Abstract
Purpose
The nucleoside analog 3′-deoxy-3′-18F-fluorothymidine (FLT) has been introduced for imaging cell proliferation with positron emission tomography (PET). We prospectively compared the diagnostic efficacy of FLT PET with that of 2-deoxy-2-18F-fluoro-d-glucose (FDG) PET for the preoperative nodal and distant metastatic staging of non-small cell lung cancer (NSCLC).
Methods
A total of 34 patients with NSCLC underwent FLT PET and FDG PET. PET imaging was performed at 60 min after each radiotracer injection. The PET images were evaluated qualitatively for regions of focally increased metabolism. For visualized primary tumors, the maximum standardized uptake value (SUV) was calculated. Nodal stages were determined by using the American Joint Committee on Cancer staging system and surgical and histologic findings reference standards.
Results
For the depiction of primary tumor, sensitivity of FLT PET was 67%, compared with 94% for FDG PET (P = 0.005). Sensitivity, specificity, positive predictive value, negative predictive value, and accuracy for lymph node staging on a per-patient basis were 57, 93, 67, 89, and 85%, respectively, with FLT PET and 57, 78, 36, 91, and 74%, respectively, with FDG PET (P > 0.1 for all comparisons). Two of the three distant metastases were detected with FLT and FDG PET.
Conclusion
In NSCLC, FLT PET showed better (although not statistically significant) specificity, positive predictive value and accuracy for N staging on a per-patient basis than FDG PET. However, FDG PET was found to have higher sensitivity for depiction of primary tumor than FLT PET.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Non-small cell lung cancer (NSCLC) accounts for 75–80% of all lung cancers and is currently the leading cause of tumor-related deaths [1]. The choice of the most appropriate therapy is determined by the tumor stage. The accurate determination of tumor size, adjacent structure invasion, mediastinal lymph node involvement, and the detection of distant metastases are of central importance.
Conventional chest radiography, computed tomography (CT), magnetic resonance imaging, radionuclide scintigraphy, and positron emission tomography (PET) are currently being used for NSCLC staging. Although tumor size and adjacent structure invasion are accurately assessed with CT [2], it is limited in the evaluation of nodal status [3, 4]. PET with 2-deoxy-2-18F-fluoro-d-glucose (FDG) is a well-established functional imaging technique for diagnostic oncologic imaging of a variety of malignancies [5]. FDG PET imaging of lung tumors has been applied for differentiation between benign and malignant lesions, staging, restaging, and determining response to treatment [6–8]. However, FDG is not a tumor-specific tracer, and false-positive findings can occur in inflammatory lesions [9]. Another problem with the technique is a decreased uptake during hyperglycemia [10].
To overcome the drawbacks of FDG, alternative radiotracers that are more closely related to cell proliferation have been investigated. 11C-thymidine is rapidly incorporated into deoxyribonucleic acid (DNA) and has been used for noninvasive imaging of tumor proliferation [11]. However, the short half-life of 11C (20 min) and the rapid in vivo degradation of 11C-thymidine make the radiotracer less suitable for routine clinical use.
Recently, a thymidine analog 3′-deoxy-3′-18F-fluorothymidine (FLT) was introduced as a stable cell proliferation imaging agent [12]. This tracer is trapped within the cytosol after being monophosphorylated by thymidine kinase-1 (TK1), a principle enzyme in the salvage pathway of DNA synthesis [13]. Therefore, the accumulation of FLT is dependent of the presence of TK1, which is closely associated with cellular proliferation [13]. FLT may reflect proliferation of lung nodules better than does FDG [14, 15]. Few data are available on the clinical comparison of FLT with FDG for staging of lung cancer [16–19]. The significance of FLT as a better diagnostic imaging tracer remains to be evaluated. Thus, the purpose of this study was to prospectively compare the diagnostic efficacy of FLT PET with that of FDG PET in the evaluation of the preoperative nodal and distant metastatic staging of NSCLC.
