Abstract
Innovations permeate healthcare settings on an ever-increasing scale. Health technology innovations (HTIs) impact our perceptions and experiences of health, care, disease, etc. Because of the fast pace these HTIs are being introduced in different healthcare settings, there is a growing societal consensus that these HTIs need to be governed by ethical reflection. This paper reports a systematic review of argument-based literature which focused on articles reporting on ethical frameworks to screen or evaluate HTIs. To do this a four step methodology was followed: (1) Literature search conducted in five electronic literature databases; (2) Identification of relevant articles; (3) Development of data-extraction tool to analyze the included articles; (4) Analysis, synthesis of data and reporting of results. Fifty-seven articles were included, each reporting on a specific ethical framework. These ethical frameworks existed out of characteristics which were grouped into five common ones: (1) Motivations for development and use of frameworks; (2) Objectives of using frameworks; (3) Specific characteristics of frameworks (background context, scope, and focus); (4) Ethical approaches and concepts used in the frameworks; (5) Methods to use the frameworks. Although this multiplicity of ethical frameworks shows an increasing importance of ethically analyzing HTIs, it remains unclear what the specific role is of these analyses. An ethics of caution, on which ethical frameworks rely, guides HTIs in their design, development, implementation, without questioning their technological paradigm. An ethics of desirability questions this paradigm, without guiding HTIs. In the end, a place needs to be found in-between, to critically assess HTIs.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
On an ever-increasing scale, technological innovations are permeating healthcare settings. Throughout history, technologies have had an enormous impact on how health, care, and disease is experienced. This technological trend has intensified in recent years and will continue in the foreseeable future (Flear et al., 2013; Lehoux, 2006; Udeh, 2000). In agreement with Warren-Jones (2013) we describe health technology innovations (HTIs) as material expressions of human knowledge in devices, techniques, procedures, and systems that are or will be used in a healthcare context. These innovations are manifest in all aspects of healthcare, from medical treatments (e.g., genetic therapy, pharmacogenetics); use of materials (e.g., nanotechnology, 3D bio-printing); medical and care support (e.g., robotics, body sensors); to data gathering and analysis (e.g., e-health, big data). All HTIs have in common the overall goal of preventing, handling, or solving healthcare problems and guaranteeing, or at least aim to increase, the quality of life of people (Banta, 2009; World Health Organization, 2010, 2019).
Although HTIs hold potential to help people cultivate the “good life”, multiple possible, sometimes unforeseen, consequences might occur. These include possible impacts on the autonomy of stakeholders (e.g. care receivers, caregivers, their social network); how HTIs will influence and mediate individual, organizational, and societal conceptions of health, disease, medicine, and care; and whether they will enhance justice and equality of access to healthcare for everyone (Calnan et al., 2005; Chan, 2018). An even more important question arises: Should cultivating the “good life” be conceived as a goal for which HTIs are developed and used or also just a possible consequence of developing and using HTIs? Indeed, have we not entered a circle of HTI adoption in which specific HTIs are developed to counter the (unforeseen) effects of other, more mature, HTIs? In this sense, HTIs can be described as “medicines”, consisting of an internal tension between being curative or toxic tools (Lemmens, 2017) or between hope and fear (Vos & Willems, 2000).
During the past few years, there is growing societal consensus that the design, development, implementation and use of HTIs needs to be governed by ethical reflection. In both clinical practice and research, there is an increasingly expressed need for user friendly ethical frameworks to screen or evaluate HTIs. Until now, most attention has focused on ethical frameworks directed at implementation or use of HTI, especially in the context of Health Technology Assessment (HTA). More specifically, focus has centered on which ethical frameworks are used in HTA (Assasi et al., 2014); on reasons to include ethics in HTA (Hofmann, 2008); on guidelines about how to integrate ethics into HTA (Assasi et al., 2014); on specific barriers and enablers to use ethical frameworks (Assasi et al., 2015; Bellemare et al., 2018). Nevertheless, there is a growing call for paying more attention to the design and development of technologies so that their ethical impact can be fully understood. This is exemplified in discourses like Value Sensitive Design (VSD) (Friedman, 1996; Winkler & Spiekermann, 2018) and Responsible Research and Innovation (RRI) (Burget et al., 2017; Stilgoe et al., 2013). Is there a trend also to consider these foci in design and development of HTIs?
Both in the literature, in clinical practice and in healthcare policy, we detect an imperative to develop a systematic and integrated account of ethical frameworks used to screen or evaluate HTIs, one going beyond the regular HTA-discourse. This review answers the following question: “Which are the ethical frameworks described in the international scientific literature that govern the ethical screening or evaluation of HTIs?”.
Methods
Design
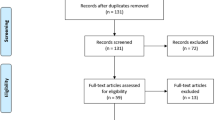
A systematic review of argument-based literature was conducted in three steps: (1) develop a search-string strategy to use in identifying relevant articles in scientific electronic literature databases; (2) assessment and inclusion of only relevant literature and; (3) extract and synthesize data for meaning (McCullough et al., 2007; McDougall 2014; Mertz et al., 2016; Sofaer & Strech, 2012). The search and selection process were conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Flow Diagram (Liberati et al., 2009).
Search Strategy
On December 6, 2019, five electronic literature databases were queried using a search strategy consisting of a string of three word-groups. The first referred to technological health innovations, the second to the fact that the literature had to be ethics literature, and the third to the fact that the literature had to report on frameworks (Supplementary Data 1). The first author (TV) developed the search strategy and discussed it repeatedly with the co-authors (CG and YD). The search strategy was first developed to be applied in Pubmed. Afterwards it was modified so it could be applied in other databases. No data range delimiters were used.
Inclusion and Exclusion Criteria
Search results were put together in a reference manager (Endnote™ version 7.4., Clarivate Analytics, Philadelphia, PA, USA). Before the process of inclusion and exclusion, all duplicates were removed. Next, we screened the publication type and excluded those publications that were not published in peer-reviewed journals.
Specific inclusion criteria are presented in Table 1. These criteria were systematically and sequentially applied to titles, abstracts, and full texts (Liberati et al., 2009). Two authors (TV and CG) independently screened the titles of publications. The eligibility of some publications was uncertain but these were discussed among the two authors until consensus was reached. During the screening of the abstracts and full-texts a third author (YD) joined the assessment of the publications about which there was doubt if these were eligible. Again, discussion took place between the three researchers (TV, CG, YD) until consensus were reached. Snowballing was used to identify possibly overlooked publications. Figure 1 presents the search and screening processes.
Search and screening process (Patterned after Liberati et al., (2009))
Data Extraction and Synthesis
We developed and used a custom, in-house tool to extract data from the included articles. The extraction tool went through several iterations of refinement by the research group (Supplementary Data 2).
Data Extraction Tool
The final version of the extraction tool consisted of two parts. The first part focused on the characteristics of each article: (1) publication year; (2) the country where the research was conducted (country of the first author); (3) research discipline of the first author; (4) research focus of the journal of publication. The second part of the extraction tool considered 10 characteristics related to the reported ethical framework to screen or evaluate HTIs: (1) name of the framework; (2) how the framework was described in the article; (3) focus of the framework (e.g., generally applicable to any HTI or aimed at one specifically); (4) context in which the framework was developed or used; (5) stage of HTI development on which the framework focuses; (6) why the framework was developed or used; (7) purpose of the ethical framework; (8) structure or method of the ethical framework; (9) ethical content of the framework; (10) ethical theory underlying the reported framework. For each of the included publications the final version of the extraction tool was used to evaluate, and document the relevant data (Supplementary Data 2).
Extraction and Synthesis of Data
Although the extraction tool gave us several focus points to extract relevant data, the synthesis of data happened inductively as it was inspired by the five preparatory stages of the coding process of the Qualitative Analysis Guide of Leuven (QUAGOL) (Dierckx de Casterlé et al., 2012, 2021). First, the first author (TV) thoroughly familiarized himself with the included articles. Second, the extraction tool was used to obtain all relevant data and to develop an individual conceptual scheme (Supplementary Data 3). Included articles were analyzed in sets of five. After each set, the resulting conceptual schemes were discussed by two authors (TV and CG). Third, common publication characteristics and the characteristics of the reported ethical frameworks were identified by comparing the developed conceptual schemes on a per-characteristic basis. Fourth, based on these comparisons, similarities and differences between the publication characteristics and the reported ethical frameworks were summarized in tables. This comparison was conducted by the first author (TV) and discussed with the co-author (CG) on a regular basis. Fifth, these tables were then consulted extensively during the writing of the results section of this review.
