Abstract
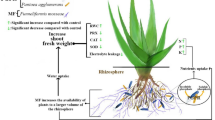
Melissa officinalis L. (Lemon balm) is one of the most important medicinal plants in the Lamiaceae family, whose essential oil compounds are affected by the inoculation of beneficial microorganisms and different irrigation regimes. In the present study, the effect of inoculation of plant growth-promoting rhizobacteria (PGPR), arbuscular mycorrhiza fungi (AMF) and PGPR + AMF (co-microbial) on growth, photosynthetic pigments, relative water content (RWC), proline and essential oil of lemon balm subjected to 100 (control) and 50% (drought stress) of field capacity (FC) in greenhouse conditions was investigated. The results showed that 50% FC irrigation increased proline content (2.9-fold) and declined chlorophyll a (40.4%), b (40.2%), carotenoids (44.6%) and RWC (17.8%) compared to the well-watered plants. Plants watered with 50% FC showed a decrease in height, dry weight of roots and shoots by 28.9, 24.1 and 25.6%, respectively, over the control. However, inoculation of PGPR, AMF and co-microbial by improving photosynthetic pigments, proline content and RWC increased plant tolerance and thus restored the growth and biomass of lemon balm under drought stress. Co-microbial inoculation also altered the chemical composition of secondary metabolites and increased the essential oil yield of lemon balm leaves. PGPR + AMF inoculation in lemon balm plants is recommended to increase plant tolerance to limited water conditions and also to improve the yield of essential oils for medical purposes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Melissa officinalis L. (Lemon balm) is one of the valuable medicinal herbs of the family Lamiaceae, which is commonly cultivated in many countries, including Iran (Younesi and Moradi 2015). The essential oil of lemon balm is widely used in the production of products such as insect repellents, natural insecticides, flavoring agents and condiments (Abbaszadeh et al. 2009; Verma et al. 2015). Hesperidin, luteolin, flavonoids, tannins, citronellal, beta-carophyllene, apigenin, linalool, isoquercetin, caffeic acid, neral, isomers of citral (geranial and geraniol), flavo-glycoside acid and rosmarinic acid are the most important secondary metabolites identified from lemon balm leaves (Argyropoulos and Müller 2014). In order to provide the essential oils needed by the market and industry, sustainable and high-quality production of lemon balm, even in unfavorable conditions of abiotic stresses, should be specially considered. In aromatic and medicinal plants, the accumulation of secondary metabolites is significantly affected by adverse environmental conditions, so that it changes the composition of essential oils and, consequently, increases or decreases secondary metabolites (Bonacina et al. 2017; Gerami et al. 2018). Inhibitory effects on growth and alterations in essential oils of medicinal plants under various abiotic stresses such as salinity and drought have been previously reported (Khorasaninejad et al. 2010; Bonacina et al. 2017). Plants activate the defense networks in response to stressful conditions to maintain cell homeostasis and alleviate the harmful effects of stress (Ghorbani et al. 2020; Ghasemi-Omran et al. 2021). Drought stress has been reported to inhibit various cell metabolisms, reduce water potential, disrupt ionic homeostasis and the activity of various enzymes, thereby reducing plant growth and yield (Bistgani et al. 2017).
Today, plant growth promoting rhizobacteria (PGPR) and arbuscular mycorrhiza fungi (AMF) are used as biofertilizers in sustainable agricultural systems in order to improve the quality and yield of crops and horticultural products, especially medicinal plants (Beattie 2006). Azospirillum brasilense is a Gram-negative free-living nitrogen-fixing bacterium and one of the most studied PGPR bacteria (Housh et al. 2021). PGPR have been shown to improve plant tolerance under drought (Zahir et al. 2008), heavy metals (Sheng 2005) and salinity (Bidgoli et al. 2019) stresses as biocontrol agents. PGPR have been reported to restore plant growth under biotic and abiotic stresses by reducing ethylene levels and increasing auxin biosynthesis as well as nitrogen fixation in roots (Patten and Glick 2002; Glick et al. 2007). AMF are also beneficial soil microorganisms that effectively improve plant tolerance under stressful conditions by establishing a mutual symbiotic relationship with the roots (Selosse et al. 2006; Wu et al. 2021). Numerous studies have shown that AMF can improve plant growth and increase plant tolerance under various environmental stresses such as drought (Essahibi et al. 2018; Zhang et al. 2018), salinity (Hajiboland et al. 2015) and heavy metals (Abdelhameed and Rabab 2019). AMF has been reported to enhance host plant adaptation to drought stress by regulating leaf stomatal conductivity, improving root hydraulic conductivity, restoring ionic homeostasis, and adjusting biochemical mechanisms (antioxidant defense system and osmotic potential regulation) (Aroca et al. 2007; Bitterlich et al. 2018). Several reports have shown that inoculation of PGPR and AMF altered the biosynthesis and amount of essential oils of medicinal plants such as lavender (Tsuro et al. 2001), Hyssop (Sharifi 2017), rosemary (Bidgoli et al. 2019) and Satureja (Zakerian et al. 2020). Non-pathogenic microorganisms have been reported to induce the synthesis of secondary metabolites in the host plant through a mechanism called induced systemic resistance (ISR) (VanLoon and Glick 2004). It has also been indicated that the biological agents released by non-pathogenic microorganisms act as effective stimuli to induce key enzymes responsible for the synthesis of secondary metabolites (Chen et al. 2000; Çakmakçı et al. 2020).
Due to the problems of water shortage in many parts of Iran (Ghorbani et al. 2009) and the promising characteristics of PGPR and AMF, as well as the importance of the medicinal plant lemon balm in the pharmaceutical and food industry, the current study was conducted to investigate the effects of moderate drought stress alone and in combination with beneficial soil microorganisms of PGPR and AMF on the growth and composition of lemon balm essential oil.
