Abstract
In this research, modified magnetic nanoparticles were prepared to be served as a magnetic dispersive solid phase extraction adsorbent for the simultaneous extraction and preconcentration of some polycyclic aromatic hydrocarbons from different wastewater samples. This method was followed by a dispersive liquid–liquid microextraction procedure to achieve high sensitivity and enrichment factors. Ultimately, the analytes were analyzed by gas chromatography–flame ionization detection. The adsorbent was the octadecylamine co-precipitated by Fe3O4 which exhibited high adsorption capability towards the target compounds. The developed process was performed by dispersion of the octadecylamine@Fe3O4 nanoparticles into an aqueous sample solution containing the target analytes by vortexing. Then, the magnetic adsorbent was collected by applying an external magnetic field followed by adding acetonitrile to elute the analytes from the adsorbent by vortexing for a few minutes. By utilizing an external magnet once again, the adsorbent was separated and the attained eluent was mixed with an extraction solvent at µL-level and speedily injected into deionized water. After centrifugation, an aliquot of the sedimented phase was injected into the separation system. The experimental parameters of both steps were investigated and optimized. Under the optimized extraction conditions, the proposed method provided low limits of detection (0.11–0.30 µg L−1) and quantification (0.40–1.0 µg L−1), and satisfactory precision with relative standard deviations in the ranges of 2–5% for intra- (n = 6) and 3–6% for inter-day (n = 4) precisions. Satisfactory extraction recoveries and high enrichment factors were obtained in the ranges of 63–84% and 506–676, respectively. The performance and results of this research unfold that the developed method is easy, practical, highly efficient, precise, and rewarding as the environmental perspective.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The term of polycyclic aromatic hydrocarbons (PAHs) covers a wide range of potent carcinogenic compounds defined to be comprised of two or more fused aromatic rings (Brazkova and Krastanov 2013; Manousi and Zachariadis 2020). These compounds are prevailing environmental contaminants that are propounded by products of the incomplete combustion of organic substances such as wood, coal, petroleum, and fossil fuels (Zhang et al. 2006). Both natural and anthropogenic activities, urban runoff, industrial processes, exhaust fumes, and wastewaters are different sources of PAHs in the environment that spread over wide areas (Srivastava et al. 2018). It has been postulated that long–term exposure to PAHs is dramatically linked to human health threats such as lung and bladder cancers, reproductive abnormalities, DNA damage, and cardiopulmonary mortality (Kuo et al. 2003; Srogi 2007; Kim et al. 2013). To sum up, considering the biological stability and bioaccumulation risk of these compounds, development of a quick, sensitive, and simple analytical method for monitoring and determination of PAHs in various industrial and environmental wastewaters is highly demanded (Bai et al. 2010; Ma et al. 2010; Ghasemi and Zolfonoun 2013). Up to now, gas chromatography (GC) (Tian et al. 2017) and high performance liquid chromatography (Zhao et al. 2013) were the most successively used analytical instruments for analysis of PAHs. Nevertheless, due to the complexity of sample matrices, endogenous interferences, and also trace levels of PAHs, high proficiency clean up, extraction, and enrichment procedures are needed prior to their analysis (Olatunji et al. 2014; Ishizaki et al. 2010). Recently, magnetic dispersive solid phase extraction (MDSPE) was used as a new form of dispersive solid phase extraction method based on magnetic particles for the extraction and preconcentration of trace analytes (Zhang et al. 2017). In this method, a magnetic adsorbent is dispersed directly into an analyte–containing aqueous solution. After adsorption of the analytes onto the adsorbent, it is separated from the suspension using an external magnetic field and subsequently the supernatant is discarded. By adding an elution solvent the analytes are desorbed from the surface of adsorbent. Compared to the conventional procedures, using magnetic adsorbents makes the sample pretreatment method simple, rapid, and cost–effective because there is no need for some time–consuming stages such as filtration or centrifugation. Low consumption of organic solvents has also great importance from the aspect of green chemistry (Asfaram et al. 2020). In addition, the developed method which is a forward–looking technique for sample preparation thanks to the use of magnetic nanoparticles (MNPs) can be proficient, stable, time–consuming, and also cost–effective (Soylak et al. 2010). Among different MNPs, magnetite (Fe3O4) is the most commonly preferred compound due to its environmentally friendly behavior, highly magnetic property, high–yield synthesis process, favorable morphology, and large surface area due to its nanostructure (Aguilar-Arteaga et al. 2010; Hemmati et al. 2018; Mirzaei et al. 2020; Nejad et al. 2019). But oxidation and low adsorption capability of these naked MNPs impede the accurate processing of them and to avoid these phenomena and also to preserve and enhance their stability, they are usually coated with several materials such as silica (Farajzadeh et al. 2019), carbon nanotubes (Rastkari and Ahmadkhaniha 2013), octadecyl (Liu et al. 2009; Synaridou et al. 2014), etc. Among the mentioned coating materials, C18 modified MNPs offer outstanding points such as excellent stability, long life-time, nontoxicity, and ultimately proposing a fast, feasible, convenient, and highly efficient extraction process (Wang et al. 2012; Katsumata et al. 2006). Moreover, in order to achieve high sensitivity and high enrichment factors (EFs), implementation of dispersive liquid microextraction (DLLME) after MDSPE is recommended (Wu et al. 2019). DLLME is a developed method offering several advantages including easiness, usefulness, high EFs, and good precision. In addition, less solvent-consumption and low cost made it as a very popular sample preparation method. This method can be modified handily and combined with other sample preparation techniques (Rezaee et al. 2006; Zgoła-Grześkowiak and Grześkowiak 2011).