Materials and methods
Patients
This prospective study was approved by our institutional review board, and written informed consent was obtained from all patients.
A total of 48 patients with histopathologically proven NSCLC were enrolled in the study. All consecutive patients referred for surgery between April 2006 and March 2007 were included, and all underwent conventional lung cancer staging on the basis of clinical information, and both FLT and FDG PET studies. Ten patients were excluded because conventional staging studies suggested extrathoracic metastasis: three because they received chemotherapy and radiation before surgical staging, and one because of a history of diabetes and serum glucose concentration of 450 mg/dl at the time of FDG injection.
Thus, 34 patients (23 men and 11 women; mean age, 69 years; range, 55–81 years) were included for preoperative primary tumor staging (Table 1). All patients also underwent surgical staging. The mean interval between FLT PET and FDG PET was 6 days (range 1–25 days; median, 4 days), whereas that between the last PET and surgical staging was 9 days (range, 1–28 days; median, 7 days). Nodal stages were classified according to the American Joint Committee on Cancer (AJCC) staging system for the classification of lung cancer [20]. Histopathologic results served as the reference standards.
FLT synthesis and PET acquisition
FLT was synthesized using the method described by Machulla et al. [21]. The radiochemical purity of the produced FLT was >95%.
All acquisitions were performed using an ECAT EXACT HR+ scanner (Siemens/CTI, Knoxville, TN). The imaging system used enabled simultaneous acquisition of 63 transverse PET images per field of view (FOV), for a total axial FOV of 15.5 cm. In-plane resolution was approximately 4.6 mm, with an axial resolution of approximately 3.5 mm full-width at half-maximum. PET scans were acquired in the 3-dimensional mode. Transmission scan was obtained using a 68Ge rod source for the purpose of attenuation correction. PET images were reconstructed with ordered subset expectation maximization (OS-EM) using two iterations and eight subsets.
Patients were instructed to fast for at least 5 h before PET imaging, although oral hydration with glucose-free water was allowed. After a normal blood glucose level in the peripheral blood was ensured on FDG PET study, patients received an intravenous administration of 3.5 MBq/kg of each radiotracer. Sixty minutes after radiotracer injection, attenuation-corrected whole-body scanning was performed from the skull base to the proximal thighs. For transmission and emission scanning, 2 and 3 min, respectively, were allowed per bed position.
PET image analysis
PET images were reviewed on a Sun Microsystems workstation (Siemens/CTI) in transverse, coronal, and sagittal planes along with maximum-intensity-projection images. Independently, two nuclear medicine physicians, who were blinded to clinical and pathologic results, prospectively evaluated FLT PET and FDG PET data. FLT and FDG PET images were read in the random, with an interval of several days between interpretations. The observers blinded to identifying information. Any difference of opinion was resolved consensus.
The PET images were evaluated qualitatively for regions of focally increased metabolism. An increased uptake to a level greater than that in the surrounding tissue was considered to characterize malignancy. For primary tumors visualized on PET, region of interest (ROI) was placed over the entire FLT- or FDG-avid lesion on all transverse planes in which the tumor appeared. The maximum standardized uptake value (SUV) was calculated by using the following formula: SUV = c dc/(d i/w), where c dc is the decay-corrected tracer tissue concentration (in Bq/g); d i, the injected dose (in Bq); and w, the patient’s body weight (in g). When no tumor-related radioactivity was discernible at visual analysis, the case was assigned a SUV of 0. The mediastinal lesions were assigned according to the Mountain and Dresler classification of regional lymph nodes [22]. Tumor staging with PET was based on the AJCC staging system for the classification of lung cancer [20].
Histopathologic analysis
The stage of regional lymph node involvement (hereafter called N stage) was determined for all patients on the basis of findings at tumor resection with mediastinal lymph node dissection. Distant metastasis stage (hereafter called M stage) was determined by means of biopsy or radiologic follow-up. Apart from assessment of tumor stage, PET images were evaluated for additional clinically important findings.
