Abstract
Two new species of Coltricia, C. subcinnamomea and C. subverrucata, are described from China based on both morphological and molecular data. Phylogenetic analyses based on nuc rDNA ITS1-5.8S-ITS2, partial 28S rDNA and partial 18S rDNA, mt partial 12S rDNA, partial sequences of genes for RNA polymerase II subunits 1 and 2, and partial sequences of genes for EF-1α confirmed the generic placement of the two new species. Coltricia subcinnamomea is characterized by stipitate, mesopodal basidiocarps, a slightly shiny pileal surface, 2–3 pores per mm, and broadly ellipsoid to subglobose basidiospores, 8.0–9.5 × 6.2–6.8 μm. Coltricia subverrucata is characterized by stipitate, mesopodal basidiocarps, a faintly concentrically zonate pileal surface with radiating lines, 0.5–2 pores per mm, strongly verrucose hyphae and broadly ellipsoid basidiospores, 7.8–9.0 × 6.0–7.0 μm.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Coltricia Gray, typified by C. perennis (L.) Murrill, and Coltriciella Murrill, typified by C. dependens, are cosmopolitan genera of Hymenochaetales. Most species are terrestrial and a few have been found associated with plant roots, and presumably mycorrhizal (Tedersoo et al. 2007), while others are found on wood (that could only be a physical support in fact).
Coltricia is characterized by poroid and stipitate basidiocarps, a monomitic hyphal system lacking clamp connections and colored, ellipsoid to subglobose, smooth basidiospores (Dai 2010). The complex habitat and exceedingly closed morphology characteristics make this genus easy to be confounded with Coltriciella. Coltriciella differs from Coltricia by the ornamented vs smooth basidiospores (Ryvarden 1991), and molecular data indicated that the two genera were sister clades, forming a single lineage. However, the monophyly of each genus is still unresolved (Wagner and Fischer 2002; Larsson et al. 2006; Tedersoo et al. 2007; Bian and Dai 2017).
The two genera have been extensively studied worldwide; so far, 46 species are recognized in Coltricia, and 13 species in Coltriciella (Burdsall 1969; Gilbertson and Ryvarden 1986; Corner 1991; Masuka and Ryvarden 1993; Rajchenberg and Wright 1998; Núñez and Ryvarden 2000; Aime et al. 2003; Niemelä 2005; Ryvarden et al. 2006; Ryvarden 2007; Gomes-Silva et al. 2009; Baltazar et al. 2010; Dai 2010; Dai et al. 2010; Dai and Li 2012; Zhou and Tedersoo 2012; Valenzuela et al. 2012; Decock 2013; Ryvarden and Melo 2014; Bian and Dai 2015; Bian et al. 2016; Vasco-Palacios 2016; Bian and Dai 2017).
During investigations of Coltricia and Coltriciella in China, several collections of Coltricia were found that could not be assigned to any described species, also forming two distinct clades in multilocus phylogenetic inferences. They are described as Coltricia subcinnamomea and Coltrica subverrucata. An identification key of Coltricia and Coltriciella observed in China is provided.
Materials and methods
Morphological studies
The studied specimens are deposited in the herbarium of the Institute of Microbiology, Beijing Forestry University (BJFC). Macromorphological descriptions were based on field notes and laboratory observations. Microscopic routine used in this study followed Dai (2010). Drawings were made with the aid of a drawing tube. Microscopic measurements were made from slide preparations stained with Cotton Blue and Melzer’s reagent. Spores were measured from section cuts from tubes. In presenting variation in basidiospore size, 5% of measurements were excluded from each end of the range, and are given in parentheses. In the text, the following abbreviations were used: KOH = 5% potassium hydroxide, IKI = Melzer’s reagent, IKI– = neither amyloid nor dextrinoid, CB = Cotton Blue, CB– = acyanophilic, L = mean basidiospore length (arithmetic average of all basidiospores), W = mean basidiospore width (arithmetic average of all basidiospores), Q = variation in the ratios of L/W between specimens studied, n = number of basidiospores measured from given number of specimens. Color terms followed Kornerup and Wanscher (1978).