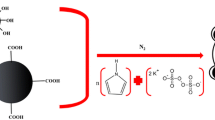
The aim of this study was to find an effective, reliable, and facile analytical method for monitoring and determination of PAHs in industrial and environmental wastewater samples. For this purpose, a combination of MDSPE based on new MNPs and DLLME was developed and optimized. To gratify the qualification of magnetic separation, Fe3O4 particles were selected as the magnetic core and octadecylamine was chosen as the modifier. The utilized magnetic octadecylamine nanoparticles were prepared by co–precipitation method wich has high and scalable reaction yield. The obtained results point that MDSPE based on octadecylamine@Fe3O4 combined with DLLME coupled to GC–flame ionization detection (FID) is a persuasive and quick method for PAHs analysis in wastewater samples.
Experimental
Chemicals and solutions
The studied PAHs consisting of acenaphthene, phenanthrene, anthracene, pyrene, acenaphtylene, and benzo[a]pyrene were obtained from Merck (Darmstadt, Germany). Ferric chloride hexahydrate (FeCl3.6H2O), ferrous sulfate heptahydrate (FeSO4.7H2O), octadecylamine, and ammonia solution (25%, w/w) used in preparation of the adsorbent were also purchased from Merck. Acetonitrile (ACN), acetone, methanol (Merck) and iso-propanol (Caledon, Canada) were tested for desorption of the selected analytes in MDSPE and as the disperser solvents in DLLME. The extraction solvents used in DLLME including 1,2-dibromoethane (1,2-DBE), chloroform, carbon tetrachloride, 1,1,1-trichloroethane (1,1,1-TCE), and 1,1,2-trichloroethane (1,1,2-TCE), all analytical grade, were acquired from Janssen (Beerse, Belgium). Sodium hydroxide, sodium sulfate, hydrochloric acid (37%, w/w), sodium chloride, and sodium nitrate were from Merck. A mixture stock solution of the selected PAHs at a concentration of 100 mg L−1 (each analyte) was prepared in ACN. This solution was diluted with deionized water to prepare working standard solutions.
Samples
Several wastewaters including pharmaceutical, mine, petrochemical, and refinery wastewaters were obtained from Dana Pharmaceutical Company (Tabriz, Iran), Sungun copper mine (Ahar, East Azarbaijan, Iran), Tabriz Petrochemical Company (Tabriz, Iran), and Tabriz Refinery (Tabriz, Iran), respectively. Other aqueous solutions including municipality wastewater and fountain water (Tabriz, Iran) were also investigated.
Apparatus
All chromatographic measurements were carried out with a Shimadzu 2014 gas chromatograph equipped with a split/splitless injector (used in a splitless/split mode, split ratio of 1:10 min, and sampling time of 1 min) and an FID set at 300 °C. Separation of the selected PAHs was carried out on a Zerbon™ capillary column (95% dimethyl, 5% diphenyl poly siloxane, 30 m × 0.25 mm i.d., and a film thickness of 0.25 µm) (Phenomenex, Torrance, CA, USA). Helium (99.999%, Gulf Cryo, United Arab Emirates) was utilized as carrier and make up gases at a linear velocity of 30 cm s−1 and flow rate of 30 mL min−1, respectively. The initial column oven temperature was set at 100 °C for 3.0 min, elevated to 300 °C at a rate of 13 °C min−1, and retained at 300 °C for 10 min. The provision of hydrogen as the fuel for FID was done by a hydrogen generator (OPGU-1500S, Shimadzu, Japan) at a flow rate of 30 mL min−1. Also, the needed air as an oxidant for FID was supplied at a flow rate of 300 mL min−1. Phase separation was carried out by a Hettich centrifuge model D-7200 (Tuttlingen, Germany) and an L46 vortex (Labinco, Breda, the Netherlands) was utilized for vortexing. All pH adjustments of aqueous solutions were performed using a Metrohm pH meter (model 654, Herisau, Switzerland). A magnetic heater stirrer (Heidolph MR 3001 K, Germany) was used in the adsorbent preparation step. X-ray diffraction (XRD) pattern of the prepared adsorbent was recorded by an X-ray diffractometer (Siemens AG, Karlsruhe, Germany). It was performed in the range of 4–73° operated at a voltage of 35 kV. Surface morphology and size of MNPs were studied by scanning electron microscopy (SEM) (Tescan, Czech) and the elemental analysis of the adsorbent was investigated by an energy dispersive X-ray (EDX) spectrometer (Tescan, Brno, Czech). Evaluating magnetic property of the prepared adsorbent was done by a Lake Shore 7304 vibrating sample magnetometer (VSM) (Lakeshore, USA). Fourier transform infrared (FTIR) spectrum was recorded by a Bruker FTIR spectrophotometer (Billerica, USA).
Preparation of sorbent
The magnetic octadecylamine nanocomposite (octadecylamine@Fe3O4) was prepared by a chemical co–precipitation method. Briefly 100 mg of octadecylamine was added to 50 mL sulfuric acid solution (2 mol L−1). In order to dissolve octadecylamine, temperature of the mixture was adjusted at 80 °C and stirred at a rate of 300 rpm for 20 min. After obtaining a clear solution, 0.53 g of FeCl3.6H2O and 0.36 g of FeSO4.7H2O were added to the solution. Afterward, 20 mL concentrated ammonia solution was added dropwise until emerging stable brownish color that is an indication of the generation of Fe3O4 nanoparticles. During the whole process, the solution temperature was maintained at 80 °C and it was stirred at 300 rpm. After 2 h, the produced MNPs were separated in the presence of an external magnet and washed with a mixture of deionized water: ethanol (50:50, v/v) and finally dried at room temperature.
