Abstract
Purpose
This cohort study was designed to compare the efficacy and safety of laparoscopic Roux-en-Y gastric bypass (LRYGB) and laparoscopic sleeve gastrectomy (LSG) for the management of morbid obesity during the 11-year experience.
Materials and Methods
This study was conducted between July 2006 and August 2019. Annually we recorded data about the weight, body mass index (BMI), percentage of excess weight loss (%EWL), percentage of weight loss (%WL), weight regain, and postoperative complications into a prospectively collected database.
Results
A total of 1146 patients (LRYGB=396 and LSG=750) aged >18 years old were included in the study. Over the first 7 years after surgery, the weight loss rate was significantly higher in the LRYGB-treated group than the LSG-treated group, and weight regain was significantly lower in the LRYGB-treated group in comparison with the LSG-treated group. Our results revealed that statistically, but not clinically, the efficacy of LRYGB and LSG is equivalent in terms of %WL, %EWL, and weight regain within 8 years and more. Besides, there were no significant differences in surgery-related mortality and severe complications between the two procedures.
Conclusion
Our study’s results indicated that the clinical efficacy of LRYGB in the management of obesity is better than LSG during the 11-year experience. But the differences in weight loss and weight regain were not statistically relevant at 8 years and more after the surgery. Also, a comparison of severe complications did not indicate significant differences between the two groups.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obesity is a major underlying cause of some chronic diseases, mental disorders, and cancers. It also has a close relationship with an increased risk of disability and mortality rate [1, 2]. The prevalence of obesity is growing worldwide, and it has become a global concern [2]. According to results of a large study conducted in 200 countries (including 2416 community-based surveys), the number of adult men with obesity has increased from 31 to 281 million, and also the number of women with obesity has increased from 69 to 390 million, during 1975–2016 worldwide [3].
Surgical interventions are more effective approaches to manage morbid obesity and its related comorbidities than non-surgical interventions, i.e., diet therapy, physical activity, and behavioral changes [4]. Based on the International Federation for the Surgery of Obesity and Metabolic Disorders (IFSO) survey, 579,517 patients underwent bariatric surgery in 2014 worldwide. Among them, 45.9% belonged to sleeve gastrectomy (SG), and 39.6% belonged to Roux-en-Y gastric bypass (RYGB) [5]. RYGB is a gold standard procedure that was previously and routinely performed in Canada and the USA in the past years. However, the number of performed RYGB surgeries has gradually decreased, and currently, it is mainly performed in South America and Europe. On the contrary, numbers of performed SG surgeries have grown in Canada, the USA, and Asia over the last decades. It has become a popular procedure due to its easy technique and good results [4].
Many longitudinal studies have assessed RYGB and SG outcomes at 1 year, 2–5 years, and 5–10 years of follow-up. However, few studies compared the effectiveness and safety of RYGB and SG in treating morbid obesity. Therefore, this cohort study was designed to compare the efficacy and safety of laparoscopic RYGB (LRYGB) and laparoscopic SG (LSG) to manage morbid obesity during the 11-year experience.
Materials and Methods
This open cohort study was conducted on eligible patients with morbid obesity who were referred to our center for obesity treatment. Eligibility criteria for surgery included body mass index (BMI) ≥35 kg/m2 with or without obesity-related comorbidity, having the age between 14–70 years old, no history of major psychiatric disorders, and no drug or alcohol addiction. Routinely we perform LRYGB for patients with higher BMI and several obesity-related metabolic disorders and patients with severe gastroesophageal reflux disease (GERD), whereas LSG is performed in high-risk patients; however, surgical procedures were individualized based on each patient’s condition and preference.
This study was ongoing, and data from all patients inserted into a prospectively collected database. In our center, 1255 patients underwent LRYGB (n=453) and LSG (n=802) surgery between July 2006 and August 2019. Patients aged ≤18 years (n=39) old and those who had a history of previous bariatric surgery (n=72) were excluded from the study. Finally, we included data of 1146 patients (LRYGB=396 and LSG=750) in analyses. Follow-up duration was calculated from surgery to the last date of weight measurement, and patients who did not complete their measurement were defined as loss of follow-up.
