Abstract
Purpose
Postoperative nausea and vomiting (PONV) is a frequent unappealing laparoscopic sleeve gastrectomy (LSG) sequel. The study’s purpose was to determine the prevalence, risk factors of PONV, and management of PONV after LSG.
Patients and Methods
This multicenter retrospective study included patients with morbid obesity who had LSG between January 2022 and April 2023. The age range for LSG was 16 to 65 years, and the eligibility requirements included morbid obesity according to international guidelines.
Results
PONV was experienced by 74.6% of patients who underwent LSG at 6 h postoperative. Multivariate analysis revealed that female gender, smokers, preoperative GERD, gastropexy, and severity of pain were found to be independent risk variables of the development of PONV, while antral preservation, opioid-free analgesia, and intraoperative combined analgesia were found to be independent protective variables against the development of PONV. Combined intravenous ondansetron and metoclopramide improved 92.6% of patients who developed PONV. Dexamethasone and antihistamines drugs are given for 42 cases with persistent PONV after using intravenous ondansetron and metoclopramide. Pain management postoperatively by opioid-free analgesia managed PONV. Helicobacter pylori status has no role in the development of PONV after LSG.
Conclusion
Female gender, smoking, presence of preoperative GERD, gastropexy, and severity of pain were found to be independent risk variables of the development of PONV, while antral preservation, opioid-free analgesia, and intraoperative combined analgesia were observed to be independent protective factors against the occurrence of PONV. Combined intravenous ondansetron and metoclopramide improved PONV. Dexamethasone and antihistamines drugs are given for persistent PONV.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Laparoscopic sleeve gastrectomy (LSG) is one of the most common and effective surgical interventions for the treatment of morbid obesity [1]. The technical feasibility of LSG, excellent results in terms of weight reduction and comorbidity improvement, and safety are all factors contributing to its rising popularity. Regardless of the numerous advantages of LSG, a variety of postoperative morbidities, such as staple line leakage, bleeding, gastroesophageal reflux disease (GERD), and postoperative nausea and vomiting (PONV), have been observed [1,2,3].
PONV is a frequent unappealing LSG sequel. This complication can result in prolonged hospital stays, increased medical costs, increased rates of hospital readmission, and increased patient dissatisfaction. According to studies, 60 to 80% of individuals who have LSG may develop PONV. The young patients, female gender, history of motion sickness, non-smoking patients, use of volatile anesthetics, postoperative opioid usage, duration of operation, induced pneumoperitoneum, antral resection, increased intragastric pressure after LSG, and dilated proximal pouch are predisposing factors for PONV [4,5,6,7]. Before surgery, the risk of PONV following bariatric surgery may be predicted using the Apfel score [7]. On the other hand, the PONV impact scale score is used to determine the real PONV after surgery [8]. Preventing and treating PONV and pain in the early postoperative phase with the proper medication regimen is essential to lowering patients’ morbidity after bariatric surgery operations [4].
PONV treatment is improved by a multimodal strategy and a combination of medications. In comparison to a single-agent method, a combined antiemetic prophylactic approach had superior efficacy in decreasing PONV [9,10,11]. The most frequently used medications in this strategy were dexamethasone, metoclopramide, droperidol, or a serotonin receptor antagonist. In several types of research, dexamethasone and antihistamines like promethazine and cyclizine were used as preventative drugs during anesthesia in bariatric procedures. Following LSG, multidisciplinary pain treatment significantly lowers postoperative opioid consumption and enhances the recovery process [6,7,8,9,10].
PONV in bariatric surgery patients continues to be a significant problem despite the use of many antiemetic prophylaxes. The chance of dehydration is decreased by increasing patients’ tolerance to drinking water and treating PONV. Despite the significant impact of PONV on this patient population, there is no consensus on the optimal approach to PONV prevention and management. PONV can cause significant discomfort and distress for patients, as well as an increased risk of other complications such as dehydration and electrolyte imbalance. This study aimed to determine the prevalence, risk factors, and management of PONV after LSG.
