Abstract
A structural molar volume model based on the silicate tetrahedral Q-species has been developed to accurately predict the molar volume of molten oxides. In this study, the molar volumes of ternary and multicomponent melts in the Li2O-Na2O-K2O-MgO-CaO-MnO-PbO-Al2O3-SiO2 system are reviewed and compared with the predicted molar volumes from the newly developed structural model. The model can accurately predict the molar volumes using binary model parameters without any ternary or multicomponent parameters. The nonlinear behavior in the molar volume of silicate melts is well predicted by the present model.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In the Part I of this series, a structural molar volume model for binary melts was introduced. The model is based on the silicate Q n species which are calculated from the Modified Quasichemical Model[1] with optimized thermodynamic data from FactSage FToxid database.[2] All the unary and binary experimental data were critically reviewed based on experimental technique and the molar volume parameters of unary oxide component and binary Q n species were optimized to reproduce the experimental data.
As Part II of this series, the predictive ability of the present molar volume model is demonstrated for ternary and multicomponent melts without any additional model parameters. All experimental data of the ternary and multicomponent melts in Li2O-Na2O-K2O-MgO-CaO-MnO-PbO-Al2O3-SiO2 system are reviewed, and the comparison of the predicted values from the present model with experimental data is presented.
Structural Molar Volume Model
The structural molar volume model is based on the molar volume of different Q-species. As discussed in the Part I, silicate tetrahedral Q n species where n = 0 to 4 are calculated from the Modified Quasichemical Model (MQM)[1] with optimized thermodynamic model parameters (stored in FactSage FToxid database[2]). The amounts of different Q n species for the binary silicate melt can be calculated considering the bond fractions of bridged oxygen O0, broken oxygen O−, and free oxygen O2−. In the MQM, the bridged, broken, and free oxygen for example in a MO-SiO2 melt are described as Si-Si, M-Si, and M-M pairs, respectively. This is demonstrated in Part I. The same approach was used to calculate the Q n species for ternary and multicomponent melts. Of course, in the ternary and multicomponent melt, the broken oxygen can be generated by different network modifiers, and the total number of all broken oxygens is counted in the calculation of Q n species.
The molar volumes of the ternary and higher order melts are predicted from the unary and binary model parameters by first calculating the quantity of each Q n species for a given melt and performing a linear extrapolation from the binary parameters weighted by the quantity of each M-Si pairs:
where \( n_{{Q^{n} }} \) and n t are the amount of Q n species and total number of oxygen in 1 mol of liquid, and i and j are cations of network modifiers. \( x_{{{\text{Si}} - i}} \) and \( x_{i - j} \) are bond fractions of broken oxygen (Si−i pairs) and free oxygen (i–j pairs) in 1 mol of liquid. The molar volume parameters of pure SiO2 \( \left( {a_{{{\text{SiO}}_{2} }} + b_{{{\text{SiO}}_{2} }} T} \right) \), \( Q^{n} \left( {a_{{n{\text{M}}}} + b_{{n{\text{M}}}} T} \right), \) where n = 0 to 3, and MO \( \left( {a_{{{\text{M}} - {\text{M}}}} + b_{{{\text{M}} - {\text{M}}}} T} \right) \) are obtained by the optimization of the molar volume of binary MO-SiO2 melt. The last term in Eq. [1] incorporates the effect of free oxygen species. The molar volume of M i –M j (a i–j + b i–j T) is the average of the molar volume of M i –M i (a i–i + b i–i T) and M j –M j (a j–j + b j–j T).
It should be noted that Al2O3, an amphoteric component, is treated as a network modifier in the present model. As seen in Part I of this study, the molar volumes of the CaO-Al2O3 and MgO-Al2O3 melts change linearly with composition which can be described by the last term in Eq. [1]. Therefore, no additional complication like Q-species was required for Al2O3. In the case of alkali oxide, A2O, A2 was used as a cation unit (for example, Na2 instead of Na) in the model.
The structure of silicate melts of ternary and higher order system can be calculated at a given composition and temperature from the MQM with thermodynamic database. This structural change is automatically taken into account in the molar volume calculations as described in Eq. [1].
Prediction of Molar Volume in Ternary and Higher Order Systems
The present structural molar volume model can predict the molar volume of ternary and higher order molten oxides from unary and binary model parameters without additional parameters. In the prediction, the structures of melts (Q i species and free oxides) are calculated from the FToxid thermodynamic database.[2]
In the following sections, each ternary system is examined individually. A ternary plot is shown to illustrate the composition ranges studied. To show the accuracy of the model, an experimental data vs prediction plot (1 to 1 plot) is also shown with 2 pct error bars. Some key systems are further examined by showing pseudo-binary sections to illustrate the trends in the experimental data and the model predictions. The results from the linear model of Lange and Carmichael[3] are also shown as dashed lines for comparison. All compositions are in mole percent unless otherwise specified. All the systems examined and the associated references are shown in Table I.
Ternary Alkali Silicate Melts
Li2O-Na2O-SiO2 system
The ternary Li2O-Na2O-SiO2 system was studied along two different iso-composition joins as shown in Figure 1(a). Din[5] used the SBA technique while Golovin and Dertev[8] used the DBA technique. Bockris et al.[4] examined a single composition with equi-molar Li2O and Na2O using the DBA technique and Sasek and Lisy[6] did the same using the SBA technique. All available experimental data were measured in the high silica region and are consistent with each other. The present model can predict all the results within ~1 pct error as shown in Figure 1(b). To better illustrate the prediction of the present model, two joins are examined in Figure 2 (constant SiO2 and constant Na2O). The present model and the linear model are nearly identical across the constant silica join. They are nearly equivalent along the constant Na2O join in the high silica region, but the present model predicts a higher molar volume in the Li2O-Na2O system because the molar volumes of Li2O and Na2O in the present model are much higher than those in the linear model of Lange and Carmichael.
