Abstract
The benthic foraminiferal abundance and diversity for the last ~145 kyr at ODP Hole 723A in the northwestern Arabian Sea have been analyzed to better understand the factors responsible for the changes in the strength of Oxygen Minimum Zone (OMZ). The decline in the abundance of dysoxic taxa and more diverse fauna during interstadials of MIS 5 (5.5 and 5.1) along with higher percentages of G. bulloides reveal that the increased advection of well-oxygenated southern deep waters enhanced the rate of remineralization of sinking organic matter and also reduced the strength of OMZ. The improved supply of organic matter to the sea floor due to southwest summer monsoon linked higher surface productivity between 114 and 108 ka and 102 and 92 ka enhanced the decay of organic matter utilizing more oxygen at intermediate depths which resulted in the development of strong OMZ. The increased inflow of well-oxygenated Sub-Antarctic Mode and Antarctic Intermediate Waters (SAMW-AAIW) and significantly reduced or negligible outflow of low oxygen Red Sea Water (RSW) in the northwestern Arabian Sea developed relatively weak OMZ and better bottom water oxygenation during early Holocene. The low abundance of G. bulloides indicates decline in the surface water productivity due to weaker southwest summer monsoon during late Holocene while more outflow of oxygen poor RSW at intermediate depths and no further inflow of well oxygenated SAMW-AAIW beyond 5°N relatively strengthens the OMZ in the northwestern Arabian Sea.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Study of the Quaternary deep-sea benthic foraminifera provides significant information about the climatically induced changes in deep-water environment. The Arabian Sea sediments have well preserved faunal and geochemical record of changes in the Indian Ocean monsoon and its influence on surface water productivity, delivery of organic matter to the seafloor and deep-sea oxygenation. There is general consensus regarding the sign of southwest summer monsoon change in several records of productivity; however, many studies show a relatively intricate nature of phase relation of oceanic response to the changing monsoon pattern (Rostek et al. 1997; Almogi-Labin et al. 2000; Budziak et al. 2000; Schmiedl and Leuschner 2005).
The main source of sub-surface oxygen for both the Arabian Sea and Bay of Bengal is from the Southern Hemisphere. Antarctic Circumpolar Current (ACC) is mainly responsible for well oxygenated, intermediate water which flows northward and spreads throughout the Indian Ocean. The outflow of high salinity and low-oxygen Red Sea Water (RSW) and Persian Gulf Water (PGW) also influences the oxygenation of intermediate water mass of Arabian Sea (Jung et al. 2001). The changing global sea level is suggested to be responsible to control the formation of deep-water in the Red Sea and the amount of outflow of RSW at glacial–interglacial scales (Rohling and Zachariasse 1996). The Indonesian Throughflow (ITF) water is also suggested to influence the upper Indian Ocean and Arabian Sea waters at thermocline depth (Haines et al. 1999; Song et al. 2004).
Deep-sea benthic foraminiferal study in the Gulf of Aden reveals that intense NE winter monsoon increased the surface water productivity during glacial times (Almogi-Labin et al. 2000). The intervals of strong NE winter monsoon are related to the increased surface water productivity in the southeastern Arabian Sea which is linked to the precession-based maxima in ice volume (Rostek et al. 1997). The intensity and extent of OMZ during late Quaternary in the northeastern Arabian Sea is directly related to the surface water productivity and associated organic matter fluxes to the sea floor. The intervals of intensified OMZ are associated with the productivity maxima and enhanced organic matter supply to the sea floor (den Dulk et al. 2000), whereas very weak or absence of OMZ are related to the periods of low surface water productivity and deep winter mixing during intense NE winter monsoon (Reichart et al. 1998). Further studies suggest intimate relation of surface water productivity and OMZ with the monsoon variation on centennial- and millennial-scale which are linked to the variability in North Atlantic climate (Schulz et al. 1998; Leuschner and Sirocko 2000; Altabet et al. 2002; Gupta et al. 2003). The out of phase relationship of deepening of OMZ with intense SW summer monsoon in the western Arabian Sea has been explained by variation in the advection of oxygen-rich deep-water masses (Schmiedl and Leuschner 2005). Recently, Das et al. (2017) have undertaken a high resolution benthic foraminiferal study of Holocene sediment in the northwestern Arabian Sea and suggested significant role of intermediate water circulation in the variation of OMZ intensity. They also found decadal to centennial scale cyclicity in the OMZ intensity linked with the solar cycles. The present study provides a high resolution benthic foraminiferal record of a sediment core in the northwestern Arabian Sea over past ~145 kyr to understand the role of Indian Ocean monsoon system and intermediate water circulation in the variability of OMZ intensity.
2 Oceanographic settings
The present day Arabian Sea shows significant seasonal variations in the biogenic particle flux due to changing monsoon winds. The formation of strong pressure gradient due to differential heating between Asian continent and Southern Indian Ocean develops southwest summer monsoon winds from June to September and northeast winter monsoon winds from November to March (Webster 1987). The development of Ekman transport along the Oman margin due to intense SW summer monsoon winds driven by Findlater Jet causes upwelling of nutrient-rich subsurface water to the surface that increases the surface water productivity. The reversal of surface circulation during NE winter monsoon causes convective mixing and cooling of surface water which also increases the surface productivity primarily in the open-ocean (Bartolacci and Luther 1999).
The decay of large amount of organic matter in the water column produced by the enhanced rate of surface water productivity consuming higher amount of dissolved oxygen at intermediate water depths along with pronounced thermohaline stratification results into the development of a constant and expanded oxygen minimum zone (OMZ) between about 200 and 1200 m (Olson et al. 1993; Schulz et al. 1996). The main sources of intermediate waters in the Arabian Sea are: (a) well-oxygenated Sub-Antarctic Mode and Antarctic Intermediate Waters (SAMW-AAIW), (b) the Indonesian Intermediate Water (IIW), and (c) less oxygenated and more saline Persian Gulf Water (PGW) and Red Sea Water (RSW). A combination of SAMW-AAIW and IIW together as Indian Ocean Central Waters (IOCW) flows in to the Arabian Sea during the SW summer monsoon along the Somali Current (Boning and Bard 2009). The present day OMZ is described as the zone having dissolved oxygen <0.5 ml/l (Levin 2003), which can also be the result of semi-enclosed character of the northern Arabian Sea and supply of the North Indian Intermediate Water from its southern source (Wyrtki 1973; Olson et al. 1993). Additionally, the outflow of more saline and oxygen poor RSW and PGW that are confined mainly at a depth of ~800 m also strengthens the OMZ. Below the OMZ, the Arabian Sea is influenced by relatively more saline and oxygen-rich North Indian Deep Water (NIDW) approximately between 1200 and 3800 m of depth. Biological and geochemical processes at the sea floor of deep Arabian Sea mainly depend upon the trophic level and deep water oxygen content (Graf 1989; Pfannkuche and Lochte 2000). Oxygenation is the primary factor to control the distribution of the less diverse benthic fauna within the depth range of OMZ in the eutrophic condition (Jannink et al. 1998; Gooday et al. 2000).
