Abstract
Background
This study was conducted to evaluate the amount of weight loss and weight regain and also remission rate of obesity-related comorbidities including diabetes mellitus (DM), hypertension (HTN), and dyslipidemia in patients who underwent LRYGB or LSG during 5 years of follow-up.
Methods
A total of 120 patients who underwent LRYGB or LSG from 2011 to 2013 were enrolled and followed-up for 5 years. Changes in weight, body mass index (BMI), percent weight loss (%WL), and percentage of excess weight loss (%EWL) were recorded.
Result
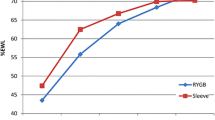
%WL and %EWL were similar between two procedures at 1 year following operation. In patients who underwent LSG, %WL and %EWL were 24.6 ± 1.3% and 61.9 ± 3.5%, respectively, and in LRYGB were 30.4 ± 1.3% and 79.4 ± 3.6%, respectively, 5 years after surgery. Weight regain was 32.0% in LSG and 9.3% in LRYGB after 5 years. The remission rate of diabetes mellitus, hypertension, and dyslipidemia was 70%, 54%, and 100%, respectively, after 5 years of surgery.
Conclusion
Comparing the 5-year success rate of these two techniques, LRYGB seems to be superior to LSG, with lower weight regain and higher weight loss. The short- and long-term effects of two procedures on remission of comorbidities were comparable.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nowadays, about one in every three persons in the world is overweight or obese which is an increased prevalence compared with the past [1]. The prevalence of obesity increased from 1980 to 2016 worldwide. In 2016, it was estimated that 281 million men and 390 million women, aged 20 years and older, were obese [2]. In 2030, the population of obese people is expected to reach 1.12 billion adults (20% of adults’ population) in the world [3]. A similar trend in the growth of the overweight population in Iran has been reported [4]. A recent study reported that the prevalence of obesity in Iranian females has increased from 31.3 to 38.6%, from 1999 to 2008 [5]. It has been shown that obesity is associated with other major diseases including diabetes mellitus type 2 (DM), hypertension (HTN), dyslipidemia, sleep apnea, asthma, fatty liver, reflux, and some types of cancer [1, 6,7,8]. Obesity and its related complications cause 2.5 million deaths annually [6].
Epidemiological studies as well as intervention studies indicate that weight loss is associated with a decreased incidence of hypertension and non-insulin-dependent diabetes mellitus [9, 10]. There are several medical and surgical interventions for the treatment of obesity. Medical treatments including diet therapy, behavior modifications, and medications are helpful. Meanwhile, surgical treatments result in more sustained weight loss in comparison with medical treatments [6, 11]. Laparoscopic Roux-en-Y gastric bypass (LRYGB) and laparoscopic sleeve gastrectomy (LSG) are the most common types of bariatric surgery [12]. LSG is only a restrictive procedure but LRYGB is a combination of the malabsorptive and restrictive procedure. There are some reports indicating that both LRYGB and LSG result in efficient weight loss [13].
Weight regain after surgery is a great concern for bariatric surgeons. It has been estimated that 15–35% of the patients will experience weight regain after LYRGB [14].
Despite previous studies on bariatric surgeries, no comprehensive study has compared the results of LRYGB and LSG in Iran. This study was conducted to evaluate the amount of weight loss and weight regain and also remission rate of obesity-related comorbidities including DM, HTN, and dyslipidemia in patients who underwent LRYGB or LSG surgeries during 5 years of follow-up.
Materials and Methods
In this prospective study, 120 morbidly obese patients who underwent LRYGB (64 patients) and LSG (56 patients) at the Erfan Hospital, Tehran, since 2011 to 2013 participated and were followed-up until 2017. The surgery was indicated, based on National Institutes of Health (NIH) criteria body mass index (BMI) ≥ 40 or ≥ 35 kg/m2 with an obesity-related comorbidities and failure to respond to non-surgical approaches was considered as indications for bariatric surgery [15]. Patients were aged < 19 years, who had major depressive disorder, psychosis, uncontrolled hypo- and hyperthyroidemia, and addiction to opioid or alcohol were excluded from the study.
The study protocol was approved by the Ethics Committee of Tehran University of Medical Sciences (No. 9311245004), and informed consent was obtained from all the participants.
