Abstract
Simple and efficient in vitro plant production systems were established for Ledebouria ovatifolia (Bak.) Jess. Adventitious shoots were best produced from leaf explants with Murashige and Skoog medium containing 5 μM thidiazuron and 2 μM naphthaleneacetic acid, and from organogenic callus with Murashige and Skoog medium containing 2 μM indole-3-acetic acid, 5 μM thidiazuron, and 30 μM glutamine. Indole-3-butyric acid and 25 μM phloroglucinol were effective for rooting of shoots. Embryogenic callus was induced on semi-solid medium containing growth regulators. The highest numbers of somatic embryos, 43.2–35.6 (globular to cotyledonary stages, respectively) from friable, embryogenic callus were obtained on liquid medium with 15 g L−1 sucrose, 10 μM glutamine, 0.1 μM picloram, and 0.2 μM thidiazuron. Seventy-three percent of somatic embryos germinated on semi-solid medium with 15 g L−1 sucrose, 0.3 μM gibberellic acid, 0.3 μM phloroglucinol, and Murashige and Skoog macronutrients. All plantlets were successfully acclimatized in the greenhouse. Production of clonal plants was confirmed by features of embryoids using light and transmission electron microscopy, which detected cytoplasmic components including many mitochondria, lipid bodies together with starch grains, chloroplasts, Golgi apparatuses, vacuoles, and nuclei. The reported developmental system reinforced the importance of nutritional and hormonal effects as well as the effect of phloroglucinol on in vitro plant production. Histological and ultrastructural studies demonstrated the bipolar structure, and viability of somatic embryoids. The micropropagation and somatic embryogenesis protocols reported here provide systems for germplasm conservation and large-scale clonal propagation, and for pharmacological, and genetic transformation studies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The monocotyledonous genus Ledebouria (Asparagaceae) consists of approximately 50 species, most of which contain bioactive compounds of medicinal value (Govaerts 2016). However, many species are threatened and require conservation efforts (SANBI 2015). Ledebouria ovatifolia (Bak.) Jess. is an African plant popularly known as Icubudwana in the native Zulu dialect. Its bulbs are used in traditional medicine to treat backache, influenza, and gastroenteritis (Hutchings et al. 1996; Sparg et al. 2002). It is also used for ethnoveterinary purposes (Gerstner 1938; Waller et al. 2013). The bulb produces bioactive compounds such as 4,4′-dihydroxy-2,6′-dimethoxychalcone; 5,7-dihydroxy-3-(4′-hydroxybenzyl)-4-chromanone; homoisoflavanones; ovatifolionone; and xanthone (Pohl et al. 2001; Waller et al. 2013). Ethanol and dichloromethane extracts of bulbs and xanthone have antibacterial and anti-inflammatory activities (Sparg et al. 2002; Waller et al. 2013).
L. ovatifolia is included in the Red Data List of South African Plants (SANBI 2015) as a result of overexploitation, and its conservation status requires the development of an efficient mass propagation system. Methods for micropropagation via organogenesis and somatic embryogenesis would be valuable for conservation, mass clonal propagation, production of artificial seeds, cryopreservation, biomass production, and for producing potential resources for new drug development and clinical research (Ramachandra and Ravishankar 2002; Sharma and Dubey 2011; Baskaran et al. 2013; Kumari et al. 2015). In vitro techniques are promising tools for research into the basic physiology and biochemistry of plant cells, and for studies of somatic mutation, protoplasts, and somatic hybridization (Orczyk et al. 2003; Georgiev et al. 2011). However, morphological abnormalities such as embryo fusion, lack of apical meristems, or loss of bipolarity have occurred during somatic embryogenesis (Benelli et al. 2010).
Although micropropagation protocols have been developed for other Ledebouria species (Shushu et al. 2009; Wetschnig et al. 2013), a micropropagation protocol for L. ovatifolia has yet to be described. A morpho-histological and cytological study during embryogenesis is therefore required to confirm the embryogenic capacity of L. ovatifolia and to describe the developmental stages of somatic embryos (SEs). The present investigation aimed to develop a simple, efficient, and rapid in vitro plant regeneration system via direct and indirect organogenesis and somatic embryogenesis for conservation, large-scale clonal commercial propagation, and genetic improvement of L. ovatifolia from leaf explants.