3 Materials and methods
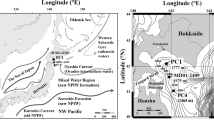
A deep-sea benthic foraminiferal study was based on samples of ODP Hole 723A (lat. 18°03.079′N; long. 57°36.566′E and water depth 807.8 m) situated very close to the Oman margin around mid-depth of present day OMZ in the northwestern Arabian Sea (figure 1) where much intense winds during summer monsoon cause increased upwelling and surface water productivity. Benthic foraminiferal census data were produced by analyzing a total of 180 samples. Samples were collected at an average of 15 cm interval from 27.2 m long core with an average time interval of about 800 years. Each sample of 10 cm3 volume was processed by treating with 10% calgon solution overnight. The processed samples were wet sieved with Tyler sieves over 125 µm size fraction and then dried at 50°C. The washed samples over 125 µm size fraction were divided by using micro splitter into an aliquot of about 300 benthic foraminiferal specimens which was used to generate census data. A total of 176 benthic foraminiferal species representing 73 genera were recorded at Hole 723A (Annexure 1). The relative abundance of significant benthic foraminiferal species and groups were presented as the percent of total population.
The faunal census data was used to determine various diversity indices such as Simple diversity (S), Shannon Index (H), Equitability (E) and Fisher’s Alpha Index (a) with the help of PAST software (Version 3.25) (Hammer et al. 2001). The mathematical expressions for these indices are:
Simple diversity (S) is the total number of species in each sample.
Shannon index (entropy) (H): This diversity index gives consideration to the number of specimens and number of species both. The value of this diversity index remains zero for community with only single species, whereas communities with several taxa, each having a few specimens show higher values.
where n is the number of individuals.
Equitability: This index measures the evenness with which individuals are distributed among the recorded species. This index can be acquired by dividing Shannon diversity with the logarithm of number of taxa.
Fisher's alpha: This diversity index is defined as coefficient a of the formula
where S is the number of species, n is the number of specimens and a is the Fisher's alpha index.
The dysoxic and oxic benthic foraminiferal species were identified on the basis of Hermelin and Schimmield (1990), Kaiho (1994), Schumacher et al. (2007), Caulle et al. (2014) Singh et al. (2015) and Das et al. (2017) in order to explain bottom water oxygen condition (Annexure 2). The dysoxic species are distributed in the oxygen range between 0.1 and 0.3 ml/l (Kaiho 1994) which include infaunal, thin-walled, elongate and flattened taxa. This group mainly includes Bolivinids, Globobuliminids, Dentalina spp. and Fursenkoina spp. which dominantly occur in extremely oxygen depleted (< 1 ml/l) deep-sea environment (Bernhard and Sen Gupta 1999). Benthic species which thrive in oxic conditions (> 1.5 ml/l) include miliolids and taxa having relatively larger test size with thick wall and planoconvex planispiral and trochospiral shapes (Kaiho 1994). The above oxic fauna do not occur in the deep sea environment with <1 ml/l of dissolved oxygen.
The relative abundance data of Globigerina bulloides was produced by counting about 300 planktic foraminiferal specimens from over 125 µm size fraction. We also used the benthic foraminiferal δ18O and δ13C data from Hole 723A of Niitsuma et al. (1991). The age of top 7.4 m section is based on the AMS 14C dates given by Naidu and Malmgren (1996). For rest of the section, we used the time–depth relationship in the oxygen isotope stratigraphy provided by Anderson and Prell (1993) which was recalculated following Lisiecki and Raymo (2005).
4 Results
The relative abundance of benthic foraminiferal species and groups, benthic δ13C and δ18O values and species diversity show distinct variations during past ~145 kyr at Hole 723A (figures 2–5). The abundance patterns of dominant benthic foraminiferal species and/or group do not exhibit any close relation with glacial and interglacial intervals (figures 2, 3), whereas G. bulloides occurs with relatively higher abundance during interglacial intervals (figure 5). During interglacial MIS 5 between 114–108 ka and 102–92 ka Bolivina spp. (mainly B. spathulata) and dysoxic fauna increased their abundance which corresponds with less diverse fauna and almost absence of miliolids (figures 2, 3, 5). The interstadials (5.5 and 5.1) and early Holocene were the periods of distinct increase in the values of species diversity and equitability and decline in the dysoxic taxa along with relatively higher abundance of G. bulloides and low values of δ13C (figure 5). The relative abundances of Fursenkoina spp., Bulimina spp. and Bolivina spp. show their decline during interstadials 5.5 and 5.1 (figure 3). The early Holocene was also characterized by distinct increase in the population of shallow infaunal species, H. balthica and intermediate to deep infaunal species, B. aculeata from the lower margin of OMZ (figure 2). Three distinct peaks of abundance of oxic taxa and miliolids along with increase in species diversity at around 130, 65 and 14 ka were noticed which correspond with relatively low abundance of G. bulloides (figures 3, 5).
Temporal variation in relative abundances of Oxic taxa (%), Dysoxic taxa (%), Fisher’s alpha (a), Globigerina bulloides (%); benthic δ13C and δ18O at ODP Hole 723A (Niitsuma et al. 1991) (even numbered grey horizontal bars represent glacial stages).
