Opinion Statement
Cannabis is a useful botanical with a wide range of therapeutic potential. Global prohibition over the past century has impeded the ability to study the plant as medicine. However, delta-9-tetrahydrocannabinol (THC) has been developed as a stand-alone pharmaceutical initially approved for the treatment of chemotherapy-related nausea and vomiting in 1986. The indication was expanded in 1992 to include treatment of anorexia in patients with the AIDS wasting syndrome. Hence, if the dominant cannabinoid is available as a schedule III prescription medication, it would seem logical that the parent botanical would likely have similar therapeutic benefits. The system of cannabinoid receptors and endogenous cannabinoids (endocannabinoids) has likely developed to help us modulate our response to noxious stimuli. Phytocannabinoids also complex with these receptors, and the analgesic effects of cannabis are perhaps the best supported by clinical evidence. Cannabis and its constituents have also been reported to be useful in assisting with sleep, mood, and anxiety. Despite significant in vitro and animal model evidence supporting the anti-cancer activity of individual cannabinoids—particularly THC and cannabidiol (CBD)—clinical evidence is absent. A single intervention that can assist with nausea, appetite, pain, mood, and sleep is certainly a valuable addition to the palliative care armamentarium. Although many healthcare providers advise against the inhalation of a botanical as a twenty-first century drug-delivery system, evidence for serious harmful effects of cannabis inhalation is scant and a variety of other methods of ingestion are currently available from dispensaries in locales where patients have access to medicinal cannabis. Oncologists and palliative care providers should recommend this botanical remedy to their patients to gain first-hand evidence of its therapeutic potential despite the paucity of results from randomized placebo-controlled clinical trials to appreciate that it is both safe and effective and really does not require a package insert.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cannabis has been used medicinally for millennia. The remains of a young woman dubbed the Siberian Ice Maiden have led anthropologists to speculate that cancer patients may have benefitted from it 2700 years ago [1]. This woman, estimated to be in her mid-20s at death, was found on MRI scanning to have metastatic breast cancer. A pouch containing cannabis was found around her waist. This generated hypotheses that she was using cannabis to treat the symptoms of her disease, or perhaps even the cancer itself. Cannabis made its way from East Asia along the Silk Road to the Indian subcontinent and further west into the Arab world where it was used medicinally for centuries [2, 3••]. Cannabis was reportedly introduced to the West by W.B. O’Shaughnessy, a surgeon working in India with the British East Indies Company who brought it to the UK. At the beginning of the twentieth century, most of the forerunners of today’s major pharmaceutical companies had cannabis products available in the USA for physicians to prescribe to their patients.
The death knell to the use of cannabis as medicine came in 1937 with the introduction of the Marihuana Tax Act. By using the Mexican name, Harry Anslinger, a prohibitionist who became the first head of the Federal Narcotics Bureau, did an end run around physicians who knew the medicine as cannabis. The Act imposed a levy of $1 an ounce for medical use and $100 an ounce for recreational use. The American Medical Association stood alone in opposing the Act which ultimately passed. Cannabis was removed from the US Pharmacopeia in 1942. In 1961, the United Nations Single Convention on Narcotic Drugs broadened its scope to include cannabis among prohibited narcotics. The Controlled Substances Act of 1970 placed cannabis in schedule I which means that it has a high potential for abuse and no accepted medical use. Studying the potential medical utility of cannabis in the USA has been difficult in that the only legal source for research has been the National Institute on Drug Abuse which has a mandate to study substances of abuse as substances of abuse and not as therapeutic agents [4, 5••].
Shortly after cannabis was placed in schedule I, the National Cancer Institute became a major supporter of clinical trials of synthetic delta-9-tetrahydrocannabinol (THC)—the main psychoactive cannabinoid in the plant—as a treatment for chemotherapy-induced nausea and vomiting [6]. Numerous studies conducted in the 1970s and 1980s eventually led to the approval of two THC pharmaceuticals for this indication in 1985–1986 [7,8,9,10,11]. Both dronabinol and nabilone were licensed and approved as the first therapeutic cannabinoids affording oncologists the opportunity to have the longest experience with recommending a cannabis-based medicine. In 1992, the US Food and Drug Administration expanded the labeling indication for dronabinol to include treatment of the anorexia associated with weight loss in patients with AIDS prompting clinical trials to investigate its utility in cancer anorexia–cachexia syndrome as well [12]. More recently, nabiximols, a whole plant extract of cannabis delivered as an oromucosal spray licensed and available in Canada and the European Union, has been studied in cancer-related pain [13,14,15,16,17]. Despite the clinical evidence on the synthetic cannabinoid and plant-derived pharmaceuticals, existing barriers to investigating the therapeutic potential of the botanical itself account for the paucity of evidence in the medical literature [5••].
The absence of evidence, however, is not evidence of absence of effect. Rather than an abundance of data from prospective, randomized, double-blind, placebo-controlled trials, increasing information is becoming available from surveys of large numbers of cancer patients accessing medicinal cannabis. A cross-sectional anonymous survey was completed by 926 patients seen at the Fred Hutchinson Cancer Research Center in Seattle, a National Cancer Institute–designated comprehensive cancer center [18•]. Nearly one-quarter of the respondents considered themselves as active cannabis users. Overall, 75% used cannabis for physical symptoms, 63% for neuropsychiatric symptoms, 35% recreationally, and 26% reported using cannabis to help treat their cancer. Cannabis was used most frequently for pain, followed by nausea and stress. Regardless of symptom, 51% scored cannabis as a major benefit and 39% felt it was of moderate benefit. Results from an analysis of cannabis use among 2970 Israeli cancer patients reported favorable effects on pain, sleep problems, nausea and vomiting, and weakness and fatigue [19•]. Two-thirds of the patients noted significant improvement after 1 month of cannabis use; 20% reported moderate improvement, and only 8% reported that cannabis was not helpful. One-third of the respondents reported a decrease in prescription medications including opioid analgesics, sedatives, hypnotics, and corticosteroids.
