 [15]. Interestingly and up to date, C. carboxidivorans and E. limosum are the only two strains reported to be able to produce caproate from syngas, and C. carboxidivorans is the only known strain able to synthetize hexanol from syngas [69]. When C. carboxidivorans was not detected in syngas-fed microbiota, it was supposed that caproate and/or caprylate production arose via a multi-species synergy with conventional CE intermediated by acetate (or butyrate) and ethanol from the Wood-Ljungdahl pathway [2].
Besides the above mentioned “syngas aided anaerobic fermentation,” the use of organic electron donors such as acetate, lactate, or ethanol can enable the production of MCCAs through microbial CE in anaerobic fermentation. For instance, the addition of 0.5 g/L of acetate improved the bioconversion of CO to ethanol and acetate in batch cultures of C. autoethanogenum [32]. However, and from a sustainable point of view, utilization of rich carbohydrates biomass is preferable to obtain such electron donors (Fig. 3C). One anaerobic fermentation option is the lactic acid fermentation. In this process, lactic acid bacteria in situ convert carbohydrates to lactate as electron donors that are subsequently consumed during CE. Simultaneously, utilizing H2, CO2, and occasionally CO in fermentation systems can facilitate the production of MCCAs through the generation of acetate, an electron acceptor in CE, and the generation of ethanol, serving as an electron donor for CE. Generally, the use of mixed cultures has reported productivities and concentrations of MCCAs comparable with those of single strain cultures of CE species and in a broader range of pH [5]. For example, Nzeteu et al. [70] suggested that a lactate-based CE community had synergy with hydrogenotrophic activity to produce about 130% more caproate (until 89.5 mM) in comparison with the H2-free fermentation (35.2 mM). A similar synergy was recently reported by Wu et al. [71], where comparing with non-H2-supplemented test, the lactate-carbon-flow used for MCCAs production was enhanced by 28.4% after H2 supply, obtaining maximum caproate production of 47.3 mM.
The strategy depicted in Fig. 3D allows the incorporation of dark fermentation to CE. Dark fermentation is the anaerobic conversion of carbohydrate-rich organic matter into some SCCAs (e.g., acetate, butyrate, lactate), alcohols, H2, and CO2, principally [72]. Continuous dark fermentation reactors have reported suitable gas productivities until 54 L/L-d (55% H2 and 45% CO2) and average acetate, butyrate and lactate concentrations of 6, 11, and 10 g/L [73, 74]. The stimulation/presence of acetogens and lactic acid bacteria was one of the main results obtained in such investigations. Nevertheless, the use of biomass with high abundance and activity of acetogens for syngas fermentation has been poorly studied. And the combination of dark fermentation to syngas aided CE has not been reported until now.
Therefore, a foreseen way forward in the field is the development of a sequential system to produce CO2, H2, SCCAs, and MCCAs. The implementation of such system could consist of two reactors. In the first reactor, the dark fermentation process will be carried out with the production of H2, CO2, and SCCAs at organic loading rates where acetogenic bacteria are naturally present. In the second reactor, the gas fermentation will be performed using the H2 and CO2 produced in the first reactor and the selected and enriched autotrophic biomass from the first reactor as a source of inoculum. This second reactor is expected to produce acetate and ethanol without the fed of an organic substrate, as well as butyrate, valerate, caproate by the CE process.
Overall, the co-feeding strategy of syngas and degradable substrates can brand mixed cultures viable for syngas-fermenting reactors. In other words, mixed microbial communities may outclass in syngas-aided CE. The use of mixed microbial communities can further add simplifications to the bioprocess of SCCAs and MCCAs production. It is known from anaerobic digestion and fermentation studies that microbial communities can operate steadily in non-sterile reactors, which can reduce operating and maintaining costs in comparison with single strain cultures [72, 75]. Besides, it is expected that mixed microbial communities can better handle the inhibitors and contaminants typically found in syngas (e.g., sulfur oxides, ethylene, acetylene) that may negatively affect the performance of the syngas fermenters [5]. This characteristic resilience to substrate quality and composition fluctuations has been decisive for the success of anaerobic digestion and wastewater treatment [72]. However, and to the best of our knowledge, studies focused on the robustness of mixed microbial communities with real syngas must be developed.
Omic-Sciences in Syngas Fermentation and Chain Elongation
To date, most studies have employed 16S rRNA gene Illumina sequencing to characterize CE and syngas fermentation microbiomes (Tables 1 and 2), which is a well-established technology but has important biases and limitations. For example, the sequencing data reported in each individual study may be affected by biases such as extraction protocol, primer choice, and sequencing approach. Also, a comprehensive and complete characterization of the microbial community structure can be hindered by the low sequencing depth of 16S rRNA sequencing. Therefore, omics-sciences stand out as excellent tools to complement and acquire information that cannot be obtained from 16S rRNA sequencing analysis. They are designed mostly at the universal detection of genes (metagenomics), mRNA (metatranscriptomics), proteins (metaproteomics), and metabolites (metabolomics) in a specific biologic sample in a non-targeted and non-biased way [76].