Materials and Methods
Plant material, organogenesis, and somatic embryogenesis
Young leaves (from 3-mo-old greenhouse-grown bulbs) of L. ovatifolia were collected from the Botanical Garden, University of KwaZulu-Natal, Pietermaritzburg, South Africa. Leaves were washed with Tween 20 for 1 min and then decontaminated with 0.1% aqueous HgCl2 for 10 min. Leaves were then rinsed five times with sterile distilled water. To induce adventitious shoot regeneration by direct organogenesis, leaf explants (approximately 15 × 10 mm) were excised and cultured for 12 wk on MS (Murashige and Skoog 1962) medium containing different concentrations and combinations of plant growth regulators (PGRs), including benzyladenine (BA), meta-topolin riboside [mTR; 6-(−3-hydroxybenzylamino)-9-β-D-ribofuranosylpurine)], thidiazuron (TDZ), naphthaleneacetic acid (NAA), indole-3-acetic acid (IAA), and phloroglucinol (PG) (Table 1). To induce indirect organogenesis, leaf explants were placed on callus induction (CI) medium for 6 wk. CI consisted of MS with various concentrations of sucrose, and of 2,4-dichlorophenoxyacetic acid (2,4-D) or picloram. To induce shoot regeneration, calluses formed on CI were then placed on media containing various concentrations and combinations of 2,4-D, picloram, NAA, IAA, BA, mTR, zeatin, TDZ, glutamine (Gln), and PG (Table 2). Shoots >2 cm in length were transferred to rooting medium containing half-strength MS and various concentrations and combinations of NAA, indole-3-butyric acid (IBA), PG, and gibberellic acid (GA3) for 6 wk (Table 3).
MS media solidified with 8 g L−1 agar (MSS) containing either 30, 40, 50, or 70 g L−1 sucrose and 10 μM picloram were tested for production of embryogenic callus (EC) from leaf explants. For maturation of embryos, EC with globular stage embryos was transferred onto MSS supplemented with 2 μM picloram and 0.5 μM BA, mTR, TDZ, GA3, or PG for 4 wk (Table 4). Matured embryos (globular to cotyledonary-initiation stages) were transferred to MSS or MSS medium containing two-fold MS macronutrients (MSN) for 8 wk to induce germination of embryos.
To establish embryogenic cell suspension cultures, 6-wk-old, friable, EC was transferred from MSS with 30 g L−1 sucrose and 10 μM picloram to 100-mL Erlenmeyer flasks containing 20 mL of ½-MSL or MSL supplemented with various PGRs (Table 5), and cultured for 4 wk. These procedures were as described previously (Baskaran and Van Staden 2012), except that the settled cell volume (SCV) was 1000 μL. Embryos at different developmental stages were then transferred onto embryo germination (EG) medium (½- MSS or MSS supplemented with various PGRs; Table 5). The embryo germination percentage was calculated after 8 wk as the number of germinated SE/total number of SE × 100. In all experiments, medium without PGRs was used as a control.
In all the experiments, analytical grade chemicals of macro and micro nutrients of MS and sucrose were obtained from Merck (Modderfontein, South Africa); mercuric chloride, vitamins, and plant growth regulators were obtained from Sigma-Aldrich® (St. Louis, Missouri, MO); agar was obtained from Oxoid (Hampshire, England); and phloroglucinol was obtained from UniLab (Muldersdrift, South Africa). All media were adjusted to pH 5.8 with 0.1 N NaOH and/or 0.1 N HCl before adding agar (8 g L−1) and autoclaved at 121°C for 20 min. Cultures were maintained at 25 ± 2°C with a light intensity of 40 μmol m−2 s−1 (provided by cool white fluorescent light; OSRAM L 58 W/740, Midrand, South Africa) and a 16-h photoperiod. Acclimatization of regenerated plantlets (approximately 50–90 mm) from organogenesis and somatic embryogenesis was conducted in a greenhouse as described previously (Baskaran and Van Staden 2012).
Microscopic studies
All major SE developmental stages (globular, pear-shaped, torpedo-shaped, and cotyledonary) were photographed using a Leica MZ 16 Stereo Microscope (Leica: OFC450 C type, Digital Camera, Mannheim, Germany). All chemicals used were of analytical grade (EMS, Hatfield Pennsylvania; Gurr, Poole, England and Agar Scientific Ltd., Essex, UK). The SEs were fixed in 3% (v/v) glutaraldehyde for 12 h and washed in 0.05 M sodium cacodylate buffer for 30 min. The SEs were then fixed in 2% osmium tetroxide for 2 h, rinsed twice in 0.05 M sodium cacodylate buffer for 30 min, and dehydrated through a graded ethanol series (10, 30, and 50% for 10 min each, 70% ethanol overnight, 90% ethanol for 15 min, and then twice with 100% ethanol for 15 min each, and rinsed twice with 100% propylene oxide for 15 min each). The SEs were then infiltrated with 50:50 Spurr’s resin:propylene oxide for 1 h, followed twice with 100% Spurr’s resin for 1 h each. Samples were allowed to polymerize in 100% Spurr’s resin at 70°C for 24 h by being placed in flat silicone embedding molds. The SEs were cut into 1-μm-thick sections with a glass knife in a ultra microtome (Leica EM UC7, Mannheim, Germany), mounted onto glass slides with a drop of water and fixed over a hot plate (approximately 60°C for 10 min). For general histology, toluidine blue stain (2% v/v) was used. Longitudinal sections (LS) were visualized and photographed using a compound light microscope (Olympus AX70, Tokyo, Japan, under bright field mode). For ultrastructural studies of somatic embryos via transmission electron microscopy (TEM), 100-nm-thick sections were cut and placed on 200-mesh copper grids, contrasted with 2% uranyl acetate for 5 min, and washed in distilled water. The sections were examined with a JEM-1400 (JEOL Ltd, Tokyo, Japan) TEM operating at 120 kV.
