Abstract
Summary
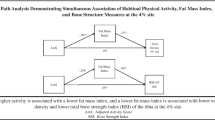
Osteoporosis is common, and physical activity is important in its prevention and treatment. Of the categories of historical physical activity (PA) examined, we found that weight-bearing and very hard physical activity had the strongest relationships with areal bone mineral density (aBMD) throughout growth and into adulthood, while for measures of strength, only grip strength proved to be an independent predictor of aBMD.
Purpose/introduction
To examine relationships between aBMD (total body, lumbar spine, proximal femur, tibial shaft, distal radius) and estimates of historical PA, current strength, and cardiovascular fitness in adult premenopausal women.
Methods
One hundred fifty-two adult premenopausal women (40 ± 9.6 years) undertook aBMD (dual-energy X-ray absorptiometry (DXA)) and completed surveys to estimate historical physical activity representative of three decades (Kriska et al. [1]), while subsets underwent functional tests of isokinetic strength (hamstrings and quadriceps), grip strength (hand dynamometer), and maximum oxygen uptake (MaxV02; cycle ergometer). Historical PA was characterized by demand (metabolic equivalents, PA > 3 METS; PA > 7 METS) and type (weight-bearing; high impact).
Results
Significant positive independent predictors varied by decade and site, with weight-bearing exercise and PA > 3 METS significant for the tibial shaft (10–19 decade) and only PA > 7 METS significant for the final two decades (20–29 and 30–39 years; total body and total hip). A significant negative correlation between high impact activity and tibial shaft aBMD appeared for the final decade. For strength measures, only grip strength was an independent predictor (total body, total hip), while MaxV02 provided a significant independent prediction for the tibial shaft.
Conclusions
Past PA > 7 METS was positively associated with aBMD, and such activity should probably constitute a relatively high proportion of all weekly PA to positively affect aBMD. The findings warrant more detailed investigations in a prospective study, specifically also investigating the potentially negative effects of high impact PA on tibial aBMD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bone mass is mainly determined by the interplay of genetic factors (range 50–90 %) with the magnitude of the contribution somewhat different for different parts of the skeleton [2]. Physical activity (PA) and diet appear to be the “major players” among the modifiable lifestyle factors that contribute to the remaining variance. In terms of PA, those forms which feature impact loading and/or strong muscular contractions appear to have the greatest positive influence [3]. An age/exercise interaction has also been adduced with the “growing years” purported to provide the most exercise-amenable skeleton with peak bone mass (PBM) usually attained at or not long after puberty [4, 5] but potentially deferred until early in the third decade [6]. Given the prevalence and morbidity associated with osteoporosis, particularly in postmenopausal women, it is important to determine whether any bone gains made in response to exercise during the years preceding menopause are preserved, providing a reserve against which to draw. Studies of retired athletes support retention [7–9] though not universally [10]. Retrospective studies of the link between life span PA and bone mass have targeted postmenopausal women [11, 12], adult premenopausal women [13–15], and “older” men [11, 15, 16] subdividing the life span in a variety of ways and using different descriptors of the type and demand of PA including “mild to strenuous” [11], weight-bearing [12–14], metabolic equivalents (METS) [17], and strain rate/ground reaction forces (GRFs) [16, 18]. These generally support a retained positive effect of weight-bearing activity, particularly that engendering high GRF, though one study [19] reported that distance walked to and from school (low impact/low metabolic cost) between 9 and 11 years made a significant independent contribution to femoral bone mass in perimenopausal women. Although adolescence is commonly posited as a “window of opportunity” for maximizing the exercise/bone response [20], Daly and Bass [16] found no relationship between adolescent PA (classified by an osteogenic index (OI)) and bone mass in older men. However, they did find significantly higher indices of femoral shaft bone strength when comparing the highest and lowest tertiles for adolescent weight-bearing impact PA. In the current work, relationships between aBMD and historical PA were explored in adult premenopausal women using data from three decades encompassing the “growing” years, young adulthood, and the period immediately following the attainment of PBM. Historical PA was characterized by MET value as “moderate plus” (all PA > 3 METS) and very hard (all PA > 7 METS); “weight-bearing” and “high impact.” Relationships between aBMD and body composition/anthropometric variables were assessed to inform the selection of covariates for regression analyses examining aBMD/activity relationships [21]. To more common assessment sites (total body, lumbar spine, total hip, distal radius), the tibial shaft (n = 88) was added to explore how this primary cortical weight-bearing site might reflect PA. Finally, subgroups underwent assessments of strength and aerobic capacity to provide data on relationships between “functional” measures and current aBMD. Given the variable results of studies into the influence of lifetime PA on bone mass in later life, our main aim was similar to previous work in attempting to elucidate what types of PA carried out in the past might have lasting positive effects on the skeleton, thereby informing exercise prescription at different ages.
Methods
Participants
Five hundred twenty-one community dwelling women initially responded to advertisements in local newspapers, local community fitness centers, and the Victorian members’ database of Australian Union of Senior Swimmers International (AUSSI). The latter were approached in relation to a study of the influence of swimming on bone mass which indicated no negative effect [22]. Exclusion criteria were menstrual status (fewer than nine normal menses in the previous 12 months), age (<25 years), use of medications known to effect bone mass, and gynaecological/medical conditions (e.g., hysterectomy, ovariectomy, polycystic ovarian disease, thyroid conditions, history of low trauma fracture). After the application of the exclusion criteria, data from 152 participants were included. The reasons for exclusion were failure to complete health and physical activity surveys (n = 209) and a combination of gynecological abnormalities, postmenopausal status, and/or use of inappropriate medications (n = 160). Of the 152 eligible participants, subsets participated in tests to examine relationships between current aBMD and muscular strength (n = 59) and/or maximum oxygen uptake (MaxV02, n = 66). All procedures were approved by the RMIT University Human Ethics Committee, and written informed consent obtained from all participants in the study.