The advent of high-throughput sequencing technologies used in metagenomics and metatranscriptomics has made it possible to obtain datasets that are commensurate to the complexity of these microbial communities. Metagenomics is a new approach to study microorganisms obtained from a specific environment by functional gene screening or sequencing analysis. Metagenomics studies focus on microbial diversity, community structure, genetic and evolutionary relationships, and interactions and relationships with the environment [77]. Metatranscriptomic methods can be used to compare a biological response to different conditions or treatments or to assess physiological responses to external stimulation. Whole transcriptome sequencing is the most widely used method for studying RNA functions, exploring and analyzing the gene structure and function, and revealing intrinsic links between gene expression and biological phenomena [78], whereas metaproteomics involves characterization of the protein components present in the environmental microbial community at a specific time. It determines the protein complement that is post-transcriptionally regulated and translated. The community protein complement also includes the proteins that interact within and among a microbial community [79]. Finally, metabolomics is a comprehensive, qualitative, and quantitative study of all the small molecules in an organism. Metabolomic tools are being increasingly used to generate an unbiased global profile of metabolites in samples or to quantify with high sensitivity a small panel of metabolites. Metabolites are the result of the interaction of the system’s genome with its environment and are not only the end product of gene expression but also form part of the regulatory system in an integrated manner [78].
In the literature, few metagenomics and metatranscriptomics studies report the microbial ecology of CE and syngas fermentation processes with mixed microbial communities. Agler et al. [80] conducted the first study of metagenomic analysis on mixed cultures to produce MCCAs using raw organic materials (yeast-fermentation beer). The metagenomic analysis suggested that more than 50% of all assigned reads were from Clostridium spp. (particularly C. kluyveri), and that these were highly correlated with caproate production. Also, they found that other genera, including Ethanoligenens, Bifidobacterium, and Desulfitobacterium, represented important pools of genes for hydrolysis and ethanol oxidation. Wu et al. [48] through a metagenomic analysis suggested that both the reverse β-oxidation and the fatty acid biosynthesis pathways contributed to the CE process in a system with waste activated sludge and alkaline fermentation liquor as a feedstock. Also, they estimated that the functional enzymes as well as the fatty acid biosynthesis and the reverse β-oxidation pathways were mainly associated with C. kluyveri, C. botulinum, and C. magnum as key species responsible for the CE process. This study also proposed that acetogenesis, via the Wood-Ljungdahl pathway, is another important bioprocess for inorganic carbon fixation. Similarly, Leng et al. [62] studied the co-production of propanediol and caproate in a 2 L mixed-cultured semi-continuous reactor. From the metagenomic analysis, the authors proposed that E. limosum is capable of converting glycerol to propanediol, ethanol and H2, and redirecting the electron potential of H2 into acetate via the Wood-Ljungdahl pathway, which is then used for caproate production. C. kluyveri worked synergistically with E. limosum by consuming ethanol and acetate for caproate production.
Han et al. [51] carried out a metagenomic analysis from a CE reactor inoculated with acclimated biogas microbiome reactor. Using the metagenomic assembly approach, they retrieved 91 draft genomes in total, 3 of which were nearly complete and were assigned to unknown strains of Methanolinea tarda, Bordetella avium, and Planctomycetaceae, which except for the methanogen M. tarda, were likely new-found active participators of CE in the mixed culture. Another relevant result in this investigation was that the microbial structure of the CE reactor was like the inoculum reactor although reactor performances differed. Therefore, a metatranscriptomic analysis was additionally performed. The RNA sequencing results demonstrated that the microbial structure was highly stable while their function was flexible. Additionally, the fatty acid biosynthesis pathway, rather than the reverse β-oxidation pathway for CE, was more active and pivotal [42]. Interestingly, the distribution of microbial abundance in the metagenomic and metatranscriptomic analysis was significantly different. For example, the Clostridium relative abundances varied from 0.0 to 6.2; however, the low genetic abundance exhibited much higher relative transcriptomic abundances, while methanogens were not assigned as the transcriptionally active genera. These results reveal that metagenome analysis at the shotgun level also had limitations to identify the reactor microbiome. Additionally, key genes with low abundance but high transcription might be ignored. Therefore, metatranscriptomic analysis is more recommended to identify microorganisms and their functional associations.
Wang et al. [44] carried out a metatranscriptomic study of Clostridium lactatifermentans for CE in batch reactors fed with glucose and lactose. Interestingly, they found that C. lactatifermentans showed robust growth on glucose but more active caproate synthesis on lactate. Comparative transcriptome revealed that the genes involved in the reverse β-oxidation for caproate synthesis and ATPase-dependent ATP generation were upregulated under lactate feeding, while several genes responsible for biomass synthesis were upregulated under glucose feeding. Thus, metatranscriptomics achieves an in-depth understanding of carbon and energy metabolism in single strain and/or mixed cultures for future genetic engineering to optimize microbial cell factory towards MCCAs production. To our knowledge, metaproteomic and metametabolomic analyses have not been used into the analysis of CE and syngas fermentation processes.