Li2O-K2O-SiO2 system
The Li2O-K2O-SiO2 system was examined along a single silica join by Sasek and Lisy[6] using the SBA method. Bockris et al.[4] examined a single composition. The accuracy of the model and corresponding compositions are shown in Figure 3. The composition closer to the K2O-SiO2 binary system by Sasek and Lisy (circled) showed nonlinear behavior with temperature.
Na2O-K2O-SiO2 system
The ternary Na2O-K2O-SiO2 system was examined by three research groups. Sasek and Lisy[6] used the SBA method, while the other two research groups, Dertev and Golovin[8] and Lange and Carmichael[11], used the DBA method. Dertev and Golovin examined an iso-SiO2 join and found the molar volume increases linearly from the Na2O to the K2O side. All the experimental data are predicted within 1 pct error, as shown in Figure 4.
Ternary Lead Oxide Silicate Melts
The ternary lead oxide silicates have been studied in very few works. Nearly all the publications were produced by Japanese research groups using the SBA method and published graphically. In addition, the majority of the data were obtained from a single research group (Hino et al.[15,19] and Ejima et al.[10]). The results in the PbO-SiO2 system by Hino et al. are not considered to be the most accurate (as shown in Part I) so there is a somewhat consistent difference between the experimental data and present predictions.
Suginohara et al.[9] also performed measurements in the PbO-SiO2 system with small additions (<5 mol pct) of other oxides, but the results have been omitted as accurate data extraction was not possible from the figures provided in their paper.
Li2O-PbO-SiO2 system
This ternary system was studied by Ejima et al.[10] along two joins (50 mol pct SiO2 and equi-molar PbO:SiO2) using the SBA method. The compositions measured and the model accuracy can be seen in Figure 5. The molar volume along the two joins is examined in Figure 6. The results suggest a linear decrease in molar volumes with increasing Li2O concentration at constant SiO2, which is well predict by the present model. At higher Li2O concentrations of the PbO/SiO2 = 1 section the experimental data shows a significant deviation from the linear trend. This abrupt change seems unlikely because it would require a significant structural change in the liquid. This might have occurred due to the formation of a solid phase. Similar observations were made in other alkali bearing lead oxide systems.
The ternary Li2O-PbO-SiO2 system: (a) compositions measured and (b) model accuracy[10]
The molar volume in the Li2O-PbO-SiO2 system across the (a) SiO2 = 50 mol pct and (b) PbO/SiO2 = 1 joins[10] [filled symbols at 1473 K (1200 °C) and open symbols at 1273 K (1000 °C)]
Na2O-PbO-SiO2 system
The molar volume of the Na2O-PbO-SiO2 system was examined by Hino et al.[15] (the same research group examined the Li2O-PbO-SiO2 system) and published their results graphically. A single composition at multiple temperatures was also measured by Kostanyan and Karapetyan.[14] Both research groups used a SBA method. As shown in Figure 7, the molar volume data by Kostanyan and Karapetyan are much higher than the data by Hino et al. The predicted molar volumes are closer to the data by Hino et al. and the increasing trend in molar volume with increasing Na2O content is well replicated. The scatter in the data is significant across both joins (constant SiO2 and equi-molar ratio of PbO and SiO2) as shown in Figure 8.
The molar volume in the Na2O-PbO-SiO2 system across the (a) SiO2 = 50 mol pct and (b) PbO/SiO2 = 1 joins[15] [filled symbols at 1473 K (1200 °C) and open symbols at 1273 K (1000 °C)]
K2O-PbO-SiO2 system
Kostanyan and Karapetyran[14] and Hino et al.[19] also examined the potassium oxide-containing system. The experimental data and the present model agree moderately well. The investigated compositions and the model accuracy are shown in Figure 9. The molar volume across two joins (50 pct SiO2 and equi-molar PbO:SiO2) increases with the addition of K2O, which are well predicted by the present model as shown in Figure 10. The one experimental point at 50 pct K2O circled in Figure 9, deviated largely from the linear trend of the experimental data and suggests a negative thermal expansion: their data were likely measured sub-liquidus, but no phase diagram information is available.
The molar volume in the K2O-PbO-SiO2 system across the (a) SiO2 = 50 mol pct and (b) PbO/SiO2 = 1 joins[19] [filled symbols at 1473 K (1200 °C) and open symbols at 1273 K (1000 °C)]
MgO-PbO-SiO2 and CaO-PbO-SiO2 systems
The molar volume of the PbO-SiO2 system with MgO or CaO additions were studied by Ouchi et al.[24] using a single bob method for the majority of measurements and the double bob method to determine the corrections required. Additions of up to 12 mol pct MgO or CaO were made. The molar volume decreased linearly with increasing CaO or MgO content. The present model is able to predict all the experimental data within 1 pct error. Ouchi et al. also examined the effect of BaO, SrO, and NiO additions to the PbO-SiO2 system. In all cases, a linear molar volume change with oxide addition was observed along constant silica joins. The compositions examined in the MgO-PbO-SiO2 and CaO-PbO-SiO2, and model accuracy are shown in Figure 11.
The ternary MgO-PbO-SiO2 and CaO-PbO-SiO2 systems: (a) compositions examined and (b) model accuracy.[24] Open symbols represent SBA measurements and filled symbols represent DBA measurements
Ternary Alkali Alkali-Earth Silicate Melts
Li2O-MgO-SiO2 and Li2O-CaO-SiO2 systems
The lithium silicate systems with either MgO or CaO additions have not been studied in depth. Only one composition was studied (72.22 SiO2-9.72 Li2O-18.06 MgO or CaO by mole percent) by Sasek et al.[6] using the SBA method at multiple temperatures. Both the linear model and the present model provide similar results and can predict the experimental data within a 1 pct error limit as shown in Figure 12.
