Abstract
Background
Anastomotic leak (AL) is a feared complication after colorectal surgery. Prompt diagnosis and treatment are crucial. C-reactive protein (CRP) and procalcitonin (PCT) have been proposed as early AL indicators. The aim of this systematic review was to evaluate the CRP and CPT predictive values for early AL diagnosis after colorectal surgery.
Methods
Systematic literature search to identify studies evaluating the diagnostic accuracy of postoperative CRP and CPT for AL. A Bayesian meta-analysis was carried out using a random-effects model and pooled predictive parameters to determine postoperative CRP and PCT cut-off values at different postoperative days (POD).
Results
Twenty-five studies (11,144 patients) were included. The pooled prevalence of AL was 8% (95 CI 7–9%), and the median time to diagnosis was 6.9 days (range 3–10). The derived POD3, POD4 and POD5 CRP cut-off were 15.9 mg/dl, 11.4 mg/dl and 10.9 mg/dl respectively. The diagnostic accuracy was comparable with a pooled area under the curve (AUC) of 0.80 (95% CIs 0.23–0.85), 0.84 (95% CIs 0.18–0.86) and 0.84 (95% CIs 0.18–0.89) respectively. Negative likelihood ratios (LR−) showed moderate evidence to rule out AL on POD 3 (LR− 0.29), POD4 (LR− 0.24) and POD5 (LR− 0.26). The derived POD3 and POD5 CPT cut-off were 0.75 ng/ml (AUC = 0.84) and 0.9 ng/ml (AUC = 0.92) respectively. The pooled POD5 negative LR (−0.18) showed moderate evidence to rule out AL.
Conclusions
In the setting of colorectal surgery, CRP and CPT serum concentrations lower than the derived cut-offs on POD3-POD5, may be useful to rule out AL thus possibly identifying patients at low risk for AL development.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Anastomotic leak (AL) is a feared complication after colorectal surgery with a reported incidence ranging from 2 to 17% depending on patient comorbidities, operating surgeon expertise, emergency/elective settings and hospital volume.1,2,3,4,5,6,7,8 AL has been shown to be associated with increased mortality, prolonged hospital stay, substantial healthcare costs and worsening oncological outcomes (i.e. increased local recurrence).9,9,11 Even if several risk factors have been identified for postoperative AL, it remains difficult to predict its development. This is because the onset of AL is insidious with a potential early or late presentation.
Prompt diagnosis and treatment are crucial to potentially limit the related consequences.12,13 Inflammatory biomarkers like C-reactive protein (CRP) and procalcitonin (PCT) have been proposed as decision-making indicators for patient discharge and proposed for early AL diagnosis.14,13,16 CRP has been shown to have a significant correlation with postoperative infectious complications while PCT seems more sensitive and reliable for AL.17,16,17,18,21 Previous meta-analyses evaluated the predictive value of both CRP and PCT for the development of AL after colorectal surgery.22,21,22,25 However, results were conflicting and heterogeneous. Therefore, since the publication of recent studies, a new evidence has become available.
Hence, the aim of the present Bayesian meta-analysis was to perform an updated systematic literature review and investigate the association of postoperative serum CRP/PCT concentrations with AL and assess their predictive role in the early diagnosis of AL after colorectal surgery.
Materials and Methods
We conducted this study according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement.26 Institutional review board approval was not required. MEDLINE, Scopus, Web of Science, Cochrane Central Library, and ClinicalTrials.gov were used.27 The last date of search was 28 February 2022. A combination of the following MeSH (Medical Subject Headings) terms was used (“C-reactive protein” (tiab), OR “CRP” (tiab)) AND (“procalcitonin” (tiab), OR “PCT” (tiab)) AND (“leak” (tiab), OR “anastom* leak” (tiab), OR “fistula”, OR “ dehiscence”) AND (“colorec*” (tiab), OR “colon” (tiab), OR “bowel”, OR “gastrointestinal”) AND (“surgery” (tiab), OR “operation” (tiab)) AND (“laparotomy” (tiab), OR “laparosc*” (tiab), OR “rob*”). All titles were initially evaluated and suitable abstracts extracted. The search was completed by consulting the listed references of each article. The study protocol was registered at the PROSPERO (International prospective register of systematic reviews) (Registration Number: CRD42020220698).
Eligibility Criteria
Inclusion criteria were as follows: (a) comparative studies investigating serum CRP (mg/dl) and PCT (ng/ml) data and their predictive values for AL at different postoperative days (POD) in patients undergoing colorectal surgery with resection and anastomosis; (b) comparative studies reporting data for open and minimally invasive (laparoscopic and robotic) approaches for both benign (i.e. inflammatory bowel disease, symptomatic diverticular disease or other indications) and malignant diseases; (c) English written; (d) when two or more papers were published by the same institution or study group or used the same data-set, or articles with the largest sample size; (e) in case of duplicate studies with accumulating numbers of patients, only the most complete reports were included for quantitative analysis. Exclusion criteria were as follows: (a) studies not reporting the predictive value for postoperative serum CRP and PCT; (b) studies not reporting AL outcome separately from other septic complications; (c) not English-written; (d) abstracts, case reports and case series with less than 10 patients.
