Abstract
Purpose of Review
This review examines recent (2016 onwards) neuroscientific findings on the mechanisms supporting mindfulness-associated pain relief. To date, its clear that mindfulness lowers pain by engaging brain processes that are distinct from placebo and vary across meditative training level. Due to rapid developments in the field of contemplative neuroscience, an update review on the neuroimaging studies focused on mindfulness, and pain is merited.
Recent Findings
Mindfulness-based therapies produce reliably reductions in a spectrum of chronic pain conditions through psychological, physiological, and neural mechanisms supporting the modulation of evaluation and appraisal of innocuous and noxious sensory events.
Summary
Neuroimaging and randomized control studies confirm that mindfulness meditation reliably reduces experimentally induced and clinical pain by engaging multiple, unique, non-opioidergic mechanisms that are distinct from placebo and which vary across meditative training level. These promising findings underscore the potential of mindfulness-based approaches to produce long-lasting improvements in pain-related symptomology.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Despite the wealth of empirical physiological, psychological, and neuroscientific evidence, the affective, cognitive, and sensory mechanisms that modulate pain remain difficult to disentangle, forming an obstacle to the effective treatment of a wide number of chronic pain conditions. Chronic pain affects over 116 million Americans [1] and 1.5 billion people worldwide [2], impacting women disproportionately [3], and with an annually increasing prevalence [4]. Many qualify chronic pain as having reached epidemic proportions [5].
Under the mistaken belief that the risk for developing opioid use disorder was low when opioids are taken for the purpose of analgesia [6], the rate of opioid prescriptions markedly increased over the past two decades [7]. In 2012 alone, 259 million opioid prescriptions were given in the USA [8]. The repeated use of opioids produces physiological adaptations that lead to tolerance and physical dependence [7], driving a downward spiral of maladaptive cognitive-affective sequelae [9] that have led to an increased rate of opioid misuse and opioid use disorder among chronic pain patients [10].
Shortcomings of Pharmacological Pain Therapies
The use of opioids as frontline analgesics has been further called into question by a recent wave of evidence demonstrating that opioid-based treatments provide little benefit and actually exacerbate chronic pain. A recent randomized clinical trial (RCT) examined 12 months of opioid vs. non-opioid treatment for chronic back, hip, or knee pain and found that individuals taking opioids reported no pain-related improvements when compared with non-opioid therapies [11]. In contrast, opioid-treated patients exhibited significantly higher pain intensity and medication-related adverse effects. A number of cohort-based studies and systematic reviews report similar results—small or no beneficial effects of opioids in treating polyneuropathy, chronic low back pain, musculoskeletal, and chronic non-cancer pain, alongside higher risks of all-cause mortality, opioid dependency, and overdose [12,13,14,15,16].
The shortcomings of other chronic pain treatments like spinal cord stimulation [17], intrathecal pumps [18], and nerve ablations [19, 20] to produce long-term pain relief have inspired a wave of cognitive therapies that integrate components of contemplative practices to strengthen self-regulation of affective processes and target the etiological factors of chronic pain, rather than the physical source of injury. Yet, the degree to which complex psychosocial factors underlie many of these conditions has made the efficacious treatment of chronic pain a slow and difficult approach. Enhancing self-regulation of arising sensory and affective experiences is a potential mechanism in promoting long-lasting reductions in pain and corresponding comorbidities [21]. To this end, self-regulatory practices such as mindfulness-based meditation reliably reduce affective pain when compared with sensory pain by targeting moment-to-moment appraisals of self and environment [22,23,24,25,26,27].
Recent advances on the unique modulatory mechanisms supporting mindfulness-based analgesia have begun to shed light on how the affective-motivational and sensory-discriminative components of pain interact. Previous reviews have outlined the neuroimaging-based evidence of the distinct mechanisms supporting brief and long-term mindfulness training-induced pain relief discovered up to 2016 [28,29,30,31]. The present review presents a short synopsis of such work and delineates recently discovered (> 2016) neuroscientific evidence that elucidates pain relief mechanisms and the manner in which interventions promote health across a broad range of disease points in greater detail. It is worth nothing that this review focuses on mindfulness meditation, one among thousands of distinct meditation techniques.
Brief Synopsis of Neurophysiology and Non-pharmacological Pain Modulation
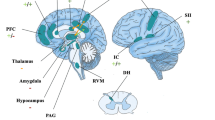
Pain is constructed and modulated through a myriad of interactions between peripheral nociceptive sensory afferents (A-delta and C-fibers), ascending nociceptive signals in the spinal cord, and a distributed network of subcortical and cortical brain areas. Nociceptive information originates in sensory primary afferents that synapse in the dorsal horn before ascending contralaterally, largely through the spinothalamic pathway, to a feedback network of lower level sensory regions, including the periaqueductal gray matter (PAG), thalamus, and primary somatosensory (SI) and secondary somatosensory (SII) cortices [32,33,34,35,36,37,38]. The anterior and posterior insular cortices then fine-tune the ascending nociceptive signal before higher order brain regions, including the dorsal anterior cingulate cortex (dACC) and prefrontal cortex (PFC), enrich it with contextual meaning [39,40,41,42].
Cognitive techniques that manipulate mood [43], expectations [40, 44], and attention [45] reduce pain via a common descending inhibitory pathway. To this extent, the endogenous opioidergic system mediates cognitively modulated pain relief engaged by placebo [46,47,48,49,50,51], conditioned pain modulation [52], acupuncture [53], hypnosis [54], and attentional control [55]. Specifically, the cognitive modulation of pain is associated with reductions of neural activity in pain-related brain regions and activation of the PAG to facilitate descending inhibition of pain at the level of the spinal cord [56,57,58,59]. In contrast, growing evidence demonstrates that mindfulness meditation reduces pain through mechanisms that may bypass PAG-mediated descending inhibition [23, 31].
Mindfulness and Mindfulness-Based Therapies
Mindfulness meditation originated 4500 years ago in Southeast Asia and is, in its modern instantiation, a loose construct whose precise definition remains a matter of debate. In the secular scientific context within which mindfulness-based therapies are studied, it refers to a self-regulated attentional state based on non-judgmental awareness of the present moment [60]. Mindfulness can be cultivated by a variety of meditation routines or can be practiced without formal training. Within the wide variety of mindfulness meditation techniques, two operationalized approaches stand out: focused attention [61] and choiceless awareness [62].