The molar volume for the (a) ternary Li2O-MgO-SiO2 and (b) ternary Li2O-CaO-SiO2 systems at various temperatures with a composition of 72.22 SiO2-9.72 Li2O-18.06 MgO or CaO in mol pct[6]
Na2O-MgO-SiO2 system
The Na2O-MgO-SiO2 system was studied in four independent works. The measurements by Adachi et al.[12] were performed using the sessile drop (SD) method. Their results are not considered to be accurate based on experimental technique and inconsistency with other measurements. The remaining data obtained from Din,[5] Sasek and Lisy[6], and Dertev and Golovin[8] are consistent with each other and are well predicted, as shown in Figure 13. Din et al.[42] published the same results of Din.[5] Two binary sections were examined in more detail: in Figure 14, the constant 66.7 mol pct silica join and the constant 14.3 mol pct Na2O join are shown. The results from the linear model and the present model are nearly equivalent in the high SiO2 region and are in good agreement with the experimental data. They provide different extrapolations at the low-SiO2 region due to the different molar volumes of pure MgO and Li2O used in the two models.
Na2O-CaO-SiO2 system
The Na2O-CaO-SiO2 system was studied in similar composition ranges as the Na2O-MgO-SiO2 system as shown in Figure 15. The work by Din,[5,42] Sasek and Lisy,[6] and Dertev and Golovin[8] are in good agreement with each other and are well predicted. Like the Na2O-MgO-SiO2 system, the results of Adachi et al.[12] (SD method) are less reliable. The results of Coenen[13] using the SBA method were inconsistent with the results of Din at the similar compositions. Dertev and Golovin reported a linear decrease in molar volume at 66.7 pct SiO2 with replacement of Na2O by CaO, which are well predicted by the present model and the linear model.
K2O-MgO-SiO2 system
Only one composition was examined in this ternary system by Sasek et al.[6] using the SBA method: 72.22 SiO2-9.72 K2O-18.06 MgO in mol pct. The experimental data as a function of temperature are well predicted by the present model as shown in Figure 16. Nearly the same results are obtained by the linear model. Both models predict the experimental temperature dependence.
The molar volume of the ternary K2O-MgO-SiO2 system at various temperatures at a composition of 72.22 SiO2-9.72 K2O-18.06 MgO[7] in mol pct
K2O-CaO-SiO2 system
Only two compositions were examined in this ternary system: Sasek and Lisy[6] (72.22 SiO2-9.72 K2O-18.06 CaO in mol pct) using the SBA method and Lange and Carmichael[11] (59.52 SiO2-9.97 K2O-30.51 CaO in mol pct) using the DBA method. Both datasets are well predicted by the present model as a function of temperature as shown in Figure 17.
Ternary Manganese Oxide-Containing Systems
Relatively few measurements have been made in the manganese containing systems with the exception of the CaO-MnO-SiO2 system. In addition, there is no experimental data using the DBA technique. Only imaging techniques and SBA technique which are prone to systematic errors have been used.
Na2O-MnO-SiO2 system
This is the only known alkali system containing manganese oxide studied. Adachi et al.[12] examined three compositions. The predictions from the present model are higher than the experimental data, as shown in Figure 18. The accuracy of the work of Adachi et al.[12] using the SD technique was questioned in several systems. The molar volume of the Na2O-SiO2 system extrapolated from the results by Adachi et al. are noticeably lower than the experimental data of the Na2O-SiO2 system as examined in Part I of this series.
The ternary Na2O-MnO-SiO2 system: (a) compositions examined and (b) the Na2O/SiO2 = 0.8 join[12]
CaO-MnO-SiO2 system
The CaO-MnO-SiO2 system has been well studied. It was examined by Kekelidze et al.[26] (SD method), Segers et al.[27] (SBA method), and more recently by Lee et al.[28] (SBA method). Segers et al. and Lee et al. provided their results in the form of equations with temperature limits, while Kekelidze et al. made measurements at a temperature of 1773 K (1500 °C) and tabulated their results. The measurements of Kekelidze et al. span a wide composition range; however, some of the lowest silica content measurements were made below the liquidus. All three datasets are in good agreement with each other, but the data of Kekelidze are the least consistent. All experimental data are well predicted by the present model and are within 2 pct error as shown in Figure 19.
To further examine this system, three iso-SiO2 sections with 30, 40, and 50 wt pct of SiO2 are presented in Figure 20. Molar volume increases with replacement of MnO with CaO at iso-SiO2 concentrations. These sections illustrate the agreement between authors and the accuracy of the present model. As mentioned above, the results of Kekelidze et al. are the least consistent.
Ternary Alkali-Earth Silicate Melts
MgO-CaO-SiO2 system
The MgO-CaO-SiO2 system is one of the most important metallurgical and geological systems. This system was investigated by Licko and Danek[21] along two pseudo-binary joins (CaMgSi2O6-Ca2MgSi2O7 and CaSiO3-Ca2MgSi2O7) using a SBA technique. The remaining authors[11,17,22,23,43] used the more accurate DBA technique. The examined compositions and the accuracy of the present model can be seen in Figure 21. The experimental data are slightly under-predicted. The deviation from the experimental results of Licko and Danek may be attributed to the SBA technique. The data of Courtial and Dingwell[43] are the least well predicted by the present model (approaching 2 pct deviation). However, it was noted previously that their measurements deviated from those of Tomlinson et al.[44] in both the binary MgO and CaO silicate systems which were used to calibrate the binary model parameters of the present model (see Part I). The two joins are further examined in Figure 22 and demonstrate the predicted trends. The present model provides better accuracy than the linear model of Lange and Carmichael,[3] as can be seen in Figure 22(a).