Patients and Characteristics
Study Design
This multicenter retrospective study included patients with obesity who had LSG between January 2022 and April 2023. The age range for LSG was 16 to 65 years, and the eligibility requirements included patients with obesity suitable for LSG according to international guidelines. Patients below the age of 16 or over 65 years, with pregnancy, patients with a history of alcohol or drug addiction, chronic opioid use, and patients who developed postoperative complications that required reexploration or patients who had inflammatory bowel disease were all excluded from the study. All patients completed an informed permission form after explaining the procedure and any potential postoperative morbidities. Data were prospectively recorded in a computer file (a computer-based patient record). Three centers participated in this study including the Gastroenterology Surgical Center, Mansoura University, Mansoura, Egypt; Mansoura University Hospital, Mansoura University, Mansoura, Egypt; and Wadi Aldawaser Armed Forces Hospital, Wadi Aldawaser, Kingdom of Saudi Arabia. The institutional review boards of each participating hospital gave their approval to the study (IRB numbers were R.22.12.2002 and R.6.01.2023).
Preoperative Assessment
All patients followed a clinical bariatric surgery protocol that involves a general physical assessment and demographic data. Comprehensive blood work, endocrine assessment, ECHO, and ECG to determine heart state, spirometry, and psychiatric evaluation. Upper GIT endoscopy, abdomen ultrasonography, and chest X-rays are common procedures. Biopsy of the stomach mucosa before surgery to determine Helicobacter pylori status. All patients received a preoperative Clexan and had an elastic bandage on both lower limbs.
Anesthesia and Analgesia Protocol
The usual ASA monitoring was performed. Induction of anesthesia was carried out using propofol 2 mg/kg and fentanyl 1 µg/kg. Rocuronium (0.6–1.2 mg/kg) was given to assist with endotracheal intubation. Maintenance of the anesthesia using sevoflurane 2% with oxygen and nitrous (FiO2 = 50%). Supplementary fentanyl 50 µg, dexamethasone 8 mg IV, ondansetron 4 mg IV, and 1 g paracetamol were administered after induction. Pain management is underway intraoperatively by giving the patient 1 mg/kg of pethidine with or without tramadol 1 mg/kg. Patients were extubated in the operating room and transported to the PACU.
Surgical Technique
The LSG procedure was carried out under general anesthesia. CO2 up to 15 mmHg caused pneumoperitoneum to form. Based on the operator’s preference, three to five ports are positioned. Above the pylorus, devascularization of the greater omentum using sealing devices started, moving on to the cardio-esophageal junction, revealing the left crus of the diaphragm. Antral resection or preservation was done depending on the operator’s choice. After gastric stapling, gastropexy, which involves stitching the sleeved stomach to the greater omentum and peripancreatic fascia, was performed in some cases.
Primary Outcome
The prevalence of PONV after LSG during the recovery time, 3 h, 6 h, 12 h, 24 h postoperatively after transferring the patients to the ward, and after 1 week was the primary outcome.
Definition
Nausea is a feeling of unease and discomfort in the stomach that often leads to the urge to vomit. Vomiting is the forceful expulsion of stomach contents through the mouth. Retching is the act of making an effort to vomit, usually accompanied by dry heaves and gagging, spasmodic, and rhythmic contraction of the respiratory muscles without expulsion of gastric contents.
Secondary Outcomes
The secondary outcomes included the assessment of risk factors for the development of PONV, the response of PONV to medications, the assessment of pain severity using the visual analog scale (VAS) score throughout recovery, and the first 24 h after transferring the patients to the ward.
A modified Aldrete score was utilized to discharge the patient from the post-anesthesia care unit (PACU) based on the score of 8 required to discharge from PACU. Modified Aldrete score included 5 variables (breathing, consciousness level, oxygen saturation, physical activity, and blood pressure) [12]. The quality of recovery using quality of recovery-40 score (QoR-40) at 24 h. The QoR-40 has been confirmed as a tool to assess the postoperative quality of recovery and comprises 40 questions linked to five parts with total scores ranging from 40 to 200 [13,14,15].