Anthropometry and body composition
Height was measured using a calibrated wall-mounted stadiometer (Tanita, Japan) to the nearest 0.1 cm with the subject lightly clad and wearing no shoes. Weight was determined using calibrated electronic digital scales (SECA, Germany) to the nearest 0.1 kg and BMI calculated (kg/m2). Total body lean mass (kg) and fat mass (kg) were derived from dual-energy X-ray absorptiometry (DXA) total body scans (Lunar DPX-IQ, GE Medical Systems, Madison, WI).
Medical history and menstrual status
Medical history was assessed by completion of a questionnaire that solicited information on general health, medication use, specific illnesses (emphasis on gynecological issues), surgical procedures, and previous fractures including any produced by low trauma. A separate section elicited frequency of menses in the preceding 12 months and the occurrence of changes typical of menopausal onset (e.g., fluid retention, hot flushes, night sweats).
Calcium intake
Daily calcium intake was approximated retrospectively through completion of a 12-month calcium frequency questionnaire (CFQ [1]) containing 22 food items for which participants were asked to specify the number of serves consumed per day or per week or per month with a standard serving size specified for each item. Information from the CFQ was analyzed using “Foodworks 2005” (Xyris Software, Brisbane, Australia) by a dietician.
Historical physical activity
To estimate historical PA, the previously validated Historical Leisure Activity Questionnaire (HLAQ) originally designed by Kriska and colleagues [23] was modified and self-administered. Participants were asked to complete a table listing 44 physical activities (including sport, occupational, domestic, and leisure activities). The list of activities was expanded to take into account the Australian context (e.g., squash, netball, hockey). Participants were asked to indicate if they had ever performed a listed activity and if so, for how many years, at what ages, whether they currently performed the activity, and finally, on average, the number of weeks/days and duration (hours) of participation. They were also asked to add activities whenever they considered them appropriate with a particular emphasis on reporting any strength-based activities like weight training. The data were subsequently compiled under the headings stated previously to reflect type and demand with MET values based upon the work of Ainsworth and colleagues [24]:
-
Weight-bearing: PA performed upright with body weight supported on the feet, e.g., walking, running, tennis, net ball.
-
High impact: PA dominated by jumping and landing with GRFs generally considered >3 BW and rapidly absorbed, e.g., basketball, gymnastics, netball, ballet.
-
Moderate plus: PA >3 METS, e.g., brisk walking >6 kph, jogging, leisurely cycling <10 kph.
-
Very hard: PA >7 MET, e.g., hill climbing, running ≥10 kph, circuit training.
Activities reported by participants were matched to those provided by Ainsworth et al. [24] wherever possible and the best match made in other circumstances.
The moderate plus category (all PA > 3 METS) was considered a gauge of total PA and used to express the proportions (%) contributed by each of the other PA categories. Similarly, the weight-bearing category was used as a baseline for estimating the proportion of high impact PA experienced. The data were subsequently assigned to three decades: 10–19 years (rapid bone formation; transition through puberty), 20–29 years (young adulthood including the attainment of PBM), and 30–39 years (normally characterized by maintenance of bone mass).
DXA measurements
aBMD was measured by DXA (Lunar DPX-IQ, GE Medical systems, Madison, WI) at the total body, total hip, lumbar spine (L2–L4), tibial shaft, and ultradistal radius (left side for all extremities). Short-term operator in vivo precision was assessed by calculating coefficients of variation for duplicate measurements over a 2-week period in 30 adult volunteers (mean age 35 years) in a separate university-approved pilot study (range 1.1–3.5 %). In addition, weight for each participant was compared with the sum of their total bone, lean, and fat masses (mean difference −680 gm, r = 0.99). The left tibial shaft was examined in 88 participants using the manufacturer’s AP spine software with the subject supine and the leg surrounded by rice bags and scanned from ankle to knee. Edge markers were used to isolate the tibia then the “ruler” facility employed to divide the tibia into proximal, middle, and distal thirds with the middle third used to represent the shaft. Different ns for DXA scans appearing in the various tables that follow were the result of the age range of the participants (particularly in the 30–39 decade) and missing scans resulting from scheduling difficulties particularly for tibial scans which required an additional visit to the laboratory.
Functional measurements
Maximum voluntary concentric and eccentric contractions of the quadriceps and hamstring muscle groups were performed on a calibrated Kin Com isokinetic muscle testing device (Isokinetic International, Chattanooga, USA) at 30° per second. Grip strength was measured in both limbs using a calibrated hand dynamometer (Smedley, Tokyo) performed in a standing position. Cardiovascular fitness was assessed by determining MaxVO2 (ml/min and ml/kg/min) and peak power output (PPO, watts) using a calibrated electronically braked cycle ergometer (Lode Excalibur Sport, Netherlands) and automated expired gas analysis apparatus (Medgraphics, USA) using a ramp protocol. Briefly, participants pedaled at an initial load equivalent to their body weight (expressed in watts) for 2 min, and the load increased by 25 or 100 W (based upon individual fitness levels) for a further 2 min until unable to continue (cadence 60–80 cpm).