Focused attention involves narrowing attention to the dynamic moment-to-moment quality of a sensory, emotional, or cognitive event, such as the breath, and re-orienting attention from distractors so as to sustain attention on the chosen object [31, 61]. Choiceless awareness, on the other hand, involves the wide non-reactive monitoring of moment-by-moment sensory, cognitive, or affective states occurring in one’s experience. The focused attention practitioner tends to naturally transition into a choiceless awareness state in which the internal sensory and cognitive milieu are monitored without evaluation [31, 61]. As choiceless awareness practice further develops, awareness may reflect back upon itself, resulting in a profound reduction of self-referential processing during a state known as non-dual awareness that limits evaluation of object from the perspective of self [63]. The large number of operational variations even within each of these techniques, however, requires that scientific studies reference the precise technique being evaluated.
Mindfulness-based stress reduction (MBSR) [64] is one of the most promising mindfulness-based therapies to demonstrate clinical efficacy across a range of health outcomes. MBSR is an 8-week course that combines a range of sitting and moving meditation practices integrated with some behavioral therapy, daily “homework,” and a silent 2-day retreat. Other more-tailored mindfulness-based interventions evolved from MBSR to target different disease points: mindfulness-based cognitive therapy (MBCT) to treat depression [65,66,67,68], mindfulness-oriented recovery enhancement (MORE) to target opioid misuse and chronic pain [69], and mindfulness-based relapse prevention, a therapy that reliably alleviates addiction and long-term recovery [70].
Mindfulness-based therapies improve pain-related symptoms across a range of conditions including fibromyalgia, migraine, and irritable bowel syndrome [69, 71,72,73,74,75,76,77,78,79,80,81,82], with recent meta-analyses on chronic pain [83], rheumatoid arthritis [84], and cancer-related pain [85] confirming earlier findings. Of the different interventions, the 8-week MBSR program is the most studied. Multiple randomized control trials report post-MBSR improvements in pain-related symptoms and quality of life for patients with a spectrum of chronic pain conditions [86] including chronic low back pain [71, 87] and, in some one case, with sustained effects after 3 years [88]. Mindfulness meditation–based pain relief has also been studied in healthy, pain-free subjects via experimentally induced pain, with results showing that bouts of training briefer than 8 weeks are efficacious. One such study using noxious heat found that after a four-session (20 min/session) mindfulness meditation–based intervention, meditation resulted in a significant 40% reduction in pain intensity and 57% reduction in pain unpleasantness ratings [22].
Importantly, across studies, the most salient effects of mindfulness-based therapies are related to the significant improvements in affective factors that accompany and often complicate pain conditions, such as pain acceptance [89], stress [90, 91], depression [68, 92,93,94,95], and catastrophizing [96]. Across most to all studies of mindfulness and pain, mindfulness practice leads to significant improvements in affective pain when compared with sensory pain [22,23,24,25,26,27]. Mindfulness-based therapies thus appear particularly effective at targeting the affective component of pain and constitute a promising avenue toward better understanding and mitigating the psychosocial components of chronic pain. Until recently, however, the specific neural mechanisms responsible for mindfulness-based analgesia remained largely unknown.
The Neuroscience of Mindfulness-Based Pain Reduction up to 2016
The advent of neuroimaging techniques such as functional magnetic resonance imaging (fMRI) opened the doors for researchers to investigate the neural mechanisms active in mindfulness meditation–based pain relief and is associated with the surge in research on mindfulness [97]. Up to 2016, a number of studies identified multiple unique neuromodulatory mechanisms for both brief and long-term mental training.
In the context of brief mental training, two studies [22, 98] found mindfulness-based pain reductions in response to painful heat that were associated with activation in three cortical areas: the subgenual ACC (sgACC) (associated with cognitive and affective pain control), the orbitofrontal cortex (OFC) (supporting contextual evaluation of sensory events), and the right anterior insula (associated with afferent nociceptive signal modulation and interoceptive awareness). In addition, they found that effortful cognitive processes mediated by executive attention downregulate ascending nociceptive signals in the thalamus through a mechanistically unique cortico-thalamic-cortical pathway that may have also facilitated lower SI corresponding to the stimulation site. Taken together, these mechanisms may relieve pain by recontextualizing pain as innocuous sensory information rather than an affectively laden threat to bodily integrity—a type of “mindful” reappraisal of the meaning of nociceptive input. Researchers showed mindfulness-related pain relief to be mechanistically distinct from placebo-based pain relief by comparing experimentally induced pain during mindfulness meditation and sham mindfulness, an intervention identical to genuine mindfulness training except omitting mindfulness-based instructions (e.g., non-judgmental attention to the breath) [98]. Instead, participants are instructed to “take deep breaths” every 2–3 min “as we sit here in mindfulness meditation.” This novel pain modulatory pathway was found to be non-opioidergically mediated [23••] and thus distinct from those of placebo-based pain relief [31, 98].
On the other hand, long-term training elicits reductions in affective pain alongside greater activation in somatosensory regions and deactivation of appraisal-related regions like vmPFC [24, 25]. This decoupling between frontal, appraisal-related areas and sensory areas may reflect a distinct non-appraisal mechanism, reflecting the ability of expert meditators to separate the sensory experience of pain from the corresponding contextualization, thereby removing the need for OFC-mediated top-down inhibitory control [24, 25, 31, 99]. That is, experienced meditators have developed the capacity to sense noxious stimuli as frankly painful without the evaluation of unpleasantness/affect that traditionally accompanies painful experiences. Needless to say, there is great potential therapeutic value of both brief and long-term mindfulness practice to treat chronic pain patients for whom pharmacological analgesia has proven ineffective.
Recent Discoveries in the Neurophysiological Mechanisms Supporting Mindfulness-Based Pain Relief
Since 2016, evidence has continued to accumulate demonstrating that mindfulness-based pain relief is modulated through multiple unique neural mechanisms that are largely insensitive to opioidergic antagonism. As the role of pain’s affective dimension in chronic disease etiology has grown in importance, so too has the accumulation of evidence demonstrating that mindfulness uniquely impacts affective pain to promote well-being. These new findings remarkably parallel recent findings suggesting that mindfulness meditation engages a novel modulatory pathway to reduce pain.