Materials and methods
Microbial inoculum
In the present study, Azospirillum brasilense (Sp245 strain) was used as a PGPR. After culturing A. brasilense on Nitrogen-fixing bacteria (NFB) medium, PGPR inoculum was prepared in a bacterial concentration of 108 CFU/mL in deionized water. Glomus mosseae was used as AM fungi in this study, which was obtained from the Laboratory of Soil and Water Research Institute, Iran. The AMF inoculum included infected root fragments, mycelium and spores of G. mosseae. In order to inoculate, 100 g of AMF inoculum was added to the soil (1.5 kg) of each pot (Pan et al. 2020).
Plant materials and treatments
Lemon balm seeds were germinated in autoclaved in pots (17 cm height and 20 cm diameter) containing autoclaved clay, sand and humus (1:3:2) after surface sterilization with ethanol (70%) and chlorox (10%) solutions for 5 and 1 min, respectively, and washing with distilled water. In each pot, five seeds were planted which, after germination, were thinned to one seedling. Biofertilizer treatments include: (a) Non-inoculation treatment: The seeds were immersed in distilled water for 30 min and autoclaved AMF inoculum was added to the potting soil, (b) PGPR inoculation: soaking the seeds in PGPR inoculum (30 min) and adding autoclaved fungal inoculum to the soil, (c) AMF inoculation: soaking the seeds in distilled water and adding AMF inoculum to the soil, (d) Co-microbial inoculation: soaking the seeds in PGPR inoculum and adding AMF inoculum to the soil. The plants were grown at a 16/8 (light/dark) photoperiod with an intensity light of 350 µmol photon m─2 s─1, 60 ± 5 relative humidity and 25/22 °C day/night. The pots were watered as needed and fed by half-strength Hoagland once a week. 30 days after sowing, lemon balm plants were treated with drought stress at two levels (100 and 50% of field capacity (FC)) according to the managed allowed depletion previously described by Allen et al. (2000). After weighing the pots daily and measuring the amount of water lost through soil leaching, evaporation and transpiration, the lost water was replaced. The plants were exposed to drought stress for 30 days and then sampling was performed.
Measurement of growth parameters
At the end of the experiment, the height and leaf numbers of lemon balm plants were measured. Then, to estimate the dry weight of roots and shoots, plant samples were divided into roots and shoots after sampling and weighed after being exposed to 70 °C for 72 h (Ghorbani et al. 2011).
Measurement of photosynthetic pigments, proline and relative water content (RWC)
The contents of chlorophyll (a and b) and carotenoids pigments were determined based on Wellburn (1994) and recording the optical density at 470, 652 and 665 nm as mg/g fresh weight.
Proline contents were measured by ninhydrin reagent (acetic acid + orthophosphoric acid (6 mM) + 125 g ninhydrin) according to Bates et al. (1973) method. After crushing the fresh leaves in sulfosalicylic acid and centrifuging at 10,000 × g for 15 min, ninhydrin reagent was added to the supernatants and incubated at 100 °C for 1 h. After cooling the samples on the ice bath and adding toluene to each, the spectrophotometric measurements were recorded at 520 nm absorbance.
The RWC leaves were obtained according to the method previously explained by Schonfeld et al. (1988) and according to the formula: (FM – DM) / (TM – DM) × 100. (FM: fresh mass weight, DM: dry mass weight, TM: turgid mass weight).
Extraction and assay of essential oil
After air-drying lemon balm leaves, the essential oil of the leaves was obtained by Clevenger device and hydrodistillation for 3 h. Gas chromatography-mass spectrometry (GC-MS) device (Varian 3400, USA) with a DB-5 fused silica column (30 m × 0.25 mm, film thickness 0.25 μm, J and W Scientific, Inc.) was used for GC-MS analysis. Dichloromethane with a ratio of 10:1 was used to dilute the essential oil. Helium was employed as the carrier gas, 1 mL/min flow rate. the injection volume and injector temperature were 2.0 µL and 220 °C, respectively. The temperature of the column was maintained at 60 °C for 5 min and then reached to 180 °C at the rate of 2 °C/min, to 260 °C at the rate of 10 °C/min, and finally heated at 40 °C/min to program 300 °C. Using the alkane series C7-C26, the retention indices (RIs) of the essential oil compounds were determined. The chemical compounds of the essential oils were identified by comparing the mass spectra of the essential oil compounds with the mass spectra of the NIST 11.0 library and comparing the IRs published in the literature.
Statistical analysis
All experiments were performed with four biological replications (mean morphological traits and RWC were calculated from 10 independent biological replications), each of which averaged three technical replications. Data analysis was carried out using SAS 9.1.3 software. The means were compared based on the least significant difference (LSD) test (p < 0.05).
