Abstract
Methionine (Met) has important functions for homeostasis of various species, including zebrafish. However, the increased levels of this amino acid in plasma, a condition known as hypermethioninemia, can lead to cell alterations. Met is crucial for the methylation process and its excesses interfere with the cell cycle, an effect that persists even after the removal of this amino acid. Some conditions may lead to a transient increase of this amino acid with unexplored persistent effects of Met exposure. In the present study, we investigated the behavioral and neurochemical effects after the withdrawal of Met exposure. Zebrafish were divided into two groups: control and Met-treated group (3 mM) for 7 days and after maintained for 8 days in tanks containing only water. In the eighth day post-exposure, we evaluated locomotion, anxiety, aggression, social interaction, and memory, as well as oxidative stress parameters, amino acid, and neurotransmitter levels in the zebrafish brain. Our results showed that 8 days after Met exposure, the treated group showed decreased locomotion and aggressive responses, as well as impaired aversive memory. The Met withdrawal did not change thiobarbituric acid reactive substances, reactive oxygen species, and nitrite levels; however, we observed a decrease in antioxidant enzymes superoxide dismutase, catalase, and total thiols. Epinephrine and cysteine levels were decreased after the Met withdrawal whereas carnitine and creatine levels were elevated. Our findings indicate that a transient increase in Met causes persistent neurotoxicity, observed by behavioral and cognitive changes after Met withdrawal and that the mechanisms underlying these effects are related to changes in antioxidant system, amino acid, and neurotransmitter levels.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Amino acids play an important role in behavioral neurobiology since they are present in various metabolic cycles and altered levels can contribute to behavioral changes such as aggression and psychopathy [1]. Methionine (Met) is an essential sulfur-containing amino acid responsible for the formation of cysteine and carnitine [2] and, through cysteine, the origin of glutathione and taurine [3]. Met also has modulatory properties in GABAergic signaling [4]. In addition, creatine and epinephrine have its methyl group derived from Met [5], as well as serotonin and dopamine [6]. Met has an important role as the universal amino acid for translation initiation [7] and is a precursor of S-adenosylmethionine, the main donor of methyl groups for various molecules such as nucleic acids, histones, and lipid derivatives [8]. Met is also involved in biological processes such as growth, development, and immune response [2]. The maintenance of its amount in the organism is a matter of concern not only for homeostasis but also from the toxicological point of view since Met is considered the most toxic amino acid according to animal studies [9]. Changes in Met plasma levels have been related to the human liver and, especially to cerebral, disorders [10,11,12,13]. Some patients remain asymptomatic even with moderate plasma Met levels [14], but perhaps behavioral changes with no apparent cause may be due to an imbalance in the levels of this amino acid or its metabolites. The connection between the metabolites of Met, S-adenosylmethionine, and homocysteine, and behavioral changes has been already shown [6].
The hypermethioninemia is defined as an excess of Met in the blood at levels above 40 μMol/L [15]. This is a pathological condition caused by excessive consumption of Met, due to supplementation or a high protein diet, liver disease, premature birth, or can occur in case of an innate metabolism error, when Met is not properly metabolized [16]. Inherited hypermethioninemia that is not associated with other metabolic disorders can be caused by an impairment of enzymes such as Methionine adenosyltransferase (MAT) [17], glycine N-methyltransferase (GNMT) [18], and S-adenosyl-homocysteine hydrolase (AHCY) [19]. Despite that, hypermethioninemia can occur due to a secondary metabolic disorder like homocystinuria induced by cystathionine beta-synthase (CBS) deficiency [20], tyrosinemia type I with mutations in the fumarylacetoacetate hydrolase (FAH) gene [21], and citrin deficiency [22]. However, the mechanisms related to neurological effects induced by hypermethioninemia are still not completely elucidated [23]. Stefanello et al. [24] demonstrated increased acetylcholinesterase (AChE) activity in the cerebral cortex of developing Wistar rats after chronic treatment with Met, which could be associated with impaired working memory performance. Met exposure also caused changes in glutamate uptake and adenosine nucleotide catabolism and in the ectonucleotidase and adenosine receptor gene expression in zebrafish brain tissues [25]. Besides, l-Met inhibits proliferation and interferes with the cell cycle of pancreatic cancer cells that remained after Met withdrawal, indicating a persistent effect of l-Met exposure [26].
Methionine is indispensable for homeostasis of the organism’s redox control, and its supplementation or restriction can impact the antioxidant capacity [2]. High levels of Met caused several biochemical changes that can influence the body’s antioxidant control [27,28,29]. Soares et al. showed that acute administration of Met induces oxidative damage and apoptosis in the cerebral cortex of rats [27]. Stefanello et al. showed that chronic subcutaneous injections of Met promote an increase of the thiobarbituric acid reactive substances (TBARs) in rat hippocampus [29] and increased inflammatory cell number and toxicity in the liver [28]. Thus, oxidative stress was pointed out as a possible mechanism involved in neurological diseases caused by hypermethioninemia.
Zebrafish (Danio rerio) has been considered an interesting model organism for several studies due to its high compatibility of physiological pathways and organs with mammals [30] and has a sequenced genome, showing high similarity with about 84% of disease-related genes in humans [31]. This species has a well-described central nervous system, presenting neurotransmission systems such as dopaminergic, glutamatergic, and serotonergic similar to mammals [32]. Many studies take advantage of zebrafish’s human translatability, facility of use, and low cost to study this animal as a model of innate errors of metabolism and amino acid disorders, including hypermethioninemia [33,34,35,36,37]. Also, zebrafish have a physiological response to stress and exhibit social and cognitive behavior and highly complex interactions such as shoaling [38, 39]. Several studies have been developed evaluating zebrafish behavioral characteristics such as locomotor activity, aggression, social interaction, learning, and memory [40,41,42].
The influence of higher levels of Met in neurotoxic processes is well understood [23]. However, behaviors that involve its misbalance have not yet been explored. In this study, we performed a wide behavioral analysis of Met-exposed zebrafish and evaluated the anxiety, social interaction, aggression, spatial, and aversive memory. The main compounds for which their biosynthesis is influenced by Met were analyzed in parallel to the determination of oxidative stress parameters. We also investigated the effects of Met’s withdrawal on behavioral and biochemical parameters in adult zebrafish to explore and discuss the mechanisms that underlie Met persistent toxic effects.
Materials and Methods
Animals
A total of 288 adult wild-type zebrafish (5–6 months, 0.2–0.4 g) from the AB background were used. The animals were obtained from our breeding colony. Until the treatment, the animals were maintained in recirculating systems (Zebtec, Tecniplast, Italy) with reverse osmosis–filtered water equilibrated to reach the species standard temperature (28 °C ± 2 °C), pH (7.0 to 7.5), conductivity (300–700 μS), ammonia (< 0.02 mg/L), nitrite (< 1 mg/L), nitrate (< 50 mg/L), and chloride levels (0 mg/L). Animals were subjected to a light–dark cycle of 14/10 h, respectively. They received paramecium between 6 and 14 days post-fertilization (dpf) and, after 14 dpf, they received commercial flakes (TetraMin Tropical Flake Fish®) three times a day that was supplemented with brine shrimp [43]. The sample number to behavioral and biochemical analyses were based on previous studies performed for our group [41, 44] and other research groups that used zebrafish as an animal model [45,46,47].