Unique Neurophysiological Mechanisms Supporting Mindfulness Meditation-Induced Pain Relief: an Update from 2016
Recent mechanistically focused clinical trials examining the effects of brief mindfulness-based mental training provide converging evidence that meditation bypasses the body’s endogenous opioid system and operates via multiple unique neural pathways. The mechanisms supporting pain relief by mindfulness meditation are postulated to be disparate across training level and experience. Yet, recent evidence demonstrates that long-term meditation practitioners reliably reduce pain via non-opioidergic mechanisms. May and colleagues [100] recorded subjects’ pain ratings in response to painful electrical stimulation, before and during choiceless awareness meditation during intravenous administration of saline or naloxone (the opioid antagonist) in a double-blind, randomized, crossover design. Naloxone infusion not only failed to antagonize meditation-induced pain relief but actually enhanced pain relief—an unexpected result. It was suggested that the opioid blockage may have upregulated a non-opioid pathway.
In order to disentangle the specific role of other underlying components of mindfulness for pain relief, Wells and colleagues [101] conducted a double-blind, placebo-controlled crossover naloxone and saline infusion study. Healthy subjects were randomized into mindfulness meditation, sham mindfulness meditation, and slow-paced breathing groups and administered noxious 49 °C heat to the back of the calf. As expected, naloxone did not reverse pain relief associated with mindfulness meditation. Surprisingly, slow-paced breathing significantly reduced pain during opioidergic antagonism. Importantly, naloxone infusion reversed analgesia produced by sham mindfulness meditation, providing supplementary evidence that mindfulness is mechanistically distinct from placebo. Since all techniques significantly lowered breathing rates, the independent pain-relieving role of slow breathing from endogenous opioids could not be ruled out. However, the authors found that self-reported “attention to the breath” was absent only from sham mindfulness practice. The shifting of executive attention between breath sensations, appraising distractions, and reorienting back to breath was hypothesized to modulate pain before nociceptive information can be elaborated into a subjective sensory experience, a phenomenon corroborated by post-hoc qualitative assessments in which greater pain relief was associated with self-reported attention to the breath [101]. Thus, the authors postulated that attending to the breath, independently of mindfulness-based reappraisal, reduces pain by bypassing opioidergically mediated descending inhibition. Crucially, since slow-paced breathing is easier to perform than mindful attention to the breath, slow attentive breathing may be a critically valuable therapy for patients seeking a less cognitively demanding, non-opioidergic, self-administered pain therapy. Taken together, three separate studies now demonstrate that mindfulness reduces pain via non-opioidergic processes independent of meditation expertise.
Mindfulness meditation involves a number of components such as choiceless awareness, attention to the breath, body awareness, and relaxation, which might underlie its effects on pain. A number of recent studies have begun to elucidate which specific components drive these effects, showing, for example, that the relaxation component alone is not the driver behind mindfulness-based pain relief [102] but that attention to breathing is a potential mechanism of action [101]. Moreover, it has been shown that the autonomic nervous system plays a unique role in mindfulness-associated affective pain relief.
Greater Granularity on Unique Mechanisms Underlying Affective Pain Relief
There is an established relationship between increased activity in the parasympathetic autonomic nervous system and pain relief. However, until recently, it was unknown to what extent mindfulness meditation-induced pain relief is associated with parasympathetic activity. A study examined high-frequency heart rate variability (HF HRV), a robust marker of parasympathetic activation, during noxious heat and mindfulness and sham mindfulness meditation to determine the role of the parasympathetic system [103]. Higher HF HRV was more strongly associated with lower pain unpleasantness (though not with pain intensity) ratings during genuine mindfulness than during sham mindfulness. Further mechanistic research is needed to characterize the precise role of the autonomic nervous system in mindfulness-based pain relief, but these results point toward a unique parasympathetic activation pattern engaged by mindfulness to relieve pain.
The neuroscientific basis for affective pain relief associated with mindfulness meditation has benefited from randomized control studies combining different forms of experimentally induced pain and pain-eliciting stimuli. In one study, meditation-naïve participants received 30 min of mindfulness training before either maintaining a mindset of mindful acceptance or reacting naturally to a series of aversive vs. neutral images and individually calibrated noxious vs. innocuous heat stimuli [104]. After each trial, participants reported subjective pain and negative emotion levels. Mindful acceptance led to lower self-reported pain, less emotional reactivity to negative images as registered in the amygdala and PFC, and lower pain-related activity in the neurological pain signature (NPS)—a machine learning–based regression technique that can reliably predict self-reported pain [105]. These findings provide novel evidence that a single mindfulness training session produces significant pain relief and engages modulatory brain mechanisms that support the evaluation of negative affect.
Mindfulness meditation may also reduce social pain. In a study by Laneri et al. [106], long-term meditators and non-meditators were presented with images of people in neutral and in socially embarrassing situations after either a choiceless awareness mindfulness meditation or rest. All subjects exhibited greater ACC, anterior insula, and mPFC activation in response to embarrassing images, a typical pattern associated with the negative affect of experiencing another’s distressing mental state [107]. Compared with the subjects who did not meditate prior to the task, long-term meditation practitioners exhibited lower anterior insula activation during the presentation of embarrassing images, and these effects were associated with higher compassion scores. These data provide supplementary evidence that the anterior insula is strongly associated with cultivating interoceptive processes and compassion, an effect that may be developed after longer bouts of training. Previous studies had shown social pain from vicarious embarrassment to be significantly associated with anterior insula activation [107,108,109], while others had shown that mindfulness decreases reactivity to affective experiences, with associated lower anterior insula activation [110, 111]. Therefore, these results suggest that meditation may reduce affective distress not only in response to one’s own pain but also to someone else’s. Moreover, this attenuation of empathic distress—feeling of another’s pain—is in line with studies showing different brain activation patterns for empathic distress and empathic care [112], with the latter associated to compassion. Thus, converging lines of evidence suggest that mindfulness meditation may also attenuate social pain, an additional health benefit [113]. Remarkably, the degree of decreased anterior insular activation was contingent on the frequency of daily practice, adding to growing evidence that compassion cultivation is a trainable skill.
Growing Therapeutic Usefulness
The studies reviewed above, conducted in healthy, pain-free individuals, have elucidated the specific pain-relieving mechanisms that may now allow for the design of more effective mindfulness-based therapies to target specific chronic pain conditions.