Statistical analysis
Detection rate of primary tumor was compared between FLT PET and FDG PET by using the McNemar’s test. The same test was also used to determine the statistical significance of differences in N stage determined with FLT PET and FDG PET. FLT SUV and FDG SUV were compared by using the two-tailed Wilcoxon signed rank test. P < 0.05 was considered to indicate a statistically significant difference. The sensitivity, specificity, positive predictive value, negative predictive value, and accuracy of the PET examinations in the assessment of lymph node involvement were calculated on a per-patient basis and on a per-nodal-station basis, for both FLT PET and FDG PET. The values were expressed as mean, with 95% confidence interval (CI).
Results
Detection of primary lung tumor
Histologic analysis revealed 20 adenocarcinomas, 12 squamous cell carcinomas, 3 pleomorphic carcinomas, and one adenosquamous carcinoma. Two patients had two primary lesions each. Thus, a total of 36 lesions were examined.
For depiction of primary lung tumor, the sensitivity of FLT PET was 67% (24 of 36), compared with 94% (34 of 36) for FDG PET (P = 0.005, McNemar’s test; Table 1). Mean (±SD) FLT SUV in primary tumor was significantly lower than that of FDG SUV [3.5 ± 3.6 vs 8.9 ± 6.5, P < 0.001 (two-tailed Wilcoxon signed rank test)].
N Staging
A total of 164 nodal groups were sampled. Of these, 11 nodal groups proved to be positive for malignancy in 7 of 34 patients.
On the basis of FLT PET data, hilar and mediastinal lymph node involvement was correctly determined in 29 of 34 patients (overstaging in two patients; understaging in three patients), whereas determination of lymph node involvement based on FDG PET data resulted in 25 correctly staged cases (overstaging in six patients; understaging in three patients; Table 1, Figs. 1 and 2).
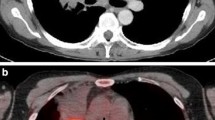
CT and PET images obtained in a 66-year-old woman with pleomorphic carcinoma of right upper lobe (Patient No. 34). a CT image shows low density tumor in right upper lobe and 8-mm (short-axis diameter) lymph node in right paratracheal area. b FDG coronal and c transverse PET images and d FLT coronal and e transverse PET images show peripheral increased metabolism in the primary tumor (arrowheads) and focally increased metabolism in N2 disease (arrows). Histopathologic analysis revealed mediastinal lymph node involvement in this case
CT and PET images obtained in a 57-year-old man with squamous cell carcinoma of right lower lobe (Patient No. 19). a CT image at level of pulmonary artery shows low density tumor in right lower lobe. Peripheral increased metabolism in the primary tumor (arrowheads) is shown in b FDG coronal and c FLT coronal PET images. d CT image at level of aortic arch shows 10-mm (short-axis diameter) lymph node in right paratracheal area. Focally increased metabolism in N2 disease is shown in e FDG coronal and f transverse PET images (arrows), but not in g FLT coronal and h transverse PET images. The false-positive FDG PET finding was corrected on the basis of FLT PET findings. Histopathologic analysis did not reveal mediastinal lymph node involvement in this case
Sensitivity, specificity, positive predictive, negative predictive, and accuracy values for N staging on a per-patient basis and on a per-nodal-station basis are summarized in Table 2. The specificity, positive predictive value and accuracy of FLT PET on a per-patient basis and the positive predictive value on a per-nodal-station basis were higher than the corresponding values for FDG PET, although the differences were not statistically significant (P > 0.1 for all comparisons).