As oncologists are understandably most convinced by results published in the medical literature, this article will summarize currently available evidence. It is important to keep in mind the existing barriers to studying the botanical and appreciate that most of the existing data to date is derived from studies of the isolated THC pharmaceuticals or, increasingly, the oromucosal whole plant extract. The US National Academies of Sciences, Engineering and Medicine published The Health Effects of Cannabis and Cannabinoids in January 2017 which provides an excellent summary of the literature from studies conducted through 2016 [5••] The National Cancer Institute’s Physician Data Query Complementary and Alternative Medicine (NCI PDQ® CAM) website continuously updates available information on cannabis and cannabinoids in cancer [3••]. Downloadable summaries on the site are available for both health professionals and patients. A comprehensive updated review is also found in the online monograph Health Canada Information for Health Care Professionals: Marihuana (marijuana, cannabis) [20••].
Nausea and vomiting
Something must have prompted the NCI and pharmaceutical companies in the 1970s to turn attention to the THC molecule as a potential antiemetic for patients receiving chemotherapy. One could hypothesize it was anecdotal testimonials from cancer patients who derived benefit from inhalation of cannabis in the treatment of this vexing symptom. The body of evidence derived from numerous studies of both dronabinol and nabilone in the 1970s and 1980s has now been subjected to numerous meta-analyses and systematic reviews [9,10,11]. All conclude that the cannabinoids were superior to placebo and equal to or superior to the benefit obtained from the then available anti-emetics. As both pharmaceuticals were approved in 1980s, the impetus to conduct further trials comparing the THC therapies to more modern antiemetics has not been great. One study, however, did evaluate dronabinol compared to ondansetron in delayed nausea and vomiting prior to the availability of today’s substance P inhibitors for this indication [21]. In this trial of 61 patients receiving highly emetogenic chemotherapy, patients received dexamethasone with ondansetron and/or dronabinol or placebo for delayed nausea. The total response was the same for ondansetron (58%), dronabinol (54%), or combination (47%), and all were superior compared to placebo (20%). The intensity of the nausea and vomiting/retching was reported to be less in the dronabinol recipients.
Botanical cannabis has only been evaluated in three controlled clinical trials. In two of them, cannabis was only made available after dronabinol had failed, so not surprisingly, efficacy was lacking [8]. The third was a small 20-patient trial where the patients received 4 doses of inhaled cannabis or 4 doses of dronabinol. Treatment was only effective in five of the patients. Seven preferred dronabinol; four preferred the cannabis, and nine had no preference. The plant-derived extract, nabiximols, was also studied in a phase II placebo-controlled trial of 16 patients where 4.8 sprays of nabiximols was found to be superior to placebo in further decreasing chemotherapy-induced nausea [22].
Despite the lack of published evidence, anecdotal reports accumulated over 36 years of treating cancer patients in San Francisco would suggest that inhaled cannabis is active as an antiemetic in patients with chemotherapy-induced nausea and vomiting. Many patients, in fact, eschew prescription antiemetics altogether in favor of using cannabis-based interventions. This allows them to avoid the troubling constipation often reported as a side effect of serotonin 5-HT3 receptor antagonists. Cannabis use prior to a visit to the chemotherapy infusion center has also been reported to be useful in decreasing anticipatory nausea [23, 24, 25•]. Cannabis is also the only antiemetic that stimulates appetite.
The National Academies of Sciences, Engineering, and Medicine (NASEM) concluded that some of the strongest published evidence supports the conclusion that in adults with chemotherapy-induced nausea and vomiting, oral cannabinoids are effective antiemetics [5••, 26•]. The American Society of Clinical Oncology Expert Panel, however, recommends that FDA-approved cannabinoids dronabinol and nabilone only be used to treat nausea and vomiting that are resistant to standard therapies [27]. The guidelines conclude that “evidence remains insufficient to recommend marijuana in this setting.”
Appetite
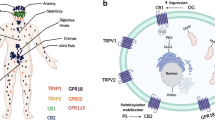
Humans, as well as all other animal species, possess an intricate system involving cannabinoid receptors and endogenous cannabinoids or endocannabinoids. The CB1 receptors is one of the most densely populated 7-transmembrane domain G protein–coupled receptor in the human brain [28]. CB1 receptors are found throughout the tissues of the body. The CB2 receptor was initially found on macrophages and the marginal zone of the spleen with high concentrations on B lymphocytes and natural killer cells suggesting that it may have something to do with immunity [29]. Anandamide and 2-arachidonoylglycerol (2-AG) are the two best characterized endocannabinoids that are made on demand and metabolized by different enzymes. CB1 receptors found in the lateral hypothalamus and limbic system locations are felt to be associated with food intake control, particularly by involvement in the motivational/reward aspects of eating. The plant cannabinoids—phytocannabinoids—can also exert their activities through complexing with the cannabinoid receptors. Early studies of smoked cannabis in a residential setting noted a 40% increase in caloric intake compared to placebo [30]. The increased intake was from snacks and not meals and was predominantly in the form of sweet solid items and not sweet liquids or savory solids.