After arriving in the PACU, hemodynamic values, VAS for pain, and modified Observer’s Assessment of alertness/Sedation Scale (OAA/S) scores were recognized every 10 min [16].
Postoperative Management
With daily reports, pulse, blood pressure, O2 saturation, and intravenous fluid were recorded. After 6 h postoperatively, clear oral fluid will start to flow. On the second postoperative day, oral gastrograffin was performed. Patients were discharged from the hospital if there were no complaints. The patient was encouraged to take vitamins, take an anticoagulant, take omeprazole for 2 months, and drink lots of water.
The Rationale for Management of the Patient with PONV Post-LSG
Prevention of PONV started with ondansetron and dexamethasone intraoperatively, with total intravenous anesthesia. Patients received antiemetics postoperatively according to the patient’s needs including intravenous ondansetron 4 mg and metoclopramide 10 mg. Dexamethasone 8 mg IV and antihistamines drugs were given to the patient with intolerable PONV.
Pain management started intraoperatively by giving the patient 1 mg/kg of pethidine with or without tramadol 1 mg/kg. Postoperative analgesia in the form of pethidine when VAS of pain is above 5 and paracetamol for patients with VAS of more than 2.
Statistical Analysis
Descriptive statistics were reported as numbers and percentages for categorical variables while a mean and standard deviation were used to describe continuous variables. All statistical evaluations were performed utilizing IBM SPSS Statistics 27. A p-value of 0.05 was found to be statistically significant after using the Student t-test for continuous variables and the chi-square test for categorical variables. A multivariate analysis was utilized to identify independent factors of the occurrence of PONV post-LSG for the significant variables discovered by univariate analysis as odds ratios (OR) with their associated 95% confidence intervals (CI). Exp (B) (odds ratio (OR) > 1 indicated the increased occurrence of the PONV (risk factors) while odds ratio (OR) < 1 indicated the decreased occurrence of the PONV (protective factors).
Results
Demographic Data
Seven hundred sixty-five patients in total, with a mean age of 33.02 ± 9.7 years (range: 16–65 years) and a 36.1% male population, were included in the research. This multicentric study was carried out in 3 centers, with center 1 undergoing 265 LSG, center II 255 LSG, and center III 245 LSG, during the study period. The mean preoperative BMI was 46.83 ± 5.05 kg/m2. Ninety-two (11.87%) patients had preoperative GERD. Perioperative data are shown in Table 1.
Intraoperative and postoperative data are shown in Table 2.
Prevalence and Risk Factors of PONV
At 6 h after LSG, 74.6% of patients had experienced PONV. According to the univariate analysis, female gender, smokers, preoperative GERD, antral resection, gastropexy, type of intraoperative analgesia, postoperative opioid, postoperative recovery status, and severity of pain were risk variables for the occurrence of PONV after LSG. Female gender, smokers, preoperative GERD, gastropexy, and severity of pain were observed to be independent risk factors for the occurrence of PONV, while antral preservation, opioid-free analgesia, and intraoperative combined analgesia were observed to be independent protective factors against the occurrence of PONV (Table 3).
Management and Outcomes of PONV After LSG
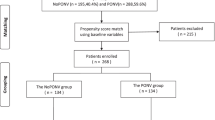
All patients, who developed PONV, received antiemetics postoperatively according to the patient’s needs including intravenous ondansetron 4 mg and metoclopramide 10 mg. Five hundred twenty-nine patients out of 571 improved with antiemetics, while the other 42 patients required dexamethasone 8 mg IV and antihistamines drugs to stop PONV (Fig. 1).
Pain management started intraoperatively by giving the patient 1 mg/kg of pethidine with or without tramadol 1 mg/kg. Postoperative analgesia in the form of pethidine when VAS of pain is above 5 and paracetamol for patients with VAS of more than 2.