M Staging
Overall, three patients had unsuspected metastatic disease (one lung metastasis, one muscle metastasis and one adrenal metastasis) which was not detected on previous examinations. Verification of distant metastatic disease was accomplished with biopsy in two patients (one lung metastasis and one muscle metastasis) and with radiologic follow-up (follow-up period, 124 days) in one patient (adrenal metastasis). FLT PET showed two sites suggestive of metastases: one muscle (true positive) and one adrenal (true positive). FDG PET showed five sites suggestive of metastases: three colons (false positive), one muscle (true positive) and one adrenal (true positive).
Additional findings
In one patient, both FLT PET and FDG PET depicted primary colon carcinoma that was not diagnosed with previous examinations.
Discussion
The present study has demonstrated that FLT PET in NSCLC shows better (although not statistically significant) specificity, positive predictive value and accuracy, on a per-patient basis and the positive predictive value on a per-nodal-station basis, for N staging than FDG PET. However, FDG PET was found to have higher sensitivity for depiction of primary tumor than FLT PET.
Inherent limitations of PET include its failure to depict anatomic landmarks and its limited spatial resolution, which restrict its use for assessing tumor staging. FDG PET has been shown to be substantially more sensitive and specific in the detection and characterization of metastases to mediastinal lymph nodes [7]. According to one report involving esophageal cancer staging, however, FDG PET is substantially less specific than CT for depicting lymph node metastasis, especially in regions of granulomatous disease [23]. Increased glucose uptake in a benign node can be caused by either reactive hyperplasia or granulomatous inflammation, which may be indistinguishable from malignancy. FDG accumulates in inflammatory cells such as activated granulocytes and macrophages and in some benign tumors because these cells require glucose as their substrate for energy production [9, 24]. Therefore, a more specific tracer that does not show uptake in inflammatory tissues might be useful.
In the search for more cancer-specific tracers, FLT was introduced as a proliferation tracer [12]. FLT permeates the cell membrane by facilitated diffusion [25] and is phosphorylated by the S-phase-specific enzyme TK1, which leads to intracellular trapping [25, 26]. Although the detailed uptake mechanism of FLT is still unknown, a pilot study demonstrated increased FLT uptake in tumor tissue [12]. In a rodent model of inflammation, van Waarde et al. recently demonstrated intense uptake of FDG but not of FLT in turpentine-induced muscle inflammation, further indicating a higher specificity of FLT [27].
With respect to primary tumor depiction, FLT PET is limited by its low sensitivity (67%), compared with FDG PET (94%) as indicated in the present study. All ten primary tumors, which showed false-negative findings on FLT PET and true-positive findings on FDG PET, were adenocarcinomas with mixed subtype (papillary adenocarcinoma + bronchioloalveolar carcinoma). This histological subtype of adenocarcinoma is known to have a better prognosis than other types of NSCLC [28]. One of the reasons for these false-negative findings may be explained by a low cell turnover. In the present study, the mean FLT SUV in primary tumors was significantly lower than the mean FDG SUV. This finding is consistent with a number of recent studies [14, 16–19]. The lower level of uptake probably increases the detection limit, making it more difficult to visualize the lesion.
The present study showed that FLT PET had low sensitivity (57%) for N staging. Recent studies have also reported lower sensitivity for N staging (33–56%) [16, 18, 19]. However, FLT PET had good specificity (93%) and accuracy (85%; FDG PET had 78 and 74%, respectively) and moderate positive predictive value (67%; FDG PET had 36%), on a per-patient basis for N staging although there was no significant difference between these two modalities. Recent studies also show higher specificity (98–100%) for N staging [16, 19]. Although the positive predictive value of FLT PET was higher than that of FDG PET, the differentiation between malignancy and focally increased metabolism caused by an inflammatory lymph node reaction remains challenging. Increased FLT uptake in inflammation and normal tissue has also been reported by Cobben et al. [29] evaluating FLT PET for staging of laryngeal cancer. These finding may appear to be related to proliferation of lymphocytes and nonspecific increase in the accumulation of FLT due to increased perfusion and vascular permeability, although the detailed uptake mechanism of FLT is still unknown. However, only a small number of patients have been examined in the present and previously reported studies. Larger patient populations need to be examined to determine the diagnostic accuracy of FLT PET for N staging.