The neural mechanisms supporting the direct alleviation of clinical pain have been brought into sharp focus by a seminal study by Seminowicz et al. [114••], which assessed the behavioral efficacy and neural mechanisms supporting enhanced MBSR (MBSR+) on episodic migraine, a severe and disabling neurological pain disorder. The 98-participant randomized clinical trial compared a 12-session 4-month-long MBSR+ (doubling the duration of the original MBSR) intervention to a time-matched stress management for headache (SMH) program. Both groups showed reductions in the number of headache days per month from baseline to 20 weeks, with 23% of the SMH group responding to treatment compared with 52% of the MBSR+ group, a response rate comparable with that of first-line pharmacologic treatment. Moreover, fMRI data showed the MBSR+ group had weaker resting-state connectivity between dorsal anterior insula and both superior parietal lobule and cuneus, an effect the authors interpreted as evidence of increased cognitive efficiency from mindfulness-based training. Promising therapeutic results like these highlight the potential of mindfulness-based pain relief to reshape the chronic pain experience without the need for lengthy or cognitively demanding interventions. In the final section, the potential of mindfulness-based interventions to reduce the risk of opioid misuse among opioid-treated chronic pain patients will be discussed.
Role of Mindfulness-Based Therapies for Opioid-Treated Chronic Pain Recovery
So far, mindfulness-based interventions have been discussed in light of their therapeutic potential to modulate pain. Important recent research also suggests that these interventions may also mitigate the risk factors causing opioid-treated chronic pain patients to develop opioid use disorder, a valuable role in light of the current opioid crisis.
Evidence suggests that prolonged opioid use may cause hedonic dysregulation, a sensitization to aversive experiences and concurrent reduction in responsiveness to natural rewards, which decreases positive affect and drives a downward spiral linking chronic pain to opioid misuse [9, 115]. In order to counteract this negative spiral and promote hedonic well-being, MORE, an intervention based on Garland et al.’s mindfulness-to-meaning theory [116], unites training in mindfulness, reappraisal, and savoring skills to simultaneously address chronic pain and addictive behavior. In two stage 2 RCTs, MORE was shown to decrease prescription opioid misuse and pain severity. In the first of these trials, in a sample of 115 chronic pain patients who had taken prescription opioids for more than 10 years, MORE reduced the occurrence of opioid misuse by 63% while decreasing opioid craving, pain severity, and pain interference [75]. The effects of MORE on pain were statistically mediated by the capacity to reappraise pain as innocuous sensory information, again suggesting that mindfulness training modifies contextual evaluations of nociceptive information. In a second RCT, MORE was evaluated as a prophylactic intervention for long-term opioid-treated chronic pain patients at risk of developing opioid misuse [117•]. Ninety-five patients were randomized to 8 weeks of MORE or to a support group intervention, after which MORE-treated patients reported significantly greater improvements in an array of positive psychological functions and reductions in pain severity and opioid misuse risk than support group patients at 3-month follow-up. The effects of MORE on reducing pain severity were statistically mediated by increases in positive psychological functioning and, most notably, by mindfulness-induced mental states and enhanced capacity to savor positive experiences. These clinical effects parallel recent EEG data from a series of randomized controlled experiments demonstrating that MORE increases neurophysiological responsivity to naturally rewarding, positive stimuli and decreases neurophysiological reactivity to opioid-related cues [118]. However, the underlying neural mechanisms supporting the effect of MORE on reducing pain and opioid misuse remain unknown. While more research is needed to confirm the precise neural mechanisms that modulate these processes, the ability of mindfulness meditation to not only target pain via multiple unique non-opioidergic modulatory pathways but to also mitigate the psychological risks of developing opioid use disease makes it an important candidate for further research.
Future Directions for Mindfulness-Based Pain Therapies
The neuroscience of mindfulness-based pain relief is in its infancy, and yet it has already proven capable of expanding the repertoire of known neural modulatory pain pathways and of shedding light on the complex affective and psychosocial components of chronic pain. The vast number of global chronic pain sufferers as well as the health and social risks of opioid use disorder underscores the potential value of developing validated self-administrable mind-body therapies that target pain multidimensionally via several unique neuromodulatory pathways. The great variability within mindfulness meditation techniques as well as the biopsychosocial complexity of chronic pain conditions, however, requires researchers to continue to apply the highest experimental standards to find and fine-tune the clinically relevant tools we need to produce long-lasting improvements in chronic pain management. By altering the meaning, interpretation, and appraisal of nociceptive information on its way to constructing the subjective experience of pain, mindfulness-based approaches may play an important role in the integrative therapeutic regimens capable of stemming the rising tide of chronic pain.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Simon LS. Relieving pain in America: a blueprint for transforming prevention, care, education, and research. J Pain Palliat Care Pharmacother. 2012;26:197–8.
Global pain management market to reach US$60 Billion by 2015, according to a new report by Global Industry Analysts, Inc. [Internet]. Glob. Ind. Anal. Inc. [cited 2020 Apr 8]. Available from: https://www.prweb.com/releases/2011/1/prweb8052240.htm
Tsang A, Von Korff M, Lee S, Alonso J, Karam E, Angermeyer MC, et al. Common chronic pain conditions in developed and developing countries: gender and age differences and comorbidity with depression-anxiety disorders. J Pain. 2008;9:883–91.
Zimmer Z, Zajacova A. Persistent, consistent, and extensive: the trend of increasing pain prevalence in older Americans. J Gerontol Ser B. 2020;75:436–47.
Domenichiello AF, Ramsden CE. The silent epidemic of chronic pain in older adults. Prog Neuro-Psychopharmacol Biol Psychiatry. 2019;93:284–90.
Porter J, Jick H. Addiction rare in patients treated with narcotics. N Engl J Med. 1980;302:123.
Volkow ND, Blanco C. The changing opioid crisis: development, challenges and opportunities. Mol Psychiatry. 2020:1–16.
Rothstein MA. The opioid crisis and the need for compassion in pain management. Am J Public Health. 2017;107:1253–4.
Garland EL, Froeliger B, Zeidan F, Partin K, Howard MO. The downward spiral of chronic pain, prescription opioid misuse, and addiction: cognitive, affective, and neuropsychopharmacologic pathways. Neurosci Biobehav Rev. 2013;37:2597–607.
Vowles KE, McEntee ML, Julnes PS, Frohe T, Ney JP, van der Goes DN. Rates of opioid misuse, abuse, and addiction in chronic pain: a systematic review and data synthesis. Pain. 2015;156:569–76.
Krebs EE, Gravely A, Nugent S, Jensen AC, DeRonne B, Goldsmith ES, et al. Effect of opioid vs nonopioid medications on pain-related function in patients with chronic back pain or hip or knee osteoarthritis pain: the SPACE randomized clinical trial. JAMA. 2018;319:872–82.
