Abstract
Thai food is popular because of its unique characteristics and health benefits, particularly the antioxidant properties of the herbs and spices added to them. To confirm its antioxidant properties, 11 common herbs and spices from Thailand were evaluated for their chemical components and radical scavenging activity. The effects of herb and spice powders on lipid oxidation in fish were evaluated. The results confirmed the presence of rich essential chemical components that are beneficial to humans. For example, ginger contained 31.21 ± 0.39% linoleic acid, sweet basil contained 52.96 ± 0.92% linolenic acid, kaffir lime leaves contained 517.15 ± 8.89 ng/mg lipid α-tocopherol, and climbing wattle contained 368.75 ± 18.2 μg/g sample adenosine monophosphate. High total polyphenol content and strong radical scavenging activity were also present in these herbs and spices, especially in turmeric, fingerroot, and ginger. Moreover, they exhibited excellent lipid oxidation prevention ability in fish, especially turmeric and ginger in lipid peroxide prevention and fingerroot and ginger in aldehyde prevention. The results showed significant differences between oxidized and non-oxidized fish samples. In addition, the results of a principal component analysis indicated a strong correlation between the chemical components and samples. In conclusion, Thai herbs and spices, which include turmeric, fingerroot, and ginger, have outstanding benefits that are essential for improving health and are suitable for use as preservatives in food.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Thai food is inspired by various cultures and cuisines to produce unique flavors in delectable combinations [1]. Sriwattana et al. summarized the popularity of Thai food, and found that the most popular were Tom Yum Kung (hot and sour shrimp soup), Pad Thai (Thai style fried noodles), and Kaeng Kew Wan (green curry) [2]. These foods contain various herbs and spices, including kaffir lime leaves, galangals, chilis, lemongrass, chives, garlic, and coriander, which make Thai food healthy. According to Khanthapok and Sukrong, Thai food contains several antioxidants and biological compounds that have benefits such as anti-aging effects [3]. Danyuthasilpe et al. studied elderly individuals in northern Thailand [4]. The survey showed that Thai cooking commonly contains a variety of organic products, such as vegetables, herbs, and spices. In addition, Siwarungson and Lertpringkop investigated Thai curry soups without coconut milk. They determined that all the soups contained several antioxidant, anti-aging, and anti-hypertensive compounds, which provide a variety of health benefits [5].
The leading health benefits in Thai foods come from the herbs and spices. Embuscado described herbs and spices as plants with unique fragrances and tastes composed of various flavonoid, phenolic, and sulfur-related compounds [6]. In addition, the differentiation between herbs and spices is based on the part used. Only the leaves of herbs are used, whereas in spices, parts of the plant other than the leaves are used [6]. Many researchers have reported the health benefits of herbs and spices, including reducing the risk of cancer, cardiovascular disease, cholesterol, and inflammation [7,8,9]. These benefits are because of the antioxidants present in herbs and spices that prevent lipid oxidation and the growth of microorganisms. These effects have inspired several researchers to pursue antioxidant-related projects, such as packaging to prevent lipid oxidation and preserve food for a longer time or adding natural extracts containing high contents of antioxidants to food [10]. All studies were aimed at preventing oxidation, maintaining food freshness, and improving food quality. Herbs and spices have antioxidant and antibacterial properties and they are mainly composed of polyphenols. Other fat- and water-soluble components are also present, but they have not been as extensively researched. It is possible that these components also have antioxidant properties, similar to polyphenols. Although a large number of Thailand’s herbs and spices are widely known, some of them still remain relatively unknown, resulting in a lack of data on their effects and benefits.
In this study, 11 herbs and spices from Thailand were evaluated for other fat- and water-soluble components and total polyphenolic content to investigate their radical scavenging activities. The effects of herb and spice powders on lipid oxidation in fish were evaluated. This study aimed to collect general data on the chemical components in herbs and spices and investigate their ability to scavenge free radicals and to prevent lipid oxidation for improved food preservation.
Materials and methods
Sample preparation
Eleven fresh herb and spice samples were obtained from a local market in Nakhon Ratchasrima, Thailand. The sample names (with scientific names), analytical parts, and obtained locations are listed in Table 1. All samples were cut and sun-dried until all water was completely removed. They were then pulverized using a blender (MX1200XTSLJ; Waring Commercial, McConnellsburg, Pennsylvania, USA) and stored at − 30 °C until further analysis. The resulting herb and spice powders are shown in Fig. 1. Raw fish of the species, Seriola dumerili (amberjack), were purchased from a local market and homogenized using a food processor (TK 435; TESCOM, Tokyo, Japan). Homogenized fish were stored in a plastic bag at − 30 °C until further analysis.
Picture of the herbs and spice powders used in this study: (1) Kaffir lime leaf (Citrus hystrix), (2) galangal (Alpinia galanga), (3) turmeric (Curcuma longa), (4) fingerroot (Boesenbergia rotunda), (5) ginger (Zingiber officinale), (6) lemongrass (Cymbopogon citratus), (7) pandan leaf (Pandanus amaryllifolius), (8) white pepper (Piper nigrum L.), (9) sweet basil (Ocimum basilicum), (10) climbing wattle (Senegalia pennata), and (11) coriander (Coriandrum sativum). Detailed information on these samples is presented in Table 1
Chemicals
Potassium persulfate, l-malic acid, l-lactic acid, Folin-Ciocalteu reagent, campesterol, β-sitosterol, desmosterol, stigmasterol, ergosterol, triphenyl phosphine, and acetonitrile were purchased from Sigma-Aldrich Ltd. (Tokyo, Japan). α-, β-, γ-, and δ-Tocopherol were purchased from Eisai Co. Ltd. (Tokyo, Japan). Triethylamine and 2,2-diphenyl-1picrylhydrazyl (DPPH) were purchased from Tokyo Chemical Industry Co. Ltd. (Tokyo, Japan). AccQ-FBB and AccQ-Flour reagents were purchased from Waters Corporation (Milford, Massachusetts, USA). 1,3-Cyclohexanedione and oxalic acid were purchased from Nacalai Tesque (Kyoto, Japan). Pyroglutamic acid was purchased from MP Biomedicals (Illkirch-Graffenstaden, France). 4-Hydroxy-trans-2-hexenal (HHE) were purchased from Cayman Chemical Company (Ann Arbor, Michigan, USA). Distilled water was purchased from Takasugi Pharmaceutical Co. Ltd. (Fukuoka, Japan). Other chemicals and solvents used in the experiments were purchased from FUJIFILM Wako Pure Chemical Industries (Osaka, Japan). All reagents used in this experiment were of reagent grade.