Despite the well-known existence of the “munchies” as described above, the NASEM report found “no or insufficient evidence” for the benefit of cannabinoids in treatment of cancer-associated anorexia cachexia syndrome [5••]. This conclusion was based largely on results of a randomized double-blind study of 469 adults with advanced cancer and weight loss [12]. Participants were assigned to receive dronabinol 2.5 mg twice daily or the progestational hormone megestrol acetate 800 mg daily. Dronabinol was inferior to megestrol in increasing appetite and weight, and the combination was less effective than the megestrol alone.
A more recent trial of nabilone was conducted in 65 patients with advanced non-small cell lung cancer in Mexico [31]. The primary endpoints were appetite, nutritional status, and quality of life in this randomized, double blind, placebo-controlled trial. At the end of 8 weeks, the patients receiving the nabilone had increased intake of calories and carbohydrates and significantly improved quality of life; changes were not appreciated in the control group. Change in weight was not mentioned in the manuscript. It must be remembered, however, that THC is just one of approximately 400 chemicals present in the plant and that there is likely synergy between THC and the other bioactive phytochemicals that is absent in the isolated cannabinoid pharmaceutical. Currently, there is much interest in the effects of cannabidiol (CBD), a cannabinoid that is felt to lack the intoxicating effect of THC [32,33,34]. Very little is known about the therapeutic effect of CBD although it is becoming increasingly available worldwide. A Dutch study queried consumers regarding their experience with three different strains of available cannabis—19% THC, < 1% CBD; 12% THC, < 1% CBD; and 6% THC, 7.5% CBD [35]. The low THC group reported less appetite stimulation. Animal models suggest that when the ratio of THC:CBD is close to 1, CBD may attenuate some of THC’s effects, apparently beneficial as well as adverse [36].
Pain
It has been postulated that the reason humans and other animal species have the system of cannabinoid receptors and endocannabinoids is to help us to modulate our response to pain [37••]. It would not be surprising that the NASEM report found the strongest, most conclusive evidence supporting the finding that, in adults with chronic pain, those treated with cannabis and cannabinoids are more likely to experience a clinically significant reduction in pain symptoms [5••, 26•]. Numerous meta-analyses and systematic reviews on this topic have recently been published, albeit with somewhat conflicting conclusions [10, 38,39,40]. Overall, one would have to conclude that cannabis is useful in reducing pain. The largest body of evidence to date with regard to the effects of the botanical is in the reduction of neuropathic pain [5••, 26•, 41, 42]. A meta-analysis of six trials in HIV-related peripheral neuropathy supports cannabis as an effective treatment for this now less frequently encountered clinical entity [43]. A small crossover study of the oromucosal 1:1 THC:CBD pharmaceutical, nabiximols, in 16 patients with chemotherapy-induced peripheral neuropathy showed no overall difference between nabiximols and placebo [44]. However, a responder analysis of five patients who improved on nabiximols suggests that the number needed to treat for one to benefit was five supporting further studies of cannabinoids for this troublesome syndrome. As well, animal models demonstrate that cannabinoids can not only treat neuropathy induced by the vinca alkaloids, the platinums, and the taxanes, but it can also prevent them in some instances as well [45,46,47]. In general, patients who have developed chemotherapy-induced peripheral neuropathy should be recommended to consider a cannabis-based treatment. There may come a day when those commencing treatment with the offending agents might also be advised to use cannabis-based medicines as prophylaxis, but more research is certainly needed.
In animal models and in a few small, short clinical trials, there appears to be evidence that cannabinoids may be synergistic with opioids in relief of pain [48]. There does not appear to be any adverse interaction—clinical or pharmacokinetic—when the two classes are administered in twain. A randomized, double-blind, placebo-controlled study of nabiximols was conducted in 397 advanced cancer patients with chronic pain [17]. The difference in median percent improvements in average pain between 199 nabiximols and 198 placebo recipients did not quite reach statistical significance (P = 0.0854). Patient testimonials over the years have suggested that many cancer patients prescribed opiates to cope with end-of-life pain feel unable to communicate with their loved ones and are able to wean off the narcotics by substituting cannabis. In states where cannabis has been made legal for medical or recreational use, decreased prescriptions for opiates have been appreciated as well as a decline in opiate-associated mortality [49, 50].
CBD is felt to be analgesic and anti-inflammatory. Clinical trials to date have not investigated CBD as an analgesic. It has been suggested by a Rachel Peyraube, an experienced cannabis clinician in Uruguay where cannabis is legal who defines herself as an endocannabinologist, that CBD is only effective as an analgesic for pain secondary to inflammation, and the usual dose for an effect is 75–100 mg although some may benefit at lower doses (personal communication). CBD, as well as THC, is now available in sprays, balms, and creams that can be applied topically with patients reporting significant reduction in pain, but again, conclusive clinical trial data is lacking to date. Providers should be aware of the significant pharmacokinetic differences between inhaled and ingested cannabis-based medicines [51•]. Inhalation produces a peak in minutes compared to a much lower peak that is reached in hours when THC is orally ingested. When taken by mouth, the delta-9-THC is metabolized into an even more psychoactive metabolite on first pass metabolism explaining why people might be more prone to excessive psychotropic effects and overdosing with oral products. For better control over onset, depth, and duration of effect, inhalation may be optimal. If an orally ingested product with desirable activity is found, it decreases the need for more frequent dosing associated with inhalation. Tinctures and oils provide some degree of rapid sublingual absorption mimicking the inhaled kinetics, and these products are then swallowed providing the more sustained effect of oral ingestion.
