Abstract
This study investigated the effect of 7-chloro-4-(phenylselanyl) quinoline (4-PSQ) to restore the cognitive impairment caused by aging in male Wistar rats. Moreover, modulation of neuroplasticity markers, acetylcholinesterase (AChE) activity, and cholesterol levels was performed. Aged rats were intragastrically treated with 4-PSQ (5 mg/kg) for 7 days. Animals were tested in behavioral tasks, and then plasma (to determine cholesterol levels), hippocampus, and cerebral cortex (to determine neural cell adhesion molecule (NCAM) and polysialyltransferase (PST) levels, and AChE activity) were removed. Our findings demonstrated that treatment of aged rats with 4-PSQ restored short-term and long-term memories in the object recognition tests. 4-PSQ treatment did not restore exploratory activity (rearings) but partially restored locomotor activity (crossings) reduced by aging in the open-field test. Moreover, the compound restored the reduction in the NCAM and PST levels, and AChE activity in cerebral structures, as well as the increase in the plasma cholesterol levels, caused by aging in rats. In conclusion, 4-PSQ restored cognitive impairment caused by aging in rats by modulating synaptic plasticity, cholinergic system, and cholesterol levels.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nowadays, the elderly are the fastest-growing segment of the population. This assumes an increase in life expectancy and a greater development of the aging process. This process occurs dynamically and progressively, and several changes determine the individual’s ability to adapt to the environment, causing greater vulnerability and a higher incidence of pathological processes [1, 2].
Neural networks are particularly vulnerable to aging. Aging impairs the functional integrity of neurons [3] associated with cognition and structural plasticity [4]. In fact, a reduction of structural and functional plasticity in brain regions, such as the hippocampus and the prefrontal cortex, causes a cognitive impairment, which is related to aging [5].
The neural cell adhesion molecule (NCAM) plays a key role in neural development and has been implicated in synaptic plasticity and cognitive processes in adulthood [6, 7]. Some studies showed that a decrease of NCAM expression was associated with poor performance in spatial learning and memory tasks, as well as reduced levels of polysialylated neural cell adhesion molecule (PSA-NCAM) in the hippocampus and prefrontal cortex of aging rodents [8].
Additionally, some authors show that a deficit of cognition is caused, at least in part, by damage to cholinergic projections from the basal forebrain to the cortex [9, 10]. One of the most ubiquitous enzymes present in the cholinergic synapses is acetylcholinesterase (AChE) [11]. Janeczek et al. [12] inferred that a decrease in AChE activity in normal elderly people reflects the neuroplasticity that optimizes the capacity of memory by the impact of presynaptically released acetylcholine.
Moreover, an increase in the risk of developing neurodegenerative diseases, including dementia and Alzheimer’s disease, has been associated with aging [13]. Along these lines, abnormalities in cholesterol metabolism are important in the pathogenesis of Alzheimer’s disease, which contribute to cognitive decline among the aged [14].
In this context, the pharmaceutical industry has developed therapeutic strategies to minimize or even slow down the effects of aging on the human brain, promoting an improvement in quality and life expectancy [15, 16]. Selenium is a trace element, which significantly influences the cognitive functions [17] and different neurological disorders [18]. As an alternative to incorporate selenium into organic molecules, synthetic approaches have been developed for the synthesis of various organoselenium derivatives. Organoselenium compounds had an effect in improving memory and learning processes in adult [19] and old [20,21,22] animals.
7-Chloro-4-(phenylselanyl) quinoline (4-PSQ) is an organoselenium compound and a quinoline derivative, which has several pharmacological properties already described in the literature [23,24,25,26,27,28,29,30]. Thus, quinoline derivatives have received the attention of researchers because of their important pharmacological actions [31, 32].
Based on the aforementioned facts and on our interest in applying the findings of the pharmacology of 4-PSQ, the purpose of the present study was to investigate the possible effect of 4-PSQ in the treatment of the cognitive impairment caused by aging in rats. In addition, we examined the effect of the compound on the expression of NCAM and two polysialyltransferases (STX and PST) involved in the neuroplasticity and in the AChE activity in the hippocampus and cerebral cortex, as well as plasma cholesterol levels of aged rats.
Material and Methods
Animals
Male young adult (3 months old, 10% lifespan completed, weighing 170–250 g) and aged (23 months old, 76.6% lifespan completed, weighing 400–700 g) Wistar rats were obtained from a local breeding colony. The animals were housed in cages with free access to food and water. They were kept in a separate air-conditioned (22 ± 2 °C) room, on a 12-h light/12-h dark cycle, with lights on at 7:00 a.m. Commercial diet and water were supplied ad libitum. Animal care and all experimental procedures were conducted in compliance with the National Institute of Health Guide for the Care and Use of Laboratory Animals (NIH publications no. 80-23, revised in 1996) [33] and in accordance with the Committee on Care and Use of Experimental Animal Resources, Federal University of Pelotas, Brazil (CEEA 4599-2016). All efforts were made to minimize the number of animals used and their suffering.