Results
Fifty-seven publications met our inclusion criteria (Supplementary Data 4). The structure of the results section follows the order in which the characteristics were listed in the extraction tool (Supplementary Data 2).
Characteristics of Included Articles
Publication characteristics are presented in Table 2. Publication dates ranged from 1991 to 2019. More than 65% of the publications were published after 2013 suggesting a recent increase of interest in ethical frameworks to screen or evaluate HTIs. The countries where the research was mainly conducted were in Europe and North America. Only two were conducted in an Asian country. No studies originated from African or South American countries.
Based on the professional background of the first authors, the majority of the included articles were written by philosophers or ethicists. The backgrounds of the first authors for the remainder were in the clinical sciences, management, policy, or technology development. These trends coincided with the scope of the journal in which the articles appeared.
Characteristics of Ethical Frameworks
Each of the 57 included articles reported on a specific ethical framework that can be used for the ethical screening or evaluation of HTIs. To gain insight into the form and content of these frameworks, we present the results on their characteristics of which some have been put under the same heading.
Motivations for Development and Use of Ethical Frameworks
There were different motivations to develop or use ethical frameworks for the screening or evaluation of HTIs (Table 3). These motivations stemmed from observations that current ethical frameworks are no longer relevant to new HTIs; from the need to gain a realistic understanding of the general impact of HTIs; the need to adequately address the perspectives on HTIs of different stakeholders; the need to analyze possible impacts of new HTIs on social, cultural, political, and religious dimensions of society; the need to implement ethical analysis in policy decisions about HTIs; and the need for ethical considerations about whether specific HTIs and their funding should be prioritized in health care. The common thread is that they are all grounded in the idea that ethical analysis is a necessary step to gain in-depth insight into HTIs, their benefits and risks to society, its institutions, its citizens, as well as to morality itself.
Objectives of Ethical Frameworks
The development or use of ethical frameworks was done with different objectives in mind (Table 4). We found that most ethical frameworks were developed to descriptively screen or normatively evaluate HTIs, or do both.
Screening of HTIs entails a descriptive method of illuminating ethical tensions. It mostly consists of a continuous reflective process that is engaged throughout the unfolding of a new technology, including during the design, development, implementation, and use stages. Evaluation refers to the normative method of developing an ethical judgment of a particular HTI. This judgement is the result of an ethical analysis and usually takes place during the stage of implementation or use of an HTI.
These two main objectives can be further subdivided into six specific objectives that emerged out of our analysis. They all share the premise that HTIs not only need to be economically and clinically beneficial, but also need to meet specific ethical standards on a societal, organizational, or individual level. These specific objectives are summarized in Table 4.
Specific characteristics of Ethical Frameworks
We identified three characteristics that provide deeper insight into the particularity of the ethical frameworks.
Background Context
Ethical frameworks are usually developed as a result of a certain project, method or strategy, i.e. a certain background context. The three major background contexts we identified are HTA, VSD, and RRI (Table 5).
Twenty-five articles reported on an ethical framework related to HTA, which entails a multidisciplinary process in which relevant and validated evidence on a specific HTI is synthesized. This evidence covers several aspects of an HTI, such as clinical effectiveness, safety, cost-efficiency, and social and ethical impact. Strikingly, all articles in this category pointed to an “ethical deficit” in HTA, which manifested in statements that ethical analyses of an HTI are often ignored, do not have a real impact, or are considered as necessary theoretically but practically are viewed as a “fifth wheel”. Therefore, the aim of most articles within an HTA context was to counter this shortfall, both in terms of content and method. The deficit was addressed in content by proposing ethical principles or visions, for example, and in method by providing suggestions on how to integrate ethical analysis into a broader HTA, for example the INTEGRATE-HTA methodology (Bond & Weeks, 2017; Lysdahl et al., 2017).
The background context of four articles were related to VSD. While the ethical frameworks in the HTA context mainly focused on implementation or routine use of HTIs, the VSD frameworks almost exclusively focused on design and development and viewed values more broadly. Besides the ethical values related to their use, HTIs also embody the values that the developers consider to be important. For example, preventive health apps embody the value of good health but also of cost-efficiency. Additionally, HTIs embody the values that developers believe potential users value (e.g. user-friendliness of health apps). Finally, HTIs embody a number of values that are considered to be important in society (e.g. health apps as a sign of universally accessible basic care). The VSD approach stresses that these different ethical values need to be given prime attention during the design and development of an HTI. For example, if respect for privacy is highly valued, then it should be given upfront attention by integrating data encryption into HTIs early on in their design.
A third context in which ethical frameworks were developed or used related to RRI; six articles were in this category. This context refers to a transparent and interactive process of HTI research and development in which all relevant stakeholders are actively involved. This transparency and interaction aimed to integrate ethical values that are widely supported in society into HTIs. Compared to VSD, RRI has a broader perspective, as it mostly focuses on the full life cycle of a technology, from conceptualization and extraction of minerals to decomposition and recycling.
Scope
The scope of the ethical frameworks varied from general and generic to a focus on a particular HTI or family of HTIs. Ethical frameworks with a general scope are those that can be applied in principle to all HTIs (Bond & Weeks, 2017; Baltussen et al., 2017; Burls et al., 2011; Giacomini, 2005; Gibson et al., 2002; Goetghebeur & Cellier, 2018; Heintz et al., 2015; Hofmann, 2005; Hofmann et al., 2014; Lysdahl et al., 2017; Marckmann et al., 2015; Pacifico Silva et al., 2018; Saarni et al., 2008; Sacchini et al., 2009). Some publications focus on one or more particular HTI as case studies, e.g. care robots (Amigoni & Schiaffonati, 2018; Coeckelbergh, 2010; Coeckelbergh, 2016; Misselhorn et al., 2013; Sharkey, 2014; Sorell & Draper, 2014; Stahl & Coeckelbergh, 2016; van der Plas et al., 2010; van Wynsberghe, 2013a; van Wynsberghe, 2013b), cochlear implants (Daniels & van der Wilt, 2016; Reuzel et al., 1999, 2001; van der Wilt et al., 2000) or telehealth (Demiris et al., 2006; Kiran et al., 2015; Perry et al., 2009) but their scope is wider than these particular cases.
Additionally, there are ethical frameworks with a specific scope (Ajunwa et al., 2016; Whedon & Ferrell, 1991; Boers et al., 2019; Denecke, 2017; Stylianou & Talias, 2017; Torous & Roberts, 2017; Stol et al., 2017; Howard et al., 2013; Kosta et al., 2010). These frameworks arise from the development, implementation or use of a particular HTI.
Focus
Based on the included literature, we distinguish four stages of technology development on which the ethical frameworks focus: design, development, implementation, and use. More than half of the reported ethical frameworks focus on the implementation and use of HTI (Ajunwa et al., 2016; Autti-Rämö & Mäkelä, 2007; Baltussen et al., 2017; Bond & Weeks, 2017; Demiris et al., 2006; Di Pietro et al., 2018; Ebbesen & Jensen, 2006; Giacomini, 2005; Gibson et al., 2002; Goetghebeur & Cellier, 2018; Goetghebeur et al., 2010; Heintz et al., 2015; Hofmann, 2005; Hofmann, 2017; Hofmann et al., 2014; Howard et al., 2013; Kosta et al., 2010; Lysdahl, et al., 2017, Perry et al., 2009; Reuzel et al., 1999; Reuzel et al., 2001; Saarni et al., 2008; Sacchini et al., 2009; Sacchini et al., 2015; Saifuddeen et al., 2013; Saifuddeen et al., 2014; Sharkey, 2014; Stol et al., 2017; Stylianou & Talias, 2017; Thorstensen, 2019; van der Plas et al., 2010; van der Wilt et al., 2000; Whedon & Ferrell, 1991). Six of the frameworks focused on the development, implementation and use of HTIs (Boers et al., 2019; Burls et al., 2011; Daniels & van der Wilt, 2016; Denecke, 2017; Gutiérrez-Ibarluzea, 2012; van Wynsberghe, 2013b) and two exclusively on HTI design (Timmermans et al., 2011; van Wynsberghe, 2013a). Seven ethical frameworks focused on all four stages of technology development (Aicardi et al., 2018; Coeckelbergh, 2010, 2016; Fothergill et al., 2019; Kiran et al., 2015; Misselhorn et al., 2013; Sorell & Draper, 2014). Three frameworks were developed to ethically analyze the full life cycle (from mineral extraction and conceptualization to recycling) of HTIs (Lipworth & Axler, 2016; Pacifico Silva et al., 2018; Stahl & Coeckelbergh, 2016).