Sleep
The NASEM report concluded that there was a moderate evidence of effect for cannabinoids improving short-term sleep outcomes in individuals with sleep disturbances associated with chronic pain, fibromyalgia, multiple sclerosis, and obstructive sleep apnea syndrome [5••]. Cancer patients frequently report difficulties with sleep. Phase I–III studies of nabiximols, with its THC:CBD ratio of 1:1, in 2000 subjects demonstrate marked improvement in subjective sleep parameters in patients with pain conditions including intractable cancer pain and peripheral neuropathic pain [13]. Many suggest that CBD-enriched cannabis preparations are particularly effective for inducing and maintaining sleep. Ethan Russo, a neurologist and ethnobotanist, finds this perplexing as he maintains that CBD is a stimulant not a soporific agent [52]. He asserts that CBD-rich cannabis chemovars (strains) are usually high in the terpene myrcene which he believes is what is beneficial for sleep. Be that as it may, many cancer patients are benefitting from cannabis-based medicines for sleep issues.
Anxiety
One of the most appreciated side effects of cannabis use is anxiety and paranoia [53]. On the other hand, many patients find cannabis useful, both medically and recreationally, to decrease anxiety. THC is more likely to be associated with increased anxiety while CBD is felt to be more anxiolytic [54]. The largest published randomized placebo-controlled trial of CBD prior to the recent Epidiolex® studies in children with refractory seizure disorders was a 24-person investigation of CBD in patients with social anxiety disorder during a simulated public speaking experience [55]. The 12 participants receiving the CBD had less anxiety during the simulation than the 12 receiving placebo. In states where medicinal cannabis is available, prescriptions for anti-anxiety medications have also decreased significantly [49].
Cannabis as an anti-cancer agent
The first evidence that cannabinoids may have was anti-cancer activity emanated from a report published in 1975 that showed that Lewis lung carcinoma cells could be inhibited in vitro by delta-9-THC, delta-8-THC, and CBD [56]. Since that time, much of the work in this area has been done in Spain and Italy with the bulk of it being done in animal glioma models [2, 25•, 57,58,59,60]. A meta-analysis of 34 in vitro studies found that cannabinoids selectively killed rodent glioma cells in all but one study leaving normal cells untouched [61]. Further investigations into the mechanism of action of cannabinoids have demonstrated antiproliferative effects via cell cycle arrest as well as cell death by way of toxicity, apoptosis, necrosis, and autophagy. In addition, cannabinoids block vascular endothelial growth factor leading to anti-angiogenesis and block matrix metaloproteinaise-2 leading to decreased invasiveness and metastasis. The anti-tumor effects of cannabinoids have been demonstrated in vitro and in animal models against a wide range of cancers [62,63,64,65].
Despite the wealth of in vitro and animal evidence, the NASEM report concluded that there was no or insufficient evidence that cannabinoids have anti-cancer effects in humans [5••]. One study conducted in nine patients with recurrent glioblastoma multiforme applied topical THC to the brain tumors via a catheter [66]. No benefit beyond that observed with chemotherapy alone was noted. A press release in February 2017 reported the results of a placebo-controlled trial of nabiximols in patients receiving chemotherapy for recurrent glioblastoma multiforme [67]. Ten of the 12 patients treated with nabiximols were alive at 1 year compared to only 5 of the 9 placebo recipients (P-0.042). At the time of the press release, the median survival of the nabiximols-treated patients was 550 days compared to 369 days in the placebo recipients. Although these results are encouraging, the study is small and has not yet been published in the peer-reviewed medical literature. It suggests that an oromucosal whole-plant extract with a THC:CBD ratio of 1:1 may augment the benefit of standard chemotherapy. As a number of other tumor types have also been shown to have increased or decreased expression of CB1, CB2 or both, this appears to be an area ripe for continued future research [68•].
Despite the lack of published evidence in the medical literature that cannabis or cannabinoids have any anti-tumor activity in humans, the Internet abounds with anecdotal reports of patients who have cured their own cancers using cannabis-derived products alone eschewing standard cancer therapies [69]. There are practitioners who specialize in developing regimens of potent cannabis oils for patients to use in hopes of shrinking their tumors. Often times, the THC doses are so high that patients spend much of their time bedridden unable even to eat. As both THC and CBD interact with hepatic cytochrome p450 isoforms, there is also a theoretical concern about botanical:pharmaceutical interactions. CBD especially is felt to be a potentially potent inhibitor of cytochrome p450 such that highly concentrated oils may lead to decreased metabolism and increased toxicity of conventional anti-cancer therapies [70]. The Food and Drug Administration issued a warning in November 2017 to companies claiming that they had cannabis-derived products that could cure cancer (https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm583295.htm).