Statistical analysis
Pearson correlations were calculated to indicate the strength of relationships for aBMD at the various sites while the major analysis involved a series of hierarchical multiple regressions using age, weight, BMI, lean mass, and percent fat mass as covariates (constants) to identify independent predictors of bone mass among the PA variables. The covariates were chosen on the basis of previously published work [25–28] and our own analysis (partial correlations with age as covariate) and were entered as a block in the first step of the regression analysis with one activity variable added at the second step. Separate analyses were applied to historical physical activity, isokinetic strength, grip strength, and aerobic fitness. Alpha was set at p < 0.01 to detect significant correlations in all hierarchical regression analyses of historical physical activity. This alpha level was selected to provide a more stringent criterion in the face of multiple correlation calculations and the concomitant increased likelihood of type 1 errors. For all other analyses, p < 0.05 was accepted for significance. All tests were performed using PASW version 18 for Windows.
Results
Participant characteristics
Table 1 summarizes general physical social characteristics, aBMD, and calcium intake for the participants. Calcium intake was low, averaging 66 % of the RDI. Mean T- and Z-scores were above average for aBMD at all sites with the exception of the distal radius where the averages were negative. All were premenopausal on the basis of nine or more normal menstrual cycles reported in the previous 12 months and the absence of any current menopausal symptoms (data not shown).
Relationships between different BMD sites
Correlations between the different sites have been summarized briefly here where we report the ranges of r for each site, the sites of the weakest and strongest correlations, and p values:
-
Lumbar spine 0.45–0.79 (tibia weakest, total body strongest, all p < 0.01).
-
Total hip 0.51–0.81 (tibia weakest, total body strongest, all p < 0.01).
-
Radius 0.24–0.65 (tibia weakest, total body strongest, 0.05 > p < 0.01).
-
Tibial shaft 0.24–0.59 (radius weakest, total body strongest, 0.05 > p < 0.01).
-
Total body 0.59–0.81 (tibia weakest, total hip strongest, all p < 0.01).
Relationships between measures of body composition/anthropometry and aBMD
Partial correlations (covariate = age) were used to explore relationships between aBMD and height, weight, BMI, total lean mass, total fat mass, and percent fat. For total body aBMD, the strongest significant correlations were provided by total lean mass (r = 0.32; p < 0.001), body weight (r = 0.25; p < 0.01), and BMI (r = 0.20; p < 0.05). None of the variables correlated significantly with aBMD at the ultradistal radius, and only two were significantly related to lumbar spine aBMD (total lean mass and height, r = 0.28 and 0.16; p < 0.001 and 0.05, respectively). Total hip aBMD correlated significantly with each of the independent variables with the exception of height (r = 0.17 to 0.34; 0.05 > p < 0001). Overall, the magnitudes of the significant correlations were comparable for total lean mass, weight, and BMI. The tibial shaft correlated significantly with weight, BMI, and total lean mass but with neither measure of fat or height (range r = 0.22 to 0.32; 0.05 < p < 0.01). Total lean mass, BMI, and %Fat were subsequently used as covariates in analyzing the relationships between PA and aBMD.
Historical physical activity
Tables 2 to 4 present the results of hierarchical multiple regression analyses exploring the relationships between historical PA and the various measures of aBMD. Each decade is represented by one table with dependent variables (aBMD at each site) displayed on the left, categories of lifetime PA and their unadjusted simple correlations with aBMD appear next, followed by the “model summary” which shows the correlation (R) and variance explained (R 2) by the combination of covariates (age, lean mass, BMI, %Fat) and any significant activity variable. The final columns display the additional variance explained by PA (change R 2) and whether the addition is significant (p) followed in such cases, by the standardized beta coefficient and 95 % confidence intervals.
10–19 years
For the earliest decade (Table 2), two significant positive correlations were detected, both at the tibial shaft: one with weight-bearing PA and the other with moderate plus PA (r = 0.27 and 0.28, respectively; both p < 0.01). The prediction of aBMD based upon the combination of covariates and PA (model summary) explained between 14.5 and 18.9 % of the variance with weight-bearing and moderate plus PA both adding significant levels of unique predictive variance (4.5 and 4.7 %, respectively, both p < 0.05; β both 0.22, both p < 0.05). In summary, mean hours of moderate plus PA were approximately 1 h per week with very hard and high impact PA contributing 27 and ~6 %, respectively, while high impact activity constituted ~3 % of weight-bearing activity.
20–29 years
In this decade (Table 3), there were two significant positive correlations for very hard PA (total body and total hip, r = 0.29 and 0.31, respectively; both p < 0.01), while both weight-bearing and moderate plus PA (portraying total PA) were related to aBMD at the tibial shaft (r = 0.29 and 0.27, respectively; both p < 0.01). After “adding” PA to the covariates, only very hard PA added significantly to the prediction of aBMD and only for total body and total hip adding 5.0 and 6.0 %, respectively (β 0.24 and 0.27, respectively, both p < 0.01). In terms of total time spent engaged in PA, moderate plus PA had risen to an average of 1.75 h per week with very hard and high impact activities contributing 12.5 and ~6 %, respectively, while the proportion of weight-bearing activity contributed by high impact PA averaged ~3.0 %.
30–39 years
Table 4 reveals that only very hard PA generated significant positive relationships at any site (total body, radius, and total hip r = 0.25 to 0.37, all p < 0.01). There were no relationships between lumbar spine aBMD and any activity variable, but a significant negative correlation between high impact activity and the tibial shaft was detected (−0.40; p < 0.001). Independent contributions to the covariate model were provided by very hard PA for total body and total hip (8.0 and 7.0 % additions, p < 0.01; β 0.30 and 0.28, respectively, both p < 0.01), while the negative relationship between high impact PA and the tibial shaft added a considerable 28 % to the prediction offered by the covariate model (p < 0.001; β −0.55, p < 0.0001). Mean reported moderate plus PA was very similar to the previous decade (~1.75 h per week) with very hard and high impact activities now contributing 21 and ~3 %, respectively; however, the proportion of weight-bearing PA made up of high impact PA was less than 1.5 %.