Treatments
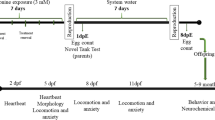
The animals were chosen randomly and divided into two groups considering a density of three animals per liter: control and Met-treated groups. The animals were placed in a glass tank with 5 L of capacity; the control group contained only water and the Met group was exposed for 7 days to a nominal concentration of 3.0 mM of this amino acid dissolved in tank water (l-methionine, Sigma-Aldrich, Ltda, Brazil). This dose was chosen according to previous studies of hypermethioninemia in rodents [48] and zebrafish [25]. We analyzed the Met levels in the water by liquid chromatography coupled to mass spectrometry and we found a mean of 2.743 mM ± 0.127 (as standard deviation) for the Met group and it was not detected Met values for the control group. The animals were kept in the same conditions as mentioned above, however, in glass tanks with reverse osmosis–filtered water previously treated with Tetra’s AquaSafe® that neutralizes chlorine, chloramine, and heavy metals. For maintenance of Met concentration and experimental conditions, in the third and fifth day, the water and Met 3.0 mM solution were renewed. To investigate if the effect of hypermethioninemia remained after the Met exposure, we maintained the animals for more 7 days in tanks containing only water. In the eighth day post-withdrawal of Met (8 dpw), the animal behavior and biochemical analysis were evaluated. The study design is outlined in Fig. 1.
Timeline of the experimental procedure. The control group contained only water and the Met group was exposed to a concentration of 3.0 mM of this amino acid dissolved in tank water for 7 days. The animals were maintained for more 8 days in tanks containing only water and in the eighth day post-exposure (8 dpw) the animal behavior and biochemical analysis were evaluated
Behavioral Analysis
Novel Tank Test
The novel tank test, an exploratory test analogous to the rodent’s open-field test, was carried out as described previously by Gerlai et al. with modifications [42, 49]. The experiment was conducted in a temperature-controlled room (27 °C ± 2 °C) between 9:30 a.m. and 12 p.m. A total of 75 animals (control 35; Met 40) were used, which are tested in experimental triplicate. The animals were placed individually in experimental tanks (30-cm length × 15-cm height × 10-cm width). After 60 s of habituation, the locomotor behavior was recorded for 5 min, for subsequent analysis with EthoVision XT software (version 11.5, Noldus). The tank was virtually divided into two zones with a horizontal line since the zebrafish tend to spend more time at the bottom of the tank when introduced into a new environment, until gradually moving to the upper zone after a few minutes [50]. The locomotor parameters analyzed were distance moved (m) and velocity (m/s, the ratio between distance traveled and movement). Time spent in upper zone (s) and latency to enter at the upper zone (s) also were measured.
Social Interaction
Social interaction was evaluated between 9:30 a.m. and 12.00 p.m. The zebrafish is a schooling fish that may exhibit a preference for its conspecifics under certain circumstances. For this test, a total of 57 animals (control 27; Met 30) were tested in experimental duplicate. Each fish was individually placed in an experimental tank (30-cm length × 15-cm height × 10-cm width). On one side of the experimental tank, an empty fish tank was placed; on the other side, an identically sized tank held 15 zebrafish, which were designated the “stimulus fish.” The fish undergoing evaluation was allowed to acclimatize to the experimental tank for a 60-s period, after which its behavior was video recorded for 5 min for subsequent analysis with EthoVision XT (version 11.5, Noldus) as described by Gerlai et al. [49] with modifications. To quantify fish preference between the “stimulus fish” side of their tank and the empty tank, the experimental tank was virtually divided into three equal sections. The amount of time the experimental fish spent closer to the stimulus zone was measured.
Mirror-Induced Aggression
Aggression behavior was evaluated between 9:30 a.m. and 12.00 p.m. The mirror-induced aggression was used to measure aggression according to the procedure described by Gerlai et al. [49], with modifications. A total of 44 (control 23; Met 21) animals were used and tested in experimental duplicate. Each fish was individually placed in an experimental tank (30 cm length × 15 cm height × 10 cm width). A mirror (45 cm × 38 cm) was placed at the side of the tank at an angle of 22.5° to the back wall of the tank so that the left vertical edge of the mirror touched the side of the tanks and the right edge was further away. Thus, when the experimental fish swam to the left side of the tank, their mirror image appeared closer to them. After 1 min of acclimatization, a 5-min session was recorded for subsequent quantification of aggression with EthoVision XT (version 11.5, Noldus). Three virtual vertical lines and 1 virtual horizontal line were used to divide the tank into six sections and allowed the researchers to record the number of entries the fish made into each section. Entry to the left-most segment indicated a preference for proximity to the “opponent,” whereas entry to the right-most segments implied avoidance. The amount of time the experimental fish spent in the segment closest to the mirror was measured. We also evaluated the number of bites against the mirror image as an aggression parameter. For the bites parameter, a blind analysis was performed by the evaluator to avoid any bias.
Inhibitory Avoidance
To assess whether Met could impair memory in adult animals, we carried out an inhibitory avoidance test between 9:00 a.m. and 12:00 p.m. [41, 51]. There were two sessions, training and test, with a 24-h interval between them. In each session, animals were placed individually in an experimental tank (18-cm length × 9-cm width × 7-cm high) with water, divided by a guillotine door into two compartments of equal size, one black (right side) and one white (left side). During the training session, the animal was placed in the white compartment with the door closed for 1 min for habituation and environment recognition. After this period, the division was lifted. Once the animal crossed into the black side of the tank, the guillotine door was closed again and through the use of two electrodes attached to an 8-V stimulator was administered a final shock pulse of 3 ± 0.2 V AC (intensity measured between electrodes and the center of the dark compartment) for 5 s. The animal was removed from the apparatus and returned to its housing tank with only water for 24 h until the test session, which consisted of the same protocol as the training session, but without the electric shock. The latency to enter the black compartment during each session was measured and the expected increase in the test session was used as an index of memory retention. A 180-s ceiling was imposed on test session latency measurements. For this test, a total of 22 animals (control 11; Met 11) was used.
Biochemical Parameters
Oxidative Stress Parameters
Tissue Preparation
Samples of brain tissue were obtained after euthanasia of each subject by hypothermic shock, in which the brains were rapidly dissected and maintained in ice. A pool of three brains (n = 6–9) was homogenized in 20 mM sodium phosphate buffer, at pH 7.4, containing 140 mM KCl, and maintained in a − 80 °C freezer until biochemical analysis. Then, the homogenates were centrifuged at 800g for 10 min at 4 °C. The supernatant was used for the measurements of oxidative stress parameters. A total of 54 animals (control 27, Met 27) were used to evaluate thiobarbituric acid reactive substances (TBARS) and dihydrodichlorofluorescein (H2DCF) oxidation assay. For the analyses of nitrite levels, superoxide dismutase (SOD) activity, catalase (CAT) activity, and total thiol content, a total of 36 animals (control 18, Met 18) were used.
2′,7′-Dihydrodichlorofluorescein Oxidation Assay
The 2′,7′-dichlorodihydrofluorescein diacetate (H2DCF-DA) is an indirect indicator for reactive oxygen species (ROS) mainly H2O2 [52, 53]. ROS production was measured according to LeBel et al. [54], which is based on the oxidation of H2DCF, a formed product when H2DCF-DA is cleaved by esterases. H2DCF is then oxidized by ROS in the samples and this reaction produces dichlorofluorescein (DCF), a fluorescent compound which is measured at λem = 488 nm and λex = 525 nm. Results were measured as nanomole of DCF per milligram of protein.
