Abstract
Ongoing rapid climate change has a major effect on marine fauna, and understanding these faunal changes analogous to future climatic periods is crucial. The Oligocene is commonly considered a critical transition period, linking the archaic world of the tropical Eocene and the more modern ecosystems of the Miocene. Here, we show the response of marine benthic foraminifera to the early Oligocene climatic changes at Ocean Drilling Program (ODP) Hole 1138A of the Southern Ocean (Indian Sector). We made use of the diversity parameters, the relative abundance of dominant benthic foraminifera, and isotopic data to understand past oceanographic changes. Our results suggest that the early Oligocene was an interval of unstable conditions dominated by the species of high oxygen, intermediate food supply, and well-ventilated, cold, corrosive bottom water conditions. The species richness abruptly decreases at the end of the studied interval, which shows the major Southern Hemisphere glaciation. During this time, species were characterized by relatively cold and carbonate-corrosive bottom water. Additionally, the present study of the benthic foraminiferal abundance and diversity indices reveals the cooling of the Southern Ocean at the early and late stages of the studied interval interrupted by a short-lived warming event. The study further enhances the understanding of paleo-marine ecology by evaluating the response of deep-sea benthic foraminifera to global climate change.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The planet Earth has witnessed extreme climate variability in its geological past, especially during the Cenozoic, i.e., the last 65 Ma. Cenozoic climate history was marked by multiple climatic changes in which the transition from Greenhouse to Icehouse was major event. This transition was marked as the Eocene–Oligocene Transition (EOT) at ~ 34 Ma (Zachos et al. 2001; Coxall et al. 2005; Anagnostou et al. 2016; Tang et al. 2020). Additionally, the early Oligocene events (Oi-1) that are the peak timing of the Antarctic glaciation at the end of the EOT (DeConto and Pollard 2003) were subdivided into two positive excursions, Oi-1a and Oi-1b (Zachos et al. 1992). Although recent studies compile many small positive excursions in their high-resolution stable isotopic analysis, these previous studies record the long Oi-event from 33.6 to 25.2 Ma with small abrupt perturbations, namely, Oi-1 (33.7 Ma and 33.4 Ma), Oi-1a (32.8 Ma), and Oi-1b (31.7 Ma) in the Southern Ocean (Miller et al. 1991; Pälike et al. 2006; Lee and Jo 2019; O’Brien et al. 2020). They suggest that a decrease in pCO2 consumption caused the short-term recovery phase resulting from a decrease in the present ice sheet cover during the Oi-1a, also known as the earliest Oligocene Glacial Maxima (EOGM) (Coxall and Wilson 2011). Additionally, these events also coincide with low eccentricity. However, the high eccentricity level between these events suggests that total solar irradiance on the ground was slightly higher than the average (Lee and Jo 2019). Other studies explain that the observed isotopic excursions between Oi-1 (33.5 Ma) and Oi-1b (31.7 Ma) events are due to the release of a short-lived pulse of CO2 at different phases due to leakage of fossil carbon (Coxall et al. 2018). These events are noticeable by observing the positive and negative excursions of oxygen isotope data (δ18O values) that mark the periods of global cooling/increase in ice volume and warming, respectively. And the decrease in the oxygen isotopic values in the austral high latitude during the recovery phase could have been caused by regional sea ice melting.
These climatic/oceanographic changes affect the living hood of marine fauna heavily. Marine protists, especially benthic foraminifera, have proven to be good indicators of such climatic events. Benthic foraminifera occur in most marine environments, eutrophic to oligotrophic settings, flourishing in epifaunal and infaunal microhabitats, different oxygenated environments, and sustaining variable temperature and energy conditions; therefore, they are ideal for paleo-reconstruction of climatic variability and global ocean circulation (Gupta and Thomas 1999, 2003; Corliss et al. 2009; De and Gupta 2010; Singh et al. 2022). Previous studies indicate a significant relationship between deep-sea benthic foraminifera diversity and high latitudinal oceanic processes (Fischer 1960; Buzas and Gibson 1969; Thomas and Gooday 1996). The diversity in deep-sea benthic foraminifera depends on numerous attributes like heterogeneity in habitat, availability of food, temperature, and rate of predation (Kaiho 1994; Gooday et al. 2010). Several studies relate the significant and gradual change in the diversity of deep-sea benthic foraminifera to cooling deep water and an oxygenated environment (CaCO3 corrosivity; Thomas 1992). Therefore, deep-sea benthic foraminifera witnessed significant oceanic and climatic turnover throughout the Cenozoic (Singh et al. 2012). Many recent and earlier studies provide critical information related to the distribution and ecological preferences of benthic foraminifera and the factors controlling it (Streeter 1973; Lohmann 1978; Corliss 1979; Thomas 1992; van der Zwaan et al. 1999; Gooday et al. 2000; Fentimen et al. 2020).
Deep-sea benthic foraminifera provide multiple ways to record climatic and oceanographic changes; however, isotopic records, faunal response, and diversity are the most distinct and reliable tools (Miller et al. 1991; Nomura1995; Singh and Gupta 2004; Zachos et al. 2001; Rai and Maurya 2009; Kumar et al. 2021). Deep-sea benthic foraminifera are also vital in deciphering oceanic environments due to their wide ecological adaptability (Schnitker 1974; Streeter 1973; Singh et al. 2021). The ecology of marine fauna gets severely affected due to the surface productivity and oxygenation of deep water throughout the Cenozoic (Rodrigues et al. 2018; Kender et al. 2019; Lu et al. 2020). During Cenozoic, the opening of the Drake Passage and the Tasman gateway caused a significant change in the oceanic circulation, triggering the initiation of ice sheet building over the Antarctic landmass, initiating the Antarctic Circumpolar Current (ACC) (Kennett 1977; Lawver and Gahagan 2003). The corresponding high thermal gradient between high and low latitudes and the decline in atmospheric pCO2 levels formed the continental ice sheet over Antarctica (DeConto and Pollard 2003; Katz et al. 2008; Bohaty et al. 2012; Anagnostou et al. 2016). The beginning of ACC and the growth of the ice sheet resulted in the Oligocene cooling more than the warmer Eocene, which affected the faunal abundance and diversity pattern at the high latitude (Pälike et al. 2012). Miller et al. (1991) further represented Oligocene as an analog for the modern icehouse world with numerous episodes of the waxing and waning of ice sheets. Consequently, the early Oligocene marks a phase of noticeable change in the volume of deep water formation in the Southern Ocean (Takata et al. 2012).
The stable isotope analysis is an excellent indicator for observing and delineating the paleoclimate and paleoceanographic conditions of the deep marine (Armstrong McKay et al. 2016). Additionally, benthic foraminiferal carbon, as well as oxygen isotope records, forms a reliable chemostratigraphic tool for correlation and a proxy to constrain the past carbon cycle and the global climatic and oceanographic perturbations (Li et al. 2022). They have been used in many discussions of change in bottom water mass type (i.e., changeover from older bottom water to younger bottom water), global ice volume, deep-sea temperature, and local productivity (Zachos et al. 2001; Ravelo and Hillaire-Marcel 2007). Furthermore, the carbon isotopic values of deep-sea benthic foraminifera species and their microhabitat preferences also reveal the deep environmental condition. The δ13C values of epifaunal taxa reflect the bottom water dissolved inorganic carbon and aging, and the infaunal taxa closely align with the more negative δ13C values of pore waters (Ravelo and Hillaire-Marcel 2007). This correlation between isotopic composition and microhabitat preference indicates that the δ13C values of deep-sea benthic foraminifera are directly/indirectly influenced by deep ambient environments and ecological preferences (Rathburn et al. 2000).
The Southern Ocean plays a crucial role in controlling the global climate due to its unique oceanography, which is a primary source of surface and deep cold water currents. The current oceanographic setting of the Southern Ocean is established after many tectonic and oceanographic changes in the past. It changed over time and became a primary source of cold deep water mass (Lawver and Gahagan 2003; Haumann et al. 2020). It behaves like a buffer zone of all the water masses; here, all the primary ocean water gets mixed and distributed throughout the world ocean by the water circulation process (thermohaline circulation). The mixing of water leads to a change in the behavior of water masses. So, the deep ocean currents play a vital role in maintaining the ocean’s heat budget, while the surface water currents and the organisms control the atmospheric carbon dioxide level (Pascual et al. 2020). To understand paleoceanography, we can analyze the deep-sea sediments, elemental analysis (stable isotope analysis) of faunas, and morphological characteristics of fossils. Also, the quantitative study, i.e., census data, factor analysis, statistics, and time series analysis of fauna abundance, can be essential for studying paleoceanography and simple climatology. Thus, the combined study of micropaleontological and paleoceanographic data can predict future biotic and climatic advancements.
The present study focuses on understanding the diversity index pattern and the relative abundance of the early Oligocene deep-sea benthic Foraminifera at the ODP hole 1138A, the Southern Ocean (Indian Sector). Due to proto-ACC initiation, the influence of cold water profoundly affected the present study site, and its implications are recorded in the deep-sea benthic foraminifera. The mainstay of this paper remains to explore benthic foraminiferal response with the help of diversity patterns and stable isotope records in the Southern Ocean and its relative abundance in response to the ocean-driven diversification during the early Oligocene.
Physiology of the study area
The Kerguelen Plateau is a large igneous province in the Indian Sector of the Southern Ocean which extends from ~ 2300 km southeast between 46°S and 64°S toward Antarctica. The shallow submarine Kerguelen Plateau is divided into Northern Kerguelen Plateau (NKP), Central Kerguelen Plateau (CKP), and Southern Kerguelen Plateau (SKP) (Fig. 1a). The estimated age of SKP is ~ 80 to 100 Ma, whereas NKP has formed from ~ 65 Ma to recent (Frey et al. 2003). It is surrounded by Australian-Antarctic, Prince Elizabeth Trough, Enderby, and Crozet basins in the northeast, south, southwest, and north, respectively (Wang et al. 2016).