Drugs
4-PSQ (Fig. 1) was prepared and characterized in our laboratory by the method previously described by Duarte et al. [25]. Analysis of the 1H nuclear magnetic resonance (NMR) and 13C NMR spectra showed analytical and spectroscopic data in full agreement with its assigned structure. The chemical purity of 4-PSQ (99.9%) was determined by gas chromatography-mass spectrometry (GC/MS). The compound was dissolved in canola oil. All other chemicals were of analytical grade and obtained from standard commercial suppliers.
Experimental Protocol
Rats were randomly divided into three experimental groups (10 animals/group) (Table 1). The animals belonging to young adult and aged groups received the canola oil (1 ml/kg) and the animals of the aged + 4-PSQ group received the compound (5 mg/kg). Treatments with canola oil or 4-PSQ were performed for 7 days, daily, by intragastric (i.g.) route, via gavage. The 4-PSQ dose was chosen based on previous studies by our group [24, 30].
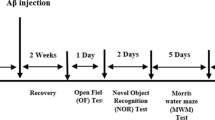
Figure 2 illustrates the experimental protocol of this study. After 7 days of treatment, animals were tested in behavioral tasks: the open-field (OFT) and object recognition (ORT) tests. Subsequent to behavioral tests, animals were allowed to rest until the 14th day in order to avoid behavioral stress, when they were euthanized by inhalation of isoflurane for ex vivo assays. The experimental protocol agreed with Bortolatto et al. [20].
The experimental protocol. Aged rats were intragastrically (i.g.) treated for 7 days with 4-PSQ (5 mg/kg) or canola oil (1 ml/kg). Young adult animals received the canola oil (1 ml/kg). Open-field test (OFT) and object recognition test (ORT) were performed on the 7th, 8th, and 9th days, respectively. On the 14th day, the animals were euthanized by inhalation of isoflurane for ex vivo assays
Behavioral Tasks
OFT
The OFT was performed on the 7th day of the experimental protocol. The open field was a 40 × 45-cm arena surrounded by 50-cm-high plywood walls. The floor of the open field was divided into 9 equal squares (3 × 3). Each animal was placed at the center of the open field and observed for 4 min to record the locomotor (number of segments crossed with the four paws) and exploratory (number of rearings on the hind limbs) activities [34]. The arena floor was cleaned between experiments and the test was carried out in a temperature- and light-controlled room. The 4-min session of OFT was also useful to familiarize the rats with the arena, as a context habituation trial for the recognition memory task.
ORT
Twenty-four hours after OFT (on the 8th day of the experimental protocol), animals were trained in the same arena in order to perform a novel ORT as previously described [35]. ORT, a non-spatial memory task, is used as a measure to evaluate the short-term (STM) and long-term (LTM) memories. Four objects were used in this task: A1, A2, B, and C. The A1 and A2 objects were two identical balls, the B object was a cube and the C object was a square. Each object presented the following color pattern: blue, red, and yellow. All objects were made of plastic material, measuring 10 × 10 cm (length × height). On the day of the task, training was conducted by placing individual rats in the center of the arena, in which two identical objects (objects A1 and A2) were positioned in two adjacent corners, slightly away from the walls. Animals were allowed to explore the objects for 5 min. Exploration was defined as sniffing or touching the object with the nose and/or forepaws. Periods in which the rat moved around, climbed over, or sat on the objects were not recorded. The STM of rats was evaluated 1.5 h after training in the presence of a familiar object (A1) and a novel object (B), and the total time spent in exploring each object was determined during 5 min as a measure of learning and recognition memory. After STM, the rats were housed in our boxes. The LTM was performed 24 h after training, on the 9th day of the experimental protocol, in which rats were placed to explore a familiar object (A1) and a novel object (C) for 5 min and the total time spent in exploring each object was determined. To prevent olfactory cues, the objects and the field were cleaned with an ethanol/water solution after each trial. The results were expressed as exploratory preference (%) according to the following formula: [time spent on the novel object ∕ (time spent on the familiar object + time spent on the novel object)] × 100.
Ex Vivo Assays
On the 14th day of the experimental protocol, rats were anesthetized with isoflurane and blood samples were collected from the heart ventricle, using heparin as anticoagulant to obtain plasma. Plasma was obtained by centrifugation (900×g for 15 min) to determine the cholesterol levels.
Cerebral structures (cortex and hippocampus) were immediately removed and dissected. The cerebral cortex and hippocampus were separated and washed with cold saline solution (0.9%) to determine AChE activity and expression of NCAM, STX, and PST.
AChE Activity
Cerebral cortex and hippocampus samples were homogenized in 0.25 M sucrose buffer (1/10, weight/volume) and centrifuged at 900×g at 4 °C for 15 min. AChE activity was performed according to the method of Ellman et al. [36], using acetylthiocholine as substrate. Enzyme activity was measured by spectrophotometry at 412 nm and expressed as micromole of acetylcholine (ACh) per hour per milligram of protein. The protein concentration was measured according to the Bradford method [37].