Results
Plant growth dynamics
The results showed that drought stress (50% FC) reduced the height of lemon balm by 28.9% compared to the control. Under non-stress conditions, the application of biofertilizers (AMF, PGPR and co-microbial inoculation) significantly improved plant height compared to non-inoculated plants. An increase of 13.8, 17.3 and 17.6% were recorded in plants inoculated with PGPR, AMF and co-microbial over the non-inoculated plants. Three microbial treatments restored the height of lemon balm in drought-stressed plants, however, there were no significant differences among the three microbial-colonized plants (p > 0.05, Table 1). Inoculation of PGPR, AMF and PGPR + AMF significantly enhanced the root dry weight (RDW) by 34.5, 46.6 and 45.7%, respectively in non-stressed plants. Drought stress declined the RDW by 24.1% over the control plants. However, three inoculation treatments significantly restored the RDW in drought-stressed plants, although there were no significant differences between them (p > 0.05, Table 1). In non-stressed plants, co-colonization of PGPR and AMF elevated the shoot dry weight (SDW) by 13%. Drought stress significantly reduced the SDW, while PGPR, AMF and PGPR + AMF colonization significantly improved the SDW of drought-stressed plants (12.5–19.3%) (Table 1). PGPR, AMF and PGPR + AMF treatments increased the RDW/SDW ratio by 27.8, 38.2 and 29.1%, respectively in comparison to no-inoculated plants under 100% FC. Drought stress had no significant effect on RDW/SDW ratio. Although the three inoculation treatments increased the RWD/SWD ratio in drought-stressed plants, however, no significant differences were observed between inoculated and non-inoculated plants under drought stress (Table 1). A 24.1% reduction was observed in the leaf numbers of lemon balm under drought stress compared to plants irrigated with 100% FC. However, inoculation of PGPR, AMF and PGPR + AMF significantly enhanced the numbers of leaves in 50% FC-stressed lemon balm plants and the highest increase was recorded in plants inoculated with PGPR + AMF (Table 1).
Photosynthetic pigments
As shown in Table 2, drought stress significantly reduced chlorophyll a and b content by 40.4 and 40.2 %, respectively in lemon balm leaves compared to non-inoculated plants watered with 100% FC. In drought stress-treated plants, PGPR, AMF and co-microbial colonization restored chlorophyll a content by 26.8, 22.8 and 42.3% and chlorophyll b by 32.7, 36.5 and 44.2%, respectively over non-inoculated plants under drought stress (Table 2). Drought stress also declined the carotenoids content over non-inoculated plants under 100% FC. However, three inoculation treatments caused a significant increase in the carotenoids content in drought stress-stressed plants over non-inoculated plants under drought stress, with the largest enhancement observed in co-microbial-inoculated plants (Table 2).
Relative water content (RWC) and proline content
Non-inoculated plants exposed to drought stress showed a 17.8% increase in RWC over non-inoculated plants watered with 100% FC. However, the colonization of PGPR, AMF and co-microbial significantly elevated RWC by 8.8, 10.3 and 11.7% in plants treated with drought stress over non-inoculated plants under 100% FC (Table 2). In Non-inoculated plants exposed to drought stress, all three inoculation treatments significantly enhanced the proline content in lemon balm leaves but there were no significant differences between them. Drought stress significantly increased the proline content over non-inoculated plants; however, the inoculation of PGPR, AMF and PGPR + AMF caused a further increase in proline content in drought-stressed plants that the highest raise in proline content was observed in co-microbial-inoculated plants (Table 2).
Yield and chemical composition of essential oil
The chemical composition and yield of essential oil have an important effect on the commercial value of medicinal plants. In the present study, the yield of essential oil in non-inoculated plants watered with 100% FC was 0.105% and the application of drought stress increased the yield of essential oil by 58.1% compared to well-irrigated non-inoculated plants. In plants watered with 100% FC, inoculation of PGPR, AMF and PGRP + AMF significantly enhanced the yield of lemon balm essential oil and the highest increase was related to the co-microbial inoculation treatment. In non-inoculated plants treated with drought stress, PGPR and AMF inoculation had no significant effects on essential oil yield, however, the co-microbial inoculation increased essential oil yield by 44.8% compared to non-inoculated plants treated with drought stress (Fig. 1).
Thirty-eight compounds were detected in the essential oil of lemon balm leaves, which belonged to four groups, i.e., oxygenated monoterpenes, oxygenated sesquiterpenes, monoterpene hydrocarbons and sesquiterpene hydrocarbons. The oxygenated monoterpenes group was the predominant class in lemon balm essential oil in all treatments. The results also showed that drought stress and all three microbial inoculation treatments increased the monoterpenes hydrocarbons and sesquiterpenes hydrocarbons class compounds compared to non-inoculated plants treated with 100% FC. However, the concentration of oxygenated monoterpenes compounds under drought stress and three microbial inoculation treatments decreased over non-inoculated plants treated with 100% FC (Table 3). The results showed that α-citral (40.25%), neral (28.11%) and citronellal (9.07%), as the most biologically active compounds in the essential oil of lemon balm leaves, were the most abundant compounds in non-inoculated plants treated with 100% FC. Drought stress diminished α-citral (36.18%), neral (25.44 %) and citronellal (7.01%) compounds over non-inoculated plants watered with 100% FC. In non-inoculated plants treated with 100% FC, microbial inoculation treatments enhanced citronellal and lowered α-citral and neral. In non-inoculated plants treated with drought stress, microbial inoculation treatments enhanced citronellal and reduced neral. However, PGPR and AMF colonizations enhanced citral content and co-microbial colonization declined citral content in non-inoculated plants treated with drought stress (Table 3).
