Abstract
Purpose of Review
The failure of bony union following a fracture, termed a fracture nonunion, has severe patient morbidity and economic consequences. This review describes current consensuses and future directions of investigation for determining why, detecting when, and effective treatment if this complication occurs.
Recent Findings
Current nonunion investigation is emphasizing an expanded understanding of the biology of healing. This has led to assessments of the immune environment, multiple cytokines and morphogenetic factors, and the role of skeletogenic stem cells in the development of nonunion. Detecting biological markers and other objective diagnostic criteria is also a current objective of nonunion research. Treatment approaches in the near future will likely be dominated by the development of specific adjunct therapies to the nonunion surgical management, which will be informed by an expanded mechanistic understanding of nonunion biology.
Summary
Current consensus among orthopedists is that improved diagnosis and treatment of nonunion hinges first on discoveries at the bench side with later translation to the clinic.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
When a fracture fails to heal, the patient, healthcare system, and economy all suffer. Fracture nonunion is associated with significant morbidity and pain for the patient as well as additional costs averaging close to $12,000 per complication in the USA, largely due to the loss of productivity that accompanies the lengthy healing course [1, 2]. Traditionally, fracture nonunion was believed to occur in 5–10% of all fractures; however, two large, recent analyses in the USA and Scotland estimate the rate more in the 2–5% range [3, 4]. The incidence of nonunion varies significantly between bones. Zura et al. analyzed 309,330 fractures at various sites and reported the highest rates of nonunion observed were in the scaphoid (15.5%), tibia and fibula (14%), and femur (13.9%). The metacarpal and radius demonstrated the lowest rates of nonunion at 1.5% and 2.1%, respectively [4].
The FDA currently defines fracture nonunion as a fracture that has failed to heal within 9 months from injury with 3 consecutive months of healing stagnation [5]. Recent evidence, however, supports shortening the failure to heal period to 6 months for a more rapid diagnosis [6]. Many clinicians disagree on when a fracture is determined a nonunion, and, therefore, the exact time point for which to distinguish delayed and arrested healing [7]. Identifying an objective way to define the transition point remains a topic of ongoing research such that nonunion can be diagnosed and treated as early as possible.
Types of Nonunion
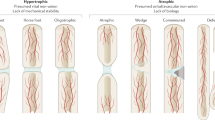
Fracture nonunions are primarily categorized as hypertrophic or atrophic based on biological viability at the fracture site determined by radiographic analysis. Hypertrophic nonunions are described as those in which biological potential has been maintained and, therefore, callus is observable radiographically. These fractures are believed to fail in the healing process because of mechanical failure at the fracture site from such stressors as inadequate stability or premature weight-bearing. Radiographically, hypertrophic nonunions can be further subdivided into elephant’s foot, horse’s hoof, or oligotrophic based on the callus pattern. Conversely, atrophic nonunions are believed to be void of biological potential as determined by a lack of callus development. In these cases, mechanical stability is not believed to be the chief causative factor [8•]. Figure 1 depicts three separate tibial fractures of which one has healed without complication, one has gone on to hypertrophic nonunion, and the last has gone on to atrophic nonunion.
Three different healing outcomes following fracture of the tibia. All images are lateral radiographs. The left image displays a distal 1/3rd tibia fracture with successful healing. The center image displays a tibial shaft fracture in which both evidence of callus as well as mechanical instability are noted. This indicates a hypertrophic nonunion. Lastly, the right image displays a distal tibial plafond fracture in which no callus is evident, indicating biologically inert boney ends and therefore atrophic nonunion
The presence of vascularity at the fracture site has traditionally been used as a marker of biologic capacity and, thus, a factor distinguishing hypertrophic from atrophic nonunions. Recent experimental evidence contradicts this, however, suggesting that adequate vascularity can persist in a believed atrophic environment [8•, 9]. Interestingly, Panteli et al. have also demonstrated that biological activity at the cellular level persists in human samples taken from nonunions that had been classified as atrophic based on classical schema [10]. These results call for revision of traditional hypertrophic vs. atrophic designation schema, or implementation of a modernized non-binary classification system such as the Non-Union Scoring System (NUSS) proposed and validated by Calori et al. [11,12,13]. These results also point to the deficiency of biological determinants for the progression of bone healing and primary reliance on radiological assessments.
Nonunion Etiology and Associated Factors
A Closer Look at the Fracture Site
The pathogenesis of fracture nonunion remains heavily researched but largely unexplained. Current insight points to either mechanical failure or disruption of the “bone-healing unit” as the chief causative factors [8•]. The concept of mechanical failure hinges on long-standing theories of bone tissue response to mechanical forces [8•]. In these cases, nonunions are preceded by a fracture site that is under either too much or too little, strain precipitated by such factors as inadequate implant use or application, insufficient anatomical reduction, large cortical defects, periosteal stripping, and erroneous load bearing [8•, 14,15,16,17,18,19,20,21]. Mechanical failure contributes to nonunion in the majority of cases [8•, 11].
Disruption of the bone-healing unit is believed to involve interruption of critical molecular and cellular regulators of fracture repair. At the molecular level, interruption of the signaling pathway of three specific classes of cytokines is believed to impact fracture healing [22, 23]. The first class is proinflammatory cytokines, which includes tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6). TNF-α has been shown in animal studies to play a role in cartilage resorption during remodeling and to regulate mesenchymal stem cell (MSC) differentiation in a concentration-dependent fashion [14, 24, 25]. IL-6 in the early posttraumatic period was recently determined to regulate a balanced immune response and subsequent bone repair in a mouse model, and has demonstrated a positive effect on callus strength and mineralization in earlier studies [26, 27]. However, contradictory results, including higher IL-6 and lower IL-6 receptor serum levels in nonunion patients, convey much is still to be known about the relationship between IL-6 signaling and nonunion [28, 29].
The second class of notable bone-healing cytokines is the transforming growth factor-β (TGF-β) superfamily that includes bone morphogenic proteins (BMPs). Although the molecular mechanisms remain unknown, BMPs have a well-established role as regulators of all four phases of fracture healing as chemotactic regulators of MSC differentiation and angiogenesis [14, 30, 31]. BMP-2 and BMP-7 specifically have garnered the most research attention since they are currently on-market as biologic adjuvant therapies. Their therapeutic efficacy will be discussed in a later section of this review.
The third and final notable class of bone-healing cytokines are the metalloproteinases and angiogenic factors. These cytokines coordinate the congruous matrix degradation and angiogenesis processes of fracture healing, regulated by matrix metalloproteinase (MMP) and vascular endothelial growth factor (VEGF), respectively [23]. MMP-13 and MMP-9 in particular have been shown to be critical regulators of cartilage breakdown and may even be detectable in urine as a non-invasive marker of nonunion [32, 33]. The role of VEGF in nonunion is more nuanced; it is suspected that although VEGF is important for angiogenesis at the fracture site and for union, hypervascularization in the initial healing period can be harmful [32, 34]. Continuing investigation is required to better elucidate the mechanisms by which all discussed cytokines affect healing in vivo, as well as their exact timing and at what concentration they act.
At a cellular level, the availability and adequate differentiation capacity of osteogenic cells, specifically skeletogenic stem cells (also known as MSCs), is a key component of the bone-healing unit [8•, 14]. MSCs are provided to the fracture site from a combination of periosteal, endosteal, bone marrow, and soft tissue origins and are critical progenitors for all but one cell type (osteoclasts) involved in the fracture repair process [14, 35, 36]. The significant soft tissue injury and periosteal stripping that can occur in high-energy fractures or from excessive surgical dissection can disrupt the availability of these MSCs and contribute to an inadequate supply of repair cells. These conditions can also disrupt vascular supply to the fracture site with detrimental consequences for healing [14, 37]. When applicable, the use of minimally invasive implants and implanting techniques may mitigate these disruptions and preserve the bone-healing unit.