Hoffman EM, Watson JC, St Sauver J, Staff NP, Klein CJ. Association of long-term opioid therapy with functional status, adverse outcomes, and mortality among patients with polyneuropathy. JAMA Neurol. 2017;74:773–9.
Ray WA, Chung CP, Murray KT, Hall K, Stein CM. Prescription of long-acting opioids and mortality in patients with chronic noncancer pain. JAMA. 2016;315:2415–23.
Abdel Shaheed C, Maher CG, Williams KA, Day R, McLachlan AJ. Efficacy, tolerability, and dose-dependent effects of opioid analgesics for low back pain: a systematic review and meta-analysis. JAMA Intern Med. 2016;176:958–68.
Chou R, Deyo R, Friedly J, Skelly A, Weimer M, Fu R, et al. Systemic pharmacologic therapies for low back pain: a systematic review for an American College of Physicians Clinical Practice Guideline. Ann Intern Med. 2017;166:480–92.
Megale RZ, Deveza LA, Blyth FM, Naganathan V, Ferreira PH, McLachlan AJ, et al. Efficacy and safety of oral and transdermal opioid analgesics for musculoskeletal pain in older adults: a systematic review of randomized, placebo-controlled trials. J Pain. 2018;19:475.e1–475.e24.
Duarte RV, Nevitt S, McNicol E, Taylor RS, Buchser E, North RB, et al. Systematic review and meta-analysis of placebo/sham controlled randomised trials of spinal cord stimulation for neuropathic pain. PAIN. 2020;161:24–35.
Deer TR, Pope JE, Hanes MC, McDowell GC. Intrathecal therapy for chronic pain: a review of morphine and ziconotide as firstline options. Pain Med Oxford Acad. 2019;20:784–98.
Daniels J, Gray R, Hills RK, Latthe P, Buckley L, Gupta J, et al. Laparoscopic uterosacral nerve ablation for alleviating chronic pelvic pain: a randomized controlled trial. JAMA Am Med Assoc. 2009;302:955–61.
McCormick ZL, Korn M, Reddy R, Marcolina A, Dayanim D, Mattie R, et al. Cooled radiofrequency ablation of the genicular nerves for chronic pain due to knee osteoarthritis: six-month outcomes. Pain Med Oxford Acad. 2017;18:1631–41.
Price DD, Harkins SW, Baker C. Sensory-affective relationships among different types of clinical and experimental pain. Pain. 1987;28:297–307.
Zeidan F, Martucci KT, Kraft RA, Gordon NS, McHaffie JG, Coghill RC. Brain mechanisms supporting the modulation of pain by mindfulness meditation. J Neurosci. 2011;31:5540–8.
•• Zeidan F, Adler-Neal AL, Wells RE, Stagnaro E, May LM, Eisenach JC, et al. Mindfulness-meditation-based pain relief is not mediated by endogenous opioids. J Neurosci Soc Neurosci. 2016;36:3391–7 Finds that mindfulness meditation-based pain relief operates via non-opioidergic mechanisms and is thus (a) mechanistically separate from other cognitive pain inhibitory mechanisms, and (b) a good candidate for chronic pain treatment given the lack of cross-tolerance with opioid-based medications.
Grant JA, Courtemanche J, Rainville P. A non-elaborative mental stance and decoupling of executive and pain-related cortices predicts low pain sensitivity in Zen meditators. Pain. 2011;152:150–6.
Gard T, Hölzel BK, Sack AT, Hempel H, Lazar SW, Vaitl D, et al. Pain attenuation through mindfulness is associated with decreased cognitive control and increased sensory processing in the brain. Cereb Cortex N Y N 1991. 2012;22:2692–702.
Brown CA, Jones AKP. Meditation experience predicts less negative appraisal of pain: electrophysiological evidence for the involvement of anticipatory neural responses. Pain. 2010;150:428–38.
Lutz A, McFarlin DR, Perlman DM, Salomons TV, Davidson RJ. Altered anterior insula activation during anticipation and experience of painful stimuli in expert meditators. NeuroImage. 2013;64:538–46.
Adler-Neal AL, Zeidan F. Mindfulness meditation for fibromyalgia: mechanistic and clinical considerations. Curr Rheumatol Rep. 2017;19:59.
Zeidan F, Grant JA, Brown CA, McHaffie JG, Coghill RC. Mindfulness meditation-related pain relief: evidence for unique brain mechanisms in the regulation of pain. Neurosci Lett. 2012;520:165–73.
Zeidan F, Baumgartner JN, Coghill RC. The neural mechanisms of mindfulness-based pain relief: a functional magnetic resonance imaging-based review and primer. PAIN Rep. 2019;4:e759.
Zeidan F, Vago D. Mindfulness meditation–based pain relief: a mechanistic account. Ann N Y Acad Sci. 2016;1373:114–27.
Wiech K, Ploner M, Tracey I. Neurocognitive aspects of pain perception. Trends Cogn Sci. 2008;12:306–13.
Tracey I, Mantyh PW. The cerebral signature for pain perception and its modulation. Neuron. 2007;55:377–91.
Derbyshire SW, Jones AK, Gyulai F, Clark S, Townsend D, Firestone LL. Pain processing during three levels of noxious stimulation produces differential patterns of central activity. Pain. 1997;73:431–45.
Basbaum AI, Fields HL. Endogenous pain control systems: brainstem spinal pathways and endorphin circuitry. Annu Rev Neurosci. 1984;7:309–38.
Coghill RC, Talbot JD, Evans AC, Meyer E, Gjedde A, Bushnell MC, et al. Distributed processing of pain and vibration by the human brain. J Neurosci. 1994;14:4095–108.
Coghill RC, Sang CN, Maisog JM, Iadarola MJ. Pain intensity processing within the human brain: a bilateral, distributed mechanism. J Neurophysiol. 1999;82:1934–43.
Coghill RC, McHaffie JG, Yen Y-F. Neural correlates of interindividual differences in the subjective experience of pain. Proc Natl Acad Sci U S A. 2003;100:8538–42.
Lobanov OV, Quevedo AS, Hadsel MS, Kraft RA, Coghill RC. Frontoparietal mechanisms supporting attention to location and intensity of painful stimuli. Pain. 2013;154:1758–68.