The molar volume in the MgO-CaO-SiO2 system across the (a) SiO2/MgO = 2 and (b) CaO/SiO2 = 1 joins. The legend is the same Fig. 21, and open and filled symbols represent 1723 K (1450 °C) and 1873 K (1600 °C), respectively
Ternary Alumino-Silicate Melts
Alumino-silicate melts have received a fair amount of attention by the modeling community due to the possibility of significant changes in structure as a function of composition. Bottinga et al.[45] devised a model which attempted to incorporate nonlinear trends as a function of the changes in coordination of aluminum. In the present model, nonlinear trends in the molar volume of alumina were introduced in the SiO2-Al2O3 system. Because of the importance of these alumino-silicate systems, many isopleths are illustrated to prove the accuracy of predictions of the present model.
Na2O-Al2O3-SiO2 system
The sodium containing alumino-silicate system was extensively studied by Riebling et al.[16] using a SBA technique. The measurements were performed along constant silica joins in addition to along the Na2O/Al2O3 = 1 join. Adachi et al.[12] (SD), Coenen[13] (SBA), Nelson and Carmichael[17] (DBA), and Stein et al.[18] (DBA) also investigated the molar volume of this ternary system using various techniques. All experimental compositions are plotted in Figure 23 along with the model accuracy. The molar volume changes in the sections with Na2O/Al2O3 = 1, and iso-SiO2 (50, 60, 75 pct) are also plotted in Figure 24. The results of Stein et al.[18] (DBA) are believed to be the most accurate and are comparable to those of Riebling within ~0.5 pct. Along the Na2O/Al2O3 = 1 section, significant curvature is predicted due to the restrictions on the Na2O-SiO2 system. These two sets of data are very well predicted by the present model as shown in Figures 23 and 24. The experimental data of Adachi et al. and Nelson and Carmichael are lower than the data by Riebling and Stein et al. The results of Coenen are systematically higher than the predicted values from the present model. Both the present model and the linear model of Lange and Carmichael predict similar values in the high SiO2 region.
K2O-Al2O3-SiO2 system
A single composition was examined by Lange and Carmichael[11] (65.84 SiO2, 5.6 Al2O3, 28.56 K2O mol pct) in this ternary system using the DBA method. The experimental data are compared with the values predicted from the present model and the linear model of Lange and Carmichael[3] in Figure 25. Both models can well predict the experimental data.
The molar volume of the ternary K2O-Al2O3-SiO2 system (65.84 SiO2, 5.6 Al2O3, 28.56 K2O mol pct) as a function of temperature[11]
MgO-Al2O3-SiO2 system
The MgO-Al2O3-SiO2 system was systematically studied by Riebling[25] with a SBA method. Measurements by Lange and Carmichael[11] and Courtial and Dingwell[43] were performed using the DBA method. The experimental compositions are shown in Figure 26. The change in molar volume at an iso-SiO2 sections (50 pct SiO2) and a section with MgO/Al2O3 = 1 by Riebling are compared with the predicted data in this study in Figure 27. All the results are reasonably well predicted except for the highest silica content measurements of Riebling (even higher than pure SiO2), circled in Figures 26 and 27.
CaO-Al2O3-SiO2 system
The CaO-Al2O3-SiO2 system has been investigated extensively. Unfortunately, there is considerable disagreement in the available experimental data. The experimental compositions (in both mole and weight percent) along with the model accuracy are shown in Figure 28. The most comprehensive studies have been performed using the less accurate experimental techniques. Kammel et al.[30] (SBA) investigated the change in molar volume of this ternary system at various iso-CaO content joins. However, their data are systematically higher than the results of more accurate DBA measurements by Lange and Carmichael,[11] Taniguchi,[22] Knoche et al.,[23], and Courtial and Dingwell.[37] The measurements in the silica poor region are performed primarily using the MBP technique,[32,33,38,40] with questionable accuracy.
The change in molar volume at constant CaO (30, 40, 45, 50 wt pct CaO) are shown in Figure 29. The molar CaO/Al2O3 = 1 join is shown in Figure 30. All the DBA measurement results are well predicted within a 2 pct error limit. The trends in the results by Kammel et al.[30] are well reproduced although systematically higher than the predictions of the present model. Comparing the present model with the linear model by Lange and Carmichael,[3] the predictions are similar in the high silica region but the model of Lange and Carmichael predicts a significantly lower molar volumes in the silica poor regions. Along the molar CaO/Al2O3 = 1 join, both models predict approximately the same molar volumes at 50 mol pct SiO2 content but the present model suggests significant curvature with silica content resulting from the curvatures observed in molar volumes of the Al2O3-SiO2 and CaO-SiO2 systems.
Ternary Aluminate Melts
Several silica free aluminate systems were examined in the literature. Bochorishvili and Yakobashvili[33] examined the effect of oxide addition to a Al2O3-CaO melt (45 wt pct Al2O3-55 wt pct CaO). Additions of Li2O, Na2O, K2O, MgO, or SiO2 were made and the density change was measured using the MBP method. The alkali bearing systems are not shown due to very limited data but are reasonably well predicted by the present model, considering the error associated with the MBP technique.