Another factor that can disrupt the bone-healing unit is the presence of infection. Infection has demonstrated a significant association with nonunion since it can create osteolysis, a prolonged inflammatory response, and necrotic soft tissue between bone fragments [11, 14, 38,39,40]. Infection does not always present with classical symptoms and has been reported to be present in as low as 5% and as high as 20% of perceived aseptic nonunions [11, 41•]. Therefore, special effort should be taken to rule out infection in all cases of nonunion, especially recalcitrant ones.
Clinical Risk Factors
Nonunion development is dependent on injury factors such as fracture severity and location as well as systemic illness and medication use in the patient. These risk factors and others were examined recently in a large cohort of patients across multiple centers by Zura et al. [4]. Regarding injury risk factors, this review demonstrated that severe fractures caused by a high-energy mechanism, that were open, and that were accompanied by multiple concomitant fractures were at the highest risk for nonunion [4]. This agrees with a recent review supporting the association between higher Gustilo-Anderson classification and nonunion development in the tibia [42]. The most notable systemic illness associated with nonunion is diabetes, showing association in both clinical reviews and animal studies [4, 43]. Smoking is likely the most long-standing modifiable risk factor associated with nonunion, which has been corroborated by numerous bed and bench-side investigations [44,45,46,47]. Nicotine’s vasoconstrictive effect and role as a disruptor of TNF-α signaling gives pathophysiological insight into the association [44, 48]. Interestingly, the review by Zura et al. did not find smoking to be a major risk factor in their analysis; however, this is likely a false negative due to underreporting [4].
The uses of certain antibiotics, anticoagulants, anticonvulsants, and bisphosphonates acutely have all demonstrated associations with nonunion [4, 49, 50•, 51, 52]. Regarding analgesics, acute as well as chronic use of opioids have been found to contribute to nonunion [4, 50•]. Given the trauma population’s susceptibility to nonunion risk factors, a confounder(s) could explain this association; however, results thus far are a cause for concern and warrant immediate further investigation. Nonsteroidal anti-inflammatory drugs (NSAIDs) have a conflicting experimental association with nonunion despite existing dogma prohibiting their use during the fracture recovery period in the USA [45, 50•, 53, 54•]. Given the recent association between opioids and nonunion, and evidence that NSAID supplementation can decrease opioid use postoperatively, more research is called for regarding NSAID safety in the acute period [54•]. The Pain Study, a large prospective multi-center randomized controlled trial, is currently investigating this question and will provide level 1 evidence in the near future [55].
It is wise not to consider any of these risk factors in a bubble. The review by Zura et al. demonstrated that the majority of significant risk factors were much less impactful as singular actors in nonunion through a comparison of univariate and multivariate odds ratios [4]. Recent work by Mills et al. supports this idea, finding that two thirds of patients with nonunion had more than one attributable cause [11]. Surgeons, therefore, should be sure to consider the interplay of all aforementioned risk factors when risk stratifying for nonunion.
Role of Genetics
With the increasing accessibility of genetic testing, identifying genetic predispositions for fracture nonunion has become a topic of hot research. Results thus far point to single nucleotide polymorphisms in inhibitors of BMP, as well as haplotypes of BMP-4, fibroblast growth factor receptor-1, and platelet-derived growth factor (PDGF), as potential causative agents of aseptic nonunion [15, 56,57,58]. Complimentary deoxyribonucleic acid array analysis has also elucidated 8 genes expressed locally at significantly higher levels in nonunion tissue when compared to fresh callus [59]. Most recently, 3 studies have also identified multiple micro-ribonucleic acids linked to both impaired and augmented fracture healing [60,61,62]. Although application of these results is limited by study design and current understanding of nonunion pathogenesis, it is reasonable to envision a future supplementary role of genetic testing in individualized nonunion risk determination.
Table 1 provides a summary of these factors as well as other biologic, injury associated, and patient comorbidity factors affecting the progression of bone healing.
Clinical Assessments
History and Physical Exam
Diagnosis of fracture nonunion first involves a careful history taking and physical exam. Pain/tenderness with palpation at the fracture site, pain/tenderness with weight-bearing, and/or an inability to bear weight altogether can be expected in cases of nonunion, extrapolated from the 3 accepted physical exam criteria by which a healed fracture is defined [64]. The clinical assessment also includes evaluation of movement at the fracture site, inspection for signs of infection, and assessment of lengthening of shortening of the fracture segment as well as alignment. Assessment of the patient’s weight-bearing status and compliance is also important since delayed weight-bearing was recently demonstrated to be associated with delayed healing [65]. This association is likely explained by the need for an optimal stress environment to induce bone healing in accordance with Perren’s strain theory [66].
Imaging
Although the physical exam can offer clues that a fracture is failing to heal, radiologic assessment is the essential element of formal nonunion diagnosis. Plain radiographs allow for assessments of callus bridging, displacement, and angulation at the fracture site, as well as implant loosening or failure hinting at a present or impending nonunion. Traditionally, radiographic determination of nonunion has been largely subjective, leading to poor reliability in determining the stage of union [67, 68]. In an effort to create more objective radiographic parameters, standardized criteria in the form of radiographic union scores for the tibia (RUST), hip (RUSH), humerus (RUSHU), and radius (RUSS) have been developed with interobserver reliability demonstrated in all scoring systems [69,70,71,72,73]. All four scoring systems utilize a modified version of the same base scoring system, in which the presence of callus and visible fracture line at anterior, posterior, medial, and lateral cortices is rated on a 3-point scale with higher scores correlating to union [70,71,72,73]. The RUSHU system most modifies this base criteria, applying separate scores for cortical bridging and fracture line visibility to each cortex in addition to two scores for trabecular consolidation [70]. A modified RUST (mRUST) score with a higher interobserver agreement than the traditional RUST has also been developed. The mRUST differs from RUST in that it further subdivides the middle “present callus” score of 2 in the RUST for more descriptive staging, making each cortical score out of four instead of three [74]. Figure 2 shows the mRUST system applied (Fig. 2). Validation work in animal models show that RUST and mRUST scoring systems are significantly correlated with healing time, bone mineral density, and biomechanical parameters [75,76,77]. The next step in widespread application of RUST, mRUST, and other union scores for nonunion involves identifying a reliable threshold score at a well-defined time point that can diagnose or even predict the complication in each bone. Recent attempts have identified a RUSH score of < 18 at 6 months after injury in the femoral neck, and a RUST score of roughly < 8 in the range of 11 to 14 weeks after injury in either the tibia or femur to distinguish a fracture that will heal from one that will progress to nonunion [78, 79]. Further research in this area is called for since identification of such a threshold score would provide an objective diagnostic and predicable value for earlier nonunion detection.
Modified RUST scoring of humerus fracture lateral radiographic series. Weeks after fracture are denoted. The images include numeric scoring for each cortex based on the Modified Radiological Union Score of Tibia criteria [74]. Scoring criteria is outlined below the radiograph series. For complete mRUST scoring, both cortices on anterior-posterior radiographs would also be scored and added to the scores as seen above
Other imaging options available for judging bone-healing progression are computed tomography (CT) scanning and ultrasound. CT scans have been posited as a more sensitive imaging study than plain radiographs for diagnosing nonunion; however, the demonstrated lack of specificity and artifact caused by implants adjacent to the fracture site limits clinical applicability [80, 81]. Alternatively, ultrasound is being considered an easily accessible, non-ionizing, and sensitive imaging technology for diagnosing nonunion. Early work by Craig et al. and Moed et al. first demonstrated ultrasound’s ability to detect developing callus before it could be visualized radiographically, implying it could be used to determine the osteogenic activity of a suspected nonunion [82, 83]. More recent results by Nicholson et al. demonstrated that at 6 weeks after injury, only 10% of patients had bridging callus on radiograph but 60% had sonographic bridging callus (SBC), of which all 60% went on to unite. Most importantly, this study showed that no SBC was present at any time point in the patients that developed nonunion [84•]. The use of contrast-enhanced ultrasound has also recently demonstrated predictive value in determining success of nonunion revision surgery in the tibia through its ability to assess perfusion at the fracture site [85]. These results, in combination with the accessibility and safety characteristic of ultrasound, suggest that this technology could be used in daily clinical practice in the near future.