Discussion
The results illustrated that plant height, total dry weight of roots and shoots and number of leaves in lemon balm showed a significant decrease under drought stress. Idrees et al. (2010) indicated that drought stress decreases plant biomass production and growth by reducing water potential, water absorption, closing stomatal apparatuses, reducing turgor pressure and cell enlargement. Negative effects of drought stress on plant growth, biomass production and leaf number have been previously reported by Saheri et al. (2020) and Khan et al. (2019). Therefore, the decrease in growth and total dry weight of lemon balm treated with drought stress can be due to the reduction in turgor pressure and water potential induced by water stress, which may reduce cell expansion and division and induce senescence in the leaves (Saheri et al. 2020). A similar reduction in growth occurred in medicinal plants, such as Thymus daenensis (Bistgani et al. 2017), Portulaca oleracea (Saheri et al. 2020), and Rosmarinus officinalis (Abbaszadeh et al. 2020). However, our results indicated that inoculation of PGPR, AMF, and PGPR + AMF improved the growth and biomass of lemon balm under drought stress, in accordance with other results Kang et al. 2014; Ganjeali et al. 2018; Mutumba et al. 2018; Zakerian et al. 2020). Boutasknit et al. (2020) showed that AM fungi restored the growth of drought-stressed Carob plants by improving efficiency of photosynthetic apparatuses, stomatal conductance, water content, and nutrient uptake. It has been reported that the positive effect of PGPR on plant growth may be due to increased activity of ACC (1-aminocyclopropane-1-carboxylic acid) deaminase enzymes and, consequently, decreased ethylene levels in the plant (Glick et al. 2007; Zhang et al. 2019) indicated PGPR increase plant tolerance and growth under drought stress by preserving the thylakoid membrane structure of chloroplasts, improving the content of photosynthetic pigments, and increasing water use efficiency (WUE). Therefore, the improvement of growth in lemon balm by inoculation of PGPR and AMF indicates the ability of beneficial microorganisms to improve plant adaptation to conditions with limited water availability. The results also showed that growth improvement in plants inoculated by co-microbial (PGPR + AMF) was greater than inoculation with PGPR or AMF alone, indicating the synergistic effects of PGPR and AMF on the adaptation of lemon balm to drought stress, according to results previously reported by Ruíz-Sánchez et al. (2011).
Photosynthesis is an important plant process and sensitive to stressful conditions that directly affect plant growth and biomass (Ghorbani et al. 2019). The results illustrated that drought stress caused an evident decline in the contents of chlorophyll a, b and carotenoids, however, PGPR, AMF and co-microbial inoculation treatments were found to restore the contents of pigments in drought-stressed plants, which is consistent with data published by Zhang et al. (2019), Al-Arjani et al. (2020) and Sharifi (2017). Guler et al. (2016) demonstrated that PGPR restored the content of photosynthetic pigments by reducing oxidative stress and reactive oxygen species (ROS) levels, thereby improving plant adaptation to drought stress. Al-Arjani et al. (2020) showed that AMF increased chlorophyll content by enhancing magnesium uptake and, consequently, improved plant growth under drought stress. Increased chlorophyll synthesis by AM fungi has also been previously documented by Zhu et al. (2017) and Hashem et al. (2016). Improving the carotenoids content by inoculation of PGPR and AMF can protect the photosynthetic apparatus and, as a result, improve the efficiency of the photosynthetic process in drought-stressed plants. Therefore, our findings demonstrated that PGPR and AMF, by restoring photosynthetic pigments in drought-stressed plants, protected the photosynthetic apparatus and increased the efficiency of the photosynthesis process (Ghorbani et al. 2018a), which could improve plant growth under drought stress.
The relative water content (RWC) is used to evaluate the osmotic stress induced by stress conditions (Ghorbani et al. 2018b), which indicates the water balance in the plant. Plants with the accumulation of osmotic compounds such as proline, maintain plant water balance and thus improve adaptation to osmotic stress induced by abiotic stress such as drought stress (Ghorbani et al. 2021). The results of the present study showed that drought stress decreased RWC and increased proline accumulation in the leaves of lemon balm. However, PGPR, AMF and co-microbial inoculations improved RWC and proline content in drought-stressed plants. Similar results of improving RWC and proline content in potato by PGPR inoculation (Batool et al. 2020) and ephedra by AMF induction (Al-Arjani et al. 2020) have been documented. In addition to osmotic regulation, proline accumulation protects membranes and proteins against oxidative stress induced by abiotic stresses. Therefore, the findings showed that inoculation of PGPR and AMF by regulating proline metabolism, caused proline accumulation and, consequently, improved RWC, which can increase the adaptation of lemon balm plants under drought stress.
Shikimic acid and mevalonic acid are two distinct pathways for the biosynthesis of plant essential oils. Terpenoids are synthesized from the mevalonic pathway under the influence of photosynthesis, growth and assimilation, while phenolic compounds are synthesized from the shikimic pathway by the activity of the phenylalanine ammonia-lyase (PAL) enzyme (Gang et al. 2001). The results showed that the yield of lemon balm essential oil increased under drought stress, which suggests that adverse conditions, even with reduced photosynthesis and less available carbon, may have a positive effect on the production of secondary metabolites (Siemens et al. 2002; Brili et al. 2007). Similar increases occurred in the essential oils of Cuminum cyminum L. (Alinian et al. 2016) and Pelargonium odoratissimum (Khalid et al. 2010) under drought stress. However, Baghalian et al. (2011) and Bannayan et al. (2008) showed that drought stress had no effect on the essential oil of black cumin and German chamomile, respectively. Therefore, these results indicate that the essential oil of medicinal plants is affected by various factors such as growth conditions, genotype, species and drought stress level. The results also showed that co-microbial inoculation increased the yield of essential oil in lemon balm treated with 100% FC and drought stress, which indicates the positive effect of co-inoculation on the biosynthesis of lemon balm essential oil. Similar results have been reported by Zakerian et al. (2020), Bidgoli et al. (2019) and Sharifi (2017) on the positive effect of PGPR and AMF inoculation on the essential oil content of medicinal plants. Due to the role of secondary metabolites as lipophilic compounds in reducing membrane lipid peroxidation and minimizing the accumulation of ROS and oxidative stress (Vickers et al. 2009), increasing the production of secondary metabolites could be a defense mechanism induced by microbial inoculation to improve plant tolerance under drought stress and alleviate oxidative stress induced by drought stress. It has been shown that the biosynthesis of secondary metabolites depends on photosynthesis, primary metabolism, and oxidation processes for the energy and carbon supply (Raklami et al. 2019; Azizi et al. 2021). Both PGPR and AMF are able to improve primary metabolites by increasing plant growth, mineral content and photosynthesis, suggesting that the improving effects of microbial inoculation on the synthesis of secondary metabolites may be due to the proper balance of carbon and nutrient supply (Vafadar et al. 2014). It has also been shown that volatile organic compounds (VOCs) released by PGPR induce the activity of enzymes involved in the synthesis of essential oils of Mentha piperita (Santoro et al. 2011). Therefore, the results revealed PGPR and AMF inoculations changed the concentration of essential oil compounds in non-inoculated plants treated with 100% FC and drought stress, which indicates the effects of microbial inoculation on the activity of enzymes involved in the synthesis of these compounds, which can be due to the production of elicitors produced by PGPR and AMF in the soil (Pan et al. 2020). However, due to the lack of available information on the role of PGPR and AMF on the expression or activity of enzymes responsible for the synthesis of essential oil in lemon balm, more accurate studies at the molecular levels are needed to accurately understand the effect of PGPR and AMF on essential oil accumulation.