Expression of NCAM, STX, and PST
RNA Extraction, cDNA Synthesis, and Quantitative Real-time Polymerase Chain Reaction
Total mRNA was extracted in the hippocampus and cerebral cortex using TRIzol reagent (Invitrogen™, Carlsbad, USA) followed by DNase treatment with DNase I Amplification Grade (Invitrogen™, Carlsbad, USA) in order to ensure minimum DNA contamination of the samples. The total RNA isolated was quantified and its purity (260/280 and 260/230 ratios) was examined by the spectrophotometer NanoVue (GE, Fairfield, CT, USA). The cDNA synthesis was performed using a High Capacity cDNA Reverse Transcription Kit (AppliedBiosystems™, UK) according to the manufacturer’s protocol. For reverse transcription, 1 μg of total RNA was used in a reaction volume of 20 μl. The amplification was performed with GoTaq® qPCR Master Mix (Promega, Madison, WI) using the Agilent Mx3005P QPCR System (Agilent Technologies Inc., Santa Clara, CA) and the sequence of primers used is indicated in Table 2. The qPCR conditions were as follows: 10 min at 95 °C to activate the hot-start Taq polymerase, followed by 35 cycles of denaturation for 15 s at 95 °C, primer annealing for 60 s at 60 °C, and extension for 30 s at 72 °C (fluorescence signals were detected at the end of every cycle). Baseline and threshold values were automatically set by the Stratagene MxPro software. The number of PCR cycles required to reach the fluorescence threshold in each sample was defined as the cycle threshold (Ct) value, and each sample was analyzed in duplicate to obtain an average Ct for each sample. The 2-ΔΔCT method was used to normalize the fold change in gene expressions, using the beta-actin (ACTB) as housekeeping gene.
Cholesterol Levels
The cholesterol levels were determined in the plasma of rats using an automated kinetic colorimetric method by Cobas Integra 400 plus. Cholesterol esters are cleaved by the action of cholesterol esterase and produce free cholesterol and fatty acids. Cholesterol oxidase catalyzes the oxidation of cholesterol to cholest-4-en-3-one and hydrogen. In the presence of peroxidase, the hydrogen peroxide formed affects the oxidative coupling of phenol and 4-aminoantipyrine, forming a quinone-imine red dye. The color intensity of the dye formed is directly proportional to the concentration of cholesterol. It is determined by measuring the increase in absorbance at 512 nm.
Statistical Analysis
Data are expressed as mean ± standard error of the mean (S.E.M.). Data were analyzed by Graphpad Prism® 5; the normality of data was evaluated by the D’Agostino and Pearson omnibus normality test. Statistical analysis was performed using a one-way analysis of variance (ANOVA) followed by the Newman-Keuls test when appropriate. Values of p < 0.05 were considered statistically significant. The Pearson correlation coefficient was used for correlation analysis.
Results
OFT
The number of rearings and crossings in the OFT is presented in Fig. 3a and b, respectively. The one-way ANOVA revealed a significant difference in the number of rearings (F(2,27) = 10.09, p < 0.001) and crossings (F(2,27) = 30.40, p < 0.0001) of rats. Aged rats had a reduction in the number of rearings (around 58%) (Fig. 3a) and crossings (around 48%) (Fig. 3b), when compared with the young adult rats. Treatment with 4-PSQ was not effective in reversing the number of rearings (Fig. 3a), but it partially restored the number of crossings (Fig. 3b).
Effect of 4-PSQ treatment on the exploratory (a) and locomotor (b) behaviors of rats in the open-field test (OFT). Values are expressed as mean ± standard error of the mean (n = 10 rats/group). **p < 0.01, ***p < 0.001, ****p < 0.0001 as compared with the young adult group. #p < 0.05, ####p < 0.0001 as compared with the aged group (one-way analysis of variance/Newman-Keuls test)
ORT
There was no significant difference among groups in the percentage of exploratory preference during the familiarization phase (F(2,27) = 1.576, p > 0.05) (data not shown).
In STM, the one-way ANOVA revealed a significant effect on the percentage of exploratory preference for a novel object in rats (F(2,27) = 11.61, p < 0.001). Aged rats had a reduction (around 43%) in the exploratory preference for the new object, when compared with young adult rats (Fig. 4a). The treatment of aged rats with 4-PSQ reversed this memory parameter in aged rats, reaching the levels of young adult rats (Fig. 4a).
Effect of 4-PSQ treatment on short-term (a) and long-term (b) memories in the object recognition test (ORT). Values are expressed as mean ± standard error of the mean (n = 10 rats/group). **p < 0.01, ***p < 0.001 as compared with the young adult group. ##p < 0.01, ###p < 0.001 as compared with the aged group (one-way analysis of variance/Newman-Keuls test)
In LTM, the one-way ANOVA had a significant effect on the exploratory preference percentage for a novel object in rats (F(2,27) = 14.61, p < 0.0001). Statistical analysis showed that the exploratory preference was reduced (around 54%) in the aged rats, when compared with the young adult rats (Fig. 4b). 4-PSQ treatment enhanced the percentage of preference for a novel object in aged rats, reaching values similar to those of young adult rats (Fig. 4b).