Extraction procedure
MDSPE
Initially, 8 mL of deionized water spiked with 50 µg L−1 of acenaphthene, phenanthrene, pyrene, and acenaphtylene, and 10 µg L−1 of anthracene, and benzo[a]pyrene, or sample (see Section “Samples”) was added into a 12-mL glass test tube and 0.468 g NaCl (1 mol L−1) was dissolved in it. Afterward, 20 mg of the prepared adsorbent was added and vortexed for 5 min. Then, the PAH-loaded octadecylamine@Fe3O4 MNPs was isolated by placing a magnet at the bottom of the tube and aqueous phase was discarded. Subsequently, 1.5 mL ACN was added on the collected adsorbent and vortexed for 3 min to elute the selected analytes. The eluent was isolated from the adsorbent in the presence of external magnetic field and utilized in the subsequent DLLME step as a disperser solvent.
DLLME
The eluent obtained from the MDSPE step was mixed with 26 µL 1,2-DBE as the extraction solvent and speedily injected into 5 mL sodium chloride solution (1 mol L−1) placed in a 10-mL conical bottom glass test tube using a 2-mL glass syringe. Accordingly, a cloudy solution was emerged and centrifuged for 5 min at 7000 rpm. Eventually, 1 µL of the sedimented phase (10 ± 0.5 µL) was taken and injected into GC–FID system.
It is worth mentioning that, DLLME is an efficient microextraction process which benefits from brief extraction procedure, being inexpensive, and containing high enrichment factors (EFs) for analytes. But, actually the principle drawback of DLLME is its non-selective extraction feature that matrix components are also migrated into the extraction solvent resulting in noisy chromatograms which eventually makes it difficult to detect and determine the target compounds at low concentrations. So, suffering from high matrix effect and incomparable relative recovery data with deionized water due to the matrix complexity of the surveyed samples of this study (sewage, wastewater, and fountain samples) makes it impossible to apply just a DLLME procedure for the extraction of the analytes. For performing a clean-up step and relatively selective sorption of the analytes, application of MDSPE is inevitable. Also, DLLME is not directly applicable in real samples having suspended solids such as sewage samples. So, there is a high necessity for another extraction method to be coupled to DLLME in order to reduce the matrix effect of complex samples. Moreover, as you know, MDSPE suffers from lack of enrichment factor. This is due to the fact that the volume of the elution solvent in this study is 1.5 mL which is not analyte-enriched enough. So, the signals of the analytes would be small and consequently achieving low LODs and LOQs would be impossible. Due to the fact that concentrations of the studied analytes are at µg L−1 level, detection and determination of such low concentrations are impossible by applying only MDSPE method. In this method, the adsorbent is directly added and dispersed into the sample solution and the clean-up goal was achieved by adsorbing the target compounds. Therefore, matrix effect is diminished and reaching lower LOD and LOQ values becomes possible. According to these facts, the developed process tries to benefit from the advantages of both MDSPE and DLLME. So, the combination of the two methods led to an effective method for the successful detection and determination of the analytes.
EF and extraction recovery (ER) calculation
EF is stated as the ratio of analyte concentration in the extracted phase (Csed) in DLLME step to its initial spiked concentration in the aqueous solution (C0).
Additionally, ER is calculated using Eq. (2) which denotes the percentage of the migrated amount of analyte into the extractant (nsed) to the amount of analyte in the initial aqueous solution (n0).
where Vsed is volume of the sedimented phase and Vaq is volume of the aqueous solution.
Results and discussion
Characterization of sorbent
Characterization of the prepared MNPs was performed using FTIR, SEM, EDX, VSM, and XRD techniques. Figure 1a, b show the FTIR spectra of Fe3O4 and octadecylamine@Fe3O4 MNPs, respectively. This technique is utilized to assure the desired chemical modification on the surface of MNPs. As it is clear from both spectra the absorption peak at 591.80 cm−1 represents the Fe–O stretching vibration in magnetite. In comparison to Fig. 1a, the advent of new peaks at 1115.06, 1405.19, and 1621.28 cm−1 in Fig. 1b is related to C–N stretching, C–H bending, and N–H bending bonds, respectively, confirming the successful modification of Fe3O4 MNPs with octadecylamine. The two absorption bonds around 3041.59 cm−1 are assigned to N–H vibrations of the primary amino group. The characteristic peaks at 2847.81 and 2916.38 cm−1 are corresponded to the asymmetric and symmetric extension vibrations of –CH2 in –(CH2)17CH3 chain, respectively (Farajzadeh et al. 2020; https://www.sigmaaldrich.com/US/en/technical-documents/technical-article/analytical-chemistry/photometry-and-reflectometry/ir-spectrum-table.html). The morphological features and size observation of the prepared adsorbent were examined by SEM technique. The obtained results show the agglomeration of nearly spherical–shaped particles (Fig. 1c) with a mean diameter of 40 nm (Fig. 1d) revealing a perfect platform for the adsorption of the analytes. Moreover, the performed elemental analysis on the product indicates the peaks denoting Fe, C, O, and N atoms with weight percentages of 21.60, 24.02, 42.82, and 9.00%, respectively (Fig. 1e). The outcome from VSM analysis illustrates the S-like magnetic hysteresis loop analysis of the adsorbent (Fig. 1f). The saturation magnetization value of the product was found to be 32 emu g−1 evincing its suitable magnetic property. Figure 1g also illustrates the XRD pattern of the final product. As can be observed, diffraction lines occur at 2θ angles of 32.84, 35.8, and 63.16°. These peaks indicate that the modification of Fe3O4 to form octadecylamine nanoparticles occurs without any phase change in the magnetite particles and the synthesized MNPs have a cubic crystalline structure (Hu et al. 2018; Maddah and Shamsi 2012).
