Abstract
Background
Vasovagal syncope (VVS) is a heterogeneous disorder that creates challenges for treatment. Metoprolol is an important therapeutic option for children with VVS.
Aims
The study examined the predictive value of 24-h urine norepinephrine (NE) levels in the assessment of the therapeutic efficacy of metoprolol for recurrent VVS in children.
Methods
Thirty-eight children with recurrent VVS and 20 healthy children were enrolled in our study. Twenty-four-hour urine NE levels were measured by LC-MS-MS. VVS children were diagnosed by BHUTT and/or SNHUTT, and received metoprolol treatment for 3 months. Symptom scoring was utilized to evaluate the therapeutic effect. A ROC curve was used to investigate the predictive value of 24-h urine norepinephrine levels.
Results
There exists significant correlation between 24-h urine NE levels and supine systolic and diastolic blood pressures. The 24-h urine NE levels of responders (40.75 ± 12.86 μg/24 h) were higher than those of nonresponders (21.48 ± 6.49 μg/24 h), and there was a significant difference between the two groups (P < 0.001). A ROC curve of the predictive value of 24 h urine NE levels revealed that the area under the curve was 0.926. A cutoff value for 24-h urine NE level of 34.84 μg/24 h produced both high sensitivity (70%) and specificity (100%) in predicting the efficacy of metoprolol therapy for VVS.
Conclusions
Patients with high 24-h urine NE levels have higher supine systolic and diastolic pressures and more effective responses to metoprolol. A 24-h urine norepinephrine level of > 34.84 μg/24 h was an indicator of the effectiveness of metoprolol therapy for VVS in children.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Vasovagal syncope (VVS) is a self-limiting syncope that is mediated by the autonomic nervous system, and it is triggered by multiple factors. Abnormalities exist in some physiological regulatory mechanisms, which create an imbalance in the coordination of heart rate, blood pressure, and blood volume, which cause vasodilation, hypotension, and bradycardia. Therefore, VVS is a functional disease. Currently, the pathogenesis of VVS is believed to involve decreased venous reflux, high catecholamine levels, imbalance of autonomic nervous function, and an abnormal Bezold-Jarish reflex [1]. However, the specific pathogenesis of VVS is not completely elucidated, and it varies across different types of patients. Numerous experimental and clinical studies have shown that many central and peripheral neurotransmitters and vasoactive molecules, such as catecholamine, 5-hydroxytryptamine, H2S, NO, CT-proAVP, and MR-proANP, play important roles in the occurrence of VVS [2,3,4,5,6].
Recurrent VVS syncope can seriously affect the quality of life and mental health of patients [7, 8]. Recently, increasing attention has been paid to the treatment of vasovagal syncope, especially drug therapy [9]. Catecholamine directly reflects autonomic nervous activity, and patients with VVS have increased presyncope adrenaline and norepinephrine secretion. Some VVS children may have increased basal catecholamine, and high catecholamine levels may be an important factor in the occurrence of VVS [10, 11]. Beta-adrenoceptor blockers (β-blockers) can block β1-receptor activity and have a negative inotropic effect on the heart. This activity reduces the stimulation of the mechanical receptors on the posterior inferior ventricular wall, which reduces the intensity of the Bezold-Jarisch reflection. Blockade of β-receptor activity can antagonize the high catecholamine levels in patients with vasovagal syncope and reduce peripheral venous congestion. Antagonism also minimizes the possibility of excessive and intense ventricular contraction [1, 12]. Based on these theoretical hypotheses and a large number of clinical studies, β-blocker agents have become common drugs in VVS treatment. Unfortunately, previous studies showed diverse effect of β-blocker on VVS [13,14,15]. Differences in noradrenergic activity between hyperadrenergic and other types of VVS may explain this inconsistency. Norepinephrine (NE) is primarily released from adrenergic nerve terminals, and it can reflect the status of the sympathetic nerve activity. NE is excreted partly in the form of a prototype through urination, and detection of 24-h urine NE can reflect sympathetic nerve activity. Therefore, we hypothesized that 24-h urine NE levels would help guide the management of β-blockers in VVS children.