Statistical analysis
All experiments were conducted three times with 25 replicates for each treatment for callus induction, shoot regeneration, and rooting, and with five replicates each for SE formation by suspension culture and germination experiments. Data were analyzed using a one-way analysis of variance (ANOVA) and are presented as mean ± standard error. Treatment means were separated using Duncan’s multiple range tests at the 5% probability level and analyzed using IBM SPSS for Windows version 23 (SPSS Inc., Chicago, IL).
Results and Discussion
Plant regeneration in vitro
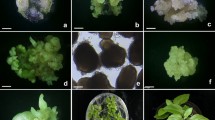
Various concentrations and combinations of PGRs and PG were investigated for their effect on in vitro adventitious shoot regeneration from leaf explants (direct organogenesis). Shoot buds were initiated at the cut edge of explants from all treatments, except the control, after 2 wk of culture (Fig. 1a ). The morphogenic response (shoot and root regeneration) varied after 5 wk of culture (Fig. 1b ). The shoot multiplication rate increased with the addition of low concentrations of BA or TDZ after 12 wk of culture (Table 1; Fig. 1c ), but did not differ significantly between the three treatments (Table 1). High concentrations of PG produced a significant number of shoots, while the formation of roots was noticed in all PG treatments (Table 1). Therefore, a lower concentration of cytokinin or higher concentration of PG is required for shoot multiplication in L. ovatifolia. Similar effects of cytokinins were reported for Muscari mirum and Drimia robusta (Nasircilar et al. 2011; Baskaran et al. 2013). In this study, root formation was significantly higher with 40 μM PG (Table 1; Fig. 1d ), indicating that PG influenced root regeneration in L. ovatifolia. PG has auxin-like activity inducing shoots and subsequent rooting, and in other plant species (Zimmerman 1984; Tallón et al. 2012), PG acts synergistically with synthetic auxins. Combinations of BA and NAA significantly increased shoot number; however, the highest number of shoots was produced with 5 μM TDZ and 2 μM NAA (Table 1; Fig. 1e ). Synergistic effects of cytokinin and auxin on shoot proliferation have been observed in other plant species (Mutanyatta et al. 2003; Ascough and Van Staden 2010; Baskaran et al. 2013).
In vitro plant regeneration via direct and indirect organogenesis from leaf explants of L. ovatifolia. (a) Induction of shoots from leaf explants on MS medium with 5 μM BA. (b) Shoot regeneration with roots on MS medium with 5 μM BA and 40 μM PG. (c) Shoot multiplication on MS medium with 5 μM TDZ. (d) Root growth of shoots in 40 μM PG. (e) Proliferation of shoots in medium with 5 μM TDZ and 2 μM NAA. (f) Production of greenish-white, compact callus on MSS with 30 g L−1 sucrose and 15 μM 2,4-D and white, compact callus on MSS with 40 g L−1 sucrose and 15 μM picloram. (g) Induction of whitish-green, compact callus with 50 g L−1 sucrose and 15 μM picloram. (h) Development of shoot regeneration from callus on MSS with 2 μM picloram, 5 μM TDZ, and 30 μM Gln. (i) Shoot regeneration from callus in medium with 2 μM picloram, 5 μM TDZ, and 25 μM PG. (j) Proliferation of shoots in medium with 2 μM IAA, 5 μM TDZ, and 30 μM Gln. (k) Rooting of shoots in 2 μM IBA. (l) Prolific rooting of shoots with 2 μM IBA and 25 μM PG. (m) Acclimatized ex vitro plants in the greenhouse after 6 mo.