Predictive Scoring
Using what is known about risk factors associated with nonunion in combination with validated imaging scoring tools, multiple predictive scoring systems have been developed. One such system integrates the RUST score, the presence of infection, and the Nonunion Risk Determination Score created by O’Halloran et al., utilizing 3 relevant paramters to stratify nonunion risk in tibia fractures [86, 87]. Another, by Zura et al., was created using a database of over 90 million participants and has demonstrated the ability to predict nonunion in 18 different bones [88••]. Overall, these scoring systems represent non-invasive, objective tools that provide the clinician and patient valuable information to help guide clinical decision-making. Efforts should be taken to validate these scores prospectively and increase accessibility for real-time use in the clinic.
Serum Analysis
The documented circulation of signaling proteins during all phases of fracture repair posits that serological differences may exist between patients with unremarkable healing and those with nonunion. Importantly, recent proof of concept work by Hussein et al. demonstrated that changes in expression levels of various proteins involved in fracture healing were detectable through serum analysis, and identified 50 candidate proteins of particular interest [89•]. Past work identifying potential markers has demonstrated differences between alkaline phosphate, osteocalcin, and various pro-inflammatory cytokines between normal and nonunion models; small sample sizes and poor controls in these studies limit current applicability [90••, 91,92,93]. Properly controlled, in vivo research is called for since identification of reliable serum markers of nonunion would provide an immensely valuable, objective, and accessible test with decisive clinical applicability.
Treatment Approaches
Surgical Approaches
Surgery is often indicated for the treatment of an established nonunion. If mechanical failure is the suspected cause of the nonunion, revision, replacement, and/or augmentation of the initial construct is indicated to optimize the strain experienced within the fracture site environment to induce healing [5, 6, 8•, 49, 94]. Optimizing the mechanical environment can also involve restoring proper limb alignment, helping recover native on-axis force transmission that can improve patient function and protect implants [5]. If deep infection is noted, surgical intervention is required to irrigate and debride necrotic bone and soft tissue to create clean bony margins capable of growth (95). These cases may also require delivery of local antibiotics in the form of antibiotic nails, cement beads, and cement spacers [94, 96,97,98]. In the case of large cortical defects, as can occur in high-grade open injuries or in the aftermath of osteomyelitis, optimizing the mechanical environment may require the use of bone transport techniques such as application of an Ilizarov external frame [99]. The Ilizarov frame in combination with distraction of 1 mm per day following a latency period may provide adequate stimulus for bone growth in these complicated cases, preventing the need for amputation [100, 101].
In large cortical defects or nonunions deemed atrophic, bone grafting may be useful to optimize both the mechanical and biologic environment. Favorable grafts are osteogenic, osteoinductive, and osteoconductive in nature. Autograft harvested from the iliac crest (ICBG) manifests these three qualities and is the current gold standard in practice. Donor site morbidity and even associated healthcare costs have prompted the search for other options [49, 94, 102, 103]. In an effort to mitigate these drawbacks, the reamer irrigator aspirator system (RIA) was developed and has since proven to be an efficacious alternative for harvesting autograft to ICBG with decreased cost and morbidity despite questions about its yield of bone-forming cells [49, 94, 102, 104]. Allograft combined with recombinant growth factors is also an option without the drawback of host harvest [105]. Demonstration of decreased efficacy compared to autograft and associated disease transmission risks, however, limits widespread implementation [94, 103]. Of note, a recent review by Maceroli et al. found the mechanism of injury, increasing body mass, cortical defect size, flap size, and insurance status to be significantly associated with failure of the bone grafting procedure itself [106].
Current research is investigating the combination of bone marrow aspirate concentrate (BMAC) with an osteoconductive scaffold as an alternative to ICBG and RIA autograft. Thus far, BMAC has proven a reliable source of concentrated MSCs that are not only osteogenic but also osteoinductive [94, 103, 107, 108]. This is superior to both autograft and growth factor supplementation, as MSCs from BMAC are able to engage in coordinated paracrine signaling at physiological levels [94, 108]. Support for the efficacy of BMAC was recently provided by Gianakos et al. in their review of animal studies, finding superior outcomes for osteoconductive scaffolds treated with BMAC compared to osteoconductive scaffolds with no progenitor cells across all measurable markers of bone healing in the vast majority of experiments [109]. An even more recent retrospective study in humans found no difference in union rates between nonunions treated with BMAC vs. ICBG [110•]. Regarding collection method, bone marrow centrifuged to BMAC is also taken from the iliac crest; however, it has demonstrated less donor site morbidity in comparison to ICBG grafting with a researched sector rule for directing safe extraction [108, 111]. Thus, BMAC, in combination with one of the many commercially available osteoconductive scaffolds, appears to be a promising future treatment option in biologically non-viable nonunions or those with large cortical defects. It should be noted, however, that recent work in the cardiovascular field demonstrated that the benefit of stem cell–based therapies was from the acute immune response they generated rather than from specific properties of the cells themselves [112]. This consequently calls into question whether MSCs and therefore BMAC is superior to other stem cell–based therapies as promoters of healing in bone; further basic science research comparing MSCs and BMAC to cell-based controls is called for.
Commercial Biologic Adjuvants
Multiple adjuvant therapies have been developed for the purpose of augmenting the biologic capacity of an atrophic nonunion. Of these, demineralized bone matrix (DBM), BMPs, PDGF, and parathyroid hormone (PTH) therapy are the four with greatest clinical traction. DBM was the first of these therapies to be researched and introduced, building off the landmark discovery in 1965 of Marshall Urist [113]. However, a review of the literature reveals only methodologically weak studies demonstrating a benefit for DBM in healing from which clinical decisions cannot be based [94, 114]. Also deriving from Urist’s experiment was the discovery of BMP’s as critical osteoinductive cytokines, from which considerable research and development has led to the commercialization of both recombinant human rhBMP-2 and rhBMP-7 [102, 115]. Regarding comparison between the two types, two recent trials have demonstrated rhBMP-2 to be superior to rhBMP-7 for the indication of nonunion [116, 117]. Few trials exist, however, supporting either type’s efficacy in comparison to the standard of care autologous bone graft [30, 94]. Questions surrounding dosage and delivery method, high cost, and potential complications from usage also raise concerns regarding the viability of BMP for clinical use [30, 94]. Similar to both rhBMP-2 and rhBMP-7, recombinant human-derived PDGF-BB (PDGF-BB) is commercially available and FDA approved as an adjuvant for nonunion. However, in contrast to both rhBMPs, PDGF-BB is only approved for use in the setting of foot and ankle arthrodesis nonunion for which it has demonstrated promising healing results [118]. Further work supporting its efficacy and safety in comparison to autograft and in nonunion models of other bones may expand its use. Lastly, PTH injection, available commercially as Teriparatide, has demonstrated promise as a healing adjuvant in osteoporosis and acute fracture but not yet reliably in nonunion and is not currently FDA approved for this indication [94, 119]. In summary, at current, each of these adjuvant therapy options hold immense potential value as local stimulators of fracture nonunion. However, a lack of strong, consistent level I evidence and questions about the logistics of their application continues to prevent their wholehearted adoption.