Thiobarbituric Acid Reactive Substances
Thiobarbituric acid reactive substances (TBARS) was determined as described by Ohkawa et al. [55]. TBARS measures malondialdehyde (MDA), a product of lipoperoxidation caused mainly by hydroxyl free radicals. A calibration curve was performed using 1,1,3,3-tetramethoxypropane and each curve point was subjected to the same treatment as that of the sample. TBARS were determined in a spectrophotometer by the absorbance at 535 nm and measured as nanomole per milligram of protein.
Nitrite Levels
Nitric oxide (NO) was indirectly measured by nitrite levels. Samples were mixed with Griess reagent (1:1 mixture of 1% sulphanilamide in 5% phosphoric acid and 0.1% naphthylethylenediamine dihydrochloride in water) forming a rosy product determined on a microplate reader (SpectraMax M5/M5 Microplate Reader; Molecular Devices, MDS Analytical Technologies) at 543 nm. Nitrite concentration was calculated using a sodium nitrite standard according to the description in the literature [56].
Superoxide Dismutase Activity Assay
Superoxide dismutase (SOD) is an enzyme specialized in eliminating superoxide anion (O2.−) radicals derived from extracellular stimulants turning into hydrogen peroxide (H2O2) [57]. SOD (EC 1.15.1.1) activity assay was performed according to Marklund and Marklund [58]. SOD activity is measured through pyrogallol autoxidation, and the activity was then indirectly assayed at 420 nm. A calibration curve was performed with purified SOD as a standard. The results were measured as units per milligram of protein.
Catalase Activity Assay
Catalase (CAT) is an enzyme specialized in quickly eliminating H2O2 from a biological medium. CAT (EC 1.11.1.6) activity was assayed based on the consumption H2O2 at 240 nm according to Aebi [59]. Samples were mixed with a 0.06% solution of H2O2 and its consumption measured between 5 min in a spectrophotometer. The results were measured as units per milligram of protein, with one CAT unit being 1 μmol of H2O2 consumed per minute. The results were measured as units per milligram of protein.
Total Thiol Content
The analysis of thiol content was used as an estimate of oxidative modifications in proteins according to the method described by Ellman [60]. The reaction uses the dithionitrobenzoic acid (DTNB) colored reagent, which is reduced by the thiols present in the sample, generating a yellowish compound (TNB) which is read on a spectrophotometer at 412 nm. Results were measured as nanomole of TNB per milligram protein.
Determination of Neurotransmitter and Amino Acids
The presence of neurotransmitters and amino acids was analyzed in zebrafish brain at 8 dpw. The samples were prepared according to [61]. Briefly, three brains (n = 6) were pooled and homogenized in 300 μL of 0.1 M formic acid (Sigma-Aldrich, St. Louis, MO) and centrifuged at 20,000g for 20 min at 4 °C. The supernatant was transferred to a 250-μL glass vial and injected into an Agilent ultra-high performance liquid chromatograph, model 1290 Infinity, coupled to an Agilent mass spectrometer, model 6460 TQQQ (Agilent, Santa Clara, CA, USA). Chromatographic separations were performed on a column Zorbax Eclipse Plus C18 RRHD (5 × 2.1 mm, 1.8 μm, Agilent, Paolo Alto, USA) using a mobile phase consisting of (A) 0.1% formic acid and (B) acetonitrile with 0.1% formic acid, in gradient mode. The gradient started with 2% of B, and then programmed to 95% of B after 4.5 min, remaining in this condition for 1.3 min before returning to the start condition. The total time of the chromatographic run was 7.5 min, the mobile phase flow was 0.2 mL/min with a column temperature of 40 °C, and the injected extract volume was 10 μL. The spectrometer was operated in MRM mode and the analytes were quantified with the following transitions (m/z): dopamine (DA, 154 > 119.1), serotonin (SER, 177 > 160), glutamic acid (GLU, 148 > 48), glutathione (GLT, 308 > 162.2), epinephrine (EPI, 184 > 166), carnitine (CAR, 162 > 60.3), methionine (MET, 150 > 61), homocysteine (HCY, 136 > 56.1), creatine (CRE, 132 > 44), taurine (TAU, 126 > 108), and cysteine (CYS, 122 > 59). Quantifications were performed by external standardization and calibration curves were obtained with the following concentrations: DA 1.0, 5.0, 10.0, 15.0, and 20.0 ng/mL; SER 1.0, 5.0, 10.0, 15.0, and 20.0 ng/mL; EPI 0.1, 0.5, 1.0, 1.5, and 2.0 ng/mL; HCY 0.1, 0.5, 1.0, 1.5, and 2.0 ng/mL; CRE 2.5, 12.5, 25.0, 37.5, and 50 ng/mL; MET 2.5, 12.5, 25.0, 37.5, and 50 ng/mL; CYS 2.5, 12.5, 25.0, 37.5, and 50 ng/mL; GLU 0.1, 0.5, 1.0, 1.5, and 2.0 μg/mL; TAU 0.1, 0.5, 1.0, 1.5, and 2.0 μg/mL; GLT 1.0, 2.5, 5.0, 7.5, and 10.0 μg/mL; CRE 1.0, 2.5, 5.0, 7.5, and 10.0 μg/mL.
Standards were prepared individually in the water at concentration of a 0.5 mg/mL. Before the analyses, they were mixed and diluted with mobile phase A and beta-mercaptoethanol was added in an equivalent concentration of the samples. The final results were corrected by the protein concentration of the samples. A total of 36 animals (control 18, Met 18) were used for the analysis.
Protein Determination
The protein content of the samples was determined according to the Bradford method [62], using a spectrophotometer with a 1-cm quartz cell. The obtained results were used to normalize both neurotransmitter and amino acid concentrations and the oxidative stress parameters, according to described by Lowry et al. [63]. Serum bovine albumin was used as a standard for the calibration curve.
Statistical Analysis
Statistical differences for novel tank test, social interaction, and mirror-induced aggression were determined by Welch’s t test. Oxidative stress parameters, neurotransmitters, and amino acid concentrations were determined by Student’s t test. In the inhibitory avoidance memory task, training, and test latencies for each group, likewise, the latencies of control and Met group were compared using Mann–Whitney U tests. All the data were expressed as the mean ± standard error (SEM). For all comparisons, the significance level was set at p < 0.05.
Results
Novel Tank Test
The behavioral pattern of adult animals was analyzed after 8 days of Met withdrawal (8 dpw). After 8 dpw, Met-treated group showed a decrease in distance moved (p = 0.0001) (Fig. 2a) and in velocity (p = 0.0001) (Fig. 2b). Also, it was observed an increase in time in the upper zone (p = 0.0206) for the treated group when compared the control (Fig. 2c). However, the latency to enter in the upper zone did not present a significant change.