Callus was initiated from the cut edge of leaf explants (100%) except from the control, after 1 wk of culture. After 6 wk, different types of calluses (white compact, and whitish green and greenish white compact) including mucilaginous substances were developed on media containing sucrose and PGR treatments (Fig. 1f ). The effectiveness of PGRs on inducing different types of callus has been reported for other plant species (Wetschnig et al. 2013). In this study, increased concentrations of sucrose (> 50 g L−1) and 15 μM picloram produced the largest quantity of organogenic callus (approx. >2.5 g FW per explant; Fig. 1g ). Higher concentrations of carbon have been reported to be beneficial for organogenesis in monocotyledonous plants (Lu et al. 1983; Jain et al. 1997). In this study, shoot buds were induced in calluses derived from CI medium containing 50 g L−1 sucrose and 15 μM picloram when transferred to regeneration medium (Table 2; Fig. 1h , i). More shoots were obtained with a combination of 2 μM IAA, 5 μM TDZ, and 30 μM Gln, but the mean for this treatment was not significantly different than that obtained with combinations of 2 μM picloram, 5 μM TDZ, and 30 μM Gln, or 25 μM PG (Table 2; Fig. 1j). The addition of PG to shoot regeneration media induced roots, approximately 6 roots, 4 cm long, per shoot. A similar response was observed with other plant species (Sarkar and Naik 2000).
The regenerated shoots (>2 cm long) were transferred to half-strength MS medium supplemented with various concentrations and combinations of NAA, IBA, PG, and GA3. The rooting of shoots varied significantly between treatments (Table 3; Fig. 1k ). Callus was observed at the base of the shoots in NAA treatments. In this study, rooting was significantly improved in media containing combinations of IBA and 0.5 μM GA3 (Table 3). However, the combination of IBA and 25 μM PG was more effective for promoting root induction and shoot growth than other treatments (Table 3; Fig. 1l ). The importance of gibberellins and PG in culture media has been established for in vitro rooting (Zimmerman 1984; George et al. 2008).
Somatic embryogenesis and plantlet conversion
Somatic embryogenesis was achieved from EC on semi-solid MS (MSS) medium with different concentrations of sucrose, PGRs, PG, and MS medium with additional macronutrients. EC developed white, globular, partially pear-shaped, and club-shaped embryoids in all treatments except the control after 4 wk (Fig. 2a ). Similar types of embryoids have been observed in Zingiber officinale and Drimia robusta (Lincy et al. 2009; Baskaran and Van Staden 2014). In this study, embryogenesis was achieved in all treatments with 100% frequency except the control (Table 4). Increased concentrations (>50 g L−1) of sucrose promoted the formation of globular embryos directly from explants as well as EC, but later EC produced different stages (globular to club-shaped cotyledon initiation) of embryoids. These embryoids were large with mucilaginous substances and later appeared profuse white and developed root hairs (Fig. 2b ).
In vitro plant regeneration from somatic embryos in semi-solid and cell suspension cultures of L. montana. (a) Formation of embryoids with EC on MSS medium with 40 g L−1 sucrose and 10 μM picloram. (b) Development of white and soft globular embryos and other stages in MSS medium with 50 g L−1 sucrose and 10 μM picloram. (c) Maturation of SEs with cotyledonary stage on MSS with 1 μM picloram and 0.5 μM TDZ. (d) Germination and plantlet conversion from SEs in MSS with MSN. (e) Induction of friable, EC on MSS medium with 30 g L−1 sucrose, 10 μM picloram, and 10 μM Gln. (f, g) Formation of embryoids with different developmental stages in MSL. (h) Production of large SEs on MSL with TDZ. (i) Induction of long radicle from cotyledonary stage embryo on MSL with GA3. (j) Formation of SEs in cluster. (k) Development of plantlets in MSS medium with 15 g L−1 sucrose, MSN, 0.3 μM GA3, and 0.3 μM PG. (l) Ex vitro plants of L. ovatifolia in the greenhouse.
SEs matured and germinated when transferred to embryo maturation and germination media (Table 4). Somatic embryos produced on medium containing sucrose at 40 g L−1 with 10 μM picloram matured (globular to cotyledonary initiation) best on medium containing 2 μM picloram and 0.5 μM TDZ (Fig. 2c ). These embryos germinated (55.4%) and produced significantly more plantlets (11.2 ± 1.65) on MSS medium containing additional macronutrients (Table 4; Fig. 2d ). The effect of macronutrient and sucrose concentration on somatic embryogenesis has been reported in other plant species (Groll et al. 2002; Ali and Lamarti 2014), while combinations of picloram and TDZ are considered the best inducers for different types of SEs in several bulbous plant species (Bakhshaie et al. 2010; Baskaran and Van Staden 2014).