Analysis of lipophilic components
Lipophilic components were extracted from the herbs and spices by following the method described by Ito et al. [11]. Each herb and spice powder (1 g) was mixed with a chloroform:methanol solution (30 mL, 2:1 v/v) and homogenized. The upper layer was centrifuged and removed using an aspirator. The lower layer was filtered and evaporated until approximately 5 mL remained. The chloroform:methanol solution (up to final volume of 10 mL, 2:1 v/v) was added to the final solution and stored at − 30 °C until further analysis. To determine the fatty acid composition, 3 g of lipid was measured, trans-methylated through saponification, and subjected to BF3-catalyzed methylation according to Ito et al. method [11]. These fatty acid methyl esters (FAMEs) were resuspended in acetone and analyzed using gas–liquid chromatography (GLC). To determine the tocopherol content, 3 mg of extracted lipid was saponified and the unsaponifiable substance was extracted according to Ito et al. [11]. The unsaponifiable substance was dissolved in an ethyl acetate:hexane solution (200 µL, 85:15 v/v) for analysis by high-performance liquid chromatography (HPLC). Sterols were analyzed following the method described by Yoshida et al. [12]. The unsaponifiable substance, which was extracted following a similar method as for tocopherol analysis, was dissolved in acetone (100 μL) and analyzed using GLC.
Analysis of water-soluble components
Water-soluble components were extracted from herbs and spices following the method described by Tanaka et al. [13]. The herb and spice powder samples (1 g) were mixed with 1% perchloric acid (20 mL) and homogenized. Then, the pH was adjusted to 6.8 using a potassium hydroxide solution before being centrifuged. Distilled water (up to final volume of 50 mL) was added to the solution, and it was filtered through a 0.8 µm filter. The final solution was stored at -30 °C until further analysis. The free amino acid content was analyzed following the method described by Tanaka et al. [13]. The processed solution was analyzed using HPLC. Purine and pyrimidine were extracted as described by Ishimaru et al. [14]. The water-soluble solution was injected into the HPLC system and analyzed at a wavelength of 270 nm. Organic acids were analyzed following the method described by Sekizawa et al. [15]. The water-soluble solution was directly injected and analyzed using HPLC at a wavelength of 210 nm.
Analysis of total polyphenol content
The total polyphenol content was compared with the lipophilic, water-soluble, and methanol-soluble components. The extraction of lipophilic and water-soluble components was previously described in the extraction of lipophilic and water-soluble component sections, respectively. Methanol-soluble substances were extracted from herbs and spices following the method described by Jongsawatsataporn and Tanaka [16].
The total polyphenol content was analyzed following a modified version of the method described by Lim et al. with modifications [17]. First, methanol (1.9 mL) was added to the extract solutions (100 μL of lipophilic, water-soluble, and methanol-soluble component solutions), followed by the addition of the Folin-Ciocalteu reagent (500 μL). The resulting solution was incubated for 5 min at 30 °C in a water bath. Thereafter, a 20% sodium carbonate solution (5 mL) was added to the solution, after which it was incubated again for 40 min at 30 °C. Finally, the solution was centrifuged at 3000 × g for 10 min. The total polyphenol content was determined by measuring the absorbance increase at 730 nm between the blank and sample solutions. The results were calculated against a calibration curve of pyrogallol, and the data was expressed in micrograms of pyrogallol equivalent per gram of sample.
Analysis of radical scavenging activity
The radical scavenging activities of the DPPH and 2,2′-azino-bis-3-ethylbenzthiazoline-6-sulphonic acids (ABTs) in the extracts were analyzed following the method described by Lim et al. [17]. For DPPH analysis, a DPPH solution (2.5 mL) was added to a mixture of the extract solutions (100 μL) and ethanol (1.9 mL). The samples were incubated for 30 min at 30 °C in a water bath. The activities were determined by measuring the decrease in absorbance of DPPH radicals at 520 nm based on the difference between the blank and sample solutions. The results were calculated against a calibration curve of α-tocopherol and the data was expressed in micrograms of α-tocopherol equivalent per gram of sample because α-tocopherol is widely used as an index of antioxidant activity.
For ABTs analysis, an ABTs solution (4.75 mL) was added to the sample extracts (0.25 mL). The samples were then incubated for 6 min at 30 °C in a water bath. The activities were determined by measuring the decrease in absorbance of ABTs radicals detected at 740 nm based on the difference between the blank and sample solutions. The results were calculated against a calibration curve of α-tocopherol and the data was expressed in micrograms of α-tocopherol equivalentper gram of sample.
Effect of herb and spice powders addition to fish on lipid oxidation
In this study, we referred to Wang et al. to investigate the effect of herb and spice powders on the suppression of lipid oxidation in fish meat [18]. To accomplish this goal, three types of fish meat samples were prepared. Fresh and frozen minced fish meat were prevented from undergoing oxidation. For the control, frozen minced fish meat (3 g) was weighed into a 50 mL glass centrifuge tube and capped. The samples were subjected to oxidation in a water bath at 30 °C for 15 h. For the preparation of the fish meat with herb and spice powder samples, minced frozen fish meat (10 g) and a powder of herb and spice (0.5 g) were weighed into a mortar and mixed using a pestle. The sample was transported into a 50 mL glass centrifuge tube, capped, and subjected to oxidation in a water bath at 30 °C for 15 h. After preparation, the samples were analyzed for lipid peroxide (LPO) and aldehydes. LPO was analyzed according to the method described by Nakamura et al. [19]. Cyclohexane and triphenylphosphine reagent (100 μL each) were added to an aliquot (3 mg) of the lipids in a screw-capped tube. The mixture was shaken gently at 30 °C in the dark for 30 min. The resulting stoichiometrically generated triphenylphosphine oxide was determined using an HPLC system at a wavelength of 260 nm. The aldehyde content was analyzed according to the method described by Tanaka et al. [20]. The samples that contained aldehydes were reacted with a 1,3-cyclohexanedione (CHD) reagent. CHD-aldehydes were analyzed using an HPLC system with a fluorescence detector at an excitation and emission wavelength of 385 and 450 nm, respectively. The results were calculated by comparing the level of lipid oxidation of the fresh fish meat, control, and fish meat with herb and spice powder samples.
Statistical analysis
In this study, the data was presented as the mean ± standard deviation (SD). Principal component analysis (PCA) was conducted using a Bell Curve in Microsoft Excel software (Social Survey Research Information Co., Ltd.). PCA was conducted on normalized data to reduce the number of actual variables (44 chemical compounds, total polyphenol contents, radical scavenging activities, and LPO and aldehyde contents in fish) and derived variables (principal components) that summarize the total variance, that is, to correlate the chemical compositions of fermented fish products with their respective characteristics.