Consistency of reporting
Bivariate correlations calculated for each category of PA for each decade (data not shown) suggested that participants had been consistent in their reporting and appeared to have largely maintained their individual PA patterns. For moderate plus PA, r ranged between 0.73 and 0.88, for very hard PA 0.65 to 0.87, for high impact 0.28 to 0.62, and for weight-bearing PA 0.72 to 0.85 (all p < 0.0001 with the exception of r = 0.28, p = 0.006). With one exception, the strength of the relationship was least between the first and last decades while for high impact PA, the weakest correlation was found between the 20–29 and 30–39 decades (r = 0.28).
Bone mass and functional measures
Strength
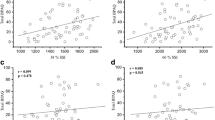
Isokinetic strength testing of the quadriceps and hamstring muscle groups revealed the expected greater mean magnitudes for the eccentric form (quadriceps 131 vs. 114; hamstrings 59 vs. 52 nm, respectively). For the quadriceps, both concentric and eccentric maximum strength correlated significantly with aBMD at the total body, spine, and total hip (range 0.23–0.35; 0.05 > p < 0.001), while the hamstring group produced only two significant positive correlations, both for the concentric mode with total body and total hip (r = 0.24 and 0.25; p < 0.05). However, no isokinetic measurement for either muscle group was an independent predictor of aBMD at any site. Grip strength (Table 5) also produced a number of significant simple correlations at various sites but none with the radius or tibial shaft. Significant correlations were lowest at the lumbar spine (r = 0.28) and highest at the total body (r = 0.41). In contrast with the lower limb strength measurements, grip strength added significant independent predictions to the covariate model for the total body and total hip adding an average (left and right sides) of 15.1 and 10.7 %, respectively (mean β 0.22, 0.36, respectively, both p < 0.01).
Cardiovascular fitness
The results for the cardiovascular tests are summarized in Table 6. Before addition to the covariate model, absolute MaxVO2 (ml/min) provided the strongest and most frequent significant relationships with aBMD, producing positive correlations with all but the distal radius (r = 0.26 to 0.46). The weakest of these was at the lumbar spine, where it was, however, the only variable to produce a significant correlation (r = 0.26). Adjusting for body weight (MaxVO2 ml/kg/min) did not produce stronger correlations than the absolute measure. Indeed, the correlations produced were always weaker than those generated by absolute MaxVO2 at the same site. PPO correlated significantly only with the tibial shaft (r = 0.33). Significant independent predictions of aBMD were only detected at the tibial shaft where both absolute and relative MaxVO2 added 10 and 9 %, respectively, to the covariate model (β 0.57, 0.55, respectively, both p < 0.05).
Discussion
Body composition, anthropometry, and aBMD
The results of the current investigation revealed a number of age-adjusted significant positive correlations between measures of body composition, anthropometry, and aBMD at various sites but none for the distal radius in concert with previous work [29, 30]. That height produced only a single small significant correlation (with the spine) agrees with the work of Lu et al. [25] who found no correlations between height and total hip BMD but a significant positive correlation with spine aBMD albeit about double the size of the one found in the present work (r = 0.31 vs. 0.16). In the current work, the magnitudes of the correlations produced by lean mass, body weight, and BMI were comparable at all sites other than the spine where only lean mass and height produced significant positive correlations. When fat and lean mass were compared, the latter consistently produced higher correlations, in agreement with previous work supporting a greater influence of lean mass on bone [21, 25, 26] though for total body aBMD, the correlation was only about half the magnitude found previously when total body BMC was used as the dependent variable [21, 27]. In connection with this result, it has been suggested that when bone mass is represented by BMC (a variable uncontrolled for body size), the relationship between bone and lean mass is strongest but diminishes with a concomitant increase in the variance explained by fat mass when body size is accounted for in some way (e.g., using aBMD or volumetric BMD [28]), and this might account for the smaller correlations found compared with previous works [21]. Despite the use of aBMD in the current work, the correlations identified for fat mass were smaller at all sites than those recorded for lean mass, weight, and BMI, a finding in agreement with Wang et al. [26] who reported similar findings and magnitudes for these relationships in a large group of younger women (N = 921, 20–25 years of age). With regard to aBMD alone, the different sites were moderately to very strongly correlated, with the exception of the relationship between the tibial shaft and the distal radius (r = 0.24). In fact, in all cases, the weakest relationships were found for the tibial shaft (r = 0.24 to 0.59), a logical finding given that it was the only cortical site measured.