Cell suspension culture
Different strengths of MSL and MSL with 15 g L−1 sucrose alone or in combinations with Gln or PGRs or PG were evaluated for the development of somatic embryos from EC derived from MS medium containing 30 g L−1 sucrose, 10 μM picloram, and 10 μM Gln (Fig. 2e ). Watery callus with white, soft, globular, and pear-shaped embryos followed by the successive appearance of thick, early torpedo-shaped, torpedo-shaped, and green-cotyledonary stage embryos was formed from cell aggregates (Table 5; Fig. 2f, g ). The number of SEs was improved significantly with a reduced concentration (15 g L−1) of sucrose and Gln treatments (Table 5). Improvement of SEs by osmoticum has been reported for other bulbous plants (Baskaran and Van Staden 2014). A significantly improved number of SEs was observed with the addition of picloram, but this did not differ significantly from the numbers observed with 2,4-D treatments (Table 5). Addition of cytokinins, GA3, and PG to the media significantly increased SE numbers (Table 5). Somatic embryos treated with mTR, TDZ, or zeatin were relatively large and green. However, a higher number of SEs developed with TDZ treatment (Table 5; Fig. 2h ). A positive effect of TDZ for SE production has also been reported for other plant species (Baskaran and Van Staden 2012, 2014; Wang et al. 2013).
Acclimatization
Well-developed plantlets from organogenesis (direct and indirect) and somatic embryogenesis (semi-solid and suspension culture) systems were transferred to plastic pots containing a 1:1 (v/v) vermiculite: sand mixture. The plantlets were successfully acclimatized in a greenhouse (Figs. 1m and 2l ) with a 100% survival rate.
The germination of SEs was best in MSL containing GA3 and PG treatments, while the radicles of cotyledonary embryos were long (approximately, 2–3 cm) (Fig. 2i ). Somatic embryos were more clustered (Fig. 2j ) with increasing concentration (>0.2 μM) of BA, mTR, TDZ, or zeatin (data not shown). This indicated that the type and combination of PGR are important for the production of SEs. Similar phenomena have also been reported for other plant species (Bakhshaie et al. 2010; Baskaran and Van Staden 2012; Wang et al. 2013).
Different developmental stages of SEs (pear-shaped, early torpedo, torpedo, and cotyledonary stages) were evaluated for conversion to plantlets on EG medium (Table 5). SEs germinated in all treatments, but torpedo and cotyledonary stages of SEs promoted rapid plantlet development. Pear-shaped and early torpedo embryos germinated slowly, and most did not germinate in PGR, MSN, or PG-free media. The frequency of germination increased significantly with a reduced concentration (15 g L−1) of sucrose and MSN in full-strength MS medium (Table 5), suggesting that alteration of osmotic stress is essential for improvement of germination in L. ovatifolia. The influence of osmoticum on the germination of SEs has been described in other plant species (Komatsuda et al. 1992).
Addition of GA3 or PG significantly improved germination frequency; however, the combination of 0.3 μM GA3 and 0.3 μM PG resulted in the highest frequency (73.3%) of germination (Table 5; Fig. 2k ). The importance of GA3 for germination of SEs has been reported for other bulbous species (Baskaran and Van Staden 2012; Yücesan et al. 2014). PG is a phenolic compound used as a growth regulator in in vitro studies; however the mechanism in somatic embryogenesis is unclear (Teixeira da Silva et al. 2013). The results of the present study suggest that PG plays a major role in germination of SEs in L. ovatifolia; however, the mechanism needs to be investigated.
Histology and ultrastructure of somatic embryos
Histological and ultra-thin sections of different developmental stages of SE were observed under a compound light microscope and TEM, respectively. The histological observations revealed bipolar SEs including both shoot (SM) and root (RM) meristems, illustrating that these were true SEs in accordance with the phenomenon as observed in other plant species (Thompson et al. 2001). Globular embryos consisted of meristematic cellular ovate regions (MCO) of smaller to larger isodiametric cells with vacuolated, large intercellular spaces (IS) and intense cell division (clusters of two to six isodiametric cells) (Fig. 3a ). Successive cell divisions led to multicellular, bipolar structures characteristic of pear-shaped to cotyledonary stage embryos. The SM regions of pear-shaped to cotyledonary stage embryos existed with epidermal cells, procambial strands, and scattered vascular bundles (VB) (Fig. 3b–g ). Somatic embryos at torpedo to cotyledonary stages showed connective tissues (ground tissues) between shoot and root meristems, while RMs occurred with aggregations of compacted and elongated cells (Fig. 3d ). These cells showed reduced cytoplasm and IS, while epidermis, cortex, quiescent center, procambium (PC), and root cap (CP) tissues appeared clearly (Fig. 3e, h ), indicating the development of roots. The present study revealed that bipolar embryos are typically normal, and shoot and root apical meristems lead to the production of a single plant (Fig. 3a–h ).