Materials and methods
Subjects
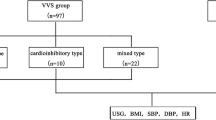
Thirty-eight children were diagnosed with VVS in the Department of Pediatrics, Qilu Hospital of Shandong University. The diagnostic process was performed in accordance with the Guidelines for the Diagnosis of Syncope (2016 Revised Edition) [16]. Twenty healthy children without syncope or presyncope syndromes served as controls. All patients had a full history recorded and underwent a physical examination, echocardiography, electroencephalography, chest X-ray, and serum testing for biochemistry to exclude diseases related to the central nervous, cardiovascular, and metabolic systems. Then, the VVS group children underwent baseline head-up tilt test (BHUTT) and/or sublingual nitroglycerin-provocative head-up tilt test (SNHUTT). This study was approved by the ethics committee of Qilu Hospital of Shandong University, and all participants’ guardians were fully informed of the purpose and methods of the study.
Protocols for BHUTT and SNHUTT
BHUTT [17]
The protocol for BHUTT is as follows. For each subject, informed parental consent was obtained. Drugs that could affect normal autonomic nervous system function were avoided for at least 3 days before the test, and patients fasted for at least 4 h before the head-up tilt test (HUTT). The tests were performed in a quiet, softly lit, temperature-controlled room equipped with medical resuscitation facilities. Patients were secured to a tilt table and monitored for 10 min in a supine position. Supine systolic BP, supine diastolic BP, and heart rate were recorded. Patients were then tilted upward at a 60° angle until a positive reaction occurred or until 45 min elapsed. Electrocardiograms were continuously recorded, and blood pressure and heart rate were monitored every 5 min by an automated sphygmomanometer (GE DASH 2000). Children were placed in a supine position as soon as the positive response occurred.
SNHUTT [18]
The BHUTT-negative children remained upright on the tilted bed at 60° and received sublingual nitroglycerin (4–6 μg/kg). The maximum dose administered was not greater than 300 μg. Children were observed for 20 min or until a positive response occurred. Electrocardiograms were continuously recorded, and blood pressure and heart rate were monitored after medication administration.
Collection of 24-h urine and the norepinephrine test
Twenty-four-hour urine samples were collected from all 38 VVS children after diagnosis. Twenty-four-hour urine samples were also collected from the 20 children in the control group. Children were asked to engage in usual activities during urine sample collection. Urine was excreted at 7 a.m., and all urine after 7 a.m. was collected in a large container (a clean spittoon or a wide mouth bottle) until 7 a.m. the following day. The specimens were mixed, and the total urine volume was measured using a graduated glass container. Then, 3–5 ml urine was removed to a clean dry tube and tested as soon as possible. Concentrated hydrochloric acid (0.5–1 ml/100 ml urine) was added to reduce degradation of 24-h urine. NE levels in 24-h urine were measured by liquid chromatography-tandem mass spectrometry (LC-MS-MS) [19].
Diagnostic Criteria
A positive response was characterized as syncope or severe presyncope during the HUT test, accompanied with any of the following responses [16, 20]: (1) systolic BP (SBP) ≤ 80 mmHg (1 mmHg = 0.133 kPa) or diastolic BP (DBP) ≤ 50 mmHg or mean pressure decrease ≥ 25%; (2) HR < 75 beats/min in 4–6-year-old children, HR < 65 beats/min for 7–8-year-old children, HR < 60 beats/min for children older than 8 years; (3) electrocardiogram showing sinus arrest and premature junctional contractions; and (4) atrioventricular block and cardiac arrest ≥ 3 s. The responses were classified as cardioinhibitory, vasoinhibitory, or mixed inhibitory. VVS-VI type was characterized as a significant BP decrease without an obvious HR reduction. VVS-CI type was characterized as a marked HR decrease without a marked decrease in systolic pressure. VVS-MI type was characterized as a decreased HR and BP.