Bone Stimulation
Bone growth stimulators are an available, non-invasive, adjuvant treatment option for nonunion. Stimulator types include direct current, capacitive coupling, inductive coupling, and low-intensity pulsed ultrasound (LIPUS). A recent review by Haglin et al. summarized the research to date on all four types for the indication of nonunion, finding no consistent level I evidence to support a clinical recommendation of the use of any stimulator type [120]. This does not mean, however, that bone stimulators hold no potential treatment value. In their review, Haglin et al. acknowledged that inductive and capacitive coupling stimulators had “fair evidence” in the form of multiple level II and level III studies with consistent findings supporting their efficacy [120]. A randomized controlled trial by Schofer et al. and meta-analysis by Leighton et al. on the efficacy of LIPUS also points to LIPUS’s success in the 3- to 6-month period following revision surgery for augmenting union, with Leighton et al. even going so far as to argue for their efficacy over surgical intervention [121, 122]. Even in the absence of substantial level I evidence, the indication may still exist for the use of LIPUS, inductive coupling, and capacitive coupling stimulators in light of a risk-benefit analysis. To date, no evidence links LIPUS, inductive or capacitive coupling bone stimulators to any harmful side effects; thus, these devices are low-risk, potentially maximum benefit adjuvant therapies that at worst have no effect. Therefore, a cost-benefit analysis would be a beneficial next step in addition to aforementioned level I evidence for the purposes of determining the need to elevate bone stimulators from offered to recommended in the clinic. Table 2 summarizes treatment options currently available to clinicians for fracture nonunion.
Conclusion
Fracture nonunion remains a debilitating complication characterized by high patient morbidity and economic burden. Best clinical estimates point to either mechanical failure or disruption of the “bone-healing unit” at the fracture site as the chief causative elements, although an understanding of what this means at a molecular level remains to be seen. What can be agreed upon is that the healing process involves a complex interaction of cellular signaling that is difficult to augment exogenously with commercial biologics such as rhBMPs or DBM. Zooming out from the cellular level reveals that progress has been made identifying clinical risk factors associated with nonunion and objective measures have improved with which to make a timely diagnosis. Clinical implementation of other promising diagnostic and therapeutic technologies has largely been limited by a lack of convincing evidence from reproducible and methodologically sound studies. Based on current trends, studies of this nature are likely in the near future; with them will hopefully come the evidence to support adoption of new solutions to what has been a long-standing clinical challenge.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Brinker MR, Trivedi A, O'Connor DP. Debilitating effects of femoral nonunion on health-related quality of life. J Orthop Trauma. 2017;31(2):e37–42. https://doi.org/10.1097/BOT.0000000000000736.
Hak DJ, Fitzpatrick D, Bishop JA, Marsh JL, Tilp S, Schnettler R, et al. Delayed union and nonunions: epidemiology, clinical issues, and financial aspects. Injury. 2014;45:S3–7. https://doi.org/10.1016/j.injury.2014.04.002.
Mills LA, Aitken SA, Simpson AH. The risk of non-union per fracture: current myths and revised figures from a population of over 4 million adults. Acta Orthop. 2017;88(4):434–9. https://doi.org/10.1080/17453674.2017.1321351.
Zura R, Xiong Z, Einhorn T, Watson JT, Ostrum RF, Prayson MJ, et al. Epidemiology of fracture nonunion in 18 human bones. JAMA Surg. 2016;151(11):e162775. https://doi.org/10.1001/jamasurg.2016.2775.
Harwood PJ, Ferguson DO. (ii) An update on fracture healing and non-union. Orthop Trauma. 2015;29(4):228–42. https://doi.org/10.1016/j.mporth.2009.12.004.
Andrzejowski P, Giannoudis PV. The ‘diamond concept’ for long bone non-union management. J Orthop Traumatol. 2019;20(1):21. https://doi.org/10.1186/s10195-019-0528-0.
Bhandari M, Fong K, Sprague S, Williams D, Petrisor B. Variability in the definition and perceived causes of delayed unions and nonunions: a cross-sectional, multinational survey of orthopaedic surgeons. J Bone Joint Surg. 2012;94(15):e109. https://doi.org/10.2106/JBJS.K.01344.
• Elliott DS, Newman KJ, Forward DP, Hahn DM, Ollivere B, Kojima K, et al. A unified theory of bone healing and nonunion: BHN theory. Bone Joint J. 2016;98(7):884–91. https://doi.org/10.1302/0301-620X.98B7.36061This review establishes the idea of the “bone-healing unit,” a critical entity that forms around the fracture site following injury. This paper posits that disruption of this unit is the mechanism behind nonunion and that mechanical instability causes such disruption in the majority of cases.
Brownlow HC, Reed A, Simpson AH. The vascularity of atrophic non-unions. Injury. 2002;33(2):145–50. https://doi.org/10.1016/s0020-1383(01)00153-x.
Panteli M, Pountos I, Jones E, Giannoudis PV. Biological and molecular profile of fracture non-union tissue: current insights. J Cell Mol Med. 2015;19(4):685–713. https://doi.org/10.1111/jcmm.12532.
Mills L, Tsang J, Hopper G, Keenan G, Simpson AH. The multifactorial aetiology of fracture nonunion and the importance of searching for latent infection. Bone Jt Res. 2016;5(10):512–9. https://doi.org/10.1302/2046-3758.510.BJR-2016-0138.
Calori GM, Colombo M, Mazza EL, Mazzola S, Malagoli E, Marelli N, et al. Validation of the non-union scoring system in 300 long bone non-unions. Injury. 2014;45:S93–7. https://doi.org/10.1016/j.injury.2014.10.030.
Calori GM, Phillips M, Jeetle S, Tagliabue L, Giannoudis PV. Classification of non-union: need for a new scoring system? Injury. 2008;39:S59–63. https://doi.org/10.1016/S0020-1383(08)70016-0.
Brinker MR, O’Connor DP. The biological basis for nonunions. JBJS Rev. 2016;4(6). https://doi.org/10.2106/JBJS.RVW.15.00078.
Copuroglu C, Calori GM, Giannoudis PV. Fracture non-union: who is at risk? Injury. 2013;44(11):1379–82. https://doi.org/10.1016/j.injury.2013.08.003.
Fong K, Truong V, Foote CJ, Petrisor B, Williams D, Ristevski B, et al. Predictors of nonunion and reoperation in patients with fractures of the tibia: an observational study. BMC Musculoskelet Disord. 2013;14(1):103. https://doi.org/10.1186/1471-2474-14-103.
Lin SJ, Chen CL, Peng KT, Hsu WH. Effect of fragmentary displacement and morphology in the treatment of comminuted femoral shaft fractures with an intramedullary nail. Injury. 2014;45(4):752–6. https://doi.org/10.1016/j.injury.2013.10.015.
Ricci WM, Streubel PN, Morshed S, Collinge CA, Nork SE, Gardner MJ. Risk factors for failure of locked plate fixation of distal femur fractures: an analysis of 335 cases. J Orthop Trauma. 2014;28(2):83–9. https://doi.org/10.1097/BOT.0b013e31829e6dd0.
Santolini E, West R, Giannoudis PV. Risk factors for long bone fracture non-union: a stratification approach based on the level of the existing scientific evidence. Injury. 2015;46:S8–19. https://doi.org/10.1016/S0020-1383(15)30049-8.
Schemitsch EH, Bhandari M, Guyatt G, Sanders DW, Swiontkowski M, Tornetta P III, et al. Prognostic factors for predicting outcomes after intramedullary nailing of the tibia. J Bone J Surg. 2012;94(19):1786–93. https://doi.org/10.2106/JBJS.J.01418.
Yiannakopoulos CK, Kanellopoulos AD, Trovas GP, Dontas IA, Lyritis GP. The biomechanical capacity of the periosteum in intact long bones. Arch Orthop Trauma Surg. 2008;128(1):117–20. https://doi.org/10.1007/s00402-007-0433-5.