Results and discussion
Lipophilic components
Fatty acid composition
The fatty acid composition of the herbs and spices is shown in Table 2. All samples contained various fatty acids except palmitoleic acid (16:1), which is commonly found in plants, especially oil crops and nuts like macadamias [21, 22]. Similarly, other researchers have rarely reported or did not mention the palmitoleic acid content in herbs and spices [23, 24]. Neffati and Marzouk reported that a small amount of palmitoleic acid content was observed in coriander [25]. The primary focus of this experiment was linolenic acid (18:3 n-3), which is a greatly beneficial fatty acid that acts as and is converted to eicosapentaenoic acid and is predominantly found in plants. Sweet basil had the highest percentage of linolenic acid (52.96 ± 0.92% fatty acid), followed by coriander (36.41 ± 0.52% fatty acid) and pandan leaf (28.28 ± 0.94% fatty acid) [26]. According to a report by Daga et al., sweet basil contains a higher concentration of linolenic acid than coriander, but that a prominent amount was still present in both herbs [27]. In addition, Ramesh et al. reported linoleic acid (18:2 n-6) in galangal, turmeric, ginger, lemongrass, white pepper, and sweet basil [28]. Most of the results from the aforementioned reports contained similar information and conclusions, including similar percentages of fatty acids in galangal, ginger, and white pepper. Moreover, there are many reports on the essential oil components of pandan leaves. However, there are only a few reports on the fatty acid composition of raw pandan leaves. Our data may be the first to show that pandan leaves contain a higher linolenic acid content than the more commonly studied herbs such as sweet basil. In case of n-6/n-3 ratio, all samples except for white pepper contained less than 6% of the n-6/n-3 ratio. A higher n-6/n-3 ratio increases the risk of obesity, inflammation, and cancer [29,30,31]. This means that most of the herbs and spices in this experiment were safe for adult consumption [32]
Tocopherol content
The tocopherol content of the herbs and spices is shown in Table 3. Plants are described as living organisms that can accumulate and store lipids in their cells. However, when compared with oil crops, including sunflower, palm, and canola oils, a lower lipid content was observed, which was scarcely reported [33]. Herbs and spices mainly contain α- and γ-tocopherols, which are the major forms of vitamin E. These tocopherols can prevent inflammation in cells and act against prostate cancer [34, 35]. In this study, kaffir lime leaves contained the highest α-tocopherol content (517.15 ± 8.89 ng/mg lipid), whereas ginger contained the highest γ-tocopherol content (86.07 ± 1.13 ng/mg lipid) among the samples. However, none of the samples contained β- or δ-tocopherols. Dertyasasa and Tunjung also reported that kaffir lime leaves contain both α- and γ-tocopherols [36]. In addition, Jelled et al. confirmed that ginger contains high levels of α- and γ-tocopherols [37]. Other herbs and spices are also consistent with other researchers’ reports, including Gómez-Coronado et al., who reported the contents of α- and γ-tocopherols in basil and coriander [38].
Sterol content
The sterol content of the herbs and spices are shown in Table 4. All samples contained stigmasterol and β-sitosterol, which are important compounds commonly found in plants [39, 40]. Stigmasterol is related to hormone synthesis in humans, whereas β-sitosterol plays an important role in anti-diabetic and anticancer activities, and can act as an antioxidant [39, 40]. However, campesterol, ergosterol, and desmosterol were present in small amounts or were absent from some samples, such as coriander, which contained stigmasterol, β-sitosterol, and campesterol. Saini et al. similarly reported the sterol content of coriander [41]. The turmeric sample contained the highest stigmasterol (8.30 ± 1.20 μg/g lipid) and β-sitosterol (14.69 ± 2.00 μg/g lipid) content with the highest among the samples. This result is consistent with the reports of Gupta et al. and Li et al. [42, 43]. Many researchers focus on the sterol content of oil crops, including palm, sunflower, and soybean oils [44]. Therefore, the data indicates that non-oil crops contain relatively small amounts of fatty acids and sterol. This is also consistent with the result that relatively small amounts of sterol content exist in the herb and spice samples that are not oil crops.
Water-soluble components
Free amino acid content
The free amino acid content of the herbs and spices is shown in Table 5. The results showed that climbing wattle contained the highest total free amino acid content among the samples, followed by sweet basil and kaffir lime leaves. Other researchers have reported that some amino acids, including arginine (Arg), glycine (Gly), lysine (Lys), alanine (Ala), tyrosine (Tyr), serine (Ser), and histidine (His), could function as antioxidants [45]. This implies that most of the herbs and spices used in the experiments contained the abovementioned amino acids, especially climbing wattle, which contained the highest Arg content (13,061.48 ± 87.9 μg/g sample). Some researchers have reported the amino acid content of other plant samples. For example, Ajayi et al. and Ifeanyi et al. reported the amino acid content in ginger and Almuhayawi et al. reported the amino acid content in lemongrass [46,47,48]. Our results are consistent with these reports. When we focused on human essential amino acids, including phenylalanine (Phe), valine (Val), tryptophan (Trp), threonine (Thr), isoleucine (Ile), methionine (Met), leucine (Leu), His, and Lys but excluding Met and Trp, which were not present in any sample, and Thr, which was not present in fingerroots, ginger, lemongrass, and white pepper, the samples contained high levels of these essential amino acids [49].
Purine and pyrimidine monophosphate contents
The purine and pyrimidine monophosphate contents of the herbs and spices are shown in Table 6. None of the herbs and spices in this experiment contained guanosine 5′-monophosphate (GMP) or inosine 5′-monophosphate (IMP), whereas small amounts of cytidine 5′-monophosphate (CMP) and adenosine 5′-monophosphate (AMP) were present in some samples. Ginger did not contain any purine or pyrimidine monophosphate. Overall, sweet basil, climbing wattle, and coriander contained significantly higher levels of purine and pyrimidine monophosphate than the other samples. Coriander had the highest CMP content (198.14 ± 4.6 μg/g sample) and climbing wattle contained the highest AMP content (368.75 ± 18.2 μg/g sample). Purine and pyrimidine contents have been used to determine the level of freshness of edible animals and fish. However, CMP, GMP, IMP, and AMP can also determine the umami taste in various foods [14]. Duan et al. reported the amounts of purine and pyrimidine monophosphate in sweet basil and coriander [50]. These results might be slightly different from those of the present study. However, they also concluded that both sweet basil and coriander contained lesser amounts of purine and pyrimidine monophosphate than the other samples. In another study, Duan et al. reported the purine and pyrimidine content of turmeric [51]. However, only CMP was present in this experiment, in contrast to Duan et al.’s reports, which reported very small amounts of all types of purine and pyrimidine monophosphate [50, 51].