Treatment and follow-up protocols
All 38 VVS children were prescribed metoprolol and a conventional therapeutic regimen, including increased intake of water and salt [21, 22], maneuvers to prevent blood from pooling in the lower extremities (e.g., leg crossing) and to abort the episode (e.g., supine posture) [23], autonomic nervous function exercise [17], psychological intervention [24], avoidance of predisposing situations, and reassurance of the non-life-threatening nature of the condition [9, 17]. The initial dosage of metoprolol was 0.25 mg/kg twice daily [16]. Follow-up was conducted for 3 months after therapy via telephone and outpatient follow-up. The patients and their parents were required to report any changes in symptoms and drug intolerance.
Criteria for evaluating the therapeutic effects
Symptom scoring was utilized to evaluate the therapeutic effect of metoprolol [25]. Scoring was based on the typical symptoms of VVS, including syncope, dizziness, nausea, palpitation, headache, tremulousness, chest tightness, and blurred vision, and graded from 0 to 4 according to the frequency of orthostatic intolerance (OI) symptoms. A score of 0 indicated no symptom occurrence during follow-up. A score of 1 indicated one episode of an OI symptom per month. A score of 2 indicated two to four episodes of symptom occurrence per month. A score of 3 indicated two to seven episodes of symptom occurrence per week, and a score of 4 indicated more than once daily symptom occurrence. The total score for each participant was the sum of all the scores derived from his or her symptoms. Symptom scoring was performed for each participant both at the beginning of the study and after 3 months of metoprolol therapy. Patients responded to the symptom queries, and parents provided the necessary information when needed. The scores were recorded at clinical visits. Therapy was deemed to be effective when the symptoms of OI disappeared or symptom scores decreased to less than 50%.
Statistical analysis
Statistical analysis was completed by SPSS software version 19.0 (SPSS, Chicago, Illinois). Data are presented as the means ± SD. Comparisons of age, height, body weight, supine SBP, supine DBP, supine heart rate, and 24-h urine NE levels were performed using independent Student’s t test. Comparisons of sex (male/female ratio) between groups were performed using Pearson’s chi-square test. A P value < 0.05 was considered statistically significant. The relationships between supine SBP, supine DBP, and supine heart rate with 24-h urine NE levels in children with VVS were analyzed using the Pearson correlation test. A receiver-operating characteristic (ROC) curve was utilized to evaluate the predictive value of 24-h urine NE level in assessing the therapeutic responses of metoprolol. The area under the curve (AUC) represents the predictive value. An AUC from 0.5 to 0.7 indicated a low predictive value, an AUC from 0.7 to 0.9 indicated a moderate predictive value, and an AUC > 0.9 indicated a high predictive value. The 95% confidence interval (CI) of AUC did not contain 0.5, or a P value < 0.05 confirmed that 24-h urine NE level was a reliable predictor of the therapeutic effect of metoprolol in children with VVS. The optimal cutoff value was determined as the maximum of the Youden index, which is defined as sensitivity plus specificity minus 1, where sensitivity and specificity were calculated as proportions.
Results
Demographics of the VVS and control groups
The mean age of the 38 VVS patients was 9.88 ± 1.92 years and ranged from 6 to 13 years. Females were predominant (females/males 20:18). The mean BMI of the 38 VVS patients was 17.78 ± 4.03. Twenty-six VVS children exhibited a vasodepressor pattern, and 12 children exhibited a mixed pattern. The mean age of the 20 control group children was 9.92 ± 1.96 years and ranged from 7 to 13 years. The mean BMI was 17.06 ± 4.71.