Lobanov OV, Zeidan F, McHaffie JG, Kraft RA, Coghill RC. From cue to meaning: brain mechanisms supporting the construction of expectations of pain. Pain. 2014;155:129–36.
Oshiro Y, Quevedo AS, McHaffie JG, Kraft RA, Coghill RC. Brain mechanisms supporting spatial discrimination of pain. J Neurosci. 2007;27:3388–94.
Oshiro Y, Quevedo AS, McHaffie JG, Kraft RA, Coghill RC. Brain mechanisms supporting discrimination of sensory features of pain: a new model. J Neurosci. 2009;29:14924–31.
Villemure C, Bushnell MC. Cognitive modulation of pain: how do attention and emotion influence pain processing? Pain. 2002;95:195–9.
Koyama T, McHaffie JG, Laurienti PJ, Coghill RC. The subjective experience of pain: where expectations become reality. Proc Natl Acad Sci U S A. 2005;102:12950–5.
Seminowicz DA, Mikulis DJ, Davis KD. Cognitive modulation of pain-related brain responses depends on behavioral strategy. Pain. 2004;112:48–58.
Amanzio M, Benedetti F. Neuropharmacological dissection of placebo analgesia: expectation-activated opioid systems versus conditioning-activated specific subsystems. J Neurosci. 1999;19:484–94.
Eippert F, Bingel U, Schoell ED, Yacubian J, Klinger R, Lorenz J, et al. Activation of the opioidergic descending pain control system underlies placebo analgesia. Neuron. 2009;63:533–43.
Grevert P, Albert LH, Goldstein A. Partial antagonism of placebo analgesia by naloxone. Pain. 1983;16:129–43.
Levine JD, Gordon NC, Fields HL. The mechanism of placebo analgesia. Lancet Lond Engl. 1978;2:654–7.
Wager TD, Scott DJ, Zubieta J-K. Placebo effects on human mu-opioid activity during pain. Proc Natl Acad Sci U S A. 2007;104:11056–61.
Zubieta J-K, Bueller JA, Jackson LR, Scott DJ, Xu Y, Koeppe RA, et al. Placebo effects mediated by endogenous opioid activity on mu-opioid receptors. J Neurosci. 2005;25:7754–62.
King CD, Goodin B, Kindler LL, Caudle RM, Edwards RR, Gravenstein N, et al. Reduction of conditioned pain modulation in humans by naltrexone: an exploratory study of the effects of pain catastrophizing. J Behav Med. 2013;36:315–27.
Harris RE, Zubieta J-K, Scott DJ, Napadow V, Gracely RH, Clauw DJ. Traditional Chinese acupuncture and placebo (sham) acupuncture are differentiated by their effects on mu-opioid receptors (MORs). NeuroImage. 2009;47:1077–85.
Stephenson JB. Reversal of hypnosis-induced analgesia by naloxone. Lancet Lond Engl. 1978;2:991–2.
Sprenger C, Eippert F, Finsterbusch J, Bingel U, Rose M, Büchel C. Attention modulates spinal cord responses to pain. Curr Biol CB. 2012;22:1019–22.
Beitz AJ. The organization of afferent projections to the midbrain periaqueductal gray of the rat. Neuroscience. 1982;7:133–59.
Mantyh PW. Connections of midbrain periaqueductal gray in the monkey. I. Ascending efferent projections. J Neurophysiol. 1983;49:567–81.
Mantyh PW. Connections of midbrain periaqueductal gray in the monkey. II. Descending efferent projections. J Neurophysiol. 1983;49:582–94.
Liebeskind JC, Guilbaud G, Besson JM, Oliveras JL. Analgesia from electrical stimulation of the periaqueductal gray matter in the cat: behavioral observations and inhibitory effects on spinal cord interneurons. Brain Res. 1973;50:441–6.
Kabat-Zinn J, Hanh TN. Full catastrophe living: using the wisdom of your body and mind to face stress, pain, and illness: Random House Publishing Group; 2009.
Lutz A, Slagter HA, Dunne JD, Davidson RJ. Attention regulation and monitoring in meditation. Trends Cogn Sci. 2008;12:163–9.
Garrison KA, Zeffiro TA, Scheinost D, Constable RT, Brewer JA. Meditation leads to reduced default mode network activity beyond an active task. Cogn Affect Behav Neurosci. 2015;15:712–20.
Josipovic Z. Neural correlates of nondual awareness in meditation. Ann N Y Acad Sci. 2014;1307:9–18.
Kabat-Zinn J. Some reflections on the origins of MBSR, skillful means, and the trouble with maps. Contemp Buddhism Routledge. 2011;12:281–306.
Kuyken W, Byford S, Taylor RS, Watkins E, Holden E, White K, et al. Mindfulness-based cognitive therapy to prevent relapse in recurrent depression. J Consult Clin Psychol. 2008;76:966–78.
Segal ZV, Williams JMG, Teasdale JD, Kabat-Zinn J. Mindfulness-based cognitive therapy for depression. New York: The Guilford Press; 2012.
Segal ZV, Bieling P, Young T, MacQueen G, Cooke R, Martin L, et al. Antidepressant monotherapy vs sequential pharmacotherapy and mindfulness-based cognitive therapy, or placebo, for relapse prophylaxis in recurrent depression. Arch Gen Psychiatry. 2010;67:1256–64.
Segal ZV, Dimidjian S, Beck A, Boggs JM, Vanderkruik R, Metcalf CA, et al. 2020 Outcomes of online mindfulness-based cognitive therapy for patients with residual depressive symptoms: a randomized clinical trial. JAMA Psychiatry [Internet]. [cited 2020 Apr 20]; Available from: https://jamanetwork.com/journals/jamapsychiatry/fullarticle/2759418
Garland EL, Howard MO. Mindfulness-oriented recovery enhancement reduces pain attentional bias in chronic pain patients. Psychother Psychosom. 2013;82:311–8.
Bowen S, Witkiewitz K, Clifasefi SL, Grow J, Chawla N, Hsu SH, et al. Relative efficacy of mindfulness-based relapse prevention, standard relapse prevention, and treatment as usual for substance use disorders: a randomized clinical trial. JAMA Psych Am Med Assoc. 2014;71:547–56.
Morone NE, Greco CM, Moore CG, Rollman BL, Lane B, Morrow LA, et al. A mind-body program for older adults with chronic low Back pain: a randomized clinical trial. JAMA Intern Med. 2016;176:329–37.