Organic acid content
The organic acid content of the herbs and spices is shown in Table 7. All samples contained fumaric acid, especially coriander, which had the highest content among the samples (54.98 ± 1.12 mg/g sample). Fumaric acid is present in several plants because it has an important function in the fixed carbon procedure. Older plants tend to have a higher content of fumaric acid compared to younger plants [52]. Other organic acids were also present in the samples, including oxalic, citric, and succinic acids. However, relatively small amounts of unpleasant smelling organic acid groups, including propionic, butyric, isovaleric, and valeric acids, were present in the herb and spice samples that were analyzed [53,54,55]. The results indicated that the galangal sample was the only one that contained butyric acid. It was also determined that sweet basil contained a relatively small amount of isobutyric acid. As previously mentioned, Duan et al. reported the purine, pyrimidine monophosphate, and organic acid contents in turmeric, sweet basil, and coriander [50, 51]. The results remained consistent with this analysis despite the fact that only a few of the organic acids were analyzed. In particular, a high oxalic acid content was present in turmeric (20.64 ± 0.35 mg/g sample). Oxalic acid is present in various plants and can act as an antioxidant [56]. A higher radical scavenging activity is typically associated with a higher oxalic acid content.
Total polyphenol content
The total polyphenol content of herbs and spices are shown in Table 8. This experiment used three types of herb and spice extracts, including lipophilic, water-, and methanol-soluble solutions. Each type of extract had different total polyphenol contents. In particular, the lipophilic extract of turmeric, and the methanol-soluble extracts of fingerroot and ginger expressed higher polyphenol contents than the other extracts. The result clearly showed that turmeric contains the highest total polyphenol content, followed by fingerroot and ginger. Herbs and spices are sources of polyphenols. When consuming herbs and spices, the level of polyphenol intake also increases [57]. Thailand’s herbs and spices are widely used in Thai cuisine and in supplementary and commercial snacks [58]. Many studies have reported the antioxidative ability and polyphenol content of turmeric and ginger. Alafiatayo et al. reported the polyphenol contents of ten selected species of Zingiberaceae rhizomes. The report also showed that turmeric and ginger contained higher polyphenol content than other samples [59]. Jongsawatsataporn and Tanaka reported higher antioxidant contents in turmeric, fingerroot, and ginger. Słowianek and Leszczynska also reported antioxidant properties in methanol-extracts of various herbs and spices, including ginger, sweet basil, and turmeric [16, 60]. Among all samples of herbs and spices, the polyphenol content of turmeric remained the highest compared to the others in this study.
Radical scavenging activity
The radical scavenging activities of herbs and spices are shown in Table 8. In this study, DPPH and ABTs radical scavenging activity in lipophilic, water- and methanol-soluble extracts were observed. Among the results, the DPPH and ABTs in water-soluble extracts exhibited the highest radical scavenging activity. Regarding DPPH, the lipophilic extract of turmeric (221.39 ± 2.65 μg α-tocopherol equivalent), water-soluble extract of kaffir lime leaves (325.63 ± 17.24 μg α-tocopherol equivalent, and methanol-soluble extract of ginger (296.65 ± 25.69 μg α-tocopherol equivalent) exhibited the highest equivalent contents. The lipophilic and methanol-soluble extracts of turmeric and the water-soluble extracts of climbing wattle exhibited the highest equivalent contents of ABTs. The lipophilic extract of turmeric contained 746.23 ± 6.61 μg α-tocopherol equivalent, the water-soluble extract of climbing wattle contained 3946.6 ± 293.20 μg α-tocopherol equivalent, and the methanol-soluble extract of turmeric contained 757.76 ± 39.98 μg α-tocopherol equivalent. Similar to polyphenols, many researchers have reported DPPH and ABTs radical scavenging activities of herbs and spices. According to the previous topic, Słowianek and Leszczynska also reported the percentage inhibition of DPPH in methanol-extracts of ginger, sweet basil, and turmeric [60]. Compared with our results, the DPPH and ABT radical scavenging activities of ginger, sweet basil, and turmeric were excellent and showed a high inhibition rate. Nikolic et al. reported on the DPPH and ABT content of sweet basil. The results showed a relatively small amount of equivalent content [61]. Assefa et al. also reported on various herbs and spices, including galangal, coriander, turmeric, sweet basil, white pepper, and ginger [62]. Lu et al. reported the DPPH activity of galangal, ginger, and white pepper [63]. In conclusion, turmeric was mostly reported because of its high efficiency in both DPPH and ABTs, equivalent to all types of extracts, and the results were consistent with findings from other researchers.
Effect of herb and spice powders addition to fish on lipid oxidation
Lipid peroxide levels in fish mixed with herbs and spices
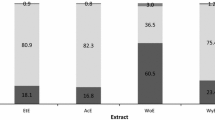
The LPO levels in fish mixed with herbs and spices are shown in Fig. 2a. This bar graph represents the LPO levels in fresh fish meat, the control, and fish meat with herb and spice powder. The LPO content of the control increased when compared with fresh fish meat. The LPO content of fish meat with kaffir lime leaves, pandan leaves, sweet basil, climbing wattle, and coriander increased. In other samples which included galangal, finger root, ginger, and lemongrass, the LPO levels decreased, whereas only turmeric and white pepper samples were not different from the control. In agreement with the total polyphenol content and radical scavenging activity sections, galangal, fingerroot, and ginger had a higher total polyphenol content and radical scavenging activity than the other samples. Normally, lipid oxidation can easily occur in food, especially food that contains considerable amounts of oil emulsion, such as salad dressing, mayonnaise, and milk, which could worsen food quality, including taste, smell, texture, and nutritional quality [64]. Notably, lipid oxidation in fish can occur anytime between feeding in the aquaculture process and the cooking of the fish, meaning that without careful handling, the fish will get spoiled [65]. Because of this condition, many researchers have studied lipid oxidants in fish and examined them by adding several components, such as tea catechins for minced meat or grape extract in fish [66, 67]. Herbs and spices are widely used to prevent lipid oxidation in food. For example, Nikousaleh and Prakash reported the effect of cloves and cinnamon on meat sausage, and Juntachote et al. used dried holy basil to cook ground pork [68, 69]. The studies all reported that the addition of a high antioxidant-containing substance could better prevent lipid oxidation. In conclusion, the results of preventing lipid oxidation by galangal, fingerroot, and ginger in this study were consistent with other researchers’ reports.