Comparison of the demographics between the VVS and control groups
The general characteristics of the groups are shown in Table 1. Age, sex, ratio, and BMI were not significantly different between groups. The supine SBP (98.79 ± 7.64 mmHg) and DBP (57.42 ± 4.85 mmHg) of the VVS group were lower than those of the control group (SBP 104.70 ± 5.60 mmHg and DBP 64.80 ± 6.26 mmHg). The supine heart rate of the VVS group (75 ± 11 bpm) was similar to that of the control group (76 ± 9 bpm). Significant differences in supine systolic and diastolic pressure were observed between the two groups (P < 0.01), but no significant difference in heart rate was observed (P > 0.05).
Comparisons of 24-h urine NE levels between the VVS group and control group
We analyzed the 24-h urine NE values of the 38 children with VVS. The mean 24-h urine NE level was 31.62 ± 14.11 μg/24 h, and the dispersion coefficient (R2) was 0.0028, which is very high. The higher R2 indicates that the individual differences between VVS children were obvious and suggest that this indicator may have a predictive value for drug treatment choice. The mean 24-h urine NE level of the control group was 35.04 ± 7.28 μg/24 h, and the R2 was 0.0002, which is lower than that of the VVS group and highlight the higher dispersion of VVS group.
Correlation analysis between 24-h urine NE level and supine blood pressure and heart rate in children with recurrent VVS
The results showed that patients with a high level of 24-h urine NE have higher supine SBP and DBP. Pearson correlation analyses revealed a significant positive correlation between 24-h urine NE level and supine SBP (r = 0.604; P < 0.001) and supine DBP (r = 0.400; P < 0.05). No significant correlation was observed with supine heart rate (r = 0.049; P > 0.05) (Fig. 1).
Scatter plots and trend lines of supine systolic pressure, diastolic pressure, heart rate, and 24-h urine NE levels of VVS children, n = 38. a Pearson correlation analyses revealed significant positive correlations between 24-h urine NE level and supine systolic BP (r = 0.604; P < 0.001). b Pearson correlation analyses revealed significant positive correlations between 24-h urine NE level and supine diastolic BP (r = 0.400; P < 0.05). c No significant correlation was observed between 24-h urine NE level and supine heart rate (r = 0.049; P > 0.05)
Comparison of baseline characteristics in vasodepressor type and mixed type
The general characteristics, including age, sex ratio, BMI, 24-h urine NE level, supine blood pressure, supine heart rate, and symptom score before treatment, are shown in Table 2. There was no statistically significant difference between the two types (P > 0.05).
Comparison of baseline characteristics between responders and nonresponders
Of the 38 patients with VVS who received metoprolol treatment, 20 had effective responses, with symptom disappearance or a symptom score decrease greater than 50%. The pretreatment data, such as age, sex, BMI, and pretreatment symptom scores, did not differ between reponders and nonresponders (P > 0.05). The mean supine systolic pressure of responders was 102.65 ± 7.50 mmHg, the mean supine diastolic pressure was 59.60 ± 4.77 mmHg, and the mean supine heart rate was 77 ± 11 bpm. The mean supine systolic pressure of nonresponders was 95.22 ± 7.23 mmHg, the mean supine diastolic pressure was 55.17 ± 3.88 mmHg, and the mean supine heart rate was 73 ± 12 bpm. Significant differences in supine systolic and diastolic pressures were observed between groups (P < 0.01), but no significant difference in supine heart rate was observed (P > 0.05). The 24-h urine NE level of responders before treatment was 40.75 ± 12.86 μg/24 h and 21.48 ± 6.49 μg/24 h in nonresponders. The difference between the two groups was significant (P < 0.001). The pretreatment symptom score of responders was 5.65 ± 2.03, and the score of nonresponders was 4.94 ± 1.89. The difference between the two was not obvious (P > 0.05). The score of responders after therapy was 1.35 ± 0.93, and the score of nonresponders was 4.28 ± 1.36. There were significant differences between the two groups (P < 0.001) (Table 3).
The prediction of the effect on metoprolol with ROC curve
The ROC curve of 24-h urine NE for predicting the therapeutic effect of metoprolol revealed an AUC of 0.926 (95%CI 0.848 to 1, p < 0.01). A cutoff value for 24-h urine NE of 34.84 μg/24 h yielded high sensitivity (70%) and specificity (100%) (Fig. 2).