Conclusions
Conclusively, drought stress (50% FC) significantly reduced the photosynthetic pigments and RWC and thus reduced the growth and biomass of lemon balm, however, the yield of essential oil under drought stress increased. Inoculation of PGPR, AMF and PGPR + AMF improved photosynthetic pigments and RWC and increased proline accumulation in drought-stressed plants, which was associated with improved plant growth and biomass, and the highest increase was observed in plants inoculated with co-microbial. Co-microbial inoculation also improved the essential oil yield in lemon balm treated with 100% FC and drought stress, which indicates the positive effect of microbial inoculation on the production of secondary metabolites in medicinal plants. Due to the synergistic effects of PGPR and AMF inoculation, co-microbial inoculation in lemon balm plants is recommended to enhance plant tolerance to drought stress and also to improve commercial value.
References
Abbaszadeh B, Farahani HA, Valadabadi SA, Darvishi HH (2009) Nitrogenous fertilizer influence on quantity and quality values of balm (Melissa officinalis L.). J Agric Ext Rural Dev 1(1):31–33. https://doi.org/10.5897/JAERD.9000054
Abbaszadeh B, Layeghhaghighi M, Azimi R, Hadi N (2020) Improving water use efficiency through drought stress and using salicylic acid for proper production of Rosmarinus officinalis L. Ind Crops Prod 144:111893. https://doi.org/10.1016/j.indcrop.2019.111893
Abdelhameed RE, Rabab AM (2019) Alleviation of cadmium stress by arbuscular mycorrhizal symbiosis. Int J Phytoremed 21(7):663–671. https://doi.org/10.1080/15226514.2018.1556584
Al-Arjani AF, Hashem A, Abd-Allah EF (2020) Arbuscular mycorrhizal fungi modulates dynamics tolerance expression to mitigate drought stress in Ephedra foliata Boiss. S J Biol Sci 27(1):380–394. https://doi.org/10.1016/j.sjbs.2019.10.008
Alinian S, Razmjoo J, Zeinali H (2016) Flavonoids, anthocynins, phenolics and essential oil produced in cumin (Cuminum cyminum L.) accessions under different irrigation regimes. Ind Crops Prod 81:49–55. https://doi.org/10.1016/j.indcrop.2015.11.040
Allen RG, Pereira LS, Raes D, Smith M (2000) Crop evapotranspiration. FAO irrigation and drainage paper, no. 56. FAO, Roma, pp 1–300
Argyropoulos D, Müller J (2014) Changes of essential oil content and composition during convective drying of lemon balm (Melissa officinalis L.). Ind Crops Prod 52:118–124. https://doi.org/10.1016/j.indcrop.2013.10.020
Aroca R, Porcel R, Ruiz-Lozano JM (2007) How does arbuscular mycorrhizal symbiosis regulate root hydraulic properties and plasma membrane aquaporins in Phaseolus vulgaris under drought, cold or salinity stresses? New Phytol 173:808–816. https://doi.org/10.1111/j.1469-8137.2006.01961.x
Azizi S, Tabari Kouchaksaraei M, Hadian J, Fallah Nosrat Abad AR, Modarres Sanavi SAM, Ammer C, Bader MKF (2021) Dual inoculations of arbuscular mycorrhizal fungi and plant growth-promoting rhizobacteria boost drought resistance and essential oil yield of common myrtle. For Ecol Manag 497:119478. https://doi.org/10.1016/j.foreco.2021.119478
Baghalian K, Abdoshah S, Khalighi-Sigaroodi F, Paknejad F (2011) Physiological and phytochemical response to drought stress of German chamomile (Matricaria recutita L.). Plant Physiol Biochem 49:201–207. https://doi.org/10.1016/j.plaphy.2010.11.010
Bannayan M, Nadjafi F, Azizi M, Tabrizi L, Rastgoo M (2008) Yield and seed quality of Plantago ovata and Nigella sativa under different irrigation treatments. Ind Crops Prod 27:11–16. https://doi.org/10.1016/j.indcrop.2007.05.002
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Batool T, Ali S, Seleiman MF, Naveed NH, Ali A, Ahmed K, Abid M, Rizwan M, Shahid MR, Alotaibi M, Al-Ashkar I, Mubushar M (2020) Plant growth promoting rhizobacteria alleviates drought stress in potato in response to suppressive oxidative stress and antioxidant enzymes activities. Sci Rep 10:16975. https://doi.org/10.1038/s41598-020-73489-z
Beattie GA (2006) Plant-associated bacteria: survey, molecularphylogeny, genomics and recent advances. In: Gnanamanickam SS (ed) Plant-associated bacteria. Springer, Dordrecht, pp 1–56. https://doi.org/10.1007/978-1-4020-4538-7_1
Bidgoli RD, Azarnezhad N, Akhbari M, Ghorbani M (2019) Salinity stress and PGPR effects on essential oil changes in Rosmarinus officinalis L. Agric Food Secur 8:2. https://doi.org/10.1186/s40066-018-0246-5
Bistgani ZE, Siadat SA, Bakhshandeh A, Ghasemi Pirbalouti A, Hashemi M (2017) Interactive effects of drought stress and chitosan application on physiological characteristics and essential oil yield of Thymus daenensis Celak. Crop J 5(5):407–415. https://doi.org/10.1016/j.cj.2017.04.003