Discussion
PONV is a common complication after LSG. It is estimated that up to 80% of patients experience PONV after LSG, making it one of the most common postoperative complications. Due to several reasons, including higher intraabdominal pressure, more air entering the digestive system, and prolonged retention of lipid-soluble, emetogenic anesthetic drugs, obesity is a recognized independent risk factor for PONV. The administration of opioids raises the risk of PONV through increased vestibular system sensitivity and direct stimulation of the mu and delta opioid receptors in the brain stem chemoreceptor trigger zone [17,18,19,20,21]. PONV can cause significant discomfort and distress for patients, as well as an increased risk of other complications such as dehydration and electrolyte imbalance. PONV can result in problems such as aspiration, suture dehiscence, subcutaneous emphysema, pneumothoraxes, and prolonged duration in PACU [17,18,19]. Therefore, it is important to understand the prevalence, risk factors, prevention, and treatment of PONV after LSG.
Several studies have been conducted to determine the prevalence of PONV after LSG. Several studies found that the overall incidence of PONV after bariatric surgery was 60–80% compared with up to 40% in the general surgical population. LSG seems to be the most common bariatric procedure causing PONV. The incidence was higher in female patients (76%) compared to male patients (64%). The incidence was also higher in patients who received general anesthesia compared to those who received regional anesthesia (77% vs. 59%) [22,23,24,25,26,27].
Several risk factors for PONV have been identified in patients undergoing LSG. These include female gender, younger age, history of motion sickness or nausea/vomiting during pregnancy, history of alcohol abuse or smoking, use of general anesthesia, use of opioid analgesics during surgery, and longer duration of surgery [24,25,26,27]. The risk factors for PONV can be divided into patient-related factors (e.g., female gender, history of motion sickness or PONV), procedure-related factors (e.g., type of anesthesia used, duration of surgery, increased gastric blood and secretions, the technique of gastropexy of the sleeved stomach, bariatric surgery patients at increased risk compared to other surgical subpopulations), and medication-related factors (e.g., opioids). Other risk factors include preoperative anxiety or stress, smoking status, age, obesity, and postoperative pain [22,23,24,25,26,27,28]. The Apfel score, which is based on factors including female gender, a history of motion sickness or PONV, quitting smoking, and opioid usage after surgery, is frequently used to evaluate the risk of PONV before surgery [23]. In this study, female gender, smoking, presence of preoperative GERD, gastropexy, and severity of pain were found to be independent risk variables of the development of PONV, while antral preservation, opioid-free analgesia, and intraoperative combined analgesia were observed to be independent protective factors against the occurrence of PONV.
Although there are established guidelines for the management of PONV in the context of general surgery, there is none for the prevention or treatment of PONV in patients who underwent bariatric surgery, for whom PONV significantly increases perioperative morbidity and hospital resource use. The multimodal pharmaceutical strategy for PONV prophylaxis proposed for high-risk surgical patients is suitable for the bariatric population, according to the American Society for Metabolic and Bariatric Surgery [25,26,27,28,29,30,31,32]. To reduce the risk of PONV after LSG, several preventive measures can be taken. Non-pharmacological strategies include preoperative education about the signs and symptoms of PONV, avoidance of preoperative fasting, avoidance of postoperative opioids, use of antiemetic prophylaxis with 5-HT3 receptor antagonists or dexamethasone, use of multimodal analgesia regimens, and avoidance of nitrous oxide during surgery. Prophylactic antiemetic approaches utilizing three or more drugs may be more effective than either single- or dual-drug regimens [25, 29].
The American Society for Metabolic and Bariatric Surgery has recently published guidelines for the prevention and management of PONV after LSG. These guidelines recommend premedication with ondansetron before induction in high-risk patients, avoidance of volatile anesthetics in favor of regional anesthesia techniques, prophylactic administration of ondansetron or granisetron during surgery in high-risk patients, use of multimodal analgesia including non-opioid medications such as acetaminophen or NSAIDs in all patients undergoing LSG, avoidance intraoperative hypotension or bradycardia in all patients undergoing LSG, early ambulation postoperatively in all patients undergoing LSG, avoidance postoperative opioid analgesics in all patients undergoing LSG if possible, and prompt treatment with antiemetics if PONV occurs despite preventive measures [32].