Except two short intervals of 30–25 and 42–38 ka, the values of species diversity remained typically low during most of the time between ~55 and 15 ka (MIS 3 and 2) which also corresponds with the increased population of dysoxic taxa (figure 5). The low-oxygen taxa such as Fursenkoina spp. and Bolivina spp. along with Uvigerina spp. also increased their abundance during this interval (figure 3). However, Bulimina spp. also shows moderately higher abundance during 55–15 ka with substantial increase in its abundance between 65 and 60 ka (figure 3). The population of deep infaunal and dysoxic taxa such as Fursenkoina reagani, Bolivina pygmaea and Bulimina arabiensis remained high between ~55 and 15 ka (figure 2). This was also an interval of lower benthic δ13C values (figure 5).
The late Holocene interval is characterized by relatively higher abundance dysoxic taxa and less diverse and equitable fauna (figure 5). The population of Fursenkoina spp., Uvigerina spp. and Uvigerina peregrina increased during this interval (figures 2, 3). However, the relative abundance of G. bulloides shows decline during the late Holocene (figure 5).
5 Discussion
5.1 Ecological significance of benthic foraminiferal taxa
The relative abundance pattern of ecologically significant benthic foraminiferal species and faunal diversity were analyzed to better explain the changes in the trophic level and bottom water oxygenation through time. The significant benthic foraminiferal groups/species (with abundance of 15% or more in at least two samples) recorded in this study are Bolivina spp., Bulimina spp., Fursenkoina spp., Uvigerina spp., Bulimina aculeata, Bulimina arabiensis, Bolivina pygmaea, Bolivina spathulata, Fursenkoina reagani, Uvigerina peregrina and Hyalinea balthica. The past changes in the dissolved oxygen conditions in the deep sea environment can be efficiently explained by analyzing the relative abundances of low-oxygen taxa.
The benthic foraminiferal assemblages mainly consisted of species of Bulimina, Bolivina, Uvigerina and Fursenkoina are abundantly distributed in the deep-sea environments characterized by less oxygenated bottom waters and increased organic matter influx to the sea floor (e.g., Lutze and Coulbourn 1984; Caralp 1989; Sen Gupta and Machain-Castillo 1993). Species of Bolivina, being deep infaunal taxa, prefer to live within the warm bottom waters of OMZ in the equatorial Indian Ocean with increased organic matter supply (Singh et al. 2012). Bolivina spathulata, a deep infaunal species occurs abundantly in sediments with increased supply of organic matter within low oxygen bottom waters (Corliss 1985; Corliss and Chen 1988; Kaiho 1994; Jorissen 1999). Both Bolivina pygmaea and Fursenkoina reagani are characterized by small, thin wall, elongate and flattened tests showing close similarity with infaunal, dysoxic taxa of Kaiho (1994) on the continental margin of the high productivity areas.
The infaunal species of Bulimina show their increased abundance in the bottom waters with higher trophic level and/or less oxygenated condition (Murray 1991). The higher abundances of Bulimina spp. were recorded from the upper part of OMZ in the Arabian Sea with characteristically high organic matter influx to the sea floor (Hermelin and Schimmield 1990). Bulimina aculeata is commonly an intermediate to deep infaunal species (Mackensen et al. 2000) that is adapted to changing oxygen content and trophic condition at the bottom and can survive both on fresh and degraded organic matter (Linke and Lutze 1993). Abundance of this species is associated with present day high surface water productivity in the Gulf of Aden and during past interglacial intervals (Almogi-Labin et al. 2000). The occurrence of B. aculeata in the Arabian Sea is not associated with the organic matter supply to the sea floor (Hermelin and Shimmield 1990), but its distribution by the surface water productivity and organic carbon flux in the south Atlantic and south China Sea (Mackensen et al. 1995; Jannink et al. 1998). In the recent sediments of northwestern Arabian Sea, B. aculeata abundantly occurs in the regions of very high surface water productivity (De and Gupta 2010). The increased population of Bulimina arabiensis in the late Quaternary sediments of the eastern Arabian Sea is suggested to indicate high organic matter supply to the sea floor and less oxygenated bottom water condition (Bharti and Singh 2013).
The genus Fursenkoina inhabits a very less oxygenated benthic environment of shallow eastern Arabian Sea (Mazumdar and Nigam 2014). In oxygen-depleted benthic environments, miliolids show their absence or rare occurrence (Mullineaux and Lohmann 1981; Nolet and Corliss 1990; Jannink et al. 1998; Jorissen 1999) and can be used as an important marker of dissolved oxygen in the bottom water. Increased abundance of miliolids in the northern Arabian Sea is considered as an evidence of increased bottom water oxygenation and thus, this group can be used as very significant indicator of oxygen and regarded as important proxy for the reconstruction of changes in the bottom and pore water oxygen (den Dulk et al. 2000).
Uvigerina peregrina is a shallow infaunal species which generally inhabits in topmost 1-cm sediment with its highest occurrence in the lower part of OMZ with high organic matter influx (Hermelin and Shimmield 1990; Jannink et al. 1998; Schmiedl 2000; Schumacher et al. 2007). This species is recorded in the recent sediments within the OMZ of northwestern Arabian Sea where surface water productivity remains very high and bottom water is much depleted in oxygen (De and Gupta 2010). Several other studies suggested the abundant occurrence of U. peregrina in fine-grained sediments with increased organic matter content, but could not show clear evidence of its occurrence in oxygen deficient deep-sea environment (e.g., Lutze and Coulbourn 1984; Rathburn and Corliss 1994). The occurrence of this species near the base or even below the OMZ indicates its preference for relatively less food supply or highly degraded organic matter (Jannink et al. 1998). The intervals of abundant occurrence of U. peregrina are explained by the increase in depth of lower boundary of OMZ in the western Arabian Sea (Schmiedl and Leuschner 2005).
Hyalinea balthica is recorded as shallow infaunal taxa from the lower part of Arabian Sea OMZ where oxygen depletion is not much severe (Hermelin and Shimmield 1990). The increased occurrence of this species is associated with the higher diversity reflecting less stressed ecological condition (Schmiedl et al. 2000). Hyalinea balthica is a suboxic species found to be associated with moderate supply of organic matter and less intense OMZ in the northwestern Arabian Sea (Das et al. 2017).