Potential risks
Compared to many of the pharmaceuticals prescribed by oncologists, cannabis is a relatively benign intervention. No overdose deaths have been reported due to a lack of cannabinoid receptors on the brainstem involved in control of respiration as opposed to opiate receptors. The addictive potential is also quite low and likely not an issue in patients with advanced malignancy receiving palliative care. The NASEM report discussed the isolated case reports of pulmonary aspergillosis in patients with a variety of malignancies and other immunocompromised states [5••]. Case series analyses in patients with HIV failed to associate cannabis use with an increased risk of pulmonary aspergillosis [71]. There is also fear that smoking cannabis could lead to increased risk of upper aerodigestive malignancies. Numerous meta-analysis have failed to demonstrate an association between recreational cannabis use and either lung or head and neck cancers [5••]. There have been a number of epidemiologic studies suggesting an association between cannabis use and the development of non-seminomatous germ cell tumors, but it is unclear whether the association implies causation or is perhaps due to the fact that young men are frequent users of recreational cannabis and also most likely to develop testicular cancers [5••, 72].
Caution is advisable in the elderly and those with a history of cardiovascular disease. Cannabis use can lead to tachycardia and hyper- or hypotension which can be a stress on the heart. In addition, postural hypotension leading to falls is also a risk in these populations. People with prior adverse psychiatric experiences to cannabis use should also best avoid using it in the cancer setting. Finally, providers should be aware of the rare hyperemesis syndrome that may accompany chronic cannabis use [73].
Oncologists are always cautious about the potential for a botanical product to interact with prescribed anti-cancer therapies either via a cytochrome P450 interaction or an oxidant–antioxidant effect. Scant data exists investigating such interactions with cannabis. Other than in some in vitro studies showing potentiation of chemotherapy effects with cannabinoids, the only human pharmacokinetic study was of a cannabis tea with docetaxel and irinotecan which showed no adverse interaction although not many patients consume cannabis in this manner [74]. With regard to newer immunotherapies, a retrospective analysis of 140 patients receiving nivolumab for a variety of advanced malignancies in Israel has reported that the 51 individuals using cannabis in association with their immunotherapy had an inferior response rate compared to the 89 who did not (15.9% compared with 37.5% (P = 0.016)) [75]. There were no differences in performance free or overall survival between the groups leading the authors to suggest a prospective clinical trial should be conducted.
Physician education
Most physicians practicing medicine today were trained during the last 75 years of relative cannabis prohibition and have very little if any knowledge of the therapeutic potential of cannabis as medicine. A recent survey of 400 US clinical oncologists found that 80% discuss the use of medical cannabis with their patients, and although nearly 50% recommend it, less than 30% consider themselves knowledgeable enough to make such recommendations [76•]. This is likely the reason that they report that the patient brings up the topic 78% of the time it is discussed. A survey of 153 oncology providers in Minnesota found that 65% supported the use of medical cannabis, but 85% desired more education on the topic [77]. Clearly, some attention should be paid to provider education. Increasingly, course modules are available on-line or provider education is being mandated in states where cannabis has been made available for medical use. Practitioners should avail themselves of these opportunities to augment their knowledgebase. Cannabis, however, is a relatively benign intervention especially in the context of the other medications prescribed by the oncologist. Hopefully, all specialty societies will increase their educational offerings on the use of cannabis as medicine. Until then, oncologists should rest assured that patients will likely be able to figure out on their own how to best use this botanical therapy that does not really require a package insert.
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Abrams DI. Integrating cannabis into clinical cancer care. Curr Oncol. 2016;23(Suppl 2):S8.
Abrams DI, Guzman M. Cannabis in cancer care. Clin Pharmacol Ther. 2015;97(6):575–86.
•• https://www.cancer.gov/about-cancer/treatment/cam/hp/cannabis-pdq. Accessed 24 May 1999. A continuously updated summary of the published literature on cannabinoids in cancer care.
Stith SS, Vigil JM. Federal barriers to cannabis research. Science. 2016;352(6290):1182.
•• National Academies of Sciences, Engineering and Medicine. The health effects of cannabis and cannabinoids: the current state of evidence and recommendations for research. Washington, DC: National Academies Press; 2017. The latest review and analysis of the health effects as gleaned from recently published peer-reviewed articles with weight of evidence scoring.
Chang AE, Shiling DJ, Stillman RC, Goldberg NH, Seipp CA, Barofsky I, et al. Delta-9-tetrahydrocannabinol as an antiemetic in cancer patients receiving high-dose methotrexate: a prospective, randomized evaluation. Ann Intern Med. 1979;91(6):819–24.
Tramèr MR, Carroll D, Campbell FA, Reynolds DJ, Moore RA, McQuay HJ. Cannabinoids for control of chemotherapy induced nausea and vomiting: quantitative systematic review. BMJ. 2001;323(7303):16.
Amar MB. Cannabinoids in medicine: a review of their therapeutic potential. J Ethnopharmacol. 2006;105(1–2):1–25.
Smith LA, Azariah F, Lavender VT, Stoner NS, Bettiol S. Cannabinoids for nausea and vomiting in adults with cancer receiving chemotherapy. Cochrane Database Syst Rev. 2015;12(11):CD009464
Whiting PF, Wolff RF, Deshpande S, Di Nisio M, Duffy S, Hernandez AV, et al. Cannabinoids for medical use: a systematic review and meta-analysis. JAMA. 2015;313(24):2456–73.
Phillips RS, Friend AJ, Gibson F, Houghton E, Gopaul S, Craig JV, et al. Antiemetic medication for prevention and treatment of chemotherapy-induced nausea and vomiting in childhood. Cochrane Database Syst Rev. 2016;(2):CD007786.