The MgO-CaO-Al2O3 system was examined by several research groups.[32,33,40,41] The difficulties with this system are primarily associated with the high melting temperatures. The compositions examined are shown in Figure 31(a). Most of the experimental data were measured up to 5 wt pct MgO composition. The accuracy of the experimental data is questionable with relatively large inconsistencies because all measurements were made using the MBP method except that of Li et al.[41] who used a SBA technique. It was found that the data of Li et al. are questionable; they made no correction for surface tension and the magnitude of their density results suggests an incredibly dense melt. The data of Li et al. are completely inconsistent with the molar volume data of the binary CaO-Al2O3. The 5 wt pct MgO join is shown in Figure 32 and illustrates the significant disagreement with this dataset. Therefore, the results by Li et al.[41] and by Xu et al.[46] from the same research group were not considered in this study. The linear model of Lange and Carmichael[3] differs from the prediction of the present model mainly because of different molar volume data of CaO, but it is not significantly different in the region of experimental studies.
Higher Order Systems
Molar volumes of multicomponent melts were examined for consistency and to ensure that the predictions remained reasonable. The components of the multicomponent systems and the experimental methods are listed in Table II. The prediction accuracy of the present model for several higher order systems can be assessed in Figure 33. The data points with filled symbols are the most accurate studies using the DBA technique and are well predicted.
The MgO-CaO-Al2O3-SiO2 system was investigated by Smolyarenko et al.[31] (MBP) Winterhager et al.[47] (SBA), Dzhincharadze et al.[48] (SD), Yakushev et al.[36] (MBP), Taniguchi[22] (DBA), and Courtial and Dingwell[43] (DBA). Coenen[13] (SBA) studied the Na2O-CaO-Al2O3-SiO2 system. Kekelidze et al.[26] (SD) studied a few compositions in the CaO-MnO-SiO2 system with small additions of Al2O3 which are not well predicted. Nelson and Carmichael[17] (DBA) studied various compositions in multiple quaternary and higher order systems, and the accuracy of their results is uncertain considering the discrepancies observed in several of the lower order systems (ex: Na2O-SiO2). Lange and Carmichael[11] (DBA) also studied various compositions in various higher order systems. Magidson et al.[38] (MBP) studied various Na2O-CaO-Al2O3-SiO2 melts and are not well reproduced; however, they quoted a large error of 4 to 5 pct with their measurements.
Predictions for Iso-molar Volume Contours of Ternary and Quaternary Systems
Iso-molar volume contour plots were calculated in this section to provide some insight into the molar volume behavior of several important ternary and quaternary systems based purely on the predictions made by the present structural molar volume model. The model assumes a single liquid melt. That is, no liquid miscibility gap or solids were considered. All the diagrams are presented in weight percent.
The predicted iso-molar volume contours for the CaO-Al2O3-SiO2 system at 1873 K (1600 °C) are presented in Figure 34(a). According to the prediction, the molar volume of the slag with constant CaO to Al2O3 ratio gradually decreases with increasing SiO2 content up to approximately 50 pct and then begins to increase again above 50 wt pct SiO2. This type of inflection is also demonstrated in Figure 30. Adding Al2O3 into CaO-SiO2 slags increases the molar volume monotonically. The molar volume of the CaO-Al2O3-SiO2 system with 10 wt pct MgO is predicted in Figure 34(b). The addition of 10 wt pct MgO does not change the general trend of the iso-contours. However, MgO addition decreases the overall molar volume with a more pronounced effect in the Al2O3-rich region. The addition of 10 wt pct Na2O to the CaO-Al2O3-SiO2 system is predicted in Figure 34(c). An increase in molar volume is predicted, with a more significant increase in the CaO rich region.
Iso-molar volume contour for the CaO-MgO-SiO2 system are predicted in Figure 35(a). Up to approximately 50 wt pct SiO2, the molar volume linearly decreases with the addition of MgO regardless of the CaO/SiO2 ratio (basicity). Above 50 wt pct SiO2, the replacement of CaO by MgO at a given SiO2 content more significantly decreases the molar volume of the melt. The effect of a 10, 20, and 30 wt pct Al2O3 addition in the CaO-MgO-SiO2 system is predicted in Figures 34(b) through (d). No significant change in iso-contour trend is observed. A general increase of 1 cm3/mol per 10 wt pct Al2O3 addition is predicted in all compositions.
Iso-molar volume contours for the CaO-MnO-SiO2 system are predicted in Figure 36(a). The general trend is similar to that of the CaO-MgO-SiO2 system. However, the molar volume is almost constant with changing SiO2 content above 50 wt pct SiO2. That is, the replacement of CaO by MnO does not significantly change the molar volume. Like the CaO-MgO-SiO2 system with Al2O3 addition, a 10 wt pct addition of Al2O3 increases the molar volume of the CaO-MnO-SiO2 system by approximately 1 cm3/mol, as shown in Figure 36(b).
The molar volume of alkali oxide alumino-silicate systems are predicted in Figure 37. The molar volume of alkali oxides changes significantly between Li2O, Na2O, and K2O, which significantly changes the iso-contour trends in each ternary system. The molar volume of pure Li2O at 1773 K (1500 °C) is predicted to be 19.37 cm3/mol which is significantly lower than that of Al2O3 and SiO2, and therefore, it induces a decreasing molar volume trend with increasing Li2O content in the Li2O-Al2O3-SiO2 system. On the other hand, K2O has a significantly higher molar volume of 53.50 cm3/mol at 1773 K (1500 °C) and therefore increases the molar volume of the K2O-Al2O3-SiO2 system with increasing K2O content. In the case of Na2O-Al2O3-SiO2 system, as Na2O is predicted to have a volume of 35.86 cm3/mole, a similar volume as Al2O3 at 1773 K (1500 °C), the molar volume of the Na2O-Al2O3 system is approximately constant and the addition of SiO2 in the Na2O-Al2O3 system decreases the molar volume.