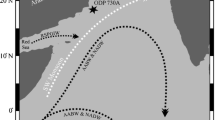
a Outline map of Kerguelen plateau with the location of hole 1138A (star). The continuous dark and light lines with arrow directions show different oceanic currents on this site. The inset map shows a broad area view of the Southern Ocean with the location of the Kerguelen Plateau. b A general schematic section of meridional ocean water circulation of the Southern Ocean illustrates modern water circulation. Acronyms: SACCF, Southern Antarctic Circumpolar Current Front; PF, Polar Front; ACC, Antarctic Circumpolar Current; FT, Fawn Trough; AABW, Antarctic Bottom Water; AAIW, Antarctic Intermediate Water; CSW, Continental Shelf Water; AASW, Antarctic Surface Water; SASW, Subantarctic Surface Water; STSW, Subtropical Surface Water. Modified after Speer et al. (2000)
The Kerguelen Plateau creates an obstacle for the ACC, therefore diverting its route (Wang et al. 2016; Wright et al. 2018). ACC is the world’s largest and most influential ocean current system driven eastward around Antarctica by prevailing westerly winds (Rintoul and Da Silva 2019). The main component of ACC is Circumpolar Deep Water (CDW). CDW is considered the highest volume water mass in the Southern Ocean and is a mixture of North Atlantic Deep Water (NADW) mass and Antarctic Bottom Water (AABW) mass, as well as some ocean fronts, i.e., Polar Front (PF), Subantarctic Front (SAF), and Southern Antarctic Circumpolar Current Front (SACCF) (Wright et al. 2018; Rintoul and Da Silva 2019) (Fig. 1a). The Kerguelen Plateau is influenced by the deep water components of the ACC that flow northward through the central part of the Kerguelen Plateau and then turn eastward toward the Australian-Antarctic Basin (Park et al. 2009). It accounts for two-thirds of the water masses driven by the ACC in all regions around Antarctica. The exact path of these water masses and fronts around the Kerguelen Plateau is still in debate (Park et al. 2009); however, the central Kerguelen Plateau is dominated by the CDW irrespective of the slight differences in PF and SACCF locations. The ODP Hole 1138A is directly under the influence of Circumpolar Deep Water (CDW) masses, and fronts are located south of the PF and north of the SACCF zones (Fig. 1a).
The modern Southern Ocean water circulation is governed by the cold deep and surface water masses (Fig. 1b). The Antarctic Bottom Water (AABW) dominates the bottom part of the Southern Ocean, while the middle water column is dominated by the Lower Circumpolar Deep Water (LCDW) and the Upper Circumpolar Deep Water (UCDW). The LCDW is colder than the UCDW, while AABW is the coldest water mass among them. Above the UCDW, a water mass known as Antarctic Intermediate Water (AAIW) dominates the Southern Ocean (Fig. 1b). In contrast, the surface regions are dominated by the different water masses, i.e., Continental Shelf Water (CSW) around the self-regions, then Antarctic Surface Water (AASW), then Subantarctic Surface Water (SASW), and the distal water mass known as Subtropical Surface Water (STSW). These water masses are also cold water mass but differ by the properties of their temperature and density (Speer et al. 2000) (Fig. 1b).
Site location, materials, and methodology
ODP site 1138 (53° 33.100′S, 75° 58.490′E; water depth, 1141 m) was drilled in the middle of the Kerguelen Plateau during the ODP leg 183 (Fig. 1a). A total of 145 samples were used from sediment cores of 35R (1–5) to 36R (1–6) of 10 cm3 volume from Ocean Drilling Program (ODP), site 1138, Hole A, located in the middle of the central Kerguelen Plateau. The studied sample belongs to calcareous nannofossil zones NP21 to NP23, planktic foraminifera zones P18 and P19, and abyssal and bathyal benthic foraminifera zones AB8 and BB5. The calcareous nannofossil zones NP21 are defined by the last occurrences (LO) of Discoaster saipanensis and Coccolithus formosus, NP22 as determined by the disappearance of Coccolithus formosus and Reticulofenestra umbilicus, and zone NP23 is defined by LO of Reticulofenestra umbilicus and first occurrences of (FO) of Sphenolithus ciperoensis (Pälike et al. 2010; Gradstein et al. 2012). The planktic foraminifera zone P18 is demarcated by the LO of Hantkenina alabamensis and Pseudohastigerina naguewichiensis, and zone P19 is defined by the LO of Pseudohastigerina naguewichiensis and Turborotalia ampliapertura (Wade et al. 2011; Gradstein et al. 2012). The bathyal benthic foraminifera zone BB5 is demarcated by the LO of Cibicidoides truncanus and Plectofrondicularia paucicostata and/or Bulimina jacksonensis, while the abyssal benthic foraminifera zone AB8 is defined by the LO of Nuttallides truempyi and Cibicidoides micrus (Berggren and Miller 1989). The 16.8 m of this early Oligocene foraminifera bearing well-preserved nannofossil chalk deposited at a water depth of ~ 1141 m (Apel et al. 2002) is used for this study. The samples cover the early Oligocene age ranging from ~ 34 to ~ 31 Ma. Apel et al. (2002) reported that ~ 80 m of the Oligocene epoch occurs in the core from 183-1138A-23R to the base of core 183-1138A-36R. The laboratory work was followed per the standard protocol used by Gupta and Thomas (1999) and Rai and Maurya (2007). The sediment samples were wet-sieved at > 125 µm and dried overnight at 50 °C. After drying, a microsplitter was used to subdivide the sample to contain ~ 300 specimens of benthic foraminifera. All benthic foraminifera have been picked from the split fraction and mounted on an assemblage slide of 48 chambers. All the specimens were identified to species level using standard literature and catalog (e.g., Nomura 1995; Holbourn et al. 2013).
The picked specimens were further computed for Shannon–Weaver (H(S)), Hurlbert’s (Sm), Fisher’s alpha (α), and equitability (E′) diversity indices (for more details, see Rai and Maurya 2009) and were plotted to study the relative abundance and the diversity pattern of the benthic foraminifera on the Kerguelen Plateau. The Shannon–Weaver diversity index explains how community individuals are distributed among existing species. So, it can also be called entropy rather than diversity. Instead of the number of species present in the community, the entropy provides the average level of uncertainty regarding the identity of a single species chosen from it (Bouchet et al. 2012). The Shannon–Weaver index defines each species dependent on its pi proportion, so the rare species make a small contribution, and Hurlbert’s diversity index contributes to reducing different size samples to a standard size so that we make them comparable in terms of the number of species. Fisher’s alpha (α) index explains the richness of the particular species in a sample, and equitability (i.e., evenness) describes high diversity when only a few species show higher abundance. It defines as the dependence of an individual on a species. Finally, species richness (S) has been used to understand the variation pattern in the species throughout the study interval. However, a variety of food resources may cause enhanced diversity. So, using the different diversity indices, we can explain the ecological preferences of the deep-sea benthic foraminifera. We have also calculated the sedimentation rate using depth and age data which remains constant (~ 20 m/Ma) at the study hole throughout the early Oligocene (Fig. 2).
The stable isotopic analysis was performed on benthic foraminifera species (Cibicidoides spp.). The carbon and oxygen isotope analyses of all the 140 samples from ODP Hole 1138A have been performed at the Stable Isotope Laboratory (SIL), Department of Earth Sciences, Indian Institute of Technology Roorkee, in India. Samples were ultrasonically washed using millipore water and dried in an oven at 50 °C. After drying, the samples were reacted with 104% H3PO4 at 70 °C in an automated ThermoFisher Kiel carbonate reaction device (Kiel IV) connected to MAT 253 plus mass spectrometer. Analytical accuracy and precision of measurements were observed by repeat analyses of laboratory standards calibrated by assigning δ18O values of -28.23‰ to in-house standard (MERCK) and -23.19‰ to NBS18 and δ13C values of -13.88‰ to in-house standard (MERCK) and -5.016‰ to NBS18. The internal and external measurement precision was 0.07‰ or better for both δ13C and δ18O in samples.
Results
Diversity indices are a well-established indicator of environmental changes and their possible effect on faunal abundance and diversity (Klootwijk and Alve 2022). The Shannon–Weaver (H(S)) index, Hurlbert’s (S100) diversity index, Fisher’s alpha (α) index, and species richness (S) vary repeatedly with significantly high values at ~ 33.7, ~ 33.4, ~ 32.8, and ~ 31.7 Ma (see Supplementary Materials (S1) for details and Fig. 3). Furthermore, the equitability (E′) shows comparatively less variability than the other diversity indices, and the values vary repeatedly (Fig. 3). The stable isotope values of Cibicidoides spp. provide quantitative estimation of the past climatic changes. Stable oxygen isotope data show a long-term decreasing trend from ~ 33.7 to ~ 33.1 Ma. The high values at ~ 33.7 Ma and ~ 33.4 Ma relate to the Oi-1 events (EOGM); furthermore, increasing trends in δ18O at ~ 32.8 Ma and ~ 31.7 Ma coincide with Oi-1a and Oi-1b events, respectively, during the early Oligocene. The oxygen isotope (δ18O) shows positive and negative excursion from time to time; it shows 0.92‰ at ~ 33.7 Ma and increases up to 1.55‰ at ~ 33.7 Ma; again, it drops to 0.48‰ at ~ 33.8 Ma and increases up to 1.89‰ at ~ 33.4 Ma. This abrupt positive excursion at ~ 33.7 Ma and ~ 33.4 Ma correlates with the Oi-1 events. At the same time, the carbon isotope increased from 0.61 to 1.63‰ at ~ 33.6 Ma and ~ 33.4 Ma, respectively, while a positive excursion of 1.29‰ also has been noted at 33.7 Ma. The decreasing trend in δ18O values from ~ 33.7 to ~ 33.6 Ma and ~ 33.4 to ~ 32.8 Ma is considered a recovery phase. Another positive excursion of δ18O is recorded at ~ 32.6 Ma and ~ 31.6 Ma with isotopic values of 1.81‰ and 2.58‰, respectively. These positive excursions are closely related to the Oi-1a and Oi-1b events during the early Oligocene. The carbon isotopic values also fluctuated from a minimum value of 0.20‰ to the highest value of 1.54‰ at ~ 32.1 Ma and ~ 31.6 Ma, respectively (Fig. 3; see S1 for details).