Optimization of parameters in MDSPE
Optimization of octadecylamine weight in the prepared nanocomposite
Fe3O4 nanoparticles were coated with octadecylamine to prevent the Fe3O4 nanoparticles oxidation in contact with air and to increase the selectivity of the adsorbent towards the studied analytes. It should be mentioned that by using bare Fe3O4 nanoparticles’ in the extraction process, the ERs of analytes were in the range of 5–10% while after modification with ODA the ERs were in the range of 63–84%. Therefore, Fe3O4 nanoparticles act as the magnetic core of the synthesized adsorbent and ODA is responsible for adsorption of the analytes in MDSPE step. So, the amount of modifier in the nanocomposite plays a critical role on ERs of the analytes. Accordingly, several amounts of octadecylamine (50, 100, 150, and 200 mg) were examined to be present in the prepared octadecylamine@Fe3O4 nanocomposite having 232 mg Fe3O4. The results (Fig. 2) indicate that ER values increase up to 100 mg and then decrease noticeably. Due to the mentioned points, excessive use of ODA, decreases the magnetic property of the nanocomposite led to the deficient collection of the adsorbent particles in the presence of external magnetic field. Hence 100 mg was sufficient to certify an adequate efficiency and hence it was selected as the optimal amount of the octadecylamine for preparation of the adsorbent.
Optimization of octadecylamine weight. Extraction conditions: MDSPE procedure: aqueous sample, 8 mL deionized water spiked with 50 µg L−1 of each PAH; adsorbent weight, 30 mg; adsorption vortex time, 5 min; elution solvent, ACN (1 mL); and vortex time in desorption step, 3 min. DLLME procedure: 5 mL deionized water; extraction solvent, Chloroform (48 µL); centrifugation rate, 7000 rpm; and centrifugation time, 5 min. Both steps were done without any salt addition or pH adjustment. The error bars indicate standard deviation of three repeated determinations
Optimization of adsorbent weight
The amount of adsorbent was considered to be correlated to the number of active sites, and quantity of the adsorbed analytes. Accordingly, to access the optimum results, several amounts of the MNPs varying in the range of 5–50 mg were studied. As shown in Fig. 3, 20 mg of the adsorbent is sufficient to assure an acceptable efficiency, hence it was selected as the optimum value. The decrease in the analytical signals at high weights occurs because of agglomeration of the adsorbent (lack of its efficient dispersion into the aqueous solution containing the analytes) or incomplete elution of the analytes during the desorption step. So, 20 mg was selected as the optimum sorbent weight to continue the optimization steps.
Optimization of octadecylamine@Fe3O4 amount. Extraction conditions: All conditions are the same as those used in Fig. 2, except 100 mg of octadecylamine was utilized in preparation of the adsorbent
Effect of salt addition
Salt addition is notably applied for the augment of extraction efficiency due to dwindling solubility of the selected analytes in aqueous solution (known as salting-out effect) and hence increasing their migration onto the sorbent surface. However, high concentrations of salt can increase viscosity of aqueous phase which can unfavorably decrease the analytes diffusion coefficients and adversely affect the extraction efficiency (known as salting-in effect). Due to the stated reasons NaCl, NaNO3, and Na2SO4 were added separately at a concentration of 1 mol L−1 (each salt) into the aqueous phase including the analytes. Among the tested salts, NaCl provides the highest ERs for the selected PAHs (Fig. 4). Moreover, to study the salt concentration, several concentrations of it (0.5, 1.0, 1.5, and 2.0 mol L−1) were studied. The obtained analytical results depicted that by increasing NaCl concentration up to 1 mol L−1, ERs of the analytes enhanced and then steadily dropped (data not shown here). It is obvious that salt addition has salting-out effect until 1 mol L−1 salt concentration and then salting–in effect emerges more dominant. With regard to the obtained results, 1 mol L−1 NaCl was chosen for the further investigations.
Study of ionic strength. Extraction conditions: are the same as those used in Fig. 3, except 20 mg of the adsorbent was utilized
Effect of solution pH
Efficiency of the proposed method in the extraction and preconcentration of the selected analytes can be altered with varying solution pH because it not only influences the adsorbent surface charge but also stability of the MNPs. Since the studied PAHs are neutral and do not ionize, it is anticipated that no considerable effect of pH would be found due to the negligible impact of pH on them. Therefore, pH of the aqueous solution was varied in the range of 3–13 (with 2-unit intervals) by using 1 mol L−1 HCl or NaOH solutions. The results (data not shown here) indicated that the highest ERs were obtainable at the pH range of 5–9 which is well assorted with the pH of all sample solutions utilized in the present study. So, as it was expected the further analyses were performed without any pH adjustment.
Optimization of vortexing time in adsorption step
In adsorptive-based analytical methods an appropriate vortexing time is necessary for acquiring high ERs because it leads to enough contact surface area between the adsorbent and analytes. To fulfill the effect of this parameter, 1–6 min vortexing time periods were tested. ERs of the analytes increased by increasing the vortex time until 5 min and remained constant in the vortex time of 6 min (data not shown here). Therefore, 5 min vortexing was selected for the further investigations.
Optimization of the type and volume of desorption/disperser solvent
In order to obtain an efficacious desorption of the adsorbed PAHs from the surface of the nanocomposite, opting an appropriate eluent is of great importance. Moreover, it can influence dispersion efficiency of the extraction solvent used in DLLME step into aqueous phase. Additionally, the chosen solvent should be miscible in both aqueous solution and extraction solvent. For this purpose, 1 mL of different solvents including ACN, methanol, acetone, and iso-propanol were tested. Referring to the results in Fig. 5, the highest ERs are achieved in the presence of ACN as the desorption/disperser solvent. Furthermore, to investigate the sufficient volume of the opted disperser solvent, its volume was studied in the range of 0.50–2.0 mL. Based on the obtained results (data not shown here), 1.5 mL of ACN was superior over the other studied volumes and it was selected as the optimum volume of the desorption/disperser solvent for the following experiments.