Surgical Technique and Postoperative Care
All patients underwent preoperative blood tests. An abdominal ultrasound scan was carried out for the patients who had no previous history of cholecystectomy. Upper gastrointestinal endoscopy was performed for all the patients who were a candidate for LRYGB and for the patients who were a candidate for LSG with a previous history of gastrointestinal disorders. Both procedures were conducted by a single bariatric surgeon (K.T) using a 5-port laparoscopic technique. LRYGB was performed by creating a 25–30 mL small pouch. The biliopancreatic limb was 50 cm. The antecolic Roux limb was between 100 and 150 cm. Gastrojejunostomy was used to perform by a linear stapler, and both anastomotic sites were sewn by absorbable sutures (PDS 3-0).
Regarding LSG, vertical sleeve gastrectomy was conducted from 5 cm proximal to the pylorus up to the angle of His. A 36-Fr bougie was inserted into the stomach to calibrate sleeve gastrectomy. Then, the stapler lines were over-sewn by running absorbable sutures (PDS 2-0). No drain and nasogastric (NG) tube were applied after both operations.
All patients were consulted by a dietitian at discharge and were re-examined at 1 week and 1, 3, 6, and 12 months postoperatively and then yearly in the surgeon’s office by the dietitian and the surgeon. Patients who were not able to attend the follow-up visits were interviewed by phone or messaging App. All patients were advised to take oral multivitamin-mineral, iron, and calcium supplements daily and receive intramuscular injections of B-complex plus B12 monthly. Nutritional deficiencies were assessed using blood tests at 6 and 12 months postoperatively and then yearly.
Measurements
Initial patients’ data, including age, sex, height, weight, body mass index (BMI), history of diseases, consumption of medications, and perioperative complications, were respectively recorded before and at the first visit after surgery. Over the follow-up visits, changes in weight and BMI and postoperative complications were noted. Percentage of excess weight loss (%EWL) was estimated as [(preoperative weight–postoperative weight)/(preoperative weight–ideal weight)]×100. Ideal body weight was determined for a BMI of 25 kg/m2. Percentage of weight loss (%WL) was calculated using the following equation: [(preoperative weight–postoperative weight)/preoperative weight]×100. Weight regain was defined as an increase in weight from the lowest recorded weight using the following formula: [(current weight–the lowest weight)/[(preoperative weight–the lowest weight)]×100 [6]. After surgery, successful weight loss was defined as %EWL ≥50% or BMI ≤35 kg/m2, and the failure rate was determined as %EWL <50% or BMI >35 kg/m2. Postoperative complications were classified based on the Clavien-Dindo (CD) classification [7].
Statistical Analysis
SPSS software (version 20.0; SPSS, Inc., Chicago, IL, USA) was applied for statistical analyses. The normal distribution of variables was assessed using the Kolmogorov-Smirnov test. Baseline variables between LRYGB- and the LSG-treated groups were analyzed using students t-test, Mann-Whitney U test, and chi-square test. To compare mean weight, BMI, %WL, and %EWL at each point between two groups, analysis of covariance (ANCOVA) was run after adjusting the baseline weight. To compare postoperative complications between LRYGB and LSG, chi-square was performed. A P-value of <0.05 was considered statistically significant.
Results
In this cohort study, a total of 1146 patients aged >18 years old were treated by LRYGB (n=396) and LSG (n=750). The mean age of participants at baseline was equal to 38.1±10.3 years old, and 78.3% were women. The initial weight and BMI were equal to 115±22 kg and 42.1±6.9 kg/m2, respectively. The median of follow-up duration was 5.21±3.50 and 4.98±2.46 years in the LRYGB and LSG groups, respectively. Table 1 presents the general characteristics of the participants. Mean initial weight and BMI were significantly higher in the LRYGB-treated group than the LSG-treated group. Table 2 shows the proportion of eligible patients who had undergone bariatric surgery and were eligible for follow-up at each postoperative year and observed patients who had completed follow-up at each postoperative year. Lower follow-up rate in the LSG group compared to the LRYGB group was related to two main reasons: (1) patient’s movement to another country and (2) patient’s unwillingness to accept follow-up due to personal reasons.
Weight Loss
Weight and BMI decreased significantly in both study groups. The lowest weight was observed at 2 years and 1 year after the surgery in the LRYGB- and the LSG-treated groups, respectively. Over the follow-up periods, there was an increasing weight and BMI trend in both study groups (Fig. 1).