5.2 Changes in the strength of OMZ
The deep-sea oxygenation plays a significant role in the benthic environments of the Arabian Sea. The paleoceanographic studies reveal that the surface water productivity and subsurface ventilation in the Arabian Sea have shown noticeable changes in the past. Earlier studies suggested several micropaleontological and geochemical evidences of changing depth and strength of OMZ in the Arabian Sea (e.g., den Dulk et al. 1998, 2000; Reichart et al. 1998; Almogi-Labin et al. 2000; Gupta et al. 2008; Rai and Das 2011). An increased occurrence of deep infaunal taxa and more organic matter supply to the sea floor at intermediate depths in the northern Arabian Sea indicate oxygen level at its minimum within the OMZ (Reichart et al. 1998; den Dulk et al. 2000). The distinct increase in the relative abundance of deep infaunal taxa having preference for oxygen depleted condition at least once during last 225,000 years suggests deepening of OMZ base up to or below 1470 m in the northern Arabian Sea (den Dulk et al. 2000). Decreased northward inflow of NADW in the Arabian Sea during glacial intervals might be responsible for the OMZ expansion, whereas relatively more inflow of oxygenated deep water results into almost present position for the OMZ base during interglacial times (den Dulk 2000). On the basis of benthic foraminiferal evidences, Schmiedl and Leuschner (2005) recognized repeated deepening of base of western Arabian Sea OMZ equal to or below 1800 m during past 190 kyr.
In general, the dominant occurrence of low-oxygen benthic foraminiferal assemblages mainly consisting of Bolivina spathulata, B. pygmaea, Bulimina arabiensis and Fursenkoina reagani during most of the last ~145 kyr suggests that except few short intervals the studied site almost continuously remained located within the middle depth of OMZ. However, some intervals of variation in the faunal abundance and diversity reflect shifts in the bottom water oxygen level at intermediate depths in the northwestern Arabian Sea. The abundant occurrence of Bolivina spp. (mainly B. spathulata) and dysoxic fauna along with less diverse fauna and almost absence of miliolids during interglacial MIS 5 particularly between 114–108 ka and 102–92 ka reveal relatively strong OMZ due to poor ventilation at intermediate depths. The organic matter supply to the sea floor during these intervals remained high due to increased monsoonal upwelling and higher surface water productivity as evidenced by the higher abundance of G. bulloides. Poor ventilation in the intermediate water is because of the utilization of oxygen due to the decay of organic matter which reduces the dissolved oxygen level and strengthens the OMZ.
The benthic foraminiferal record at ODP Hole 723A during interstadials 5.5 and 5.1 and early Holocene shows decreased percentages of dysoxic taxa, increased abundance of oxic taxa and higher values of species diversity suggest relatively better oxygenation of bottom water at intermediate depths during this time. Decreased abundance of B. spathulata along with moderate occurrence of H. balthica during interstadials 5.5 and 5.1 also reflects better oxygenation at the intermediate depths during this time. On the other hand, abundant occurrences of G. bulloides during these intervals indicate increase in the organic matter supply to the sea floor due to intense southwest monsoon led upwelling (Naidu and Malmgren 1996; Gupta et al. 2003). Increased abundance of U. peregrina during this time also indicates increased supply of organic matter to the sea floor. During interstadials, occurrence of increased abundances of epifaunal and shallow infaunal taxa along with higher faunal diversity and abundance of high oxygen indicator species in the western Arabian Sea suggest more advection of oxygen-rich NADW in the Circumpolar and Indian Ocean (Schmiedl and Leuschner 2005). The increased advection of well-oxygenated deep waters during interstadials was responsible for the enhanced rates of remineralization of organic matter settled at the sea floor and significant reduction in burial strength of organic matter in the Arabian Sea sediment (Schmiedl and Leuschner 2005). We also believe that in the study area, the enhanced lateral advection of southern source, oxygen-rich water at intermediate depths during interstadials 5.5 and 5.1 reduced the strength of OMZ due to regular oxygen replenishment.
Recently, in a high resolution benthic foraminiferal study of the northwestern Arabian Sea, Das et al. (2017) suggested stable deep-sea environment with stable faunal diversity during the early Holocene which coincided with strong southwest monsoon winds increasing the primary productivity and organic carbon influx. However, instead of increased abundance of dysoxic taxa, they recorded the occurrence of repetitive pulses of oxic taxa reflecting oxygenated bottom waters during intense southwest summer monsoon and increased influence of SAMW-AAIW in the northwestern Arabian Sea. We also recorded the low abundance of dysoxic taxa, increased occurrence of oxic and porcelaneous taxa along with more diverse fauna which coincided with increased abundance of G. bulloides during the early Holocene indicating relatively better deep-sea oxygenation and higher surface water productivity. Moreover, Gupta et al. (2008) suggested that the influence of increased production due to strong SW monsoon upwelling in the northwestern Arabian Sea during early Holocene was reduced by the enhanced Circumpolar Deep Water (CDW) admixture and resulted in to a weak OMZ.
Most of the duration between MIS 3 and MIS 2 (~55 to 15 ka) was characterized by the increased percentages of dysoxic taxa, Bulimina spp., Bolivina spp., Fursenkoina spp., B. pygmaea and F. reagani along with low values of species diversity representing strong OMZ with further decline of oxygen level at intermediate depths in the northwestern Arabian Sea. Uvigerina spp. associated with increased flux of organic matter to the sea floor shows moderately higher abundance during 55–42 ka and 30–15 ka. However, two short intervals of 42–38 ka and 30–25 ka were marked with decreased population of dysoxic taxa and more diverse benthic fauna along with low abundance of G. bulloides showing better oxygen condition and low surface productivity during this time. The reduced oxygen content in the deep-sea and deepening of the OMZ in the western Arabian Sea during ~50 to 15 ka was also reflected by the increased relative abundance of deep OMZ indicator benthic foraminifera and less diverse fauna (see figure 3 in Schmiedl and Leuschner 2005). The higher organic matter supply to the sea floor and/or reduced oxygenation of deep-sea were responsible for less oxygenated bottom water in the northern Arabian Sea during MIS 3 and 2 (den Dulk et al. 1998). A widespread increase in the surface water productivity between ~60 and ~15 ka was also identified in various parts of Arabian Sea utilizing different proxy records (e.g., Zahn and Pedersen 1991; Hermelin and Schimmield 1995; Rostek et al. 1997; Reichart et al. 1997; den Dulk et al. 1998, 2000). We suggest that the OMZ was possibly intensified due to enhanced organic matter supply to the sea floor and its decay consuming oxygen reduced the ventilation at the intermediate depths.