Historical physical activity and bone mass
10–19 years
For the first decade surveyed, only weight-bearing and moderate plus PA were independent predictors of aBMD and only for the tibial shaft (~4.5 % addition to the covariate model by each). Others have reported positive independent effects of weight-bearing activity graded by GRF during adolescence on bone mass in later life, though at different sites [16, 17]. In terms of the moderate plus category, Rideout et al. [12] also reported that a measure of total PA during a similar period (12–18 years) was a significant independent predictor of aBMD (total hip) in postmenopausal women. In contrast, Daly and Bass [16] found that an estimate of total PA for the periods 13 to 50+ years (“lifetime”) and 19 to 50 years (“mid-adulthood”) was not related to aBMD at any site. Similarly, Van Langendonck et al. [18] reported that only an “impact score” (based upon estimates of GRFs) achieved during adulthood provided a significant independent predictor of total body and lumbar spine BMD in men after a follow-up of 27 years. Interestingly, very hard PA constituted a reasonably large proportion of moderate plus PA in this decade (27 %) which may account for its positive association with tibial shaft aBMD. There would appear to be few studies where the tibia has been investigated separately, and therefore, few data are available for comparison; however, our results agree to some extent with Sayers et al. [31] who studied the middle tibia by pQCT in a large cross-sectional study of bone mass and habitual physical activity in boys and girls (mean age 15.5 years). They found that only vigorous activity (6 METS) was positively related to cortical bone mineral content and geometry. The failure of high impact exercise to leave an “impression” in the current study might simply be due to too little of this form of activity being performed. High impact PA expressed as a percentage of moderate plus and weight-bearing exercise provided figures of 7 and 3 %, respectively, for this decade. Hence, the strain and strain rates evoked by this volume of exercise may have been insufficient to stimulate modeling.
20–29 years
Examination of the results from the second decade highlights the potential importance of strenuous physical activity to aBMD with very hard PA as the only independent predictor (total hip; total body). For this period, the proportion of moderate plus activity contributed by very hard activity was ~13 %, about half of its contribution in the 10–19 decade. Hence, though moderate plus activity had increased (from 1.1 to ~1.75 h per week), it appears that only the more demanding components were associated with an osteogenic affect since moderate plus activity by itself produced no significant correlations with aBMD. This time, there were no independent predictors for the tibial shaft, perhaps indicative of a greater responsiveness at this site during the growing years [20]. High impact activities had dropped to around 6 and 3.4 % of moderate plus and weight-bearing PA, respectively, and were therefore likely too few to produce any detectable relationship with aBMD.
30–39 years
Very hard activity continued to dominate in the final decade, continuing to produce highly significant independent contributions at the total hip (7 % addition) and total body (8 % addition), reinforcing the notion that some degree of arduous physical activity is needed to produce and/or maintain a positive adaptation in bone. In this case, that proportion was around 20 % (very hard activity as a proportion of moderate plus PA). The relatively strong negative correlation between high impact PA and tibial shaft aBMD is counterintuitive given that this form of exercise constituted only about 3 % of the weekly hours reported for moderate plus activity and even less of the of weight-bearing activity (1.3 %). Perhaps the tibial shaft is less able to mount a positive response (e.g., increased intracortical resorption coupled with perisosteal deposition) when presented with high impact PA on an inconsistent basis after the age of PBM. Lending some support to this notion, Shedd et al. [32] found that a peak strain score (based upon GRFs) calculated for PA over 12 months in postmenopausal women was negatively related to indices of bone strength (polar moment of inertia; strength-strain index) though the measurement site was the distal tibia.
Some degree of consistency in the relationship between historical PA and aBMD was found for both bone site and type of PA with very hard PA adding significant contributions to the prediction of aBMD at both total body and total hip in each of the final two decades. The tibial shaft featured in both the first and last decades but with different types of PA generating positive independent contributions to the covariate model for weight-bearing and moderate plus PA during the first decade and a large negative contribution in the final decade with high impact PA.
As previously alluded to (Statistical analysis section), calculating a large number of correlations increases the risk of detecting significant correlations that are not “real.” Adjusting the alpha level using the Bonferroni approach would have provided a much more stringent “test,” but we considered the resulting alpha level (p < 0.0008) overly conservative.
Strength and bone mass
Isokinetic
In the current work, concentric quadriceps torque failed to provide any independent predictions of aBMD at any site, findings congruent with some previous work using both younger and older participants [30, 33] and divergent with others [34]. In one study employing isometric dynamometry in older men and women [35], initial significant unadjusted correlations of 0.395 and 0.353 for knee extension and flexion, respectively, “disappeared” after normalization for height and weight, prompting the suggestion that in older adults, body size is responsible for the relationship between aBMD of the proximal femur and maximum isometric strength. Interestingly, the tibia has been the target of a number of studies into bone mass/strength relationships. In early work, Madsen et al. [36] found strong positive correlations between quadricep isokinetic strength and proximal tibial bone mass in a group of 66 women (21–78 years of age) and concluded that quadricep strength was a better predictor of tibial BMD than age, height, or weight. Similarly, Vico et al. [37] found that isokinetic thigh muscle torques (flexion and extension) were the best predictors of tibial cortical bone (QCT) in 55 postmenopausal women. In contrast, Sööt et al. [38], employing both isokinetic and isometric measures of quadricep strength in 129 women (17–40 years), found that BMI and lean body mass were the strongest predictors of “leg” bone mass with strength the best predictor only for those classed as overweight. It is difficult to draw conclusions from the foregoing discussion given the varying populations and measurement sites; however, on the basis of the current and previous research, it is reasonable to suggest that there is no advantage provided by testing both maximum eccentric and concentric quadricep isokinetic strength to predict proximal femoral bone mass despite the fact that maximum eccentric strength exceeds maximum concentric strength. Maximum isokinetic strength of the hamstring muscle group did not appear to provide a useful indicator of femoral or tibial bone mass. It should be said that none of the correlations detected in the current work were more than moderate at best and even the covariate model explained a maximum of 22.8 % of the variance for the total hip. However, in the current work, concentric isokinetic quadricep strength provided correlations that were at least as strong at the total hip as those provided by each of the individual covariates and being a functional measurement, provides information pertinent to fall prevention in addition to any relationships with bone mass.