With respect to M staging, both FLT PET and FDG PET detected the two true-positive lesions. PET is known to have poor sensitivity to small (less than 1 cm) pulmonary metastasis [30], which resulted in false-negative results for one patient in the present study. Furthermore, only FDG PET showed three false-positive results owing to physiological bowel activity and/or inflammation. However, our data do not show a clear-cut result for M staging because of small number of metastatic lesions.
Most PET studies carried out today in the field of oncology involve the acquisition of whole-body scans, the majority of which are carried out in the two-dimensional (2D) mode. However, recently, with the advent of new detector technology and the implementation of model-based scatter correction techniques in the majority of commercial PET systems, there is an increasing interest in the clinical performance of three-dimensional (3D) studies for whole-body PET imaging. 3D PET leads to higher sensitivity but is more subject to artifacts caused by random and scattered coincidence events [31]. However, noise equivalent count rates for a 3D scan of the thorax after injection of low activity of FDG are similar to a 2D scan after injection of high activity of FDG [32], and that image contrast is not significantly different [33]. To address these issues, we are planning further studies to clarify the suitability of this protocol.
There are limitations in our study. PET images did not correlate with corresponding CT images. When no tumor-related radioactivity was discernible at visual analysis, the case was assigned a SUV of 0. The coregistration of CT and PET images or integrated PET/CT devices may help to improve some diagnostic problems including SUV analysis.
Because of its low sensitivity, FLT PET does not appear to be capable of replacing FDG PET for preoperative staging in NSCLC. Nevertheless, the significant correlation between FLT uptake and tumor cell proliferation, which has now been confirmed in several studies [14, 15], suggests future clinical trials to evaluate the use of FLT PET for assessing changes of tumor cell proliferation during therapy. Furthermore, it will also be important to study whether FLT uptake is correlated with patient survival, as several studies have reported that the proliferation index is a prognostic factor in NSCLC [34–37].
Conclusion
Our evaluation of preoperative staging in NSCLC using FLT PET showed better (although not statistically significant) specificity, positive predictive value and accuracy on a per-patient basis for N staging than FDG PET. On the other hand, FDG PET was found to have higher sensitivity for depiction of primary tumor than FLT PET.
References
Tanaka F, Yanagihara K, Otake Y, Miyahara R, Kawano Y, Nakagawa T, et al. Surgery for non-small cell lung cancer: postoperative survival based on the revised tumor-node-metastasis classification and its time trend. Eur J Cardiothorac Surg 2000;18:147–55.
Barker JM, Silvestri GA. Lung cancer staging. Curr Opin Pulm Med 2002;8:287–93.
McLoud TC, Bourgouin PM, Greenberg RW, Kosiuk JP, Templeton PA, Shepard JA, et al. Bronchogenic carcinoma: analysis of staging in the mediastinum with CT by correlative lymph node mapping and sampling. Radiology 1992;182:319–23.
Scott WJ, Gobar LS, Terry JD, Dewan NA, Sunderland JJ. Mediastinal lymph node staging of non-small-cell lung cancer: a prospective comparison of computed tomography and positron emission tomography. J Thorac Cardiovasc Surg 1996;111:642–8.
Rohren EM, Turkington TG, Coleman RE. Clinical applications of PET in oncology. Radiology 2004;231:305–32.
Demura Y, Tsuchida T, Ishizaki T, Mizuno S, Totani Y, Ameshima S, et al. 18F-FDG accumulation with PET for differentiation between benign and malignant lesions in the thorax. J Nucl Med 2002;44:540–8.
Gould MK, Kuschner WG, Rydzak CE, Maclean CC, Demas AN, Shigemitsu H, et al. Test performance of positron emission tomography and computed tomography for mediastinal staging in patients with non-small-cell lung cancer: a meta-analysis. Ann Intern Med 2003;139:879–92.