A prominent decline in the faunal diversity and equitability during late Holocene (last ~4 kyr) along with the relative increase in the population of dysoxic taxa indicates stronger OMZ in the northwestern Arabian Sea. This period is also characterized by the increased relative abundance of Fursenkoina spp., Uvigerina spp. and Uvigerina peregrina along with almost absence of oxic taxa and miliolids reflecting less oxygenated condition. However, low abundance of G. bulloides during late Holocene indicates less upwelling and surface water productivity due to weak southwest summer monsoon. The oxygen-rich SAMW-AAIW could not flow beyond 5°N due to increased outflow of more saline, low-oxygen RSW in the western Arabian Sea reducing the vertical mixing of water masses which resulted in the formation of better OMZ condition at intermediate depths (Das et al. 2017). The development of weaker southwest summer monsoon and an arid condition in the Indian region at ~4.2 ka was suggested to be responsible for the de-urbanization of Indus valley civilization (Dixit et al. 2014; Böll et al. 2015). However, the bottom water at deeper site GeoB3004 (1803 m water depth) behaved differently than at intermediate depth (ODP Hole 723A) in the western Arabian Sea where increased percentage of high O2 indicator benthic fauna reflects well-oxygenated deep water during the late Holocene (Schmiedl and Leuschner 2005). The outflow of oxygen-poor RSW settled around 1600 m off Somalia due to changes in the evapoprecipitation equilibrium in the Red Sea (Jung et al. 2001) and could not influence the oxygenation at deeper site in the western Arabian Sea.
6 Conclusions
The benthic foraminiferal record for the last ~145 kyr in the northwestern Arabian Sea suggests that changes in the upwelling and surface water productivity due to Indian Ocean monsoon system and outflow of oxygen poor RSW from the north are mainly responsible for the variations in the OMZ intensity. The strength of OMZ was noticeably reduced during warmest intervals of MIS 5 (5.5 and 5.1) due to increased lateral advection of well-oxygenated deep water and regular oxygen replenishment. The development of significantly strong OMZ during time intervals of 114–108 ka and 102–92 ka is perhaps linked to the increased oxygen utilization for the decay of large amount of organic matter fluxes due to increased southwest summer monsoon upwelling and surface water productivity. The higher inflow of oxygen-rich SAMW-AAIW towards the northwestern Arabian Sea and almost non-existence of the outflow of oxygen poor RSW during early Holocene reduced the effect of production due to intensified southwest monsoon upwelling that resulted into a weak OMZ. The increased outflow of low oxygen RSW and almost complete shutdown of inflow of oxygen-rich SAMW-AAIW intensified the OMZ during late Holocene while southwest summer monsoon remains weak.
References
Almogi-Labin A, Schmiedl G, Hemleben C, Siman-Tov R, Segl M and Meischner D 2000 The influence of the NE winter monsoon on productivity changes in the Gulf of Aden, NW Arabian Sea, during the last 530 ka as recorded by benthic foraminifera; Mar. Micropaleontol. 40 295–319.
Altabet M A, Higginson M J and Murray D W 2002 The effect of millennial-scale changes in Arabian Sea denitrification on atmospheric CO2; Nature 415 159–162.
Andersn D M and Prell W L 1993 A 300 kyr record of upwelling off Oman during the late Quaternary: Evidence of the Asian Southwest Monsoon; Paleoceanography 8(2) 193–208.
Bartolacci D M and Luther M E 1999 Patterns of covariability between physical and biological parameters in the Arabian Sea; Deep-Sea Res. Part II 46 1933–1964.
Bernhard J M and Sen Gupta B 1999 Foraminifera of oxygen depleted environment; In: Modern Foraminifera (ed.) Sen Gupta B, Kluwer Academic, Dordrecht, pp. 200–216.
Bharti S K and Singh A D 2013 Bulimina arabiensis, a new species of benthic foraminifera from the Arabian Sea; J. Foram. Res. 43 255–261.
Böll A, Schulz H, Munz P, Rixen T, Gaye B and Emeis K C 2015 Contrasting sea surface temperature of summer and winter monsoon variability in the northern Arabian Sea over the last 25 ka; Palaeogeogr. Palaeoclimatol. Palaeoecol. 426 10–21.
Boning P and Bard E 2009 Millennial/centennial-scale thermocline ventilation changes in the Indian Ocean as reflected by aragonite preservation and geochemical variations in Arabian Sea sediments; Geochim. Cosmochim. Acta 73(22) 6771–6788.
Budziak D, Schneide R R, Rostek F, Muller P J, Bard E and Wefer G 2000 Late Quaternary insolation forcing on total organic carbon and C37 alkenone variations in the Arabian Sea; Paleoceanography 15 307–321.
Caralp M H 1989 Abundance of Bulimina exilis and Melonis barleeanum: Relationship to the quality of organic matter; Geo-Mar. Lett. 9 37–43.
Caulle C, Koho K A, Mojtahid M, Reichart G J and Jorissen F J 2014 Live (Rose Bengal stained) foraminiferal faunas from the northern Arabian Sea: Faunal succession within and below the OMZ; Biogeosci. 11 1155–1175.
Corliss B H 1985 Microhabitats of benthic foraminifera within deep-sea sediments; Nature 314 435–438.
Corliss B H and Chen C 1988 Morphotype patterns of Norwegian Sea deep-sea benthic foraminifera and ecological implications; Geology 16 716–719.
Das M, Singh R K, Gupta A K and Bhaumik A K 2017 Holocene strengthening of the Oxygen Minimum Zone in the northwestern Arabian Sea linked to changes in intermediate water circulation or Indian monsoon intensity; Palaeogeogr. Palaeoclimatol. Palaeoecol. 483 125–135.
den Dulk M 2000 Benthic foraminiferal response to Late Quaternary variations in surface water productivity and oxygenation in the northern Arabian Sea; Geol. Ultraiectina 188 1–205.
den Dulk M, Reichart G J, Memon G M, Roelofs E M P, Zachariasse W J and van der Zwaan G J 1998 Benthic foraminiferal response to variations in surface water productivity and oxygenation in the northern Arabian Sea; Mar. Micropal. 35 43–66.
den Dulk M, Reichart G J, van Heys S, Zachariasse W J and Van der Zwaan G J 2000 Benthic foraminifera as proxies of organic matter flux and bottom water oxygenation? A case history from the northern Arabian Sea; Palaeogeogr. Palaeoclimatol. Palaeoecol. 161 337–359.