Claes L, Recknagel S, Ignatius A. Fracture healing under healthy and inflammatory conditions. Nat Rev Rheumatol. 2012;8(3):133–43. https://doi.org/10.1038/nrrheum.2012.1.
Gerstenfeld LC, Cullinane DM, Barnes GL, Graves DT, Einhorn TA. Fracture healing as a post-natal developmental process: molecular, spatial, and temporal aspects of its regulation. J Cell Biochem. 2003;88(5):873–84. https://doi.org/10.1002/jcb.10435.
Gerstenfeld LC, Cho TJ, Kon T, Aizawa T, Tsay A, Fitch J, et al. Impaired fracture healing in the absence of TNF-α signaling: the role of TNF-α in endochondral cartilage resorption. J Bone Miner Res. 2003;18(9):1584–92. https://doi.org/10.1359/jbmr.2003.18.9.1584.
Loi F, Córdova LA, Pajarinen J, Lin TH, Yao Z, Goodman SB. Inflammation, fracture and bone repair. Bone. 2016;86:119–30. https://doi.org/10.1016/j.bone.2016.02.020.
Prystaz K, Kaiser K, Kovtun A, Haffner-Luntzer M, Fischer V, Rapp AE, et al. Distinct effects of IL-6 classic and trans-signaling in bone fracture healing. Am J Pathol. 2018;188(2):474–90. https://doi.org/10.1016/j.ajpath.2017.10.011.
Yang X, Ricciardi BF, Hernandez-Soria A, Shi Y, Camacho NP, Bostrom MP. Callus mineralization and maturation are delayed during fracture healing in interleukin-6 knockout mice. Bone. 2007;41(6):928–36. https://doi.org/10.1016/j.bone.2007.07.022.
Huang L, Liu S, Song T, Zhang W, Fan J, Liu Y. Blockade of interleukin 6 by rat anti-mouse interleukin 6 receptor antibody promotes fracture healing. Biochem Mosc. 2017;82(10):1193–9. https://doi.org/10.1134/S0006297917100121.
Mathieu M, Rigutto S, Ingels A, Spruyt D, Stricwant N, Kharroubi I, et al. Decreased pool of mesenchymal stem cells is associated with altered chemokines serum levels in atrophic nonunion fractures. Bone. 2013;53(2):391–8. https://doi.org/10.1016/j.bone.2013.01.005.
Krishnakumar GS, Roffi A, Reale D, Kon E, Filardo G. Clinical application of bone morphogenetic proteins for bone healing: a systematic review. Int Orthop. 2017;41(6):1073–83. https://doi.org/10.1007/s00264-017-3471-9.
Wang T, Zhang X, Bikle DD. Osteogenic differentiation of periosteal cells during fracture healing. J Cell Physiol. 2017;232(5):913–21. https://doi.org/10.1002/jcp.25641.
Ding ZC, Lin YK, Gan YK, Tang TT. Molecular pathogenesis of fracture nonunion. J Orthop Translat. 2018;14:45–56. https://doi.org/10.1016/j.jot.2018.05.002.
Wigner NA, Kulkarni N, Yakavonis M, Young M, Tinsley B, Meeks B, et al. Urine matrix metalloproteinases (MMPs) as biomarkers for the progression of fracture healing. Injury. 2012;43(3):274–8. https://doi.org/10.1016/j.injury.2011.05.038.
Clarkin CE, Gerstenfeld LC. VEGF and bone cell signalling: an essential vessel for communication? Cell Biochem Funct. 2013;31(1):1–1. https://doi.org/10.1002/cbf.2911.
Bielby R, Jones E, McGonagle D. The role of mesenchymal stem cells in maintenance and repair of bone. Injury. 2007;38(1):S26–32. https://doi.org/10.1016/j.injury.2007.02.007.
De Bari C, Dell’Accio F, Vanlauwe J, Eyckmans J, Khan IM, Archer CW, et al. Mesenchymal multipotency of adult human periosteal cells demonstrated by single-cell lineage analysis. Arthritis Rheum. 2006;54(4):1209–21. https://doi.org/10.1002/art.21753.
Zaghloul A, Haddad B, Barksfield R, Davis B. Early complications of surgery in operative treatment of ankle fractures in those over 60: a review of 186 cases. Injury. 2014;45(4):780–3. https://doi.org/10.1016/j.injury.2013.11.008.
Calori GM, Albisetti W, Agus A, Iori S, Tagliabue L. Risk factors contributing to fracture non-unions. Injury. 2007;38:S11–8. https://doi.org/10.1016/s0020-1383(07)80004-0.
Rodriguez EK, Boulton C, Weaver MJ, Herder LM, Morgan JH, Chacko AT, et al. Predictive factors of distal femoral fracture nonunion after lateral locked plating: a retrospective multicenter case-control study of 283 fractures. Injury. 2014;45(3):554–9. https://doi.org/10.1016/j.injury.2013.10.042.
Westgeest J, Weber D, Dulai SK, Bergman JW, Buckley R, Beaupre LA. Factors associated with development of nonunion or delayed healing after an open long bone fracture: a prospective cohort study of 736 subjects. J Orthop Trauma. 2016;30(3):149–55. https://doi.org/10.1097/BOT.0000000000000488.
• Olszewski D, Streubel PN, Stucken C, Ricci WM, Hoffmann MF, Jones CB, et al. Fate of patients with a “surprise” positive culture after nonunion surgery. J Orthop Trauma. 2016;30(1):e19–23. https://doi.org/10.1097/BOT.0000000000000417This case series of 666 nonunions elucidated that in 20% of aseptic, but at risk, nonunions had a “surprise” positive intraoperative culture. This suggests the collection of intraoperative cultures for all nonunion cases regardless of inflammatory markers.
Thakore RV, Francois EL, Nwosu SK, Attum B, Whiting PS, Siuta MA, et al. The Gustilo–Anderson classification system as predictor of nonunion and infection in open tibia fractures. Eur J Trauma Emerg Surg. 2017;43(5):651–6. https://doi.org/10.1007/s00068-016-0725-y.
Shibuya N, Humphers JM, Fluhman BL, Jupiter DC. Factors associated with nonunion, delayed union, and malunion in foot and ankle surgery in diabetic patients. J Foot Ankle Surg. 2013;52(2):207–11. https://doi.org/10.1053/j.jfas.2012.11.012.
Hernigou J, Schuind F. Smoking as a predictor of negative outcome in diaphyseal fracture healing. Int Orthop. 2013;37(5):883–7. https://doi.org/10.1007/s00264-013-1809-5.
Jeffcoach DR, Sams VG, Lawson CM, Enderson BL, Smith ST, Kline H, et al. Nonsteroidal anti-inflammatory drugs’ impact on nonunion and infection rates in long-bone fractures. J Trauma Acute Care Surg. 2014;76(3):779–83. https://doi.org/10.1097/TA.0b013e3182aafe0d.
Moghaddam A, Zimmermann G, Hammer K, Bruckner T, Grützner PA, von Recum J. Cigarette smoking influences the clinical and occupational outcome of patients with tibial shaft fractures. Injury. 2011;42(12):1435–42. https://doi.org/10.1016/j.injury.2012.08.017.
Pearson RG, Clement RG, Edwards KL, Scammell BE. Do smokers have greater risk of delayed and non-union after fracture, osteotomy and arthrodesis? A systematic review with meta-analysis. BMJ Open. 2016;6(11):e010303. https://doi.org/10.1136/bmjopen-2015-010303.
Chen Y, Guo Q, Pan X, Qin L, Zhang P. Smoking and impaired bone healing: will activation of cholinergic anti-inflammatory pathway be the bridge? Int Orthop. 2011;35(9):1267–70.
Bell A, Templeman D, Weinlein JC. Nonunion of the femur and tibia: an update. Orthop Clin N Am. 2016;47(2):365–75. https://doi.org/10.1007/s00264-011-1243-5.