Novel tank test parameters of zebrafish 8 days after Met withdrawal. a distance moved; b velocity; c time in the upper zone; d latency to enter in the upper zone. Data are expressed as mean ± S.E.M from 35 to 40 animals analyzed individually for each group. Statistical analysis was performed by Welch's t test. *p < 0.05, ****p < 0.0001
Social Interaction and Mirror-Induced Aggression
Met withdrawal did not induce any social interaction changes in zebrafish and all treated animals presented the same preference for the stimulus area as their respective controls (Fig. 3a). However, Met altered aggressive behavior, evaluated by the time spent in the segment nearest to the mirror and number of bites in this zone. Met group at 8 dpw remained for less time in the mirror when compared with the control group (p = 0.0413) (Fig. 3b), demonstrating an effect of Met in decreasing aggressive behavior. There were no changes in the number of bites in the Met group when compared with that in the control (p = 0.1116) (Fig. 3c).
Effects of Met withdrawal in social interaction test and aggression parameters. a Time in interaction zone. b Time in mirror zone. c Number of bites. The data are expressed as the mean ± S.E.M from 25 to 35 animals analyzed individually for each group. Statistical analyses were performed by Welch's t test. *p < 0.05
Memory Test
The effects of Met withdrawal on inhibitory avoidance memory were analyzed in zebrafish. At 8 dpw, there were no differences in the latency for training and test sessions in Met-treated zebrafish, indicating an impairment of aversive memory (p > 0.9999), whereas there was a significant difference in the training and test latencies in the control group (p = 0.0024) (Fig. 4).
Inhibitory avoidance task performance on training and long-term memory test sessions of control and Met-treated adult zebrafish after 8 dpw. The data are expressed as the mean ± SEM from 11 animals analyzed individually for each group. Statistical analyses were performed by the Mann–Whitney test. **p < 0.01 indicates the differences between training and test sessions for each group. No differences were found between training performance among groups
Oxidative Stress Parameters
At 8 dpw, DCFH oxidation, TBARS, and nitrite levels did not change significantly when compared with the control (Fig. 5a–c). However, Met exposure followed by withdrawal was able to impair the antioxidant enzymes, since SOD (p = 0.0466) (Fig. 5d) and CAT (p = 0.0284) (Fig. 5e) activities were decreased. The content of total thiols was also reduced in the Met-treated group in comparison with that in the control (p = 0.0358) (Fig. 5f).
Effect of Met exposure on oxidative stress parameters at 8 dpw. a DCFH oxidation. b TBARS. c Nitrites. d SOD. e CAT. f Total thiols in the zebrafish brain. Data from DCFH oxidation are expressed as nmol of DCF formed per mg of protein; data from TBARS are expressed as nmol of TBARS formed per mg of protein. Enzyme activities are expressed as units per mg protein. One CAT unit is defined as 1 μmol of oxygen peroxide consumed per minute. One SOD unit is defined as the amount of SOD necessary to inhibit 50% of pyrogallol autoxidation; data from total thiols are expressed as nmol of TNB per mg of protein. Data are expressed as mean ± SEM for 6–9 independent experiments (pool of three adult brain per n) performed in triplicate. Statistical analyses were performed by Student’s t test. *p = < 0.05
Neurotransmitter and Amino Acid Analysis
The animals at 8 dpw did not show changes in concentration of the neurotransmitters dopamine, serotonin, and glutamate (Fig. 6a–d), while there was a significant decrease in epinephrine of the Met group at 8 dpw (p = 0.003) (Fig. 6c). The levels of Met and its toxic metabolite homocysteine presented no significant changes in the Met group when compared with that in the control (Fig. 6e, f), as well as glutathione and taurine (Fig. 6g, j). However, carnitine (p = 0.0258) and creatine (p = 0.0285) levels had a significant increase in the Met withdrawal group (Fig. 6h, i), while cysteine (p = 0.0098) was lower compared with that in the control group (Fig. 6k).
Effect of Met exposure on neurotransmitters and amino acids at 8 dpw. a Dopamine. b Serotonin. c Epinephrine. d Glutamate. e Methionine. f Homocysteine. g Glutathione. h Carnitine. i Creatine. j Taurine. k Cysteine. The parameters are expressed as ng/mg of protein and showed as mean ± SEM of 6 independent experiments (pool of three adult brain per n) performed in triplicate. Statistical analyses were performed by Student’s t test. *p = < 0.05 and **p < 0.01
Discussion
In the present study, we observed changes in locomotion, anxiety-like behavior, aggression, memory, oxidative stress markers, and amino acid and neurotransmitter levels after the withdrawal of Met exposure. It has been reported that citrin deficiency, MAT1A mutation, low birth weight and/or prematurity, acute liver disease, or protein intake lead to a transient hypermethioninemia state in humans subjects [16]. However, the effects of Met exposure after its withdrawal have been poorly studied. Therefore, the wide and toxic actions of Met led us to explore behavioral and neurochemical parameters after the withdrawal of exposure to this amino acid.
Changes in Met levels modify the methylation processes in the brain and thus affect behavior [64]. Withdrawal effects following Met exposure include an increase in the time spent in the upper tank zone, indicating a decrease in the anxiety-like behavior in the novel tank test. It is important to note that this phenotype is one of the indicators of anxiety-like behavior. Further analyses are required to confirm this phenotype, such as the light–dark test and the measure of cortisol levels. Furthermore, Met exposure for 7 days followed by its withdrawal promoted changes in locomotion parameters, including a decrease in the distance traveled and velocity. A generally slower swimming phenotype is in line with anxiolytic effects caused by antidepressant drugs as amitriptyline [65]. In a previous study from our group, we evaluated locomotion and anxiety-like behavior in zebrafish immediately after exposure to Met and did not observe significant alterations in these parameters [36]. Thus, our data show that withdrawal of Met exposure possibly could cause a late effect on anxiety-like behavior. Besides, our findings in the mirror-induced aggression test showed a significant decrease in time spent close to the mirror, but there were no changes in the number of bites by animals submitted to Met withdrawal. Taken together, these parameters indicate a decrease of aggression, even after Met withdrawal. Also, social interaction did not present changes. These findings indicated that the changes observed in aggression parameters were not influenced by social interaction behavior.
Encephalopathies are a common characteristic of patients with hypermethioninemia [10, 11]. The memory impairment from exposure to Met had already been studied in rats and zebrafish [24, 36]. In our study, we showed that withdrawal Met impaired aversive memory. The inhibitory avoidance task demonstrated that animals previously exposed to Met and followed by its removal had a decrease in latency to entry to the dark side of apparatus, indicating that they did not remember the shock received 1 day before. Taken together, our findings observed in novel tank test, mirror-induced aggression test, and inhibitory avoidance task reflect a persistent neurotoxic effect induced by Met exposure, since the animal lost its natural state of exploring the new area with caution, decreased its ability to attack or defend itself and had decreased its cognitive capacity.
Studies demonstrated that oxidative stress is responsible for behavioral changes in anxiety/depression [66], aggression [67], and memory [67]. Indeed, oxidative damage is one of the most important mechanisms responsible for Met toxicity [23]. Previous studies demonstrated that high levels of this amino acid cause a decrease of CAT and a consequent increase of ROS besides inducing lipid peroxidation in rat brain [27, 29]. However, our study investigated the effects on oxidative stress markers after Met withdrawal. At 8 dpw, brain lipid damage was no longer observed since TBARS did not change significantly between the groups as well as nitrites and ROS levels, demonstrating that this effect may already be normalized. However, the effects of Met on oxidative stress mechanisms are still persistent, since the levels of total thiols are decreased, showing that after Met removal, oxidative modifications in proteins are still occurring, probably due to a radical not measured in our study, like O2−. The analysis of DCFH has the limitation of not reacting with the radical O2− [53]. In addition, the decrease of SOD activity in the Met-treated group may be contributing to a possible increase in O2−. Since CAT and SOD have a balanced functioning, that is, when one enzyme is reduced, the other in a physiological condition tends to be reduced as well [68], our findings demonstrated a decrease in CAT activity. The results also demonstrated an increased level of creatine in the Met-withdrawal group, which could be acting as an antioxidant agent protecting the increase of ROS and iNOS production [69, 70]. These data demonstrated the persistent effects that this amino acid exerted on control mechanisms of oxidative stress.