Histological (LS) and ultrastructural (TEM) analyses of different developmental stages of somatic embryos of L. ovatifolia. (a) Globular embryo containing meristematic cellular ovate region (MCO) composed of smaller to larger isodiametric cells with vacuolated, large, intercellular spaces (IS), and intense cell division (Bar, 100 μm). (b) Pear-shaped embryo showing clear shoot meristems (SM) containing vacuolated cells, vascular tissues (VT), and IS (Bar, 100 μm). (c) Torpedo-shaped (Bar, 500 μm) and (f) cotyledonary-stage (Bar, 500 μm) embryos showing bipolar structures with SM and root meristem (RM). (d) Torpedo-shaped embryo showing connective tissues (CT) between SM and RM region (Bar, 100 μm), (e) the RM with procambium (PC), epidermis (ED), and root cap (RC) (Bar, 100 μm). (g) Cotyledonary embryo showing SM with vascular bundles (VB) (Bar, 100 μm) and (h) compacted and elongated cells with vascular tissues (VT) in RM regions (Bar, 50 μm). (i) TEM analyses showing ultrastructural peculiarities of the formation of cell with large vacuole (V) and condensed cytoplasm in a globular embryo (Bar, 0.5 μm), (j) reduction of central vacuole and more vacuolated with cytoplasmic organelles in pear-shaped (Bar, 0.5 μm), (k) torpedo-shaped (Bar, 1 μm), and (l) cotyledonary-stage (Bar, 0.2 μm) embryos. (m) The thick cell wall (CW) (Bar, 0.2 μm), (n) large intercellular spaces (IS) (Bar, 0.5 μm), and (o) development of mitochondria (M) (Bar, 0.2 μm) in a globular embryo. (p) Development of cytoplasmic components: nucleus (N), nucleolus (NU), Golgi apparatuses (GA), mitochondria (M), smooth endoplasmic reticulum (SER), and chloroplasts (CP) in SM regions of torpedo (Bar, 0.2 μm), and (q–s) cotyledonary embryos. (t–v) Formation of numerous mitochondria and starch grains (SG) and plastids (PS) in RMs of cotyledonary embryo. Bar q,r 0.5 μm; s 0.2 μm; t 0.5 μm; and u, v 0.2 μm.
Ultrastructural analyses of different developmental stages of SEs by TEM revealed typical eukaryote cellular cytoplasmic components, such as nuclei, mitochondria, starch grains or plastids, endoplasmic reticulum, ribosomes, Golgi bodies, chloroplasts, and vacuoles in the cytoplasm enclosed by cell walls (Fig. 3i–v ). In globular stage embryos, the vacuoles (V) were large, and the cell components were closely attached to the cell wall (CW). However, the size of the central vacuoles was reduced, but more vacuoles and cytoplasmic organelles were present in SM regions of pear-shaped to cotyledonary stages of embryo (Fig. 3j–l ). Vacuoles have physical and metabolic functions that are essential to plant growth as put forth by Marty (1999). The parenchymatous cells had large IS and a thick CW (Fig. 3m, n ), while the cell components were present in the globular to cotyledonary embryos (Fig. 3o–s ). The SMs and RMs regions of torpedo to cotyledonary stage of embryos revealed the presence of numerous mitochondria, plastids, Golgi bodies, large nuclei and nucleoli, and vacuoles with electron-dense substances (Fig. 3p–v ), indicating metabolically active cells, which suggests higher cellular energy that might lead to subsequent cell division. Similar phenomena have been reported in peach palm (Steinmacher et al. 2011). In this study, RM regions of cotyledonary stage embryos revealed clusters of mitochondria and the accumulation of starch grains in plastids of the parenchymatous cells (Fig. 3t–v ), indicating embryoid differentiation, and an energy source for subsequent cell divisions in forming plantlets and storage of carbohydrates in roots or bulbs of L. ovatifolia. Starch is rapidly used during the formation of embryogenic regions with the exception of suspensor-zone cells (Quiroz-Figueroa et al. 2002). The considerable accumulation of starch or starch grains is essential for morphogenesis and differentiation of embryoids. Similar phenomena have been reported in other monocotyledonous species (Seldimirova and Kruglova 2013).
Conclusions
A simple and highly efficient in vitro plant regeneration system via organogenesis (direct and indirect) and somatic embryogenesis (semi-solid and suspension culture) from leaf explants has been established for the first time in L. ovatifolia by manipulating various plant growth regulators, and osmotic stress by regulating the sucrose and macronutrient concentrations. This study indicates the concentrations and combinations of PGR and osmoticum are essential for improving plant regeneration in vitro. The histology and ultrastructural studies demonstrate the bipolar structure and viability somatic embryoids. The developed protocols have great potential for large-scale propagation, clonal propagation for commercial applications, conservation strategies, and medicines. In addition, the system provides a source of embryonic cells and a regeneration system for genetic transformation.