Time series plot of species diversity at ODP hole 1138A of Shannon–Weaver (H(S)) index, Hurlbert’s (S100) diversity, equitability (E′), species richness (S), and Fisher’s alpha (α) index with stable isotope data. Highlighted portion in the figure shows early Oligocene cooling events Oi-1 (pink and brown color), Oi-1a (violet color), and Oi-1b (blue color). The black arrow shows the recovery phase. Abyssal (AB) and bathyal (BB) benthic foraminiferal zones of Berggren and Miller (1989) are correlated with planktic (P) foraminiferal zones after Wade et al. (2011) and calcareous nannofossil (NN) zones after Pälike et al. (2010). Isotope data is from ODP site 744A (Salamy and Zachos 1999) with Bulk CaCO3 contents from ODP site 748B (Zachos et al. 1992). Numerical ages after GTS 2012
We also made use of the relative abundance of seven different species of deep-sea benthic foraminifera within the studied time interval. Here, we have selected high abundance species (> 5% at least in 3 samples) to describe the deep ocean environments. The benthic foraminifera species Oridorsalis umbonatus is abundant throughout the early Oligocene but attained a maximum at ~ 31.8 Ma, ~ 32.7 Ma, and from ~ 33.4 to ~ 33.7 Ma (Table 1, Fig. 4). The second species, Nuttallides umbonifer, is also present throughout the earliest Oligocene interval; however, it is dominant from ~ 33.4 Ma, ~ 32.8 Ma, and ~ 31.6 Ma (Fig. 4, Table 1). The third species, Cibicidoides kullenbergi, exhibits a significant high from ~ 33.4 to ~ 32.6 Ma and from ~ 32.0 to ~ 31.7 Ma; the latter peak is not perpetual (Table 1; Fig. 4). The fourth species, Pullenia bulloides, occurs throughout the studied interval with a significant increase from 33.3 Ma, which attains the maximum at ~ 32.8 Ma (Fig. 4). The fifth species, Globocassidulina subglobosa, increases upward throughout the interval; however, it is abundant at ~ 33.4 Ma and ~ 32.8 Ma and attains the maximum from ~ 31.5 to ~ 31.3 Ma (Table 1, Fig. 4). The sixth species, Astrononion echolsi, exhibits a decreasing trend from ~ 33.7 to ~ 33.4 Ma and has maximum abundances recorded at ~ 31.9 Ma (Fig. 4; Table 1). The last species, Uvigerina peregrina, showed maximum abundance at ~ 32.2 Ma and ~ 31.6 Ma (Fig. 4, Table 1; for details, see S1).
Time series plots of the relative abundance of dominated benthic foraminifera at ODP hole 1138A with Bulk CaCO3 contents from ODP site 748B (Zachos et al. 1992). Highlighted portion in the figure shows early Oligocene cooling events Oi-1 (pink and brown color), Oi-1a (violet color), and Oi-1b (blue color). The black arrow shows increasing and decreasing trends of the Shannon–Weaver diversity index and species abundance. Abyssal (AB) and bathyal (BB) benthic foraminiferal zones of Berggren and Miller (1989) are correlated with planktic (P) foraminiferal zones after Wade et al. (2011) and calcareous nannofossil (NN) zones after Pälike et al. (2010). Numerical ages after GTS 2012
Discussion
Paleoceanography and paleoenvironmental reconstructions based on fossil record profoundly rely on new proxies and their correlation. Geochemical data (e.g., stable isotope data and trace element data), faunal species diversity, and abundance are essential to understanding past deep marine environmental conditions. Our deep-sea benthic foraminifera records at ODP Hole 1138A showed significant variation, coinciding with the changes in marine temperature, ocean productivity, global ice volume, and global ocean circulation. The initiation of proto-ACC and the influence of cold water profoundly affected our study site. These changes are observed at this site, captured by the deep-sea fauna (benthic foraminifera). A correlative study between benthic foraminiferal species diversity and the relative abundance of dominant species helps us understand the ocean-driven diversification in this faunal group in the Southern Ocean (Indian Sector) of the early Oligocene.
Environmental preferences of deep-sea benthic foraminifera in the Southern Ocean
Oridorsalis umbonatus is an opportunistic, shallow infaunal dweller species that can live in various marine environments (Fig. 4). This taxon prefers both eutrophic and oligotrophic settings, and the depth preference is known from lower bathyal to abyssal in the Antarctic Ocean (Uchio 1960), the Atlantic Ocean (Lohmann 1978; Streeter and Shackleton 1979), the Indian Ocean (Corliss 1979; Gupta 1994), and the Sulu and South China Sea (Miao and Thunell 1993). O. umbonatus dominates above the carbonate lysocline around Antarctica, which may point toward the presence of AABW or similar water mass environments (Mackensen et al. 1990). Some researchers mentioned it as an indicator of oligotrophic settings with carbonate-corrosive bottom currents found in lower temperatures and intermediate oxygen conditions (Bremer and Lohmann 1982; Nomura 1991). The association of O. umbonatus with high carbonate levels and low carbon is also reported from the deep bottom water of the Sulu Sea (Miao and Thunell 1993). O. umbonatus choose well-oxygenated, low organic carbon environments associated with regions of low surface productivity (Burke et al. 1993), whereas Woodruff (1985) noticed that O. umbonatus is more related to regions with high productivity in the surface area, and Kaiho (1999) reported suboxic bottom waters. The abundance of this species is higher at the studied site 1138A during the early Oligocene time (~ 33.7 to ~ 33.4 Ma; Table 1 and Fig. 4), reflecting a colder water mass than the Eocene. Additionally, a higher abundance of this species was also observed at DSDP sites 516 and 522 in the South Atlantic; ODP sites 744 and 689 in the Southern Ocean; and ODP sites 1218, 1219, and U1334 in the eastern equatorial Pacific Ocean during the early Oligocene interval (Tjalsma 1983; Coxall and Wilson 2011; Takata et al. 2012). During the early stage of the Oligocene, the dominance of the species was slightly higher than the other species as the nutrient condition was possibly high and well-oxygenated conditions have been reflected by this species.
Nuttallides umbonifer is an epifaunal species that live in various environments and is associated with AABW (Fig. 4; Streeter 1973; Lohmann 1978; Bremer and Lohmann 1982; Mackensen et al. 1995). The presence of this species in the ocean indicates a cold, carbonate-corrosive oligotrophic environment between CCD and calcite lysocline deep water mass (e.g., AABW or its near equivalents) (Bremer and Lohmann 1982; Woodruff 1985; Mackensen et al. 1990, 1995; Nomura 1995; Bordiga et al. 2015). A close association between relatively high oxygenated, low temperature, and low food supply in the deep bottom water of this species is reported in the Indian Ocean (De and Gupta 2010; Singh et al. 2012). It is observed that N. umbonifer is an indicator of a low-productivity oceanic regime, while, Woodruff and Savin (1989) reported it as a high-productivity surface area species. It is a characteristic benthic fauna of all the major ocean abyssal zone of the Pacific Ocean, the Atlantic (Schnitker 1974; Streeter 1973; Lohmann 1978; Streeter and Shackleton 1979), and the Indian Oceans (Corliss 1983). The high abundance of this species dominated during the early Oligocene at ~ 33.4 Ma, ~ 32.8 Ma, and ~ 31.6 Ma at the studied site 1138A, indicating a cold, carbonate-corrosive, relatively high oxygenated, and low food supply environment at the studied site in the Southern Ocean. However, this species achieved dominance during Oligocene in the South Atlantic Ocean at DSDP sites 522 and 516 and became an important taxon (Tjalsma 1983). Other than that, higher abundance of this species was also observed at ODP sites 744 and 689 in the Southern Ocean and 1218, 1219, and U1334 in the eastern equatorial Pacific Ocean (Nomura 1995; Takata et al. 2012).
Cibicidoides kullenbergi species have preferred a substrate with low organic carbon content below the low surface productivity area (Burke et al. 1993). The Cape region also reported that this species has low carbonate contents suggesting higher carbonate dissolution rates (Eberwein and Mackensen 2006). This species is associated with the high δ13C suggesting a low carbon influx to the seafloor (Nomura 1995). The stratigraphic distribution of C. kullenbergi is confined to oxygen-rich and nutrient-poor North Atlantic Deep Water (NADW) (Lohmann 1978; Mackensen et al. 1995). The warm water association of this species is noted in the Pacific Ocean (Woodruff 1985), the Atlantic Ocean (Lutze 1979), and the Indian Ocean (Maurya and Rai 2006). These species got their higher abundance during the middle age of the early Oligocene at ~ 33.3 to ~ 32.6 Ma and ~ 32.0 to ~ 31.4 Ma as the deep water condition changed to warmer for a brief interval possibly due to the release of a short-lived pulse of CO2 at different phases due to the leakage of fossil carbon (Coxall et al. 2018). This species indicates an oxygen-rich but nutrient-poor marine environment. The abundance of this species dominated during the early Oligocene at the studied site 1138A, indicating a short interval of relatively high temperature, less oxygenated, and intermediate food supply environment at the studied site in the Southern Ocean. However, this species achieved dominance during the late Oligocene but was less or absent during the early Oligocene in the South Atlantic Ocean at DSDP sites 522 and 516 and became a vital taxon (Tjalsma 1983). Additionally, a higher abundance of this species was also observed during the late Oligocene, while a low abundance of this species was recorded during the early Oligocene at ODP sites 747, 757, and 758 in the eastern Indian Ocean (Nomura 1995).