Overall, the information obtained via several omics is not only important to understand the ecology and function in microbiomes; nonetheless, will also become crucial to choose the right chain elongator microorganism. Some microbial species or genera may have faster growth rates or higher tolerances for substrates, making them more suitable as “biocatalysts” in a given process/application, or conversely, such features might need to be taken into consideration in the bioprocess design [2].
Research Gaps and Future Perspectives
The biotechnological processes involving syngas fermentation and CE with mixed cultures hold immense potential for biofuel production, chemical synthesis, and waste utilization. Nevertheless, and as highlighted in previous sections and resumed in Fig. 4, several research gaps and challenges in these domains require attention to unlock their full potential. The main research gaps relate on the scarce understanding of the selection and interaction of microorganisms within mixed cultures. Overall, the microbial communities in open or mixed-culture fermentation are inherently more complex than single strain cultures. Despite the identification of single strains, the specific roles of satellite communities within the mixed-culture fermentation system still need clarified. The use of high-throughput sequencing technologies is imperative to gain a deeper insight into the criteria for selecting microbial cultures and to elucidate how various consortia perform under diverse conditions. This understanding is crucial for elucidating their cooperative interactions and the competition among microbial groups for substrates. Similarly, ensuring the stability of mixed cultures over prolonged fermentation periods is of paramount relevance for their industrial applicability. Future research actions should prioritize enhancing these cultures resilience, robustness, and durability while devising strategies for effective recovery from disturbances or upsets.
Reactor and process design for coupling syngas fermentation and CE are critical to advance these biotechnological processes. Effective mass transfer of syngas components and products within the reactor remains challenging. Research should focus on investigating reactor designs that facilitate efficient gas-liquid-solid interactions, optimizing the conversion of syngas into desired products while minimizing waste. Interestingly, current research often needs more integrated reactor systems that can simultaneously support both syngas fermentation and CE processes. Therefore, future research should focus on (i) the design of multi-compartment reactors that can house distinct microbial consortia optimized for syngas fermentation and CE, enabling enhanced process control; (ii) the integration of reactor design with downstream processing techniques, such as product recovery and separation, to create a holistic and efficient production chain; and (iii) the transition from batch to continuous reactor operation to improve productivity and stability. Continuous processes can minimize downtime and enhance product consistency.
Finally, more investigation is needed on utilizing waste streams, such as agricultural residues and industrial byproducts, as supplemental feedstocks for syngas fermentation, and CE, promoting sustainability and circular economy principles. Furthermore, the operational expenses associated with the extraction equipment pose a significant challenge to be addressed in full-scale applications. These costs encompass factors such as the price of the extractant and electricity consumption. Subsequent research efforts may focus on identifying cost-effective extractants and advancing the development of novel membrane materials. This pursuit aims to minimize operational investments and enhance product selectivity.
Conclusions
Emerging syngas fermentation and CE process for biofuel production has both environmental and sustainable advantages. In this review, we summarized recent advances when using mixed cultures for syngas fermentation and MCCAs production via CE process. Overall, the use of mixed microbial communities offers advantages over single strain cultures because the microbial diversity in mixed cultures improves the process stability and resilience under changing conditions. However, some negative reactions such as methanogenesis also compete for substrates with the CE.
In addition, the metabolic pathways of syngas fermentation and CE processes has been investigated by various molecular biology technologies. In general, for CE the reverse β-oxidation is widely recognized as the main metabolic pathway, but current studies revealed a new potential CE process, e.g., the fatty acid biosynthesis process. Future studies should aim at improving fatty acid biosynthesis pathway to produce MCCAs. Although the syngas fermentation aimed CE process from organic wastes to obtain caproate and/or long-chain acids is attractive, its physiological understanding is insufficient, and further omics studies are required for stable operation or trouble shooting bioreactors. Therefore, future work should build on the expanding molecular information from mixed cultures and characterization efforts to eventually unravel the flows of substrates and products within microbial community members.
Data Availability
All data generated or analysed during this study are included in this published article.