Jatoi A, Windschitl HE, Loprinzi CL, Sloan JA, Dakhil SR, Mailliard JA, et al. Dronabinol versus megestrol acetate versus combination therapy for cancer-associated anorexia: a North Central Cancer Treatment Group study. J Clin Oncol. 2002;20(2):567–73.
Russo EB, Guy GW, Robson PJ. Cannabis, pain, and sleep: lessons from therapeutic clinical trials of Sativex®, a cannabis-based medicine. Chem Biodivers. 2007;4(8):1729–43.
Johnson JR, Burnell-Nugent M, Lossignol D, Ganae-Motan ED, Potts R, Fallon MT. Multicenter, double-blind, randomized, placebo-controlled, parallel-group study of the efficacy, safety, and tolerability of THC: CBD extract and THC extract in patients with intractable cancer-related pain. J Pain Symptom Manag. 2010;39(2):167–79.
Portenoy RK, Ganae-Motan ED, Allende S, Yanagihara R, Shaiova L, Weinstein S, et al. Nabiximols for opioid-treated cancer patients with poorly-controlled chronic pain: a randomized, placebo-controlled, graded-dose trial. J Pain. 2012;13(5):438–49.
Johnson JR, Lossignol D, Burnell-Nugent M, Fallon MT. An open-label extension study to investigate the long-term safety and tolerability of THC/CBD oromucosal spray and oromucosal THC spray in patients with terminal cancer-related pain refractory to strong opioid analgesics. J Pain Symptom Manag. 2013;46(2):207–18.
Lichtman AH, Lux EA, McQuade R, Rossetti S, Sanchez R, Sun W, et al. Results of a double-blind, randomized, placebo-controlled study of nabiximols oromucosal spray as an adjunctive therapy in advanced cancer patients with chronic uncontrolled pain. J Pain Symptom Manag. 2018;55(2):179–88.
• Pergam SA, Woodfield MC, Lee CM, Cheng GS, Baker KK, Marquis SR, et al. Cannabis use among patients at a comprehensive cancer center in a state with legalized medicinal and recreational use. Cancer. 2017;123(22):4488–97 Excellent overview of cannabis use by cancer patients at a comprehensive cancer center in a state where recreational cannabis is available.
• Schleider LB, Mechoulam R, Lederman V, Hilou M, Lencovsky O, Betzalel O, et al. Prospective analysis of safety and efficacy of medical cannabis in large unselected population of patients with cancer. Eur J Intern Med. 2018;1(49):37–43 Information obtained from 2000 Israeli cancer patients using government licensed cannabis for symptom management.
•• https://www.canada.ca/en/health-canada/services/drugs-medication/cannabis/information-medical-practitioners/information-health-care-professionals-cannabis-cannabinoids.html. Accessed 24 May 1999. An updated comprehensive review of cannabis as medicine.
Meiri E, Jhangiani H, Vredenburgh JJ, Barbato LM, Carter FJ, Yang HM, et al. Efficacy of dronabinol alone and in combination with ondansetron versus ondansetron alone for delayed chemotherapy-induced nausea and vomiting. Curr Med Res Opin. 2007;23(3):533–43.
Duran M, Pérez E, Abanades S, Vidal X, Saura C, Majem M, et al. Preliminary efficacy and safety of an oromucosal standardized cannabis extract in chemotherapy-induced nausea and vomiting. Br J Clin Pharmacol. 2010;70(5):656–63.
Wilkie G, Sakr B, Rizack T. Medical marijuana use in oncology: a review. JAMA Oncol. 2016;2(5):670–5.
Turgeman I, Bar-Sela G. Cannabis use in palliative oncology: a review of the evidence for popular indications. In: International Conference on Medical Cannabis 2016
• Turgeman I, Bar-Sela G. Cannabis for cancer–illusion or the tip of an iceberg: a review of the evidence for the use of Cannabis and synthetic cannabinoids in oncology. Expert Opin Investig Drugs. 20 A comprehensive review of cannabis in cancer care.
• Abrams DI. The therapeutic effects of cannabis and cannabinoids: an update from the National Academies of Sciences, Engineering and Medicine report. Eur J Intern Med. 2018;49:7–11 A concise summary of the main findings with regard to the therapeutic effects of cannabis as summarized in the National Academies of Sciences, Engineering and Medicine’s report.
Hesketh PJ, Kris MG, Basch E, Bohlke K, Barbour SY, Clark-Snow RA, et al. Antiemetics: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2017;35(28):3240–61.
Busquets-Garcia A, Bains J, Marsicano G. CB1 receptor signaling in the brain: extracting specificity from ubiquity. Neuropsychopharmacology. 2017;43(1):4–20.
Malfitano AM, Basu S, Maresz K, Bifulco M, Dittel BN. What we know and do not know about the cannabinoid receptor 2 (CB2). Semin Immunol. 2014;26(5):369–79.
Foltin RW, Fischman MW, Byrne MF. Effects of smoked marijuana on food intake and body weight of humans living in a residential laboratory. Appetite. 1988;11(1):1–14.
Turcott JG, Núñez MD, Flores-Estrada D, Oñate-Ocaña LF, Zatarain-Barrón ZL, Barrón F, et al. The effect of nabilone on appetite, nutritional status, and quality of life in lung cancer patients: a randomized, double-blind clinical trial. Support Care Cancer. 2018;26(9):3029–38.
Mechoulam R, Peters M, Murillo-Rodriguez E, Hanuš LO. Cannabidiol–recent advances. Chem Biodivers. 2007;4(8):1678–92.