Pullenia bulloides is a shallow infaunal species that have well-established environmental preferences. It has shown the association with low temperature and highly oxygenated deep water with a top to the moderate influx of nutrient supply (Maurya and Rai 2006). Gupta and Thomas (1999) observed this species as an indicator of the average flux of organic in poorly ventilated bottom water conditions. This species allied with low oxygen conditions with intermediate to the high flux of organic matter in the Indian Ocean (Gupta et al. 2006). P. bulloides may be associated with areas of low surface productivity and, therefore, a small flux of organic matter to the seafloor (Burke et al. 1993), and observed the abundant occurrence of this species in postglacial sediments on the Ontong Java Plateau. P. bulloides is a representative species associated with high-salinity NADW in the southwest Indian Ocean (Corliss 1983). It has also been associated with assemblages that indicate the cold, low to intermediate organic flux, strongly pulsed, and high seasonality in the Indian Ocean (Gupta and Thomas 2003). In contrast, Bhaumik et al. (2007) found that this species was associated with NADW-like water mass with high oxygen and carbon at northeastern Indian Ocean DSDP site 216. This species is observed with glacial/interglacial intervals, which indicates the species can survive in low dissolved oxygen in the northwestern Atlantic Ocean. At the studied site, a higher abundance of P. bulloides was recorded during the earliest Oligocene at ~ 33.3 to ~ 32.8 Ma. The abundance of this species shows the cold with a highly oxygenated deep-sea environment. Additionally, this species commonly occurred during the early Oligocene in the South Atlantic Ocean at DSDP sites 522 and 516 but not as a vital taxon (Tjalsma 1983).
Globocassidulina subglobosa is an opportunistic cosmopolitan infaunal species associated with several different water masses with a wide bathymetry and long stratigraphic distributions. Species feeds on a fresh phytodetritus (Giusberti et al. 2016). It is reported from ODP site 758 in the eastern Indian Ocean as cold water dwelling and high food exploiting taxa (Nomura 1995). A high abundance of this species is linked to the strengthening of ocean bottom currents (Nagai et al. 2009). Takata et al. (2012) reported it as indicator of well-oxygenated bottom water with high organic carbon influx to the bottom of ocean. Fariduddin and Loubere (1997) reported that the association of G. subglobosa with NADW indicates low-productivity species in the Atlantic Ocean. As mentioned above, the association of this species with high seasonal food pulses is the characteristic feature this taxon (Eberwein and Mackensen 2006). High abundance of this taxon was also reported from submarine ridges and hills of the South Atlantic with an oligotrophic water mass of Circumpolar Deep Water (Mackensen et al. 1995). Gupta and Thomas (2003) have suggested that G. subglobosa may flourish in oligotrophic condition of the northeastern and southeastern Indian Ocean. It has also been reported that warm water condition is unfavorable to this species, and it has been observed to prefer cold water masses (Miao and Thunell 1993). Corliss (1979) reported it in close association with AABW. In this study, the abundance of G. subglobosa is high at ~ 33.4 to ~ 32.8 Ma in the early and ~ 31.5 to ~ 31.8 Ma of later stages of the studied interval (Table 1; Fig. 4) which indicates relatively colder bottom water. Additionally, a common abundance of this species was also observed at DSDP sites 516 and 522 in the South Atlantic; ODP sites 1218, 1219, and U1334 in the eastern equatorial Pacific Ocean; and ODP sites 747, 757, and 758 in the eastern Indian Ocean during the early Oligocene interval marked presence of cold bottom water (Tjalsma 1983; Nomura 1995; Takata et al. 2012).
Astrononion echolsi (synonymous with Astrononion umbilicatulum, also close ally with Astrononion pusillum Hornibrook) is a shallow infaunal benthic foraminifera, whose ecological information is minimal. A. echolsi is reported as a common species at ODP site 748 (Kerguelen Plateau) throughout the early Oligocene (Mackensen and Berggren 1992), and the first occurrences are recorded in the lower Oligocene by Nomura (1995). This species prefers a low-productivity zone on the Ontong Java Plateau (Burke et al. 1993). The association of this species with the lowest primary productivity and well-ventilated water is also reported in the Gulf of Aden (Almogi-Labin et al. 2000). This species prefers high-saline waters in the Red Sea (Gupta 1994). Singh and Gupta (2004) have mentioned that this species is associated with low organic influx and the well-ventilated deep marine environment in the southeastern Indian Ocean. A. echolsi is a representative taxon associated with hot and high-salinity NADW in the southwest Indian Ocean (Corliss 1983). ODP sites in the Indian Ocean reported that the occurrence of A. echolsi is widespread and influenced by the AABW (Corliss 1983; Nomura 1995). It is also reported from the Polar Front region of the southwest Atlantic Ocean (Mead and Kennett 1987). Miao and Thunell (1993) described its close association with oxygenated bottom water with low organic carbon influx of the South China Sea. The peak abundance of this species recorded at ~ 33.6 Ma and ~ 31.9 Ma at the studied site indicates higher production of the cold bottom water in the Southern Ocean. Additionally, this species was reported as common faunal element at ODP sites 1218, 1219, and U1334 in the eastern equatorial Pacific Ocean and ODP sites 747, 757, and 758 in the eastern Indian Ocean during the Oligocene at marked intervals (Nomura 1995; Takata et al. 2012).
Uvigerina peregrina is an infaunal species commonly described as a high-productivity taxon. The occurrence of U. peregrina in the Southeast Indian Ocean suggests the existence of Circumpolar Deep Water (Corliss 1983). The environmental preferences of this species are high organic carbon and low oxygen, commonly occurring below the upwelling zone, including the area of high concentration with bacteria, meiofauna, and exoenzymes (Altenbach and Sarnthein 1989). Jannink et al. (1998) proposed that this taxon can survive in low food supply and organic areas without any relation to oxygen paucity. Geslin et al. (2004) reported that the responses of this species vary with the oxygen condition of the deep-sea environment. A close relation of U. peregrina with the continuous organic carbon flux irrespective of the oxygen level was also reported southern Atlantic Ocean (Mackensen et al. 1995). The dominance of this species is higher at the Oxygen Minimum Zones (OMZ) reported from the northwestern Indian Ocean (Hermelin and Shimmield 1990), the northeastern Indian Ocean (Arabian Sea) with high organic flux (Jannink et al. 1998). De and Gupta (2010) reveals that the species U. peregrina is associated with warm bottom water, intermediate to high organic carbon, high surface productivity and low oxygen level in the northwest Arabian Sea. At ODP Hole 1138A, the U. peregrina encountered the warm water interruptions at ~ 32.2 Ma and ~ 31.6 Ma (Table 1; Fig. 4). In comparison, this species is reported to be less common or absent at ODP sites 747, 757, and 758 in the eastern Indian Ocean during the Oligocene interval (Nomura 1995).
The relative abundance of species has been also correlated with diversity indices which change their abundance as the temperature fluctuation occurs at the bottom of the ocean (Shimadzu et al. 2013). The successive peaks of P. bulloides and N. umbonifer (Fig. 4) indicate a change in the environment from the well-ventilated low organic flux to the dominance of carbonate-corrosive AABW (Nomura 1995; Bordiga et al. 2015). The ups and down in the diversity parameters from ~ 33.7 Ma to ~ 33.1 Ma suggests that conditions were unstable during this interval. This event coincides with one of the most abrupt changes during the earliest Oligocene between ~ 33.7 to ~ 33.4 Ma, known as the EOGM (Oi-1, Zachos et al. 1993). The oxygen isotope (δ18O) values also show an abrupt increase from 0.48 to 1.89‰ during this interval (Fig. 3). The change in isotopic values during this interval demonstrates that the continental ice suddenly appeared or increased on the Antarctic continent with a decrease in regional sea surface temperature (Kennett and Shackleton 1976; Zachos et al. 2001; Coxall et al. 2005; Lee and Jo 2019). This glaciation event was a result of the establishment of strong thermal isolation between the high (Antarctic continent) to the low latitudes and followed by opening of the Drake Passage and the establishment of the Antarctic Circumpolar Current (ACC) (Mackensen and Ehrmann 1992; Zachos et al. 2001; Lawver and Gahagan 2003; Livermore et al. 2007; Sijp et al. 2009), and hopefully polar front around Antarctica was more stronger at ~ 33.4 Ma. The cooling of the Antarctica as well as surrounding area by diverting warm western boundary currents away from the high southern latitudes (Salamy and Zachos 1999) further facilitated the ice buildup in the southern pole. The first appearance of species A. echolsi is recorded at this interval (Fig. 4), which showed significant abundance during the Oi-1, and a similar faunal event was also noted by Nomura (1995). This event occurred just after the EOT indicating a profound global change from a warm Eocene greenhouse to an icehouse Oligocene. The carbon isotopic (δ13C) record also shows an abrupt increase from 0.61 to 1.63‰. The increment in δ13C during this interval demonstrates as increased regional productivity. O. umbonatus showed a significant peak with a higher diversity index during the Oi-1 and Oi-1a interval (Fig. 4). This species also indicates the increased influence of AABW, intermediate to the high food supply, and presence of cold deep water in the Southern Ocean of the Indian sector. However, Oi-1 and Oi-1a were a phase change environment of the deep-sea benthic foraminifera, which maximizes surface water productivity in the Southern Ocean (Salamy and Zachos 1999; Diester-Haass and Zahn 2001).