De S and Gupta A K 2010 Deep-sea faunal provinces and their inferred environments in the Indian Ocean based on distribution of recent benthic foraminifera; Palaeogeogr. Palaeoclimatol. Palaeoecol. 291 429–442.
Dixit Y, Hodell D A and Petrie C A 2014 Abrupt weakening of the summer monsoon in Northwest India ~4100 yr ago; Geology 42 339–342.
Gooday A J, Bernhard J M, Levin L A and Suhr S B 2000 Foraminifera in the Arabian Sea oxygen minimum zone and other oxygen deficient settings: Taxonomic composition, diversity, and relation to metazoan faunas; Deep Sea Res. Part II 47 25–54.
Graf G 1989 Benthic-pelagic coupling in a deep-sea benthic community; Nature 341 437–439.
Gupta A K, Anderson D M and Overpeck J T 2003 Abrupt changes in the Asian southwest monsoon during the Holocene and their links to the North Atlantic Ocean; Nature 421 354–357.
Gupta A K, Das M, Clemens S C and Mukherjee B 2008 Benthic foraminiferal faunal and isotopic changes as recorded in Holocene sediments of the Northwest Indian Ocean; Paleoceanography 23 PA2214, https://doi.org/10.1029/2007PA001546.
Haines M A, Fine R A, Luther M E and Ji Z 1999 Particle trajectories in an Indian Ocean model and sensitivity to seasonal forcing; J. Phys. Oceanogr. 29 584–598.
Hammer Ø, Harper D A T and Ryan P D 2001 PAST: paleontological statistics software package for education and data analysis; Palaeontol. Electron. 4(9), http://palaeo-electronica.org/2001_1/past/issue1_01.htm.
Hermelin J O R and Shimmield G B 1990 The importance of the oxygen minimum zone and sediment geochemistry in the distribution of Recent benthic foraminifera in the northwest Indian Ocean; Mar. Geol. 91 1–29.
Hermelin J O R and Shimmield G B 1995 Impact of productivity events on the benthic foraminiferal fauna in the Arabian Sea over the last 150,000 years; Paleoceanography 10 85–116.
Jannink N T, Zachariasse W J and van der Zwaan G J 1998 Living (Rose Bengal stained) benthic foraminifera from the Pakistan continental margin (northern Arabian Sea); Deep-Sea Res. Part I 45 1483–1513.
Jorissen F J 1999 Benthic foraminiferal successions across late Quaternary Mediterranean sapropels; Mar. Geol. 153 91–101, https://doi.org/10.1016/S0025-3227(98)00088-7.
Jung S J A, Ganssen G M and Davies G R 2001 Multi-decadal variations in the early Holocene outflow of Red Sea Water into the Arabian Sea; Paleoceanography 16 658–668, https://doi.org/10.1029/2000PA000592.
Kaiho K 1994 Benthic foraminiferal dissolved oxygen index and dissolved oxygen levels in the modern ocean; Geology 22 719–722.
Leuschner D C and Sirocko F 2000 The low latitude monsoon climate during Dansgaard–Oeschger cycles and Heinrich events; Quat. Sci. Rev. 19 243–254.
Levin L A 2003 Oxygen minimum zone benthos: Adaptation and community response to hypoxia; Oceanogr. Mar. Biol.: Ann. Rev. 41 1–45.
Linke P and Lutze G F 1993 Microhabitat preferences of benthic foraminifera – a static concept or a dynamic adaptation to optimize food acquisition; Mar. Micropaleontol. 20 215–234.
Lisiecki L E and Raymo M E 2005 A Pliocene–Pleistocene stack of 57 globally distributed benthic δ18O records; Paleoceanography 20 PA1003, https://doi.org/10.1029/2004PA001071.
Lutze G F and Coulbourn W T 1984 Recent benthic foraminifera from the continental margin of northwest Africa: Community structure and distribution; Mar. Micropaleontol. 8 361–401.
Mackensen A, Schmiedl G, Harloff J and Giese M 1995 Deep-sea foraminifera in the South Atlantic Ocean: Ecology and assemblage generation; Micropaleontology 41 242–258.
Mackensen A, Schumacher S, Radke J and Schmidt D N 2000 Microhabitat preferences and stable carbon isotopes of endo-benthic foraminifera: Clue to quantitative reconstruction of oceanic new production; Mar. Micropaleontol. 40 233–258.
Mazumdar A and Nigam R 2014 Bathymetric preference of four major genera of rectilinear benthic foraminifera within oxygen minimum zone in Arabian Sea off central west coast of India; J. Earth Syst. Sci. 123 633–639.
Mullineaux L S and Lohmann G P 1981 Late Quaternary Stagnations and recirculation of the eastern Mediterranean: Changes in the deep water recorded by fossil benthic foraminifera; J. Foram. Res. 11 20–39.
Murray J W 1991 Ecology and Palaeoecology of Benthic Foraminifera; Longman Scientific & Technical, Harlow, Essex, 397p.
Naidu P D and Malmgren B A 1996 A high-resolution record of late Quaternary upwelling along the Oman Margin, Arabian Sea based on planktonic foraminifera; Paleoceanography 11(1) 129–140.
Niitsuma N, Oba T and Okada M 1991 Oxygen and carbon isotope stratigraphy at site 723, Oman Margin; In: Proceedings ODP (eds) Prell W L et al., Scientific Results 117 321–341.
Nolet G J and Corliss B H 1990 Benthic foraminiferal evidence for reduced deep-water circulation during sapropel deposition in the eastern Mediterranean; Mar. Geol. 94 109–130.
Olson D B, Hitchcock G L, Fine R A and Warren B A 1993 Maintenance of the low oxygen layer in the central Arabian Sea; Deep Sea Res; Part II 40 673–685.