ROC curve of the predictive value of 24-h urine NE level for distinguishing responders and nonresponders to metoprolol treatment. The y-axis represents the sensitivity to predict the therapeutic response to metoprolol treatment in VVS children. The x-axis represents the false positive rate (1-specificity) of prediction. The green line in the graph is the reference line, which indicates the sensitivity being equal to the false positive rate. The blue curve is farther from the green line and nearer to the upper left corner of the graph. The area under the curve is 0.926 (95% confidence interval 0.848 to 1, P < 0.01)
Discussion
This study found significant differences in 24-h urine NE levels among children with recurrent VVS, which indicates that not all VVS children have a high catecholamine status. Basal NE levels were distinctly different, which provides a possible theoretical basis for the choice of metoprolol. In our research, we also found that there was no difference in the means ± SD of the 24-h urine NE levels in children between control group and VVS group. To analyze this result in depth, we can indicate that there exist differences of hyperadrenergic status and hypoadrenergic status in VVS children relatively, and this may be one of the reasons for the diverse results in the therapeutic effect of β-blockers, while 24-h urine NE levels in healthy children are relatively stable in their daily life. Moreover, there is no difference in means ± SD of the 24-h urine NE levels in the children with VVS between vasodepressor VVS and mixed type VVS. This result indicates that there exist similarities between the two types of VVS. During the process of HUTT, when to place the patients in a supine position depends on the operator’s judging, the time point at which to place down usually determines the types of VVS. Vaddadi et al. found that patients with recurrent postural vasovagal syncope might have sympathetic nervous system phenotypes [26], and they can be divided into a low-pressure phenotype (systolic blood pressure < 100 mmHg) and a normal-pressure phenotype (systolic blood pressure > 100 mmHg). NE spillover in the low-pressure phenotype was associated with low tyrosine hydroxylase levels, which probably reduced NE synthesis. Our research further confirmed this outcome and found that patients with a high level of 24-h urine NE have higher supine systolic and diastolic pressure, but no significant correlation with supine heart rate. The responses of VVS children with different 24-h urine NE levels to metoprolol differed significantly, and children with high levels of 24-h urine NE responded more effectively. Patients with low levels of 24-h urine NE did not respond as effectively, and some children may even have symptom aggravations. Our results indicate that 24-h urine NE level > 34.84 μg is an indicator of metoprolol effectiveness in children with recurrent VVS.
The difference in clinical manifestations between VVS and other types of syncope is that most children have presyncope symptoms [27], such as dizziness, headache, chest tightness, shortness of breath, palpitation, pale, and blurred vision. These symptoms may be the only complaint in some VVS patients without a syncope episode. VVS does not increase the prevalence or mortality of cardiovascular diseases, and patients usually have a good prognosis. However, the recurrent symptoms create physical and psychological stresses in children’s daily lives, and quality of life is seriously affected. Therefore, it is very necessary to provide appropriate treatment and preventive measures in VVS children.
Beta blockers are commonly used in the treatment of vasovagal syncope. Biffi et al. [28] studied 20 patients with recurrent vasovagal syncope in a double-blind randomized controlled trial and revealed that the efficacy of metoprolol was better than that of the α2-adrenergic receptor agonist clonidine. Klingenheben et al. involved 30 recurrent VVS patients and found that the efficiency of a single use of metoprolol reached 53% [29]. When metoprolol was ineffective, the combined therapy of the α1-adrenergic receptor agonist midodrine was used, and the efficiency was up to 77%. Blockade of beta receptors antagonizes high-level catecholamine in patients with vasovagal syncope, alleviates peripheral venous stasis, and reduces the possibility of excessive ventricular contraction. However, some statistical data also indicate that beta blockers are not always effective. Sheldon et al. [13] explored the preventive effect of metoprolol in adult VVS patients using a double-blinded, randomized, placebo-controlled design. A total of 208 cases (42 ± 18 years) of VVS were treated with metoprolol (n = 108, 25~200 mg/day) and placebo (n = 100). The results demonstrated that there was no significant difference between the two groups in preventing syncope. Based on our research, we believe that this situation is closely related to the lack of effective biological reference indicators.