Effect of herbs and spices powders on lipid oxidation of fish. (a) LPO (lipid oxidation), (b) HHE (4-hydroxy-trans-2-hexenal), and (c) propanal. Fresh fish meat; frozen minced fish meat was not subjected to oxidation. Control; ten grams of frozen minced fish meat was subjected to oxidation in a water bath at 30℃ for 15 h. Fish meat with herbs and spices powder; ten grams of frozen minced fish meat and 0.5 g of herbs and spices powder were mixed well, then the mixed was subjected to oxidation in a water bath at 30℃ for 15 h. Each data are presented as the mean ± SD (standard deviation)
Aldehyde level in fish mixed with herbs and spices
The aldehyde levels in fish mixed with herbs and spices are shown in Figs. 2b and c. As mentioned in the lipid peroxide level in the fish mixed with herbs and spices section, Figs. 2b and c represent the amounts of HHE and propanal in fresh fish meat, the control, and fish meat with herb and spice powder. In this study, we focused on HHE and propanal as oxidation indicators in fish. These aldehydes are produced by the oxidation of n-3 fatty acids, which are abundant in fish products, and their levels increase owing to quality deterioration. In a previous study, we evaluated the quality of frozen saury, frozen tuna, and seaweed by using HHE and propanal levels as oxidative indicators [70,71,72]. In the present study, the HHE and propanal levels of the samples with herbs and spices were decreased. Moreover, aldehyde levels in most of the samples decreased. Lemongrass contained the highest aldehyde content among the herbs and spices that were examined, but still showed a significant difference between the fresh fish meat and control. Pushpakumari, Mathew, and Joseph reported that lemongrass had a high aldehyde content. These results can explain the higher HHE and propanal levels in lemongrass-mixed samples compared to the others [73, 74]. Aldehyde group components were produced as the main product during lipid oxidation, indicating that higher lipid oxidation occurred and higher contents of the aldehyde group were present [75, 76]. As mentioned in the section on lipid peroxide levels in fish mixed with herbs and spices, if lipid oxidation prevention by herbs and spices was successful, the aldehyde content in fish with herbs and spices with high polyphenol contents and radical scavenging activities should be lower than that of the control and fresh fish samples. In particular, the results of this study are presented in a similar manner and a superior result for lipid oxidation could be observed. This suggests that herb and spice powders could potentially prevent aldehyde creation during lipid oxidation in fish or reduce the lipid oxidation activity.
Correlation between antioxidant compounds and antioxidant capacity
The correlation between antioxidant compounds and antioxidant capacity is shown in Table 9. This table presents the correlation coefficient (r) between antioxidant capacity (DPPH or ABTs radical scavenging activity) and antioxidative compounds (total polyphenol, tocopherol, and amino acids). The correlation between ABTs radical scavenging activity and the polyphenol content in lipophilic extracts had the strongest positive correlation with r = 0.968, followed by the correlation between ABTs radical scavenging activity and the polyphenol content in methanol-extracts (r = 0.954), ABTs radical scavenging activity of water-soluble Arg (r = 0.815), and ABT radical scavenging activity of water-soluble Gly (r = 0.750). According to Gogtay and Thatte, these strong positive correlations imply that the factors are positively related to each other [77]. In this case, if the total polyphenol content of the lipophilic—or methanol-soluble extract is high, the ABTs radical scavenging activity results of the lipophilic—or methanol-soluble extracts are also high. As mentioned in the amino acid analysis results section, this also confirms that Arg and Gly act as antioxidants that can affect the ABTs radical scavenging activity of the water-soluble extracts. Similarly, an r-value higher than 0.5 also confirmed that the relationship between the two factors was still present [77]. Thus, the results of the total polyphenol content and DPPH radical scavenging activity of lipophilic extracts (r = 0.618) and ABTs radical scavenging activity of water-soluble Lys (r = 0.614) showed positive relationships with each other. However, the results of other interactions, including negative relationships, showed a weak relationship with each other due to an r-value near 0, which means that they are not affected by each other.
Assortment by principal component analysis
PCA was used to evaluate the relationship between 56 variable chemical substances and 11 herbs and spices from Thailand (Fig. 3). An estimated 48.52% of the variability could be attributed to the first two dimensions. PC1 and PC2 accounted for 29.99% and 18.93% of the variance, respectively. The results were divided into three groups: G1, G2, and G3 (Fig. 3a). G1, which included pandan leaves, sweet basil, climbing wattle, and coriander, was located to the right of the y-axis. G2, including turmeric, was located at the top of the x-axis and to the left of the y-axis. G3, which is comprised of the remaining samples, was located at the bottom of the x-axis and to the left of the y-axis. Compared with Fig. 3b G1, contained a larger variety of amino acids, organic acids, AMP, linoleic acid (18:3n-3), and ABTs in the water-soluble extracts. However, this area also included LPO, which represents a higher lipid oxidation content in fish. For G2, most of the radical scavenging activities, including that of ABTs in lipophilic and methanol-soluble extracts and the total polyphenol content of lipophilic, water-, and methanol-extracts, were located along with stearic acid (18:0), oxalic acid, and campesterol. G3 contained various sterol groups, DPPH of water- and methanol-soluble extracts, oleic acid (18:1n-9), asclepic acid (18:1n-7), α- tocopherol, acetic acid, butyric acid, and cysteine (Cys-Cys). This group also showed elevated levels of HHE and propanal. As shown in Fig. 3b, most components located in G1 were water-soluble. This result is consistent with the prediction that the ABTs radical scavenging activity of the water-soluble extracts in G1 were higher than that of G2 and G3. The G2 extracts, excluding those that were water-soluble, had a strong relationship with the total polyphenol content and ABTs radical scavenging activity, which agreed with the result that turmeric had the highest radical scavenging activities. In addition, most of the G3 extracts contained a variety of chemical components and the position of the DPPH radical scavenging activity could be determined, excluding lipophilic extracts, acetic acid, butyric acid, Cys-Cys, and Pro, which represented the components in water- and methanol-soluble extracts, and α- tocopherol, 18:1n-7, and ergosterol, which represented the components in lipophilic extracts. Both kaffir lime leaves and lemongrass were used as aroma oils, meaning that they were dominant in lipophilic extracts, whereas galangal, fingerroot, and ginger were more dominant in water-soluble extracts. White pepper was the only sample that showed favorable results for the methanol-soluble extract.