Bitterlich M, Sandmann M, Graefe J (2018) Arbuscular mycorrhiza alleviates restrictions to substrate water flow and delays transpiration limitation to stronger drought in tomato. Front Plant Sci 9:154. https://doi.org/10.3389/fpls.2018.00154
Bonacina C, Trevizan CB, Stracieri J, dos Santos TB, Gonçalves JE, Gazim ZC, de Souza SGH (2017) Changes in growth, oxidative metabolism and essential oil composition of lemon balm (Melissa officinalis L.) subjected to salt stress. Aust J Crop Sci 11(12):1665–1674. https://doi.org/10.21475/ajcs.17.11.12.pne921
Boutasknit A, Baslam M, Ait-El-Mokhtar M, Anli M, Ben-Laouane R, Douira A, El Modafar C, Mitsui T, Wahbi S, Meddich A (2020) Arbuscular mycorrhizal fungi mediate drought tolerance and recovery in two contrasting Carob (Ceratonia siliqua L.) ecotypes by regulating stomatal, water relations, and (In)organic adjustments. Plants 9:80. https://doi.org/10.3390/plants9010080
Brilli F, Barta C, Fortunati A, Lerdau M, Loreto F, Centritto M (2007) Responses of isoprene emission and carbon metabolism to drought in White poplar (Populus alba) saplings. New Phytol 175(2):244–254. https://doi.org/10.1111/j.1469-8137.2007.02094.x
Çakmakçı R, Mosber G, Milton AH, Alatürk F, Ali B (2020) The effect of auxin and auxin-producing bacteria on the growth, essential oil yield, and composition in medicinal and aromatic plants. Curr Microbiol 77:564–577. https://doi.org/10.1007/s00284-020-01917-4
Chen C, Belanger R, Benhamou N, Paulitz TC (2000) Defense enzymes induced in cucumber roots by treatment with plant growth promoting rhizobacteria (PGPR) and Pythioum aphanidermatum. Physiol Mol Plant Pathol 56:13–23. https://doi.org/10.1006/pmpp.1999.0243
Essahibi A, Benhiba L, Babram MA, Ghoulam C, Qaddoury A (2018) Influence of arbuscular mycorrhizal fungi on the functional mechanisms associated with drought tolerance in carob (Ceratonia siliqua L.). Trees 32:87–97. https://doi.org/10.1007/s00468-017-1613-8
Gang DR, Wang J, Dudareva N, Hee NK, Simon JE (2001) An investigation of the storage and biosynthesis of phenylpropenes in sweet basil. Plant Physiol 125:539–555. https://doi.org/10.1104/pp.125.2.539
Ganjeali A, Ashiani E, Zare M, Tabasi E (2018) Influences of the arbuscular mycorrhizal fungus Glomus mosseae on morphophysiological traits and biochemical compounds of common bean (Phaseolus vulgaris) under drought stress. S Afr J Plant Soil 35(2):121–127. https://doi.org/10.1080/02571862.2017.1340982
Gerami M, Ghorbani A, Karimi S (2018) Role of salicylic acid pretreatment in alleviating cadmium-induced toxicity in Salvia officinalis L. Iran J Plant Biol 10(1):81–95
Ghasemi-Omran VO, Ghorbani A, Sajjadi-Otaghsara SA (2021) Melatonin alleviates NaCl-induced damage by regulating ionic homeostasis, antioxidant system, redox homeostasis, and expression of steviol glycosides-related biosynthetic genes in in vitro cultured Stevia rebaudiana Bertoni. Vitro Cell Dev Biol- Plant 57:319–331. https://doi.org/10.1007/s11627-021-10161-9
Ghorbani A, Ghasemi Omran VO, Razavi SM, Pirdashti H, Ranjbar M (2019) Piriformospora indica confers salinity tolerance on tomato (Lycopersicon esculentum Mill.) through amelioration of nutrient accumulation, K+/Na+ homeostasis and water status. Plant Cell Rep 38:1151–1163. https://doi.org/10.1007/s00299-019-02434-w
Ghorbani A, Pishkar L, Roodbari N, Pehlivan N, Wu C (2021) Nitric oxide could allay arsenic phytotoxicity in tomato (Solanum lycopersicum L.) by modulating photosynthetic pigments, phytochelatin metabolism, molecular redox status and arsenic sequestration. Plant Physiol Biochem 167:337–348. https://doi.org/10.1016/j.plaphy.2021.08.019
Ghorbani A, Razavi SM, Ghasemi Omran VO, Pirdashti H (2018a) Piriformospora indica alleviates salinity by boosting redox poise and antioxidative potential of tomato. Russ J Plant Physiol 65:898–907. https://doi.org/10.1134/S1021443718060079
Ghorbani A, Razavi SM, Ghasemi Omran VO, Pirdashti H (2018b) Piriformospora indica inoculation alleviates the adverse effect of NaCl stress on growth, gas exchange and chlorophyll fluorescence in tomato (Solanum lycopersicum L.). Plant Biol 20:729–736. https://doi.org/10.1111/plb.12717
Ghorbani A, Tafteh M, Roudbari N, Pishkar L, Zhang W, Wu C (2020) Piriformospora indica augments arsenic tolerance in rice (Oryza sativa) by immobilizing arsenic in roots and improving iron translocation to shoots. Ecotoxicol Environ Saf 209:111793. https://doi.org/10.1016/j.ecoenv.2020.111793