At 8 dpw, no significant changes in Met content were observed in the zebrafish brain, which could indicate that Met levels were already normalized, but some consequences of this exposure remained. Epinephrine levels were decreased after Met withdrawal. Epinephrine is a neurotransmitter of the sympathetic nervous system that regulates the “flight or fight” behavior. Anxiety and locomotion are positively correlated with epinephrine levels [71,72,73] and emotional responses of aggression are also regulated by this neurotransmitter [74]. Besides that, epinephrine has a fundamental role in memory and cognition processing [75, 76]. The changes observed in behavioral parameters can be attributed to a decrease in epinephrine levels, once that deregulation in the NE system is a mechanism involved in the occurrence of pathological behaviors like anxiety and depression [77]. Also, dopamine was unchanged in Met-withdrawal animals. This decrease in epinephrine is probably related to a tendency of S-adenosylmethionine to donate its methyl groups for the synthesis of creatine [78].
Carnitine is biosynthesized in humans by lysine methylation and has Met as its methyl group donor via S-adenosylmethionine and its main function is to transport fatty acids to mitochondria [79]. It is possible to suggest that the decline in aversive memory observed in our study could be caused by the increase in carnitine levels. Previous studies reported that chronic sleep interruption–induced mice increased the carnitine levels and showed a decline in cognitive function [80]. Besides, the l-carnitine has modulatory properties in GABAergic activities, with agonist actions in the GABAA receptor [4], which could be related to an impaired behavioral response showed by the Met-withdrawal group. The creatine, in turn, is synthesized from arginine, Met, and glycine and several studies indicate the potential of this compound in the treatment of neurodegenerative disorders. Creatin regenerates ATP for the clearance of glutamate (the main mediator of excitotoxicity) [81]. Patients with Huntington’s disease supplemented with creatine had their brain glutamate levels decreased [82]. In addition, the role of creatine as an antioxidant reinforces the application of this amino acid as a therapeutic agent for the management of several neurological diseases (for a review, see [70]). The increased creatine concentration after Met withdrawal may be involved in the control of the oxidative damage markers (TBARS, ROS, and nitrites), as observed in our study. Homocysteine, the main toxic product of the Met cycle, is responsible for vascular disease [83] and cognition deficits [13]. A previous study demonstrated that creatine may control homocysteine levels [84]. Also, hyperhomocysteinemia increases the levels of TBARS and ROS, but creatine prevents this imbalance [85], besides that an increase of nitrite levels caused by glutamate administration is completely prevented by creatine [69]. Therefore, the normalized homocysteine levels may be due to the action of creatine, which is increased in our study.
Our data showed a decrease in total thiol and cysteine levels after Met withdrawal; however, glutathione was not altered in this condition. Since glutathione is a non-proteic thiol, the reduced total thiols levels may be correlated with the decrease of proteic thiols, such as cysteine, in Met withdrawal. Cysteine is a nonessential amino acid that can be biosynthesized through Met and is necessary to produce glutathione and taurine. However, the decrease of cysteine showed by us did not influence glutathione and taurine levels, which could suggest that the cysteine content is enough for the production of these amino acids. The cystine/cysteine cycle can be considered an important redox system that regulates cell survival and death [86]. The supplementation of this amino acid is responsible for increasing the levels of CAT and SOD after Met treatment [87]. Therefore, the cysteine depletion may be involved in the decrease of CAT and SOD activities observed in zebrafish submitted to Met withdrawal. Our study shows that attention should be paid to amino acid levels beyond Met since exposure to this compound leads to changes in the biochemical cycles of other amino acids while Met levels are already normalized. However, this study does not rule out the possibility that other pathways have their mechanisms compromised with Met exposure. A long-term Met exposure affects the cholinergic system increasing ACh hydrolysis and decreasing AChE expression [36]. The administration of Met decreased dendritic spine density of layer III pyramidal neurons in the frontal cortex of mice [88]. If these changes are sustained after the Met withdrawal is unknown and, for this reason, more studies are necessary to evaluate other biochemical pathways and different behavioral parameters. Besides, it is important to evaluate how long these effects can be sustained after Met withdrawal.
We concluded that Met caused important neurotoxicological effects that persist even after its removal in zebrafish, with impacts in locomotion, anxiety, aggression, memory, protein damage and enzyme antioxidant system, amino acids, and neurotransmitter levels. Individuals with diagnosed hypermethioninemia and users of dietary supplements not indicated by professionals, especially l-Met or protein blends; premature babies; or patients with liver disease are examples of individuals who deserve special attention since there are transient high levels of Met exposure in these conditions. Although the zebrafish play an important role in translational research, future studies should be conducted in humans to confirm the sustained Met effects.