Data Extraction
The following data were collected: authors, year of publication, country, study design, number of patients, sex, age, body mass index (BMI), American Society of Anesthesiologists (ASA) physical status, comorbidities, surgical indication, emergent vs. urgent procedure, surgical approach (open vs. minimally invasive), area under the receiver operating characteristics (ROC) curve, sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV). Furthermore, serum CRP (mg/dl) and CPT (ng/ml) values were collected at different PODs. To obtain a summary graph of postoperative CRP and CPT levels, data reported in the text, graphs or figures of the included studies were used and/or digitalized to obtain the median or mean values. The outcome of interest was AL, which was counted per event and defined as reported in the included studies (Supplementary Table 1). In general, AL was defined as radiological, endoscopic or operative evidence of defect in the enteric wall at the site of the anastomosis. If necessary, corresponding authors were contacted to obtain the missing data if not retrievable from the article. All data were computed independently by four investigators (AA, AS, LC, GB) and compared at the end of the reviewing process. A fifth author (DB) reviewed the database and determined discrepancies.
Outcomes
The primary outcome was the assessment of postoperative serum CPR (mg/dl) values at different intervals (from POD1 to POD5) and its predictive value for AL. The secondary outcome was postoperative serum CPT (ng/ml) assessment at POD3 and POD5 and its predictive value for AL.
Quality Assessment
Three investigators (AA, AS, LC) independently evaluated the methodological quality of the papers using the Quality Assessment of Diagnostic Accuracy Studies (QUADAS-2) tool.28,29 This assessed the risk of bias and concerns about applicability by evaluating four key domains: patient selection, index test, reference standard and flow of patients through the study and timing of tests.
Statistical Analysis
Bivariate meta-analysis was conducted using a fully Bayesian approach via integrated nested Laplace approximations (INLA). Compared to traditional meta-analyses, the Bayesian approach takes into account all sources of variation and reflects these variations in pooled results.30,31 Furthermore, the Bayesian approach can provide more accurate estimates for small samples.32 Chu and Cole’s bivariate generalized linear mixed effect with exact binomial likelihood model was used to summarize the results of several diagnostic studies by modelling sensitivity and specificity jointly (binomial-normal model).33 We assume that both sensitivity and specificity were modelled with the same logit link function. Normal prior with zero mean and 100 variance is used for the fixed effects. Variance components of random effect were modelled using penalized complexity priors choosing the parameters believing that the sensitivities or specificities lie in the interval [0.5, 0.95] with probability 0.95, according to Wakefield.34 The binomial-normal model was also used to calculate the hierarchical summary receiver operating characteristic (HSROC) model according to Rutter et al.35 Uniform distribution on [−1,1] was the choice for a vague prior of the random effects correlation parameter. Bayesian sensitivity analysis was performed changing the variance component priors. Pooled estimates of likelihood ratios (LR) and diagnostic odds ratio (DOR) were estimated according to Zwinderman and Bossuyt.36 The geometric mean of the reported CRP and CPT cut-off values at each POD was used to derive the pooled CRP and CPT cut-off values.19 The pooled prevalence of anastomotic leak was calculated according to Bona et al.37 Credible intervals (CIs) at 95% were computed. All analyses and figures were carried out using R software package version 3.4.3.38
Results
Systematic Review
Twenty-five studies met the inclusion criteria (Figure 1). The results of quality assessment using the QUADAS-2 tool are shown in Figure 2. The applicability of included studies was good. Overall, 11,144 patients were included (range 32–2501). All reports were observational; fifteen were of prospective while ten were of retrospective design. Demographic, clinical and operative variables of the patient sample are shown in Table 1. The age of the patient population ranged from 18 to 93 years, and half were males (55.9%). The preoperative ASA score was reported in ten studies39,38,39,40,41,42,43,44,45,48 and BMI in five studies.20,41,45,49,50 Elective surgery was performed in 93.3% of patients. Colorectal resections were performed via open (49.1%) and minimally invasive (laparoscopic or robotic) approaches in 50.9%. The type of anastomosis was specified in seven studies15,21,40,41,51,50,53 with stapled anastomosis being fashioned in the majority of patients (76.2%). Ileocolic, colocolic and rectal anastomoses were performed in 35.1%, 31.8% and 33.1% respectively. Surgery for cancer was performed in 75.6% of patients with almost one-fourth of procedures being performed for benign diseases (i.e. diverticulitis, inflammatory bowel disease, etc.). Tumour stage was reported in five studies,16,42,41,44,54 and the use of neoadjuvant chemoradiotherapy was specified in two studies.15,42
The included studies reported measuring CRP and CPT in the postoperative period according to different institutional protocols. The median time to AL diagnosis was 6.9 days (range 3–10). Patients were stratified according to the presence of anastomotic leak (AL group) vs. no AL (no AL group). Overall, 12 studies reported CRP levels on POD1, 13 studies on POD2, 17 studies on POD3, 13 studies on POD4 and 11 studies on POD5. The CRP serum concentration peak was observed on POD3. The pooled postoperative CRP serum levels in the two groups are shown in Figure 3. There was a statistically significant difference comparing the mean CRP concentration between the two groups on POD3 (20.5 vs. 11.5; p = 0.021), POD4 (17.8 vs. 9.6; p = 0.017) and POD5 (16.3 vs. 7.4; p = 0.024). Similarly, eight studies reported serum CPT concentrations on POD3 and 4 studies on POD5. The median PCT concentrations were significantly higher for AL vs. no AL on POD3 (3.7 vs. 0.59; p = 0.001) and POD5 (4.25 vs. 0.36; p < 0.001).
