Abstract
Objective
Cryptococcus is an opportunistic yeast with a worldwide distribution that primarily causes significant infections in immunocompromised individuals, generally by affecting the respiratory tract. But primary cutaneous cryptococcosis (PCC) without systemic infection is rare. We report a case of PCC in a patient with nephrotic syndrome.
Methods
The 23-year-old man developed severe necrotising cellulitis on both the anterior and posterior of his trunk following a massage. He had been treated with systemic corticosteroids over 20 months for nephrotic syndrome. A skin biopsy of the wound area revealed cutaneous vasculitis and chronic inflammation with yeast-like organisms. Periodic acid-Schiff (PAS) staining indicated that the structures were consistent with Cryptococcus. A Cryptococcus neoformans infection was confirmed by culture. Azole therapy was begun, and the skin ulcers gradually stopped disseminating. However, the patient died following continuous capillary haemorrhage on the 22 day since admission.
Conclusion
Cryptococcus is crucial to be considered in the differential diagnosis of subcutaneous necrosis in any patient on immunosuppressive therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cryptococcus is a type of opportunistic encapsulated yeast with a worldwide distribution [1]. Cryptococcosis continues to cause significant morbidity and mortality, especially in immunocompromised patients such as those with AIDS, organ transplants, haematological malignancies, and corticosteroid treatment [2]. Primary cutaneous cryptococcosis (PCC), lesions associated with a skin portal of entry without systemic infection, is rare but life threatening [3]. Here, we report one case of rare fatal PCC in an immunosuppressed patient. The clinical diagnosis corroborated histological findings and causative agent was confirmed as Cryptococcus neoformans in culture isolate.
Case report
A 23-year-old man was admitted to our hospital with fever and swelling, painful lesions in both the anterior and the posterior of his trunk, with no history of cough, headache or vomiting. He received a massage half a month prior to admission.
In his 20-month history of nephrotic syndrome, the patient was diagnosed with IgA nephropathy by renal biopsy and treated with prednisone (30–60 mg/day) for more than 1 year. Leflunomide and cyclophosphamide were each added once for a brief period. He had no known history of human immunodeficiency virus (HIV) infection and reported no contact with doves, poultry or other types of animals.
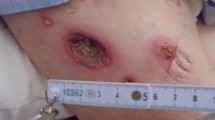
On physical examinations, the patient had erythema, tenderness, edema and soft swelling skin lesions around the trunk. Cutaneous ulcers developed with time, with necrotic subcutaneous soft tissue and perilesional edema. The laboratory examination showed that peripheral white blood cell, CD4+ T cell count, CD8+ T cell count, CD20+ B cell count and the CD4+/CD8+ T cell ratio were 23.9 (3.5–9.5) × 109/l, 143 (651.3 ± 273.6)/μl, 236 (452.62 ± 210.83)/μl, 159 (125.22 ± 51.55)/μl and 0.6, respectively, and that total blood immunoglobulin and immunoglobulin G were 17.9 (20–30) g/l and 4.8 (7–16) g/l, respectively. The serum creatinine level was elevated at 1.86 mg/dl, and the cystatin C level increased to 2.50 mg/dl. Antibodies to HIV and to hepatitis B and C were all negative, and blood cultures were negative. The serum cryptococcal antigen latex agglutination test (Immuno-Mycologics, Inc., Norman, OK, USA) was positive at a titer of 1:32. Chest computed tomography (CT) scan on Hospital Day 1 revealed signs of mild pulmonary infection without nodules. A cultural examination of necrotizing tissue on Sabouraud glucose agar at 37 °C for 3 days yielded cream-like colonies. The isolate was identified as C. neoformans by API 20C AUX (Biomerieux, Marcy, France). The antifungal susceptibility test showed that the fungus was sensitive to itraconazole but resistent to fluconazole. Skin biopsy revealed chronic inflammation with necrosis and numerous variably sized, round-to-oval budding organisms. Periodic acid-Schiff (PAS) staining of the dermis and soft tissue revealed blue, positive capsulated organisms (Fig. 1) that were consistent with Cryptococcus. Cerebrospinal fluid examination, brain magnetic resonance imaging and abdominal ultrasound examination did not show any abnormality.
Fungal culture of the soft tissue revealed the growth of Cryptococcus and confirmed our clinical diagnosis of PCC. The patient was started on fluconazole (600 mg/day for 4 days) intravenously and then changed to itraconazole (500 mg/day for 5 days) intravenously according to the result of antifungal sensitivity test. Surgical debridement was performed every day (Fig. 2). There was no evidence of disseminated intravascular coagulation. However, on the 22 day since admission, the patient died following continuous capillary haemorrhage.