Foundational Ethical Approaches and Concepts Used in Ethical Frameworks
Ethical frameworks for HTIs used different ethical concepts that are part of different predominant traditions of ethical approaches (Table 6). These concepts and approaches are the cornerstones on which the ethical frameworks rely. Of eight articles, the foundational ethical approach and/or concepts were not specifically stated nor were they inferable (Table 6).
Principles of Biomedical Ethics Approach
The four principles of biomedical ethics—“respect for autonomy,” “beneficence,” “non-maleficence” and “justice”—are integrated in a number of ethical frameworks (Beauchamp & Childress, 2001) (Table 6).
With “respect for autonomy”, the included articles pointed to the importance of individual freedom (of choice) (Amigoni & Schiaffonati, 2018; Demiris et al., 2006; Ebbesen & Jensen, 2006; Gutiérrez-Ibarluzea, 2012; Heintz et al., 2015; Kosta et al., 2010; Marckmann et al., 2015; Perry et al., 2009; Sorell & Draper, 2014; Soril et al., 2016; Torous & Roberts, 2017; Whedon & Ferrell, 1991). Respect for autonomy means that everyone must be supported in making autonomous decisions and that an individual’s choices should not be limited. If limits are imposed they always arise from a conflict with one of the other principles (Ebbesen & Jensen, 2006; Perry et al., 2009).
Both the positive and negative formulations of respect for autonomy are used to support healthcare recipients in their choice on whether or not to use HTIs (Amigoni & Schiaffonati, 2018; Demiris et al., 2006; Ebbesen & Jensen, 2006; Gutiérez-Ibarluzea, 2012; Marckmann et al., 2015; Perry et al., 2009; Sorell & Draper, 2014). In addition, potential healthcare recipients are seen as stakeholders in the technology decision-making process (Amigoni & Schiaffonati, 2018; Heintz et al., 2015; Torous & Roberts, 2017). Respect for autonomy is the ethical foundation for many other reported principles and criteria such as protecting privacy when using health technology (Demiris et al., 2006; Heintz et al., 2015; Kosta et al., 2010; Marckmann et al., 2015; Perry et al., 2009; Sorell & Draper, 2014) and providing informed consent (Amigoni & Schiaffonati, 2018; Demiris et al., 2006; Ebbesen & Jensen, 2006; Marckmann et al., 2015; Perry et al., 2009; Torous & Roberts, 2017).
The second principle “beneficence” refers to actions that are good for others based on the premise that these actions are good in themselves. “Others” can be individuals, groups, organizations, or society as a whole. The third principle “non-maleficence” refers to a minimum of doing good, namely avoiding harmful risks or actions. This principle is grounded in the moral intuition that intentionally harming a person is fundamentally wrong (Ebbesen & Jensen, 2006; Perry et al., 2009). Although beneficence and non-maleficence are two separate principles, they are often grouped in the analyzed articles, more specifically in balancing the risks and benefits associated with HTIs (Ebbesen & Jensen, 2006; Gutiérrez-Ibarluzea, 2012; Heintz et al., 2015; Marckmann et al., 2015; Perry et al., 2009; Saarni et al., 2008; Sorell & Draper, 2014; Soril et al., 2016; Torous & Roberts, 2017; Whedon & Ferrell, 1991). In the context of HTI use, it is not always clear what exactly should be considered as risks and benefits (Howard et al., 2013). Therefore, many authors recommend doing a thorough examination of HTIs, case-by-case, before using one routinely. In this way, the risks and benefits at an individual and social level are gradually identified (Amigoni & Schiaffonati, 2018; Gutiérrez-Ibarluzea, 2012; Heintz et al., 2015; Howard et al., 2013; Marckmann et al., 2015; Saarni et al., 2008; Soril et al., 2016; Whedon & Ferrell, 1991). Some of the included articles posed specific points of attention: who decides what a good balance is between risks and benefits (Saarni et al., 2008); what is the impact of the use of HTIs on the relationship between healthcare recipients and caregivers (Demiris et al., 2006; Perry et al., 2009; Whedon & Ferrell, 1991) and on the social network of care recipients (Ebbesen & Jensen, 2006; Howard et al., 2013; Perry et al., 2009; Sorell & Draper, 2014; Whedon & Ferrell, 1991); what can we say about the reliability of HTIs (Ebbesen & Jensen, 2006; Kosta et al., 2010); and what is the impact of HTIs on care recipients’ dignity and integrity (Ebbesen & Jensen, 2006; Gutiérrez-Ibarluzea, 2012; Perry et al., 2009)?
The fourth principle “justice” ensures that everyone is treated fairly and equally and that risks and benefits of an HTI are fairly distributed among the involved stakeholders. Depending on the context in which it is used, the principle is given a specific interpretation (Ebbesen & Jensen, 2006; Perry et al., 2009). Next to the fair distribution of risks and benefits (Amigoni & Schiaffonati, 2018; Marckmann et al., 2015; Saarni et al., 2008), the principle mainly points to the question of equal access to HTIs, regardless of one’s status, financial capabilities and physical or cognitive abilities (Demiris et al., 2006; Ebbesen & Jensen, 2006; Heintz et al., 2015; Howard et al., 2013; Kosta et al., 2010; Marckmann et al., 2015; Perry et al., 2009; Saarni et al., 2008; Soril et al., 2016; Whedon & Ferrell, 1991). In addition, some authors highlight the responsible use of resources (including public finances) to develop HTIs (Heintz et al., 2015; Howard et al., 2013; Soril et al., 2016), while others highlight the need to critically examine the effect of HTIs on the stigmatization of certain populations (Gutiérrez-Ibarluzea, 2012; Perry et al., 2009; Sorell & Draper, 2014); to provide compensation for any damage caused by an HTI (Amigoni & Schiaffonati, 2018; Marckmann et al., 2015); and to examine the digital divide between different population groups (Demiris et al., 2006).
Two publications did not use the four biomedical ethical principles to conduct analyses, but rather applied them to gain deeper insight into and structure existing ethical tensions with regard to HTIs which were brought forward through literature studies (Raimond et al., 2018) or stakeholder meetings (Autti-Rämö & Mäkelä, 2007).
Deliberative Democratic Approaches
In several articles, the ethical framework for HTIs was based on deliberative approaches such as: Wide Reflective Equilibrium (WRE), Accountability for Reasonableness (A4R) and Interactive Technology Assessment (ITA). Because of their emphasis on interaction, deliberation, and basic democratic principles, these different approaches are grounded in deliberative or democratic ethics (Table 6). In this approach, ethics is essentially conceived as an interactive process between different stakeholders in which their ethical values are deepened and a consensus is sought. The focus is on creating conditions that guarantee that previously disengaged or disenfranchised individuals can comment on social evolutions, such as HTI development.
WRE is the most common deliberative approach. Its aim was to produce insight into the moral principles and viewpoints that stakeholders use to make their moral judgment about HTIs. Through interaction, a coherent whole between all these applied principles and viewpoints was made. This coherent whole forms a consensus among the different stakeholders concerning the development or use of a particular HTI. This consensus can then be translated to recommendation(s) to policymakers.
The A4R approach applies four basic conditions that guarantee the deliberative democratic nature of the interactive WRE process and the reasonableness of the consensus. These conditions are:
-
Publicity: Publicity holds that the decision-making process regarding the HTI is transparent. Due attention must be paid to the motives driving the decisions.
-
Relevance: Relevance means that the stated motives are appropriate and related to an HTI and likely acceptable to all stakeholders.
-
Revisability: Revisability implies that every deliberative process comprises procedures and methods that make it possible to review and correct previous decisions about an HTI in light of new evidence or new arguments.
-
Enforcement: Enforcement means that all of the above criteria must be met during a WRE process. All three of these criteria are of equally importance.
Our analyses of several articles showed that WRE and A4R can be too abstract to be applied solely for concrete evaluations of HTIs. To avoid this abstractness, WRE and A4R were combined with other approaches. These articles emphasized the importance of empirical evidence, such as medical efficacy and cost-effectiveness, and advocated reaching a balance between facts and values (Baltussen et al., 2017; Gibson et al., 2002; Goetghebeur & Cellier, 2018).
Religiously Inspired Approaches
Personalist Approaches
Three articles make explicit use of a personalist approach (Di Pietro et al., 2018; Sacchini et al., 2009, 2015) (Table 6). Emerging from a Judeo-Christian context and inspired by an Aristotelian-Thomistic ethical perspective, personalism states that the human person, in all her/his dimensions (physical, psychological, relational, social, and spiritual) should be the basic reference value in any ethical evaluation, and so also those of HTIs.