Selection of desorption/disperser solvent type. Extraction conditions: are the same as those utilized in Fig. 4, except 1 mol L−1 NaCl was used in MDSPE step
Optimization of vortexing time in desorption step
To obtain an optimum time for desorption of the analytes, various vortexing times were tested in the range of 1–5 min. Considering the outcomes (data not shown here) vortexing for 3 min was the best time for elution of the selected PAHs from the surface of the adsorbent.
Optimization of parameters in DLLME
Selection of the type and volume of extraction solvent
Selection of a suitable extraction solvent is the pivotal parameter in DLLME-based methods. It has to acquire various criteria including low solubility in aqueous phase, having higher density than water, high extraction efficiency towards the target PAHs, and forming a stable cloudy state in the presence of ACN. Based on these points 1,2-DBE, 1,1,1-TCE, 1,1,2-TCE, chloroform, and carbon tetrachloride with the volumes of 26, 31, 30, 48, and 26 µL, respectively, were examined to get 10 ± 0.5 µL of the sedimented phase after centrifugation. Regarding the outcomes illustrated in Fig. 6, 1,2-DBE is preferred over the surveyed extractants due to resulting higher ER values. Subsequently, the effect of extraction solvent volume on the EF values was tested using several volumes of 1,2-DBE (26, 31, 36, 41, and 46 µL). It is noted that increasing volume of the extraction solvent in the mentioned range led to obtaining 10, 13, 15, 19, and 23 µL of the sedimented phase, respectively, at the end of the procedure. So, the EF values decreased by increasing the extractant volume due to dilution effect (data not shown here). Accordingly, the volume of 26 µL was chosen as the optimum volume of the extraction solvent.
Selection of extraction solvent type in DLLME. Extraction conditions: are the same as those used in Fig. 5, except 1.5 mL ACN was used as the desorption/disperser solvent
Ionic strength study
As explained before, salt addition can improve extraction efficiency of the method by decreasing solubility of the selected analytes in aqueous solution and also it can decrease mass transfer rate of the analytes via increasing viscosity of the aqueous solution which results in decreasing the extraction efficiency. To investigate this parameter, several salts including NaCl, Na2SO4, and NaNO3 (1 mol L−1 of each) were studied in which NaCl utilization provided higher ERs (data not shown here) similar to the DSPE step. Then various concentrations of NaCl (0.5, 1.0, 1.5, and 2.0 mol L−1) were added to aqueous solution and in order to keep volume of the sedimented phase constant, different volumes of 1,2-DBE (31, 36, 41, and 46 µL) were used for 0.5, 1.0, 1.5, and 2.0 mol L−1 of NaCl, respectively. The outcomes (Fig. 7) evince that ERs of the target PAHs increase by increasing the concentration of NaCl up to 1 mol L−1 and then decrease slightly by the further increasing NaCl concentration. Hence, 1 mol L−1 NaCl was selected.
Study of NaCl concentration in DLLME step. Extraction conditions: All conditions are the same as those used in Fig. 6, except 1,2-DBE was used as the extraction solvent
Reusability of the sorbent
Reusability is an important factor when evaluating the efficiency of sorbents. In order to investigate the reusability of the sorbent, repetitive extractions were done using the same sorbent according to Section “Extraction procedure”. Before repeating the experiment, the recycled sorbent was washed twice using ACN (each time by 1 mL along with vortexing for 3 min) to prevent carry-over. The obtained results indicated that the sorbent was capable to adsorb the analytes after three desorption cycles without a significant change in the ERs of the analytes which indicates that octadecylamine@Fe3O4 is a suitable sorbent for usage in repetitive analytical cycles.
Validation of the developed analytical method
The practical applicability of the proposed MDSPE–DLLME–GC–FID method was validated through evaluating various analytical values including ER, EF, linear range (LR), limits of detection (LOD) and quantification (LOQ), and relative standard deviation (RSD). The LOD is the lowest concentration of the analyte in a sample that can still be detected by applying the analytical method and using the analytical detection system but should not be quantified as an appropriate value. However, the LOQ is the lowest concentration of the sample that can still be quantitatively determined with acceptable precision and accuracy. The most common method to estimate the LOD of a chromatographic method is to measure the signal of the smallest detected analyte peak with a height equals to three times of the neighbor noise \(\left( {\frac{S}{N} = 3} \right)\). The concentration related to this signal is called LOD. On the other hand, LOQ is determined by a signal-to-noise ratio of 10. So, the concentration in which the resulted signal is 10 times taller than the neighbor noise is called LOQ. To evaluate these figures of merit several solutions with different spiked concentration of the analytes were prepared and injected into GC–FID after performing the method on them. For each of analytes, the spiked concentrations in which the signal-to-noise ratios found to be 3 and 10, were chosen as LODs and LOQs, respectively.
The obtained results of the above–mentioned figures of merit are listed in Table 1. The LODs and LOQs for the selected PAHs (based on signal-to-noise ratios of 3 and 10, respectively) were found to be in the ranges of 0.11–0.30 and 0.40–1.0 µg L−1, respectively. EF values of the proposed approach (calculated from Eq. 1) were in the range of 506–676. Also, the ERs (calculated from Eq. 2) were between 63 and 84%. Intra- (n = 6) and inter-day (n = 4) RSDs at a concentration of 10 µg L−1 of each PAH were in the ranges of 2–5 and 3–6%, respectively. The method presented wide LRs with a good linearity (r2 ≥ 0.991) for the target analytes. The listed validation parameters including low LODs and LOQs, good repeatability, and high EFs and ERs are the significant assurance of an acceptable and reliable approach.