Grip strength
In the current work, grip strength was an independent predictor of aBMD at both total body and total hip adding about 15 and 11 %, respectively, to the variance explained by the covariates. Moderate significant positive correlations between grip strength and both mid-radial and lumbar spine aBMD have been reported in older subjects (age range 61–84 years) in early work [39] with the magnitude of the correlation at the spine comparable to that found in the current study (r = 0.37 vs. 0.32 current). Discordant findings for the forearm itself are logically explained by the use of different measurement sites (mid-radius [39], ultradistal radius current). Kritz-Silverstein and Barrett-Connor [40] concluded that grip strength was a marker of “overall” bone fragility in a large group of postmenopausal women (n = 649), a proposition extended to children by Chan et al. [41] who studied over 300, 11–12-year-old boys and girls finding grip strength to be an independent predictor for spine and total body bone mass in boys and for spine and hip in girls after controlling for weight, height, pubertal development, weight-bearing activity, and calcium intake. Hence, there is support for grip strength as a general indicator of bone “strength” with the present work indicating that grip strength in combination with age, BMI, lean mass, and %Fat explained ~25 % of the variance in aBMD at the total hip and the total body. The fact that each of the body composition variables produced correlations of similar magnitude suggests that using a simple measure like BMI in combination with grip strength would be useful in screening for low bone mass at these two sites. Somewhat paradoxically, grip strength did not reflect aBMD in the distal radius itself, possibly due to the small number and size of muscles directly inserting here (two) and a general lack of impact loading. In contrast, Di Monaco et al. [42] found grip strength to be the strongest independent predictor of distal radial BMD in a group of postmenopausal women after controlling for a large number of covariates (age, weight, height, BMI, calcium intake, years since menopause) a finding at odds with the present results and that of several others [14, 33, 43] with none finding a significant relationship between grip strength and aBMD or “bone strength” at this site whether measured isometrically or isokinetically.
Cardiovascular
Early work suggested a link between aerobic capacity and aBMD in both premenopausal and postmenopausal women with significant independent relationships found between aBMD of subregions of the proximal femur and both predicted (submaximal effort) MaxVO2 [44] and MaxVO2 (ml/min) based upon maximal effort [37]. Indeed, absolute MaxVO2 (ml/min) was the best predictor of aBMD in postmenopausal women explaining between 10.8 and 18.3 % of the variance at proximal femoral sites [37], while for lumbar spine, both positive [44] and negative [39] findings have been reported in this population. In the current work, both measures of MaxVO2 were significantly positively correlated with aBMD at the total body and tibial shaft, while the absolute measure also correlated significantly with aBMD at the lumbar spine and total hip. However, they were independent predictors only for the tibial shaft, each adding about 10 % to the covariate model. The tibial shaft was also the only site where PPO produced a significant correlation though it made no independent contribution. These results should focus interest on the tibial shaft as a site for further investigation considering that it appeared to “reflect” some aspects of both historical PA and current aerobic status. The fact that unadjusted MaxVO2 was a better predictor than the weight-adjusted value probably reflects the positive relationship between body weight and bone mass in general. Improving aerobic capacity may not necessarily lead to improvement in lower extremity bone mass even when combined with strength training in older adults [45] which suggests a need to use higher impact style exercises when seeking to improve aerobic capacity and bone mass simultaneously (using weighted vests during walking/running) particularly in light of work finding that endurance running may be associated with low bone mass despite the associated multiple moderate impacts [46].
Summary
Before summarizing the findings, two important limitations are acknowledged: First the ability to accurately recall physical activity patterns over many years is an obvious limitation; however, the tool applied in the current work was based upon the HLAQ designed by Kriska et al. [1] which has proven reliability; second, aBMD as measured in the current work, does not “capture” the geometric features of bone that contribute to bone strength so that aBMD is an incomplete surrogate for the latter [e.g., [47]]. With these factors in mind, we found that the relationship between historical PA and aBMD varied by type and site across three decades with only the tibial shaft “reflecting” positive effects of weight-bearing PA and PA > 3 METS (moderate plus) during adolescence (10–19 years) then “switching” to very hard PA at the total hip and total body over the last two decades (20–29 and 30–39 years). In general, PA > 7 METS was positively associated with aBMD, and therefore, we speculate that total PA should have a relatively high contribution from strenuous PA (e.g., netball, squash, running ~10 km/h, hiking with a pack). Similarly, it would appear that weight-bearing PA requires a relatively higher contribution from high impact PA (≥4BW) than found in the current work though it might have a negative effect on the tibia after the attainment of PBM perhaps due to a reduced ability to adapt to this form of loading. The fact that tibial shaft aBMD was related to aspects of both historical PA and current aerobic status (MaxVO2) suggests a need for further investigation into the tibia as a potential “barometer” of the bone/PA relationship. Clearly, all of these suggestions should be tested in prospective studies.
With regard to relationships between functional measures and current aBMD, we found no value accrued to the prediction of bone mass by measuring maximum isokinetic strength in the quadriceps and hamstrings; hence, the utility of making such measures is related more to their association with both fall prevention and femoral bone mass combined. In contrast, we found that grip strength provided significant independent additions to the predictions of aBMD at the total body and total hip thereby supporting the notion that this simple measure is a useful indicator of aBMD/bone strength particularly when combined with a body composition indicator such as BMI.