AChE Activity
The one-way ANOVA showed a significant difference in the AChE activity in the rat cerebral cortex (F(2,27) = 6.102, p < 0.01) and hippocampus (F(2,27) = 5.371, p < 0.05). The enzymatic activity was reduced in both brain structures of aged rats (around 40% for the cerebral cortex and 28% for the hippocampus), when compared with the young adult rats (Fig. 5a and b, respectively). Treatment with 4-PSQ was effective to reverse the inhibition on the AChE activity in the cerebral cortex (Fig. 5a) and hippocampus (Fig. 5b) of aged rats.
Effect of 4-PSQ treatment on the acetylcholinesterase (AChE) activity in the cerebral cortex (a) and hippocampus (b) of rats. Values are expressed as mean ± standard error of the mean (n = 10 rats/group). *p < 0.05 as compared with the young adult group. #p < 0.05, ##p < 0.01 as compared with the aged group (one-way analysis of variance/Newman-Keuls test)
The correlation analysis (Pearson’s correlation coefficient) revealed a positive correlation between the AChE activity in both cerebral structures and STM (r = 0.533; p < 0.05 for the cerebral cortex and r = 0.527; p < 0.05 for the hippocampus). Moreover, the correlation analysis (Pearson’s correlation coefficient) demonstrated a positive correlation between the AChE activity in both cerebral structures and LTM (r = 0.478; p < 0.05 for the cerebral cortex and r = 0.527; p < 0.05 for the hippocampus).
Expression of NCAM, STX, and PST
The one-way ANOVA of NCAM expression revealed significant changes in the cerebral cortex (F(2,18) = 5.256, p < 0.05) and hippocampus (F(2,18) = 11.26, p < 0.001) of rats. Aged rats had a reduction in cerebral cortex and hippocampus NCAM expression (around 24 and 14%, respectively) compared with young adult rats (Fig. 6a and b, respectively). Treatment of aged rats with 4-PSQ reversed the reduction of NCAM expression in both cerebral structures (Fig. 6a and b).
Effect of 4-PSQ treatment on the neural cell adhesion molecule (NCAM) levels in the cerebral cortex (a) and hippocampus (b) of rats. Values are expressed as mean ± standard error of the mean (n = 7 rats/group). *p < 0.05 as compared with the young adult group. #p < 0.05, ###p < 0.001 as compared with the aged group (one-way analysis of variance/Newman-Keuls test)
The one-way ANOVA of PST expression showed a significant difference in the cerebral cortex (F(2,18) = 11.25, p < 0.001) and hippocampus (F(2,18) = 5.554, p < 0.05) of rats. PST expression was reduced in both cerebral structures (around 32% for the cerebral cortex and 54% for the hippocampus) of aged rats, when compared with the young adult rats (Fig. 7a and b). 4-PSQ treatment was effective to restore the reduction of the PST expression in the cerebral cortex (Fig. 7a) and hippocampus (Fig. 7b) of aged rats.
Effect of 4-PSQ treatment on the polysialyltransferase PST levels in the cerebral cortex (a) and hippocampus (b) of rats. Values are expressed as mean ± standard error of the mean (n = 7 rats/group). *p < 0.05 as compared with the young adult group. #p < 0.05, ###p < 0.001 as compared with the aged group (one-way analysis of variance/Newman-Keuls test)
The one-way ANOVA demonstrated changes in STX expression in the cerebral cortex (F(2,18) = 6.000, p < 0.05), but there was no alteration in the hippocampus (F(2,18) = 0.6449, p > 0.05) of rats. STX expression did not change in the cerebral cortex and hippocampus of aged rats compared with young adult rats (Fig. 8a and b, respectively). The treatment of aged rats with 4-PSQ increased (around 45%) the STX expression in the cerebral cortex, when compared with aged and young adult rats (Fig. 8a), but there was no alteration in the hippocampus (Fig. 8b).
Effect of 4-PSQ treatment on the polysialyltransferase STX levels in the cerebral cortex (a) and hippocampus (b) of rats. Values are expressed as mean ± standard error of the mean (n = 7 rats/group). *p < 0.05 as compared with the young adult group. ##p < 0.01 as compared with the aged group (one-way analysis of variance/Newman-Keuls test)
Cholesterol Levels
The one-way ANOVA revealed a significant difference in cholesterol levels in the plasma of rats (F(2,18) = 6.249, p < 0.01). Cholesterol levels increased (around 54%) in the plasma of aged rats and treatment with 4-PSQ was effective to restore this biochemical marker (Fig. 9).
Discussion
In the present study, we provided evidence for the effect of 4-PSQ on the age-related cognitive impairment in rats. These results are strongly related to the improvement of synaptic plasticity, cholinergic system, and cholesterol levels of aged rats. Moreover, this study also demonstrated the influence of age on locomotor and exploratory activities.
Aging is associated with a decline in cognitive function, including learning and memory [38]. In the current study, we analyzed recognition memory by ORT, a non-spatial memory type based on the natural tendency of rodents to explore novel objects, which mainly relies on the function of the perirhinal cortex and hippocampus [39]. Our results demonstrated that aged rats had an age-related deficit in STM and LTM, as evidenced in ORT.