Analysis of real samples
In order to evaluate efficiency of the modified Fe3O4 MNPs for the successful clean up and also applicability of the MDSPE–DLLME procedure, the validated method was further assessed with analyzing several real samples including pharmaceutical sewage, sewage of the refinery unit of copper mine, sewage of distillation unit (petrochemical and refinery wastewaters), municipality wastewater, and fountain water samples. According to the outcomes, none of the target analytes was detected. Figure 8 illustrates GC–FID chromatograms of standard solution of the analytes (100 mg L−1 of each PAHs), aqueous standard solution (50 µg L−1 of acenaphthene, phenanthrene, pyrene, and acenaphtylene, and 10 µg L−1 of anthracene, and benzo[a]pyrene) and all real samples after performing the method on them, except the standard solution prepared in 1,2-DBE in which direct injection without preconcentration was done. Moreover, to investigate the method’s accuracy and matrix effect in various samples, added-found method was employed. According to the relative recovery values (86–114%) (Table 2) obtained at the spiked concentrations of 5 and 10 µg L−1 of ach PAH, the matrices of samples have a negligible effect on the method’s performance. Hence, the proposed method is applicable for the determination of the target PAHs in the studied samples.
Typical GC–FID chromatograms of a direct injection of standard solution (100 mg L−1 in 1,2-DBE, each PAH), b deionized water spiked with 50 µg L−1 of acenaphthene, phenanthrene, pyrene, and acenaphtylene, and 10 µg L−1 of anthracene, and benzo[a]pyrene, c pharmaceutical sewage, d mine wastewater e petrochemical wastewater f refinery wastewater g municipality wastewater and h fountain water. In all cases, except chromatogram (a) the proposed method was performed and 1 µL of the sedimented phase was injected into the separation system. Peaks identification: (1) acenaphthene, (2) phenanthrene, (3) anthracene, (4) pyrene, (5) acenaphtylene, and (6) benzo[a]pyrene
Comparison of the method with other approaches
The analytical figures of merit of the developed extraction method in combination with GC–FID for the determination of the selected PAHs were compared with the other similar analytical methods. As it can be concluded from the listed results in Table 3, the proposed method has better RSDs, lower LODs (in most cases), and wider LRs in comparison to the other methods. Also the obtained EF values of the method are higher than the EFs of the mentioned methods. It is worthwhile to notify that the presented method benefits from several aspects such as good precision, easy operation, good repeatability, high EFs, and low LODs and LOQs. Moreover, utilizing a magnetic and efficient adsorbent accelerates separation process via avoiding laborious and time-consuming centrifugation or filtration procedures. Regarding to all aforementioned points, the developed method seems to be accessible, quick, and sensitive, and it can be applied effectively for monitoring the selected PAHs in various industrial wastewater samples.
Conclusions
The proposed MDSPE method based on a new magnetic adsorbent (octadecylamine@Fe3O4) was an effective, convenient, and rapid method for the simultaneous extraction and preconcentration of PAHs in several obscure matrices such as environmental and industrial wastewater samples. On account of effectual hydrophobic–hydrophobic interactions, PAHs were adsorbed by the C18 group of the nanoparticles. The GC–FID method combined with MDSPE–DLLME acquired low LODs (0.11–0.30 µg L−1) and LOQs (0.40–1.0 µg L−1), satisfactory repeatability (RSD ≤ \(6\% )\), and high EFs (506–676). Taking into account that the desired adsorbent inherits virtues of being cost–effective, easy for preparation, having higher efficiency, and easy to separate and dispersion, this method is considered to be promising for the detection and determination of PAHs and other pollutants from various real samples.
Change history
04 June 2024
A Correction to this paper has been published: https://doi.org/10.1007/s11696-024-03483-6
Abbreviations
- DLLME:
-
Dispersive liquid–liquid microextraction
- EF:
-
Enrichment factor
- ER:
-
Extraction recovery
- FID:
-
Flame ionization detection
- GC:
-
Gas chromatography
- LOD:
-
Limit of detection
- LOQ:
-
Limit of quantification
- MDSPE:
-
Magnetic dispersive solid phase extraction
- PAHs:
-
Polycyclic aromatic hydrocarbons
- RSD:
-
Relative standard deviation
References
Aguilar-Arteaga K, Rodriguez JA, Barrado E (2010) Magnetic solids in analytical chemistry: a review. Anal Chim Acta 674:157–165. https://doi.org/10.1016/J.ACA.2010.06.043
Asfaram A, Dil EA, Arabkhani P, Sadeghfar F, Ghaedi M (2020) Magnetic Cu: CuO–GO nanocomposite for efficient dispersive micro-solid phase extraction of polycyclic aromatic hydrocarbons from vegetable, fruit, and environmental water samples by liquid chromatographic determination. Talanta 218:121131. https://doi.org/10.1016/j.talanta.2020.121131
Bai L, Mei B, Guo QZ, Shi ZG, Feng YQ (2010) Magnetic solid-phase extraction of hydrophobic analytes in environmental samples by a surface hydrophilic carbon-ferromagnetic nanocomposite. J Chromatogr A 1217:7331–7336. https://doi.org/10.1016/j.chroma.2010.09.060
Brazkova M, Krastanov A (2013) Polycyclic aromatic hydrocarbons: sources, effects and biodegradation. In: International scientific conference of university of Ruse, Razgrad, Bulgaria, vol 52