References
Gulsun M, Oznur T, Aydemir E, Ozcelik F, Erdem M, Zincir S, Akgul O, Kurt Y (2016) Possible relationship between amino acids, aggression and psychopathy. Int J Psychiatry Clin Pract 20:91–100. https://doi.org/10.3109/13651501.2016.1144771
Martínez Y, Li X, Liu G, Bin P, Yan W, Más D, Valdivié M, Hu CAA et al (2017) The role of methionine on metabolism, oxidative stress, and diseases. Amino Acids 49:2091–2098. https://doi.org/10.1007/s00726-017-2494-2
Stipanuk MH (2004) Role of the liver in regulation of body cysteine and taurine levels: a brief review. Neurochem Res 29:105–110. https://doi.org/10.1023/B:NERE.0000010438.40376.c9
Wu C, Gopal KV, Moore EJ, Gross GW (2014) Antioxidants l-carnitine and d-methionine modulate neuronal activity through GABAergic inhibition. J Neural Transm 121:683–693. https://doi.org/10.1007/s00702-014-1170-x
Keller E, Boisonnas R, Vigneaud V (1950) The origin of methil group of epinephrine. J Biol Chem 183:627–632
Millar AL (2008) The methylation, neurotransmitter, and antioxidant connections between folate and depression. Altern Med Rev 13:216–226
Bhattacharyya S, Varshney U (2016) Evolution of initiator tRNAs and selection of methionine as the initiating amino acid. RNA Biol 13:810–819. https://doi.org/10.1080/15476286.2016.1195943
Lu SC (2000) S-Adenosylmethionine. Int J Biochem Cell Biol 32:391–395. https://doi.org/10.1016/S1357-2725(99)00139-9
Garlick PJ (2006) Toxicity of methionine in humans. Rev Rhum:1722–1725
Allen J, Power B, Abedin A, Purcell O, Knerr I, Monavari A (2019) Plasma methionine concentrations and incidence of hypermethioninemic encephalopathy during infancy in a large cohort of 36 patients with classical homocystinuria in the Republic of Ireland. JIMD Rep 47:41–46. https://doi.org/10.1002/jmd2.12029
Bjursell MK, Blom HJ, Cayuela JA, Engvall ML, Lesko N, Balasubramaniam S, Brandberg G, Halldin M et al (2011) Adenosine kinase deficiency disrupts the methionine cycle and causes hypermethioninemia, encephalopathy, and abnormal liver function. Am J Hum Genet 89:507–515. https://doi.org/10.1016/j.ajhg.2011.09.004
Chien Y, Abdenur JE, Baronio F et al (2015) Mudd’s disease (MAT I/III deficiency): a survey of data for MAT1A homozygotes and compound heterozygotes. Orphanet J Rare Dis 10:99. https://doi.org/10.1186/s13023-015-0321-y
Sklirou E, Lichter-Konecki U (2018) Inborn errors of metabolism with cognitive impairment: metabolism defects of phenylalanine, homocysteine and methionine, purine and pyrimidine, and creatine. Pediatr Clin N Am 65:267–277. https://doi.org/10.1016/j.pcl.2017.11.009
Furujo M, Kinoshita M, Nagao M, Kubo T (2012) Methionine adenosyltransferase I/III deficiency: neurological manifestations and relevance of S-adenosylmethionine. Mol Genet Metab 107:253–256. https://doi.org/10.1016/j.ymgme.2012.08.002
Perry TL, Hardwick DF, Dixon GH et al (1965) Hypermethioninemia: a metabolic disorder associated with cirrhosis, islet cell hyperplasia, and renal tubular degeneration. Pediatrics 36:236 LP – 250
Mudd SH (2011) Hypermethioninemias of genetic and non-genetic origin: a review. Am J Med Genet Part C Semin Med Genet 157:3–32. https://doi.org/10.1002/ajmg.c.30293
Muriello MJ, Viall S, Bottiglieri T, Cusmano-Ozog K, Ferreira CR (2017) Confirmation that MAT1A p.Ala259Val mutation causes autosomal dominant hypermethioninemia. Mol Genet Metab Rep 13:9–12. https://doi.org/10.1016/j.ymgmr.2017.07.004
Barić I, Erdol S, Saglam H, et al (2016) Glycine N-methyltransferase deficiency: a member of dysmethylating liver disorders? In: JIMD Reports. pp 101–106
Strauss KA, Ferreira C, Bottiglieri T, Zhao X, Arning E, Zhang S, Zeisel SH, Escolar ML et al (2015) Liver transplantation for treatment of severe S-adenosylhomocysteine hydrolase deficiency. Mol Genet Metab 116:44–52. https://doi.org/10.1016/j.ymgme.2015.06.005
Tran C, Bonafé L, Nuoffer J-M, Rieger J, Berger MM (2018) Adult classical homocystinuria requiring parenteral nutrition: pitfalls and management. Clin Nutr 37:1114–1120. https://doi.org/10.1016/j.clnu.2017.07.013
Park H-D, Lee DH, Choi T-Y, Lee YK, Kim JW, Ki CS, Lee YW (2009) Clinical, biochemical, and genetic analysis of a Korean neonate with hereditary tyrosinemia type 1. Clin Chem Lab Med 47:930–933. https://doi.org/10.1515/CCLM.2009.223
Ohura T, Kobayashi K, Abukawa D, Tazawa Y, Aikawa JI, Sakamoto O, Saheki T, Iinuma K (2003) A novel inborn error of metabolism detected by elevated methionine and/or galactose in newborn screening: neonatal intrahepatic cholestasis caused by citrin deficiency. Eur J Pediatr 162:317–322. https://doi.org/10.1007/s00431-003-1171-5
Schweinberger BM, Wyse ATS (2016) Mechanistic basis of hypermethioninemia. Amino Acids 48:2479–2489. https://doi.org/10.1007/s00726-016-2302-4
Stefanello FM, Monteiro SC, Matté C, Scherer EBS, Netto CA, Wyse ATS (2007) Hypermethioninemia increases cerebral acetylcholinesterase activity and impairs memory in rats. Neurochem Res 32:1868–1874. https://doi.org/10.1007/s11064-007-9464-0
Vuaden FC, Savio LEB, Rico EP, Mussulini BHM, Rosemberg DB, de Oliveira DL, Bogo MR, Bonan CD et al (2016) Methionine exposure alters glutamate uptake and adenine nucleotide hydrolysis in the zebrafish brain. Mol Neurobiol 53:200–209. https://doi.org/10.1007/s12035-014-8983-3
Benavides MA, Bosland MC, da Silva CP, Gomes Sares CT, Cerqueira de Oliveira AM, Kemp R, dos Reis RB, Martins VR et al (2014) L-Methionine inhibits growth of human pancreatic cancer cells. Anti-Cancer Drugs 25:200–203. https://doi.org/10.1097/CAD.0000000000000038
Soares MSP, Viau CM, Saffi J, Costa MZ, da Silva TM, Oliveira PS, Azambuja JH, Barschak AG et al (2017) Acute administration of methionine and/or methionine sulfoxide impairs redox status and induces apoptosis in rat cerebral cortex. Metab Brain Dis 32:1693–1703. https://doi.org/10.1007/s11011-017-0054-9
Stefanello FM, Matté C, Pederzolli CD, Kolling J, Mescka CP, Lamers ML, de Assis AM, Perry ML et al (2009) Hypermethioninemia provokes oxidative damage and histological changes in liver of rats. Biochimie 91:961–968. https://doi.org/10.1016/j.biochi.2009.04.018
Stefanello FM, Scherer EB, Kurek AG et al (2007) Effect of hypermethioninemia on some parameters of oxidative stress and on Na(+),K (+)-ATPase activity in hippocampus of rats. Metab Brain Dis 22:172–182. https://doi.org/10.1007/s11011-007-9052-7
Chakraborty HJ, Rout AK, Behera BK, Parhi J, Parida PK, Das BK (2018) Insights into the aquaporin 4 of zebrafish (Danio rerio) through evolutionary analysis, molecular modeling and structural dynamics. Gene Rep 11:101–109. https://doi.org/10.1016/j.genrep.2018.03.001