Bayesian Meta-Analysis
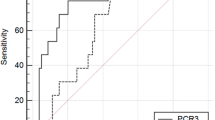
In addition to a systematic review, we performed a study-level fully Bayesian meta-analysis. Considering the random effect bivariate model, the estimated pooled POD3 CRP cut-off, resulting from 17 studies (6807 patients), is 15.9 mg/dl. The estimated pooled AUC is 0.80 (95% CIs 0.23–0.85) (Figure 4A, B, C). The calculated correlation between sensitivity and specificity was 0.09 suggesting no threshold effect. The estimated pooled POD4 CRP cut-off, resulting from 14 studies (7366 patients), is 11.4 mg/dl (Figure 5A, B, C). The estimated pooled AUC is 0.84 (95% CIs 0.18–0.86). The calculated correlation between sensitivity and specificity was −0.13 suggesting no threshold effect. The estimated pooled POD5 CRP cut-off, resulting from 12 studies (3943 patients), is 10.9 mg/dl. The estimated pooled AUC is 0.84 (95% CIs 0.18–0.89) (Figure 6A, B, C). The calculated correlation between sensitivity and specificity was −0.11 suggesting no threshold effect.
The estimated pooled POD3 CPT cut-off, resulting from 9 studies (5791 patients), is 0.75 ng/ml. The estimated pooled AUC is 0.84 (95% CIs 0.18–0.89). The calculated correlation between sensitivity and specificity was −0.59 suggesting the presence of threshold effect. The estimated pooled POD5 CPT cut-off, resulting from 4 studies (1009 patients), is 0.90 ng/ml. The estimated pooled AUC is 0.92 (95% CIs 0.89–0.97). The calculated correlation between sensitivity and specificity was −0.45 suggesting the presence of threshold effect. Pooled sensitivity, specificity, positive LR, negative LR and DOR are reported in Table 2.
The estimated pooled prevalence of anastomotic leak resulting from 25 studies (11,144 patients) is 8% (95% CI = 7–9%) (Figure 7). The Fagans’ nomograms for CRP on POD3, POD4 and POD5 are shown in Figure 8A, B, C.
Discussion
This meta-analysis shows that postoperative CRP serum concentrations on POD3, POD4 and POD5 and the PCT serum concentration on POD5 may be useful tests to rule out AL. These markers should be considered as negative tests while values below the cut-off may theoretically help in the identification of patients who are unlikely to develop AL.
The incidence of AL after colorectal surgery has been reported ranging from 2 to 17% depending on surgeon experience, hospital volumes, surgical indications and technical approaches.1,2,3,4,5 AL remains a disastrous consequence with increased resources utilization, costs, hospital length of stay, morbidity and related mortality.6,7,8,9,11 In the era of fast-track recovery, early detection and treatment of AL are even more critical to optimizing perioperative care, minimizing surgical complications and expediting recovery thus possibly reducing the rate of surgical revision. In the present study, the estimated pooled prevalence of postoperative AL was 8% (95% CIs 7–9%).