The patients were visited preoperatively by a surgeon (K.T) and the surgical procedure and its benefits and side effects were explained to them thoroughly. Type of surgery was chosen by the surgeon based on baseline weight, metabolic disorders, and eating behaviors. Preoperative work-up and postoperative recommendations were described previously [16]. Patients were visited 1, 3, 6, and 12 months after the operation and annually afterwards to examine the trend of weight loss, dietary intake, and remission of comorbidities. Preoperative data collected from the patients included weight, height, level of education, marital status, and obesity-related comorbidities including HTN, DM, and dyslipidemia. Drugs history and biochemical tests (i.e., fasting blood sugar, HbA1c, and lipid profile) were used for analysis of obesity-related comorbidities. Remission of the comorbidities was defined as comorbidities controlled with less medication or no drugs needed to control them. At each visit, weight was measured and BMI was calculated. Percent weight loss (%WL) was calculated as follows: [(weightpreoperative − weightpostoperative)/(weightpreoperative)] × 100. Percentage of excess weight loss (%EWL) was computed using the following formulas [17]: [(weightpreoperative − weightpostoperative)/(weightpreoperative − ideal weight)] × 100. Ideal body weight (IBW) was defined as the weight in a BMI of 25 kg/m2. The success rate was defined as %EWL > 50%. Weight regain was determined based on two definitions as follows: (1) 25% increase in lost weight from the first 1 year postoperation or (2) weight regain more than 10 kg from the weight at 1 year after surgery [18].
Surgical Techniques
The LRYGB procedure involved creating gastric pouch by one transverse and usually three 3.5 staple loads (Covidien). Antecolic end to side gastrojejunostomy with a Roux-en-Y limb of 150 cm was carried out using 3.5 staple load and completed by hand-sewn continuous sutures. End to side jejunojejunostomy was also created using 2.5-mm staple loads and completed by hand-sewn sutures. Finally, Petersen space was closed by Prolene continuous sutures. The LSG procedure began with gastrolysis 5 cm proximal to pylorus and was continued dividing gastrosplenic ligament up to the angel of His. Gastrectomy was carried out by linear stapler and an orogastric tube (a 36-Fr bougie) was placed in the stomach for guidance. Size of the cartridges was selected based on stomach thickness. The stapler line was routinely reinforced by running sutures (PDS II, Ethicon).
Statistical Analysis
SPSS version 20.0 (SPSS Inc., Chicago, IL) was used for statistical analysis. Recorded data were presented as mean with standard deviation (SD) and number (%). The student t test and chi-square were used to compare the baseline continuous and non-continuous variables between two groups, respectively. The analysis of covariance (ANCOVA) was applied to compare the means of both groups after adjustment for baseline weight and BMI. Chi-square test was used to compare the remission rate of diabetes, HTN, and dyslipidemia 1 year and 5 years after surgery between two groups. The level of significance was defined as P < 0.05 [19].
Results
A total number of 120 morbidly obese patients underwent bariatric surgeries. Of those, 56 patients (46.7%) underwent LSG and 64 patients (53.3%) underwent LRYGB. They were followed-up annually for 5 years. 15.4% were lost to follow-up after 5 years (18.5% in LSG and 12.0% in LRYGB).
Table 1 shows the demographic characteristics and the prevalence of comorbidities based on the procedure. Mean age of the patients was 36.8 ± 11.3 years and 78.3% of the patients were female (n = 94). Mean body weight and BMI were 109 ± 19 kg and 40.0 ± 5.8 kg/m2 in the LSG group and were 131 ± 25 kg and 47.0 ± 7.3 kg/m2, in the LRYGB group, respectively. The rate of obesity-related comorbidities was as follows: DM 10%, HTN 15%, dyslipidemia 22.5%, obstructive sleep apnea 24.2%, reflux 22.5%, gallbladder stones 12.5%, hypothyroidism 22.5%, and fatty liver 30%.
One year postoperation, %WL and %EWL were similar between two groups. Five years after the operation, %WL (30.4 ± 1.3% vs. 24.6 ± 1.3%, P = 0.005, respectively) and %EWL (79.4 ± 3.6% vs. 61.9 ± 3.5%, P = 0.001, respectively) were significantly higher in LRYGB than LSG. The success rate was 85.7% in the LSG group and 90.5% in the LRYGB group 1 year postoperative (P = 0.30) and reached to 76.0% in LSG group and 85.7% in LRYGB 5 years after surgery (P = 0.15).