Molecular phylogeny
The collections and sequences of the two new species and other fungal taxa used in this study are listed in Table 1. Eighteen new sequences were generated for this study and deposited to GenBank. The methods of DNA extraction and amplification in this study followed Chen et al. (2016). The CTAB rapid plant genome extraction kit-DN14 (Aidlab Biotechnologies Co., Ltd., Beijing) was used to extract total genomic DNA from dried specimens according to the manufacturer’s instructions. The primers ITS5/ITS4 for nuc rDNA ITS1-5.8S-ITS2 (ITS), LR0R/LR7 for nuc 28S rDNA (nLSU), NS1/NS4 for nuc 18S rDNA (nSSU), MS1/MS2 for mt 12S rDNA (mtSSU), RPB1-Af/RPB1-Cr for RNA polymerase II subunit 1 gene (RPB1), fRPB2-5f/fRPB2-7cR for RNA polymerase II subunit 2 gene (RPB2), and EF1-983F/EF1-1567F for EF-1α (TEF-1) were used for PCR amplifications (Vilgalys and Hester 1990; White et al. 1990; Matheny 2005). The PCR products were sequenced in Beijing Genomics Institute, China, with the same primers. The newly generated sequences were deposited at GenBank.
For the phylogenetic analyses, the combined dataset of ITS, nLSU, nSSU, mtSSU, RPB1, RPB2 and TEF-1 included 73 sequences representing 31 taxa, in which 23 taxa of Coltricia and 6 taxa of Coltriciella are involved. Fomitiporella chinensis (Pilát) Y.C. Dai, X.H. Ji & Vlasák and Inonotus griseus L.W. Zhou were chosen as outgroup. Sequences were aligned with MAFFT (Katoh and Toh 2008), BioEdit (Hall 1999), and Clustal X (Thompson et al. 1997). Sequence alignments were deposited at TreeBASE (submission ID 25951, www.treebase.org). The best-fit evolutionary model was estimated using MrModeltest 2.3 (Posada and Crandall 1998) as GTR + I + G for the combined dataset.
Maximum parsimony (MP) analyses were applied to the combined dataset. The construction was performed in PAUP* version 4.0b10 (Swofford 2002). All characters were equally weighted, and gaps were treated as missing data. Trees were inferred using the heuristic search option with TBR branch swapping and 1000 random sequence additions. Max-trees was set to 5000, branches of zero length were collapsed, and all parsimonious trees were saved. Clade robustness was assessed by a bootstrap (BT) analysis with 1000 replicates (Felsenstein 1985). Descriptive tree statistics, i.e., tree length (TL), consistency index (CI), retention index (RI), rescaled consistency index (RC), and homoplasy index (HI) were calculated for each maximum parsimonious tree (MPT) generated.
RAxML v.7.2.8 was used to construct maximum likelihood (ML) trees with the GTR + I + G model of site substitution including estimation of Gamma-distributed rate heterogeneity and a proportion of invariant sites (Stamatakis 2006). The branch support was evaluated with a bootstrapping method of 1000 replicates (Hillis and Bull 1993).
BI analyses were calculated with MrBayes3.1.2 with a general time reversible (GTR) model of DNA substitution and a gamma distribution rate variation across sites (Ronquist and Huelsenbeck 2003). Four Markov chains were run for 2 runs from random starting trees for 500,000 generations, and trees were sampled every 100 generations. The first 25% of sampled trees were set as burn-in. A majority rule consensus tree of all remaining trees was calculated. BS (bootstrap support for MP and ML) values and BPPs (Bayesian posterior probabilities for BI) simultaneously not less than 75% and 0.95, respectively, are shown at the nodes.
Results
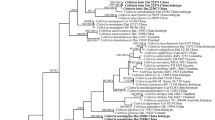
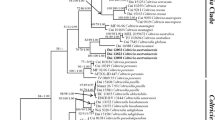
The combined dataset had an aligned length of 7839 characters, of which 4233 characters are constant, 596 are variable and parsimony-uninformative, and 3010 are parsimony-informative. MP analysis yielded four equally most-parsimonious trees (TL = 13,236, CI = 0.456, RI = 0.754, RC = 0.344, HI = 0.544). Bayesian analysis and ML analysis resulted in a similar topology to MP analysis. The ML tree is provided in Fig. 1.
The sampled specimens of C. subcinnamomea and C. subverrucata nested within the “main” Coltricia lineage, forming two well-supported terminal clades (C. subcinnamomea 100% in MP, 100% in ML, 1.00 in BI; C. subverrucata 100% in MP, 100% in ML, 1.00 in BI).