Various risk factors have been reported to be associated with AL; however, its prediction remains challenging in individual patients.4,5,6 Different strategies for early AL detection have been proposed using serum inflammatory biomarkers. Specifically, postoperative CRP and CPT serum concentrations at different POD have been proposed with contrasting results. CRP is a serum acute-phase reactant synthesized almost exclusively in the liver and is released in response to stimulation by proinflammatory cytokines such as interleukin 6 and tumour necrosis factor α.59 CRP is a reliable, but non-specific, marker of acute inflammation and has been investigated as an early indicator of infectious complications following abdominal surgery. In contrast, PCT is a peptide precursor of the hormone calcitonin produced by parafollicular cells (C cells) of the thyroid and by the neuroendocrine cells of the lung and the intestine.60 PCT is classified as a sensitive and reliable acute-phase reactant while its serum concentration rises in response to pro-inflammatory stimulus, especially of bacterial origin.61 It has been suggested that the identification of clinically relevant CRP serum concentrations and cut-off may be helpful to fast-track pathways by providing an early alert for leakage.62,63 In our study, we noticed significantly higher serum CRP concentrations in patients with AL compared to patients with no AL with a peak value on POD3 (Figure 3). The derived pooled CRP cut-off values on POD3, POD4 and POD5 were 15.9 mg/dl, 11.4 mg/dl and 10.9 mg/dl respectively. The diagnostic accuracy of these values is comparable and supported by the pooled AUC ROC curves of 0.80 (POD3), 0.84 (POD4) and 0.84 (POD5) respectively. The accuracy of the cut-off was also assessed with the analysis of LRs. Compared to predictive values that are useful to measure the accuracy of a predictive test, LRs are more precise in estimating the diagnostic probability of a single test, thus providing a more precise individual risk assessment. For these reasons, positive LRs are used in clinical practice to confirm (rule in) while negative LRs are used to exclude (rule out) a specific outcome or complication.64,65 In the present article, reflecting the poor CRP specificity, positive LRs were associated with weak evidence for AL diagnosis on POD 3 (LR+ 3.21), POD4 (LR+ 3.29) and POD 5 (LR+ 3.81). On the other hand, negative LRs showed moderate evidence to exclude (rule out) leakage on POD 3 (LR− 0.29), POD4 (LR− 0.24) and POD5 (LR− 0.26). This means that for a patient with a pre-test probability of 8%, a serum CRP concentration below the identified cut-off values on POD3 (CRP <15.9 mg/dl), POD4 (11.4 mg/dl) and POD5 (10.9 mg/dl), the post-test probability of AL is about 2.3% (Figure 8A, B, C). Notably, the lower limit of CIs of negative LR for CRP on POD3, POD4 and POD5 were 0.21, 0.19 and 0.18, respectively. This suggests that, in the absence of clinical and/or radiological suspicion, CRP may provide reasonable evidence to exclude (rule out) leakage and identify patients at low risk of AL development. By including a larger patient sample, our results broaden and further corroborate findings of previous meta-analyses. Specifically, Singh et al. in their 2013 meta-analysis included seven studies (2483 patients).19 The authors identified different CRP cut-offs on POD3 (172 mg/l), POD4 (124 mg/l) and POD5 (144 mg/l) with comparable diagnostic accuracy and remarkable associated negative LR. The authors concluded that CRP is a useful negative predictive test to rule out AL following colorectal surgery. Similarly, Yeung et al. in their 2021 analysis included 23 articles (6647 patients).22 The authors found a significantly higher serum CRP concentration in patients with AL compared to patients without AL and identified specific CRP cut-offs for POD3 (148 mg/l), POD4 (123 mg/l) and POD5 (115 mg/l) useful to predict AL after colorectal surgery. However, with the noteworthy diagnostic accuracy tested with AUC, the authors failed to assess and report pooled LRs. Compared to predictive values, these are more accurate for an individual risk assessment.
Postoperative serum CPT concentrations were significantly higher on POD3 (3.7 vs. 0.59; p = 0.001) and POD5 (4.25 vs. 0.36; p < 0.001) in patients with AL compared to patients with no AL. Pooled CPT cut-off values were 0.75 mg/dl and 0.9 ng/ml respectively. The related AUC ROC curves were 0.81 and 0.92. Interestingly, negative LR (−0.18) showed moderate evidence to rule out leakage on POD 5. Again, this means that for a patient with a pre-test probability of 8%, a serum CPT concentration below the identified cut-off value of 0.9 ng/ml (POD5), the post-test probability of AL is almost 2%. This result is similar to what previously reported by Su’a et al. and Cousin et al., who defined a high diagnostic PCT accuracy for AL on POD5.25,66
The main result of the present study is the association among high postoperative serum CRP/CPT concentrations and AL. The identification of postoperative cut-offs at various POD may be useful to exclude (rule out) AL or to identify patients at low risk for AL development. Therefore, in the context of a fast-track recovery protocol, CRP and CPT may be possibly useful for early diet advancement and safe discharge.29 However, owing to differences in patient population, study design and methodology, our results are limited by the heterogeneity of the included studies. The different surgical approaches (open vs. minimally invasive), emergency or elective settings, benign or malignant pathology, different definitions of anastomotic leak, level of anastomosis (ileocolic vs. colocolic vs. rectal), type of anastomosis (hand-sewn or stapled), severity of AL with different degrees of peritoneal contamination and effect of neoadjuvant treatment may contribute to inter-study heterogeneity. Furthermore, few of the included studies were of retrospective design while postoperative CRP and CPT measurements were not performed sequentially on each POD. Although meta-analysis is not a widely approved method for summarizing predictive data, this study may provide a useful guide for the interpretation of CRP and CPT measurements following colorectal surgery. These cut-offs are not a panacea and their isolated use and dichotomous interpretations are not advisable, while a parallel consideration of trends in conjunction with clinical and radiological signs seems prudent.50 Therefore, cut-offs should be considered as complementary tools and additional arrows and in any surgeons’ quiver.