Abbreviations
- aBMD:
-
Areal bone mineral density
- BMC:
-
Bone mineral content
- BMI:
-
Body mass index
- CFQ:
-
Calcium frequency questionnaire
- DXA:
-
Dual-energy X-ray absorptiometry
- GRF:
-
Ground reaction forces
- HLAQ:
-
Historical Leisure Activity Questionnaire
- MaxV02 :
-
Maximum oxygen uptake
- METS:
-
Metabolic equivalents
- OI:
-
Osteogenic index
References
Nowson CS, Green AJ, Wark JD (1995) Limitations of dietary calcium assessment in female twins of different ages. In Burckhardt P and Heaney RP(eds) Challenges of Modern Medicine 7:97–104
Lei SF, Chen Y, Xiong DH, Li LM, Deng HW (2006) Ethnic difference in osteoporosis-related phenotypes and its potential underlying genetic determination. J Musculoskelet Neuronal Interac 6(1):36–46
Nikander R, Kannus P, Dastidar P, Hannula M, Harrison L, Cervinka T, Narra NG, Akltour R, Arola T, Eskola H, Soimakallio S, Heinonen A, Hyttinen J, Sievanen H (2009) Targeted exercises against hip fragility. Osteoporos Int 20(8):1321–1328
Kannus P, Haapasalo H, Sankelo M, Sievänen H, Pasanen M, Heinonen A, Oja P, Vuori I (1995) Effect of the starting age of physical activity on bone mass in the dominant arm of tennis and squash players. Ann Intern Med 123:27–31
Sabatier JP, Guaydier-Souquières G, Laroche D, Benmalek A, Fournier L, Guillon-Metz F, Delavenne J, Ay D (1996) Bone mineral acquisition during adolescence and early adulthood: a study in 574 healthy females 10–24 years of age. Osteoporos Int 6(2):141–148
Recker RR, Davies KM, Hinders SM, Heaney RP, Stegman MR, Kimmel DB (1992) Bone gain in young adult women. Jama 268(17):2403–2408
Nordstrom A, Olsson T, Nordstrom P (2005) Bone gained from physical activity and lost through detraining: a longitudinal study in young males. Osteoporos Int 16(7):835–841
Welch JM, Rosen CJ (2005) Older women track and field athletes have enhanced calcaneal stiffness. Osteoporos Int 16(8):871–878
Pollock NK, Laing EM, Modlesky CM, O'Connor PJ, Lewis RD (2006) Former college artistic gymnasts maintain higher BMD: a nine-year follow-up. Osteoporos Int 17(11):1691–1697
Karlsson MK, Linden C, Karlsson C, Johnell O, Obrant K, Seeman E (2002) Exercise during growth and bone mineral density and fractures in old age. Lancet 355:469–470
Greendale GA, Barrett-Connor E, Edelstein S, Ingles S, Haile R (1995) Lifetime leisure exercise and osteoporosis. The Rancho Bernardo study. Am J Epidemiol 141(10):951–959
Rideout CA, McKay HA, Barr SI (2006) Self-reported lifetime physical activity and areal bone mineral density in healthy postmenopausal women: the importance of teenage activity. Calcif Tissue Int 79(4):214–222
Ulrich CM, Georgiou CC, Gillis DE, Snow CM (1999) Lifetime physical activity is associated with bone mineral density in premenopausal women. J Womens Health 8(3):365–375
Ishikawa K, Ohta T, Tahara Y, Suzuki T (2000) The relationship between spinal and appendicular bone mass modified by physical, historical, and lifestyle factors. J Bone Miner Metab 18(3):170–175
Brahm H, Mallmin H, Michaelsson K, Strom H, Ljunghall S (1998) Relationships between bone mass measurements and lifetime physical activity in a Swedish population. Calcif Tissue Int 62(5):400–412
Daly RM, Bass SL (2005) Lifetime sport and leisure activity participation is associated with greater bone size, quality and strength in older men. Osteoporos Int 17(8):1258–67
van Mechelen W, Twisk JW, Kemper HC, Snel J, Post GB (1999) Longitudinal relationships between lifestyle and cardiovascular and bone health status indicators in males and females between 13 and 27 years of age: a review of findings from the Amsterdam growth and health longitudinal study. Public Health Nutr 2(3A):419–427
Van Langendonck L, Lefevre J, Claessens AL, Thomis M, Philippaerts R, Delvaux K, Lysens R, Renson R, Vanreusel B, Vanden Eynde B, Dequeker J, Beunen G (2003) Influence of participation in high-impact sports during adolescence and adulthood on bone mineral density in middle-aged men: a 27-year follow-up study. Am J Epidemiol 158(6):525–33
Rikkonen T, Tuppurainen M, Kröger H, Jurvelin J, Honkanen R (2006) Distance of walking in childhood and femoral bone density in perimenopausal women. Eur J Appl Physiol 5:509–515
MacKelvie KJ, Khan KM, McKay HA (2002) Is there a critical period for bone response to weight-bearing exercise in children and adolescents? A systematic review. Br J Sports Med 36(4):250–257
Kohrt WM, Barry DW, Schwartz RS (2009) Muscle forces or gravity: what predominates mechanical loading on bone? Med Sci Sports Exerc 41(11):2050–2055
Greenway KG, Walkley JW, Rich PA (2012) Does long-term swimming participation have a deleterious effect on the adult female skeleton? Eur J Appl Physiol 112(9):3217–25
Kriska AM, Sandler RB, Cauley JA, LaPorte RE, Hom DL, Pambianco G (1988) The assessment of historical physical activity and its relation to adult bone parameters. Am J Epidemiol 127(5):1053–1063
Ainsworth BE, Haskell WL, Whitt MC, Irwin ML, Swartz AM, Strath SJ, Leon AS (2000) Compendium of physical activities: an update of activity codes and MET intensities. Med Sci Sports Exerc 32(9 Suppl):S498–504
Lu LJW, Nayeem F, Anderson KE, Grady JJ, Nagamani M (2009) Lean body mass, not estrogen or progesterone, predicts peak bone mineral density in premenopausal women. J Nutr 139(2):250–256