This basic premise translates into four operational principles. The first principle, “defense of human physical life,” expresses the need to conceive the human body in a holistic way which deserves ongoing and never ending respect. The second principle, “safeguard of the therapeutic principle,” holds that an HTI is ethically acceptable when it meets specific conditions e.g., a sole focus on the sick part of the body, invasive treatments only when none-invasive treatments are ineffective (proportionality) (Sacchini et al., 2015). The third principle is “freedom and responsibility,” where “freedom” refers to the need of care recipients’ giving their informed consent, or their non-consent, in a free way before implementing an HTI (Sacchini et al., 2009, 2015). “Responsibility” holds that the involved parties, e.g., care recipients and caregivers, take responsibility for the use of an HTI and its possible consequences (Sacchini et al., 2015). The fourth principle stresses the importance of “sociality and subsidiarity,” which means that all citizens must, among other things, show respect for themselves and each other and that society—both government and private initiatives —must offer support to those who cannot meet their own needs without undermining the place of citizens' initiatives (Di Pietro et al., 2018; Sacchini et al., 2009, 2015).
Islamic Principles
Two articles used a specific Islamic approach in their ethical framework. Saifuddeen et al., (2013, 2014) relied on five principles of the maqassid al shariah in evaluating HTIs.
“Protection of faith” means practically that any HTI is consistent with Islamic teachings as outlined in foundational Islamic sources. “Protection of life” means, practically, that any HTI must contribute to a flourishing life. HTIs cannot pose any risk to human life and the environment in which people live. “Protection of intellect” holds that HTIs may not adversely affect the human mind. “Protection of progeny” forbids HTIs that could harm or endanger human offspring. “Protection of property” holds that HTIs may not result in the destruction of human property. These five principles of the maqassid al shariah have their origins in the foundational Islamic sources: Quran, Sunnah of the Prophet (tradition), Ijtihad, and the Shariah (Islamic divine law).
AREA Framework
Six articles advanced an ethical framework that is grounded in RRI (Table 5). Three of these articles explicitly refer to the four dimensions of the AREA framework: “Anticipate,” “Reflect,” “Engage,” and “Act” (Aicardi et al., 2018; Fothergill et al., 2019; Stahl & Coeckelbergh, 2016). It is argued that these dimensions should be incorporated into the full life cycle of HTIs in order to ensure a positive ethical impact (Table 6). These dimensions are often translated into critical questions that are addressed throughout the different stages of an HTI (i.e., design, development, implementation and use).
Lipworth and Axler (2016) and Pacifico Silva et al., (2018) do not explicitly refer to these dimensions. Nevertheless, we contend that to a certain extent, Pacifico Silva et al.’s (2018) nine dimensions (health relevance, ethical, legal and social implications (ELSI), health equity, inclusiveness, responsiveness, level of care, frugality, business model, eco-responsibility) belonging to their proposed five value domains related to HTIs (population health, health system, economic domain, organizational, and environmental) and Lipworth and Axler’s (2016) four dimensions of a bioethics of innovation (anticipation, reflexivity, inclusiveness, responsiveness) can be placed into the AREA framework. The four dimensions of the AREA framework are:
-
1.
Anticipate: Anticipate holds that during the development of HTIs, one must be constantly aware of all possible difficulties, tensions and impact associated with its use. Strategies must be developed that enable us to deal with possible negative impacts. For example, what will happen if “big data” systems in healthcare are hacked (Fothergill et al., 2019)? Are certain population groups advantaged or disadvantaged by the use of a particular HTI (Lipworth & Axler, 2016; Pacifico Silva et al., 2018; Stahl & Coeckelbergh, 2016)?
-
2.
Reflect: Reflect challenges HTI developers and users to identify in advance the motivations behind the products they develop or use and to identify the results they want to achieve (Aicardi et al., 2018; Fothergill et al., 2019; Lipworth & Axler, 2016; Pacifico Silva et al., 2018; Stahl & Coeckelbergh, 2016). Where does the idea come from to develop or use a particular HTI (Fothergill et al., 2019; Lipworth & Axler, 2016)? Why are certain objectives chosen in spite of other? Who benefits from the use of that HTI (Pacifico Silva et al., 2018; Stahl & Coeckelbergh, 2016)?
-
3.
Engage: Engage refers to the need to involve all real and potential stakeholders related to a particular HTI (Aicardi et al., 2018; Fothergill et al., 2019; Lipworth & Axler, 2016; Pacifico Silva et al., 2018; Stahl & Coeckelbergh, 2016). Likewise, the natural environment and climate must be recognized as stakeholders (Pacifico Silva et al., 2018). This involvement has a critical function as it can reveal arbitrary presuppositions that are deemed to be self-evident, unconsciously held prejudices, or lop-sided power relations between stakeholders (Lipworth & Axler, 2016; Stahl & Coeckelbergh, 2016). Do stakeholders have the choice to use an HTI, or will its use become mandatory? This involvement of stakeholders aims not only to create public support for an HTI, but also to make it more socially responsible (Aicardi et al., 2018; Fothergill et al., 2019; Pacifico Silva et al., 2018).
-
4.
Act: Act refers to the idea that during the complete HTI life-cycle needs to actively incorporate the insights developed during the ethical process of anticipate, reflect and engage. It is a call to conceive ethical assessment as an integrated part of the design, development, implementation and use of HTIs (Aicardi et al., 2018; Fothergill et al., 2019; Lipworth & Axler, 2016; Pacifico Silva et al., 2018; Stahl & Coeckelbergh, 2016) and not simply as a necessary add-on.
These dimensions do not necessarily unfold chronologically. As their description indicates, their content overlaps. They are preferably integrated in a cyclical process in which the dimension of “act” provides new input for the continuous application of the three other dimensions (Aicardi et al., 2018; Fothergill et al., 2019; Pacifico Silva et al., 2018; Stahl & Coeckelbergh, 2016).
Some authors who use the AREA framework admit that it is abstract. Therefore, they complement it with specific elements of other frameworks such as the 4P’s of technology development (process, product, purpose, people) (Stahl & Coeckelbergh, 2016); the data cycle in healthcare (Fothergill et al., 2019); or bioethics (Lipworth & Axler, 2016).
Capabilities Approaches
In four articles, the ethical framework was based on the central human capabilities (Tables 6 and 7) of the capabilities approach (Nussbaum & Sen, 1993). This approach focuses on social justice and aims to show what it means for people to live a dignified life within a fair and just society. Dignity is analyzed from the perspective of the human person in relation to the different social contexts (e.g., family, organization, society, nation, world) in which we live.
For HTIs, Nussbaum’s (2006) interpretation of the capabilities approach serves as the basis for some of the described ethical frameworks. It develops a fine-tuned picture of what constitutes a dignified human life that flourishes through 10 central human capabilities (Tables 6 and 7). Through the lens of the capabilities approach, care is understood as a practice that contributes to a dignified and flourishing human life. To achieve this, procedures and structures must be developed (Sharkey, 2014), of which HTIs can be one. HTIs can then be evaluated via these capabilities.
The capabilities approach makes clear that the option of HTIs (e.g. care robots) should not a priori be evaluated in a negative way, but should always be evaluated through the lens of a particular context. This also means that if an HTI no longer contributes to the ideal of good and dignified care, there must exist the possibility to stop using that HTI without compromising the quality of care (Coeckelbergh, 2010, 2016; Misselhorn et al., 2013).
The list of central human capabilities (Table 7) is not considered to be exhaustive. The ethical frameworks indicate that technology influences these capabilities, even to the extent that some of them could disappear and others could manifest themselves in the future (Coeckelbergh, 2016; Misselhorn et al., 2013). For example, digital literacy could be seen as a newly emerged capability which is essential to achieve a flourishing dignified life.
Care Ethical Approaches
Van Wynsberghe, (2013a, 2013b) proposed the Care-Centered Framework for the ethical evaluation of HTIs. It comprises five components and can be applied prospectively (during the design and development stage) (Wynsberghe, 2013a, 2013b) or retrospectively (after implementation) (van Wynsberghe, 2013b) (Table 6). The evaluation of HTIs is based on the following components: (1) the context in which HTI is used (e.g., care for older adults, a medical intervention); (2) the medical or care treatment for which the HTI is developed or used (e.g., diagnostic research, surgical interventions, distribution of lunch in a nursing home); (3) the stakeholders involved in the healthcare act (e.g., What roles do healthcare actors take on during the act and how are responsibilities divided during the act before HTI implementation? How could the use of an HTI affect these roles and responsibilities?); (4) the HTI that is being developed or used; (5) the evaluation is also based on four moral attitudes that need to be present in every healthcare act.