Pfannkuche O and Lochte K 2000 The biogeochemistry of the deep Arabian Sea: Overview; Deep Sea Res. Part II 47 2615–2628.
Rai A K and Das S S 2011 Late Quaternary changes in surface productivity and oxygen minimum zone (OMZ) in the northwestern Arabian Sea: Micropaleontologic and sedimentary record at ODP site 728A; J. Earth Syst. Sci. 120 113–121.
Rathburn A E and Corliss B H 1994 The ecology of living (stained) deep-sea benthic foraminifera from the Sulu Sea; Paleoceanography 9 87–150.
Reichart G J, den Dulk M, Visser H J, van der Weijden C H and Zachariasse W J 1997 A 225 kyr record of dust supply and the oxygen minimum zone from the Murray Ridge (northern Arabian Sea); Palaeogeogr. Palaeoclimatol. Palaeoecol. 134 149–169.
Reichart G J, Lourens L J and Zachariasse W J 1998 Temporal variability in the northern Arabian Sea Oxygen Minimum Zone (OMZ) during the last 225,000 years; Paleoceanography 13 607–621.
Rohling J and Zachariasse W J 1996 Red Sea outflow during the last glacial maximum; Quat. Int. 31 77–83.
Rostek F, Bard E, Beaufort L, Sonzogni C and Ganssen G 1997 Sea surface temperature and productivity records for the past 240 kyr in the Arabian Sea; Deep Sea Res. Part II 44 1461–1480.
Schmiedl G and de Bovée F, Buscail R, Charrìère B, Hemleben C, Medernach L and Picon P 2000 Trophic control of benthic foraminiferal abundance and microhabitat in the bathyal Gulf of Lions, western Mediterranean Sea; Mar. Micropaleontol. 40 167–188.
Schmiedl G and Leuschner D C 2005 Oxygenation changes in the western Arabian Sea during the last 190,000 years: Productivity versus Deep water circulation; Paleoceanography 20 PA2008, https://doi.org/10.1029/2004PA001044.
Schulz H, von Rad U and Erlenkeuser H 1998 Correlation between Arabian Sea and Greenland climate oscillations of the past 110,000 years; Nature 393 54–57.
Schulz H, von Rad U and Von Stackelberg U 1996 Laminated sediments from the oxygen minimum zone of the northeastern Arabian Sea; Geol. Soc. Lond. Spec. Publ. 116 185–207.
Schumacher S, Jorissen F J, Dissard D, Larkin K E and Gooday A J 2007 Live (Rose Bengal stained) and dead benthic foraminifera from the oxygen minimum zone of the Pakistan continental margin (Arabian Sea); Mar. Micropaleontol. 62 45–73.
Sen Gupta B K and Machain-Castillo M L 1993 Benthic foraminifera in oxygen-poor habitats; Mar. Micropaleontol. 20 183–201.
Singh A D, Rai A K, Verma K, Das S and Bharti S K 2015 Benthic foraminiferal diversity response to the climate induced changes in the eastern Arabian Sea oxygen minimum zone during the last 30 ka BP; Quat. Int. 374 118–125.
Singh R K, Gupta A K and Das M 2012 Paleoceanographic significance of deep-sea benthic foraminiferal species diversity at southeastern Indian Ocean Hole 752A during the Neogene; Palaeogeogr. Palaeoclimatol. Palaeoecol. 361–362 94–103.
Song Q, Gordon A L and Visbeck M 2004 Spreading of the Indonesian Throughflow in the Indian Ocean; J. Phys. Oceanogr. 34 772–792.
Webster P J 1987 The elementary monsoon; In: Monsoons (eds) Fein J S and Stephens P L, John Wiley, Hoboken N J, pp. 3–32.
Wyrtki K 1973 Physical oceanography of the Indian Ocean; In: The Biology of the Indian Ocean (ed.) Zeitschel B, Springer, New York, pp. 18–36.
Zahn R and Pedersen T F 1991 Late Pleistocene evolution of surface and mid-depth hydrography at the Oman Margin: Planktonic and benthic isotope records at Site 724; In: Proceedings ODP (eds) Prell W L et al., Scientific Results 117 291–303.
Acknowledgements
The Ocean Drilling Program (ODP) is gratefully acknowledged to provide the core samples for the present work. Authors are very thankful to the anonymous reviewer whose thoughtful comments improved the quality of this manuscript. The grants provided to VKP as INSPIRE Fellowship by the DST (Grant No. DST/INSPIRE Fellowship/IF150618), New Delhi partially supported the present research work.
Author information
Authors and Affiliations
Contributions
Vishwesh Kumar Pathak has generated the benthic foraminiferal census data and helped in the calculations, interpretation and writing of the manuscript. Ashish Kharwar has processed the core samples to separate the foraminiferal tests and generated the planktic foraminiferal data. He also assisted in identification of taxa and preparing figures. Ajai Kumar Rai has procured the ODP core samples and was also involved in identification of foraminiferal taxa and interpretation of the paper.
Corresponding author
Additional information
Communicated by Maripi Dileep.