Yamamoto Y, Nishiyama Y, Monden T, Sasakawa Y, Ohkawa M, Gotoh M, et al. Correlation of FDG-PET findings with histopathology in the assessment of response to induction chemoradiotherapy in non-small cell lung cancer. Eur J Nucl Med Mol Imaging 2006;33:140–7.
Shreve PD, Anzai Y, Wahl RL. Pitfalls in oncologic diagnosis with FDG PET imaging: physiologic and benign variants. Radiographics 1999;19:61–77.
Langen KJ, Braun U, Rota Kops E, Herzog H, Kuwert T, Nebeling B, et al. The influence of plasma glucose levels on fluorine-18-fluorodeoxyglucose uptake in bronchial carcinomas. J Nucl Med 1993;34:355–9.
Shields AF, Larson SM, Grunbaum Z, Graham MM. Short-term thymidine uptake in normal and neoplastic tissues: studies for PET. J Nucl Med 1984;25:759–64.
Shields AF, Grierson JR, Dohmen BM, Machulla HJ, Stayanoff JC, Lawhorn-Crews JM, et al. Imaging proliferation in vivo with [F-18]FLT and positron emission tomography. Nat Med 1998;4:1334–6.
Rasey JS, Grierson JR, Wiens LW, Kolb PD, Schwartz JL. Validation of FLT uptake as a measure of thymidine kinase-1 activity in A549 carcinoma cells. J Nucl Med 2002;43:1210–7.
Buck AK, Halter G, Schirrmeister H, Kotzerke J, Wurziger I, Glatting G, et al. Imaging proliferation in lung tumors with PET: 18F-FLT versus 18F-FDG. J Nucl Med 2003;44:1426–31.
Vesselle H, Grierson J, Muzi M, Pugsley JM, Schmidt RA, Rabinowitz P, et al. In vivo validation of 3′deoxy-3′-[18F]fluorothymidine ([18F]FLT) as a proliferation imaging tracer in humans: correlation of [18F]FLT uptake by positron emission tomography with Ki-67 immunohistochemistry and flow cytometry in human lung tumors. Clin Cancer Res 2002;8:3315–32.
Buck AK, Hetzel M, Schirrmeister H, Halter G, Moller P, Kratochwil C, et al. Clinical relevance of imaging proliferative activity in lung nodules. Eur J Nucl Med Mol Imaging 2005;32:525–33.
Dittmann H, Dohmen BM, Paulsen F, Eichhorn K, Eschmann SM, Horger M, et al. [18F]FLT PET for diagnosis and staging of thoracic tumours. Eur J Nucl Med Mol Imaging 2003;30:1407–12.
Cobben DC, Elsinga PH, Hoekstra HJ, Suurmeijer AJ, Vaalburg W, Maas B, et al. Is 18F-3′-Fluoro-3′-Deoxy-L-Thymidine Useful for the Staging and Restaging of Non-Small Cell Lung Cancer? J Nucl Med 2004;45:1677–82.
Yap CS, Czernin J, Fishbein MC, Cameron RB, Schiepers C, Phelps ME, et al. Evaluation of thoracic tumors with 18F-Fluorothymidine and 18F-Fluorodeoxyglucose-positron emission tomography. Chest 2006;129:393–401.
AJCC cancer staging manual. 6th ed. New York, NY: Springer, 2002; pp 165–77.
Machulla HJ, Blocher A, Kuntzsch M, Grierson JR. Simplified labeling approach for synthesizing 3′-deoxy-3′-[18F]fluorothymidine ([18F]FLT). J Radioanal Nucl Chem 2000;24:843–6.
Mountain CF, Dresler CM. Regional lymph node classification for lung cancer staging. Chest 1997;111:1718–23.