References
Sobieraj K, Stegenta-Dąbrowska S, Luo G, Koziel JA, Białowiec A (2022) Carbon monoxide fate in the environment as an inspiration for biorefinery industry: a review. Front Environ Sci 10. https://doi.org/10.3389/fenvs.2022.822463
Candry P, Ganigue R (2021) Chain elongators, friends, and foes. Curr Opin Biotechnol 67:99–110. https://doi.org/10.1016/j.copbio.2021.01.005
Ashokkumar V, Venkatkarthick R, Jayashree S, Chuetor S, Dharmaraj S, Kumar G, Chen WH, Ngamcharussrivichai C (2022) Recent advances in lignocellulosic biomass for biofuels and value-added bioproducts - a critical review. Bioresour Technol 344(Pt B):126195. https://doi.org/10.1016/j.biortech.2021.126195
Andreides D, Fliegerova KO, Pokorna D, Zabranska J (2022) Biological conversion of carbon monoxide and hydrogen by anaerobic culture: prospect of anaerobic digestion and thermochemical processes combination. Biotechnol Adv 58:107886. https://doi.org/10.1016/j.biotechadv.2021.107886
Baleeiro FCF, Kleinsteuber S, Neumann A, Strauber H (2019) Syngas-aided anaerobic fermentation for medium-chain carboxylate and alcohol production: the case for microbial communities. Appl Microbiol Biotechnol 103(21-22):8689–8709. https://doi.org/10.1007/s00253-019-10086-9
He Y, Kennes C, Lens PNL (2022) Enhanced solventogenesis in syngas bioconversion: role of process parameters and thermodynamics. Chemosphere 299:134425. https://doi.org/10.1016/j.chemosphere.2022.134425
Chang Y-J, Chang J-S, Lee D-J (2023) Gasification of biomass for syngas production: research update and stoichiometry diagram presentation. Bioresour Technol 387:129535. https://doi.org/10.1016/j.biortech.2023.129535
Li H, Wang J, Chen C, Jia L, Hou B, Li D (2017) Effects of macropores on reducing internal diffusion limitations in Fischer–Tropsch synthesis using a hierarchical cobalt catalyst. RSC Advances 7(16):9436–9445. https://doi.org/10.1039/C6RA27166A
Sivalingam V, Winkler D, Haugen T, Wentzel A, Dinamarca C (2022) Syngas fermentation and microbial electrosynthesis integration as a single process unit. Bioresour Technol 356:127314. https://doi.org/10.1016/j.biortech.2022.127314
Yang C, Dong L, Gao Y, Jia P, Diao Q (2021) Engineering acetogens for biofuel production: from cellular biology to process improvement. Renew Sust Energ Rev 151. https://doi.org/10.1016/j.rser.2021.111563
Zhang L, Zhao R, Jia D, Jiang W, Gu Y (2020) Engineering Clostridium ljungdahlii as the gas-fermenting cell factory for the production of biofuels and biochemicals. Curr Opin Chem Biol 59:54–61. https://doi.org/10.1016/j.cbpa.2020.04.010
Wainaina S, Horvath IS, Taherzadeh MJ (2018) Biochemicals from food waste and recalcitrant biomass via syngas fermentation: a review. Bioresour Technol 248(Pt A):113–121. https://doi.org/10.1016/j.biortech.2017.06.075
Angenent LT, Richter H, Buckel W, Spirito CM, Steinbusch KJ, Plugge CM, Strik DP, Grootscholten TI, Buisman CJ, Hamelers HV (2016) Chain elongation with reactor microbiomes: open-culture biotechnology to produce biochemicals. Environ Sci Technol 50(6):2796–2810. https://doi.org/10.1021/acs.est.5b04847
Brodowski F, Lezyk M, Gutowska N, Kabasakal T, Oleskowicz-Popiel P (2022) Influence of lactate to acetate ratio on biological production of medium chain carboxylates via open culture fermentation. Sci Total Environ 851(Pt 1):158171. https://doi.org/10.1016/j.scitotenv.2022.158171
Fernandez-Blanco C, Veiga MC, Kennes C (2022) Efficient production of n-caproate from syngas by a co-culture of Clostridium aceticum and Clostridium kluyveri. J Environ Manage 302(Pt A):113992. https://doi.org/10.1016/j.jenvman.2021.113992
Shi X, Wu L, Wei W, Ni B-J (2021) Insights into the microbiomes for medium-chain carboxylic acids production from biowastes through chain elongation. Crit Rev Environ Sci Technol 52(21):3787–3812. https://doi.org/10.1080/10643389.2021.1957342
Liberato V, Benevenuti C, Coelho F, Botelho A, Amaral P, Pereira N, Ferreira T (2019) Clostridium sp. as bio-catalyst for fuels and chemicals production in a biorefinery context. Catalysts 9(11). https://doi.org/10.3390/catal9110962
Xue C, Zhao J, Chen L, Yang ST, Bai F (2017) Recent advances and state-of-the-art strategies in strain and process engineering for biobutanol production by Clostridium acetobutylicum. Biotechnol Adv 35(2):310–322. https://doi.org/10.1016/j.biotechadv.2017.01.007