The most important finding of this study was that treatment with 4-PSQ restored cognitive deficits impaired by age in rats, enhancing STM, LTM, and spatial memory, since it caused an increase of the exploratory preference by the novel object and by location of the object in aging rats. The compound investigated in this study, 4-PSQ, is a quinoline derivative with an organoselenium substituent. Indeed, quinoline compounds have been widely studied and have presented promising biological activities, being found in pharmacologically active natural products, and extensively used in clinical practice [40]. In addition, it is known that the selenium status decreases with age and may contribute to impairment of neuropsychological functions [41], such as cognitive functions [17]. Thus, incorporating selenium into organic molecules is a good alternative to supplement with this element. In this context, studies have demonstrated that organoselenium compounds are good alternatives to improve the cognitive deficit caused by aging in rats [42]. Therefore, 4-PSQ is a promising molecule, since it combines the properties of quinoline derivatives with organic selenium compounds. In fact, our research group has demonstrated a significant number of pharmacological properties of this compound, such as being antioxidant, antinociceptive, anti-inflammatory, and anxiolytic-like [24, 26, 28,29,30].
In addition, the possibility of a biological and/or clinical connection between movement, cognition, and emotion has been investigated in the aging process [43]. In the present study, crossing and rearing behaviors of aged rats in the OFT were reduced compared with young adult rats, indicating that advanced age could cause an impairment in the locomotor and exploratory activities of the animals. Indeed, studies have shown that aging causes a reduction in these behaviors [20, 42]. Treatment with 4-PSQ for 7 days did not protect against the impairment in the locomotor and exploratory activities caused by aging. In accordance, other studies have shown the failure in the treatment of locomotor and exploratory disorders in aged rats [20, 42].
Cognitive processes, mediated through the hippocampus and the prefrontal cortex, are affected by aging [3, 5]. In the present study, we measured the AChE activity in the cerebral cortex and hippocampus of aged rats in an attempt to relate cognitive functions to the cholinergic system. Here, a decline in the AChE activity in aged rats as compared with the young adult rats was observed in both regions of the brain. In fact, a reduction on the AChE activity is an important marker of age-related cholinergic neurotransmission dysfunction. The central cholinergic system is constituted by neurotransmitter acetylcholine, which is involved in the regulation of cognitive functions [44]. In this way, studies have shown that a decrease of AChE activity by aging is due to acetylcholine content in the brain, since acetylcholine synthesis and release are diminished in aged animals [45]. Moreover, Richter et al. [46] in 2017 showed a negative correlation between older people and AChE activity.
Hence, the cholinergic system might be recommended as a cognitive dysfunction treatment strategy target [47]. Importantly, in the current study, treatment with 4-PSQ reversed the inhibition of AChE activity in both cerebral structures caused by aging. This finding suggests that an increase in AChE activity caused by 4-PSQ might be related to improvement obtained in behavioral tests and it is useful in the treatment of age-related memory impairments.
Evidences indicated that age-related cognitive decline is more likely associated with alterations in synaptic connectivity than with neuronal loss [3, 48]. In this sense, NCAMs have played a key role in neural development and have been implicated in synaptic plasticity and cognitive and emotional processes in adulthood [6, 7]. In the present investigation, our objective was verified whether 4-PSQ alleviates age-induced alterations in NCAMs. The aging process significantly decreases NCAM mRNA expression in the cerebral cortex and hippocampus of rats. These results are in accordance with those obtained in the behavioral tests, given that the memory of aged rats was impaired. Indeed, the literature suggests that NCAMs are indispensable for the formation and consolidation of learning and memory [7]. Treatment with 4-PSQ significantly enhanced NCAM levels in the cerebral cortex and hippocampus of aged rats, suggesting that this compound could reverse the inhibition of the transcription and translation of NCAMs induced by aging. Few studies have demonstrated the effect of organoselenium compounds on the expression of NCAMs. Wang et al. [49] showed that an organoselenium compound, selenomethionine, reversed the changes of NCAMs induced by lead.
In addition, aging reduced the mRNA levels of PST in the hippocampus and cerebral cortex of rats as compared with the young adult rats; however, no change was found in the mRNA levels of STX in both cerebral structures. PST (or ST8SiaIV) and STX (or ST8SiaII) are polysialyltransferases responsible for synthetizing α-2,8-polysialic acid and adding it to the extracellular domains of NCAM [50, 51]. During embryonic development and the early postnatal period, NCAM is heavily polysialylated (PSA-NCAM) and STX is dominant in these periods [52]. In adulthood, PSA-NCAM is restricted to specific regions of the brain, such as the hippocampus and cerebral cortex, which have roles in synaptic plasticity and general cell migration, PST being the main one associated with α-2,8-polysialylation in the postnatal brain [52, 53]. Our results suggested that aging downregulated PST mRNA in the hippocampus and cerebral cortex of rats, this being mainly found in the old ones, reducing polysialylation of NCAM, and decreasing synaptic plasticity in these cerebral regions. Importantly, treatment with 4-PSQ significantly enhanced PST levels in both cerebral structures of aged rats, and it upregulated STX levels in the cerebral cortex. These results indicated that 4-PSQ restored polysialyltransferases, mainly PST, increasing polysialylation of NCAM, restoring synaptic plasticity in cerebral structures of aged rats, improving the cognitive functions in the behavioral tasks.