Discussion
Cryptococcus neoformans is an opportunistic yeast discovered from soil, decaying wood, fruits and vegetables in the environment worldwide. The most characteristic feature of the yeast is its polysaccharide capsule, which is the most important virulence factor and can be visualised with India Ink, methylene blue and mucicarmine staining. Four serotypes of C. neoformans have been identified: serotype A, D and serotype B and C. Serotype A (C. neoformans var.grubii) distributes ubiquitously, serotype D (C. neoformans var. neoformans) is found mainly in Europe, and serotypes B and C (C. neoformans var. gattii) are limited to tropical and subtropical areas [1]. The clinical isolate of C. neoformans var. neoformans predominates in non-HIV patients in China [4].
Primary cutaneous cryptococcosis often affects patients from rural areas with trauma or pre-existing cutaneous lesions and is defined as Cryptococcosis in the skin lesion biopsy specimen or by culture and either clinical criteria or histological criteria, together with the absence of dissemination [1]. Its common dermatological features include cellulitis, ulceration, whitlow, abscesses, lupus erythematosus, eczema and nodules in the face, hand, arm, leg or any limited portion of the unclothed body [3]. Routine laboratory test results were usually normal for immunocompetent patients and most case were treated by azoles such as itraconazole and fluconazole and showed good outcome (Table 1). The reported case was immunosuppressed due to treatment for nephropathy, and clinically documented diagnose was made based on histological findings and culture isolate.
The immune response to C. neoformans is probably efficient, for the low frequency of PCC in the whole population [9]. Both the innate and adaptive immune systems attack established cryptococcal infections, but the organism employs several specific strategies to neutralise the host’s immune system [26]. In the AIDS population, Cryptococcosis has been well analysed and considered as AIDS-defining illness. Steroids and immunosuppressive therapy are widely used to treat nephrotic syndrome, which caused CD4+ T cell count falling down in many patients [22]. Possible causes of infection in this described patient were down-regulation of T cell response and a possible micro-trauma on his thinning skin, which served as the portal of entry during massage in a moist and contaminated environment. Even low-dosage (e.g. 10 mg daily) Corticosteroid monotherapy can increase the possibility of a Cryptococcal infection (Table 1). A similar case of PCC on a 55-year-old renal transplant recipient with multiple cutaneous lesions on left thigh and nasal bleeding has been described [7]. Many other cases have been attributed to consumption of steroids or other immunosuppressant agents following solid organ transplantation [12, 15, 21–23]. The majority of these patients showed an excellent outcome upon treatment with azoles, and multiple successful regimens have also been described [6, 7, 21].
This is, to our best knowledge, the most severe case of fatal PCC due to C. neoformans in an immunocompromised patient. There is a possibility that cryptococcosis affected coagulation then caused death of the described patient, confirmation of which awaits further study. PCC should be considered as a possible cause of skin lesion resistant to empirical antibiotic therapy in immunosuppressed patients and appropriate investigations and antifungal treatment should be initiated at an early stage.
Consent
Written informed consent was obtained from next of kin for publication of this case report and accompanying images. A copy of the parental written consent is available for reviewing by the editors of this journal.