Millar SA, Stone NL, Yates AS, O'Sullivan SE. A systematic review on the pharmacokinetics of cannabidiol in humans. Front Pharmacol. 2018;9:1365.
Boggs DL, Nguyen JD, Morgenson D, Taffe MA, Ranganathan M. Clinical and preclinical evidence for functional interactions of cannabidiol and Δ 9-tetrahydrocannabinol. Neuropsychopharmacology. 2018;43(1):142–54.
Brunt TM, van Genugten M, Höner-Snoeken K, van de Velde MJ, Niesink RJ. Therapeutic satisfaction and subjective effects of different strains of pharmaceutical-grade cannabis. J Clin Psychopharmacol. 2014;34(3):344–9.
Hayakawa K, Mishima K, Hazekawa M, Sano K, Irie K, Orito K, et al. Cannabidiol potentiates pharmacological effects of Δ9-tetrahydrocannabinol via CB1 receptor-dependent mechanism. Brain Res. 2008;1188:157–64.
•• Vučković S, Srebro D, Vujović KS, Vučetić Č, Prostran M. Cannabinoids and pain: new insights from old molecules. Front Pharmacol. 2018;9:1259. An excellent summary and review of the use of cannabis in pain.
Nugent SM, Morasco BJ, O'Neil ME, Freeman M, Low A, Kondo K, et al. The effects of cannabis among adults with chronic pain and an overview of general harms: a systematic review. Ann Intern Med. 2017;167(5):319–31.
Häuser W, Petzke F, Fitzcharles MA. Efficacy, tolerability and safety of cannabis-based medicines for chronic pain management–an overview of systematic reviews. Eur J Pain. 2018 Mar;22(3):455–70.
Aviram J, Samuelly-Leichtag G Efficacy of cannabis-based medicines for pain management: a systematic review and meta-analysis of randomized controlled trials. Pain Physician. 2017;20(6):E755–E796
Abrams DI, Jay CA, Shade SB, Vizoso H, Reda H, Press S, et al. Cannabis in painful HIV-associated sensory neuropathy: a randomized placebo-controlled trial. Neurology. 2007;68(7):515–21.
Donvito G, Nass SR, Wilkerson JL, Curry ZA, Schurman LD, Kinsey SG, et al. The endogenous cannabinoid system: a budding source of targets for treating inflammatory and neuropathic pain. Neuropsychopharmacology. 2018;43(1):52–79.
Andreae MH, Carter GM, Shaparin N, Suslov K, Ellis RJ, Ware MA, et al. Inhaled cannabis for chronic neuropathic pain: a meta-analysis of individual patient data. J Pain. 2015;16(12):1221–32.
Lynch ME, Cesar-Rittenberg P, Hohmann AG. A double-blind, placebo-controlled, crossover pilot trial with extension using an oral mucosal cannabinoid extract for treatment of chemotherapy-induced neuropathic pain. J Pain Symptom Manag. 2014;47(1):166–73.
Rahn EJ, Makriyannis A, Hohmann AG. Activation of cannabinoid CB1 and CB2 receptors suppresses neuropathic nociception evoked by the chemotherapeutic agent vincristine in rats. Br J Pharmacol. 2007;152(5):765–77.
Khasabova IA, Khasabov S, Paz J, Harding-Rose C, Simone DA, Seybold VS. Cannabinoid type-1 receptor reduces pain and neurotoxicity produced by chemotherapy. J Neurosci. 2012;32(20):7091–101.
Ward SJ, McAllister SD, Kawamura R, Murase R, Neelakantan H, Walker EA. Cannabidiol inhibits paclitaxel-induced neuropathic pain through 5-HT1A receptors without diminishing nervous system function or chemotherapy efficacy. Br J Pharmacol. 2014;171(3):636–45.
Abrams DI, Couey P, Shade SB, Kelly ME, Benowitz NL. Cannabinoid–opioid interaction in chronic pain. Clin Pharmacol Ther. 2011;90(6):844–51.
Bradford AC, Bradford WD. Medical marijuana laws reduce prescription medication use in Medicare Part D. Health Aff. 2016;35(7):1230–6.
Bachhuber MA, Saloner B, Cunningham CO, Barry CL. Medical cannabis laws and opioid analgesic overdose mortality in the United States, 1999-2010. JAMA Intern Med. 2014;174(10):1668–73.
• MacCallum CA, Russo EB. Practical considerations in medical cannabis administration and dosing. Eur J Intern Med. 2018;49:12–9. A nice review of the different effects of inhaled versus ingested cannabis and other plant-related issues.
Russo EB. Cannabidiol claims and misconceptions. Trends Pharmacol Sci. 2017;38(3):198–201.
Volkow ND, Baler RD, Compton WM, Weiss SR. Adverse health effects of marijuana use. N Engl J Med. 2014;370(23):2219–27.
Shannon S, Lewis N, Lee H, Hughes S. Cannabidiol in anxiety and sleep: a large case series. Perm J. 2019;23:18–041.
Bergamaschi MM, Queiroz RH, Chagas MH, De Oliveira DC, De Martinis BS, Kapczinski F, et al. Cannabidiol reduces the anxiety induced by simulated public speaking in treatment-naive social phobia patients. Neuropsychopharmacology. 2011;36(6):1219–26.
Munson AE, Harris LS, Friedman MA, Dewey WL, Carchman RA. Antineoplastic activity of cannabinoids. J Natl Cancer Inst. 1975;55:597–602.