In an attempt to explain the other mechanisms involved in 4-PSQ effects in the aged rats, cholesterol was measured since it plays an important role in maintaining the function of the central nervous system. In the present study, aged rats had an increase in the plasma cholesterol levels, when compared with young adult rats. Indeed, high cholesterol levels have detrimental effects on cognitive performance in animal models [54]. Additionally, there is an association between Alzheimer’s disease and cholesterol levels, potentially by increasing the neuronal content of cholesterol, promoting cleavage of the amyloid protein precursor, and an impaired working memory [55], affecting directly the neurodegeneration [56]. Several studies on humans investigated a relation between plasma lipids and dementia, showing that elderly people with neurodegenerative disease, such as Alzheimer’s disease, or dementia, and cognitive deficits have higher plasma total cholesterol [57]. Our results verified that 4-PSQ was able to restore the cholesterol levels in aged rats. Indeed, clinical studies suggested that statins might reduce the risk and progression of cognitive deficit in Alzheimer’s disease [58]. Therefore, the hypocholesterolemic effect of 4-PSQ is beneficial to restore the cognitive impairment caused by aging.
Conclusions
In conclusion, our findings demonstrated that 4-PSQ restored the cognitive impairment caused by aging in rats. In addition, the compound modulated the synaptic plasticity, cholinergic system, and cholesterol levels of aged rats. Thus, these results strongly contribute to the research of a novel therapeutic agent for geriatric memory deficit.
References
Antoniou M, Wright S (2017) Uncovering the mechanisms responsible for why language learning may promote healthy cognitive aging. Front Psychol 8:2217
Drachman DA (2006) Aging of the brain and Alzheimer’s disease. Neurology 67:1340–1352
Morrison JH, Baxter MG (2012) The ageing cortical synapse: hallmarks and implications for cognitive decline. Nat Rev Neurosci 13:240–250
Bloss EK, Janssen WG, Ohm DT, Yuk FJ, Wadsworth S, Saardi KM, McEwen BS, Morrison JH (2011) Evidence for reduced experience-dependent dendritic spine plasticity in the aging prefrontal cortex. J Neurosci 31:7831–7839
Driscoll I, Howard SR, Stone JC, Monfils MH, Tomanek B, Brooks WM, Sutherland RJ (2006) The aging hippocampus: a multi-level analysis in the rat. Neurosci 139:1173–1185
Conboy L, Bisak R, Markram K, Sandi C (2010) Role of NCAM in emotion and learning. Adv Exp Med Biol 663:271–296
Muller D, Mendez P, Deroo M, Klauser P, Steen S, Poglia L (2010) Role of NCAM in spine dynamics and synaptogenesis. Adv Exp Med Biol 663:245–256
Varea E, Castillo-Gomez E, Gomez-Climent MA, Guirado R, Blasco-Ibanez JM, Crespo C, Martinez-Guijarro FJ, Nacher J (2009) Differential evolution of PSA-NCAM expression during aging of the rat telencephalon. Neurobiol Aging 30:808–818
Pratt RD, Perdomo CA (2002) Donepezil-treated patients with probable vascular dementia demonstrate cognitive benefits. Ann N Y Acad Sci 977:513–522
Bohnen NI, Muller ML, Kuwabara H, Constantine GM, Studenski AS (2009) Age-associated leukoaraiosis and cortical cholinergic deafferentation. Neurology 72:1411–1416
Koenigsberger C, Chiappa S, Brimijoin S (1997) Neurite differentiation is modulated in neuroblastoma cells engineered for altered acetylcholinesterase expression. J Neurochem 69:1389–1397
Janeczek M, Gefen T, Samimi M, Kim G, Weintraub S, Bigio E, Rogalski M, Mesulam M et al (2018) Variations in acetylcholinesterase activity within human cortical pyramidal neurons across age and cognitive trajectories. Cereb Cortex 28:1329–1337
Mendez MF (2017) Early-onset Alzheimer disease. Neurol Clin 35(2):263–281
Martins IJ, Berger T, Sharman MJ, Verdile G, Fuller SJ, Martins RN (2009) Cholesterol metabolism and transport in the pathogenesis of Alzheimer’s disease. J Neurochem 111:1275–1308
Aisen PS, Cummings J, Jack CR et al (2017) On the path to 2025: understanding the Alzheimer’s disease continuum. Alzheimers Res Ther 9:5
Majkutewicz I, Kurowska E, Podlacha M, Myślińska D, Grembecka B, Ruciński J, Pierzynowska K, Wrona D (2018) Age-dependent effects of dimethyl fumarate on cognitive and neuropathological features in the streptozotocin-induced rat model of Alzheimer’s disease. Brain Res 1686:19–33
Rayman MP (2012) Selenium and human health. Lancet 379:1256–1268
Dominiak A, Wilkaniec A, Wroczynski P, Adamczyk A (2016) Selenium in the therapy of neurological diseases. Where is it going? Curr Neuropharmacol 14:282–299