Howe K, Clark MD, Torroja CF, Torrance J, Berthelot C, Muffato M, Collins JE, Humphray S et al (2013) The zebrafish reference genome sequence and its relationship to the human genome. Nature 496:498–503. https://doi.org/10.1038/nature12111
Rico EP, Rosemberg DB, Seibt KJ, Capiotti KM, da Silva RS, Bonan CD (2011) Zebrafish neurotransmitter systems as potential pharmacological and toxicological targets. Neurotoxicol Teratol 33:608–617. https://doi.org/10.1016/j.ntt.2011.07.007
Capiotti KM, Fazenda L, Nazario LR, Menezes FP, Kist LW, Bogo MR, da Silva RS, Wyse AT et al (2013) Arginine exposure alters ectonucleotidase activities and morphology of zebrafish larvae (Danio rerio). Int J Dev Neurosci 31:75–81. https://doi.org/10.1016/j.ijdevneu.2012.09.002
Quintana AM, Yu HC, Brebner A, Pupavac M, Geiger EA, Watson A, Castro VL, Cheung W et al (2017) Mutations in THAP11 cause an inborn error of cobalamin metabolism and developmental abnormalities. Hum Mol Genet 26:2838–2849. https://doi.org/10.1093/hmg/ddx157
Savio LEB, Vuaden FC, Kist LW, Pereira TC, Rosemberg DB, Bogo MR, Bonan CD, Wyse ATS (2013) Proline-induced changes in acetylcholinesterase activity and gene expression in zebrafish brain: reversal by antipsychotic drugs. Neuroscience 250:121–128. https://doi.org/10.1016/j.neuroscience.2013.07.004
Vuaden FC, Savio LEB, Piato AL, Pereira TC, Vianna MR, Bogo MR, Bonan CD, Wyse ATS (2012) Long-term methionine exposure induces memory impairment on inhibitory avoidance and alters acetylcholinesterase activity and expression in zebrafish (Danio rerio). Neurochem Res 37:1545–1553. https://doi.org/10.1007/s11064-012-0749-6
Wager K, Mahmood F, Russell C (2014) Modelling inborn errors of metabolism in zebrafish. J Inherit Metab Dis 37:483–495. https://doi.org/10.1007/s10545-014-9696-5
Miller AH, Haroon E, Raison CL, Felger JC (2013) Cytokine targets in the brain: Impact on neurotransmitters and neurocircuits. Depress Anxiety 30:297–306. https://doi.org/10.1002/da.22084
Parker MO, Annan LV, Kanellopoulos AH, Brock AJ, Combe FJ, Baiamonte M, Teh MT, Brennan CH (2014) The utility of zebrafish to study the mechanisms by which ethanol affects social behavior and anxiety during early brain development. Prog Neuro-Psychopharmacology Biol Psychiatry 55:94–100. https://doi.org/10.1016/j.pnpbp.2014.03.011
Giacomini ACVV, Abreu MS, Giacomini LV, Siebel AM, Zimerman FF, Rambo CL, Mocelin R, Bonan CD et al (2016) Fluoxetine and diazepam acutely modulate stress induced-behavior. Behav Brain Res 296:301–310. https://doi.org/10.1016/j.bbr.2015.09.027
Nabinger DD, Altenhofen S, Bitencourt PER, Nery LR, Leite CE, Vianna MRMR, Bonan CD (2018) Nickel exposure alters behavioral parameters in larval and adult zebrafish. Sci Total Environ 624:1623–1633. https://doi.org/10.1016/j.scitotenv.2017.10.057
Zanandrea R, Abreu MSMS, Piato A et al (2018) Lithium prevents scopolamine-induced memory impairment in zebrafish. Neurosci Lett 664:34–37. https://doi.org/10.1016/j.neulet.2017.11.010
Westerfield M (2000) The Zebrafish book. A Guide for The Laboratory Use of Zebrafish (Danio rerio)
Altenhofen S, Nabinger DD, Wiprich MT, Pereira TCB, Bogo MR, Bonan CD (2017) Tebuconazole alters morphological, behavioral and neurochemical parameters in larvae and adult zebrafish (Danio rerio). Chemosphere 180:483–490. https://doi.org/10.1016/j.chemosphere.2017.04.029
Mocelin R, Marcon M, da Rosa Araujo AS, Herrmann AP, Piato A (2019) Withdrawal effects following repeated ethanol exposure are prevented by N-acetylcysteine in zebrafish. Prog Neuro-Psychopharmacology Biol Psychiatry 93:161–170. https://doi.org/10.1016/j.pnpbp.2019.03.014
Bevilaqua F, Sachett A, Chitolina R, Garbinato C, Gasparetto H, Marcon M, Mocelin R, Dallegrave E et al (2020) A mixture of fipronil and fungicides induces alterations on behavioral and oxidative stress parameters in zebrafish. Ecotoxicology 29:140–147. https://doi.org/10.1007/s10646-019-02146-7
Agostini JF, Toé HCZD, Vieira KM, Baldin SL, Costa NLF, Cruz CU, Longo L, Machado MM et al (2018) Cholinergic system and oxidative stress changes in the brain of a zebrafish model chronically exposed to ethanol. Neurotox Res 33:749–758. https://doi.org/10.1007/s12640-017-9816-8
Stefanello FM, Matté C, Scherer EB, Wannmacher CMD, Wajner M, Wyse ATS (2007) Chemically induced model of hypermethioninemia in rats. J Neurosci Methods 160:1–4. https://doi.org/10.1016/j.jneumeth.2006.07.029
Gerlai R, Lahav M, Guo S, Rosenthal A (2000) Drinks like a fish: Zebra fish (Danio rerio) as a behavior genetic model to study alcohol effects. Pharmacol Biochem Behav 67:773–782. https://doi.org/10.1016/S0091-3057(00)00422-6
Cachat J, Stewart A, Grossman L, Gaikwad S, Kadri F, Chung KM, Wu N, Wong K et al (2010) Measuring behavioral and endocrine responses to novelty stress in adult zebrafish. Nat Protoc 5:1786–1799. https://doi.org/10.1038/nprot.2010.140
Blank M, Guerim LD, Cordeiro RF, Vianna MRM (2009) A one-trial inhibitory avoidance task to zebrafish: Rapid acquisition of an NMDA-dependent long-term memory. Neurobiol Learn Mem 92:529–534. https://doi.org/10.1016/j.nlm.2009.07.001
Wang X, Roper MG (2014) Measurement of DCF fluorescence as a measure of reactive oxygen species in murine islets of Langerhans. Anal Methods 6:3019–3024. https://doi.org/10.1039/C4AY00288A
Chen X, Zhong Z, Xu Z, Chen L, Wang Y (2010) 2′,7′-Dichlorodihydrofluorescein as a fluorescent probe for reactive oxygen species measurement: Forty years of application and controversy. Free Radic Res 44:587–604. https://doi.org/10.3109/10715761003709802
LeBel CP, Ali SF, McKee M, Bondy SC (1990) Organometal-induced increases in oxygen reactive species: the potential of 2′,7′-dichlorofluorescin diacetate as an index of neurotoxic damage. Toxicol Appl Pharmacol 104:17–24. https://doi.org/10.1016/0041-008X(90)90278-3
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal Biochem 126:131–138. https://doi.org/10.1016/0003-2697(82)90118-x
Miao L, St. Clair DK (2009) Regulation of superoxide dismutase genes: Implications in disease. Free Radic Biol Med 47:344–356. https://doi.org/10.1016/j.freeradbiomed.2009.05.018
MARKLUND S, MARKLUND G (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 47:469–474. https://doi.org/10.1111/j.1432-1033.1974.tb03714.x
Aebi H (1984) Oxygen radicals in biological systems. Methods Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77. https://doi.org/10.1016/0003-9861(59)90090-6
Altenhofen S, Wiprich MT, Nery LR, Leite CE, Vianna MRMR, Bonan CD (2017) Manganese(II) chloride alters behavioral and neurochemical parameters in larvae and adult zebrafish. Aquat Toxicol 182:172–183. https://doi.org/10.1016/j.aquatox.2016.11.013