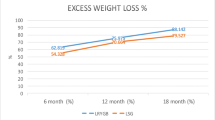
In the LRYGB-treated group, the highest %WL and %EWL were equal to 32.4±0.5 and 82.8±1.5%, respectively, 2 years after the surgery. In the LSG-treated group, the highest %WL and %EWL were equal to 29.6±0.3 and 77.6±1.0%, respectively, at 1 year after the surgery. %WL and %EWL were more significant in the LRYGB-treated group than the LSG-treated group over the first 7 years after the surgery. Postoperative %WL and %EWL were higher in the LRYGB-treated group than the LSG-treated group at 8 years and more, but there were no significant differences between the two groups (Fig. 2).
Weight regain was significantly less in the LRYGB-treated group than the LSG-treated group during the first 6 years postoperatively. However, weight regain was similar between the two study groups at 7 years and more after the surgery (Fig. 3).
After stratification analysis based on sex, there were no significant differences in the rate of WL and weight regain between both sexes in the study groups (data not shown).
Complications
Three cases of early death (<30 days) (LRYGB=2 and LSG=1) and six late deaths (>30 days) (LRYGB=4 and LSG=2) occurred after surgery. However, five deaths occurred as a result of surgery-related complications. The occurrence of bleeding was significantly higher in the LRYGB-treated group than the LSG-treated group, but most were treated by blood transfusion. Also, patients in the RYGB group experienced more marginal ulcers and internal hernias compared with those in the LSG group. We found no significant differences in other complications between the two procedures (Table 3).
According to the CD classification, a comparison of severe complications with the potential for re-operation, life-threatening complications, and death was similar between the two procedures. Nevertheless, CD grade II was significantly higher in the LRYGB-treated group than the LSG-treated group (Table 4).
Obesity-Related Comorbidities
All obesity-related comorbidities were remitted after surgery (Table 5). The remission rate was more than 70% for diabetes, hyperlipidemia, sleep apnea, fatty liver, and hyperuricemia both groups. Besides, there was 51% and 48% remission of hypertension following LRYGB and LSG, respectively.
Discussion
In this cohort study, we compared changes in weight and rate of postoperative complications between patients who underwent LRYGB and those who underwent LSG during the 11-year experience. The weight loss quality was significantly higher in the LRYGB-treated group than the LSG-treated group 1 year after the surgery, and it remained unchanged until 7 years. The weight regain rate from weight nadir was also significantly less in the LRYGB-treated group than the LSG-treated group during the first 7 years. Our results indicated clinically that the rate of weight loss was higher in the LRYGB-treated group than the LSG-treated group, and the percentage of weight regain was lower in the LRYGB-treated group than the LSG-treated group at 8 years and more after the surgery. Still these differences were not statistically significant between the two study groups. Comparison of severe complications did not indicate significant differences between the two procedures.
Our results obtained at the first 8 years after the surgery agree with randomized or non-randomized cohort studies [8,9,10,11,12,13,14], indicating that LRYGB leads to significantly more weight loss than LSG. Results of two meta-analyses on comparative cohort studies conducted by Yang et al. [15], including 15 studies (1381 subjects) and Shoar et al. [16], including 14 studies (1381 subjects), also confirmed the advantage of LRYGB over LSG in the achievement of weight loss. Regarding the long-term efficacy of LRYGB and LSG, several studies had assessed postoperative weight changes during ≥10 years of follow-up [6, 17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34]. But due to single series data, they did not compare trajectories of weight changes between the two procedures mentioned above. Weight regain is also somewhat familiar after performing RYGB and SG, but a few studies compared its value between the two surgeries. Most previous long-term studies have not reported mean weight to regain, and they just indicated it by a decrease in %EWL over time. In our study, baseline weight and BMI were significantly higher in patients who underwent LRYGB than those who underwent LSG; thus, we performed ANCOVA analysis with adjustment for baseline BMI to validate comparison and compensate for the selection bias. However, after adjustment, the LRYGB-treated group indicated clinically better results regarding weight loss and weight regain than the LSG-treated group. Further, well-designed cohort studies with a larger sample size are suggested to compare the long-term efficacy of LRYGB vs. LSG.