Guzman M. Cannabinoids: potential anticancer agents. Nat Rev Cancer. 2003;3(10):745–55.
Velasco G, Sánchez C, Guzmán M. Towards the use of cannabinoids as antitumour agents. Nat Rev Cancer. 2012;12(6):436–44.
Massi P, Solinas M, Cinquina V, Parolaro D. Cannabidiol as potential anticancer drug. Br J Clin Pharmacol. 2013;75(2):303–12.
Dumitru CA, Sandalcioglu E, Karsak M. Cannabinoids in glioblastoma therapy: new applications for old drugs. Front Mol Neurosci. 2018;11:159.
Rocha FC, dos Santos Júnior JG, Stefano SC, da Silveira DX. Systematic review of the literature on clinical and experimental trials on the antitumor effects of cannabinoids in gliomas. J Neuro-Oncol. 2014;116(1):11–24.
McAllister SD, Christian RT, Horowitz MP, Garcia A, Desprez PY. Cannabidiol as a novel inhibitor of Id-1 gene expression in aggressive breast cancer cells. Mol Cancer Ther. 2007;6(11):2921–7.
Cridge BJ, Rosengren RJ. Critical appraisal of the potential use of cannabinoids in cancer management. Cancer Manag Res. 2013;5:301.
Raup-Konsavage WM, Johnson M, Legare CA, Yochum GS, Morgan DJ, Vrana KE. Synthetic cannabinoid activity against colorectal cancer cells. Cannabis Cannabinoid Res. 2018;3(1):272–81.
Sharafi G, He H, Nikfarjam M. Potential use of cannabinoids for the treatment of pancreatic cancer. J Pancreat Cancer. 2019;5(1):1–7.
Guzman M, Duarte MJ, Blazquez C, Ravina J, Rosa MC, Galve-Roperh I, et al. A pilot clinical study of Δ 9-tetrahydrocannabinol in patients with recurrent glioblastoma multiforme. Br J Cancer. 2006;95(2):197–203.
https://www.gwpharm.com/about-us/news/gw-pharmaceuticals-achieves-positive-results-phase-2-proof-concept-study-glioma. Accessed 24 May 2019.
• Ladin DA, Soliman E, Griffin L, Van Dross R. Preclinical and clinical assessment of cannabinoids as anti-cancer agents. Front Pharmacol. 2016;7:361 Summary of the expression of cannabinoid receptors in various tumor types that may have implications for future treatment protocols.
Cathcart P, de Giorgio A, Stebbing J. Cannabis and cancer: reality or pipe dream? Lancet Oncol. 2015;16(13):1291–2.
Yamaori S, Okamoto Y, Yamamoto I, Watanabe K. Cannabidiol, a major phytocannabinoid, as a potent atypical inhibitor for CYP2D6. Drug Metab Dispos. 2011;39(11):2049–56.
Wallace JM, Lim R, Browdy BL, Hopewell PC, Glassroth J, Rosen MJ, et al. Pulmonary Complications of HIV Infection Study Group. Risk factors and outcomes associated with identification of aspergillus in respiratory specimens from persons with HIV disease. Chest. 1998;114(1):131–7.
Gurney J, Shaw C, Stanley J, Signal V, Sarfati D. Cannabis exposure and risk of testicular cancer: a systematic review and meta-analysis. BMC Cancer. 2015;15(1):897.
Sontineni SP, Chaudhary S, Sontineni V, Lanspa SJ. Cannabinoid hyperemesis syndrome: clinical diagnosis of an underrecognised manifestation of chronic cannabis abuse. World J Gastroenterol: WJG. 2009;15(10):1264.
Engels FK, De Jong FA, Sparreboom A, Mathot RA, Loos WJ, Kitzen JJ, et al. Medicinal cannabis does not influence the clinical pharmacokinetics of irinotecan and docetaxel. Oncologist. 2007;12(3):291–300.
Taha T, Meiri D, Talhamy S, Wollner M, Peer A, Bar-Sela G. Cannabis impacts tumor response rate to nivolumab in patients with advanced malignancies. Oncologist. 2019;theoncologist-2018.18 31:1–2.
• Braun IM, Wright, Peteet J, Meyer FL, Yuppa DP, Bolcic-Jankovic D, et al. Medical oncologists’ beliefs, practices and knowledge regarding marijuana used therapeutically: a nationally representative survey study. J Clin Oncol. 2018;36:1957–62 Results of a survey of oncologists knowledge, attitudes and beliefs regarding cannabis in cancer patients and a call for more education on the topic.
Zylla D, Steele G, Eklund J, Mettner J, Arneson T. Oncology Clinicians and the Minnesota Medical Cannabis Program: a survey on medical Cannabis practice patterns, barriers to enrollment, and educational needs. Cannabis Cannabinoid Res. 2018;3(1):195–202.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Donald I. Abrams has received compensation from AXIM Biotechnologies, Insys Therapeutics, Intec, Maui Grown Therapies, Scriptyx, Spectrum, Tikun Olam, and VIVO Cannabis for service as a consultant.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Palliative and Supportive Care
Rights and permissions
About this article
Cite this article
Abrams, D.I. Should Oncologists Recommend Cannabis?. Curr. Treat. Options in Oncol. 20, 59 (2019). https://doi.org/10.1007/s11864-019-0659-9
Published:
DOI: https://doi.org/10.1007/s11864-019-0659-9