•• Buchheit T, Zura R, Wang Z, Mehta S, Della Rocca GJ, Steen RG. Opioid exposure is associated with nonunion risk in a traumatically injured population: an inception cohort study. Injury. 2018;49(7):1266–71. https://doi.org/10.1016/j.injury.2018.05.004This cohort study of 45,000 fractures found that an elevated nonunion odds ratio was associated with acute and chronic use of schedule 2 opioids. Given the prevalence of opioid prescription in the aftermath of fracture, this raises intense concern about the effect opioids could potentially be having on healing.
Pountos I, Georgouli T, Bird H, Kontakis G, Giannoudis PV. The effect of antibiotics on bone healing: current evidence. Expert Opin Drug Saf. 2011;10(6):935–45. https://doi.org/10.1517/14740338.2011.589833.
Pountos I, Georgouli T, Blokhuis TJ, Pape HC, Giannoudis PV. Pharmacological agents and impairment of fracture healing: what is the evidence? Injury. 2008;39(4):384–94. https://doi.org/10.1016/j.injury.2007.10.035.
Kurmis AP, Kurmis TP, O’Brien JX, Dalén T. The effect of nonsteroidal anti-inflammatory drug administration on acute phase fracture-healing: a review. J Bone Joint Surg. 2012;94(9):815–23. https://doi.org/10.2106/JBJS.J.01743.
•• EL MD, Daniel JN, Rogero RG, Shakked RJ, Nicholson K, Pedowitz DI, et al. How does perioperative ketorolac effect opioid consumption and pain management after ankle fracture surgery? Clin Orthop Relat Res. 2019. https://doi.org/10.1097/CORR.0000000000000978This level 1 trial demonstrated that the addition of ketorolac to ankle fracture patients’ standardized postoperative pain regimen decreased opioid use with no difference in healing at 12 weeks. These results both suggest ketorolac as a safer analgesic alternative to opioids in the early postoperative period and oppose dogma that NSAIDs blunt healing.
Castillo RC, Raja SN, Frey KP, Vallier HA, Tornetta P III, Jaeblon T, et al. Improving pain management and long-term outcomes following high-energy orthopaedic trauma (pain study). J Orthop Trauma. 2017;31:S71–7. https://doi.org/10.1097/BOT.0000000000000793.
Dimitriou R, Carr IM, West RM, Markham AF, Giannoudis PV. Genetic predisposition to fracture non-union: a case control study of a preliminary single nucleotide polymorphisms analysis of the BMP pathway. BMC Musculoskelet Disord. 2011;12(1):44.
Guimarães JM, Guimarães IC, Duarte ME, Vieira T, Vianna VF, Fernandes MB, et al. Polymorphisms in BMP4 and FGFR1 genes are associated with fracture non-union. J Orthop Res. 2013;31(12):1971–9. https://doi.org/10.1186/1471-2474-12-44.
Zeckey C, Hildebrand F, Glaubitz LM, Jürgens S, Ludwig T, Andruszkow H, et al. Are polymorphisms of molecules involved in bone healing correlated to aseptic femoral and tibial shaft non-unions? J Orthop Res. 2011;29(11):1724–31. https://doi.org/10.1002/jor.21443.
Zimmermann G, Schmeckenbecher KH, Boeuf S, Weiss S, Bock R, Moghaddam A, et al. Differential gene expression analysis in fracture callus of patients with regular and failed bone healing. Injury. 2012;43(3):347–56. https://doi.org/10.1016/j.injury.2011.10.031.
He B, Zhang ZK, Liu J, He YX, Tang T, Li J, et al. Bioinformatics and microarray analysis of miRNAs in aged female mice model implied new molecular mechanisms for impaired fracture healing. Int J Mol Sci. 2016;17(8):1260. https://doi.org/10.3390/ijms17081260.
Waki T, Lee SY, Niikura T, Iwakura T, Dogaki Y, Okumachi E, et al. Profiling microRNA expression in fracture nonunions: potential role of microRNAs in nonunion formation studied in a rat model. Bone Joint J. 2015;97(8):1144–51. https://doi.org/10.1302/0301-620x.97B8.34966.
Waki T, Lee SY, Niikura T, Iwakura T, Dogaki Y, Okumachi E, et al. Profiling microRNA expression during fracture healing. BMC Musculoskelet Disord. 2016;17(1):83–8. https://doi.org/10.1186/s12891-016-0931-0.
Reverte MM, Dimitriou R, Kanakaris NK, Giannoudis PV. What is the effect of compartment syndrome and fasciotomies on fracture healing in tibial fractures? Injury. 2011;42(12):1402–7. https://doi.org/10.1016/j.injury.2011.09.007.
Morshed S, Corrales L, Genant H, Miclau T III. Outcome assessment in clinical trials of fracture-healing. J Bone J Surg. 2008;90(Supplement_1):62–7. https://doi.org/10.2106/JBJS.G.01556.
Houben IB, Raaben M, Batenburg MV, Blokhuis TJ. Delay in weight bearing in surgically treated tibial shaft fractures is associated with impaired healing: a cohort analysis of 166 tibial fractures. Eur J Orthop Surg Traumatol. 2018;28:1–8. https://doi.org/10.1007/s00590-018-2190-2.
Perren SM, Boitzy A. Cellular differentiation and bone biomechanics during consolidation of a fracture. Anat Clin. 1978;1(1):13–28.
Davis BJ, Roberts PJ, Moorcroft CI, Brown MF, Thomas PB, Wade RH. Reliability of radiographs in defining union of internally fixed fractures. Injury. 2004;35(6):557–61. https://doi.org/10.1016/S0020-1383(03)00262-6.
Hammer RR, Hammerby S, Lindholm B. Accuracy of radiologic assessment of tibial shaft fracture union in humans. Clin Orthop Relat Res. 1985;199:233–8. https://doi.org/10.1097/00003086-198510000-00033.
Bhandari M, Chiavaras MM, Parasu N, Choudur H, Ayeni O, Chakravertty R, et al. Radiographic union score for hip substantially improves agreement between surgeons and radiologists. BMC Musculoskelet Disord. 2013;14(1):70. https://doi.org/10.1186/1471-2474-14-70.
Chiavaras MM, Bains S, Choudur H, Parasu N, Jacobson J, Ayeni O, et al. The Radiographic Union Score for Hip (RUSH): the use of a checklist to evaluate hip fracture healing improves agreement between radiologists and orthopedic surgeons. Skelet Radiol. 2013;42(8):1079–88. https://doi.org/10.1007/s00256-013-1605-8.
Christiano AV, Goch AM, Leucht P, Konda SR, Egol KA. Radiographic union score for tibia fractures predicts success with operative treatment of tibial nonunion. J Clin Orthop Trauma. 2019;10(4):650–4. https://doi.org/10.1016/j.jcot.2018.10.010.
Oliver WM, Smith TJ, Nicholson JA, Molyneux SG, White TO, Clement ND, et al. The Radiographic Union Score for HUmeral fractures (RUSHU) predicts humeral shaft nonunion. Bone Joint J. 2019;101(10):1300–6. https://doi.org/10.1302/0301-620X.101B10.BJJ-2019-0304.R1.
Whelan DB, Bhandari M, Stephen D, Kreder H, McKee MD, Zdero R, et al. Development of the radiographic union score for tibial fractures for the assessment of tibial fracture healing after intramedullary fixation. J Trauma Acute Care Surg. 2010;68(3):629–32. https://doi.org/10.1097/TA.0b013e3181a7c16d.
Litrenta J, Tornetta P III, Mehta S, Jones C, O'Toole RV, Bhandari M, et al. Determination of radiographic healing: an assessment of consistency using RUST and modified RUST in metadiaphyseal fractures. J Orthop Trauma. 2015;29(11):516–20. https://doi.org/10.1097/BOT.0000000000000390.