References
Neuville S, Dromer F, Morin O, Dupont B, Ronio O, Lortholary O (2003) Primary cutaneous cryptococcosis: a distinct clinical entity. Clin Infect Dis 36:337–347
Hafner C, Linde HJ, Vogt T, Breindl G, Tintelnot K, Koellner K, Landthaler M, Szeimies RM (2005) Primary cutaneous cryptococcosis and secondary antigenemia in a patient with long-term corticosteroid therapy. Infection 33:86–89
Dinato SLM, Dinato MM, Nakanishi CP, Almeida JRP, Romiti N (2006) Disseminated cutaneous cryptcoccosis in a patient with AIDS. Rev Inst Med Trop Sao Paulo 48:353–358
Li A, Nishimura K, Taguchi H, Tanaka R, Wu S, Miyaji M (1993) The isolation of Cryptococcus neoformans from pigeon droppings and serotyping of naturally and clinically sourced isolates in China. Mycopathologia 124:1–5
Molina-Leyva A, Ruiz-Carrascosa JC, Leyva-Garcia A, Husein-Elahmed H (2013) Cutaneous Cryptococcus laurentii infection in an immunocompetent child. Int J Infect Dis 17:e1232–e1233
Marques SA, Bastazini IJR, Martins AL, Barreto JA, Barbieri D’Elia MP, Lastória JC, Marques ME (2012) Primary cutaneous cryptococcosis in Brazil: report of 11 cases in immunocompetent and immunosuppressed patients. Int J Dermatol 51:780–784
Anuja K, Mahua S (2012) Primary cutaneous cryptococcosis due to Cryptococcous laurentii in a renal transplant recipient. Saudi J Kidney Dis Transpl 23(1):102–105
Pasa CR, Chang MR, Hans-Filho G (2012) Post-trauma primary cutaneous cryptococcosis in an immunocompetent host by Cryptococcus gattii VGII. Mycoses 55:e1–e3
Spiliopoulou A, Anastassiou ED, Christofidou M (2011) Primary cutaneous cryptococcosis in immunocompetent hosts. Mycoses 55:e45–e47
Nilton N, Nilton NF, Andreza GV (2011) Primay cutaneous cryptococcosis in an immunocompetent patient. An Bras Dermatol 86:1178–1180
Lingegowda BP, Koh TH, Ong HS, Tan TT (2011) Primary cutaneous cryptococcosis due to Cryptococcus gattii in Singapore. Singap Med J 52:e160–e162
Ferry T, Moos D, Radenne S, Bienvenu AL, Kanitakis J (2011) Primary cutaneous cryptococcosis in a liver transplant recipient. BMJ Case Rep. 2011:bcr0220113814
Chrystiane RP, Marilene RC, Günter H (2011) Post-trauma primary cutaneous cryptococcosis in an immunocompetent host by Cryptococcus gattii VGII. Mycoses 55:e1–e3
Leão CA, Ferreira-Paim K, Andrade-Silva L, Mora DJ, Da-Silva PR, Machado AS, Neves PF, Pena GS et al (2011) Primary cutaneous cryptococcosis caused by Cryptococcus gattii in an immunocompetent host. Med Mycol 49:352–355
Zorman JV, Zupanc TL, Parac Z, Cucek I (2010) Primary cutaneous cryptococcosis in a renal transplant recipient: case report. Mycoses 53:535–537
Pau M, Lallai C, Aste N, Aste N, Atzori L (2010) Primary cutaneous cryptococcosis in an immunocompetent host. Mycoses 53:256–258
Mehta V, De A, Balachandran C, Monga P (2009) Mucocutaneous histoplasmosis in HIV with an atypical ecthyma like presentation. Dermatol Online J 15:10
Jasch KC, Hermes B, Scheller U, Harth W (2008) Pyoderma gangrenosum-like primary cutaneous cryptococcosis. Acta Derm Venereol 88:76–77
Emily AM, Brain BA, Stephen MK (2008) Cutaneous cryptococcosis in a patient on corticosteroid therapy for rheumatoid arthritis. Int J Dermatol 47:630–632
Allegue F, de Lis MP, Pérez-Alvarez R (2007) Primary cutaneous cryptococcosis presenting as a whitlow. Acta Derm Venereol 87:443–444
Lu HC, Yang YY, Huang YL, Chen TL, Chuang CL, Lee FY, Lee SD (2007) Disseminated cryptococcosis initially presenting as cellulitis in a rheumatoid arthritis patient. J Chin Med Assoc 70:249–252
Ogami N, Horino T, Arii K, Takao T, Hashimoto K, Seike M, Kodama H (2005) Primary cutaneous cryptococcosis in a patient with minimal change nephrotic syndrome. Nephrol Dial Transpl 20:2578–2579
Hafner C, Linde HJ, Vogt T, Breindl G, Tintelnot K, Koellner K, Landthaler M, Szeimies RM (2005) Primary cutaneous cryptococcosis and secondary antigenemia in a patient with long-term corticosteroid therapy. Infection 33:86–89
Xia XJ, Xu AE (2005) Two cases of cutaneous cryptococcosis. Mycoses 48:238–241
Werchniak AE, Baughman RD (2004) Primary cutaneous cryptococcosis in an elderly man. Clin Exp Dermatol 29:159–160
Voelz K, May RC (2010) Cryptococcal interactions with the host immune system. Eukaryot Cell 9:835–846
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Liu, Y., qunpeng, H., Shutian, X. et al. Fatal primary cutaneous cryptococcosis: case report and review of published literature. Ir J Med Sci 185, 959–963 (2016). https://doi.org/10.1007/s11845-015-1346-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-015-1346-x