Treatment for PONV should be initiated as soon as possible after onset. If PONV occurs despite preventive measures, pharmacological treatment should be initiated as soon as possible. The first-line treatment includes 5-HT3 receptor antagonists such as ondansetron or granisetron, dopamine antagonists such as metoclopramide or prochlorperazine, corticosteroids such as dexamethasone or methylprednisolone, antihistamines such as diphenhydramine or promethazine, benzodiazepines such as lorazepam or midazolam, and NK1 receptor antagonists such as aprepitant or fosaprepitant. If these treatments fail to control symptoms, then second-line treatments may be considered including anticholinergics such as scopolamine or glycopyrrolate, cannabinoids such as nabilone or dronabinol, neurokinin 1 receptor antagonists such as aprepitant or fosaprepitant, serotonin receptor agonists such as ondansetron or granisetron, anticholinergics/antihistamines combinations, benzodiazepines combined with antiemetics (lorazepam/ondansetron combination), and cannabinoids combined with antiemetics (nabilone/ondansetron combination) [26,27,28,29,30,31,32,33]. In our study, 571 patients, who developed PONV, received antiemetics postoperatively including intravenous ondansetron and metoclopramide. Five hundred twenty-nine patients out of 571 improved with antiemetics drugs, while the other 42 patients required dexamethasone 8 mg IV and antihistamines drugs to stop PONV.
There are a few limitations in this multicentric study. The fact that different surgical teams and anesthesia teams performed the procedures is the most obvious of these but the teams have the same experiences that abolish this limitation. The study’s second limitation is that it is retrospective, although this drawback has been eliminated because all data were entered into a computer file prospectively.
Conclusion
PONV was experienced by 74.6% of patients who underwent LSG. Female gender, smoking, presence of preoperative GERD, gastropexy, and severity of pain were found to be independent risk variables of the development of PONV, while antral preservation, opioid-free analgesia, and intraoperative combined analgesia were observed to be independent protective factors against the occurrence of PONV. Combined intravenous ondansetron and metoclopramide improved the majority of patients who developed PONV. Dexamethasone and antihistamines drugs are given for cases with persistent PONV after using combined intravenous ondansetron and metoclopramide.
References
Sakran N, Raziel A, Goiten O, et al. Laparoscopic sleeve gastrectomy for morbid obesity in 3003 patients: results at a high-volume bariatric center. Obes Surg. 2016;26:2045–50.
Abdallah E, El Nakeeb A, Youssef T, et al. Impact of extent of antral resection on surgical outcomes of sleeve gastrectomy for morbid obesity (a prospective randomized study). Obes Surg. 2014;24(10):1587–94.
Rosenthal RJ, International Sleeve Gastrectomy Expert Panel. International Sleeve Gastrectomy Expert Panel Consensus Statement: best practice guidelines based on experience of >12,000 cases. Surg Obes Relat Dis. 2012;8(1):8–19.
Halliday TA, Sundqvist J, Hultin M, et al. Post-operative nausea and vomiting in bariatric surgery patients: an observational study. Acta Anaesthesiol Scand. 2017;61(5):471–9.
Bamgbade OA, Oluwole O, Khaw RR. Perioperative antiemetic therapy for fast-track laparoscopic bariatric surgery. Obes Surg. 2018;28(5):1296–301.
Naeem Z, Chen IL, Pryor AD, et al. Antiemetic prophylaxis and anesthetic approaches to reduce postoperative nausea and vomiting in bariatric surgery patients: a systematic review. Obes Surg. 2020;30(8):3188–200.
Apfel CC, Laara E, Koivuranta M, et al. A simplified risk score for predicting postoperative nausea and vomiting: conclusions from crossvalidations between two centers. Anesthesiology. 1999;91:693–700.