Farajzadeh MA, Afshar Mogaddam MR, Feriduni B (2016) Simultaneous synthesis of a deep eutectic solvent and its application in liquid–liquid microextraction of polycyclic aromatic hydrocarbons from aqueous samples. RSC Adv 6:47990–47996. https://doi.org/10.1039/C6RA04103E
Farajzadeh MA, Safi R, Yadeghari A (2019) Combination of QuEChERS extraction with magnetic solid phase extraction followed by dispersive liquid–liquid microextraction as an efficient procedure for the extraction of pesticides from vegetable, fruit, and nectar samples having high content of solids. Microchem J 147:571–581. https://doi.org/10.1016/j.microc.2019.03.074
Farajzadeh MA, Pezhhanfar S, Zarei M, Mohebbi A (2020) Simultaneous elimination of diethyl phthalate, butylated hydroxy toluene and butylated hydroxy anisole from aqueous medium by an adsorption process on pretreated waste material; investigation of isotherms and neural network modeling. J Iran Chem Soc 17:1377–1386. https://doi.org/10.1007/s13738-020-01863-9
Ghasemi JB, Zolfonoun E (2013) Simultaneous spectrophotometric determination of trace amount of polycyclic aromatic hydrocarbons in water samples after magnetic solid-phase extraction by using projection pursuit regression. Environ Monit Assess 185:2297–2305. https://doi.org/10.1007/s10661-012-2709-7
Hemmati M, Rajabi M, Asghari A (2018) Magnetic nanoparticle based solid-phase extraction of heavy metal ions: a review on recent advances. Microchim Acta. https://doi.org/10.1007/s00604-018-2670-4
Hosseini MH, Rezaee M, Akbarian S, Mizani F, Pourjavid MR, Arabieh M (2013) Homogeneous liquid-liquid microextraction via flotation assistance for rapid and efficient determination of polycyclic aromatic hydrocarbons in water samples. Anal Chim Acta 762:54–60. https://doi.org/10.1016/j.aca.2012.10.030
Hou L, Lee HK (2002) Application of static and dynamic liquid-phase microextraction in the determination of polycyclic aromatic hydrocarbons. J Chromatogr A 976:377–385. https://doi.org/10.1016/S0021-9673(02)01152-4
https://www.sigmaaldrich.com/US/en/technical-documents/technical-article/analytical-chemistry/photometry-and-reflectometry/ir-spectrum-table.html. Accessed 1 October 2021
Hu K, Qiao J, Wu X, Yang H, Huang Y, Zhang S (2018) Poly (calixarene ionic liquid) modified Fe3O4 nanoparticles as new sorbent for extraction of flavonoids in fruit juice and green tea. Microchem J 143:39–46. https://doi.org/10.1016/j.microc.2018.07.029
Ishizaki A, Saito K, Hanioka N, Narimatsu S, Kataoka H (2010) Determination of polycyclic aromatic hydrocarbons in food samples by automated on-line in-tube solid-phase microextraction coupled with high-performance liquid chromatography-fluorescence detection. J Chromatogr A 117:5555–5563. https://doi.org/10.1016/j.chroma.2010.06.068
Juan L, Jared L, Chisvert A (2017) Trace determination of volatile polycyclic aromatic hydrocarbons in natural waters by magnetic ionic liquid-based stir bar dispersive liquid microextraction. Talanta 17:30813–30815. https://doi.org/10.1016/j.talanta.2017.07.091
Katsumata H, Asai H, Kaneco S, Suzuki T, Ohta K (2006) Determination of linuron in water samples by high performance liquid chromatography after preconcentration with octadecyl silanized magnetite. Microchem J 85:285–289. https://doi.org/10.1016/j.microc.2006.07.001
Kim KH, Jahan SA, Kabir E, Brown RJC (2013) A review of airborne polycyclic aromatic hydrocarbons (PAHs) and their human health effects. Environ Int 60:71–80. https://doi.org/10.1016/j.envint.2013.07.019
Kuo CY, Hsu YW, Lee HS (2003) Study of human exposure to particulate PAHs using personal air samplers. Arch Environ Contam Toxicol 44:454–459. https://doi.org/10.1007/s00244-002-1177-4
Liu Y, Li H, Lin JM (2009) Magnetic solid-phase extraction based on octadecyl functionalization of monodisperse magnetic ferrite microspheres for the determination of polycyclic aromatic hydrocarbons in aqueous samples coupled with gas chromatography–mass spectrometry. Talanta 77:1037–1042. https://doi.org/10.1016/j.talanta.2008.08.013
Ma J, Xiao R, Li J, Yu J, Zhang Y, Chen L (2010) Determination of 16 polycyclic aromatic hydrocarbons in environmental water samples by solid-phase extraction using multi-walled carbon nanotubes as adsorbent coupled with gas chromatography–mass spectrometry. J Chromatogr A 1217:5462–5469. https://doi.org/10.1016/j.chroma.2010.06.060
Maddah B, Shamsi J (2012) Extraction and preconcentration of trace amounts of diazinon and fenitrothion from environmental water by magnetite octadecylsilane nanoparticles. J Chromatogr A 1256:40–45. https://doi.org/10.1016/j.chroma.2012.07.085
Manousi N, Zachariadis GA (2020) Recent advances in the extraction of polycyclic aromatic hydrocarbons from environmental samples. Molecules 25:1–29. https://doi.org/10.3390/molecules25092182
Mirzaei F, Mohammadi Nilash M, Sepahvand H, Fakhari AR, Shaabani A (2020) Magnetic solid-phase extraction based on fluconazole-functionalized Fe3O4@SiO2 nanoparticles for the spectrophotometric determination of cationic dyes in environmental water samples. J Iran Chem Soc 17:1591–1600. https://doi.org/10.1007/s13738-020-01889-z
Moja SJ, Mtunzi F, Madlanga X (2013) Determination of polycyclic aromatic hydrocarbons (PAHs) in river water samples from the Vaal Triangle area in South Africa. J Environ Sci Health Part A Toxic/hazard Subst Environ Eng 48:847–854. https://doi.org/10.1080/10934529.2013.761477