Yoon YC, Lee KS, Shim YM, Kim BT, Kim K, Kim TS. Metastasis to regional lymph nodes in patients with esophageal squamous cell carcinoma: CT versus FDG PET for presurgical detection-prospective study. Radiology 2003;227:764–70.
Kubota R, Yamada S, Kubota K, Ishiwata K, Tamahashi N, Ido T. Intratumoral distribution of fluorine-18-fluorodeoxyglucose in vivo: high accumulation in macrophages and granulation tissues studied by microautoradiography. J Nucl Med 1992;33:1972–80.
Mier W, Haberkorn U, Eisenhut M. [18F]FLT; portrait of a proliferation marker. Eur J Nucl Med Mol Imaging 2002;29:165–9.
Grierson JR, Shields AF. Radiosynthesis of 3′-deoxy-3′-[18F]fluorothymidine: [18F]FLT for imaging of cellular proliferation in vivo. Nucl Med Biol 2000;27:143–56.
van Waarde A, Cobben DC, Suurmeijer AJ, Maas B, Vaalburg W, de Vries EF, et al. Selectivity of 18F-FLT and 18F-FDG for differentiating tumor from inflammation in a rodent model. J Nucl Med 2004;45:695–700.
Higashi K, Ueda Y, Yagishita M, Arisaka Y, Sakurai A, Oguchi M, et al. FDG PET measurement of the proliferative potential of non-small cell lung cancer. J Nucl Med 2000;41:85–92.
Cobben DC, van der Laan BF, Maas B, Vaalburg W, Suurmeijer AJ, Hoekstra HJ, et al. 18F-FLT PET for visualization of laryngeal cancer: comparison with 18F-FDG PET. J Nucl Med 2004;45:226–31.
Gould MK, Maclean CC, Kuschner WG, Rydzak CE, Owens DK. Accuracy of positron emission tomography for diagnosis of pulmonary nodules and mass lesions. JAMA 2001;285:914–24.
Wienhard K, Eriksson L, Grootoonk S, Casey M, Pietrzyk U, Heiss WD. Performance evaluation of the positron scanner ECAT EXACT. J Comput Assist Tomogr 1992;16:804–13.
Lartizien C, Comtat C, Kinahan PE, Ferreira N, Bendriem B, Trebossen R. Optimization of injected dose based on noise equivalent count rates for 2- and 3-dimensional whole body PET. J Nucl Med 2002;43:1268–78.
Raylman R, Kison P, Wahl RL. Capabilities of two- and three-dimensional FDG-PET for detecting small lesions and lymph nodes in the upper torso: a dynamic phantom study. Eur J Nucl Med 1999;26:39–45.
Costa A, Silvestrini R, Mochen C, Leguaglie C, Boracchi P, Faranda A, et al. P53 expression, DNA ploidy and S-phase cell fraction in operable locally advanced non-small-cell lung cancer. Br J Cancer 1996;73:914–9.
Puglisi F, Minisini AM, Aprile G, Barbone F, Cataldi P, Artico D, et al. Balance between cell division and cell death as predictor of survival in patients with non-small-cell lung cancer. Oncology 2002;63:76–83.
Dosaka-Akita H, Hommura F, Mishina T, Ogura S, Shimizu M, Katoh H, et al. A risk-stratification model of non-small cell lung cancers using cyclin E, Ki-67, and ras p21: different roles of G1 cyclins in cell proliferation and prognosis. Cancer Res 2001;61:2500–4.
Pence JC, Kerns BJ, Dodge RK, Iglehart JD. Prognostic significance of the proliferation index in surgically resected non-small-cell lung cancer. Arch Surg 1993;128:1382–90.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamamoto, Y., Nishiyama, Y., Kimura, N. et al. Comparison of 18F-FLT PET and 18F-FDG PET for preoperative staging in non-small cell lung cancer. Eur J Nucl Med Mol Imaging 35, 236–245 (2008). https://doi.org/10.1007/s00259-007-0613-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-007-0613-0