Wilhelm EA, Souza AC, Gai BM, Chagas PM, Roehrs JA, Nogueira CW (2012) Hyperthermic seizures enhance responsiveness to pentylenetetrazole and induce cognitive dysfunction: protective effect of 3-alkynyl selenophene. Life Sci 90:666–672
Bortolatto CF, Wilhelm EA, Chagas PM, Nogueira CW (2012) p-Chloro-diphenyl diselenide, an organoselenium compound, with antidepressant-like and memory enhancer actions in aging male rats. Biogerontology 13:237–249
Bortolatto CF, Guerra Souza AC, Wilhelm EA, Nogueira CW (2013) Acute treatment with bis selenide, an organic compound containing the trace element selenium, prevents memory deficits induced by reserpine in rats. Biol Trace Elem Res 151:92–99
Jardim NS, Sartori G, Sari MHM, Müller SG, Nogueira CW (2017) Bisphenol A impairs the memory function and glutamatergic homeostasis in a sex-dependent manner in mice: beneficial effects of diphenyl diselenide. J Biol Inorg Chem 329:75–84
Savegnago L, Vieira AI, Seus N, Goldani BS, Castro MR, Lenardão EJ, Alves D (2013) Synthesis and antioxidant properties of novel quinoline–chalcogenium compounds. Tetrahedron Lett 54:40–44
Pinz M, Reis AS, Duarte V, da Rocha MJ, Goldani BS, Alves D, Savegnago L, Luchese C et al (2016) 4-Phenylselenyl-7-chloroquinoline, a new quinoline derivative containing selenium, has potential antinociceptive and anti-inflammatory actions. Eur J Pharmacol 780:122–128
Duarte LFB, Oliveira RL, Rodrigues KC, Voss GT, Godoi B, Schumacher RF, Perin G, Wilhelm EA et al (2017) Organoselenium compounds from purines: synthesis of 6-arylselanylpurines with antioxidant and anticholinesterase activities and memory improvement effect. Bioorg Med Chem 25:6718–6723
Reis AS, Pinz M, Duarte LFB, Roehrs JA, Alves D, Luchese C, Wilhelm EA (2017) 4-Phenylselenyl-7-chloroquinoline, a novel multitarget compound with anxiolytic activity: contribution of the glutamatergic system. J Psychiatr Res 84:191–199
Salgueiro WG, Goldani BS, Peres TV, Miranda-Vizuete A, Aschner M, Rocha JBT, Alves D, De Ávila DS (2017) Insights into the differential toxicological and antioxidant effects of 4-phenylchalcogenil-7-chloroquinolines in Caenorhabditis elegans. Free Radic Biol Med 110:133–141
Silva VDG, Reis AS, Pinz MP, da Fonseca CAR, Duarte LFB, Roehrs JA, Alves D, Luchese C et al (2017) Further analysis of acute antinociceptive and anti-inflammatory actions of 4-phenylselenyl-7-chloroquinoline in mice. Fundam Clin Pharmacol 31:513–525
Vogt AG, Voss GT, de Oliveira RL, Paltian JJ, Duarte LFB, Alves D, Jesse CR, Roman SS et al (2018) Organoselenium group is critical for antioxidant activity of 7-chloro-4-phenylselenyl-quinoline. Chem Biol Interact 282:7–12
Voss GT, Oliveira RL, De Souza JF, Duarte LFB, Fajardo AR, Alves D, Luchese C, Wilhelm EA (2018) Therapeutic and technological potential of 7-chloro-4-phenylselanyl quinoline for the treatment of atopic dermatitis-like skin lesions in mice. Mater Sci Eng C Mater Biol Appl 84:90–98
Wilhelm EA, Machado NC, Pedroso AB, Goldani BS, Seus N, Moura N, Savegnago L, Jacob RG et al (2014) Organocatalytic synthesis and evaluation of 7-chloroquinoline-1,2,3-triazoyl carboxamides as potential antinociceptive, antiinflammatory and anticonvulsant agent. RSC Adv 4:41437–41445
Saraiva MT, Krüger R, Baldinotti RS, Lenardão EJ, Luchese C, Savegnago L, Wilhelm EA, Alves D (2016) 7-Chloroquinoline-1,2,3-triazoyl carboxylates: organocatalytic synthesis and antioxidant properties. J Braz Chem Soc 27:41–53
Guide for the Care and Use of Laboratory Animals (1996) National Research Council (US) Committee for the Update of the Guide for the Care and Use of Laboratory Animals
Walsh RN, Cummins RA (1976) The open-field test: a critical review. Psychol Bull 83:482–504
De Lima MN, Laranja DC, Caldana F, Bromberg E, Roesler R, Schroder N (2005) Reversal of age-related deficits in object recognition memory in rats with l-deprenyl. Exp Gerontol 40:506–511
Ellman GL, Courtney KD, Andres V Jr, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Haradaa CN, Marissa C, Lovec N, Triebeld K (2013) Normal cognitive aging. Clin Geriatr Med 29:737–752
Barker GR, Warburton EC (2011) When is the hippocampus involved in recognition memory? J Neurosci 31:10721–10731
Vandekerckhove S, D’hooghe M (2015) Quinoline-based antimalarial hybrid compounds. Bioorg Med Chem 23:5098–5119
Akbaraly TN, Hinninger-Favier I, Carriere I, Arnaud J, Gourlet V, Roussel AM, Berr C (2007) Plasma selenium over time and cognitive decline in the elderly. Epidemiol 18:52–58
Cechella JL, Leite MR, Rosario AR, Sampaio TB, Zeni G (2014) Diphenyl diselenide-supplemented diet and swimming exercise enhance novel object recognition memory in old rats. Age 36:9666