The junctional transmitter between sympathetic ganglion cells and effectors is norepinephrine, and NE is partly excreted in the urine in the form of a prototype [30]. Therefore, 24-h urine NE levels reflect the functional state of the sympathetic nervous system. Based on this theory, we hypothesized that 24-h urine NE levels can be used as a predictive marker for the efficacy of beta blockers in the treatment of VVS, and the corresponding research was carried out. The study found that metoprolol was more effective in treating VVS children with higher 24-h urinary NE levels. To examine the possible predictive value of 24-h urine NE levels, we used ROC analysis and showed that 24-h urine NE levels exhibited a sensitivity of 70% and specificity of 100% in predicting of metoprolol efficacy for the treatment of VVS. Therefore, 24-h urine NE levels could be used as a reference index in choosing medication for children with VVS. For children with orthostatic intolerance, 24-h urine samples are routinely collected to detect 24-h urine Na+ content, which is used as a reference index for the treatment of ORS [31]. Therefore, 24-h urine specimens are easy to obtain, and it is noninvasive. Basal catecholamine status can be assessed by this test, and the effectiveness of metoprolol treatment can be predicted. Our study suggests that this test has a positive predictive value for the treatment of VVS. The samples are convenient to collect, and this method is worth popularizing.
The present study also has limitations. The age range of our patients was relatively concentrated. The small sample size might lead to bias. We also lacked follow-up data of 24-h urine NE after treatment. Further research is needed to increase the number of cases and perform a multicenter joint study, whether 24-h urine NE reduction is more suitable for the application of midodrine is also worth further research.
Conclusions
Our study demonstrated a significant difference in 24-h urine NE levels between children with recurrent VVS. Patients with high levels of 24-h urine NE have higher supine systolic and diastolic pressure and responded more effectively to metoprolol. This finding is of great clinical significance in the selection of metoprolol for VVS children.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- NE:
-
norepinephrine
- VVS:
-
vasovagal syncope
- LC-MS-MS:
-
liquid chromatography-tandem mass spectrometry
- BHUTT:
-
baseline head-up tilt test
- SNHUTT:
-
sublingual nitroglycerin-provocative head-up tilt test
- ROC:
-
receiver-operating characteristic
- HR:
-
heart rate
- BP:
-
blood pressure
- SBP:
-
systolic blood pressure
- DBP:
-
diastolic blood pressure
- OI:
-
orthostatic intolerance
- AUC:
-
area under the curve
- CI:
-
confidence interval
- ORS:
-
oral rehydration salts
References
Grubb BP (2005) Clinical practice. Neurocardiogenic syncope. N Engl J Med 352(10):1004–1010. https://doi.org/10.1056/NEJMcp042601
Brignole M, Alboni P, Benditt D et al (2001) Guidelines on management (diagnosis and treatment) of syncope. Eur Heart J 22(15):1256–1306. https://doi.org/10.1053/euhj.2001.2739
Nair N, Padder FA, Kantharia BK (2003) Pathophysiology and management of neurocardiogenic syncope. Am J Manag Care 9(4):327–334 quiz 335-326
Shi Y, Tian H, Gui YH et al (2008) Association of nitric oxide and eNOS with the pathogenesis of vasovagal syncope. Zhongguo Dang Dai Er Ke Za Zhi 10(4):478–480
Hamrefors V, Spahic JM, Nilsson D, Senneby M, Sutton R, Melander O, Fedorowski A (2017) Syndromes of orthostatic intolerance and syncope in young adults. Open Heart 4(1):e000585. https://doi.org/10.1136/openhrt-2016-000585