Weight regain was found in 9.3% of patients who underwent LRYGB and 32.0% of those who underwent LSG (P = 0.004). Based on the first definition, weight regain was observed in 17.3% of the patients 5 years postoperative (30.0% in the LSG group and 5.6% in the LRYGB group, P = 0.001). It was found that 20.0% of the patients who underwent LSG and 7.4% of the patients who underwent LRYGB experienced weight regain of more than 10 kg 4 years after the surgery (P = 0.05). Table 2 reveals %WL, %EWL, and weight regain 1 and 5 years after the surgery in two groups.
Comorbidities remission rate in different surgery groups is shown in Figs. 1, 2, and 3. It was indicated that the prevalence of dyslipidemia was decreased from 22.5 to 0.0%, 1 year after the surgery, and this rate remained unchanged for the next 5 years in all the patients (Fig. 1). Remissions were also observed in patients with DM and HTN after 1 year of surgery. Remission of HTN was 49.0% in LSG and 52.0% in LRYGB 1 year after surgery. At 5 years postoperative, HTN remission was 49.0% and 55.0% in LSG and LRYGB, respectively (Fig. 2). Moreover, the remission rate of DM 1 and 5 years after the surgery was recorded to be 77.0% and 77.0%, respectively in LSG and 73.0% and 82.0%, respectively in LRYGB (Fig. 3). There were no significant differences between LRYGB and LSG on treatment of DM, HTN, and dyslipidemia at 1 year and 5 years after surgery.
Discussions
This study was designed to evaluate the amount of weight loss and weight regain and also remission rate of obesity-related comorbidities including DM, HTN, and dyslipidemia in patients who underwent LRYGB or LSG during 5 years of follow-up. The small sample size was the main limitation of the present study. In the present study, the %WL and %EWL in LSG and LRYGB group were comparable in the first 1 year after the operation. However, %WL and %EWL in LRYGB were significantly higher than LSG after 5 years. Our results were consistent with Brethauer et al. [20] that indicated higher %WL (28.1 ± 10.9% vs. 22.2 ± 9.3%, P = 0.01) and %EWL (60.5 ± 24.6% vs. 49.5 ± 24.9%, P = 0.04) in LRYGB compared with LSG 5 years postoperation. Bhandari et al. [21] have revealed that LRYGB leads to more %EWL than LSG (61% vs. 50%) over 6 years of follow-up. In contrary, Dakour Aridi et al. [13] showed a higher %EWL for the LSG group in comparison with the LRYGB group 1 year and 5 years after the surgery. In another study, %EWL (69.8% vs. 67.3%) and rate of successful weight loss (87.3% vs 77.8) were similar between LRYGB and LSG 5 years after operation [22].
In this study, 20.2% of the patients had weight regain 5 years after the operation. Baig et al. [18] have indicated that weight regain was 35.1% in LSG and 14.6% in LRYGB. Cooper et al. [23] have reported that the mean of weight regain after LRYGB was 23.4% of maximum weight loss. It was illustrated in the present study that 20.0% of the patients who underwent LSG and 7.4% of the patients who underwent LRYGB experienced weight regain of more than 10 kg 5 years after the surgery. There are reports indicating a 19% weight regain of more than 10 kg 5 years after SG [24]. King et al. [25] have shown that patients in the LSG group had more weight regain, compared with the LRYGB group, 5 years after the surgery (P = 0.003). To our knowledge, other risk factors for weight regain after SG have not been reported. However, studies evaluating weight regain after RYGBP have shown multiple conditions associated with this adverse result, including dietary noncompliance, frequency of follow-up visits to a dietitian, physical inactivity, mental health disturbances, and hormonal and metabolic factors. Poor diet quality, characterized by excessive intake of calories, snacks, sweets, and fatty foods, and higher intake of high glycemic index carbohydrates, was reported in patients who regained weight after RYGB [26].
In the present study, significant improvement in diabetes was observed 1 year and 5 years after surgery in both procedures (77% and 82% in LSG and LRYGB, respectively) with no significant differences between two groups. The past cohort studies have revealed the promotion of glycemic index after bariatric surgeries [27, 28]. Our results were consistent with Peterli et al. [29] that indicated remission of diabetes 3 years after LSG (60%) and LRYGB (77%) was similar between two surgeries. In two other prospective studies, Dicker et al. [30] and Salminen et al. [31] also reported that the remission rate of DM at 5 years after LRYGB is similar to LSG. The effect of LRYGB on glycemic control may be explained as a result of patient’s weight loss, decrease in calorie intake caused by changes in gastrointestinal anatomy, and hormonal changes [17].