According to the phylogeny analyses, the two new species are distinctive from the other sampled species.
Taxonomy
Coltricia subcinnamomea L.S. Bian & Y.C. Dai, sp. nov. Fig. 2
MycoBank no.: MB 834952
Diagnosis: this species is characterized by stipitate, mesopodal basidiocarps, a slightly shiny pileal surface, 2–3 pores per mm, 11–14-μm-wide contextual hyphae and broadly ellipsoid to subglobose basidiospores, 8.0–9.5 × 6.2–6.8 μm (average).
Holoype: CHINA. Shanxi Province: Qinshui County, Lishan National Nature Reserve, on ground, 24 Aug 2016, Dai 17016 (BJFC 023121).
Etymology: subcinnamomea (Lat.): referring to similarity to Coltricia cinnamomea.
Basidiocarps: annual, centrally stipitate, solitary, soft, and without odor or taste when fresh, becoming soft corky when dry. Pilei more or less circular, flat to infundibuliform, up to 2 cm in diam and 3 mm thick at center. Pileal surface honey-yellow to grayish brown when dry, hirsute in the center with bristles erected, velutinate to glabrous, slightly shiny, distinctly concentrically zonate and sulcate; margin thin and obtuse, curving down upon drying. Pore surface cinnamon-buff to yellowish brown upon drying; pores angular, 2–3 per mm; dissepiments thin, lacerate. Context fawn, leathery, up to 0.5 mm thick. Tubes buff-yellow, distinctly paler than context, fragile or slightly brittle when dry, up to 2 mm long. Stipe reddish brown, corky, finely velutinate, up to 1.5 cm long, 3 mm in diam.
Hyphal structure: hyphal system monomitic; generative hyphae simple septate; tissue darkening but otherwise unchanged in KOH.
Context: contextual hyphae cinnamon-buff to yellowish brown, thick-walled with a wide lumen, rarely branched, frequently simple septate, more or less straight, more or less regularly arranged, 11–14 μm in diam; hyphae in stipe similar to those in context, parallel along the main axe, rarely branched, sometimes collapsed, 10–14 μm in diam.
Tubes: tramal hyphae cinnamon-buff to yellowish brown, thin- to slightly thick-walled with a wide lumen, rarely branched, frequently simple septate, straight, subparallel along the tubes, 5–7 μm in diam. Cystidia and cystidioles absent; basidia broadly clavate, thin-walled, with four sterigmata and a simple septum at the base, 15–20 × 7–9 μm; basidioles similar in shape but slightly smaller.
Spores: basidiospores broadly ellipsoid to subglobose, yellowish, thick-walled, smooth, IKI–, CB–, (6.2–)8.0–9.5 × (5.2–)6.2–6.8(–7.4) μm, L = 8.12 μm, W = 6.47 μm, Q = 1.26–1.32 (n = 60/2).
Additional material (paratype) examined: CHINA. Shanxi Province, Qinshui County, Lishan National Nature Reserve, on ground, 24 Aug 2016, Dai 17022 (BJFC 023127).
Coltricia subverrucata L.S. Bian & Y.C. Dai, sp. nov. Figs. 3 and 4
MycoBank no.: MB 834953.
Diagnosis: this species is characterized by centrally stipitate basidiocarps, an indistinctly concentrically zonate pileal surface with radially aligned lines, 0.5–2 pores per mm, strongly verrucose hyphae and broadly ellipsoid basidiospores, 7.8–9.0 × 6.0–7.0 μm (average).
Holotype: CHINA. Guizhou Province, Lvyang County, Kuankuoshui National Nature Reserve, on ground of angiosperm forest, 27 Sep 2014, Dai 15600 (BJFC 019704).
Etymology: subverrucata (Lat.): referring to similarity to Coltricia verrucata.
Basidiocarps: annual, centrally stipitate, solitary, soft and without odor or taste when fresh, becoming soft corky when dry. Pilei more or less circular, flat to infundibuliform, up to 2.5 cm in diam and 2 mm thick at center. Pileal surface fawn to grayish brown when dry, velutinate, with indistinct concentric zones and radially aligned lines; margin thin and obtuse, curved down when dry. Pore surface honey-yellow to yellowish brown upon drying; pores angular, 0.5–2 per mm; dissepiments thin, lacerate. Context deep olive, leathery, up to 0.5 mm thick. Tubes curry-yellow, distinctly paler than context, fragile or slightly brittle when dry, up to 1.5 mm long. Stipe reddish brown, corky, finely velutinate, up to 2.5 cm long, 2 mm in diam, swollen near the base.