Ghorbani A, Zarinkamar F, Fallah A (2009) The effect of cold stress on the morphologic and physiologic characters of tow rice varieties in seedling stage. J Crop Breed 1:50–66
Ghorbani A, Zarinkamar F, Fallah A (2011) Effect of cold stress on the anatomy and morphology of the tolerant and sensitive cultivars of rice during germination. J Cell Tissue 2(3):235–244
Glick BR, Cheng Z, Czarny J, Duan J (2007) Promotion of plant growth by ACC deaminase-producing soil bacteria. Eur J Plant Pathol 119:329–339. https://doi.org/10.1007/s10658-007-9162-4
Guler NS, Pehlivan N, Karaoglu SA, Guzel S, Bozdeveci A (2016) Trichoderma atroviride ID20G inoculation ameliorates drought stress-induced damages by improving antioxidant defence in maize seedlings. Acta Physiol Plant 38(6):132. https://doi.org/10.1007/s11738-016-2153-3
Hajiboland R, Dashtebani F, Aliasgharzad N (2015) Physiological responses of halophytic C4 grass, Aeluropus littoralis to salinity and arbuscular mycorrhizal fungi colonization. Photosynthetica 53(4):572–584. https://doi.org/10.1007/s11099-015-0131-4
Hashem A, Abd-Allah EF, Alqarawi AA, Al-Huqail AA, Wirth S, Egamberdieva D (2016) The interaction between arbuscular mycorrhizal fungi and endophytic bacteria enhances plant growth of Acacia gerrardii under salt stress. Front Microbiol 7:1089. https://doi.org/10.3389/fmicb.2016.01089
Housh AB, Powell G, Scott S, Anstaett A, Gerheart A, Benoit M, Waller S, Powell A, Guthrie JM, Higgins B, Wilder SL, Schueller MJ, Ferrieri RA (2021) Functional mutants of Azospirillum brasilense elicit beneficial physiological and metabolic responses in Zea mays contributing to increased host iron assimilation. ISME J 15:1505–1522. https://doi.org/10.1038/s41396-020-00866-x
Idrees M, Khan MMA, Aftab T, Naeem M, Hashmi N (2010) Salicylic acid-induced physiological and biochemical changes in lemongrass varieties under water stress. J Plant Interact 5(4):293–303. https://doi.org/10.1080/17429145.2010.508566
Kang SM, Khan AL, Waqas M, You YH, Kim JH, Kim JG, Hamayun M, Lee IJ (2014) Plant growth-promoting rhizobacteria reduce adverse effects of salinity and osmotic stress by regulating phytohormones and antioxidants in Cucumis sativus. J Plant Interact 9(1):673–682. https://doi.org/10.1080/17429145.2014.894587
Khalid KA, Teixeira da Silva JA, Caic W (2010) Water deficit and polyethylene glycol 6000 affects morphological and biochemical characters of Pelargonium odoratissimum L. Sci Hortic 125:159–166. https://doi.org/10.1016/j.scienta.2010.03.009
Khan N, Bano A, Rahman MA, Guo J, Kang Z, Babar MA (2019) Comparative physiological and metabolic analysis reveals a complex mechanism involved in drought tolerance in Chickpea (Cicer arietinum L.) induced by PGPR and PGRs. Sci Rep 9:2097. https://doi.org/10.1038/s41598-019-38702-8
Khorasaninejad S, Mousavi A, Soltanloo H, Hemmati K, Khalighi A (2010) The effect of salinity stress on growth parameters, essential oil yield and constituent of peppermint (Mentha piperita L.). World Appl Sci J 11(11):1403–1407
Mutumbam FA, Zagal E, Gerding M, Castillo-Rosales D, Paulino L, Schoebitz M (2018) Plant growth promoting rhizobacteria for improved water stress tolerance in wheat genotypes. J Soil Sci Plant Nutr 18(4):1080–1096. https://doi.org/10.4067/S0718-95162018005003003
Pan J, Huang C, Peng F, Zhang W, Luo J, Ma S, Xue X (2020) Effect of arbuscular mycorrhizal fungi (AMF) and plant growth-promoting bacteria (PGPR) inoculations on Elaeagnus angustifolia L. in saline soil. Appl Sci 10:945. https://doi.org/10.3390/app10030945
Patten CL, Glick BR (2002) Role of Pseudomonas putida in idoleacetic acid in development of the host plant root system. Appl J Environ Microbiol 68:3795–3801. https://doi.org/10.1128/AEM.68.8.3795-3801.2002
Raklami A, Bechtaoui N, Tahiri AI, Anli M, Meddich A, Oufdou K (2019) Use of rhizobacteria and mycorrhizae consortium in the open field as a strategy for improving crop nutrition, productivity and soil fertility. Front Microbiol 10:1106. https://doi.org/10.3389/fmicb.2019.01106
Ruíz-Sánchez M, Armada E, Muñoz Y, García de Salamone IE, Aroca R, Ruíz-Lozano JM, Azcón R (2011) Azospirillum and arbuscular mycorrhizal colonization enhance rice growth and physiological traits under well-watered and drought conditions. J Plant Physiol 168(10):1031–1037. https://doi.org/10.1016/j.jplph.2010.12.019
Saheri F, Barzin G, Pishkar L, Boojar MMA, Babaeekhou L (2020) Foliar spray of salicylic acid induces physiological and biochemical changes in purslane (Portulaca oleracea L.) under drought stress. Biologia 75:2201. https://doi.org/10.2478/s11756-020-00571-2