The Shannon–weaver index [H(S)], Hurlbert's diversity index (S100), species richness (S), equitability (\(E{\prime}\)), and fisher’s alpha index (α) showed high values at ~ 33.7, ~ 33.4, ~ 32.8 and ~ 31.7 Ma. Also, a decreasing trend of species richness and fisher’s alpha index have been observed after ~ 31.7 Ma. The variability in diversity indices express the change in the bottom water conditions from warm to cold and vice versa. This time interval coincides with the progressive influence of ACC, AABW, and AAIW due to opening Drake Passage which is responsible thermal isolation of Antarctic landmass from rest of the world. The ACC intensified, and AAIW flow increased during this interval, which enhanced erosional strength (Pagani et al. 2000) and was marked by numerous hiatuses that appeared on Kerguelen Plateau (Barker and Burrell 1977). The dotted line marks hiatus at the study site (Fig. 3) are indicative of intense circulation due production of AABW. Again, the parameters H(S), S100, S, E′, and α decrease up to ~ 33.1 Ma after that, showing a gradual increase from ~ 33.1 Ma to 32.8 Ma. After 32.8 Ma, all the diversity indices showed a constant increase up to 32.0 Ma, and after that, a sharp decrease in the parameters followed by a high peak at ~ 31.7 Ma and an increasing pattern up to ~ 33.1 Ma, coinciding with the Early Oligocene Postglacial Maximum (EOPM; 33.1 Ma to ~ 32.6 Ma) (Coxall and Wilson 2011). These positive excursions of the different diversity indices indicate the opening of Drake Passage and the abrupt formation of the Antarctic ice sheet, while the negative peaks show the deglaciation period (Miller et al. 1991; Pälike et al. 2006; Lee and Jo 2019) that correlated with short warming intervals.
During EOGM, the marine environment gradually changed, and conditions became more stable as the species diversity also showed increased values (Fig. 3), coinciding with decreased productivity. δ18O values of Cibicidoides spp. at ~ 33.0 Ma, a recovery phase may be triggered by a deglaciation event, causing a rising sea level. The carbon isotopic values also show decreasing δ13C values (Fig. 3). This event also marks the high abundance of C. kullenbergi, with an increase in the number of species as well as species diversity parameters. However, a brief interval of increased abundance of N. umbonifer at ~ 32.8 Ma was noticed (Fig. 4). This sudden increased abundance of N. umbonifer indicates the presence of more corrosive bottom waters or less arrival of food at the seafloor or both after the second step of cooling (Fig. 4). At the same time interval (~ 32.8 Ma), the species O. umbonatus shows a high abundance (~ 18%), representing the cold and corrosive environments with high oxygen bottom conditions due. The low occurrence of G. subglobosa from ~ 32.2 Ma to ~ 31.5 Ma indicates the expansion of the Oxygen Minimum Zone (OMZ), eutrophic, anoxic, and stagnant (Fig. 4). U. peregrina (Fig. 4) is inferred to indicate the warm deep water and intermediate to high organic carbon, high surface productivity, and low oxygen level associated with OMZ. It was shown in high abundance from 32.2 to 31.5 Ma. Simultaneously, higher occurrence of these species indicates a mixed type of deep environment. After ~ 32.2 Ma, it is observed that the species diversity index drastically decreases with an increase in δ13C values indicating higher productivity (Fig. 3). This event is recorded in the species A. echolsi and O. umbonatus (Fig. 4). This event at ~ 31.7 Ma was also known as Oi-1b (Lee and Jo 2019) in Oligocene. The high abundance of G. subglobosa from ~ 31.5 to ~ 31.2 Ma indicates a well-oxygenated, warmer temperature with a low to intermediate food supply (Fig. 4). This interval was recognized as the increasing δ13C with diversity indices. We have also correlated our diversity index data to the bulk calcium carbonate (CaCO3) content data of ODP site 748B (Zachos et al. 1992) and the stable isotopic data of ODP Hole 744A (Salamy and Zachos 1999) of the Southern Kerguelen Plateau (Figs. 3 and 4). The stable isotope data of ODP Hole 1138A and 744A reveals a decreasing trend in δ18O, whereas δ13C values exhibit a decreasing trend up to ~ 32 Ma, followed by an increasing trend (Fig. 3). These data show a decreasing trend from ~ 33.7 to ~ 33.1 Ma to a gradual increase in productivity, confirmed by the increase of stable carbon isotopic values. The peak at ~ 33.7 Ma and ~ 33.4 Ma relates to the Oi-1 event, whereas ~ 32.8 Ma and ~ 31.7 Ma coincide with Oi-1a and Oi-1b events, respectively, of early Oligocene (Fig. 3). In contrast, the carbonate contents of whole-rock samples from ODP site 748B reveals the decreasing trend from ~ 33.7 Ma followed by an increasing trend which attains maximum peak at ~ 33.2 Ma (Fig. 3); it starts decreasing again and got minimum value at ~ 32.7 Ma followed by increasing trend up to the end of the studied interval. These fluctuations in bulk calcium carbonate content in whole rock data also suggest that the influence of substantially warm water bathed the area for a brief periods during the studied interval (Fig. 3).
Ocean circulation during early Oligocene
We interpreted that the Oligocene oceanographic setting of the Southern Ocean was very different to the modern ocean. The deep water mass was dominated by AABW, intermediate water mass was dominated by AAIW, and the upper part of the Southern Ocean was dominated by surface water mass (Fig. 5). Kennett and Stott (1990) reveal the strong influence of the proto-AABW and Antarctic Intermediate Water (AAIW) on the Southern Ocean during this time; however, this study reveals that at this interval, AABW came in to full existence (Figs. 3, 4, and 5). The cooling climate initiated the formation of cold and dense bottom water (AABW), followed by sea and continental ice formation in the early Oligocene. These water masses led to the bathing of the Kerguelen Plateau and its surroundings in a stepwise manner during the earliest Oligocene and predominantly influenced the deep-sea benthic foraminifera as shown in Figs. 3 and 4. Our findings suggested that diversity-based indices and isotopic data derived from the benthic foraminifera quickly responded to the deep water mass variation during the early Oligocene and leads to production and dominance of AABW in Southern Ocean.
A general schematic section of meridional ocean water circulation of the Southern Ocean illustrates surface and deep water circulation patterns during early Oligocene. Acronyms: AABW, Antarctic Bottom Water (modified after Kennett and Stott 1990)
Conclusions
By most standards, the rapid cooling of the sub-polar seas and the appearance of a full-scale continental ice sheet on Antarctica was a major climate event that should have had a significant impact on Southern Ocean ecology and global climate. The isotopic records and the diversity parameters of early Oligocene benthic foraminiferal species such as Shannon–Weaver (H(S)) index, Hurlbert’s (S100) diversity index, Fisher’s alpha (α) index, equitability (E′), and species richness (S) combined with population abundance of dominating benthic foraminifera at ODP Hole 1138A are correlated with the increased glaciation on the Antarctic and related ocean wide productivity. Species diversity decreased during the glaciation and increased during interglacial intervals in the Southern Ocean. The decrease in the species diversity parameters of the Southern Ocean is also related to the occurrences of low oxygen species and high seasonality as observed in the North Atlantic and Southeastern Indian Oceans (Corliss et al. 2009; Singh et al. 2012). The increase in the species diversity was observed between Oi-1 (33.4 Ma) and Oi-1b (31.7 Ma) events due to the release of the short-lived pulse of CO2 at different phases due to leakage of fossil carbon (Coxall et al. 2018) or release of trap gas hydrates (i.e., methane, ethane, or carbon dioxide) or short-lived volcanic eruptions, which release CO2 and warm the deep ocean. The low values of species diversity amplified by high seasonality and relatively cold, strong bottom water currents after the Eocene–Oligocene Transition (33.9 Ma) and after the Oi-1b (31.7 Ma) event correspond to the substantial buildup of the Southern Hemisphere glaciation and intensification of ACC and AABW, and early Oligocene have also noticed marked intervals of warming.
Data availability
The authors declare that the data supporting the findings of this study are available within the article and its supplementary information files. Also, all the core samples and all raw datasets were systematically cataloged and lodged in the Micropaleontology Laboratory, Department of Earth Sciences, Indian Institute of Technology Roorkee (IITR), Uttarakhand.
Code availability
Not applicable.