Hyphal structure: hyphal system monomitic; generative hyphae simple septate; tissue darkening but otherwise unchanged in KOH.
Context: contextual hyphae yellowish brown to fawn, thick-walled with a wide lumen, occasionally branched, frequently simple septate, more or less straight, strongly verrucose, more or less regularly arranged, 17–20 μm in diam; hyphae in stipe similar to those in context, parallel along the main axe, rarely branched, sometimes collapsed, 17–20 μm in diam.
Tubes: tramal hyphae honey-yellow to yellowish brown, thin- to slightly thick-walled with a wide lumen, occasionally branched, frequently simple septate, straight, strongly verrucose, subparallel along the tubes, 5–7 μm in diam. Cystidia and cystidioles absent; basidia broadly clavate, thin-walled, with four sterigmata and a simple septum at the base, 20–25 × 7–10 μm; basidioles similar in shape but slightly smaller.
Spores: basidiospores broadly ellipsoid, yellowish, thick-walled, smooth, IKI–, CB–, 7.8–9.0(–9.2) × (5.5–)6.0–7.0(–7.5) μm, L = 8.40 μm, W = 6.40 μm, Q = 1.31–1.32 (n = 60/2).
Additional material (paratype) examined: CHINA. Sichuan Province, Liangshan, Mianning County, Lingshan Temple, on ground of Castanopsis forest, 17 Sep 2012, Dai 12919 (BJFC 013183).
Discussion
Coltricia subcinnamomea has centrally stipitate basidiocarps, large pores, wide contextual hyphae, and broadly ellipsoid to subglobose basidiospores. These features are also shared by C. abieticola Y.C. Dai, C. austrosinensis L.S. Bian & Y.C. Dai and C. wenshanensis L.S. Bian & Y.C. Dai. However, C. abieticola has a duplex stipe and grows in conifer forest (Dai 2010). Coltricia austrosinensis can be distinguished by indistinct concentric zones, pale pore surface (buff-yellow), and distinctly narrower basidiospores (8–10 × 5.5–6.5 μm; Bian et al. 2016). Coltricia wenshanensis has larger basidiocarps (up to 5 cm in diam), larger pores (0.5–2 per mm) and smaller basidiospores (7.5–8 × 6–7; Bian and Dai 2017). Coltricia subcinnamomea shares C. cinnamomea these macromorphological characters, including the slightly shiny pileal surface. And the two species are closely related in the phylogenetic analyses (Fig. 1). However, C. cinnamomea has narrower contextual hyphae (up to 10 μm in diam) and smaller basidiospores (6.9–8.1 × 5.5–6.4 μm, L = 7.7 μm, W = 6.0 μm, Niemelä 2005).
Coltricia subverrucata resembles C. crassa Y.C. Dai and C. montagnei (Fr.) Murrill in having centrally stipitate basidiocarps, large pores and broadly ellipsoid basidiospores. However, C. crassa has thicker basidiocarps (up to 2 cm at the base), paler pore surface (cream to buff-yellow), dendrohyphidia-like hyphae, and larger basidiospores (9–12 × 5.9–7 μm; Dai 2010). Coltricia montagnei differs from C. subverrucata by larger basidiocarps (up to 12 cm wide and 1–2 cm thick), lamellate hymenophore, wavy margin, and distinctly longer but narrower basidiospores (9–12 × 5–6 μm; Murrill 1920). Coltricia subverrucata is phylogenetically related to C. verrucata Aime, T.W. Henkel & Ryvarden (Fig. 1). Both species have verrucose hyphae, but C. verrucata has smaller pores (2–5 per mm), narrower contextual hyphae (8–13 μm) and narrower basidiospores (7–9 × 5–6 μm; Aime et al. 2003).