In this study, the remission rate of HTN was 47% and 53% 1 and 5 years after the surgery, respectively. All the patients who suffered from dyslipidemia were recovered 1 year after the surgery, and the results remained constant for the next 5 years in both groups. The effect of both procedures in the treatment of HTN and dyslipidemia was equal. Peterli et al. [29] have shown promotion of lipid profile after bariatric surgeries but this improvement was higher in LRYGB than LSG. In another study, the remission rate of 74% and 66% has been reported for HTN and dyslipidemia, respectively [28]. There are also reports of a 16% decrease in the prevalence of HTN in a 7-year follow-up time [27]. Other study indicated that the prevalence of HTN reduced from 49 to 35% and the prevalence of dyslipidemia decreased from 51 to 40% over long-term periods [9]. The SLEEVEPASS randomized clinical trial have shown that after 5 years of follow-up, remission rate of dyslipidemia was 47% in LSG and 60% in LRYGB. This ratio was reported to be 19% and 26% for HTN in the LSG and LRYGB group, respectively [31]. Results of a systematic review and meta-analysis stated that per 5-unit decrease in BMI, declined diabetes by 33%, HTN by 27%, and hyperlipidemia by 20% [32].
Conclusion
Both LRYGB and LSG are an effective approach to treat morbid obesity. In the rate of successful weight loss, %WL and %EWL were equal between two groups at the first 1 year postoperatively. However, LRYGB was more effective than LSG in terms of weight loss maintenance and weight regain prevention over the long-term periods. The efficacy of LRYGB in remission of obesity-related comorbidities such as DM, HTN, and dyslipidemia was the same as LSG over the follow-up periods.
References
Hruby A, Hu FB. The epidemiology of obesity: a big picture. Pharmacoeconomics. 2015;33:673–89.
Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: a pooled analysis of 2416 population-based measurement studies in 128.9 million children, adolescents, and adults. Lancet. 2017;390:2627–42.
Kelly T, Yang W, Chen CS, et al. Global burden of obesity in 2005 and projections to 2030. Int J Obes. 2008;32:1431–7.
Esteghamati A, Khalilzadeh O, Mohammad K, et al. Secular trends of obesity in Iran between 1999 and 2007: National Surveys of risk factors of non-communicable diseases. Metab Syndr Relat Disord. 2010;8:209–13.
Hosseinpanah F, Barzin M, Amiri P, et al. The trends of metabolic syndrome in normal-weight Tehranian adults. Ann Nutr Metab. 2011;58:126–32.
Buchwald H, Avidor Y, Braunwald E, et al. Bariatric surgery: a systematic review and meta-analysis. JAMA. 2004;292:1724–37.
Stenard F, Iannelli A. Laparoscopic sleeve gastrectomy and gastroesophageal reflux. World J Gastroenterol. 2015;21:10348–57.
Li Z, Bowerman S, Heber D. Health ramifications of the obesity epidemic. Surg Clin North Am. 2005;85:681–701.
Capoccia D, Coccia F, Guarisco G, et al. Long-term metabolic effects of laparoscopic sleeve gastrectomy. Obes Surg. 2018;28:2289–96.
Fuchs T, Loureiro M, Both GH, et al. The role of the sleeve gastrectomy and the management of type 2 diabetes. Arq Bras Cir Dig. 2017;30:283–6.
Golzarand M, Toolabi K, Farid R. The bariatric surgery and weight losing: a meta-analysis in the long-and very long-term effects of laparoscopic adjustable gastric banding, laparoscopic roux-en-Y gastric bypass and laparoscopic sleeve gastrectomy on weight loss in adults. Surg Endosc. 2017;31:4331–45.
Gloy VL, Briel M, Bhatt DL, et al. Bariatric surgery versus non-surgical treatment for obesity: a systematic review and meta-analysis of randomised controlled trials. BMJ. 2013;347:f5934.
Dakour Aridi H, Khazen G, Safadi BY. Comparison of outcomes between laparoscopic roux-en-Y gastric bypass and sleeve gastrectomy in a Lebanese bariatric surgical practice. Obes Surg. 2018;28:396–404.
Jirapinyo P, Abu Dayyeh BK, Thompson CC. Weight regain after roux-en-Y gastric bypass has a large negative impact on the bariatric quality of life index. BMJ Open Gastroenterol. 2017;4:e000153.
Cummings DE, Cohen RV. Beyond BMI: the need for new guidelines governing the use of bariatric and metabolic surgery. Lancet Diabetes Endocrinol. 2014;2:175–81.