References
Ali NB, Lamarti A (2014) Macronutrients effect on secondary somatic embryogenesis of Moroccan cork oak (Quercus suber L.). Am J Plant Sci 5:1851–1861
Ascough GD, Van Staden J (2010) Micropropagation of Albuca bracteata and A. nelsonii—indigenous ornamentals with medicinal value. S Afr J Bot 76:579–584
Bakhshaie M, Babalar M, Mirmasoumi M, Khalighi A (2010) Somatic embryogenesis and plant regeneration of Lilium ledebourii (Baker) Boiss., an endangered species. Plant Cell Tissue Organ Cult 102:229–235
Baskaran P, Singh S, Van Staden J (2013) In vitro propagation, proscillaridin A production and antibacterial activity in Drimia robusta. Plant Cell Tissue Organ Cult 114:259–267
Baskaran P, Van Staden J (2012) Somatic embryogenesis of Merwilla plumbea (Lindl.) Speta. Plant Cell Tissue Organ Cult 109:517–524
Baskaran P, Van Staden J (2014) Plant regeneration via somatic embryogenesis in Drimia robusta. Plant Cell Tissue Organ Cult 119:281–288
Benelli C, Germana MA, Camino T, Beghe D, Fabbri A (2010) Morphological and anatomical observations of abnormal somatic embryos from anther cultures of Citrus reticulate. Biol Plant 54:224–230
George EF, Hall MA, De Klerk GJ (2008) Plant propagation by tissue culture. The background, vol 1. Springer Verlag, Dordrecht, The Netherlands
Georgiev V, Ivanov I, Berkov S, Pavlov A (2011) Alkaloids biosynthesis by Pancratium maritimum L. shoots in liquid culture. Acta Physiol Plant 33:927–933
Gerstner J (1938) A preliminary check list of Zulu names of plants, with short notes. Bantu Stud 12:321–342
Govaerts R (2016) Species details: Ledebouria ovatifolia (Baker) Jessop. In: Catalogue of Life. Plants people possibilities. http://www.catalogueoflife.org/col/details/species/id/bc54be7751b8a546f599937a208fad64. Cited 29 Jan 2016
Groll J, Mycock DJ, Gray VM (2002) Effect of medium salt concentration on differentiation and maturation of somatic embryos of Cassava (Manihot esculenta Crantz). Ann Bot 89:645–648
Hutchings A, Scott AH, Lewis G, Cunningham AB (1996) Zulu medicinal plants. An inventory. University of Natal Press, Pietermaritzburg, pp 38–44
Jain RK, Davey MR, Cocking EC, Wu R (1997) Carbohydrate and osmotic requirements for high frequency plant regeneration from protoplast-derived colonies of indica and japonica rice varieties. J Exp Bot 48:751–758
Komatsuda T, Lee W, Oka S (1992) Maturation and germination of somatic embryos as affected by sucrose and plant growth regulators in soybeans Glycine gracilis Skvortz and Glycine max (L.) Merr. Plant Cell Tissue Organ Cult 28:103–113
Kumari A, Baskaran P, Van Staden J (2015) Enhanced HIV-1 reverse transcriptase inhibitory and antibacterial properties in callus of Catha edulis Forsk. Phytother Res 29:840–843
Lincy AK, Remashree AB, Sasikumar B (2009) Indirect and direct somatic embryogenesis from aerial stem explants of ginger (Zingiber officinale Rosc.). Acta Bot Croat 68:93–103
Lu CY, Vasil V, Vasil IK (1983) Improved efficiency of somatic embryogenesis and plant regeneration in tissue cultures of maize (Zea mays L.). Theor Appl Genet 66:285–289
Marty F (1999) Plant vacuoles. Plant Cell 11:587–599
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Mutanyatta J, Matapa PG, Shushu DD, Abegaz BM (2003) Homoisoflavonoids and xanthones from the tubers of wild and in vitro regenerated Ledebouria graminifolia and cytotoxic activities of some of the homoisoflavonoids. Phytochemistry 62:794–804
Nasircilar AG, Mirci S, Karaguzel O, Eren O, Baktir I (2011) In vitro propagation of endemic and endangered Muscari mirum from different explant types. Turk J Bot 35:37–43
Orczyk W, Przetakiewicz J, Nadolska-Orczyk A (2003) Somatic hybrids of Solanum tuberosum—application to genetics and breeding. Plant Cell Tissue Organ Cult 74:1–13
Pohl T, Koorbanally C, Crouch NR, Mulholland D (2001) Secondary metabolites of Scilla plumbea, Ledebouria cooperi and Ledebouria ovatifolia (Hyacinthaceae). Biochem Syst Ecol 29:857–860