Cooke ME, Hussein AI, Lybrand KE, Wulff A, Simmons E, Choi JH, et al. Correlation between RUST assessments of fracture healing to structural and biomechanical properties. J Orthop Res. 2018;36(3):945–53. https://doi.org/10.1002/jor.23710.
Fiset S, Godbout C, Crookshank MC, Zdero R, Nauth A, Schemitsch EH. Experimental validation of the Radiographic Union Score for Tibial fractures (RUST) using micro-computed tomography scanning and biomechanical testing in an in-vivo rat model. J Bone Joint Surg. 2018;100(21):1871–8. https://doi.org/10.2106/JBJS.18.00035.
Litrenta J, Tornetta P III, Ricci W, Sanders RW, O'toole RV, Nascone JW, et al. In vivo correlation of radiographic scoring (radiographic union scale for tibia fractures) and biomechanical data in a sheep osteotomy model: can we define union radiographically? J Orthop Trauma. 2017;31(3):127–30. https://doi.org/10.10997/BOT.0000000000000753.
Frank T, Osterhoff G, Sprague S, Garibaldi A, Bhandari M, Slobogean GP, et al. The radiographic union score for hip (RUSH) identifies radiographic nonunion of femoral neck fractures. Clin Orthop Relat Res. 2016;474(6):1396–404. https://doi.org/10.1007/s11999-015-4680-4.
Perlepe V, Cerato A, Putineanu D, Bugli C, Heynen G, Omoumi P, et al. Value of a radiographic score for the assessment of healing of nailed femoral and tibial shaft fractures: a retrospective preliminary study. Eur J Radiol. 2018;98:36–40. https://doi.org/10.1016/j.ejrad.2017.10.020.
Morshed S. Current options for determining fracture union. Adv Med. 2014;2014. https://doi.org/10.1155/2014/708574.
Bhattacharyya T, Bouchard KA, Phadke A, Meigs JB, Kassarjian A, Salamipour H. The accuracy of computed tomography for the diagnosis of tibial nonunion. J Bone Joint Surg. 2006;88(4):692–7. https://doi.org/10.2106/JBJS.E.00232.
Craig JG, Jacobson JA, Moed BR. Ultrasound of fracture and bone healing. Radiol Clin N Am. 1999;37(4):737–51. https://doi.org/10.1016/s0033-8389(05)70126-3.
Moed BR, Subramanian S, van Holsbeeck M, Watson JT, Cramer KE, Karges DE, et al. Ultrasound for the early diagnosis of tibial fracture healing after static interlocked nailing without reaming: clinical results. J Orthop Trauma. 1998;12(3):206–13. https://doi.org/10.1097/00005131-199803000-00013.
• Nicholson JA, Oliver WM, LizHang J, MacGillivray T, Perks F, Robinson CM, et al. Sonographic bridging callus: an early predictor of fracture union. Injury. 2019. https://doi.org/10.1016/j.injury.2019.09.027This prospective study demonstrated a reproducible use of ultrasound as a more sensitive technique for determining callus formation than radiograph at 6 weeks. This has applicability as a tool for nonunion risk stratification at an early time point.
Krammer D, Schmidmaier G, Weber MA, Doll J, Rehnitz C, Fischer C. Contrast-enhanced ultrasound quantifies the perfusion within tibial non-unions and predicts the outcome of revision surgery. Ultrasound Med Biol. 2018;44(8):1853–9. https://doi.org/10.1016/j.ultrasmedbio.2018.04.013.
O’Halloran K, Coale M, Costales T, Zerhusen T, Castillo RC, Nascone JW, et al. Will my tibial fracture heal? Predicting nonunion at the time of definitive fixation based on commonly available variables. Clin Orthop Rel Res. 2016;474(6):1385–95. https://doi.org/10.1007/s11999-016-4821-4.
Ross KA, O’Halloran K, Castillo RC, Coale M, Fowler J, Nascone JW, et al. Prediction of tibial nonunion at the 6-week time point. Injury. 2018;49(11):2075–82. https://doi.org/10.1016/j.injury.2018.07.033.
•• Zura R, Watson JT, Einhorn T, Mehta S, Della Rocca GJ, Xiong Z, et al. An inception cohort analysis to predict nonunion in tibia and 17 other fracture locations. Injury. 2017;48(6):1194–203. https://doi.org/10.1016/j.injury.2017.03.036This study represents the largest cohort analysis on nonunion to date, assessing the clinical outcomes of over 90 million patients to determine which factors contribute significantly to development of the complication. This information was then applied to create applicable models for nonunion risk stratification and prediction.
• Hussein AI, Mancini C, Lybrand KE, Cooke ME, Matheny HE, Hogue BL, et al. Serum proteomic assessment of the progression of fracture healing. J Orthop Res. 2018;36(4):1153–63. https://doi.org/10.1002/jor.23754This study demonstrated that serum proteomic analysis is a capable technique for detecting the differing expression of signaling proteins involved in the healing cascade. This provides valuable proof of concept work in the hunt for serum markers indicative of impaired healing.
•• Cheng A, Krishnan L, Pradhan P, Weinstock LD, Wood LB, Roy K, et al. Impaired bone healing following treatment of established nonunion correlates with serum cytokine expression. J Orthop Res. 2019;37(2):299–307. https://doi.org/10.1002/jor.24186This study utilized serum cytokine analyses to demonstrate an increased expression of inflammatory cytokines in nonunions failing to respond to treatment, both indicating a potential mechanism for nonunion and identifying detectable markers. These are valuable results as the orthopedic community pushes for identification of objective biological markers of nonunion.
Komnenou A, Karayannopoulou M, Polizopoulou ZS, Constantinidis TC, Dessiris A. Correlation of serum alkaline phosphatase activity with the healing process of long bone fractures in dogs. Vet Clin Path. 2005;34(1):35–8. https://doi.org/10.1111/j.1939-165x.2005.tb00006.x.
Pountos I, Georgouli T, Pneumaticos S, Giannoudis PV. Fracture non-union: can biomarkers predict outcome? Injury. 2013;44(12):1725–32. https://doi.org/10.1016/j.injury.2013.09.009.
van Baardewijk LJ, van der Ende J, Lissenberg-Thunnissen S, Romijn LM, Hawinkels LJ, Sier CF, et al. Circulating bone morphogenetic protein levels and delayed fracture healing. Int Orthop. 2013;37(3):523–7. https://doi.org/10.1007/s00264-012-1750-z.
Nauth A, Lee M, Gardner MJ, Brinker MR, Warner SJ, Tornetta P III, et al. Principles of nonunion management: state of the art. J Orthop Trauma. 2018;32:S52–7. https://doi.org/10.1097/BOT.00000000000001122.
Yin P, Zhang L, Li T, Zhang L, Wang G, Li J, et al. Infected nonunion of tibia and femur treated by bone transport. J Orthop Surg Res. 2015;10(1):49. https://doi.org/10.1186/s13018-015-0189-5.
Koury KL, Hwang JS, Sirkin M. The antibiotic nail in the treatment of long bone infection: technique and results. Orthop Clin North Am. 2017;48(2):155–65. https://doi.org/10.1016/j.ocl.2016.12.006.
McKee MD, Li-Bland EA, Wild LM, Schemitsch EH. A prospective, randomized clinical trial comparing an antibiotic-impregnated bioabsorbable bone substitute with standard antibiotic-impregnated cement beads in the treatment of chronic osteomyelitis and infected nonunion. J Orthop Trauma. 2010;24(8):483–90. https://doi.org/10.1097/BOT.0b013e3181df91d9.
Selhi HS, Mahindra P, Yamin M, Jain D, William G Jr, Singh J. Outcome in patients with an infected nonunion of the long bones treated with a reinforced antibiotic bone cement rod. J Orthop Trauma. 2012;26(3):184–8. https://doi.org/10.1097/BOT.0b013e318225f77c.