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Lowry OH, Rosebrough A, Farr L, Randall RJ (1994) Protein measurement with the folin phenol reagent. Anal Biochem 217:220–230. https://doi.org/10.1016/0304-3894(92)87011-4
Weaver ICG, Champagne FA, Brown SE, Dymov S, Sharma S, Meaney MJ, Szyf M (2005) Reversal of maternal programming of stress responses in adult offspring through methyl supplementation: altering epigenetic marking later in life. J Neurosci 25:11045–11054. https://doi.org/10.1523/JNEUROSCI.3652-05.2005
Meshalkina DA, Kysil EV, Antonova KA, Demin KA, Kolesnikova TO, Khatsko SL, Gainetdinov RR, Alekseeva PA et al (2018) The effects of chronic amitriptyline on zebrafish behavior and monoamine neurochemistry. Neurochem Res 43:1191–1199. https://doi.org/10.1007/s11064-018-2536-5
Salim S (2017) Oxidative stress and the central nervous system. J Pharmacol Exp Ther 360:201 LP – 205. https://doi.org/10.1124/jpet.116.237503
Coccaro EF, Lee R, Gozal D (2016) Elevated plasma oxidative stress markers in individuals with intermittent explosive disorder and correlation with aggression in humans. Biol Psychiatry 79:127–135. https://doi.org/10.1016/j.biopsych.2014.01.014
Yenkoyan K, Harutyunyan H, Harutyunyan A (2018) A certain role of SOD/CAT imbalance in pathogenesis of autism spectrum disorders. Free Radic Biol Med 123:85–95. https://doi.org/10.1016/j.freeradbiomed.2018.05.070
Cunha MP, Lieberknecht V, Ramos-Hryb AB, Olescowicz G, Ludka FK, Tasca CI, Gabilan NH, Rodrigues ALS (2016) Creatine affords protection against glutamate-induced nitrosative and oxidative stress. Neurochem Int 95:4–14. https://doi.org/10.1016/j.neuint.2016.01.002
Marques EP, Wyse ATS (2019) Creatine as a neuroprotector: an actor that can play many parts. Neurotox Res 36:411–423. https://doi.org/10.1007/s12640-019-00053-7
Kopin IJ (1984) Avenues of investigation for the role of catecholamines in anxiety. Psychopathology 17:83–97. https://doi.org/10.1159/000284081
Henrique AJ, Gabrielloni MC, Rodney P, Barbieri M (2018) Non-pharmacological interventions during childbirth for pain relief, anxiety, and neuroendocrine stress parameters: a randomized controlled trial. Int J Nurs Pract 24:e12642. https://doi.org/10.1111/ijn.12642
Winter B, Breitenstein C, Mooren FC, Voelker K, Fobker M, Lechtermann A, Krueger K, Fromme A et al (2007) High impact running improves learning. Neurobiol Learn Mem 87:597–609. https://doi.org/10.1016/j.nlm.2006.11.003
Dennis RL (2016) Adrenergic and noradrenergic regulation of poultry behavior and production. Domest Anim Endocrinol 56:S94–S100. https://doi.org/10.1016/j.domaniend.2016.02.007
Morris KA, Gold PE (2013) Epinephrine and glucose modulate training-related CREB phosphorylation in old rats: relationships to age-related memory impairments. Exp Gerontol 48:115–127. https://doi.org/10.1016/j.exger.2012.11.010
Toth M, Ziegler M, Sun P, Gresack J, Risbrough V (2013) Impaired conditioned fear response and startle reactivity in epinephrine-deficient mice. Behav Pharmacol 24:1–9. https://doi.org/10.1097/FBP.0b013e32835cf408
Goddard AW, Ball SG, Martinez J, Robinson MJ, Yang CR, Russell JM, Shekhar A (2010) Current perspectives of the roles of the central norepinephrine system in anxiety and depression. Depress Anxiety 27:339–350. https://doi.org/10.1002/da.20642
Brosnan JT, Brosnan ME, Bertolo RFP, Brunton JA (2007) Methionine: a metabolically unique amino acid. Livest Sci 112:2–7. https://doi.org/10.1016/j.livsci.2007.07.005
Ferreira GC, McKenna MC (2017) L-Carnitine and acetyl-l-carnitine roles and neuroprotection in developing brain. Neurochem Res 42:1661–1675. https://doi.org/10.1007/s11064-017-2288-7
Feng L, Wu H, Song G, Lu C, Li YH, Qu LN, Chen SG, Liu XM et al (2016) Chronical sleep interruption-induced cognitive decline assessed by a metabolomics method. Behav Brain Res 302:60–68. https://doi.org/10.1016/j.bbr.2015.12.039
Oliet SHR, Piet R, Poulain DA (2001) Control of glutamate clearance and synaptic efficacy by glial coverage of neurons. Science 292(80):923 LP – 926. https://doi.org/10.1126/science.1059162
Bender A, Auer DP, Merl T et al (2005) Creatine supplementation lowers brain glutamate levels in Huntington’s disease. J Neurol 252:36–41. https://doi.org/10.1007/s00415-005-0595-4
Lentz SR (2005) Mechanisms of homocysteine-induced atherothrombosis. J Thromb Haemost 3:1646–1654. https://doi.org/10.1111/j.1538-7836.2005.01364.x
Taes YEC, Delanghe JR, De Vriese AS et al (2003) Creatine supplementation decreases homocysteine in an animal model of uremia. Kidney Int 64:1331–1337. https://doi.org/10.1046/j.1523-1755.2003.00206.x
Kolling J, Scherer EBS, Siebert C, Marques EP, dos Santos TM, Wyse ATS (2014) Creatine prevents the imbalance of redox homeostasis caused by homocysteine in skeletal muscle of rats. Gene 545:72–79. https://doi.org/10.1016/j.gene.2014.05.005
Banjac A, Perisic T, Sato H, Seiler A, Bannai S, Weiss N, Kölle P, Tschoep K et al (2008) The cystine/cysteine cycle: a redox cycle regulating susceptibility versus resistance to cell death. Oncogene 27:1618–1628. https://doi.org/10.1038/sj.onc.1210796
Stojanović M, Šćepanović L, Todorović D, Mitrović D, Šćepanović V, Šćepanović R, Ilić S, Šćepanović T et al (2018) Suppression of methionine-induced colon injury of young rats by cysteine and N-acetyl-l-cysteine. Mol Cell Biochem 440:53–64. https://doi.org/10.1007/s11010-017-3155-1
Tueting P, Davis JM, Veldic M, Pibiri F, Kadriu B, Guidotti A, Costa E (2010) L-Methionine decreases dendritic spine density in mouse frontal cortex. Neuroreport 21:543–548. https://doi.org/10.1097/WNR.0b013e3283373126
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior-Brasil (CAPES)-finance code 001, Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS), and Instituto Nacional de Ciências e Tecnologia para Doenças Cerebrais, Excitotoxicidade e Neuroproteção. R.Z and C.D.B. (Proc. 305035/2015-0) were the recipients of a fellowship from CNPq.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Ethics Approval
All protocols were approved by the Animal Care Committee of the Pontifical Catholic University of Rio Grande do Sul (8758-CEUA-PUCRS). This study was registered in the Sistema Nacional de Gestão do Patrimônio Genético e Conhecimento Tradicional Associado - SISGEN (Protocol No. A3B073D).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zanandrea, R., Wiprich, M.T., Altenhofen, S. et al. Withdrawal Effects Following Methionine Exposure in Adult Zebrafish. Mol Neurobiol 57, 3485–3497 (2020). https://doi.org/10.1007/s12035-020-01970-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-020-01970-x