Conclusions
CRP and CPT values lower than the derived cut-offs on POD3-POD5 may be useful tools to rule out leak after colorectal surgery thus identifying patients at low risk for AL development. In the context of enhanced recovery after surgery protocols, the integration of a CRP and CPT-based diagnostic algorithm as complementary instruments to clinical assessment may be valuable to reduce global cost, improve outcomes and patient care.
References
Thornton M, Joshi H, Vimalachandran C, Heath R, Carter P, Gur U, Rooney P. Management and outcome of colorectal anastomotic leaks. Int J Colorectal Dis. 2011;26(3):313–20.
Hyman N, Manchester TL, Osler T, Burns B, Cataldo PA. Anastomotic leaks after intestinal anastomosis: it’s later than you think. Ann Surg. 2007;245:254–258.
Alves A, Panis Y, Pocard M, Regimbeau JM, Valleur P. Management of anastomotic leakage after nondiverted large bowel resection. J Am Coll Surg. 1999;189:554–559.
McDermott FD, Heeney A, Kelly ME, Steele RJ, Carlson GL, Winter DC. Systematic review of preoperative, intraoperative and postoperative risk factors for colorectal anastomotic leaks. Br J Surg. 2015;102(5):462–79.
Sánchez–Guillén L, Frasson M, García–Granero Á, Pellino G, Flor–Lorente B, Álvarez–Sarrado E, García–Granero E. Risk factors for leak, complications and mortality after ileocolic anastomosis: comparison of two anastomotic techniques. Ann R Coll Surg Engl. 2019;101(8):571–578.
van Rooijen SJ, Huisman D, Stuijvenberg M, Stens J, Roumen RMH, Daams F, Slooter GD. Intraoperative modifiable risk factors of colorectal anastomotic leakage: Why surgeons and anesthesiologists should act together. Int J Surg. 2016;36(Pt A):183–200.
Huo YR, Phan K, Morris DL, Liauw W. Systematic review and a meta–analysis of hospital and surgeon volume/outcome relationships in colorectal cancer surgery. J Gastrointest Oncol. 2017;8(3):534–546.
Rausa E, Zappa MA, Kelly ME, Turati L, Russo L, Aiolfi A, Bonitta G, Sgroi LG (2019) A standardized use of intraoperative anastomotic testing in colorectal surgery in the new millennium: is technology taking over? A systematic review and network meta–analysis. Techniques in Coloproctology 23(7) 625–631. https://doi.org/10.1007/s10151-019-02034-6
Sciuto A, Merola G, De Palma GD, Sodo M, Pirozzi F, Bracale UM, Bracale U. Predictive factors for anastomotic leakage after laparoscopic colorectal surgery. World J Gastroenterol. 2018;7;24(21):2247–2260.
Kastora SL, Osborne LL, Jardine R, Kounidas G, Carter B, Myint PK. Non–steroidal anti–inflammatory agents and anastomotic leak rates across colorectal cancer operations and anastomotic sites: A systematic review and meta–analysis of anastomosis specific leak rate and confounding factors. Eur J Surg Oncol. 2021;47(11):2841–2848.
Koedam TWA, Bootsma BT, Deijen CL, van de Brug T, Kazemier G, Cuesta MA, Fürst A, Lacy AM, Haglind E, Tuynman JB, Daams F, Bonjer HJ; COLOR COLOR II study group. Oncological Outcomes After Anastomotic Leakage After Surgery for Colon or Rectal Cancer: Increased Risk of Local Recurrence. Ann Surg. 2022;1;275(2):e420–e427.
Macarthur DC, Nixon SJ, Aitken RJ. Avoidable deaths still occur after large bowel surgery. Scottish Audit of Surgical Mortality, Royal College of Surgeons of Edinburgh. Br J Surg. 1998;85(1):80–3.
Aiolfi A, Bona B, Guerrazzi G, Bonitta G, Rausa E, Panizzo V, Campanelli G, Micheletto G (2020) Intracorporeal Versus Extracorporeal Anastomosis in Laparoscopic Right Colectomy: An Updated Systematic Review and Cumulative Meta–Analysis. Journal of Laparoendoscopic & Advanced Surgical Techniques. 30(4)402–412. https://doi.org/10.1089/lap.2019.0693
Lane JC, Wright S, Burch J, Kennedy RH, Jenkins JT. Early prediction of adverse events in enhanced recovery based upon the host systemic inflammatory response. Colorectal Dis. 2013;15(2):224–30.