These four moral attitudes, originally described by Tronto, (1993), are attentiveness, responsibility, competency, and responsiveness. They are the foundations of care ethics, an ethical theory which considers care acts as those human acts that are always value-loaded since they always take place in a relational context. The use of any HTI should help support the development of these moral attitudes in healthcare (van Wynsberghe, 2013a, 2013b). Thus, HTIs can be evaluated against those four attitudes.
Casuistic Approaches
Some articles advanced the idea that specific HTIs can only be assessed on the premises of the context in which these are being used (Table 6). Ethical principles and values can be used as a guide in the ethical review, but this can never be done in a universal way. How certain ethical principles and values were implemented in previous cases and contexts can at most indicate a certain direction for the evaluation of new HTIs.
Eclectic Approaches
Several articles based their ethical framework for HTIs on a combination of the above and other approaches and principles (Table 6). Hence, these ethical frameworks are grounded in a broad and diverse range of sources. These articles can be divided into two groups. The largest group draws the ethical concepts from various ethical theories such as principlism, axiology, value ethics, consequentialism or utilitarianism. Another group develop their ethical framework from concepts of sociological disciplines that are supplemented by insights from ethics, bioethics, and philosophy.
Outliers
Some articles based their ethical framework on procedures and principles from theories not used in others, namely, post-phenomenology (Kiran et al., 2015) and research/data ethics (Ajunwa et al., 2016).
Methods Used in Ethical Frameworks
We identified several methods that the authors of the included articles used in applying ethical frameworks to screen or evaluate HTIs. These methods are closely related to the above-described ethical concepts and approaches and the contextual background of the frameworks (Table 8).
The most commonly used method was systematically applying principles, capabilities, or attitudes, as ethical criteria to assess the implementation or use of HTIs. The method mostly evaluated the impact (e.g., on care recipients’ autonomy or dignity) and the possible consequences of a particular HTI (e.g. reinterpretation of the relationship between caregivers and care recipients, or redistribution of subsidies).
Another method often used comprised a deliberative procedure. The objective was to interactively involve all relevant stakeholders in the implementation and/or use of HTIs through a deliberative reflection exercise. This interaction was intended to produce a broad ethical view of HTIs with the aim of identifying ethical tensions that are often not visible using a systematic application of, for example, ethical principles. Although these interactions often presuppose a consensus about HTIs, a deliberative procedure usually has a screening goal (descriptive) rather than an evaluative (normative) goal.
Other methods aim to systematically integrate ethical principles or values into the design or during the entire development process of HTIs. This method was usually used in the context of VSD or RRI. By closely scrutinizing the ethical tensions related to an HTI and then by developing strategies to deal with them at an earlier stage (e.g., robust encryption keys for personal data, compensation payments for possible environmental damage) the mitigation of these ethical tensions is aimed for. Ethical principles or values which are highly values are then guaranteed from an early stage in the life-cycle of an HTI.
Some articles proposed ethical questionnaires as the methodology of their ethical frameworks. These questionnaires were mostly inspired by ethical concepts rooted in different ethical theories. They are aimed to highlight ethical principles or values that stakeholders considered vis-à-vis an HTI. The questions covered all dimensions regarding the use of an HTI, such as the impact that it could have on users (e.g., in terms of privacy) or on the end-user organization. Although these questionnaires could be used individually to analyze and evaluate an HTI, in most articles it is argued that they are used as a discussion tool in a deliberative setting.
Lastly, there were authors that used their ethical framework not specifically to evaluate a particular HTI, but rather to organize the collected evidence about it. In the same vein other authors relied on previous ethical screenings or evaluations of HTIs to guide their own.
Discussion and Conclusion
Methodological Discussion
This systematic review analyzed several ethical frameworks reported in the literature related to the screening and evaluation of HTIs. We restricted our search to peer-review articles in academic journals and used a rigorous procedure to identify 57 relevant articles. Guided by the PRISMA statement (Liberati et al., 2009), we first developed and applied an all-encompassing search string. Second, we applied pre-defined inclusion and exclusion criteria for candidate publications. Third, we constructed and used a novel data-extraction tool to analyze the different characteristics of the ethical specific ethical frameworks reported.
Included articles were diverse, originating in several disciplines including philosophy, ethics, technology development, clinical sciences and policy or management. Five common characteristics of the various ethical frameworks were discernable: (1) motivations behind the development and use; (2) objectives of their development and use; (3) characteristics related to their background context, scope, and focus; (4) ethical concepts and approaches on which the various frameworks rely; and (5) methodologies to use the frameworks in the real world. Despite the thoroughness of the current review, it needs to be said that the included publications mainly stem from western countries. This territorial narrowness can be due to the used electronic databases to find the relevant literature. Moreover, it can also point to a technological deficit in non-western countries in comparison with western countries by which ethical frameworks would not be necessary. Hence, the question arises, and remains, what this deficit specifically entails. Finally, this review’s exclusive focus on ethical frameworks should not blind us of the fact that ethical screening or evaluation of HTIs does not happen in isolation. In applied situations, they are accompanied and influenced by medical studies, effectiveness studies, economic analysis, etc., as project, such as the INTEGRATE-HTA project, shows (Bond & Weeks, 2017; Gerhardus et al., 2017; Lysdahl et al., 2017; Wahlster et al., 2017).
Discussion of the Ethics of Ethical Frameworks
This review’s scope contrasted that of most existing reviews of ethical frameworks developed for screening or evaluating HTIs (Assasi et al., 2014, 2015; Bellemare et al., 2018) as it went beyond the specific context of HTA. Similar to Reijers et al., (2018), we focused on frameworks developed or used for emerging technologies (ex ante), for technology design (intra), and for existing technologies (ex post). Hence, this review highlighted several ethical theories and discourses underlying existing frameworks. Other studies have also highlighted principlism, casuistry, deliberative models, triangular model, axiology, context-sensitive approaches, eclectic approaches, among others (Assasi et al., 2014; Hofmann, 2008). However, our review also identified other theories and discourses, for example the AREA framework, care ethics approach, capabilities approach, Islamic theology, and post-phenomenology.
The included articles applied ethics in several different ways to screen or evaluate HTIs. Nevertheless, it remains unclear what the precise function could be of such an ethical screening or evaluation. This lack of clarity is because, in relation to HTIs, the nature of ethics as a discourse has become muddled. It is unclear if the purpose of ethics is to scrutinize HTIs in the sense of assessing its desirability, or whether the purpose of ethics is to focus attention on those aspects of HTIs that should meet certain ethical standards, in the sense of implementing HTIs in a cautious and responsible manner (Have, 2004)? The difference made in this review between “ethical evaluation” and “ethical screening” already referred to this tension to a certain extent. Whereas the ethics of desirability places itself outside the technological paradigm so to be able to critically question HTIs, the ethics of caution places itself inside the technological paradigm so to guide this’ design, development, implementation, and use. It is clear from this review that most existing ethical frameworks are aligned with the ethics of caution.
Moreover, with the influence of technological mediation discourses, it seems that the emphasis on an ethics of caution has intensified. Although ethical analysis used to focus on the use of HTIs, it now is increasingly focused on the design and development stages. This trend is clear for those frameworks used in the context of VSD and RRI. The drive behind this shift is how HTIs are viewed. Whereas they used to be viewed as neutral instruments meant to be used for reaching certain healthcare goals, they are now viewed as embodiments of certain values and principles held by the different stakeholders in the technology life-cycle (designer, developer, user, society etc.). Moreover, it is now recognized that HTIs also shape and reshape our conceptions of life, health, care, and even ethics, according to those embodied values and principles. Indeed, as Stilgoe et al., (2013, p. 1570) state, in technology governance a shift has taken place from a product focus, to a process and integrity focus, and now to a process focus which includes “[…] questions of uncertainty (in its multiple forms), purposes, motivations, social and political constitutions, trajectories and directions of innovation”. By including ethics from the design and development stage onwards, it is argued that HTIs will become more ethically responsive once they are used.