Cho S, Heiby EM, McCracken LM, Lee S-M, Moon D-E. Pain-related anxiety as a mediator of the effects of mindfulness on physical and psychosocial functioning in chronic pain patients in Korea. J Pain Off J Am Pain Soc. 2010;11:789–97.
Fox SD, Flynn E, Allen RH. Mindfulness meditation for women with chronic pelvic pain: a pilot study. J Reprod Med. 2011;56:158–62.
Ussher M, Spatz A, Copland C, Nicolaou A, Cargill A, Amini-Tabrizi N, et al. Immediate effects of a brief mindfulness-based body scan on patients with chronic pain. J Behav Med. 2014;37:127–34.
Garland EL, Manusov EG, Froeliger B, Kelly A, Williams JM, Howard MO. Mindfulness-oriented recovery enhancement for chronic pain and prescription opioid misuse: results from an early-stage randomized controlled trial. J Consult Clin Psychol. 2014;82:448–59.
de Boer MJ, Steinhagen HE, Versteegen GJ, Struys MMRF, Sanderman R. Mindfulness, acceptance and catastrophizing in chronic pain. PLoS One. 2014;9:e87445.
Morone NE, Rollman BL, Moore CG, Li Q, Weiner DK. A mind-body program for older adults with chronic low back pain: results of a pilot study. Pain Med Malden Mass. 2009;10:1395–407.
Morone NE, Lynch CS, Greco CM, Tindle HA, Weiner DK. “I Felt Like a New Person.” The Effects of Mindfulness Meditation on Older Adults With Chronic Pain: Qualitative Narrative Analysis of Diary Entries. J Pain Off J Am Pain Soc. 2008;9:841–8.
McCracken LM, Keogh E. Acceptance, mindfulness, and values-based action may counteract fear and avoidance of emotions in chronic pain: an analysis of anxiety sensitivity. J Pain Off J Am Pain Soc. 2009;10:408–15.
McCracken LM, Velleman SC. Psychological flexibility in adults with chronic pain: a study of acceptance, mindfulness, and values-based action in primary care. Pain. 2010;148:141–7.
McCracken LM, Gauntlett-Gilbert J, Vowles KE. The role of mindfulness in a contextual cognitive-behavioral analysis of chronic pain-related suffering and disability. Pain. 2007;131:63–9.
Rosenzweig S, Greeson JM, Reibel DK, Green JS, Jasser SA, Beasley D. Mindfulness-based stress reduction for chronic pain conditions: variation in treatment outcomes and role of home meditation practice. J Psychosom Res. 2010;68:29–36.
Hilton L, Hempel S, Ewing BA, Apaydin E, Xenakis L, Newberry S, et al. Mindfulness meditation for chronic pain: systematic review and meta-analysis. Ann Behav Med Oxford Acad. 2017;51:199–213.
DiRenzo D, Crespo-Bosque M, Gould N, Finan P, Nanavati J, Bingham CO. Systematic review and meta-analysis: mindfulness-based interventions for rheumatoid arthritis. Curr Rheumatol Rep. 2018;20:75.
Johannsen M, O’Connor M, O’Toole MS, Jensen AB, Zachariae R. Mindfulness-based cognitive therapy and persistent pain in women treated for primary breast cancer [Internet]. Wolters Kluwer; 2018 [cited 2020 Apr 3]. Available from: https://www.ingentaconnect.com/content/wk/cjpn/2018/00000034/00000001/art00009
Kabat-Zinn J. An outpatient program in behavioral medicine for chronic pain patients based on the practice of mindfulness meditation: theoretical considerations and preliminary results. Gen Hosp Psychiatry. 1982;4:33–47.
Cherkin DC, Sherman KJ, Balderson BH, Cook AJ, Anderson ML, Hawkes RJ, et al. Effect of mindfulness-based stress reduction vs cognitive behavioral therapy or usual care on back pain and functional limitations in adults with chronic low back pain: a randomized clinical trial. JAMA. 2016;315:1240–9.
Kabat-Zinn J, Lipworth L, Burney R. The clinical use of mindfulness meditation for the self-regulation of chronic pain. J Behav Med. 1985;8:163–90.
Ruskin DA, Gagnon MM, Kohut SA, Stinson JN, Walker KS. A mindfulness program adapted for adolescents with chronic pain [Internet]. Wolters Kluwer; 2017 [cited 2020 Apr 3]. Available from: https://www.ingentaconnect.com/content/wk/cjpn/2017/00000033/00000011/art00009
Taren AA, Gianaros PJ, Greco CM, Lindsay EK, Fairgrieve A, Brown KW, et al. Mindfulness meditation training alters stress-related amygdala resting state functional connectivity: a randomized controlled trial. Soc Cogn Affect Neurosci. 2015;10:1758–68.
Creswell JD, Taren AA, Lindsay EK, Greco CM, Gianaros PJ, Fairgrieve A, et al. Alterations in resting-state functional connectivity link mindfulness meditation with reduced interleukin-6: a randomized controlled trial. Biol Psychiatry. 2016;80:53–61.
de Jong M, Peeters F, Gard T, Ashih H, Doorley J, Walker R, et al. A randomized controlled pilot study on mindfulness-based cognitive therapy for unipolar depression in patients with chronic pain. J Clin Psychiatry [Internet]. 2018 [cited 2020 Apr 3];79. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6020018/
Eisendrath S, Chartier M, McLane M. Adapting mindfulness-based cognitive therapy for treatment-resistant depression: a clinical case study. Cogn Behav Pract. 2011;18:362–70.
Eisendrath SJ, Gillung EP, Delucchi KL, Chartier M, Mathalon DH, Sullivan JC, et al. Mindfulness-based cognitive therapy (MBCT) versus the health-enhancement program (HEP) for adults with treatment-resistant depression: a randomized control trial study protocol. BMC Complement Altern Med. 2014;14:95.
Williams JMG, Crane C, Barnhofer T, Brennan K, Duggan DS, Fennell MJV, et al. Mindfulness-based cognitive therapy for preventing relapse in recurrent depression: a randomized dismantling trial. J Consult Clin Psychol. 2014;82:275–86.
Turner JA, Anderson ML, Balderson BH, Cook AJ, Sherman KJ, Cherkin DC. Mindfulness-based stress reduction and cognitive-behavioral therapy for chronic low back pain: similar effects on mindfulness, catastrophizing, self-efficacy, and acceptance in a randomized controlled trial. Pain. 2016;157:2434–44.