Lagoutte N, Facy O, Ravoire A, Chalumeau C, Jonval L, Rat P, Ortega–Deballon P. C–reactive protein and procalcitonin for the early detection of anastomotic leakage after elective colorectal surgery: pilot study in 100 patients. J Visc Surg. 2012;149(5):e345–9.
Platt JJ, Ramanathan ML, Crosbie RA, Anderson JH, McKee RF, Horgan PG, McMillan DC. C–reactive protein as a predictor of postoperative infective complications after curative resection in patients with colorectal cancer. Ann Surg Oncol. 2012;19(13):4168–77.
Warschkow R, Beutner U, Steffen T, Muller SA, Schmied BM, Guller U et al. Safe and early discharge after colorectal surgery due to C–reactive protein: a diagnostic meta–analysis of 1832 patients. Ann Surg. 2012;256:245–250.
Bona D, Micheletto G, Bonitta G, Panizzo V, Cavalli M, Rausa E, Cirri S, Aiolfi A. Does C–reactive Protein Have a Predictive Role in the Early Diagnosis of Postoperative Complications After Bariatric Surgery? Systematic Review and Bayesian Meta–analysis. Obes Surg. 2019;29(11):3448–3456.
Singh PP, Zeng IS, Srinivasa S, Lemanu DP, Connolly AB, Hill AG. Systematic review and meta–analysis of use of serum C–reactive protein levels to predict anastomotic leak after colorectal surgery. Br J Surg. 2014;101(4):339–46.
Facy O, Paquette B, Orry D, Binquet C, Masson D, Bouvier A, Fournel I, Charles PE, Rat P, Ortega–Deballon P; IMACORS Study. Diagnostic Accuracy of Inflammatory Markers As Early Predictors of Infection After Elective Colorectal Surgery: Results From the IMACORS Study. Ann Surg. 2016;263(5):961–6.
Sala Hernandez A, Frasson M, García–Granero A, Hervás Marín D, Laiz Marro B, Alonso Pardo R, Aldrey Cao I, Alvarez Perez JA, Roque Castellano C, García González JM, Tabet Almeida J, García–Granero E; EDEN study group. Diagnostic accuracy of C–reactive protein, procalcitonin and neutrophils for the early detection of anastomotic leakage after colorectal resection: a multicentric, prospective study. Colorectal Dis. 2021;23(10):2723–2730.
Yeung DE, Peterknecht E, Hajibandeh S, Hajibandeh S, Torrance AW. C–reactive protein can predict anastomotic leak in colorectal surgery: a systematic review and meta–analysis. Int J Colorectal Dis. 2021;36(6):1147–1162.
Adamina M, Steffen T, Tarantino I, Beutner U, Schmied BM, Warschkow R. Meta–analysis of the predictive value of C–reactive protein for infectious complications in abdominal surgery. Br J Surg. 2015;102(6):590–8.
Gans SL, Atema JJ, van Dieren S, Groot Koerkamp B, Boermeester MA. Diagnostic value of C–reactive protein to rule out infectious complications after major abdominal surgery: a systematic review and meta–analysis. Int J Colorectal Dis. 2015;30(7):861–73.
Cousin F, Ortega–Deballon P, Bourredjem A, Doussot A, Giaccaglia V, Fournel I. Diagnostic Accuracy of Procalcitonin and C–reactive Protein for the Early Diagnosis of Intra–abdominal Infection After Elective Colorectal Surgery: A Meta–analysis. Ann Surg. 2016 Aug;264(2):252–6.
Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta–analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009;339:b2700. https://doi.org/10.1136/bmj.b2700.
Goossen K, Tenckhoff S, Probst P, et al. Optimal literature search for systematic reviews in surgery. Langenbecks Arch Surg. 2018 Feb;403(1):119–129.
Whiting PF, Rutjes AW, Westwood ME, Mallett S, Deeks JJ, Reitsma JB, Leeflang MM, Sterne JA, Bossuyt PM; QUADAS–2 Group. QUADAS–2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med. 2011;18;155(8):529–36.
Aiolfi A, Asti E, Rausa E, Bonavina G, Bonitta G, Bonavina L. Use of C–reactive protein for the early prediction of anastomotic leak after esophagectomy: Systematic review and Bayesian meta–analysis. PLoS One. 2018;17;13(12):e0209272.
Sutton AJ, Abrams KR. Bayesian methods in meta–analysis and evidence synthesis. Stat. Methods Med. Res. 2001;10:277–303.
Aiolfi A, Tornese S, Bonitta G, Rausa E, Micheletto G, Bona D. Roux–en–Y gastric bypass: systematic review and Bayesian network meta–analysis comparing open, laparoscopic, and robotic approach. Surg Obes Relat Dis. 2019;15(6):985–994.
Higgins JP, Thompson SG, Spiegelhalter DJ. A re–evaluation of random–effects meta–analysis. J R Stat Soc Ser A Stat Soc. 2009;172:137–159.