Granholm AC, Boger H, Emborg ME (2008) Mood, memory and movement: an age-related neurodegenerative complex? Curr Aging Sci 1:133–139
Kilimann I, Hausner L, Fellgiebel A, Filippi M, Wurdemann TJ, Heinsen H, Teipel SJ (2017) Parallel atrophy of cortex and basal forebrain cholinergic system in mild cognitive impairment. Cereb Cortex 27:1841–1848
Das A, Dikshit M, Nath C (2001) Profile of acetylcholinesterase in brain areas of male and female rats of adult and old age. Life Sci 68:1545–1555
Richter N, Michel A, Onur OA, Kracht L, Dietlein M, Tittgemeyer M, Neumaier B, Fink GR et al (2017) White matter lesions and the cholinergic deficit in aging and mild cognitive impairment. Neurobiol Aging 53:27–35
Schliebs R, Arendt T (2011) The cholinergic system in aging and neuronal degeneration. Behav Brain Res 221:555–563
Peters A, Sethares C, Luebke JI (2008) Synapses are lost during aging in the primate prefrontal cortex. Neurosci 152:970–981
Wang M, Fu H, Xiao Y, Ai B, Wei Q, Wang S, Liu T, Ye L et al (2013) Effects of low-level organic selenium on lead-induced alterations in neural cell adhesionmolecules. Brain Res 1530:76–81
Nakayama J, Fukuda MN, Fredette B, Ranscht B, Fukuda M (1995) Expression cloning of a human polysialyltransferase that forms the polysialylated neural cell adhesion molecule present in embryonic brain. Proc Natl Acad Sci U S A 92:7031–7035
Scheidegger EP, Sternberg LR, Roth J, Lowe JB (1995) A human STX cDNA confers polysialic acid expression in mammalian cells. J Biol Chem 270:22685–22688
Hildebrandt H, Becker C, Murau M, Gerardy-Schahn R, Rahmann H (1998) Heterogenous expression of the polysialyltransferases ST8Sia II and ST8Sia IV during postnatal rat brain development. J Neurochem 71:2339–2348
Rutishauser U (2008) Polysialic acid in the plasticity of the developing and adult vertebrate nervous system. Nat Rev Neurosci 9:26–35
Cordner ZA, Tamashiro KL (2015) Effects of high-fat diet exposure on learning & memory. Physiol Behav 152:363–371
Paradis E, Clavel S, Julien P, Murthy MR, de Bilbao F, Arsenijevic D, Giannakopoulos P, Vallet P et al (2004) Lipoprotein lipase and endothelial lipase expression in mouse brain: regional distribution and selective induction following kainic acid induced lesion and focal cerebral ischemia. Neurobiol Dis 15:312–325
Ricciarelli R, Canepa E, Marengo B, Marinari UM, Poli G, Pronzato MA, Domenicotti C (2012) Cholesterol and Alzheimer’s disease: a still poorly understood correlation. IUBMB Life 64:931–935
He Q, Li Q, Zhao J, Wu J, Ji L, Huang G, Ma F (2016) Relationship between plasma lipids and mild cognitive impairment in the elderly Chinese: a case-control study. Lipids Health Dis 15:146
Haag MD, Hofman A, Koudstaal PJ, Stricker BH, Breteler MM (2009) Statins are associated with a reduced risk of Alzheimer disease regardless of lipophilicity. The Rotterdam Study. J Neurol Neurosurg Psychiatry 80:13–17
Funding
This study received financial support and scholarships from the following Brazilian agencies: Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (UNIVERSAL 408874/2016-3), Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS) (PRONEM 16/2551-0000240-1, PRONUPEQ 16/2551- 0000526-5, PqG 17/2551-0001013-2). This study was also financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível superior – Brasil (CAPES) - Finance Code 001. A.G.V. is the recipient of a FAPERGS fellowship. D.A., V.F.C., E.A.W., and C. L are recipients of CNPq fellowship. This study also received financial assistance from L’ORÉAL-UNESCO-ABC for Women in Science.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Animal care and all experimental procedures were conducted in compliance with the National Institute of Health Guide for the Care and Use of Laboratory Animals (NIH publications no. 80-23, revised in 1996) [33] and in accordance with the Committee on Care and Use of Experimental Animal Resources, Federal University of Pelotas, Brazil (CEEA 4599-2016). All efforts were made to minimize the number of animals used and their suffering.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(DOCX 968 kb)
Rights and permissions
About this article
Cite this article
Barth, A., Vogt, A.G., dos Reis, A.S. et al. 7-Chloro-4-(Phenylselanyl) Quinoline with Memory Enhancer Action in Aging Rats: Modulation of Neuroplasticity, Acetylcholinesterase Activity, and Cholesterol Levels. Mol Neurobiol 56, 6398–6408 (2019). https://doi.org/10.1007/s12035-019-1530-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-019-1530-5