Zhang F, Li X, Stella C, Chen L, Liao Y, Tang C, Jin H, du J (2012) Plasma hydrogen sulfide in differential diagnosis between vasovagal syncope and postural orthostatic tachycardia syndrome in children. J Pediatr 160(2):227–231. https://doi.org/10.1016/j.jpeds.2011.08.008
Varga E, Worum F, Szabo Z et al (2002) Motor vehicle accident with complete loss of consciousness due to vasovagal syncope. Forensic Sci Int 130(2–3):156–159
Chen L, Li X, Todd O, Wang C, Jin H, du J (2014) A clinical manifestation-based prediction of haemodynamic patterns of orthostatic intolerance in children: a multi-centre study. Cardiol Young 24(4):649–653. https://doi.org/10.1017/S1047951113000929
Shen WK, Sheldon RS, Benditt DG, Cohen MI, Forman DE, Goldberger ZD, Grubb BP, Hamdan MH, Krahn AD, Link MS, Olshansky B, Raj SR, Sandhu RK, Sorajja D, Sun BC, Yancy CW (2017) 2017 ACC/AHA/HRS guideline for the evaluation and Management of Patients with Syncope: a report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines and the Heart Rhythm Society. Circulation 136(5):e60–e122. https://doi.org/10.1161/CIR.0000000000000499
Chosy JJ, Graham DT (1965) Catecholamines in vasovagal fainting. J Psychosom Res 9(2):189–194
Benditt DG, Ermis C, Padanilam B et al (2003) Catecholamine response during haemodynamically stable upright posture in individuals with and without tilt-table induced vasovagal syncope. Europace 5(1):65–70
Parry SW, Kenny RA (1999) The management of vasovagal syncope. QJM 92(12):697–705
Sheldon R, Connolly S, Rose S, Klingenheben T, Krahn A, Morillo C, Talajic M, Ku T, Fouad-Tarazi F, Ritchie D, Koshman ML (2006) Prevention of Syncope trial (POST): a randomized, placebo-controlled study of metoprolol in the prevention of vasovagal syncope. Circulation 113(9):1164–1170. https://doi.org/10.1161/CIRCULATIONAHA.105.535161
Ventura R, Maas R, Zeidler D et al (2002) A randomized and controlled pilot trial of beta-blockers for the treatment of recurrent syncope in patients with a positive or negative response to head-up tilt test. Pacing Clin Electrophysiol 25(5):816–821
Sheldon R, Rose S, Connolly S (2003) Prevention of Syncope Trial (POST): a randomized clinical trial of beta blockers in the prevention of vasovagal syncope; rationale and study design. Europace 5(1):71–75
Chinese Pediatric Cardiology Society (2016) (CPCS),Editorial Committee of Chinese Journal of Pediatrics,Pediatric cardiology society, Beijing pediatric society, Beijing medical association(BMA),committee on pediatric Syncope, pediatricians branch, Chinese medical doctor association (CMDA). Guidelines for the diagnosis of syncope (2016 revised edition) [J]. Chin J Pediatr 54(4):246–250 (Chinese)
Sheldon RS, Grubb BP, Olshansky B et al (2015) 2015 heart rhythm society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome, inappropriate sinus tachycardia, and vasovagal syncope. Heart Rhythm 12(6):e41–e63. https://doi.org/10.1016/j.hrthm.2015.03.029
Vlahos AP, Tzoufi M, Katsouras CS, Barka T, Sionti I, Michalis LK, Siamopoulou A, Kolettis TM (2007) Provocation of neurocardiogenic syncope during head-up tilt testing in children: comparison between isoproterenol and nitroglycerin. Pediatrics 119(2):e419–e425. https://doi.org/10.1542/peds.2006-1862
Bicker J, Fortuna A, Alves G, Falcão A (2013) Liquid chromatographic methods for the quantification of catecholamines and their metabolites in several biological samples—a review. Anal Chim Acta 768:12–34. https://doi.org/10.1016/j.aca.2012.12.030
Chen L, Yang YY, Wang C et al (2006) A multi-center study of hemodynamic characteristics exhibited by children with unexplained syncope. Chin Med J 119(24):2062–2068