Chu H, Cole SR. Bivariate meta–analysis of sensitivity and specificity with sparse data: A generalized linear mixed model approach. J Clin Epidemiol. 2006;59(12):1331–1332.
Wakefield J. Disease mapping and spatial regression with count data. Biostatistics. 2007;8(2):158–183.
Rutter CM, Gatsonis CA. A hierarchical regression approach to meta–analysis of diagnostic test accuracy evaluations. Statistics in Medicine. 2021;20(19):2865–2884.
Zwinderman AH, Bossuyt PM. We should not pool diagnostic likelihood ratios in systematic reviews. Stat Med. 2008;27(5):687–97.
Bona D, Lombardo F, Matsushima K, Cavalli M, Panizzo V, Mendogni P, Bonitta G, Campanelli G, Aiolfi A. Diaphragmatic herniation after esophagogastric surgery: systematic review and meta–analysis. Langenbecks Arch Surg. 2021;406(6):1819–1829.
R Core Team. R: a language and environment for statistical computing. R Foundation for Statistical Computing [cited 2022 Mar 14].
Kørner H, Nielsen HJ, Søreide JA, Nedrebø BS, Søreide K, Knapp JC. Diagnostic accuracy of C–reactive protein for intraabdominal infections after colorectal resections. J Gastrointest Surg. 2009;13(9):1599–606.
Garcia–Granero A, Frasson M, Flor–Lorente B, Blanco F, Puga R, Carratalá A, Garcia–Granero E. Procalcitonin and C–reactive protein as early predictors of anastomotic leak in colorectal surgery: a prospective observational study. Dis Colon Rectum. 2013;56(4):475–83.
Ramos Fernández M, Rivas Ruiz F, Fernández López A, Loinaz Segurola C, Fernández Cebrián JM, de la Portilla de Juan F. C reactive protein as a predictor of anastomotic leakage in colorectal surgery. Comparison between open and laparoscopic surgery. Cir Esp. 2017;95(9):529–535.
Reynolds IS, Boland MR, Reilly F, Deasy A, Majeed MH, Deasy J, Burke JP, McNamara DA. C–reactive protein as a predictor of anastomotic leak in the first week after anterior resection for rectal cancer. Colorectal Dis. 2017;19(9):812–818.
Mik M, Dziki L, Berut M, Trzcinski R, Dziki A. Neutrophil to Lymphocyte Ratio and C–Reactive Protein as Two Predictive Tools of Anastomotic Leak in Colorectal Cancer Open Surgery. Dig Surg. 2018;35(1):77–84.
Zawadzki M, Krzystek–Korpacka M, Gamian A, Witkiewicz W. Serum cytokines in early prediction of anastomotic leakage following low anterior resection. Wideochir Inne Tech Maloinwazyjne. 2018;13(1):33–43.
Muñoz JL, Alvarez MO, Cuquerella V, Miranda E, Picó C, Flores R, Resalt–Pereira M, Moya P, Pérez A, Arroyo A. Procalcitonin and C–reactive protein as early markers of anastomotic leak after laparoscopic colorectal surgery within an enhanced recovery after surgery (ERAS) program. Surg Endosc. 2018;32(9):4003–4010.
Pantel HJ, Jasak LJ, Ricciardi R, Marcello PW, Roberts PL, Schoetz DJ Jr, Read TE. Should They Stay or Should They Go? The Utility of C–Reactive Protein in Predicting Readmission and Anastomotic Leak After Colorectal Resection. Dis Colon Rectum. 2019;62(2):241–247.
Pantoja Pachajoa DA, Gielis M, Palacios Huatuco RM, Benitez MN, Avila MN, Doniquian AM, Alvarez FA, Parodi M. Neutrophil–to–lymphocyte ratio vs C–reactive protein as early predictors of anastomotic leakage after colorectal surgery: A retrospective cohort study. Ann Med Surg (Lond). 2021;5;64:102201.
Baeza–Murcia M, Valero–Navarro G, Pellicer–Franco E, Soria–Aledo V, Mengual–Ballester M, Garcia–Marin JA, Betoret–Benavente L, Aguayo–Albasini JL. Early diagnosis of anastomotic leakage in colorectal surgery: prospective observational study of the utility of inflammatory markers and determination of pathological levels. Updates Surg. 2021;73(6):2103–2111.
Warschkow R, Steffen T, Beutner U, Müller SA, Schmied BM, Tarantino I. Diagnostic accuracy of C–reactive protein and white blood cell counts in the early detection of inflammatory complications after open resection of colorectal cancer: a retrospective study of 1,187 patients. Int J Colorectal Dis. 2012;27(10):1377.
Stephensen BD, Reid F, Shaikh S, Carroll R, Smith SR, Pockney P; PREDICT Study Group collaborators. C–reactive protein trajectory to predict colorectal anastomotic leak: PREDICT Study. Br J Surg. 2020;107(13):1832–1837.