References
Almogi-Labin A, Schmiedl G, Hemleben C, Siman-Tov R, Segl M, Meischner D (2000) The influence of the NE winter monsoon on productivity changes in the Gulf of Aden, NW Arabian Sea, during the last 530 ka as recorded by foraminifera. Marine micropaleontology 40:295–319. https://doi.org/10.1016/S0377-8398(00)00043-8
Altenbach AV, Sarnthein M (1989) Productivity record in benthic foraminifera, In: Berger, W.H., Smetacek, V.S., Wefer, G. (Eds.), Productivity of the oceans: present and past. Springer-Verlag, New York 8:255–269
Anagnostou E, John EH, Edgar KM, Foster GL, Ridgwell A, Inglis GN, Pancost RD, Lunt DJ, Pearson PN (2016) Changing atmospheric CO2 concentration was the primary driver of early Cenozoic climate. Nature 533:380–384. https://doi.org/10.1038/nature17423
Apel M, Kiessling W, Bohm F, Lazarus D (2002) Radiolarian faunal characteristics in Oligocene sediments of the Kerguelen Plateau, Leg 183, Site 1138. Proc Ocean Drill Program Sci Results 183:48
Armstrong McKay DI, Tyrrell T, Wilson PA (2016) Global carbon cycle perturbation across the Eocene-Oligocene climate transition. Paleoceanography 31:311–329. https://doi.org/10.1002/2015PA002818
Barker PF, Burrell J (1977) The opening of Drake Passage. Mar Geol 25:15–34. https://doi.org/10.1016/0025-3227(77)90045-7
Berggren WA, Miller KG (1989) Cenozoic bathyal and abyssal calcareous benthic foraminiferal zonation. Micropaleontology 35:308–320. https://doi.org/10.2307/1485674
Bhaumik AK, Gupta AK, Sundar Raj M, Mohan K, De S, Sarkar S (2007) Paleoceanographic evolution of the northeastern Indian Ocean during the Miocene: evidence from deep-sea benthic foraminifera (DSDP Hole 216A). Indian J Mar Sci 36:332–341
Bohaty SM, Zachos JC, Delaney ML (2012) Foraminiferal Mg/Ca evidence for Southern Ocean cooling across the Eocene-Oligocene transition. Earth Planet Sci Lett 317–318:251–261. https://doi.org/10.1016/j.epsl.2011.11.037
Bordiga M, Henderiks J, Tori F, Monechi S, Fenero R, Legarda-Lisarri A, Thomas E (2015) Microfossil evidence for trophic changes during the Eocene-Oligocene transition in the South Atlantic (ODP Site 1263, Walvis Ridge). Clim Past 11:1249–1270. https://doi.org/10.5194/cp-11-1249-2015
Bouchet VMP, Alve E, Rygg B, Telford RJ (2012) Benthic foraminifera provide a promising tool for ecological quality assessment of marine waters. Ecol Indic 23:66–75. https://doi.org/10.1016/j.ecolind.2012.03.011
Bremer ML, Lohmann GP (1982) Evidence for primary control of the distribution of certain Atlantic Ocean benthonic foraminifera by degree of carbonate saturation. Deep Sea Res. Part A. Oceanogr Res Pap 29:987–998. https://doi.org/10.1016/0198-0149(82)90022-X
Burke SK, Berger WH, Coulbourn WT, Vincent E (1993) Benthic foraminifera in box core ERDC 112, Ontong Java Plateau. J Foraminifer Res 23:19–39. https://doi.org/10.2113/gsjfr.23.1.19
Buzas MA, Gibson TG (1969) Species diversity: benthonic foraminifera in Western North Atlantic. Science 163:72–75. https://doi.org/10.1126/science.163.3862.72
Corliss BH (1979) Response of deep-sea benhtonic foraminifera to development of psychrosphere near Eocene/Oligocene boundary. Nature 282:63–65
Corliss BH (1983) Distribution of Holocene deep-sea benthonic foraminifera in the southwest Indian Ocean. Deep Sea Res Part A Oceanogr Res Pap 30:95–117. https://doi.org/10.1016/0198-0149(83)90064-X
Corliss BH, Brown CW, Sun X, Showers WJ (2009) Deep-sea benthic diversity linked to seasonality of pelagic productivity. Deep Res Part I Oceanogr Res Pap 56:835–841. https://doi.org/10.1016/j.dsr.2008.12.009
Coxall HK, Wilson PA (2011) Early Oligocene glaciation and productivity in the eastern equatorial Pacific: insights into global carbon cycling. Paleoceanography 26:n/a-n/a. https://doi.org/10.1029/2010PA002021
Coxall HK, Wilson PA, Pälike H, Lear CH, Backman J (2005) Rapid stepwise onset of Antarctic glaciation and deeper calcite compensation in the Pacific Ocean. Nature 433:53–57. https://doi.org/10.1038/nature03135
Coxall HK, Huck CE, Huber M, Lear CH, Legarda-Lisarri A, O’Regan M, Sliwinska KK, van de Flierdt T, de Boer AM, Zachos JC, Backman J (2018) Export of nutrient rich Northern Component Water preceded early Oligocene Antarctic glaciation. Nat Geosci 11:190–196. https://doi.org/10.1038/s41561-018-0069-9
De S, Gupta AK (2010) Deep-sea faunal provinces and their inferred environments in the Indian Ocean based on distribution of Recent benthic foraminifera. Palaeogeogr Palaeoclimatol Palaeoecol 291:429–442. https://doi.org/10.1016/j.palaeo.2010.03.012
DeConto RM, Pollard D (2003) Rapid Cenozoic glaciation of Antarctica induced by declining atmospheric CO2. Nature 421:245–249. https://doi.org/10.1038/nature01290
Diester-Haass L, Zahn R (2001) Paleoproductivity increase at the Eocene - Oligocene climatic transition: ODP/DSDP sites 763 and 592. Palaeogeogr Palaeoclimatol Palaeoecol 172:153–170. https://doi.org/10.1016/S0031-0182(01)00280-2
Eberwein A, Mackensen A (2006) Regional primary productivity differences off Morocco (NW-Africa) recorded by modern benthic foraminifera and their stable carbon isotopic composition. Deep Res Part I Oceanogr Res Pap 53:1379–1405. https://doi.org/10.1016/j.dsr.2006.04.001
Fariduddin M, Loubere P (1997) The surface ocean productivity response of deeper water benthic foraminifera in the Atlantic Ocean. Mar Micropaleontol 32:289–310. https://doi.org/10.1016/S0377-8398(97)00026-1
Fentimen R, Lim A, Rüggeberg A, Wheeler AJ, Van Rooij D, Foubert A (2020) Impact of bottom water currents on benthic foraminiferal assemblages in a cold-water coral environment: the Moira Mounds (NE Atlantic). Mar Micropaleontol 154:101799. https://doi.org/10.1016/j.marmicro.2019.101799
Fischer A (1960) Latitudinal Variations in Organic Diversity. Evolution 14:64. https://doi.org/10.2307/2405923
Frey FA, Coffin MF, Wallace PJ, Weis D (2003) Leg 183 summary: Kerguelen Plateau-Broken Ridge—a large Igneous Province. Proc Ocean Drill Program 183 Initial Rep 183:1–48. https://doi.org/10.2973/odp.proc.ir.183.101.2000
Geslin E, Heinz P, Jorissen F, Hemleben C (2004) Migratory responses of deep-sea benthic foraminifera to variable oxygen conditions: laboratory investigations. Mar Micropaleontol 53:227–243. https://doi.org/10.1016/j.marmicro.2004.05.010
Giusberti L, Boscolo Galazzo F, Thomas E (2016) Variability in climate and productivity during the Paleocene-Eocene Thermal Maximum in the western Tethys (Forada section). Clim Past 12:213–240. https://doi.org/10.5194/cp-12-213-2016
Gooday AJ, Bernhard JM, Levin LA, Suhr SB (2000) Foraminifera in the Arabian Sea oxygen minimum zone and other oxygen-deficient settings: taxonomic composition, diversity, and relation to metazoan faunas. Deep Res Part II Top Stud Oceanogr 47:25–54. https://doi.org/10.1016/S0967-0645(99)00099-5
Gooday AJ, Bett BJ, Escobar E, Ingole B, Levin LA, Neira C, Raman AV, Sellanes J (2010) Habitat heterogeneity and its influence on benthic biodiversity in oxygen minimum zones. Mar Ecol 31:125–147. https://doi.org/10.1111/j.1439-0485.2009.00348.x
Gradstein FM, Ogg JG, Hilgen FJ (2012) The Geologic Time Scale 2012. Elsevier. https://doi.org/10.1127/0078-0421/2012/0020
Gupta AK (1994) Taxonomy and bathymetric distribution of Holocene deep-sea benthic foraminifera in the Indian Ocean and the Red Sea. Micropaleontology 40:351–367. https://doi.org/10.2307/1485940
Gupta AK, Thomas E (1999) Latest Miocene-Pleistocene productivity and deep-sea ventilation in the northwestern Indian Ocean (Deep Sea Drilling Project Site 219). Paleoceanography 14:62–73. https://doi.org/10.1029/1998PA900006
Gupta AK, Thomas E (2003) Initiation of Northern Hemisphere glaciation and strengthening of the northeast Indian monsoon: ocean drilling program site 758, eastern equatorial Indian Ocean. Geology 31:47–50. https://doi.org/10.1130/0091-7613(2003)031%3c0047:IONHGA%3e2.0.CO;2
Gupta AK, Das M, Bhaskar K (2006) South Equatorial Current (SEC) driven changes at DSDP Site 237, Central Indian Ocean, during the Plio-Pleistocene: evidence from Benthic Foraminifera and Stable Isotopes. J Asian Earth Sci 28:276–290. https://doi.org/10.1016/j.jseaes.2005.10.006
Haumann FA, Moorman R, Riser SC, Smedsrud LH, Maksym T, Wong APS, Wilson EA, Drucker R, Talley LD, Johnson KS, Key RM, Sarmiento JL (2020) Supercooled Southern Ocean Waters. Geophys Res Lett 47:e2020GL090242. https://doi.org/10.1029/2020GL090242
Hermelin JOR, Shimmield GB (1990) The importance of the oxygen minimum zone and sediment geochemistry in the distribution of Recent benthic foraminifera in the northwest Indian Ocean. Mar Geol 91:1–29. https://doi.org/10.1016/0025-3227(90)90130-C