In this study, the long-term mortality rate was 1.9% (1.5% in the LRYGB-treated group and 0.4% in the LSG-treated group), from which 1.1% were surgery-related complications. Early death (<30 days) occurred among three patients (LRYGB=2 and LSG=1). One patient died from bleeding, and the reason for death in two other patients was embolism. Late death (>30 days) was also observed among six patients during follow-up. In the LRYGB-treated group (n=4), one patient died following intussusception. The reason for death for the other three cases was unknown. In the LSG-treated group, one patient died after the rejection of intestinal transplantation, and one patient died due to suicide. There are a few studies reported long-term mortality rate after administration of RYGB (between 3.3 and 8.3% in the literature) [6, 21, 35] and SG (0–1.8% in the literature) [31, 32]. Our results are in line with those reported by Bruschi Kelles et al. [35]. They indicated a 0.55% early mortality (<30 days) and 1% late mortality following administration of RYGB over 10 years of follow-up. However, Thereaux et al. [36] showed a decline in mortality rate by 36 and 68% for RYGB and SG, respectively, compared to the patients with obesity in the control group over 7-year follow-up.
In our series, bleeding was observed in 3.5 and 1.1% of patients in LRYGB and the LSG-treated groups, respectively, which is in line with previous studies reporting 0.6–6.7% and 0.4–2.6% bleeding after administration of RYGB [20, 22, 29, 36, 37] and SG [32, 33], respectively. In the present study, the bleeding source was intra-luminal (from anastomotic sites) and intra-abdominal (from omentum or port site) in the LRYGB-treated group. They were managed by conservative treatment. Among patients who underwent LSG, the source of bleeding was only intra-abdominal, and no case of intra-luminal bleeding was observed. Marginal ulcers were found in six patients after performing LRYGB (1.5%) over a follow-up period that was in agreement with that reported in the literature (1.4–4.0%) [6, 21]. The reason for marginal ulcer in two patients was alcohol intake and in one patient was frequent hookah usage. The cause of marginal ulcer was unclear in other patients. The marginal ulcer was managed by medical treatment in all patients.
An internal hernia in our study was comparable with the study by Kothari et al. [21], who reported a 1.3% incidence of an internal hernia 10 years after administration of LRYGB. All affected underwent re-operation to repair an internal hernia. In one patient who underwent LRYGB, an internal hernia resulted in gangrene of the small bowel; thus we reversed gastric bypass by attaching the small pouch to the remnant stomach and removed the small bowel. The low incidence of internal hernias in our patients is related to closing mesenteric defects and Peterson’s space.
Two gastrojejunostomy stenosis cases (0.5%) were observed in this study treated using endoscopic balloon dilation. Incidence of stenosis has been shown to vary between 0.5 and 15.6% in the literature [20,21,22, 29, 36]. As documented in previous studies, using a linear stapler to create gastrojejunostomy instead of a circulatory stapler could significantly reduce the occurrence of stenosis [24], justifying the low incidence of stenosis in our study.
The gastric leak is a major complication after performing SG accompanied by infection and mortality. The evidence reports 0.7–18% of leak incidence post-SG [32, 33, 38]. In the current study, 0.9% of patients experienced gastric leaks over the first 7 days postoperatively. The last case of the gastric leak was observed in 2014. Since then, the gastric stapler lines are over-sown by sutures, and no case of the gastric leak has been observed until now.
We used CD classification to compare surgery-related complications between the two groups. CD classification is a standard tool to assess surgical complications and is approved for all surgeries [39]. A comparison of complications between LSG and LRYGB did not show significant differences in severe compilations.
There were some limitations. First, our study was not a randomized trial; thus, there was a significant difference in weight and BMI between the two groups at baseline. However, the ANCOVA test was used along with adjustment for baseline BMI to decrease selection bias and increase the validity of comparison. Second, we could not evaluate some of the events handled at other hospitals, especially in patients who have moved to another country. Third, the low number of patients at long-term follow-up was another limitation of our study.
In conclusion, our study indicated that the clinical efficacy of LRYGB in the management of obesity is better than LSG during the 11-year experience. But the differences in weight loss and weight regain were not statistically relevant at 8 years and more after the surgery. Besides, a comparison of severe complications did not indicate significant differences between the two groups.
References
Zhang S, de la Haye K, Ji M, et al. Applications of social network analysis to obesity: a systematic review. Obes Rev. 2018;19(7):976–88.
Lung T, Jan S, Tan EJ, et al. Impact of overweight, obesity and severe obesity on life expectancy of Australian adults. Int J Obes (Lond). 2019;43(4):782–9.
Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: a pooled analysis of 2416 population-based measurement studies in 128.9 million children, adolescents, and adults. Lancet. 2017;390(10113):2627-42.
Angrisani L, Santonicola A, Iovino P, et al. Bariatric surgery worldwide 2013. Obes Surg. 2015;25(10):1822–32.
Angrisani L, Santonicola A, Iovino P, et al. Bariatric surgery and endoluminal procedures: IFSO worldwide survey 2014. Obes Surg. 2017;27(9):2279–89.
Duvoisin C, Favre L, Allemann P, et al. Roux-en-Y gastric bypass: ten-year results in a cohort of 658 patients. Ann Surg. 2018;268(6):1019–25.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240(2):205–13.
Bhandari M, Reddy M, Kosta S, et al. Laparoscopic sleeve gastrectomy versus laparoscopic gastric bypass: a retrospective cohort study. Int J Surg. 2019;67:47–53.
Guraya SY, Strate T. Effectiveness of laparoscopic Roux-en-Y gastric bypass and sleeve gastrectomy for morbid obesity in achieving weight loss outcomes. Int J Surg. 2019;70:35–43.
Pekkarinen T, Mustonen H, Sane T, et al. Long-term effect of gastric bypass and sleeve gastrectomy on severe obesity: do preoperative weight loss and binge eating behavior predict the outcome of bariatric surgery? Obes Surg. 2016;26(9):2161–7.
Perrone F, Bianciardi E, Ippoliti S, et al. Long-term effects of laparoscopic sleeve gastrectomy versus Roux-en-Y gastric bypass for the treatment of morbid obesity: a monocentric prospective study with minimum follow-up of 5 years. Updates Surg. 2017;69(1):101–7.
van de Laar AW, Nienhuijs SW, Apers JA, et al. The Dutch bariatric weight loss chart: a multicenter tool to assess weight outcome up to 7 years after sleeve gastrectomy and laparoscopic Roux-en-Y gastric bypass. Surg Obes Relat Dis. 2019;15(2):200–10.
Zhang Y, Zhao H, Cao Z, et al. A randomized clinical trial of laparoscopic Roux-en-Y gastric bypass and sleeve gastrectomy for the treatment of morbid obesity in China: a 5-year outcome. Obes Surg. 2014;24(10):1617–24.
Ahmed B, King WC, Gourash W, et al. Long-term weight change and health outcomes for sleeve gastrectomy (SG) and matched Roux-en-Y gastric bypass (RYGB) participants in the Longitudinal Assessment of Bariatric Surgery (LABS) study. Surgery. 2018;164:774–83.
Yang P, Chen B, Xiang S, et al. Long-term outcomes of laparoscopic sleeve gastrectomy versus Roux-en-Y gastric bypass for morbid obesity: results from a meta-analysis of randomized controlled trials. Surg Obes Relat Dis. 2019;15(4):546–55.
Shoar S, Saber AA. Long-term and midterm outcomes of laparoscopic sleeve gastrectomy versus Roux-en-Y gastric bypass: a systematic review and meta-analysis of comparative studies. Surg Obes Relat Dis. 2017;13(2):170–80.
Adams TD, Davidson LE, Litwin SE, et al. Weight and metabolic outcomes 12 years after gastric bypass. N Engl J Med. 2017;377(12):1143–55.
Artero A, Martinez-Ibanez J, Civera M, et al. Anthropometric parameters and permanent remission of comorbidities 10 years after open gastric bypass in a cohort with high prevalence of super-obesity. Endocrinol Diabetes Nutr. 2017;64(6):310–6.
Christou NV, Look D, Maclean LD. Weight gain after short- and long-limb gastric bypass in patients followed for longer than 10 years. Ann Surg. 2006;244(5):734–40.
Higa K, Ho T, Tercero F, et al. Laparoscopic Roux-en-Y gastric bypass: 10-year follow-up. Surg Obes Relat Dis. 2011;7(4):516–25.
Kothari SN, Borgert AJ, Kallies KJ, et al. Long-term (>10-year) outcomes after laparoscopic Roux-en-Y gastric bypass. Surg Obes Relat Dis. 2017;13(6):972–8.
Mehaffey JH, LaPar DJ, Clement KC, et al. 10-year outcomes after Roux-en-Y gastric bypass. Ann Surg. 2016;264(1):121–6.