Wang MC, Bachrach LK, Van Loan M, Hudes M, Flegal KM, Crawford PB (2005) The relative contributions of lean tissue mass and fat mass to bone density in young women. Bone 37(4):474–481
Makovey J, Naganathan V, Sambrook P (2005) Gender differences in relationships between body composition components, their distribution and bone mineral density: a cross-sectional opposite sex twin study. Osteoporos Int 16(12):1495–1505
Reid IR (2002) Relationships among body mass, its components, and bone. Bone 31(5):547–555
Travison TG, Araujo AB, Esche GR, Beck J, McKinlay JB (2008) Lean mass and not fat mass is associated with male proximal femur strength. J Bone Miner Res 23(2):189–198
Miller LE, Nickols-Richardson SM, Wootten DF, Ramp WK, Herbert WG (2004) Relationships among bone mineral density, body composition, and isokinetic strength in young women. Calcif Tissue Int 74(3):229–235
Sayers A, Mattocks C, Deere K, Ness A, Riddoch C, Tobias JH (2011) Habitual levels of vigorous, but not moderate or light, physical activity is positively related to cortical bone mass in adolescents. J Clin Endocrinol Metab 96(5): Epub 2011 Feb doi:E793-802
Shedd KM, Hanson KB, Alekel DL, Schiferl DJ, Hanson LN, Van Loan MD (2007) Quantifying leisure physical activity and its relation to bone density and strength. Med Sci Sports Exerc 39(12):2189–2198
Bayramoğlu M, Sözaym S, Karataş M, Kilinç S (2005) Relationships between muscle strength and bone mineral density of three body regions in sedentary postmenopausal women. Rheumatol Int 25(7):513–517
Ribom E, Ljunggren Ö, Piehl-aulin K, Ljunghall S, Bratteby L-E, Samuelson G, Mallmin H (2004) Muscle strength correlates with total body bone mineral density in young women but not in men. Scand J Med Sci Sports 14(1):24–29
Owings TM, Pavol MJ, Grabiner MD (2002) Lower extremity muscle strength does not independently predict proximal femur bone mineral density in healthy older adults. Bone 30(3):515–520
Madsen OR, Schaadt O, Bliddal H, Egsmose C, Sylvest J (1993) Relationship between quadriceps strength and bone mineral density of the proximal tibia and distal forearm in women. J Bone Miner Res 8(12):1439–1444
Vico L, Pouget JF, Calmels P, Chatard JC, Rehailia M, Minaire P, Alexandre C (1995) The relations between physical ability and bone mass in women aged over 65 years. J Bone Miner Res 10(3):374–383
Sööt T, Jürimäe T, Jürimäe J, Gapeyeva H, Pääsuke M (2005) Relationship between leg bone mineral values and muscle strength in women with different physical activity. J Bone Miner Metab 23(5):401–406
Bevier WC, Wiswell RA, Pyka G, Kozak KC, Newhall KM, Marcus R (1989) Relationship of body composition, muscle strength, and aerobic capacity to bone mineral density in older men and women. J Bone Miner Res 4(3):421–432
Kritz-Silverstein D, Barrett-Connor E (1994) Grip strength and bone mineral density in older women. J Bone Miner Res 9(1):45–51
Chan DC, Lee WT, Lo DH, Leung JC, Kwok AW, Leung PC (2008) Relationship between grip strength and bone mineral density in healthy Hong Kong adolescents. Osteoporos Int 19(10):1485–1495
Di Monaco M, Di Monaco R, Manca M, Cavanna A (2000) Handgrip strength is an independent predictor of distal radius bone mineral density in postmenopausal women. Clin Rheumatol 19(6):473–476
Frank AW, Lorbergs AL, Chilibeck PD, Farthin JP, Kontulainen SA (2010) Muscle cross sectional area and grip torque contraction types are similarly related to pQCT derived bone strength indices in the radii of older healthy adults. J Musculoskelet Neuronal Interact 10(2):136–141
Pocock NA, Eisman JA, Yeates MG, Sambrook PN, Eberl S (1986) Physical fitness is a major determinant of femoral neck and lumbar spine bone mineral density. J Clin Invest 78(3):618–621
Stewart KJ, Bacher AC, Hees PS, Tayback M, Ouyang P, Jan de Beur S (2005) Exercise effects on bone mineral density relationships to changes in fitness and fatness. Am J Prev Med 28(5):453–460
Hind K, Truscott JG, Evans JA (2006) Low lumbar spine bone mineral density in both male and female endurance runners. Bone 39(4):880–885
Haapasalo H, Kontulainen S, Sievänen H, Kannus P (2000) Exercise-induced bone gain is due to enlargement in bone size without a change in volumetric bone density: a peripheral quantitative computed tomography study of the upper arms of male tennis players. Bone 27(3):351–357
Acknowledgments
The authors wish to express their appreciation to the all of the participants and to the Shepherd Foundation of Victoria for funding that supported the project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Kathleen Greenway, Jeff Walkley, and Peter Rich declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
All procedures were approved by the RMIT University Human Ethics Committee, and written informed consent obtained from all participants in the study.
Rights and permissions
About this article
Cite this article
Greenway, K.G., Walkley, J.W. & Rich, P.A. Relationships between self-reported lifetime physical activity, estimates of current physical fitness, and aBMD in adult premenopausal women. Arch Osteoporos 10, 34 (2015). https://doi.org/10.1007/s11657-015-0239-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11657-015-0239-y