Santoro MV, Zygadio J, Giordano W, Banchio E (2011) Volatile organic compounds from rhizobacteria increase biosynthesis of essential oils and growth parameters in peppermint (Mentha piperita). Plant Physiol Biochem 49:1177–1182. https://doi.org/10.1016/j.plaphy.2011.07.016
Schonfeld MA, Johnson RC, Carwer BF, Mornhinweg DW (1988) Water relations in winter wheat as drought resistance indicators. Crop Sci 28:526–531. https://doi.org/10.2135/cropsci1988.0011183X002800030021x
Selosse MA, Richard F, He X, Simard SW (2006) Mycorrhizal networks: des liaisons dangereuses? Trends Ecol Evol 21(11):621–628. https://doi.org/10.1016/j.tree.2006.07.003
Sharifi P (2017) The Effect of plant growth promoting rhizobacteria (PGPR), salicylic acid and drought stress on growth indices, the chlorophyll and essential oil of Hyssop (Hyssopus officinalis). Biosci Biotechnol Res Asia 14(3):1033–1042. https://doi.org/10.13005/bbra/2538
Sheng XF (2005) Growth promotion and increased potassium uptake of cotton and rape by a potassium releasing strain of Bacillus edaphicus. J Soil Biol Biochem 37:1918–1922. https://doi.org/10.1016/j.soilbio.2005.02.026
Siemens DH, Garner SH, Mitchell-Olds T, Callaway RM (2002) Cost of defense in the context of plant competition: Brassica rapa may grow and defend. Ecology 83(2):505–517. https://doi.org/10.2307/2680031
Tsuro M, Inoue M, Kameoka H (2001) Variation in essential oil components in regenerated lavender (Lavandula vera DC) plants. Sci Hortic 88(4):309–317. https://doi.org/10.1016/S0304-4238(00)00214-4
Vafadar Fa, Amooaghaie R, Otroshy M (2014) Effects of plant-growth-promoting rhizobacteria and arbuscular mycorrhizal fungus on plant growth, stevioside, NPK, and chlorophyll content of Stevia rebaudiana. J Plant Interact 9:128–136. https://doi.org/10.1080/17429145.2013.779035
VanLoon LC, Glick BR (2004) Increased plant fitness by rhizobacteria. In: Sandermann H (ed) Molecular Ecotoxicology of Plants, Ecological Suites. Springer-Verlag, Berlin, pp 178–205. https://doi.org/10.1007/978-3-662-08818-0_7
Verma RS, Rajendra CP, Chauhan A (2015) Evaluation of essential oil quality of lemon balm (Melissa officinalis L.) grown in two locations of northern India. J Essent Oil Res 27(5):412–416. https://doi.org/10.1080/10412905.2015.1015692
Vickers CE, Gershenzon J, Lerdau MT, Loreto F (2009) A unified mechanism of action for volatile isoprenoids in plant abiotic stress. Nat Chem Biol 5(5):283–291. https://doi.org/10.1038/nchembio.158
Wellburn AR (1994) The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J Plant Physiol 144:307–313. https://doi.org/10.1016/S0176-1617(11)81192-2
Wu C, Qu J, Liu L, Kang H, Sun H, Zhang Y, Ghorbani A, Pehlivan N (2021) Quo vadis: signaling molecules and small secreted proteins from mycorrhizal fungi at the early stage of mycorrhiza formation. Symbiosis 85:123–143. https://doi.org/10.1007/s13199-021-00793-1
Younesi O, Moradi A (2015) Effect on different priming methods on germination and seedling establishment of two medicinal plants under salt stress conditions. Cercet Agro Moldova 48(3):43–51. https://doi.org/10.1515/cerce-2015-0040
Zahir ZA, Munir A, Asghar HN, Shaharoona B, Arshad M (2008) Effectiveness of rhizobacteria containing ACC deaminase for growth promotion of peas (Pisum sativum) under drought conditions. J Microbiol Biotechnol 18:958–963
Zakerian F, Sefidkon F, Abbaszadeh B, Kalate Jari S (2020) Effects of water stress and mycorrhizal fungi on essential oil content and composition of Satureja sahendica Bornm. J Agric Sci Tech 22(3):789–799
Zhang F, Zou YN, Wu QS (2018) Quantitative estimation of water uptake by mycorrhizal extraradical hyphae in citrus under drought stress. Sci Hortic 229:132–136. https://doi.org/10.1016/j.scienta.2017.10.038
Zhang W, Xie Z, Zhang X, Lang D, Zhang X (2019) Growth-promoting bacteria alleviates drought stress of G. uralensis through improving photosynthesis characteristics and water status. J Plant Interact 14(1):580–589. https://doi.org/10.1080/17429145.2019.1680752
Zhu XQ, Tang M, Zhan HQ (2017) Arbuscular mycorrhizal fungi enhanced the growth, photosynthesis, and calorific value of black locust under salt stress. Photosynthetica 55(2):378–385. https://doi.org/10.1007/s11099-017-0662-y
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Eshaghi Gorgi, O., Fallah, H., Niknejad, Y. et al. Effect of Plant growth promoting rhizobacteria (PGPR) and mycorrhizal fungi inoculations on essential oil in Melissa officinalis L. under drought stress. Biologia 77, 11–20 (2022). https://doi.org/10.1007/s11756-021-00919-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-021-00919-2