Score and loading plots of principal component analysis of 11 herbs and spices in Thailand a Score plot showing 11 herbs and spices from Thailand. The detail information each sample were shown in Table 1. b Loading plot showing the distribution of the 56 functional chemical compounds analyzed in this study: 14:0 (myristic acid), 15:0 (pentadeaylic acid), 16:0 (palmitic acid), 18:0 (stearic acid), 18:1 n-9 (oleic acid), 18:1 n-7 (vaccenic acid), 18:2 n-6 (linoleic acid), 18:3 n-3 (α-linolenic acid), α-Toc (α-Tocopherol), γ-Toc (γ -Tocopherol), Desmosterol (desmosterol), Ergosterol (ergosterol), Campesterol (campesterol), Stigmasterol (stigmasterol), β-Sitosterol (β-sitosterol), Asp (asparagine), Glu (glutamic acid), Ser (serine), Gly (glycine), His (histidine), Tau (taurine), Thr (threonine), Ala (alanine), Arg (arginine), Pro (proline), Tyr (tyrosine), Cys-Cys (cysteine), Val (valine), Ile (isoleucine), Leu (leucine), Lys (lysine), Phe (phenylalanine), CMP (cytidine monophosphate), AMP (adenosine monophosphate), Oxalic (oxalic acid), Maleic (maleic acid), Citric (citric acid), Tartaric (tartaric acid), Succinic (succinic acid), Fumaric (fumaric acid), Acetic (acetic acid), Pyroglutamic (pyroglutamic acid), Isobutyric (isobutyric acid), Butyric (butyric acid), P-Lipid (total polyphenol of the lipid-soluble extracts), P-Water (total polyphenol of the water-soluble extracts), P-Methanol (total polyphenol of the methanol-soluble extracts), D-Lipid (DPPHs scavenging activity of the lipid-soluble extracts), D-Water (DPPH scavenging activity of the water-soluble extracts), D-Methanol (DPPH scavenging activity of the methanol-soluble extracts), A-Lipid (ABTs scavenging activity of the lipid-soluble extracts), A-Water (ABTs scavenging activity of the water-soluble extracts), A-Methanol (ABTs scavenging activity of the methanol-soluble extracts), LPO (lipid oxidation), HHE (4-hydroxy-trans-2-hexenal), and propanal
Conclusion
The lipophilic and water-soluble components, total polyphenol content, and radical scavenging activity of 11 herbs and spices from Thailand were determined. In particular, there are few reports on water-soluble components, including amino acids, purine, pyrimidine, and organic acids, as well as fat-soluble components, such as fatty acids, sterols, and tocopherol. These chemical components may have an antioxidant capacity and contain polyphenols. When these herbs and spices were added to fish and oxidized, most of the herbs and spices prevented lipid oxidation and aldehyde production. This result involved the polyphenols present in herbs, spices, and other antioxidant substances. The PCA results also showed a strong correlation between the chemical components and samples. Among the samples, turmeric, fingerroot, and ginger showed the best results, especially for antioxidant-related compounds. All the results were consistent with those of other researchers. Herbs and spices are used in food processing to handle livestock and fish to prevent quality deterioration and suppress odors. In addition, the nutritional components of the herbs and spices may be ingested by eating livestock and fish with herbs and spices. In particular, it is possible to ingest plant sterols that are rarely found in livestock and fish. This data was used for the development of food preservation methods and other applications that could improve human health. More data on herbs and spices is required for future experimentation on, for example, the differentiation of each type, including fresh or edible extracts that produce similar results or other herbs and spices that were not mentioned in this report because most of the local herbs and spices in Thailand are not well known.
Data availability
All data generated or analyzed during this study are included in this published article.
References
H.T.W. Tan, in Thai Cuisine, In ed. By L.W. Shan (Times Editions - Marshall Cavendish, Hougang Singapore), p. 8–9 (2005)
S. Sriwattana, V. Haruthaithanasan, P. Chompreeda, A.V.A. Resurreccion, Kasetsart J. Soc Sci. 23, 139–150 (2002)
P. Khanthapok, S. Sukrong, J Food Health Bioenviron Sci. 12, 54–67 (2019)
C. Danyuthasilpe, K. Amnatsatsue, C. Tanasugarn, P. Kerdmongkol, A.B. Steckler, Health Prom. Int. 24, 394–403 (2009). https://doi.org/10.1093/heapro/dap038
N. Siwarungson, Int J Food Sci Nutr. 2, 20–22 (2017)
M.E. Embuscado, J Funct Food. 18, 811–819 (2015). https://doi.org/10.1016/j.jff.2015.03.005
A. Jiang, J. AOAC Int. (2019). https://doi.org/10.5740/jaoacint.18-0418
E.I. Opara, M. Chohan, Int J Mol Sci. 15, 19183–19202 (2014). https://doi.org/10.3390/ijms151019183
L.C. Tapsell, I. Hemphill, L. Cobiac, C.S. Patch, D.R. Sullivan, M. Fenech, S. Roodenrys, J.B. Keogh, P.M. Clifton, P.G. Williams, V.A. Fazio, K.E. Inge, Med. J Aust. 185, s1–s24 (2006). https://doi.org/10.5694/j.1326-5377.2006.tb00548.x
J. Gómez-Estaca, C. López-de-Dicastillo, P. Hernández-Muñoz, R. Catalá, R. Gavara, Trends Food Sci. Technol. 35, 42–51 (2014). https://doi.org/10.1016/j.tifs.2013.10.008