Myles PS, Wengritzky R. Simplified postoperative nausea and vomiting impact scale for audit and post-discharge review. Br J Anaesth. 2012;108(3):423–9.
Uribe AA, Bergese SD. What is the ideal combination antiemetic regimen? Best Pract Res Clin Anaesthesiol. 2020;34(4):701–12.
Varner KL, March AL. Prevention of nausea and vomiting after laparoscopic sleeve gastrectomy: are we doing enough? AANA J. 2020;88(2):142–7.
Pourfakhr P, Aghabagheri M, ZabihiMahmoudabadi H, et al. Prophylactic administration of diphenhydramine/acetaminophen and ondansetron reduced postoperative nausea and vomiting and pain following laparoscopic sleeve gastrectomy: a randomized controlled trial. Obes Surg. 2021;31(10):4371–5.
Aldrete JA. The post-anesthesia recovery score revisited. J Clin Anesth. 1995;7(1):89–91.
Gornall BF, Myles PS, Smith CL, et al. Measurement of quality of recovery using the QoR-40: a quantitative systematic review. Br J Anaesth. 2013;111(2):161–9.
Myles PS, Weitkamp B, Jones K, et al. Validity and reliability of a postoperative quality of recovery score: the QoR-40. Br J Anaesth. 2000;84(1):11–5.
Zeeni C, Aouad MT, Daou D, et al. The effect of intraoperative dexmedetomidine versus morphine on postoperative morphine requirements after laparoscopic bariatric surgery. Obes Surg. 2019;29(12):3800–8.
Chernik DA, Gillings D, Laine H, et al. Validity and reliability of the Observer’s Assessment of Alertness/Sedation Scale: study with intravenous midazolam. J Clin Psychopharmacol. 1990;10(4):244–51.
Bremner WG, Kumar CM. Delayed surgical emphysema, pneumomediastinum and bilateral pneumothoraces after postoperative vomiting. Br J Anaesth. 1993;71(2):296–7.
Kurnoshina P, Bykovskaia A, Akhtyamov G. Preemptive analgesia: strategies to prevent postoperative nausea and vomiting. Minerva Anestesiol. 2012;78(4):539–50.
Dikkers W, Fokkens W, Goeree R, et al. Multimodal preoperative analgesia for prevention of postoperative nausea and vomiting following laparoscopic sleeve gastrectomy: a systematic review and meta-analysis. Obes Surg. 2019;29(2):397–404.
Watcha MF, White PF. Postoperative nausea and vomiting. Its etiology, treatment, and prevention. Anesthesiology. 1992;77(1):162–84.
Khansari M, Sohrabi M, Zamani F. The useage of opioids and their adverse effects in gastrointestinal practice: a review. Middle East J Dig Dis. 2013;5(1):5–16.
Fathy M, Abdel-Razik MA, Elshobaky A, et al. Impact of pyloric injection of magnesium sulfate-lidocaine mixture on postoperative nausea and vomiting after laparoscopic sleeve gastrectomy: a randomized-controlled trial. Obes Surg. 2019;29(5):1614–23.
Apfel CC, Läärä E, Koivuranta M, Greim CA, Roewer N. A simplified risk score for predicting postoperative nausea and vomiting. Anesthesiology. 1999;91:693–700.
Mendes MN, Monteiro RS, Martins FANC. Prophylaxis of postoperative nausea and vomiting in morbidly obese patients undergoing laparoscopic gastroplasties. A comparative study among three methods. Rev Bras Anestesiol. 2009;59(5):570–6.
Gan TJ, Diemunsch P, Habib AS, et al. Consensus guidelines for the management of postoperative nausea and vomiting. Anesth Analg. 2014;118(1):85–113.
Naeem Z, Chen IL, Pryor AD, et al. Antiemetic prophylaxis and anesthetic approaches to reduce postoperative nausea and vomiting in bariatric surgery patients: a systematic review. Obes Surg. 2020;30(8):3188–200.