Simpson AH, Robiati L, Jalal MM, Tsang ST. Non-union: indications for external fixation. Injury. 2019;50:S73–8. https://doi.org/10.1016/j.injury.2019.03.053.
McNally M, Ferguson J, Kugan R, Stubbs D. Ilizarov treatment protocols in the management of infected nonunion of the tibia. J Orthop Trauma. 2017;31:S47–54. https://doi.org/10.1097/BOT.0000000000000987.
Saridis A, Panagiotopoulos E, Tyllianakis M, Matzaroglou C, Vandoros N, Lambiris E. The use of the Ilizarov method as a salvage procedure in infected nonunion of the distal femur with bone loss. J Bone Joint Surg British volume. 2006;88(2):232–7. https://doi.org/10.1302/0301-620X.88B2.16976.
Nauth A, Lane J, Watson JT, Giannoudis P. Bone graft substitution and augmentation. J Orthop Trauma. 2015;29:S34–8. https://doi.org/10.1097/BOT.0000000000000464.
Perez JR, Kouroupis D, Li DJ, Best TM, Kaplan L, Correa D. Tissue engineering and cell-based therapies for fractures and bone defects. Front Bioeng Biotech. 2018;6:105. https://doi.org/10.3389/fbioe.2018.00105.
Dawson J, Kiner D, Warren Gardner II, Swafford R, Nowotarski PJ. The reamer–irrigator–aspirator as a device for harvesting bone graft compared with iliac crest bone graft: union rates and complications. J Orthop Trauma. 2014;28(10):584–90. https://doi.org/10.1097/BOT.0000000000000086.
Baboolal TG, Boxall SA, El-Sherbiny YM, Moseley TA, Cuthbert RJ, Giannoudis PV, et al. Multipotential stromal cell abundance in cellular bone allograft: comparison with fresh age-matched iliac crest bone and bone marrow aspirate. Regen Med. 2014;9(5):593–607.
Maceroli MA, Gage MJ, Wise BT, Connelly D, Ordonio K, Castillo RC, et al. Risk factors for failure of bone grafting of tibia nonunions and segmental bone defects: a new preoperative risk assessment score. J Orthop Trauma. 2017;31:S55–9. https://doi.org/10.2217/rme.14.17.
Jones EA, Giannoudis PV, Kouroupis D. Bone repair with skeletal stem cells: rationale, progress to date and clinical application. Ther Adv Musculoskelet Dis. 2016;8(3):57–71. https://doi.org/10.1177/1759720X16642372.
Imam MA, Holton J, Ernstbrunner L, Pepke W, Grubhofer F, Narvani A, et al. A systematic review of the clinical applications and complications of bone marrow aspirate concentrate in management of bone defects and nonunions. Int Orthop. 2017;41(11):2213–20. https://doi.org/10.1007/s00264-017-3597-9.
Gianakos A, Ni A, Zambrana L, Kennedy JG, Lane JM. Bone marrow aspirate concentrate in animal long bone healing: an analysis of basic science evidence. J Orthop Trauma. 2016;30(1):1–9. https://doi.org/10.1097/BOT.0000000000000453.
•• Lin K, VandenBerg J, Putnam SM, Parks CD, Spraggs-Hughes A, CM MA, et al. Bone marrow aspirate concentrate with cancellous allograft versus iliac crest bone graft in the treatment of long bone nonunions. Ota Int. 2019;2(1):e012. https://doi.org/10.1097/OI9.0000000000000012Results from this study proport that BMAC with allograft is an efficacious alternative to autologous ICBG with decreased patient morbidity. This challenges the dogma that autologous ICBG is the gold standard for treatment of nonunions that are atrophic and/or have large cortical defects.
Hernigou J, Picard L, Alves A, Silvera J, Homma Y, Hernigou P. Understanding bone safety zones during bone marrow aspiration from the iliac crest: the sector rule. Int Orthop. 2014;38(11):2377–84. https://doi.org/10.1007/s00264-014-2343-9.
Vagnozzi RJ, Maillet M, Sargent MA, Khalil H, Johansen AK, Schwanekamp JA, et al. An acute immune response underlies the benefit of cardiac stem cell therapy. Nature. 2020;577(7790):405–9. https://doi.org/10.1038/s41586-019-1802-2.
Urist MR. Bone: formation by autoinduction. Science. 1965;150(3698):893–9. https://doi.org/10.1126/science.150.3698.893.
Drosos GI, Touzopoulos P, Ververidis A, Tilkeridis K, Kazakos K. Use of demineralized bone matrix in the extremities. World J Orthop. 2015;6(2):269–77. https://doi.org/10.5312/wjo.v6.i2.269.
Friedlaender GE, Perry CR, Cole JD, Cook SD, Cierny G, Muschler GF, et al. Osteogenic protein-1 (bone morphogenetic protein-7) in the treatment of tibial nonunions: a prospective, randomized clinical trial comparing rhOP-1 with fresh bone autograft. J Bone J Surg. 2001;83(Pt 2):S151.
Conway JD, Shabtai L, Bauernschub A, Specht SC. BMP-7 versus BMP-2 for the treatment of long bone nonunion. Orthopedics. 2014;37(12):e1049–57. https://doi.org/10.3928/01477447-20141124-50.
Haubruck P, Tanner MC, Vlachopoulos W, Hagelskamp S, Miska M, Ober J, et al. Comparison of the clinical effectiveness of bone morphogenic protein (BMP)-2 and-7 in the adjunct treatment of lower limb nonunions. Orthop Traumatol Surg Res. 2018;104(8):1241–8. https://doi.org/10.1016/j.ostr.2018.08.008.
Scott RT, McAlister JE, Rigby RB. Allograft bone: what is the role of platelet-derived growth factor in hindfoot and ankle fusions. Clin Podiatr Med Surg. 2018;35(1):37–52. https://doi.org/10.1016/j.cpm.2017.08.008.
Wojda SJ, Donahue SW. Parathyroid hormone for bone regeneration. J Orthop Res. 2018;36(10):2586–94. https://doi.org/10.1002/jor.24075.
Haglin JM, Jain S, Eltorai AE, Daniels AH. Bone growth stimulation: a critical analysis review. JBJS Rev. 2017;5(8):e8. https://doi.org/10.2106/JBJS.RVW.16.00117.
Leighton R, Watson JT, Giannoudis P, Papakostidis C, Harrison A, Steen RG. Healing of fracture nonunions treated with low-intensity pulsed ultrasound (LIPUS): a systematic review and meta-analysis. Injury. 2017;48(7):1339–47. https://doi.org/10.1016/j.injury.2017.05.016.
Schofer MD, Block JE, Aigner J, Schmelz A. Improved healing response in delayed unions of the tibia with low-intensity pulsed ultrasound: results of a randomized sham-controlled trial. BMC Musculoskelet Disord. 2010;11(1):229. https://doi.org/10.1186/1471-2474-11-229.
Griffin XL, Costa ML, Parsons N, Smith N. Electromagnetic field stimulation for treating delayed union or non-union of long bone fractures in adults. Cochrane Database Syst Rev. 2011;4. https://doi.org/10.1002/14651858.CD008471.pub2.
Hughes MS, Anglen JO. The use of implantable bone stimulators in nonunion treatment. Orthopedics. 2010;33(3). https://doi.org/10.3928/01477447-20100129-15.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
G. Bradley Reahl, Louis Gerstenfeld, and Michael Kain declare no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Orthopedic Management of Fractures
Rights and permissions
About this article
Cite this article
Reahl, G.B., Gerstenfeld, L. & Kain, M. Epidemiology, Clinical Assessments, and Current Treatments of Nonunions. Curr Osteoporos Rep 18, 157–168 (2020). https://doi.org/10.1007/s11914-020-00575-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11914-020-00575-6