M. Ito, K. Koba, R. Hikihara, M. Ishimaru, T. Shibata, H. Hatate, R. Tanaka, Food Chem. 255, 147–156 (2018). https://doi.org/10.1016/j.foodchem.2018.02.070
M. Yoshida, C.A. Vanstone, W.D. Parsons, J. Zawistowski, P.J. Jones, Eur. J. Clin Nutr. 60, 529–537 (2006). https://doi.org/10.1038/sj.ejcn.1602347
R. Tanaka, K. Uchida, M. Ishimaru, M. Ito, N. Matsumoto, Y. Taoka, H. Hatate, J. Food Meas. Charact. 12, 200–208 (2018). https://doi.org/10.1007/s11694-017-9631-1
M. Ishimaru, M. Haraoka, H. Hatate, R. Tanaka, Food Anal Method. 9, 1606–1615 (2016). https://doi.org/10.1007/s12161-015-0341-1
H. Sekizawa, S. Yamashita, K. Tanji, S. Okoshi, K. Yoshioka, Nippon Shokuhin Kagaku Kogaku Kaishi 60, 212–217 (2013). https://doi.org/10.3136/nskkk.60.212
N. Jongsawatsataporn, R. Tanaka, Food Anal. Method. 15, 1331–1340 (2022). https://doi.org/10.1007/s12161-021-02199-7
S. Lim, A.-H. Choi, M. Kwon, E.-J. Joung, T. Shin, S.-G. Lee, N.-G. Kim, H.-R. Kim, Food Chem. 278, 178–184 (2019). https://doi.org/10.1016/j.foodchem.2018.11.058
X. Wang, M. Xu, J. Cheng, W. Zhang, X. Liu, P. Zhou, Meat Sci. 154, 22–28 (2019). https://doi.org/10.1016/j.meatsci.2019.04.003
T. Nakamura, R. Tanaka, Y. Higo, K. Taira, T. Takeda, Fish Sci. 64, 617–620 (1998). https://doi.org/10.2331/fishsci.64.617
R. Tanaka, Y. Sugiura, T. Matsushita, J Liq. Chromatogr. Rel. Technol. 36, 881–896 (2013). https://doi.org/10.1080/10826076.2012.678454
E.N. Aquino-Bolaños, L. Mapel-Velazco, S.T. Martín-del-Campo, J.L. Chávez-Servia, A.J. Martínez, I. Verdalet-Guzmán, Int. J. Food Prop. 20, 1262–1269 (2017). https://doi.org/10.1080/10942912.2016.1206125
E. Reszczyńska, A. Hanaka, Cell Biochem. Biophys. 78, 401–414 (2020). https://doi.org/10.1007/s12013-020-00947-w
Z.R. Nengroo, A. Rauf, Ind Crop Prod. 140, 111596 (2019). https://doi.org/10.1016/j.indcrop.2019.111596
A. Savych, R. Basaraba, N. Muzyka, P. Ilashchuk, Pharmacia. 68, 433–439 (2021)
M. Neffati, B. Marzouk, Acta Physiol. Plantarum. 31, 455–461 (2009). https://doi.org/10.1007/s11738-008-0253-4
S. Rajaram, Am. J. Clin. Nutr. 100, 443S-448S (2014). https://doi.org/10.3945/ajcn.113.071514
P. Daga, S.R. Vaishnav, A. Dalmia, A.W. Tumaney, J. Food Sci. Tech. 59, 518–531 (2022). https://doi.org/10.1007/s13197-021-05036-1
R.K. Saini, A.D. Assefa, Y.-S. Keum, Foods. 10, 854–867 (2021). https://doi.org/10.3390/foods10040854
A.P. Simopoulos, Nutrients 8, 128–129 (2016). https://doi.org/10.3390/nu8030128
L. Yang, Z. Song, W. Cao, Y. Wang, H. Lu, F. Guo, H. Yang, J. Chen, S. Wang, G. Sun, J. Hyg. Res. 45, 436–441 (2016)
S.-H. Xia, J. Wang, J.X. Kang, Carcinogenesis 26, 779–784 (2005). https://doi.org/10.1093/carcin/bgi019
V. Wijendran, K.C. Hayes, Annu. Rev Nutr. 24, 597–615 (2004). https://doi.org/10.1146/annurev.nutr.24.012003.132106
S.D. Singer, J. Zou, R.J. Weselake, Plant Sci. 243, 1–9 (2016). https://doi.org/10.1016/j.plantsci.2015.11.003
Q. Jiang, S. Christen, M.K. Shigenaga, B.N. Ames, Am J Clin Nutr. 74, 714–722 (2001). https://doi.org/10.1093/ajcn/74.6.714
E. Reiter, Q. Jiang, S. Christen, Mol Aspects Med. 28, 668–691 (2007). https://doi.org/10.1016/j.mam.2007.01.003
E. Dertyasasa, W. Tunjung, Biosci. Biotech. Res. Asia 14, 1235–1250 (2017). https://doi.org/10.13005/bbra/2566
A. Jelled, Â. Fernandes, L. Barros, H. Chahdoura, L. Achour, I.C.F.R. Ferreira, H.B. Cheikh, Ind. Crop Prod. 77, 30–35 (2015). https://doi.org/10.1016/j.indcrop.2015.08.052
D.J.M. Gómez-Coronado, E. Ibañez, F.J. Rupérez, C. Barbas, J Chromatogr. 1054, 227–233 (2004). https://doi.org/10.1016/j.chroma.2004.08.072
S. Babu, S. Jayaraman, Biomed. Pharmacother. 131, 110702 (2020). https://doi.org/10.1016/j.biopha.2020.110702
J. Chaudhary, A. Jain, N. Kaur, L. Kishore, Int J Pharm Sci Res. 2, 2259–2265 (2011). https://doi.org/10.13040/IJPSR.0975-8232.2(9).2259-65
R.K. Saini, M.-H. Song, J.-W. Yu, X. Shang, Y.-S. Keum, Foods 10, 2378–2395 (2021). https://doi.org/10.3390/foods10102378
S.C. Gupta, B. Sung, J.H. Kim, S. Prasad, S. Li, B.B. Aggarwal, Mol Nutr Food Res. 57, 1510–1528 (2013). https://doi.org/10.1002/mnfr.201100741
S. Li, Pharm Crop. 5, 28–54 (2011). https://doi.org/10.2174/2210290601102010028
B. Derek, P.F. Walter, L.N. Rosamond in Soybean Production and Supply Chains in the Tropics. In ed. By B. Derek, P.F. Walter, L.N. Rosamond (Oxford University Press, New York) pp 66–91 (2017)
M. Martínez-Tomé, F. García-Carmona, M.A. Murcia, J. Sci. Food Agric. 81, 1019–1026 (2001). https://doi.org/10.1002/jsfa.889
O.B. Ajayi, S.F. Akomolafe, F.T. Akinyemi, ISRN Nutrition. 2013, 359727 (2013). https://doi.org/10.5402/2013/359727
O. Ifeanyi, P.-D. Jiwuba, C. Ezeokeke, M. Uchegbu, O. Ifeanyi Charles, M. Iloeje, Int. J. Agr. Rural. Dev. 17, 1663–1670 (2014)
M.S. Almuhayawi, S.K. Al Jaouni, S.M. Almuhayawi, S. Selim, M. Abdel-Mawgoud, Food Chem. 357, 129730 (2021). https://doi.org/10.1016/j.foodchem.2021.129730