Nascimento TR, Cavalcante WA, de Oliveira GHD, Zaiat M, Ribeiro R (2022) Modeling dark fermentation of cheese whey for H2 and n-butyrate production considering the chain elongation perspective. Bioresour Technol Rep 17. https://doi.org/10.1016/j.biteb.2021.100940
Stoll IK, Boukis N, Sauer J (2020) Syngas fermentation to alcohols: reactor technology and application perspective. Chem Ing Tech 92(1-2):125–136. https://doi.org/10.1002/cite.201900118
Bolívar Caballero JJ, Zaini IN, Yang W (2022) Reforming processes for syngas production: a mini-review on the current status, challenges, and prospects for biomass conversion to fuels. Appl Energy Combust Sci 10:100064. https://doi.org/10.1016/j.jaecs.2022.100064
Annie Modestra J, Katakojwala R, Venkata Mohan S (2020) CO2 fermentation to short chain fatty acids using selectively enriched chemolithoautotrophic acetogenic bacteria. Chem Eng J 394. https://doi.org/10.1016/j.cej.2020.124759
Ramio-Pujol S, Ganigue R, Baneras L, Colprim J (2018) Effect of ethanol and butanol on autotrophic growth of model homoacetogens. FEMS Microbiol Lett 365(10). https://doi.org/10.1093/femsle/fny084
Asimakopoulos K, Gavala HN, Skiadas IV (2018) Reactor systems for syngas fermentation processes: a review. Chem Eng J 348:732–744. https://doi.org/10.1016/j.cej.2018.05.003
Phillips J, Huhnke R, Atiyeh H (2017) Syngas fermentation: a microbial conversion process of gaseous substrates to various products. Fermentation 3(2). https://doi.org/10.3390/fermentation3020028
Gunes B (2021) A critical review on biofilm-based reactor systems for enhanced syngas fermentation processes. Renew Sust Energ Rev 143. https://doi.org/10.1016/j.rser.2021.110950
Sun X, Atiyeh HK, Huhnke RL, Tanner RS (2019) Syngas fermentation process development for production of biofuels and chemicals: a review. Bioresour Technol Rep 7. https://doi.org/10.1016/j.biteb.2019.100279
Schulz S, Molitor B, Angenent LT (2023) Acetate augmentation boosts the ethanol production rate and specificity by Clostridium ljungdahlii during gas fermentation with pure carbon monoxide. Bioresour Technol 369:128387. https://doi.org/10.1016/j.biortech.2022.128387
Riegler P, Chrusciel T, Mayer A, Doll K, Weuster-Botz D (2019) Reversible retrofitting of a stirred-tank bioreactor for gas-lift operation to perform synthesis gas fermentation studies. Biochem Eng J 141:89–101. https://doi.org/10.1016/j.bej.2018.09.021
Tanner RS, Miller LM, Yang D (1993) Clostridium ljungdahlii sp. nov., an acetogenic species in clostridial rRNA homology group I. Int J Syst Bacteriol 43(2):232–236. https://doi.org/10.1099/00207713-43-2-232
Whitham JM, Schulte MJ, Bobay BG, Bruno-Barcena JM, Chinn MS, Flickinger MC, Pawlak JJ, Grunden AM (2017) Characterization of Clostridium ljungdahlii OTA1: a non-autotrophic hyper ethanol-producing strain. Appl Microbiol Biotechnol 101(4):1615–1630. https://doi.org/10.1007/s00253-016-7978-6
Xu H, Liang C, Yuan Z, Xu J, Hua Q, Guo Y (2017) A study of CO/syngas bioconversion by Clostridium autoethanogenum with a flexible gas-cultivation system. Enzyme Microb Technol 101:24–29. https://doi.org/10.1016/j.enzmictec.2017.03.002
Islam MS, Guo C, Liu C-Z (2018) Enhanced hydrogen and volatile fatty acid production from sweet sorghum stalks by two-steps dark fermentation with dilute acid treatment in between. Int J Hydrog Energy 43(2):659–666. https://doi.org/10.1016/j.ijhydene.2017.11.059
Xiang Y, Luo H, Liu G, Zhang R (2022) Improvement of organic acid production with sulfate addition during syngas fermentation using mixed cultures. Water Cycle 3:26–34. https://doi.org/10.1016/j.watcyc.2022.02.001
Shen N, Dai K, Xia X-Y, Zeng RJ, Zhang F (2018) Conversion of syngas (CO and H2) to biochemicals by mixed culture fermentation in mesophilic and thermophilic hollow-fiber membrane biofilm reactors. J Clean Prod 202:536–542. https://doi.org/10.1016/j.jclepro.2018.08.162
Zhang F, Ding J, Zhang Y, Chen M, Ding ZW, van Loosdrecht MC, Zeng RJ (2013) Fatty acids production from hydrogen and carbon dioxide by mixed culture in the membrane biofilm reactor. Water Res 47(16):6122–6129. https://doi.org/10.1016/j.watres.2013.07.033