Golzarand M, Toolabi K, Hedayati M, Azam K, Douraghi M, Djafarian K. Comparative study of resting metabolic rate and plasma amino acid profile in patients who underwent laparoscopic roux-en-Y gastric bypass and laparoscopic sleeve gastrectomy: 6-month follow-up study. Obes Surg. 2019
Toolabi K, Arefanian S, Golzarand M, et al. Effects of laparoscopic roux-en-Y gastric bypass (LRYGB) on weight loss and biomarker parameters in morbidly obese patients: a 12-month follow-up. Obes Surg. 2011;21:1834–42.
Baig SJ, P P, Mahawer KK, et al. Weight regain after bariatric surgery-a multicenter study of 9617 patients from Indian bariatric surgery outcome reporting group. Obes Surg. 2019;29:1583–92.
Fleiss JL. Design and analysis of clinical experiments: John Wiley & Sons; 2011.
Brethauer SA, Aminian A, Romero-Talamas H, et al. Can diabetes be surgically cured? Long-term metabolic effects of bariatric surgery in obese patients with type 2 diabetes mellitus. Ann Surg. 2013;258:628–36.
Bhandari M, Reddy M, Kosta S, et al. Laparoscopic sleeve gastrectomy versus laparoscopic gastric bypass: a retrospective cohort study. Int J Surg. 2019;67:47–53.
Leyba JL, Llopis SN, Aulestia SN. Laparoscopic roux-en-Y gastric bypass versus laparoscopic sleeve gastrectomy for the treatment of morbid obesity. A prospective study with 5 years of follow-up. Obes Surg. 2014;24:2094–8.
Cooper TC, Simmons EB, Webb K, et al. Trends in weight regain following roux-en-Y gastric bypass (RYGB) bariatric surgery. Obes Surg. 2015;25:1474–81.
Bohdjalian A, Langer FB, Shakeri-Leidenmuhler S, et al. Sleeve gastrectomy as sole and definitive bariatric procedure: 5-year results for weight loss and ghrelin. Obes Surg. 2010;20:535–40.
King WC, Hinerman AS, Belle SH, et al. Comparison of the performance of common measures of weight regain after bariatric surgery for association with clinical outcomes. JAMA. 2018;320:1560–9.
Alvarez V, Carrasco F, Cuevas A, et al. Mechanisms of long-term weight regain in patients undergoing sleeve gastrectomy. Nutrition. 2016;32:303–8.
Courcoulas AP, King WC, Belle SH, et al. Seven-year weight trajectories and health outcomes in the longitudinal assessment of bariatric surgery (LABS) study. JAMA Surg. 2018;153:427–34.
Inge TH, Courcoulas AP, Jenkins TM, et al. Weight loss and health status 3 years after bariatric surgery in adolescents. N Engl J Med. 2016;374:113–23.
Peterli R, Wolnerhanssen B, Vetter D, et al. Laparoscopic sleeve gastrectomy versus roux-en-Y gastric bypass for morbid obesity-3-year outcomes of the preospective randomized Swiss multicenter bypass or sleeve study (SM-BOSS). Ann Surg. 2017;265:446–73.
Dicker D, Yahalom R, Comaneshter DS, et al. Long-term outcomes of three types of bariatric surgery on obesity and type 2 diabetes control and remission. Obes Surg. 2016;26:1814–20.
Salminen P, Helmio M, Ovaska J, et al. Effect of laparoscopic sleeve gastrectomy vs laparoscopic roux-en-Y gastric bypass on weight loss at 5 years among patients with morbid obesity: the SLEEVEPASS randomized clinical trial. JAMA. 2018;319:241–54.
Ricci C, Gaeta M, Rausa E, et al. Early impact of bariatric surgery on type II diabetes, hypertension, and hyperlipidemia: a systematic review, meta-analysis and meta-regression on 6,587 patients. Obes Surg. 2014;24:522–8.
Acknowledgment
The authors acknowledged Mrs. Roya Farid for critical editing of English grammar of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study protocol was approved by the Ethics Committee of Tehran University of Medical Sciences (No. 9311245004), and informed consent was obtained from all the participants.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Toolabi, K., Sarkardeh, M., Vasigh, M. et al. Comparison of Laparoscopic Roux-en-Y Gastric Bypass and Laparoscopic Sleeve Gastrectomy on Weight Loss, Weight Regain, and Remission of Comorbidities: A 5 Years of Follow-up Study. OBES SURG 30, 440–445 (2020). https://doi.org/10.1007/s11695-019-04183-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-019-04183-x