Discussions
There are a few systems that have very little or no experimental data in the binary systems that are deemed accurate enough to calibrate the binary systems required for extrapolations. For example, the MnO bearing systems are all measured using SBA measurements which may have systematic errors. In addition, few measurements have been made in the binary MnO-SiO2 system. The temperature dependence of this system requires improvement as is apparent during the examination of the CaO-MnO-SiO2 extrapolation. There is still disagreement in the experimental data of the Al2O3-SiO2 system. The measurements of Aksay et al.[49] have been questioned, the accuracy of the results of Anisimov et al.[50] is uncertain, and the single measurement of Courtial and Dingwell[37] did not provide further insight concerning the accuracy of either of these datasets. More accurate experiments are necessary to resolve the disagreement.
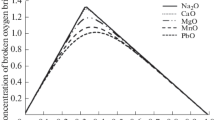
As shown in the part I of this study, a large change in the composition dependency of molar volume \( \left( {\frac{\partial V}{{\partial X_{{{\text{SiO}}_{2} }} }}} \right) \) and thermal expansivity \( (\partial /(\partial X\,{\text{SiO}}_{2} )(\partial V/\partial T)) \) is predicted in many binary silicate melt near the orthosilicate composition (\( X_{{{\text{SiO}}_{2} }} \) = 0.33). This molar volume behavior of binary silicate melt also results in nonlinear behavior of molar volume in ternary and multicomponent melts. For example, the predicted iso-molar volume contours in Figure 34 through Figure 37 clearly show nonlinear behavior of molar volume. In particular, a minimum in molar volume can be predicted in CaO-Al2O3-SiO2-based melts at constant CaO/Al2O3 join. Unfortunately, few experimental data are available for the validation of such predicted behavior at low SiO2 region due to experimental difficulties, (i) high melting temperatures in the systems containing CaO and MgO, and (ii) high vaporization tendency in the systems containing alkali oxides. The molar volume data in the MgO-CaO-SiO2 system across the SiO2/MgO = 2 (see Figure 22) shows this trend.
The present structural model can reproduce the molar volume of binary aluminate melts and ternary and multicomponent alumina containing melts which was poorly predicted by the linear model of Lange and Carmichael.[3] However, it should be noted that the treatment of Al2O3 in the present model would be over simplification of the nature of Al2O3 in liquid slag. Aluminum is known to have various coordinations in oxide melts[45] but was simplified as a network modifier connected to three nearest neighboring oxygens. The molar volumes of ternary alumino-silicate melts show more nonlinear behavior than other systems. Although the present model can reasonably predict the experimental data within experimental scatter, it might require additional ternary parameters. From the structural view point, it may be required to consider the possibility of associated formation like NaAlO2 and Ca0.5AlO2 in liquid slags like the case for structural viscosity model.[51] This should be further investigated with the availability of more accurate experimental data covering wide composition ranges.
Summary
A structural molar volume model was applied to calculate the molar volume of ternary and multicomponent molten slags of the Li2O-Na2O-K2O-MgO-CaO-MnO-PbO-Al2O3-SiO2 system. After the amount of different silicate tetrahedral Q-species in ternary and multicomponent slag are obtained from the quasichemical bond fractions computed from the Modified Quasichemical Model with the FactSage FToxid database, the molar volume of a liquid slag can be calculated using this structural information and unary and binary molar volume parameters. The present model accurately predicted the molar volume of the Li2O-Na2O-K2O-MgO-CaO-MnO-PbO-Al2O3-SiO2 melts at any composition and temperature under 1 atmosphere pressure without any additional ternary parameters. In particular, nonlinear molar volume behavior of liquid slag was well predicted which was impossible from a linear molar volume model.
References
A. D. Pelton and M. Blander, Metallurgical and Materials Transactions B, vol. 17B, pp. 805-815 (1986).
C. W. Bale, E. Bélisle, P. Chartrand, S. A. Decterov, G. Eriksson, K. Hack, I. H. Jung, Y. B. Kang, J. Melançon, A. D. Pelton, C. Robelin, and S. Petersen, “FactSage thermochemical software and databases — recent developments,” Calphad, vol. 33, pp. 295-311 (2009).
R. L. Lange and I. S. E. Carmichael, “Thermodynamic properties of silicate liquids with emphasis on density, thermal expansion and compressibility,” Reviews in Mineralogy and Geochemistry, vol. 24, pp. 25-64 (1990).
J. O. M. Bockris, J. W. Tomlinson, and J. L. White, “The structure of the liquid silicates: Partial molar volumes and expansivities,” Transactions of the Faraday Society, vol. 52, pp. 299-310 (1956).
A. Din: Untersuchungen zum strukturellen Aufbau der Gläser Na2O*xMeO*(6-x)SiO2 aus der Temperaturabhängigkeit der Dichte, der Viskosität und davon abgeleiteter Größen., Technische Universitaet Clausthal (Germany), Ph.D. Thesis (1968).
L. Sasek and A. Lisy, “Structure and properties of silicate melts. I. Effect of the size of ions on glass melt density at high temperatures,” Sbornik Vysoke Skoly Chemicko-Technologicke v Praze, L: Chemie a Technologie Silikatu, vol. L2, pp. 165-215 (1972).
L. Sasek and A. Lisy, “Structure and properties of silicate melts. II. Density of binary alkali-silica glass melts,” Sbornik Vysoke Skoly Chemicko-Technologicke v Praze, L: Chemie a Technologie Silikatu, vol. L2, pp. 217-254 (1972).
N. K. Dertev and E. P. Golovin, “Analytical method of calculation of density of a three component silicate melt,” Trudy Gor’kovskogo Politekhnicheskogo Instituta im. A. A. Zhdanova, vol. 29, pp. 27-30 (1973).