Rao Y, Wan J, Liu Y, Angelidaki I, Zhang S, Zhang Y, Luo G (2018) A novel process for volatile fatty acids production from syngas by integrating with mesophilic alkaline fermentation of waste activated sludge. Water Res 139:372–380. https://doi.org/10.1016/j.watres.2018.04.026
Liu H, Shi J, Zhan X, Zhang L, Fu B, Liu H (2017) Selective acetate production with CO2 sequestration through acetogen-enriched sludge inoculums in anaerobic digestion. Biochem Eng J 121:163–170. https://doi.org/10.1016/j.bej.2017.02.008
Chakraborty S, Rene ER, Lens PNL, Veiga MC, Kennes C (2019) Enrichment of a solventogenic anaerobic sludge converting carbon monoxide and syngas into acids and alcohols. Bioresour Technol 272:130–136. https://doi.org/10.1016/j.biortech.2018.10.002
Calvo DC, Ontiveros-Valencia A, Krajmalnik-Brown R, Torres CI, Rittmann BE (2021) Carboxylates and alcohols production in an autotrophic hydrogen-based membrane biofilm reactor. Biotechnol Bioeng 118(6):2338–2347. https://doi.org/10.1002/bit.27745
Liu C, Luo G, Wang W, He Y, Zhang R, Liu G (2018) The effects of pH and temperature on the acetate production and microbial community compositions by syngas fermentation. Fuel 224:537–544. https://doi.org/10.1016/j.fuel.2018.03.125
Kim H, Kang S, Sang BI (2022) Metabolic cascade of complex organic wastes to medium-chain carboxylic acids: a review on the state-of-the-art multi-omics analysis for anaerobic chain elongation pathways. Bioresour Technol 344(Pt A):126211. https://doi.org/10.1016/j.biortech.2021.126211
Spirito CM, Richter H, Rabaey K, Stams AJ, Angenent LT (2014) Chain elongation in anaerobic reactor microbiomes to recover resources from waste. Curr Opin Biotechnol 27:115–122. https://doi.org/10.1016/j.copbio.2014.01.003
Wang H, Zhou W, Gao J, Ren C, Xu Y (2022) Revealing the characteristics of glucose- and lactate-based chain elongation for caproate production by Caproicibacterium lactatifermentans through transcriptomic, bioenergetic, and regulatory analyses. mSystems 7(5):e0053422. https://doi.org/10.1128/msystems.00534-22
Li L, Liu C, Xu L, Zhuang H, He J, He Q, Zhang J (2023) Acclimation of anaerobic fermentation microbiome with acetate and ethanol for chain elongation and the biochemical response. Chemosphere 320:138083. https://doi.org/10.1016/j.chemosphere.2023.138083
Grootscholten TI, Steinbusch KJ, Hamelers HV, Buisman CJ (2013) Improving medium chain fatty acid productivity using chain elongation by reducing the hydraulic retention time in an upflow anaerobic filter. Bioresour Technol 136:735–738. https://doi.org/10.1016/j.biortech.2013.02.114
Ji X, Zhu K, Zhang Y, Ullah F, Li A, Zhang L (2022) Mixed culture chain elongation for consumption of acetate and ethanol in anaerobic fermentation: the impact of salt type, dosage and acclimation. Waste Manag 152:48–58. https://doi.org/10.1016/j.wasman.2022.08.005
Wu SL, Sun J, Chen X, Wei W, Song L, Dai X, Ni BJ (2020) Unveiling the mechanisms of medium-chain fatty acid production from waste activated sludge alkaline fermentation liquor through physiological, thermodynamic and metagenomic investigations. Water Res 169:115218. https://doi.org/10.1016/j.watres.2019.115218
Wang J, Yin Y (2022) Biological production of medium-chain carboxylates through chain elongation: an overview. Biotechnol Adv 55:107882. https://doi.org/10.1016/j.biotechadv.2021.107882
Magdalena JA, Ballesteros M, González-Fernández C (2020) Acidogenesis and chain elongation for bioproduct development. In: Olivares JA, Puyol D, Melero JA, Dufour J (eds) Wastewater treatment residues as resources for biorefinery products and biofuels. Elsevier, pp 391–414. https://doi.org/10.1016/B978-0-12-816204-0.00017-5
Han W, He P, Shao L, Lu F (2018) Metabolic interactions of a chain elongation microbiome. Appl Environ Microbiol 84(22). https://doi.org/10.1128/AEM.01614-18
Wallace RJ, Chaudhary LC, Miyagawa E, McKain N, Walker ND (2004) Metabolic properties of Eubacterium pyruvativorans, a ruminal 'hyper-ammonia-producing' anaerobe with metabolic properties analogous to those of Clostridium kluyveri. Microbiology (Reading) 150(Pt 9):2921–2930. https://doi.org/10.1099/mic.0.27190-0