M.J. Lopez, S.S. Mohiuddin, Biochemistry, Essential Amino Acids. (StatPearls Publishing, 2021), https://www.ncbi.nlm.nih.gov/books/NBK557845/. Accessed 28 April 2022
W. Duan, Y. Huang, J. Xiao, Y. Zhang, H. Zhang, Int. J. Food Prop. 23, 340–353 (2020). https://doi.org/10.1080/10942912.2020.1720712
W. Duan, Y. Huang, J. Xiao, Y. Zhang, Y. Tang, Food Sci. Nutr. 8, 3777–3792 (2020). https://doi.org/10.1002/fsn3.1667
D. Chia, T. Yoder, W.-D. Reiter, S. Gibson, Planta 211, 743–751 (2000). https://doi.org/10.1007/s004250000345
J.E. Amoore, Chem. Senses. 2, 267–281 (1977). https://doi.org/10.1093/chemse/2.3.267
K. Hey, S. Juran, M. Schäper, S. Kleinbeck, E. Kiesswetter, M. Blaszkewicz, K. Golka, T. Brüning, C. van Thriel, Neurotoxicology 30, 1223–1232 (2009). https://doi.org/10.1016/j.neuro.2009.08.009
G. Turner-Walker, The Removal of Fatty Residues from a Collection of Historic Whale Skeletons in Bergen: An Aqueous Approach to Degreasing. (Researchgate, 2012), https://doi.org/10.13140/2.1.1996.8969. Accessed 28 April 2022
T. Kayashima, T. Katayama, BBA-Gen Subjects. 1573, 1–3 (2002). https://doi.org/10.1016/S0304-4165(02)00338-0
S. Tantipopipat Jr., A. Boonpraderm, S. Charoenkiatkul, E. Wasantwisut, P. Winichagoon, Malays J. Nutr. 16, 137–148 (2010)
W. Wangcharoen, T. Ngarmsak, B.H.P. Wilkinson, Kasetsart J. Nat. Sci. 36, 426–434 (2002)
A.A. Alafiatayo, A. Syahida, M. Mahmood, Afr. J. Tradit. Complem. 11, 7–13 (2014). https://doi.org/10.4314/ajtcam.v11i3.2
M. Słowianek, J. Leszczynska, Herba Polonica. (2016). https://doi.org/10.1515/hepo-2016-0003
J.S. Nikolic, V.D. Mitic, V.P. Stankov Jovanovic, M.V. Dimitrijevic, G.S. Stojanovic, J. Food Meas. Charact. 13, 216–217 (2019). https://doi.org/10.1007/s11694-019-00137-0
A.D. Assefa, Y.-S. Keum, R.K. Saini, J. Food Meas. Charact. 12, 1548–1555 (2018). https://doi.org/10.1007/s11694-018-9770-z
M. Lu, B. Yuan, M. Zeng, J. Chen, Food Res. Int. 44, 530–536 (2011). https://doi.org/10.1016/j.foodres.2010.10.055
T. Waraho, D.J. McClements, E.A. Decker, Trends Food Sci. Technol. 22, 3–13 (2011). https://doi.org/10.1016/j.tifs.2010.11.003
G. Secci, G. Parisi, Ital J. Anim Sci. 15, 124–136 (2016). https://doi.org/10.1080/1828051x.2015.1128687
S. Tang, D. Sheehan, D.J. Buckley, P.A. Morrissey, J.P. Kerry, Int. J. Food Sci. Tech. 36, 685–692 (2001). https://doi.org/10.1046/j.1365-2621.2001.00497.x
I. Sánchez-Alonso, A. Jiménez-Escrig, F. Saura-Calixto, A.J. Borderías, Food Chem. 101, 372–378 (2007). https://doi.org/10.1016/j.foodchem.2005.12.058
A. Nikousaleh, J. Prakash, J Food Sci Technol. 53, 1993–2000 (2016). https://doi.org/10.1007/s13197-015-2113-8
T. Juntachote, E. Berghofer, S. Siebenhandl, F. Bauer, Food Chem. 100, 129–135 (2007). https://doi.org/10.1016/j.foodchem.2005.09.033
R. Tanaka, M. Ishimaru, H. Hatate, Y. Sugiura, T. Matsushita, Food Chem. 212, 104–109 (2016). https://doi.org/10.1016/j.foodchem.2016.05.166
R. Tanaka, K. Naiki, K. Tsuji, H. Nomata, Y. Sugiura, T. Matsushita, I. Kimura, J Food Process Preserv. 37, 325–334 (2013). https://doi.org/10.1111/j.1745-4549.2011.00652.x
R. Tanaka, N. Nakazawa, T. Maeda, H. Fukushima, R. Wada, Y. Sugiura, T. Matsushita, H. Hatate, Y. Fukuda, J Aquat Food Prod Technol. 25, 1073–1085 (2016). https://doi.org/10.1080/10498850.2015.1010679
K.N. Pushpakumari, V.A. Paul, Studies on Lemongrass Oil. (Cochin University of Science and Technology, 1987), https://dyuthi.cusat.ac.in/xmlui/bitstream/handle/purl/3314/Dyuthi-T1314.pdf?sequence=1. Accessed 28 April 2022
S. Mathew, B. Joseph (2008). Specifications on essential oils. In: J. P.P, G. Mathew, A. Joseph, S. Mathew, & B. P. Skaria, National workshop on grower - industry linkage for promotion of medicinal and aromatic plants cultivation (pp. 63–68). Cochin, India: Peevee printers.
T. Miyasaki, M. Hamaguchi, S. Yokoyama, J Food Sci. 76, C1319–C1325 (2011). https://doi.org/10.1111/j.1750-3841.2011.02388.x
Q. Zhang, Y. Ding, S. Gu, S. Zhu, X. Zhou, Y. Ding, Food Res Int. 137, 109339 (2020). https://doi.org/10.1016/j.foodres.2020.109339
N. Gogtay, U. Thatte, J Assoc Physician I. 65, 78–81 (2017). https://www.japi.org/q274a4c4/principles-of-correlation-analysis. Accessed 22 July 2022
Acknowledgements
The authors would like to thank Editage (www.editage.com) for English language editing.
Funding
This study was funded by JSPS KAKENHI Grant Number 21K05785.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. NJ: conceptualization, methodology, investigation, data curation, and writing (original draft preparation). SK: methodology, investigation, and data curation. RT: conceptualization, methodology, investigation, data curation, writing (original draft preparation), and supervision.
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jongsawatsataporn, N., Kido, S. & Tanaka, R. Evaluation of chemical components of herbs and spices from Thailand and effect on lipid oxidation of fish during storage. Food Measure 17, 346–361 (2023). https://doi.org/10.1007/s11694-022-01624-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-022-01624-7