Holbourn A, Hrnderson A, Macleod N (2013) Atlas of benthic foraminifera. Natural History Museum, UK. https://doi.org/10.1002/9781118452493
Jannink NT, Zachariasse WJ, van der Zwaan GJ (1998) Living (Rose Bengal stained) benthic foraminifera from the Pakistan continental margin (northern Arabian Sea). Deep Res Part I Oceanogr Res Pap 45:1483–1513. https://doi.org/10.1016/S0967-0637(98)00027-2
Jorissen FJ, Fontanier C, Thomas E (2007) Paleoceanographical proxies based on deep-sea benthic foraminiferal assemblage characteristics. Developments in Marine Geology 1:263–325. https://doi.org/10.1016/S1572-5480(07)01012-3
Kaiho K (1994) Benthic foraminiferal dissolved oxygen index and dissolved oxygen levels in the modern ocean. Geology 22:719–722
Kaiho K (1999) Effect of organic carbon flux and dissolved oxygen on the benthic foraminiferal oxygen index (BFOI). Mar Micropaleontol 37:67–76. https://doi.org/10.1016/S0377-8398(99)00008-0
Katz ME, Miller KG, Wright JD, Wade BS, Browning JV, Cramer BS, Rosenthal Y (2008) Stepwise transition from the Eocene greenhouse to the Oligocene icehouse. Nat Geosci 1:329–334. https://doi.org/10.1038/ngeo179
Kender S, Aturamu A, Zalasiewicz J, Kaminski MA, Williams M (2019) Benthic foraminifera indicate Glacial North Pacific Intermediate Water and reduced primary productivity over Bowers Ridge, Bering Sea, since the Mid-Brunhes Transition. J Micropalaeontol 38:177–187. https://doi.org/10.5194/jm-38-177-2019
Kennett JP (1977) Cenozoic evolution of Antarctic glaciation, the circum-Antarctic ocean, and their impact on global paleoceanography. J Geophys Res 82:3843–3860. https://doi.org/10.1029/jc082i027p03843
Kennett JP, Shackleton NJ (1976) Oxygen isotopic evidence for the development of the psychrosphere 38 Myr ago. Nature 260:513–515. https://doi.org/10.1038/260513a0
Kennett JP, Stott LD (1990) Proteus and proto-oceanus: ancestral Paleogene oceans as revealed from Antarctic stable isotopic results; ODP Leg 113. Proc Sci Results ODP Leg 113 Wedd Sea Antarct 113:865–880. https://doi.org/10.2973/odp.proc.sr.113.188.1990
Klootwijk AT, Alve E (2022) Does the analysed size fraction of benthic foraminifera influence the ecological quality status and the interpretation of environmental conditions? Indications from two northern Norwegian fjords. Ecol Indic 135:108423. https://doi.org/10.1016/j.ecolind.2021.108423
Kumar R, Maurya AS, Singh DP (2021) Effect of early Oligocene cooling on the deep-sea benthic foraminifera at IODP hole 1138A, Kerguelen Plateau (Southern Ocean). EGU EGU21–6414. https://doi.org/10.5194/egusphere-egu21-6414
Lawver LA, Gahagan LM (2003) Evolution of cenozoic seaways in the circum-antarctic region. Palaeogeogr Palaeoclimatol Palaeoecol 198:11–37. https://doi.org/10.1016/S0031-0182(03)00392-4
Lee H, Jo K (2019) Oligocene paleoceanographic changes based on an interbasinal comparison of Cibicidoides spp. δ18O records and a new compilation of data. Palaeogeogr Palaeoclimatol Palaeoecol 514:800–812. https://doi.org/10.1016/j.palaeo.2018.09.016
Li Q, Ruhl M, Wang YD, Xie XP, An PC, Xu YY (2022) Response of Carnian pluvial episode evidenced by organic carbon isotopic excursions from western Hubei, South China. Palaeoworld 31:324–333. https://doi.org/10.1016/j.palwor.2021.08.004
Livermore R, Hillenbrand CD, Meredith M, Eagles G (2007) Drake Passage and Cenozoic climate: an open and shut case? Geochem. Geophys. Geosyst. 8:Q01005. https://doi.org/10.1029/2005GC001224
Lohmann GP (1978) Abyssal benthonic foraminifera as hydrographic indicators in the western South Atlantic Ocean. J Foraminifer Res 8:6–34. https://doi.org/10.2113/gsjfr.8.1.6
Lu W, Rickaby REM, Hoogakker BAA, Rathburn AE, Burkett AM, Dickson AJ, Martínez-Méndez G, Hillenbrand CD, Zhou X, Thomas E, Lu Z (2020) I/Ca in epifaunal benthic foraminifera: a semi-quantitative proxy for bottom water oxygen in a multi-proxy compilation for glacial ocean deoxygenation. Earth Planet Sci Lett 533:116055. https://doi.org/10.1016/j.epsl.2019.116055
Lutze GF (1979) Benthic foraminifers at site 397: faunal fluctuations and ranges in the Quaternary. Initial reports Deep Sea Drill. Proj. Leg 47, Las Palmas, Canar. Islands to Vigo, Spain, 1976, (Scripps Inst. Oceanogr. UK Distrib. IPOD Committee, NERC, Swindon) 47:419–431. https://doi.org/10.2973/dsdp.proc.47-1.111.1979
Mackensen A, Berggren WA (1992) Paleogene benthic foraminifers from the southern indian ocean (kerguelen plateau): biostratigraphy and paleoecology. Proc. Ocean Drill. Program. Sci Results 120:603–631. https://doi.org/10.2973/odp.proc.sr.120.168.1992
Mackensen A, Ehrmann WU (1992) Middle Eocene through Early Oligocene climate history and paleoceanography in the Southern Ocean: stable oxygen and carbon isotopes from ODP Sites on Maud Rise and Kerguelen Plateau. Mar Geol 108:1–27. https://doi.org/10.1016/0025-3227(92)90210-9
Mackensen A, Grobe H, Kuhn G, Fiitterer DK (1990) Benthic foraminiferal assemblages from the eastern Weddell Sea between 68 and 73° S: distribution, ecology and fossilization potential. Mar Micropaleontol 16:241–283. https://doi.org/10.1016/0377-8398(90)90006-8
Mackensen A, Schmiedl G, Harloff J, Giese M (1995) Deep-sea foraminifera in the South Atlantic Ocean: ecology and assemblage generation. Micropaleontology 41:342. https://doi.org/10.2307/1485808
Maurya AS, Rai AK (2006) Paleoceanographic significance of Early Miocene deep sea benthic foraminifera at the Wombat Plateau, eastern Indian Ocean. Natl Acad Sci Lett 29:213–220
Mead GA, Kennett JP (1987) The distribution of recent benthic foraminifera in the Polar Front region, southwest Atlantic. Mar Micropaleontol 11:343–360. https://doi.org/10.1016/0377-8398(87)90006-5
Miao Q, Thunell RC (1993) Recent deep-sea benthic foraminiferal distributions in the South China and Sulu Seas. Mar Micropaleontol 22:1–32. https://doi.org/10.1016/0377-8398(93)90002-F
Miller KG, Wright JD, Fairbanks RG (1991) Unlocking the ice house: Oligocene-Miocene oxygen isotopes, eustasy, and margin erosion. J Geophys Res 96:6829–6848. https://doi.org/10.1029/90JB02015
Nagai RH, Sousa SHM, Burone L, Mahiques MM (2009) Paleoproductivity changes during the Holocene in the inner shelf of Cabo Frio, southeastern Brazilian continental margin: benthic foraminifera and sedimentological proxies. Quat Int 206:62–71. https://doi.org/10.1016/j.quaint.2008.10.014
Nomura R (1991) Oligocene to Pleistocene benthic foraminifer assemblages at Sites 754 and 756, eastern Indian Ocean. Proc., Sci. results, ODP, Leg 121. Broken Ridge Ninetyeast Ridge 121:31–75. https://doi.org/10.2973/odp.proc.sr.121.139.1991
Nomura R (1995) Paleogene to Neogene deep-sea paleoceanography in the Eastern Indian Ocean: benthic foraminifera from ODP sites 747, 757 and 758. Micropaleontology 41:251–290
O’Brien CL, Huber M, Thomas E, Pagani M, Super JR, Elder LE, Hull PM (2020) The enigma of Oligocene climate and global surface temperature evolution. Proc Natl Acad Sci U S A 117:25302–25309. https://doi.org/10.1073/pnas.2003914117
Pagani M, Arthur MA, Freeman KH (2000) Variations in Miocene phytoplankton growth rates in the Southwest Atlantic: evidence for changes in ocean circulation. Paleoceanography 15:486–496. https://doi.org/10.1029/1999PA000484
Pälike H, Norris RD, Herrle JO, Wilson PA, Coxall HK, Lear CH, Shackleton NJ, Tripati AK, Wade BS (2006) The heartbeat of the oligocene climate system. Science 314(80):1894–1898. https://doi.org/10.1126/science.1133822
Pälike H, Lyle MW, Nishi H, Raffi I, Ridgwell A, Gamage K, Klaus A, Acton G, Anderson L, Backman J et al (2012) A Cenozoic record of the equatorial Pacific carbonate compensation depth. Nature 488:609–614
Pälike H, Lyle M, Nishi H, Raffi I, Gamage K, Klaus A (2010) the Expedition 320/321 Scientists, 2010. Proceedings of the Integrated Ocean Drilling Program (IODP), 320/321. Integr. Ocean Drill. Progr Manag Int Tokyo 320/321:1–21. https://doi.org/10.2204/iodp.proc.320321.101.2010
Park YH, Vivier F, Roquet F, Kestenare E (2009) Direct observations of the ACC transport across the Kerguelen Plateau. Geophys Res Lett 36:L18603 https://doi.org/10.1029/2009GL039617
Pascual A, Rodríguez-Lázaro J, Martínez-García B, Varela Z (2020) Palaeoceanographic and palaeoclimatic changes during the last 37,000 years detected in the SE Bay of Biscay based on benthic foraminifera. Quat Int 566–567:323–336. https://doi.org/10.1016/j.quaint.2020.03.043