San-Valero P, Fernández-Naveira Á, Veiga MC, Kennes C (2019) Influence of electron acceptors on hexanoic acid production by Clostridium kluyveri. J Environ Manage 242:515–521. https://doi.org/10.1016/j.jenvman.2019.04.093
Zhu X, Zhou Y, Wang Y, Wu T, Li X, Li D, Tao Y (2017) Production of high-concentration n-caproic acid from lactate through fermentation using a newly isolated Ruminococcaceae bacterium CPB6. Biotechnol Biofuels 10:102. https://doi.org/10.1186/s13068-017-0788-y
Jeon BS, Choi O, Um Y, Sang BI (2016) Production of medium-chain carboxylic acids by Megasphaera sp. MH with supplemental electron acceptors. Biotechnol Biofuels 9:129. https://doi.org/10.1186/s13068-016-0549-3
Kim BC, Seung Jeon B, Kim S, Kim H, Um Y, Sang BI (2015) Caproiciproducens galactitolivorans gen. nov., sp. nov., a bacterium capable of producing caproic acid from galactitol, isolated from a wastewater treatment plant. Int J Syst Evol Microbiol 65(12):4902–4908. https://doi.org/10.1099/ijsem.0.000665
Flaiz M, Baur T, Brahner S, Poehlein A, Daniel R, Bengelsdorf FR (2020) Caproicibacter fermentans gen. nov., sp. nov., a new caproate-producing bacterium and emended description of the genus Caproiciproducens. Int J Syst Evol Microbiol 70(7):4269–4279. https://doi.org/10.1099/ijsem.0.004283
Diender M, Stams AJ, Sousa DZ (2016) Production of medium-chain fatty acids and higher alcohols by a synthetic co-culture grown on carbon monoxide or syngas. Biotechnol Biofuels 9:82. https://doi.org/10.1186/s13068-016-0495-0
Liu Y, Lü F, Shao L, He P (2016) Alcohol-to-acid ratio and substrate concentration affect product structure in chain elongation reactions initiated by unacclimatized inoculum. Bioresour Technol 218:1140–1150. https://doi.org/10.1016/j.biortech.2016.07.067
Jourdin L, Raes SMT, Buisman CJN, Strik DPBTB (2018) Critical biofilm growth throughout unmodified carbon felts allows continuous bioelectrochemical chain elongation from CO2 up to caproate at high current density. Front Energy Res 6. https://doi.org/10.3389/fenrg.2018.00007
Wang Q, Zhang P, Bao S, Liang J, Wu Y, Chen N, Wang S, Cai Y (2020) Chain elongation performances with anaerobic fermentation liquid from sewage sludge with high total solid as electron acceptor. Bioresour Technol 306:123188. https://doi.org/10.1016/j.biortech.2020.123188
Leng L, Nobu MK, Narihiro T, Yang P, Amy Tan GY, Lee PH (2019) Shaping microbial consortia in coupling glycerol fermentation and carboxylate chain elongation for Co-production of 1,3-propanediol and caproate: Pathways and mechanisms. Water Res 148:281–291. https://doi.org/10.1016/j.watres.2018.10.063
Palomo-Briones R, Xu J, Spirito CM, Usack JG, Trondsen LH, Guzman JJL, Angenent LT (2022) Near-neutral pH increased n-caprylate production in a microbiome with product inhibition of methanogenesis. Chem Eng J 446:137170. https://doi.org/10.1016/j.cej.2022.137170
Contreras-Davila CA, Esveld J, Buisman CJN, Strik D (2021) nZVI impacts substrate conversion and microbiome composition in chain elongation from D- and L-lactate substrates. Front Bioeng Biotechnol 9:666582. https://doi.org/10.3389/fbioe.2021.666582
Zhang L, Wang X, Chen Y, Zhang B, Xu H, Li C, Zhou Y (2023) Medium-chain fatty acid production from thermal hydrolysed sludge without external electron donor supplementation. Bioresour Technol 374:128805. https://doi.org/10.1016/j.biortech.2023.128805
Weimer PJ, Nerdahl M, Brandl DJ (2015) Production of medium-chain volatile fatty acids by mixed ruminal microorganisms is enhanced by ethanol in co-culture with Clostridium kluyveri. Bioresour Technol 175:97–101. https://doi.org/10.1016/j.biortech.2014.10.054
Vasudevan D, Richter H, Angenent LT (2014) Upgrading dilute ethanol from syngas fermentation to n-caproate with reactor microbiomes. Bioresour Technol 151:378–382. https://doi.org/10.1016/j.biortech.2013.09.105
Gildemyn S, Molitor B, Usack JG, Nguyen M, Rabaey K, Angenent LT (2017) Upgrading syngas fermentation effluent using Clostridium kluyveri in a continuous fermentation. Biotechnol Biofuels 10:83. https://doi.org/10.1186/s13068-017-0764-6
Phillips JR, Atiyeh HK, Tanner RS, Torres JR, Saxena J, Wilkins MR, Huhnke RL (2015) Butanol and hexanol production in Clostridium carboxidivorans syngas fermentation: Medium development and culture techniques. Bioresour Technol 190:114–121. https://doi.org/10.1016/j.biortech.2015.04.043