Y. Suginohara, T. Yanagase, and H. Ito, “The effects of oxide additions on the structure-sensitive properties of lead silicate melts,” Transactions of the Japan Institute of Metals, vol. 3, pp. 227-233 (1962).
T. Ejima, M. Hino, and M. Kameda, “Surface tension, density and viscosity of PbO-SiO2-Li2O ternary melts,” Journal of the Japan Institute of Metals, vol. 34, pp. 546-550 (1970).
R. A. Lange and I. S. E. Carmichael, “Densities of Na2O-K2O-MgO-MgO-FeO-Fe2O3-Al3O3-TiO2-SiO2 liquids: New measurements and derived partial molar properties,” Geochimica et Cosmochimica Acta, vol. 51, pp. 2931-2946 (1987).
A. Adachi, K. Ogino, and H. Toritani, “The surface tension of molten silicates,” Technology Reports of the Osaka University, vol. 10, pp. 149-156 (1960).
M. Coenen, “Density of cord glasses at high temperatures,” Glastechnische Berichte, vol. 39, pp. 81-89 (1966).
K. A. Kostanyan and E. K. Karapetyan, “Density of molten glasses,” Armyanskii Khimicheskii Zhurnal, vol. 20, pp. 253-256 (1967).
M. Hino, T. Ejima, and M. Kameda, “Surface tension, density and viscosity of PbO-Na2O-SiO2 ternary melts,” Journal of the Japan Institute of Metals, vol. 32, pp. 809-814 (1968).
E. F. Riebling, “Structure of sodium aluminosilicate melts containing at least 50 mol pct SiO2 at 1500C,” The Journal of Chemical Physics, vol. 44, pp. 2857-2865 (1966).
S. A. Nelson and I. S. E. Carmichael, “Partial molar volumes of oxide components in silicate liquids,” Contributions to Mineralogy and Petrology, vol. 71, pp. 117-124 (1979).
D. J. Stein, J. F. Stebbins, and I. S. E. Carmichael, “Density of molten sodium aluminosilicates,” Journal of the American Ceramic Society, vol. 69, pp. 396-399 (1986).
M. Hino, T. Ejima, and M. Kameda, “Surface tension, density and viscosity of PbO-K2O-SiO2 ternary melts,” Journal of the Japan Institute of Metals, vol. 33, pp. 617-622 (1969).
E. V. Ermolaeva, “Surface tension, viscosity, and density relations of three-component refractory oxide melts,” Ogneupory, vol. 20, pp. 221-228 (1955).
T. Licko and V. Danek, “Densities of melts in the system calcium silicate-calcium magnesium silicate (CaMgSi2O6)-calcium magnesium silicate (Ca2MgSi2O7),” Physics and Chemistry of Glasses, vol. 23, pp. 67-71 (1982).
H. Taniguchi, “Densities of melts in the system CaMgSi2O6-CaAl2Si2O8 at low and high pressures, and their structural significance,” Contributions to Mineralogy and Petrology, vol. 103, pp. 325-334 (1989).
R. Knoche, D. B. Dingwell, and S. L. Webb, “Temperature-dependent thermal expansivities of silicate melts: The system anorthite-diopside,” Geochimica et Cosmochimica Acta, vol. 56, pp. 689-699 (1992).
Y. Ouchi, T. Yoshida, and E. Kato, “Densities of ternary lead-silicate melts,” Journal of the Japan Institute of Metals, vol. 41, pp. 865-874 (1977).
E. F. Riebling, “Structure of magnesium aluminosilicate liquids at 1700 °C,” Canadian Journal of Chemistry, vol. 42, pp. 2811-2821 (1964).
T. M. Kekelidze, S. M. Mikiashvili, T. I. Dzhincharadze, and R. V. Khomeriki, “Density and surface tension of oxide melts of the manganese oxide-calcium oxide-silica and manganese oxide-calcium oxide-silica-alumina systems,” Izvestiya Akademii Nauk Gruzinskoi SSR, Seriya Khimicheskaya, vol. 4, pp. 240-244 (1978).
L. Segers, A. Fontana, and R. Winand, “Poids specifiques et volumes molaires de melanges d’oxydes fondus du systeme CaO-SiO2-MnO,” Electrochimica Acta, vol. 23, pp. 1275-1280 (1978).
J. Lee, L. T. Hoai, J. Choe, and J. H. Park, “Density measurements of CaO-MnO-SiO2 slags,” ISIJ International, vol. 52, pp. 2145-2148 (2012).
L. R. Barrett and A. G. Thomas, “Surface tension and density measurements on molten glasses in the CaO-Al2O3-SiO2 system,” Journal of the Society of Glass Technology, vol. 43, pp. 179-190 (1959).
R. Kammel and H. Winterhager, “Structure and properties of metallurgical slags. V. Density determinations and electrical conductivity measurements on melts in the system lime-alumina-silica,” Zeitschrift fuer Erzbergbau und Metallhuettenwesen, vol. 18, pp. 9-17 (1965).
V. D. Smolyarenko, A. M. Yakushev, and F. P. Edneral, “Density and surface tension of lime-alumina slags with additions of SiO2, MgO, and Na3AlF6,” Izvestiya Vysshikh Uchebnykh Zavedenii, Chernaya Metallurgiya, vol. 8, pp. 55-60 (1965).
P. P. Evseev and A. F. Filippov, “Physical and chemical properties of slags of the CaO-Al2O3-MxAy system. II. Surface tension and density of the slags,” Izvestiya Vysshikh Uchebnykh Zavedenii, Chernaya Metallurgiya, vol. 10, pp. 55-59 (1967).