Monaco-Ferreira DV, Leandro-Merhi VA. Weight regain 10 years after Roux-en-Y gastric bypass. Obes Surg. 2017;27(5):1137–44.
Nguyen NT, Kim E, Vu S, et al. Ten-year outcomes of a prospective randomized trial of laparoscopic gastric bypass versus laparoscopic gastric banding. Ann Surg. 2018;268(1):106–13.
Obeid NR, Malick W, Concors SJ, et al. Long-term outcomes after Roux-en-Y gastric bypass: 10- to 13-year data. Surg Obes Relat Dis. 2016;12(1):11–20.
Sugerman HJ, Wolfe LG, Sica DA, et al. Diabetes and hypertension in severe obesity and effects of gastric bypass-induced weight loss. Ann Surg. 2003;237(6):751–8.
Valezi AC, de Almeida MM, Mali Jr J. Weight loss outcome after Roux-en-Y gastric bypass: 10 years of follow-up. Obes Surg. 2013;23(8):1290–3.
Sjostrom L, Lindroos A, Peltonen M, et al. Life style, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med. 2004;352:2683–93.
Spivak H, Abdelmelek MF, Beltran OR, et al. Long-term outcomes of laparoscopic adjustable gastric banding and laparoscopic Roux-en-Y gastric bypass in the United States. Surg Endosc. 2012;26(7):1909–19.
Edholm D, Svensson F, Naslund I, et al. Long-term results 11 years after primary gastric bypass in 384 patients. Surg Obes Relat Dis. 2013;9(5):708–13.
Arman GA, Himpens J, Dhaenens J, et al. Long-term (11+years) outcomes in weight, patient satisfaction, comorbidities, and gastroesophageal reflux treatment after laparoscopic sleeve gastrectomy. Surg Obes Relat Dis. 2016;12(10):1778–86.
Castagneto Gissey L, Casella Mariolo JR, Genco A, et al. 10-year follow-up after laparoscopic sleeve gastrectomy: outcomes in a monocentric series. Surg Obes Relat Dis. 2018;14(10):1480–7.
Chang DM, Lee WJ, Chen JC, et al. Thirteen-year experience of laparoscopic sleeve gastrectomy: surgical risk, weight loss, and revision procedures. Obes Surg. 2018;28(10):2991–7.
Felsenreich DM, Langer FB, Kefurt R, et al. Weight loss, weight regain, and conversions to Roux-en-Y gastric bypass: 10-year results of laparoscopic sleeve gastrectomy. Surg Obes Relat Dis. 2016;12(9):1655–62.
Bruschi Kelles SM, Diniz MF, Machado CJ, et al. Mortality rate after open Roux-in-Y gastric bypass: a 10-year follow-up. Braz J Med Biol Res. 2014;47(7):617–25.
Thereaux J, Lesuffleur T, Czernichow S, et al. Long-term adverse events after sleeve gastrectomy or gastric bypass: a 7-year nationwide, observational, population-based, cohort study. Lancet Diabetes Endocrinol. 2019;7(10):786–95.
Shah K, Nergard BJ, Frazier KS, et al. Long-term effects of laparoscopic Roux-en-Y gastric bypass on metabolic syndrome in patients with morbid obesity. Surg Obes Relat Dis. 2016;12:1449–56.
Frattini F. delpini R, Inversini D, et al. Gastric leaks after sleeve gastrectomy: focus on pathogenetic factors. Surg Technol Int. 2017;31:123–6.
Miyamoto S, Nakao J, Higashino T, et al. Clavien-Dindo classification for grading complications after total pharyngolaryngectomy and free jejunum transfer. PLoS One. 2019;14(9):e0222570.
Author information
Authors and Affiliations
Contributions
K.T and M.G conceived and carried out the experiments. M.G analyzed and interpreted data. All authors were involved in writing the paper and had final approval of the submitted and published versions.
Corresponding author
Ethics declarations
Ethics Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent Statement
Informed consent was obtained from all individual participants included in the study.
Conflict of Interest
The authors declare no competing interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Toolabi, K., Golzarand, M. & Farid, R. Laparoscopic Roux-en-Y Gastric Bypass and Laparoscopic Sleeve Gastrectomy in Terms of Efficacy and Safety: a Comparative Study During 11-Year Experience. OBES SURG 31, 2489–2496 (2021). https://doi.org/10.1007/s11695-021-05313-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-021-05313-0