Co N, Coelho F, Trego AC, Abram F, Ramiro-Garcia J, Paulo L, O'Flaherty V (2022) Development of an enhanced chain elongation process for caproic acid production from waste-derived lactic acid and butyric acid. J Clean Prod 338. https://doi.org/10.1016/j.jclepro.2022.130655
Wu Q, Guo W, You S, Bao X, Luo H, Wang H, Ren N (2019) Concentrating lactate-carbon flow on medium chain carboxylic acids production by hydrogen supply. Bioresour Technol 291:121573. https://doi.org/10.1016/j.biortech.2019.121573
Castelló E, Nunes Ferraz-Junior AD, Andreani C, Anzola-Rojas MP, Borzacconi L, Buitrón G, Carrillo-Reyes J, Gomes SD, Maintinguer SI, Moreno-Andrade I, Palomo-Briones R, Razo-Flores E, Schiappacasse-Dasati M, Tapia-Venegas E, Valdez-Vázquez I, Vesga-Baron A, Zaiat M, Etchebehere C (2020) Stability problems in the hydrogen production by dark fermentation: possible causes and solutions. Renew Sust Energ Rev 119:109602. https://doi.org/10.1016/j.rser.2019.109602
Montoya-Rosales JJ, Palomo-Briones R, Celis LB, Etchebehere C, Cházaro-Ruiz LF, Escobar-Barrios V, Razo-Flores E (2022) Coping with mass transfer constrains in dark fermentation using a two-phase partitioning bioreactor. Chem Eng J 445. https://doi.org/10.1016/j.cej.2022.136749
Montoya-Rosales JJ, Palomo-Briones R, Celis LB, Etchebehere C, Razo-Flores E (2020) Discontinuous biomass recycling as a successful strategy to enhance continuous hydrogen production at high organic loading rates. Int J Hydrog Energy 45(35):17260–17269. https://doi.org/10.1016/j.ijhydene.2020.04.265
Dzulkarnain ELN, Audu JO, Wan Dagang WRZ, Abdul-Wahab MF (2022) Microbiomes of biohydrogen production from dark fermentation of industrial wastes: current trends, advanced tools and future outlook. Bioresour Bioprocess 9(1):16. https://doi.org/10.1186/s40643-022-00504-8
Deshmukh R, Sonah H, Patil G, Chen W, Prince S, Mutava R, Vuong T, Valliyodan B, Nguyen HT (2014) Integrating omic approaches for abiotic stress tolerance in soybean. Front Plant Sci 5:244. https://doi.org/10.3389/fpls.2014.00244
Zhang L, Chen F, Zeng Z, Xu M, Sun F, Yang L, Bi X, Lin Y, Gao Y, Hao H, Yi W, Li M, Xie Y (2021) Advances in metagenomics and its application in environmental microorganisms. Front Microbiol 12:766364. https://doi.org/10.3389/fmicb.2021.766364
Chakraborty D, Sharma N, Kour S, Sodhi SS, Gupta MK, Lee SJ, Son YO (2022) Applications of omics technology for livestock selection and improvement. Front Genet 13:774113. https://doi.org/10.3389/fgene.2022.774113
Srivastava N, Gupta B, Gupta S, Danquah MK, Sarethy IP (2019) Analyzing functional microbial diversity: an overview of techniques. In: Das S, Dash HR (eds) Microbial Diversity in the Genomic Era. Academic Press, pp 79–102. https://doi.org/10.1016/B978-0-12-814849-5.00006-X
Agler MT, Spirito CM, Usack JG, Werner JJ, Angenent LT (2012) Chain elongation with reactor microbiomes: upgrading dilute ethanol to medium-chain carboxylates. Energy Environ Sci 5(8). https://doi.org/10.1039/c2ee22101b
Acknowledgements
E. Razo-Flores acknowledges the “Cátedra Rodolfo Quintero-Ramírez” granted by the Universidad Autónoma Metropolitana-Cuajimalpa. P. Núñez-Valenzuela is thankful for the postgraduate scholarship provided by CONAHCYT.
Funding
This investigation was financially supported by SEP-CONACYT project A1-S-37174.
Author information
Authors and Affiliations
Contributions
J.J.M.-R.: conceptualization, investigation, data curation, formal analysis, writing-original draft. P.N.-V.: investigation, data curation, formal analysis, writing-original draft. A.O.-V.: conceptualization, writing-review and editing. M.M.-I.: writing-review and editing. S.R.: writing-review and editing. E.R.-F.: conceptualization, writing-review and editing, supervision, funding acquisition.
Corresponding author
Ethics declarations
Competing Interests
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Jesús Montoya-Rosales, J., Núñez-Valenzuela, P., Ontiveros-Valencia, A. et al. From Syngas Fermentation to Chain Elongation: the Role of Key Microorganisms and Multi-omics Analysis. Bioenerg. Res. 17, 897–911 (2024). https://doi.org/10.1007/s12155-023-10696-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12155-023-10696-2