Rai AK, Maurya AS (2007) Paleoceanographic significance of changes in Miocene deep-sea benthic foraminiferal diversity on the Wombat Plateau, Eastern Indian Ocean. J Geol Soc India 70:837–845
Rai AK, Maurya AS (2009) Effect of miocene paleoceanographic changes on the benthic foraminiferal diversity at ODP site 754A (southeastern Indian Ocean). Indian J Mar Sci 38:423–431
Rathburn AE, Levin LA, Held Z, Lohmann KC (2000) Benthic foraminifera associated with cold methane seeps on the northern California margin: ecology and stable isotopic composition. Mar Micropaleontol 38:247–266. https://doi.org/10.1016/S0377-8398(00)00005-0
Ravelo AC, Hillaire-Marcel C (2007) Chapter eighteen the use of oxygen and carbon isotopes of foraminifera in paleoceanography. Developments in Marine Geology 1:735–764 https://doi.org/10.1016/S1572-5480(07)01023-8
Rintoul SR, Da Silva CE (2019) Antarctic circumpolar current, in: Encyclopedia of Ocean Sciences 3:248–261. https://doi.org/10.1016/B978-0-12-409548-9.11298-9
Rodrigues AR, Pivel MAG, Schmitt P, de Almeida FK, Bonetti C (2018) Infaunal and epifaunal benthic foraminifera species as proxies of organic matter paleofluxes in the Pelotas Basin, south-western Atlantic Ocean. Mar Micropaleontol 144:38–49. https://doi.org/10.1016/j.marmicro.2018.05.007
Salamy KA, Zachos JC (1999) Latest Eocene-Early Oligocene climate change and Southern Ocean fertility: inferences from sediment accumulation and stable isotope data. Palaeogeogr Palaeoclimatol Palaeoecol 145:61–77. https://doi.org/10.1016/S0031-0182(98)00093-5
Schnitker D (1974) West Atlantic abyssal circulation during the past 120,000 years. Nature 248:385–387. https://doi.org/10.1038/248385a0
Shimadzu H, Dornelas M, Henderson PA, Magurran AE (2013) Diversity is maintained by seasonal variation in species abundance. BMC Biol 11:98. https://doi.org/10.1186/1741-7007-11-98
Sijp WP, England MH, Toggweiler JR (2009) Effect of Ocean gateway changes under greenhouse warmth. J Clim 22:6639–6652. https://doi.org/10.1175/2009JCLI3003.1
Singh RK, Gupta AK (2004) Late Oligocene-Miocene paleoceanographic evolution of the southeastern Indian Ocean: evidence from deep-sea benthic foraminifera (ODP Site 757). Mar Micropaleontol 51:153–170. https://doi.org/10.1016/j.marmicro.2003.10.003
Singh RK, Gupta AK, Das M (2012) Paleoceanographic significance of deep-sea benthic foraminiferal species diversity at southeastern Indian Ocean Hole 752A during the Neogene. Palaeogeogr Palaeoclimatol Palaeoecol 361–362:94–103. https://doi.org/10.1016/j.palaeo.2012.08.008
Singh DP, Saraswat R, Nigam R (2021) Untangling the effect of organic matter and dissolved oxygen on living benthic foraminifera in the southeastern Arabian Sea. Mar Pollut Bull 172:112883. https://doi.org/10.1016/j.marpolbul.2021.112883
Singh DP, Saraswat R, Pawar R (2022) Distinct environmental parameters influence the abundance of living benthic foraminifera morphogroups in the southeastern Arabian Sea. Environ Sci Pollut Res 1:1–18. https://doi.org/10.1007/s11356-022-21492-4
Speer K, Rintoul SR, Sloyan B (2000) The diabatic Deacon cell. J Phys Oceanogr 30:3212–3222. https://doi.org/10.1175/1520-0485(2000)030%3c3212:TDDC%3e2.0.CO;2
Streeter SS (1973) Bottom water and benthonic foraminifera in the North Atlantic-Glacial-interglacial contrasts. Quat Res 3:131–141. https://doi.org/10.1016/0033-5894(73)90059-8
Streeter SS, Shackleton NJ (1979) Paleocirculation of the Deep North Atlantic: 150,000-Year Record of Benthic Foraminifera and Oxygen-18. Science 203:168–171. https://doi.org/10.1126/science.203.4376.168
Takata H, Nomura R, Tsujimoto A, Khim BK (2012) Late early Oligocene deep-sea benthic foraminifera and their faunal response to paleoceanographic changes in the eastern Equatorial Pacific. Mar Micropaleontol 96–97:123–132. https://doi.org/10.1016/j.marmicro.2012.09.002
Tang H, Li SF, Su T, Spicer RA, Zhang ST, Li SH, Liu J, Lauretano V, Witkowski CR, Spicer TEV, Deng WYD, Wu MX, Ding WN, Zhou ZK (2020) Early Oligocene vegetation and climate of southwestern China inferred from palynology. Palaeogeogr Palaeoclimatol Palaeoecol 560:109988. https://doi.org/10.1016/j.palaeo.2020.109988
Thomas E (1992) Middle Eocene - Late Oligocene bathyal benthic foraminifera (Weddell Sea): faunal changes and implications for ocean circulation. In: Prothero DA, Berggren WA (eds) Eocene-Oligocene Climatic and Biotic Evolution. Princeton Unive, Princeton, pp 245–271
Thomas E, Gooday A (1996) Cenozoic deep-sea benthic foraminifers: tracers for changes in oceanic productivity? Geology 24:355–358. https://doi.org/10.1130/0091-7613(1996)024%3c0355:CDSBFT%3e2.3.CO;2
Tjalsma RC (1983) Eocene to Miocene benthic foraminifers from deep sea drilling project site 516, Rio Grande Rise, South Atlantic. Initial Rep DSDP Leg 72 Santos Brazil 72:731–755
Uchio T (1960) Ecology of living benthonic foraminifera from the Sand Diego, California Area. Cushman Found Foraminifer Res 5:5–70
van der Zwaan GJ, Duijnstee IAP, Den Dulk M, Ernst SR, Jannink NT, Kouwenhoven TJ (1999) Benthic foraminifers: proxies or problems? A review of paleocological concepts. Earth Sci Rev 46:213–236. https://doi.org/10.1016/S0012-8252(99)00011-2
Wade BS, Pearson PN, Berggren WA, Pälike H (2011) Review and revision of Cenozoic tropical planktonic foraminiferal biostratigraphy and calibration to the geomagnetic polarity and astronomical time scale. Earth-Science Rev 104:111–142. https://doi.org/10.1016/j.earscirev.2010.09.003
Wang J, Mazloff MR, Gille ST (2016) The effect of the Kerguelen Plateau on the ocean circulation. J Phys Oceanogr 46:3385–3396. https://doi.org/10.1175/JPO-D-15-0216.1
Woodruff F (1985) Changes in Miocene deep-sea benthic foraminiferal distribution in the Pacific Ocean: relationship to paleoceanography. Mem Geol Soc Am 163:131–175. https://doi.org/10.1130/MEM163-p131
Woodruff F, Savin SM (1989) Miocene deep-water oceanography. Paleoceanography 4:87–140. https://doi.org/10.1029/PA004i001p00087
Wright NM, Scher HD, Seton M, Huck CE, Duggan BD (2018) No change in Southern Ocean circulation in the Indian Ocean from the Eocene through Late Oligocene. Paleoceanogr Paleoclimatology 33:152–167. https://doi.org/10.1002/2017PA003238
Zachos JC, Lohmann KC, Walker JCG, Wise SW (1993) Abrupt climate change and transient climates during the Paleogene: a marine perspective. J Geol 101:191–213. https://doi.org/10.1086/648216
Zachos JC, Pagani H, Sloan L, Thomas E, Billups K (2001) Trends, rhythms, and aberrations in global climate 65 Ma to present. Science 292:686–693. https://doi.org/10.1126/science.1059412
Zachos JC, Berggren WA, Aubry MP, Mackensen A (1992) Isotope and trace element geochemistry of Eocene and Oligocene foraminifers from site 748, Kerguelen Plateau. Proc. Ocean Drill. Program, 120:839–854 https://doi.org/10.2973/odp.proc.sr.120.183.1992
Funding
The authors acknowledge the Ocean Drilling Program (ODP) for providing samples to ASM against sample request number 22594A for the present study; the Council of Scientific and Industrial Research, New Delhi (Govt. of India), for financial support, project number 24(0331)/14/EMR-II; and Indian Institute of Technology Roorkee (IIT Roorkee) for infrastructural facilities. RK is thankful to IIT Roorkee for financial support as MHRD fellowship.
Author information
Authors and Affiliations
Contributions
ASM conceived the presented idea. ASM provides the sample, ASM and RK performs the data processing, analysis, interpretation and computations. ASM and DPS encouraged RK to investigate the response of benthic foraminifera during the early Oligocene. RK wrote the manuscript with support from DPS and ASM. DPS and ASM did the critical revision of the article. All authors discussed the results and contributed to the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
The authors give consent for the publication of identifiable details, which can include photograph(s) and/or data and/or case history and/or details within the text to be published in the above journal and article.
Consent for publication
The authors confirm that this work is original and has not been published elsewhere, nor is it currently under consideration for publication elsewhere.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumar, R., Singh, D.P. & Maurya, A.S. Deep-sea benthic foraminiferal response to the early Oligocene cooling: a study from the Southern Ocean ODP Hole 1138A. Geo-Mar Lett 43, 11 (2023). https://doi.org/10.1007/s00367-023-00753-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00367-023-00753-2