Nathan N. Management of postoperative nausea and vomiting: the 4th Consensus Guidelines. Anesth Analg. 2020;131(2):410.
Ziemann-Gimmel P, Goldfarb AA, Koppman J, et al. Opioid-free total intravenous anaesthesia reduces postoperative nausea and vomiting in bariatric surgery beyond triple prophylaxis. Br J Anaesth. 2014;112(5):906–11.
Ruiz-Tovar J, Zubiaga L, Muñoz JL, et al. Incidence of postoperative nausea and vomiting after laparoscopic sleeve gastrectomy with staple line reinforcement with oversewing and staple line inversion vs buttressing material: a randomized clinical trial. Int J Surg. 2018;59:75–9.
Bataille A, Letourneulx JF, Charmeau A, et al. Impact of a prophylactic combination of dexamethasone-ondansetron on postoperative nausea and vomiting in obese adult patients undergoing laparoscopic sleeve gastrectomy during closed-loop propofol-remifentanil anaesthesia: a randomised double-blind placebo-controlled study. Eur J Anaesthesiol. 2016;33(12):898–905.
Riley CL. Anesthesia and enhanced recovery after surgery in bariatric surgery. Anesthesiol Clin. 2022;40(1):119–42.
Schumann R, Ziemann-Gimmel P, Sultana A, et al. Postoperative nausea and vomiting in bariatric surgery: a position statement endorsed by the ASMBS and the ISPCOP. Surg Obes Relat Dis. 2021;17(11):1829–33.
Pourfakhr P, Aghabagheri M, ZabihiMahmoudabadi H, Najjari K, Talebpour M, Khajavi MR. Prophylactic administration of diphenhydramine/acetaminophen and ondansetron reduced postoperative nausea and vomiting and pain following laparoscopic sleeve gastrectomy: a randomized controlled trial. Obes Surg. 2021;31(10):4371–5.
Author information
Authors and Affiliations
Contributions
Study conception and design: Aly E.Rashad, Ali Salem, Mohamed Attia, Ayman El Nakeeb, Mohammed N. Mohammed, Emad El Hefnawy, Mohamed Elmorshedi. Acquisition of data: Aly E. Rashad, Ayman El Nakeeb, Hassan Addossary, Ahmed Zaid, Emad El Hefnawy, Yousif Abdelmonem Abuyousif, Mohammed N. Mohammed, Mohamed Elmorshedi, Ali Salem, Mohamed Attia. Analysis and interpretation of data: Aly E. Rashad, Ayman El Nakeeb, Emad El Hefnawy, Mohamed Elmorshedi, Mohammed N. Mohammed, Emad El Hefnawy. Critical revision of the manuscript: Aly E. Rashad, Ayman El Nakeeb, Mohamed Elmorshedi, Emad El Hefnawy, Hassan Maged Aldossary, Mohammed N. Mohammed, Emad El Hefnawy, Ali Salem, Mohamed Attia.
Corresponding author
Ethics declarations
Declarations
Informed consent was obtained from all individual participants included in the study. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The manuscript has been seen and approved by all authors. This manuscript has not been submitted for publication in any other journal and will not subsequently be submitted for potential publication in another journal until a decision has been made, nor has it been published previously in any media.
Key Points
• Female, smokers, GERD, gastropexy, and pain were risk factors for PONV after LSG.
• Antral preservation, opioid-free analgesia, and combined analgesia are protective against PONV.
• HP status has no role in the development of PONV after LSG.
• Dexamethasone improved refractory PONV.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rashad, A.E., El Hefnawy, E., Elmorshedi, M. et al. Prevalence, Risk Factors, and Management of Postoperative Nausea and Vomiting After Laparoscopic Sleeve Gastrectomy (a Retrospective Multicentric Study). OBES SURG 33, 3237–3245 (2023). https://doi.org/10.1007/s11695-023-06803-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-023-06803-z