Corresponding editor: Maripi Dileep
Appendices
Annexure 1: List of benthic foraminiferal species recorded at ODP Hole 723A
Amphicoryna hirsuta | Amphicoryna intercellularis |
Amphicoryna proxima | Amphicoryna scalaris |
Angulogerina carinata | Anomalina globulosa |
Anomalina semipunctata | Astrononion umbilicatulum |
Biloculina inflata | Biloculina lucernula |
Biloculina murrhina | Bolivina bradyi |
Bolivina italica | Bolivina pseudopunctata |
Bolivina pygmaea | Bolivina robusta |
Bolivina spathulata | Bolivina subspinescens |
Bolivina tortuosa | Bolivinita pseudoplicata |
Bolivinita quadrilatera | Brizalina alata |
Brizalina capitata | Brizalina variabilis |
Bulimina aculeata | Bulimina alazanensis |
Bulimina arabiensis | Bulimina gibba |
Bulimina marginata | Bulimina striata |
Bulimina subornata | Cancris auriculus |
Cancris oblongus | Cassidulina laevigata |
Cassidulina bradyi | Cassidulina carinata |
Cassidulina crassa | Cassidulina oblonga |
Cassidulina pacifica | Ceratobulimina pacifica |
Chilostomella oolina | Chrysalidinella dimorpha |
Cibicides bradyi | Cibicides cicatricosus |
Cibicides kullenbergi | Cibicides lobatulus |
Cibicides mundulus | Cibicides pachyderma |
Cibicides pseudoungerianus | Cibicides wuellerstorfi |
Dentalina advena | Dentalina communis |
Dentalina filiformis | Dentalina guttifera |
Dentalina intorta | Dentalina subsoluta |
Discopulvinulina baconica | Discopulvinulina subbertheloti |
Eggrella bradyi | Ehrenbergina trigona |
Epinoides bradyi | Epinoides schreibersi |
Epistominella exigua | Fissurina alveolata |
Fissurina crebra | Fissurina quadrata |
Fissurina seguenziana | Fissurina trigono-marginata |
Fursenkoina bradyi | Fursenkoina cornuta |
Fursenkoina reagani | Fursenkoina rotundata |
Fursenkoina texturata | Gavelinopsis lobatulus |
Globobulimina pacifica | Globocassidulina pacifica |
Globocassidulina subglobosa | Gyroidinoides cibaoensis |
Gyroidinoides nitidula | Gyroidinoides torulus |
Heronallenella boltovoskoyi | Hoeglundina elegans |
Hyalinea balthica | Karreriella bradyi |
Karreriella subrotundata | Lagena gracillima |
Lagena hispida | Lagena laevis |
Lagena striata | Lagena sulcata |
Lenticulina altifrons | Lenticulina calcar |
Lenticulina convergens | Lenticulina peregrina |
Loxostomum limbatum | Marginulina obesa |
Martinottiella bradyana | Martinottiella communis |
Melonis barleeanum | Miliolina oblonga |
Miliolina rotundata | Neoconorbina torquemi |
Nodosaria calomorpha | Nodosaria flinti |
Nodosaria inflexa | Nodosaria lamnulifera |
Nodosaria radicula | Nodosaria simplexa |
Nonionella amplilabrata | Nonionella bradyi |
Nummoloculina irregularis | Nuttallids umbonifera |
Oolina apiculata | Oolina globosa |
Oridorsalis tenera | Oridorsalis umbonatus |
Osangularia culter | Planularia australis |
Praeglobobulimina pyrula | Protoglobobulimina affinis |
Protoglobobulimina pupoides | Pullenia bulloides |
Pullenia quadriloba | Pullenia quinqueloba |
Pullenia salisburyi | Pyrgo depressa |
Pyrgoella sphaera | Pyrulina gutta |
Quinqueloculina seminulum | Quinqueloculina tropicalis |
Quinqueloculina venusta | Quinqueloculina weaveri |
Rectoglandulina aequalis | Rectoglandulina redicula |
Rectoglandulina rotundata | Rectoglandulina torrida |
Robertina bradyi | Robertina oceanica |
Robulus costatus | Robulus crassus |
Robulus pliocaenicus | Rosalina bradyi |
Rosalina globularis | Rosalina vilardeboana |
Rotaliatinopsis semiinvoluta | Russella simplexa |
Saracenaria latifrons | Sigmoilina tenuis |
Sigmoilopsis schlumbergeri | Siphotextularia rolshauseni |
Sphaeroidina bulloides | Spiroloculina asperula |
Spiroloculina depressa | Spiroloculina rotundata |
Textularia lythostrota | Textularia pseudogramen |
Textularia sagitulla | Trifarina bradyi |
Triloculina rupertiana | Triloculina trigonula |
Tritaxia tricarinata | Uvigerina carariensis |
Uvigerina crassicostata | Uvigerina cushmani |
Uvigerina flinti | Uvigerina hispida |
Uvigerina hispidocostata | Uvigerina nitidula |
Uvigerina peregrina | Uvigerina porrecta |
Uvigerina proboscidea | Uvigerina schwageri |
Vaginulina patens | Vaginulina subelegans |
Vaginulinopsis tasmanica | Virgulina davisi |
Annexure 2: List of characteristic oxic and dysoxic benthic foraminiferal species recorded at ODP Hole 723A
Oxic species | Dysoxic species |
|---|---|
Biloculina inflata | Bolivina bradyi |
Biloculina lucernula | Bolivina italica |
Biloculina murrhina | Bolivina pseudopunctata |
Cibicides bradyi | Bolivina pygmaea |
Cibicides cicatricosus | Bolivina robusta |
Cibicides kullenbergi | Bolivina spathulata |
Cibicides lobatulus | Bolivina subspinescens |
Cibicides mundulus | Bolivina tortuosa |
Cibicides pachyderma | Brizalina alata |
Cibicides pseudoungerianus | Brizalina capitata |
Cibicides wuellerstorfi | Brizalina variabilis |
Globocassidulina subglobosa | Chilostomella oolina |
Pyrgo depressa | Dentalina advena |
Quinqueloculina weaveri | Dentalina communis |
Quinqueloculina seminulum | Dentalina filiformis |
Quinqueloculina tropicalis | Dentalina guttifera |
Quinqueloculina venusta | Dentalina intorta |
Siphotextularia rolshauseni | Dentalina subsoluta |
Spiroloculina asperula | Fissurina alveolata |
Spiroloculina depressa | Fissurina crebra |
Spiroloculina rotundata | Fissurina quadrata |
Triloculina rupertiana | Fissurina seguenziana |
Triloculina trigonula | Fissurina trigono-marginata |
Fursenkoina reagani | |
Fursenkoina bradyi | |
Fursenkoina cornuta | |
Fursenkoina rotundata | |
Fursenkoina texturata | |
Globobulimina pacifica | |
Globocassidulina pacifica | |
Praeglobobulimina pyrula | |
Protoglobobulimina affinis | |
Protoglobobulimina pupoides | |
Virgulina davisi |
Rights and permissions
About this article
Cite this article
Pathak, V.K., Kharwar, A. & Rai, A.K. Benthic foraminiferal response to changes in the northwestern Arabian Sea oxygen minimum zone (OMZ) during past \(\sim\)145 kyr. J Earth Syst Sci 130, 163 (2021). https://doi.org/10.1007/s12040-021-01659-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12040-021-01659-2