Although introducing ethics in the complete life cycle of HTIs has been a necessary and welcomed shift in focus, it still does not lead to answer the question whether a particular HTI is desirable or not. Referring to Joseph Schumpeter’s basic characterization of innovation as “creative destruction” (Schumpeter, 1943), Blok and Lemmens, (2015, p. 30) argue that the “[…] construction of new and innovative solutions is accompanied by the annihilation or destruction of the old rules and the old order (including the ‘writing off’ of ‘obsolescent’ skills, technologies and capital stocks), i.e. the positive impact of responsible innovation and contribution to the public good, is therefore accompanied by negative impacts elsewhere”. Indeed, even if an HTI is responsive to all the ethical sensitivities of stakeholders in a specific context and does justice to important values in that context, the question remains whether all possible negative impacts were considered to their fullest extent. In an increasingly connected world, it is necessary to consider the possible impacts of the development and use of an HTI across social and societal borders. What is the environmental and climate impact of that HTI? Under which labor conditions will the natural and technical elements necessary to make and use that HTI be collected and manufactured? Moreover, this connected ethical view on HTIs also challenges us to view healthcare and medical settings in a much more integrated way instead of viewing them as being divorced from any broader social, societal, and environmental ties. In the end, the notion of innovation as “creative destruction” provokes the question whether our current concept of innovation in health is indeed meant to be inherently good (Blok & Lemmens, 2015).
The tension between the ethics of caution and the ethics of desirability in relation to HTIs is grounded in the difference between working from inside the technological paradigm—(taking the HTI for granted)—or from outside it—(not taking the HTI for granted)—(Have, 2004). Despite the broadening of the ethics of caution, by focusing on the design and development stage of HTIs, it seems that the ethics of desirability is inescapable. The lesson that we can learn from this, is that the best position for ethics is the place between caution and desirability. This in-between place ensures that HTIs will not be left without ethical guidance and also avoids the perception of HTIs as being self-evident. Moreover, this place lowers the risk of “ethics washing”, namely a too superficial interpretation of what ethics is and should be.
Future research on ethical frameworks for HTIs should have the goal of broadening their focus to include all stages of the HTI life-cycle, environmental aspects, global social aspects, and intergenerational aspects, among others. We believe that this can be achieved by continuing to expand the groups of relevant stakeholders and to include stakeholders that represent those that cannot “speak for themselves” (e.g. environment, future generations). More importantly it will be critical to embrace the idea that HTIs may not be a self-evident necessity.
Availability of Data and Materials
Not applicable.
Code Availability
Not applicable.
References
Aicardi, C., Fothergill, B. T., Rainey, S., Stahl, B. C., & Harris, E. (2018). Accompanying technology development in the Human Brain Project: From foresight to ethics management. Futures, 102, 114–124. https://doi.org/10.1016/j.futures.2018.01.005
Ajunwa, I., Craford, K., & Ford, J. S. (2016). Health and big data: An ethical framework for health information collection by corporate wellness programs. Journal of Law, Medicine & Ethics, 44(3), 474–480. https://doi.org/10.1177/1073110516667943
Amigoni, F., & Schiaffonati, V. (2018). Ethics for robots as experimental technologies. Pairing anticipation with exploration to evaluate the social impact of robotics. IEEE Robotics & Automation Magazine, 25(1), 30–36. https://doi.org/10.1109/MRA.2017.2781543
Assasi, N., Schwartz, L., Tarride, J.-E., Campbell, K., & Goeree, R. (2014). Methodological guidance documents for evaluation of ethical considerations in health technology assessment: A systematic review. Expert Review of Pharmacoeconomics & Outcomes Research, 14(2), 203–220. https://doi.org/10.1586/14737167.2014.894464
Assasi, N., Schwartz, L., Tarride, J.-E., O’Reilly, D., & Goeree, R. (2015). Barriers and facilitators influencing ethical evaluation in health technology assessment. International Journal of Technology Assessment in Health Care, 31(3), 113–123. https://doi.org/10.1017/S026646231500032X
Autti-Rämö, I., & Mäkelä, M. (2007). Ethical evaluation in health technology assessment reports: An eclectic approach. International Journal of Technology Assessment in Health Care, 23(1), 1–8. https://doi.org/10.1017/S0266462307051501
Baltussen, R., Jansen, M. P. J., Bijlmakers, L., Grutters, J., Kluytmans, A., Reuzel, R., Tummers, M., & van der Wilt, G.-J. (2017). Value assessment frameworks for HTA agencies: The organization of evidence-informed deliberative processes. Value in Health, 20(2), 256–260. https://doi.org/10.1016/j.jval.2016.11.019
Banta, D. (2009). What is technology assessment? International Journal of Technology Assessment in Health Care, 25(S1), 7–9. https://doi.org/10.1017/S0266462309090333
Beauchamp, T. L., & Childress, J. F. (2001). Principles of biomedical ethics (5th ed.). Oxford University Press.
Bellemare, C. A., Dagenais, P., Suzanne, K., Béland, J. P., Bernier, L., Daniel, C. É., Daniel, C. É., Gagnon, H., Legault, G. A., Parent, M., & Patenaude, J. (2018). Ethics in health technology assessment: A systematic review. International Journal of Technology Assessment in Health Care, 34(5), 447–457. https://doi.org/10.1017/S0266462318000508
Blok, V., & Lemmens, P. (2015). The emerging concept of responsible innovation. Three reasons why it is questionable and calls for a radical transformation of the concept of innovation. In B.-J. Koops, I. Oosterlaken, H. Romijn, T. Swierstra, & J. van den Hoven (Eds.), Responsible innovation 2. Concepts approaches, and applications (pp. 19–35). Springer.
Boers, S. N., van Delden, J. J. M., & Bredenoord, A. L. (2019). Organoids as hybrids: Ethical implications for the exchange of human tissues. Journal of Medical Ethics, 45(2), 131–139. https://doi.org/10.1136/medethics-2018-104846
Bond, K., & Weeks, L. (2017). Using the INTEGRATE-HTA guidance: Experience from CADTH. International Journal of Technology Assessment in Health Care, 33(5), 591–596. https://doi.org/10.1017/S0266462317000940
Burget, M., Bardone, E., & Pedaste, M. (2017). Definitions and conceptual dimensions of responsible research and innovation: A literature review. Science and Engineering Ethics, 23(1), 1–19. https://doi.org/10.1007/s11948-016-9782-1
Burls, A., Caron, L., Cleret de Langavant, G., Dondorp, W., Harstall, C., Pathak-Sen, E., & Hofmann, B. (2011). Tackling ethical issues in health technology assessment: A proposed framework. International Journal of Technology Assessment in Health Care, 27(3), 230–237. https://doi.org/10.1017/S0266462311000250
Calnan, M., Montaner, D., & Horne, R. (2005). How acceptable are innovative health-care technologies? A survey of public beliefs and attitudes in England and Wales. Social Science & Medicine, 60(9), 1937–1948. https://doi.org/10.1016/j.socscimed.2004.08.058
Chan, S. (2018). Research translation and emerging health technologies: Synthetic biology and beyond. Health Care Analysis, 26(4), 310–325. https://doi.org/10.1007/s10728-016-0334-2
Coeckelbergh, M. (2010). Health care, capabilities, and AI assistive technologies. Ethical Theory and Moral Practice, 13, 181–190. https://doi.org/10.1007/s10677-009-9186-2
Coeckelbergh, M. (2016). Care robots and the future of ICT-mediated elderly care: A response to doom scenarios. AI & Society, 31(4), 455–462. https://doi.org/10.1007/s00146-015-0626-3
Daniels, N., & van der Wilt, G. J. (2016). Health technology assessment, deliberative process, and ethically contested issues. International Journal of Technology Assessment in Health Care, 32(1), 10–15. https://doi.org/10.1017/S0266462316000155
Demiris, G., Parker Oliver, D., & Courtney, K. L. (2006). Ethical considerations for the utilization of telehealth technologies in home and hospice care by the nursing profession. Nursing Administration Quarterly, 30(1), 56–66. https://doi.org/10.1097/00006216-200601000-00009
Denecke, K. (2017). An ethical assessment model for digital disease detection technologies. Life Sciences, Society and Policy, 13(1), 1–11. https://doi.org/10.1186/s40504-017-0062-x
Di Pietro, M. L., Teleman, A. A., Gonzalez-Melado, F. J., Zace, D., Di Raimo, F. R., Lucidi, V., & Refolo, P. (2018). Implementing carrier screening for cystic fibrosis outside the clinic: Ethical analysis in the light of the personalist view. La Clinical Terapeutica, 169(2), e71–e76. https://doi.org/10.7417/T.2018.2057
Dierckx de Casterlé, B., De Vliegher, K., Gastmans, C., & Mertens, E. (2021). Complex qualitative data analysis: Lessons learned from the experiences with the qualitative analysis guide of Leuven. Qualitative Health Research, 31(6), 1083–1093. https://doi.org/10.1177/1049732320966981
Dierckx de Casterlé, B., Gastmans, C., Bryon, E., & Denier, Y. (2012). QUAGOL: A guide for qualitative data analysis. International Journal of Nursing Studies, 49(3), 360–371. https://doi.org/10.1016/j.ijnurstu.2011.09.012
Ebbesen, M., & Jensen, T. G. (2006). Nanomedicine: Techniques, potentials, and ethical implications. Journal of Biomedicine and Biotechnology. https://doi.org/10.1155/JBB/2006/51516
Flear, M. L., Farrell, A.-M., Hervey, T. K., & Murphy, T. (Eds.). (2013). European law and new health technologies. Oxford University Press.