Tang Y-Y, Hölzel BK, Posner MI. The neuroscience of mindfulness meditation. Nat Rev Neurosci Nature Publish Group. 2015;16:213–25.
Zeidan F, Emerson NM, Farris SR, Ray JN, Jung Y, McHaffie JG, et al. Mindfulness meditation-based pain relief employs different neural mechanisms than placebo and sham mindfulness meditation-induced analgesia. J Neurosci. 2015;35:15307–25.
Perlman DM, Salomons TV, Davidson RJ, Lutz A. Differential effects on pain intensity and unpleasantness of two meditation practices. Emot Wash DC. 2010;10:65–71.
May LM, Kosek P, Zeidan F, Berkman ET. Enhancement of meditation analgesia by opioid antagonist in experienced meditators. Psychosom Med. 2018;80:807–13.
Wells RE, Collier J, Posey G, Morgan F, Auman T, Strittameter B, et al. Attention to breath sensations does not engage endogenous opioids to reduce pain. PAIN [Internet]. 2020 [cited 2020 Apr 4];Articles in Press. Available from: https://journals.lww.com/pain/Abstract/publishahead/Attention_to_breath_sensations_does_not_engage.98447.aspx
Smith KE, Norman GJ. Brief relaxation training is not sufficient to alter tolerance to experimental pain in novices. PLoS ONE [Internet]. 2017 [cited 2020 Apr 9];12. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5426711/
Adler-Neal AL, Waugh CE, Garland EL, Shaltout HA, Diz DI, Zeidan F. The role of heart rate variability in mindfulness-based pain relief. J Pain [Internet]. 2019 [cited 2020 Apr 4]; Available from: http://www.sciencedirect.com/science/article/pii/S1526590019307734
Kober H, Buhle J, Weber J, Ochsner KN, Wager TD. Let it be: mindful acceptance down-regulates pain and negative emotion. Soc Cogn Affect Neurosci Oxford Acad. 2019;14:1147–58.
Wager TD, Atlas LY, Lindquist MA, Roy M, Woo C-W, Kross E. An fMRI-based neurologic signature of physical pain. N Engl J Med. 2013;368:1388–97.
Laneri D, Krach S, Paulus FM, Kanske P, Schuster V, Sommer J, et al. Mindfulness meditation regulates anterior insula activity during empathy for social pain. Hum Brain Mapp. 2017;38:4034–46.
Paulus FM, Müller-Pinzler L, Jansen A, Gazzola V, Krach S. Mentalizing and the role of the posterior superior temporal sulcus in sharing others’ embarrassment. Cereb Cortex N Y N 1991. 2015;25:2065–75.
Fan Y, Duncan NW, de Greck M, Northoff G. Is there a core neural network in empathy? An fMRI based quantitative meta-analysis. Neurosci Biobehav Rev. 2011;35:903–11.
Lamm C, Decety J, Singer T. Meta-analytic evidence for common and distinct neural networks associated with directly experienced pain and empathy for pain. NeuroImage. 2011;54:2492–502.
Haase L, Thom NJ, Shukla A, Davenport PW, Simmons AN, Stanley EA, et al. Mindfulness-based training attenuates insula response to an aversive interoceptive challenge. Soc Cogn Affect Neurosci. 2016;11:182–90.
Paul NA, Stanton SJ, Greeson JM, Smoski MJ, Wang L. Psychological and neural mechanisms of trait mindfulness in reducing depression vulnerability. Soc Cogn Affect Neurosci Oxford Acad. 2013;8:56–64.
Ashar YK, Andrews-Hanna JR, Dimidjian S, Wager TD. Empathic care and distress: predictive brain markers and dissociable brain systems. Neuron. 2017;94:1263–1273.e4.
Edwards RR, Dworkin RH, Sullivan MD, Turk DC, Wasan AD. The role of psychosocial processes in the development and maintenance of chronic pain. J Pain. 2016;17:T70–92.
•• Seminowicz DA, Burrowes SAB, Kearson A, Zhang J, Krimmel SR, Samawi L, et al. Enhanced mindfulness-based stress reduction in episodic migraine: a randomized clinical trial with magnetic resonance imaging outcomes. PAIN. 2020;Articles in Press. Available from: https://journals.lww.com/pain/Abstract/publishahead/Enhanced_mindfulness_based_stress_reduction_in.98445.aspx. Finds that Enhanced Mindfulness-Based Stress Relief reduces episodic migraine significantly more than a stress management for headache (SMH) program at a therapeutic response rate comparable to that of first-line pharmacologic treatment. fMRI data suggests increased cognitive efficiency from mindfulness-based training.
Elman I, Borsook D. Common brain mechanisms of chronic pain and addiction. Neuron. 2016;89:11–36.
Garland EL, Farb NA, Goldin PR, Fredrickson BL. The mindfulness-to-meaning theory: extensions, applications, and challenges at the attention–appraisal–emotion Interface. Psychol Inq Routledge. 2015;26:377–87.
• Garland EL, Hanley AW, Riquino MR, Reese SE, Baker AK, Salas K, et al. Mindfulness-oriented recovery enhancement reduces opioid misuse risk via analgesic and positive psychological mechanisms: a randomized controlled trial. J Consult Clin Psychol Am Psycholog Assoc. 2019;87:927–40 Finds that Mindfulness-Oriented Recovery Enhancement (MORE) is effective as a prophylactic to prevent long-term opioid-treated chronic pain patients from developing opioid misuse disorder while reducing pain severity and significantly improving an array of positive psychological functions.
Garland EL, Atchley RM, Hanley AW, Zubieta J-K, Froeliger B. Mindfulness-oriented recovery enhancement remediates hedonic dysregulation in opioid users: neural and affective evidence of target engagement. Sci Adv. 2019;5:eaax1569.
Funding
This work was supported by the NIH’s National Center for Complementary and Integrative Health (NCCIH) (K99/R00-AT008238; R21-AT007247; R01-AT009693; R21-AT010352, FZ).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Psychological and Behavioral Aspects of Headache with Pain
Rights and permissions
About this article
Cite this article
Jinich-Diamant, A., Garland, E., Baumgartner, J. et al. Neurophysiological Mechanisms Supporting Mindfulness Meditation–Based Pain Relief: an Updated Review. Curr Pain Headache Rep 24, 56 (2020). https://doi.org/10.1007/s11916-020-00890-8
Published:
DOI: https://doi.org/10.1007/s11916-020-00890-8