Ortega–Deballon P, Radais F, Facy O, d'Athis P, Masson D, Charles PE, Cheynel N, Favre JP, Rat P. C–reactive protein is an early predictor of septic complications after elective colorectal surgery. World J Surg. 2010;34(4):808–14.
Messias BA, Botelho RV, Saad SS, Mocchetti ER, Turke KC, Waisberg J. Serum C–reactive protein is a useful marker to exclude anastomotic leakage after colorectal surgery. Sci Rep. 2020;3;10(1):1687.
El Zaher HA, Ghareeb WM, Fouad AM, Madbouly K, Fathy H, Vedin T, Edelhamre M, Emile SH, Faisal M. Role of the triad of procalcitonin, C–reactive protein, and white blood cell count in the prediction of anastomotic leak following colorectal resections. World J Surg Oncol. 2022;12;20(1):33.
Jin D, Chen L. Early prediction of anastomotic leakage after laparoscopic rectal surgery using creactive protein. Medicine (Baltimore). 2021;4;100(22):e26196.
Almeida AB, Faria G, Moreira H, Pinto–de–Sousa J, Correia–da–Silva P, Maia JC. Elevated serum C–reactive protein as a predictive factor for anastomotic leakage in colorectal surgery. Int J Surg. 2012;10(2):87–91.
Waterland P, Ng J, Jones A, Broadley G, Nicol D, Patel H, Pandey S. Using CRP to predict anastomotic leakage after open and laparoscopic colorectal surgery: is there a difference? Int J Colorectal Dis. 2016 Apr;31(4):861–8.
Giaccaglia V, Salvi PF, Antonelli MS, Nigri G, Pirozzi F, Casagranda B, Giacca M, Corcione F, de Manzini N, Balducci G, Ramacciato G. Procalcitonin Reveals Early Dehiscence in Colorectal Surgery: The PREDICS Study. Ann Surg. 2016;263(5):967–72.
Italian ColoRectal Anastomotic Leakage (iCral) Study Group. Anastomotic leakage after elective colorectal surgery: a prospective multicentre observational study on use of the Dutch leakage score, serum procalcitonin and serum C–reactive protein for diagnosis. BJS Open. 2020;4(3):499–507.
Pepys MB, Hirschfield GM. C–reactive protein: a critical update. J Clin Invest. 2003;111(12):1805–12.
Wacker C, Prkno A, Brunkhorst FM, Schlattmann P. Procalcitonin as a diagnostic marker for sepsis: a systematic review and meta–analysis. Lancet Infect Dis. 2013;13(5):426–35.
Hamade B, Huang DT. Procalcitonin: Where Are We Now? Crit Care Clin. 2020;36(1):23–40.
Giaccaglia V, Salvi PF, Cunsolo GV, Sparagna A, Antonelli MS, Nigri G, Balducci G, Ziparo V. Procalcitonin, as an early biomarker of colorectal anastomotic leak, facilitates enhanced recovery after surgery. J Crit Care. 2014;29(4):528–32.
Sparreboom CL, Komen N, Rizopoulos D, Verhaar AP, Dik WA, Wu Z, van Westreenen HL, Doornebosch PG, Dekker JWT, Menon AG, Daams F, Lips D, van Grevenstein WMU, Karsten TM, Bayon Y, Peppelenbosch MP, Wolthuis AM, D'Hoore A, Lange JF. A multicentre cohort study of serum and peritoneal biomarkers to predict anastomotic leakage after rectal cancer resection. Colorectal Dis. 2020;22(1):36–45.
Akobeng AK. Understanding diagnostic tests 2: likelihood ratios, pre– and post–test probabilities and their use in clinical practice. Acta Paediatr. 2007;96(4):487–91.
Halkin A, Reichman J, Schwaber M, Paltiel O, Brezis M. Likelihood ratios: getting diagnostic testing into perspective. QJM. 1998;91(4):247–58.
Su'a B, Tutone S, MacFater W, Barazanchi A, Xia W, Zeng I, Hill AG. Diagnostic accuracy of procalcitonin for the early diagnosis of anastomotic leakage after colorectal surgery: a meta–analysis. ANZ J Surg. 2020;90(5):675–680.
Author information
Authors and Affiliations
Contributions
AA, AS, LC and MS did the literature search. AA and DB formed the study design. Data collection was done by AA, MS and FL. AA, GB, GC and DB analysed the data. AA, PD and DB interpreted the data. AA, GB, and DB wrote the manuscript. All authors critically reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bona, D., Danelli, P., Sozzi, A. et al. C-reactive Protein and Procalcitonin Levels to Predict Anastomotic Leak After Colorectal Surgery: Systematic Review and Meta-analysis. J Gastrointest Surg 27, 166–179 (2023). https://doi.org/10.1007/s11605-022-05473-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-022-05473-z