Fothergill, B. T., Knight, W., Stahl, B. C., & Ulnicane, E. (2019). Responsible data governance of neuroscience big data. Frontiers in Neuroinformatics. https://doi.org/10.3389/fninf.2019.00028
Friedman, B. (1996). Value-sensitive design. Interactions, 3(6), 16–23. https://doi.org/10.1145/242485.242493
Gerhardus, A., Oortwijn, W., & van der Wilt, G. J. (2017). How to avoid giving the right answers to the wrong questions: The need for integrated assessments of complex health technologies. International Journal of Technology Assessment in Health Care, 33(5), 541–543. https://doi.org/10.1017/S0266462317001040
Giacomini, M. (2005). One of these things is not like the others: The idea of precedence in health technology assessment and coverage decisions. The Milbank Quarterly, 83(2), 193–223. https://doi.org/10.1111/j.1468-0009.2005.00344.x
Gibson, G. L., Martin, D. K., & Singer, P. A. (2002). Priority setting for new technologies in medicine: A transdisciplinary study. BMC Health Services Research. https://doi.org/10.1186/1472-6963-2-14
Goetghebeur, M. M., & Cellier, M. S. (2018). Can reflective multicriteria be the new paradigm for healthcare decision-making? The EVIDEM journey. Cost Effectiveness and Resource Allocation. https://doi.org/10.1186/s12962-018-0116-9
Goetghebeur, M. M., Wagner, M., Khoury, H., Rindress, D., Grégoire, J.-P., & Deal, C. (2010). Combining multicriteria decision analysis, ethics and health technology assessment: Applying the EVIDEM decisionmaking framework to growth hormone for Turner syndrome patients. Cost Effectiveness and Resource Allocation. https://doi.org/10.1186/1478-7547-8-4
Gutiérrez-Ibarluzea, I. (2012). Personalised health care, the need for reassessment. A HTA perspective far beyond cost-effectiveness. Italian Journal of Public Health, 9(4), e8653. https://doi.org/10.2427/8653
Heintz, E., Lintamo, L., Hultcrantz, M., Jacobson, S., Levi, R., Munthe, C., Tranæus, S., Östlund, P., & Sandman, L. (2015). Framework for systematic identification of ethical aspects of healthcare technologies: The SBU approach. International Journal of Technology Assessment in Health Care, 31(3), 124–130. https://doi.org/10.1017/S0266462315000264
Hofmann, B. (2005). Toward a procedure for integrating moral issues in health technology assessment. International Journal of Technology Assessment in Health Care, 21(3), 312–318. https://doi.org/10.1017/S0266462305050415
Hofmann, B. (2008). Why ethics should be part of health technology assessment. International Journal of Technology Assessment in Health Care, 24(4), 423–429. https://doi.org/10.1017/S0266462308080550
Hofmann, B. (2017). Toward a method for exposing and elucidating ethical issues with human cognitive enhancement technologies. Science and Engineering Ethics, 23(2), 413–429. https://doi.org/10.1007/s11948-016-9791-0
Hofmann, B., Droste, S., Oortwijn, W., Cleemput, I., & Sacchini, D. (2014). Harmonization of ethics in health technology assessment: A revision of the socratic approach. International Journal of Technology in Health Care, 30(1), 3–9. https://doi.org/10.1017/S0266462313000688
Howard, H. C., Swinnen, E., Douw, K., Vondeling, H., Cassiman, J.-J., Cambon-Thomsen, A., & Borry, P. (2013). The ethical introduction of genome-based information and technologies into public health. Public Health Genomics, 16(3), 100–109. https://doi.org/10.1159/000346474
Kiran, A. H., Oudshoorn, N., & Verbeek, P.-P. (2015). Beyond checklists: Toward an ethical-constructive technology assessment. Journal of Responsible Innovation, 2(1), 5–19. https://doi.org/10.1080/23299460.2014.992769
Kosta, E., Pitkänen, O., Niemelä, M., & Kaasinen, E. (2010). Mobile-centric ambient intelligence in health- and homecare—anticipating ethical and legal challenges. Science and Engineering Ethics, 16(2), 303–323. https://doi.org/10.1007/s11948-009-9150-5
Lehoux, P. (2006). The problem of health technology. Policy implications for modern health care systems. Routledge.
Lehoux, P., & Blume, S. (2000). Technology assessment and the sociopolitics of health technologies. Journal of Health Politics, Policy and Law, 25(6), 1083–1112. https://doi.org/10.1215/03616878-25-6-1083
Lemmens, P. (2017). Social Autonomy and heteronomy in the age of ICT: The digital pharmakon and the (dis)empowerment of the general intellect. Foundations of Science, 22(2), 287–296. https://doi.org/10.1007/s10699-015-9468-1
Liberati, A., Altman, D. G., Tetzlaff, J., Mulrow, C., Gøtzsche, P. C., Ioannidis, J. P. A., Clarke, M., Devereaux, P. J., Kleijnen, J., & Moher, D. (2009). The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ, 339, b2700. https://doi.org/10.1136/bmj.b2700
Lipworth, W., & Axler, R. (2016). Towards a bioethics of innovation. Journal of Medical Ethics, 42(7), 445–449. https://doi.org/10.1136/medethics-2015-103048
Lysdahl, K. B., Mozygemba, K., Burns, J., Brönneke, J. B., Chilcott, J. B., Ward, S., & Hofmann, B. (2017). Comprehensive assessment of complex technologies: Integrating various aspects in health technology assessment. International Journal of Technology Assessment in Health Care, 33(5), 570–576. https://doi.org/10.1017/S0266462317000678
Marckmann, G., Schmidt, H., Sofaer, N., & Strech, D. (2015). Putting public health ethics into practice: A systematic framework. Frontiers in Public Health. https://doi.org/10.3389/fpubh.2015.00023
McCullough, L., Coverdale, J. H., & Chervenak, F. A. (2007). Constructing a systematic review for argument-based clinical ethics literature: The example of concealed medications. The Journal of Medicine and Philosophy, 32(1), 65–76. https://doi.org/10.1080/03605310601152206
McDougall, R. (2014). Systematic reviews in bioethics: Types, challenges, and value. Journal of Medicine and Philosophy, 39(1), 89–97. https://doi.org/10.1093/jmp/jht059
Mertz, M., Kahrass, H., & Strech, D. (2016). Current state of ethics literature synthesis: A systematic review of reviews. BMC Medicine, 14(1), 152. https://doi.org/10.1186/s12916-016-0688-1
Misselhorn, C., Pompe, U., & Stapleton, M. (2013). Ethical considerations regarding the use of social robots in the fourth age. GeroPsych, 26(2), 121–133. https://doi.org/10.1024/1662-9647/a000088
Nussbaum, M. (2006). Frontiers of justice: Disability, natinality, species membership. The Belknap Press of Harvard University Press.
Nussbaum, M., & Sen, A. (Eds.). (1993). The quality of life. Clarendon Press.