Younoszai AK, Franklin WH, Chan DP et al (1998) Oral fluid therapy. A promising treatment for vasodepressor syncope. Arch Pediatr Adolesc Med 152(2):165–168
Li H, Wang Y, Liu P, Chen Y, Feng X, Tang C, du J, Jin H (2016) Body mass index (BMI) is associated with the therapeutic response to oral rehydration solution in children with postural tachycardia syndrome. Pediatr Cardiol 37(7):1313–1318. https://doi.org/10.1007/s00246-016-1436-1
Zeng H, Ge K, Zhang W, Wang G, Guo L (2008) The effect of orthostatic training in the prevention of vasovagal syncope and its influencing factors. Int Heart J 49(6):707–712
Anderson JB, Czosek RJ, Knilans TK, Marino BS (2012) The effect of paediatric syncope on health-related quality of life. Cardiol Young 22(5):583–588. https://doi.org/10.1017/S1047951112000133
Winker R, Barth A, Bidmon D, Ponocny I, Weber M, Mayr O, Robertson D, Diedrich Á, Maier R, Pilger A, Haber P, Rüdiger HW (2005) Endurance exercise training in orthostatic intolerance: a randomized, controlled trial. Hypertension 45(3):391–398. https://doi.org/10.1161/01.HYP.0000156540.25707.af
Vaddadi G, Guo L, Esler M, Socratous F, Schlaich M, Chopra R, Eikelis N, Lambert G, Trauer T, Lambert E (2011) Recurrent postural vasovagal syncope: sympathetic nervous system phenotypes. Circ Arrhythm Electrophysiol 4(5):711–718. https://doi.org/10.1161/CIRCEP.111.962332
Qingyou Z, Junbao D, Jianjun C, Wanzhen L (2004) Association of clinical characteristics of unexplained syncope with the outcome of head-up tilt tests in children. Pediatr Cardiol 25(4):360–364. https://doi.org/10.1007/s00246-003-0513-4
Biffi M, Boriani G, Sabbatani P, Bronzetti G, Frabetti L, Zannoli R, Branzi A, Magnani B (1997) Malignant vasovagal syncope: a randomised trial of metoprolol and clonidine. Heart 77(3):268–272
Klingenheben T, Credner S, Hohnloser SH (1999) Prospective evaluation of a two-step therapeutic strategy in neurocardiogenic syncope: midodrine as second line treatment in patients refractory to beta-blockers. Pacing Clin Electrophysiol 22(2):276–281
Von Euler US, Hellner S, Purkhold A (1954) Excretion of noradrenaline in urine in hypertension. Scand J Clin Lab Invest 6(1):54–59
Zhang Q, Liao Y, Tang C, du J, Jin H (2012) Twenty-four-hour urinary sodium excretion and postural orthostatic tachycardia syndrome. J Pediatr 161(2):281–284. https://doi.org/10.1016/j.jpeds.2012.01.054
Acknowledgements
We thank the Beijing Harmony Health Medical Diagnostics Co., Ltd., for contributing to these studies.
Funding
This study was supported in part by grants from the National Natural Science Foundation of China (No. 30900730) and Technology Development Plan of Shandong Province (2014GSF118066). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
QYK: data acquisition and analysis, writing. ZFC, MMW: performed the experiments. YYP, MML: data acquisition. CFZ: review and editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Consent for publication
Not applicable.
Ethics approval and consent to participate
This study was approved by the ethics committee of Qilu Hospital of Shandong University, and all participants’ guardians were fully informed of the purpose and methods of the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kong, Q., Yang, X., Cai, Z. et al. Twenty-four-hour urine NE level as a predictor of the therapeutic response to metoprolol in children with recurrent vasovagal syncope. Ir J Med Sci 188, 1279–1287 (2019). https://doi.org/10.1007/s11845-019-01979-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-019-01979-9