A. I. Bochorishvili and S. B. Yakobashvili, “Effect of metal oxide on the surface tension of lime-alumina slags,” Svarochnoe Proizvodstvo, vol. 1, pp. 13-15 (1968).
A. N. Sokolov, V. V. Baidov, and L. L. Kunin, “Velocity of ultrasound density, and surface tension as information on the structure of calcium oxide-alumina-silica system,” Fizicheskaya Khimiya i Elektrokhimiya Rasplavlennykh Solei i Shlakov, vol. No. 1, pp. 299-307 (1969).
E. V. Krinochkin, K. T. Kurochkin, and P. V. Umrikhin, “Surface tension and density of oxide melts in the calcium oxide-silica-alumina system,” Fizicheskaya Khimiya Poverkhnostnykh Yavlenii v Rasplavakh, vol. 1, pp. 179-183 (1971).
A.M. Yakushev, V.M. Romashin, and N. Ivanova, Izvestiya Vysshikh Uchebnykh Zavedenii, Chernaya Metallurgiya pp. 47–50 (1985).
P. Courtial and D. B. Dingwell, “Nonlinear composition dependence of molar volume of melts in the CaO-Al2O3-SiO2 system,” Geochimica et Cosmochimica Acta, vol. 59, pp. 3685-3695 (1995).
I. A. Magidson, A. V. Basov, and N. A. Smirnov, “Density and molar volume of CaO-Al2O3-SiO2 (+Na2O) melts,” Izvestiya Vysshikh Uchebnykh Zavedenii, Chernaya Metallurgiya, vol. 5, pp. 8-13 (2007).
L. Muhmood and S. Seetharaman, “Density measurements of low silica CaO-SiO2-Al2O3 slags,” Metallurgical and Materials Transactions B, vol. 41, pp. 833-840 (2010).
M. Zielinski and B. Sikora, “Surface tension of the calcium oxide-alumina system with additions of silica, iron(III) oxide, calcium fluoride, sodium fluoride, and magnesia,” Prace Instytutu Metalurgii Zelaza im. Stanislawa Staszica, vol. 29, pp. 157-165 (1977).
J. Li, T. Zeng, J. Xu, C. Jie, J. Zhang, and K. Chou, in 4th International Symposium on High-Temperature Metallurgical Processing, pp. 95–102 (2013).
A. Din, M. R. Sheikh, and H. W. Hennicke, “Coordination number of magnesium ions in silicate glasses,” Pakistan Journal of Scientific and Industrial Research, vol. 17, pp. 93-95 (1974).
P. Courtial and D. B. Dingwell, “Densities of melts in the CaO-MgO-Al2O3-SiO2 system,” American Mineralogist, vol. 84, pp. 465-476 (1999).
J. W. Tomlinson, M. S. R. Heynes, and J. O. M. Bockris, “The structure of liquid silicates. Part 2.-Molar volumes and expansivities,” Transactions of the Faraday Society, vol. 54, pp. 1822-1833 (1958).
Y. Bottinga, D. Weill, and P. Richet, “Density calculations for silicate liquids. I. Revised method for aluminosilicate compositions,” Geochimica et Cosmochimica Acta, vol. 46, pp. 909-919 (1982).
J.-F. Xu, J.-Y. Zhang, C. Jie, L. Tang, and K.-C. Chou, “Measuring and modeling of density for selected CaO-MgO-Al2O3-SiO2 slag with low silica,” Journal of Iron and Steel Research, International, vol. 19, pp. 26-32 (2012).
H. G. L. K. R. Winterhager, “Untersuchungen über die Dichte und die elektrische Leitfähigkeit von Schmelzen der Systeme CaO-Al2O3-SiO2 und CaO-MgO-Al2O3-SiO2.” Westdeutscher Verlag: Köln u. Opladen, (1966).
T.I. Dzhincharadze, S.M. Mikiashvili, and R.V. Khomeriki, Pererab. Zhelezn. i Margantsev. Rud Zakavraz’ya, Tbilisi, pp. 141–48 (1979).
I. A. Aksay, J. A. Pask, and R. F. Davis, “Densities of SiO2-Al2O3 melts,” Journal of the American Ceramic Society, vol. 62, pp. 332-336 (1979).
Y. S. Anisimov, E. F. Grits, and B. S. Mitin, “Surface tension and density of aluminum oxide-silicon dioxide and aluminum oxide-chromic oxide melts,” Izvestiya Akademii Nauk SSSR, Neorganicheskie Materialy, vol. 13, pp. 1444-1446 (1977).
W.-Y. Kim, Modelling viscosity of molten slags and glasses, École Polytechnique de Montréal, Ph.D. Thesis (2011).
Acknowledgments
Financial support from Hyundai Steel, JFE Steel Corporation, Nippon Steel & Sumitomo Metal, Nucor Steel, Posco, RioTinto, RHI, RIST, Tata Steel Europe, Voestalpine and the Natural Science and Engineering Research Council of Canada (NSERC) is gratefully acknowledged. One of authors (E.T.) also acknowledges the scholarship from the Fonds de Recherche du Quebec—Nature et Technologies (FRQNT) supporting his master study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted July 2, 2015.
Rights and permissions
About this article
Cite this article
Thibodeau, E., Gheribi, A.E. & Jung, IH. A Structural Molar Volume Model for Oxide Melts Part II: Li2O-Na2O-K2O-MgO-CaO-MnO-PbO-Al2O3-SiO2 Melts—Ternary and Multicomponent Systems. Metall Mater Trans B 47, 1165–1186 (2016). https://doi.org/10.1007/s11663-015-0543-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-015-0543-3