Quiroz-Figueroa FR, Fuentes-Cerda CFJ, Rojas-Herrera R, Loyola-Vargas VM (2002) Histological studies on the developmental stages and differentiation of two different somatic embryogenesis systems of Coffea arabica. Plant Cell Rep 20:1141–1149
Ramachandra RS, Ravishankar GA (2002) Plant cell cultures: chemical factories of secondary metabolites. Biotechnol Adv 20:101–153
SANBI (2015) Statistics: red list of South African plants version 2015.1. http://redlist.sanbi.org/genus.php?genus=3800-10
Sarkar D, Naik PS (2000) Phloroglucinol enhances growth and rate of axillary shoot proliferation in potato shoot tip cultures in vitro. Plant Cell Tissue Organ Cult 60:139–149
Seldimirova QA, Kruglova NN (2013) Properties of the initial stages of embryoidogenesis in vitro in wheat calli of various origin. Biol Bull 40:447–454
Sharma K, Dubey S (2011) Biotechnology and conservation of medicinal plants. J Exp Sci 2:60–61
Shushu DD, Comar JM, Abegaz BM (2009) Somaclonal variation in in vitro regenerated Ledebouria graminifolia (Hyacinthaceae), an indigenous bulb in Botswana and its potential exploitation as an ornamental plant. J Biol Sci 9:152–158
Sparg SG, Van Staden J, Jager AK (2002) Pharmacological and phytochemical screening of two Hyacinthaceae species: Scilla natalensis and Ledebouria ovatifolia. J Ethnopharmacol 80:95–101
Steinmacher DA, Guerra MP, Saare-Surminski K, Lieberei R (2011) A temporary immersion system improves in vitro regeneration of peach palm through secondary somatic embryogenesis. Ann Bot 108:1463–1475
Tallón CI, Porras I, Pérez-Tornero O (2012) Efficient propagation and rooting of three citrus rootstocks using different plant growth regulators. In Vitro Cell Dev Biol Plant 48:488–499
Teixeira da Silva JA, Dobránszki J, Ross S (2013) Phloroglucinol in plant tissue culture. In Vitro Cell Dev Biol Plant 49:1–16
Thompson D, Harrington F, Douglas G, Hennerty MJ, Nakhshab N, Long R (2001) Vegetative propagation techniques for oak, ash, sycamore and spruce. COFORD, Dublin
Waller CP, Thumser AE, Langat MK, Crouch NR, Mulholland DA (2013) COX-2 inhibitory activity of homoisoflavanones and xanthones from the bulbs of the Southern African Ledebouria socialis and Ledebouria ovatifolia (Hyacinthaceae: Hyacinthoideae). Phytochemisty 95:284–290
Wang S, Yang F, Jiu L, Zhang W, Zhang W, Tian Z, Wang F (2013) Plant regeneration via somatic embryogenesis from leaf explants of Muscari armeniacum. Biotechnol Biotechnol Equip 27:4243–4247
Wetschnig W, Brosch U, Andriatiana J, Dutta S, Knirsch W (2013) In vitro propagation and ex situ conservation of Drimia cryptopoda and Ledebouria nossibeensis, two endangered endemic Hyacinthaceae from Madagascar. Scr Bot Belg 50:33–36
Yücesan BB, Çiçek F, Gürel E (2014) Somatic embryogenesis and encapsulation of immature bulblets of an ornamental species, grape hyacinths (Muscari armeniacum Leichtlin ex Baker). Turk J Agric For 38:716–722
Zimmerman RH (1984) Rooting apple cultivars in vitro: interactions among light, temperature, phloroglucinol and auxin. Plant Cell Tissue Organ Cult 3:301–311
Acknowledgments
Financial support by the National Research Foundation (NRF), Pretoria and the University of KwaZulu-Natal, Pietermaritzburg is gratefully acknowledged. The authors are grateful to Subashen Naidu and all other Staff of the Microscopy & Microanalysis Unit (MMU), UKZN, Pietermaritzburg for microscopic assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Jorge Canhoto
Rights and permissions
About this article
Cite this article
Baskaran, P., Kumari, A., Naidoo, D. et al. In vitro propagation and ultrastructural studies of somatic embryogenesis of Ledebouria ovatifolia . In Vitro Cell.Dev.Biol.-Plant 52, 283–292 (2016). https://doi.org/10.1007/s11627-016-9762-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-016-9762-9