Nascimento MM, Rocha GO, Andrade JB (2019) Simple and effective dispersive micro-solid phase extraction procedure for simultaneous determination of polycyclic aromatic compounds in fresh and marine waters. Talanta 204:776–791. https://doi.org/10.1016/j.talanta.2019.06.061
Nejad LM, Pashaei Y, Daraei B, Forouzesh M, Shekarchi M (2019) Graphene oxide-based dispersive-solid phase extraction for preconcentration and determination of ampicillin sodium and clindamycin hydrochloride antibiotics in environmental water samples followed by HPLC–UV detection. Iran J Pharm Res 18:642–657. https://doi.org/10.22037/ijpr.2019.1100676
Olatunji OS, Fatoki OS, Opeolu BO, Ximba BJ (2014) Determination of polycyclic aromatic hydrocarbons (PAHs) in processed meat products using gas chromtography–flame ionzation detector. Food Chem 156:296–300. https://doi.org/10.1016/j.foodchem.2014.01.120
Ramos-Dorta CV, Pino V, Afonso AM (2013) Monitoring polycyclic aromatic hydrocarbons in seawaters and wastewaters using a dispersive liquid-liquid microextraction method. Environ Technol 34:607–616. https://doi.org/10.1080/09593330.2012.710255
Rastkari N, Ahmadkhaniha R (2013) Magnetic solid-phase extraction based on magnetic multi-walled carbon nanotubes for the determination of phthalate monoesters in urine samples. J Chromatogr A 1286:22–28. https://doi.org/10.1016/j.chroma.2013.02.070
Rezaee M, Assadi Y, Milani Hosseini MR, Aghaee E, Ahmadi F, Berijani S (2006) Determination of organic compounds in water using dispersive liquid–liquid microextraction. J Chromatogr A 1116:1–9. https://doi.org/10.1016/j.chroma.2006.03.007
Soylak M, Ozalp O, Uzcan F (2010) Magnetic nanomaterials for the removal, separation and preconcentration of organic and inorganic pollutants at trace levels and their practical applications: a review. Trends Environ Anal Chem 29:e00109. https://doi.org/10.1016/j.teac.2020.e00109
Srivastava P, Sreekrishnan TR, Nema AK (2018) Polyaromatic hydrocarbons: review of a global environmental issue. J Hazard Toxic Radioact Waste 22:04018004. https://doi.org/10.1061/(asce)hz.2153-5515.0000391
Srogi K (2007) Monitoring of environmental exposure to polycyclic aromatic hydrocarbons: a review. Environ Chem Lett 5:169–195. https://doi.org/10.1007/s10311-007-0095-0
Synaridou MES, Sakkas VA, Stalikas CD, Albanis TA (2014) Evaluation of magnetic nanoparticles to serve as solid-phase extraction sorbents for the determination of endocrine disruptors in milk samples by gas chromatography mass spectrometry. J Chromatogr A 1348:71–79. https://doi.org/10.1016/j.chroma.2014.04.092
Tian Y, Zhou J, Feng J, Wang X, Luo C, Sun M (2017) A silver fibre prepared by a facile method for solid-phase microextraction of polycyclic aromatic hydrocarbons. Environ Chem 14:451–457. https://doi.org/10.1071/EN17138
Wang Q, Huang L, Yu P, Wang J, Shen S (2012) Magnetic solid–phase extraction and determination of puerarin in rat plasma using C18-functionalized magnetic silica nanoparticles by high performance liquid chromatography. J Chromatogr B Anal Technol Biomed Life Sci 912:33–37. https://doi.org/10.1016/j.jchromb.2012.11.009
Wu J, Zhi S, Jia C, Li X, Zhu X, Zhao E (2019) Dispersive solid-phase extraction combined with dispersive liquid–liquid microextraction for simultaneous determination of seven succinate dehydrogenase inhibitor fungicides in watermelon by ultra high performance liquid chromatography with tandem mass spectrometry. J Sep Sci 42:3688–3696. https://doi.org/10.1002/jssc.201900862
Zgoła-Grześkowiak A, Grześkowiak T (2011) Dispersive liquid–liquid microextraction. TrAC Trends Anal Chem 30:1382–1399. https://doi.org/10.1016/j.trac.2011.04.014
Zhang HB, Luo YM, Wong MH, Zhao QG, Zhang GL (2006) Distributions and concentrations of PAHs in Hong Kong soils. Environ Pollut 141:107–114. https://doi.org/10.1016/j.envpol.2005.08.031
Zhang Y, Zhou H, Zhang Zh, Wu X, Chen W, Zhu Y, Fang Ch, Zhao Y (2017) Three-dimensional ionic liquid functionalized magnetic graphene oxide nanocomposite for the magnetic dispersive solid phase extraction of 16 polycyclic aromatic hydrocarbons in vegetable oils. J Chromatogr A 1489:29–38. https://doi.org/10.1016/j.chroma.2017.02.010
Zhao WJ, Bin Chen X, Fang L, Li CL, Zhao DY (2013) Determination of light–medium–heavy polycyclic aromatic hydrocarbons in vegetable oils by solid-phase extraction and high-performance liquid chromatography with diode array and fluorescence detection. J Agric Food Chem 61:1804–1809. https://doi.org/10.1021/jf3052779
Acknowledgements
The authors appreciatively acknowledge the University of Tabriz for financial support.
Funding
Mir Ali Farajzadeh has received research grants from the University of Tabriz.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Mir Ali Farajzadeh declares that he has no competing interest. Nasim Fazli declares that she has no competing interest. Sakha Pezhhanfar declares that he has no competing interest. Mohammad Reza Afshar Mogaddam declares that he has no competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Farajzadeh, M.A., Fazli, N., Pezhhanfar, S. et al. Facile and rapid preparation of magnetic octadecylamine nanocomposite and its application as a capable adsorbent in magnetic dispersive solid phase extraction of some polycyclic aromatic hydrocarbons from wastewater samples. Chem. Pap. 77, 781–794 (2023). https://doi.org/10.1007/s11696-022-02493-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-022-02493-6