Pacifico Silva, H., Lehoux, P., Miller, F. A., & Denis, H.-L. (2018). Introducing responsible innovation in health: A policy-oriented framework. Health Research Policy and Systems, 16(1), 90. https://doi.org/10.1186/s12961-018-0362-5
Perry, J., Beyer, S., & Holm, S. (2009). Assistive technology, telecare, and people with intellectual disabilities: Ethical considerations. Journal of Medical Ethics, 35(2), 81–86. https://doi.org/10.1136/jme.2008.024588
Raimond, V., Sambuc, C., & Pibouleau, L. (2018). Ethics evaluation revealing decision-maker motives: A case of neonatal screening. International Journal of Technology Assessment in Health Care, 34(2), 189–195. https://doi.org/10.1017/S0266462318000181
Reijers, W., Wright, D., Brey, P., Weber, K., Rodrigues, R., O’Sullivan, D., & Gordijn, B. (2018). Methods for practising ethics in research and innovation: A literature review, critical analysis and recommendations. Science and Engineering Ethics, 24(5), 1437–1481. https://doi.org/10.1007/s11948-017-9961-8
Reuzel, R. P. B., van de Wilt, G.-J., ten Have, H. A. M. J., & de Vries, R. P. F. (2001). Interactive technology assessment and wide reflective equilibrium. Journal of Medicine and Philosophy, 26(3), 245–261. https://doi.org/10.1076/jmep.26.3.245.3015
Reuzel, R. P. B., van der Wilt, G.-J., ten Have, H. A. M. J., & de Vries Robbé, P. F. (1999). Reducing normative bias in health technology assessment: Interactive evaluation and casuistry. Medicine, Health Care and Philosophy, 2(3), 255–263. https://doi.org/10.1023/A:1009963018813
Saarni, S. I., Hofmann, B., Lampe, K., Lühmann, D., Mäkelä, M., Velasco-Garrido, M., & Autti-Rämö, I. (2008). Ethical analysis to improve decision-making on health technologies. Bulletin of the World Health Organization, 86(8), 617–623. https://doi.org/10.2471/BLT.08.051078
Sacchini, D., Craxì, L., Refolo, P., Minacori, R., Cicchetti, A., Gasbarrini, A., Cammà, C., & Spagnolo, A. G. (2015). Ethical assessment of hepatitis C virus treatment: The lesson from first generation protease inhibitors. Digestive and Liver Disease, 47(5), 351–355. https://doi.org/10.1016/j.dld.2014.11.011
Sacchini, D., Virdis, A., Refolo, P., Pennacchini, M., & Carrasco de Paula, I. (2009). Health technology assessment (HTA): Ethical aspects. Medicine, Health Care and Philosophy, 12(4), 453–457. https://doi.org/10.1007/s11019-009-9206-y
Saifuddeen, S. M., Lee Wei, C., Ibrahim, A. H., & Khotib, A. M. (2013). Islamic ethical framework to tackle scientific and technological dilemmas. Journal of Dharma, 38(4), 373–386.
Saifuddeen, S. M., Rahman, N. N. A. R., Isa, N. M., & Baharuddin, A. (2014). Maqasid al-Shariah as complementary framework to conventional bioethics. Science and Engineering Ethics, 20(2), 317–327. https://doi.org/10.1007/s11948-013-9457-0
Schumpeter, J. A. (1943). Capitalism, socialism & democracy. Routledge.
Sharkey, A. (2014). Robots and human dignity: A consideration of the effects of robot care on the dignity of older people. Ethics and Information Technology, 16(1), 63–75. https://doi.org/10.1007/s10676-014-9338-5
Sofaer, N., & Strech, D. (2012). The need for systematic reviews of reasons. Bioethics, 26(6), 315–328. https://doi.org/10.1111/j.1467-8519.2011.01858.x
Sorell, T., & Draper, H. (2014). Robot carers, ethics, and older people. Ethics and Information Technology, 16(3), 183–195. https://doi.org/10.1007/s10676-014-9344-7
Soril, L. J. J., Clement, F. M., & Nosworthy, T. W. (2016). Bioethics, health technology reassessment, and management. Healthcare Management Forum, 29(6), 275–278. https://doi.org/10.1177/0840470416659385
Stahl, B. C., & Coeckelbergh, M. (2016). Ethics of healthcare robotics: Towards responsible research and innovation. Robotics and Autonomous Systems, 86, 152–161. https://doi.org/10.1016/j.robot.2016.08.018
Stilgoe, J., Owen, R., & Macnaghten, P. (2013). Developing a framework for responsible innovation. Research Policy, 42(9), 1568–1580. https://doi.org/10.1016/j.respol.2013.05.008
Stol, Y., Schermer, M. H. N., & Asscher, A. C. A. (2017). Omnipresent health checks may result in over-responsibilization. Public Health Ethics, 10(1), 35–48. https://doi.org/10.1093/phe/phw034
Stylianou, A., & Talias, M. A. (2017). Big data in healthcare: A discussion on the big challenges. Health and Technology, 7(1), 97–107. https://doi.org/10.1007/s12553-016-0152-4
ten Have, H. (2004). Ethical perspectives on health technology assessment. International Journal of Technology Assessment in Health Care, 20(1), 71–76. https://doi.org/10.1017/S0266462304000819
Thorstensen, E. (2019). Stakeholders’ views on responsible assessments of assistive technologies through an ethical HTA matrix. Societies, 9(3), 51. https://doi.org/10.3390/soc9030051
Timmermans, J., Zhao, Y., & van den Hoven, J. (2011). Ethics and nanopharmacy: Value sensitive design of new drugs. NanoEthics, 5(3), 269–283. https://doi.org/10.1007/s11569-011-0135-x
Torous, J., & Roberts, L. W. (2017). The ethical use of mobile health technology in clinical psychiatry. The Journal of Nervous and Mental Disease, 205(1), 4–8. https://doi.org/10.1097/NMD.0000000000000596
Tronto, J. (1993). Moral boundaries: A political argument for an ethic of care. Routledge.
Udeh, C. I. (2000). New technologies in medicine. JAMA, 283(19), 2592. https://doi.org/10.1001/jama.283.19.2592
van der Plas, A., Smits, M., & Wehrmann, C. (2010). Beyond speculative robot ethics: A vision assessment study on the future of the robotic caretaker. Accountability in Research, 17(6), 299–315. https://doi.org/10.1080/08989621.2010.524078
van der Wilt, G. J., Reuzel, R., & Banta, D. H. (2000). The ethics of assessing health technologies. Theoretical Medicine and Bioethics, 21(1), 101–113. https://doi.org/10.1023/A:1009934700930
van Wynsberghe, A. (2013a). A method for integrating ethics into the design of robots. Industrial Robot: An International Journal, 40(5), 433–440. https://doi.org/10.1108/IR-12-2012-451
van Wynsberghe, A. (2013b). Designing robots for care: Care centered value-sensitive-design. Science and Engineering Ethics, 19(2), 407–433. https://doi.org/10.1007/s11948-011-9343-6
Vos, R., & Willems, D. L. (2000). Technology in medicine: Ontology, epistemology, ethics and social philosophy at the crossroads. Theoretical Medicine and Bioethics, 21(1), 1–7. https://doi.org/10.1023/A:1009903321229
Wahlster, P., Brereton, L., Burns, J., Hofmann, B., Mozygemba, K., Oortwijn, W., Pfadenhauer, L., Polus, S., Rehfuess, E., Schilling, I., & Gerhardus, A. (2017). An integrated perspective on the assessment of technologies: INTEGRATE HTA. International Journal of Technology Assessment in Health Care, 33(5), 544–551. https://doi.org/10.1017/S0266462317000484
Warren-Jones, A. (2013). Mapping science and new health technologies: In search of a definition. In M. L. Flear, A.-M. Farrell, T. K. Hervey, & T. Murphy (Eds.), European law and new health technologies (pp. 70–100). Oxford University Press.
Whedon, M., & Ferrell, B. R. (1991). Professional and ethical considerations in the use of high-tech pain management. Oncology Nursing Forum, 18(7), 1135–1143.
Winkler, T., & Spiekermann, S. (2018). Twenty years of value sensitive design: A review of methodological practices in VSD projects. Ethics and Information Technology, 23(1), 17–21. https://doi.org/10.1007/s10676-018-9476-2
World Health Organization. (2010). Innovative technologies that address global health concerns. Outcome of the call Global Initiative on Health Technologies. WHO.
World Health Organization (2019). What is a health technology? Retrieved December 11, 2019 from https://www.who.int/health-technology-assessment/about/healthtechnology/en/.
Acknowledgements
This review was done as part of a larger research project about ethical frameworks used to screen or evaluate health technology innovations. The research group wants to explicitly thank Flanders’ Care (https://www.flanderscare.be) for its ongoing support during the planning and execution of this research as well as all members of the advisory board of this research project.
Funding
This study is carried out with funding from the Flemish Government, under grant no. EF47.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Vandemeulebroucke, T., Denier, Y., Mertens, E. et al. Which Framework to Use? A Systematic Review of Ethical Frameworks for the Screening or Evaluation of Health Technology Innovations. Sci Eng Ethics 28, 26 (2022). https://doi.org/10.1007/s11948-022-00377-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11948-022-00377-2