In the phylogenetic analyses of combined dataset (Fig. 1), Coltricia species are scattered into two lineages, with Coltriciella species placed in between. This questions the monophyly of Coltricia or the pertinence of keeping Coltricia and Coltriciella as distinct generic entities. Coltriciella was originally established for a species with tiny and pendent basidiocarps (Murrill 1904), a feature also shared by Coltricia tsugicola Y.C. Dai & B.K. Cui (Dai and Cui 2006; Vasco-Palacios 2016). With more and more species found, Coltriciella is hard to distinguish from Coltricia but for the ornamented basidiospores. The confounding of the two genera in morphology is also indicated by our phylogenetic analyses.
Key to species of Coltricia and Coltriciella in China
1 Basidiospores smooth .....................................................2
1* Basidiospores finely ornamented or verrucose ..........24
2 Hymenophore more or less concentrically lamellate ...... ..........................................................Coltricia montagnei
2* Hymenophore poroid ....................................................3
3 Basidiocarps pendent .........................Coltricia tsugicola
3* Basidiocarps erect .........................................................4
4 Basidiocarps laterally stipitate ........................................5
4* Basidiocarps eccentrically to centrally stipitate ............8
5 Mature pilei < 0.5 cm in diam ................Coltricia minor
5* Mature pilei > 0.5 cm in diam .......................................6
6 Basidiocarps woody-hard; basidiospores subglobose to globose, < 7 μm long ..............................Coltricia rigida
6* Basidiocarps soft to corky; basidiospores ellipsoid to broadly ellipsoid, > 7 μm long ........................................7
7 Basidiocarps up to 1 cm thick; basidiospores 8–10 × 6– 7 μm .....................................................Coltricia duportii
7* Basidiocarps up to 2 mm thick; basidiospores 7–8 × 5.2–6 μm ...............................................Coltricia lateralis
8 Stipe duplex; on fallen trunk of Abies .............................................................Coltricia abieticola
8* Stipe homogeneous; on ground of mixed forests .........9
9 Pores 0.5–3 per mm ......................................................10
9* Pores 3–6 per mm .......................................................17
10 Hyphae strongly verrucose .........................................11
10* Hyphae smooth .........................................................12
11 Basidiocarps up to 1 cm in diam, pores 2–3 per mm; basidiospores 7.5–9 × 4.8–5 μm .....Coltricia verrucata
11* Basidiocarps up to 2.5 cm in diam, pores 0.5–2 per mm; basidiospores 7.8–9 × 6–7 μm ......................................................Coltricia subverrucata
12 Context thick, up to 12 mm thick; dendrohyphidia- like hyphae present ..............................Coltricia crassa
12* Context thin, up to 1 mm thick; dendrohyphidia- like hyphae absent .......................................................13
13 Basidiocarps eccentrically stipitate, with unpleasant odor ................................................Coltricia macropora
13* Basidiocarps centrally stipitate, without odor ..........14
14 Basidiospores oblong-ellipsoid, < 5.5 μm wide .............................................................Coltricia focicola
14* Basidiospores broadly ellipsoid to subglobose, > 5.5 μm wide ......................................................................15
15 Contextual hyphae > 10 μm wide ................................................Coltricia subcinnamomea
15* Contextual hyphae < 10 μm wide ............................16
16 Stipe more or less uniform; basidiospores 7.5–8.2 × 6–6.8 μm ..................................Coltricia wenshanensis
16* Stipe up to 8 mm diam at the apex; basidiospores 8– 10 × 5.5–6.5 μm .......................Coltricia austrosinensis
17 Stipe bearing numerous spines .....Coltricia strigosipes
17* Stipe smooth or velutinate ........................................18
18 Growing in gymnosperm forests; basidiospores ellipsoid .............................................Coltricia perennis
18* Growing in angiosperm forests; basidiospores broadly ellipsoid to globose ......................................................19
19 Tramal hyphae 3–4 μm in diam .................................20
19* Tramal hyphae 4–9 in diam ......................................21
20 Mature pilei > 1.5 cm in diam; basidiospores 7.8–9 × 5.2–6 μm .....................................Coltricia subperennis
20* Mature pilei < 1.5 cm in diam; basidiospores 6–7 × 4– 5 μm .....................................................Coltricia minima
21 Basidiospores < 4 μm wide .............Coltricia pyrophila
21* Basidiospores > 4 μm wide .......................................22
22 Pileal surface hyphae dichotomously branched ...................................................................Coltricia weii
22* Pileal surface hyphae unbranched .............................23
23 Mature pilei > 2 cm in diam; basidiospores 7–8 × 5.5– 6.5 μm ........................................Coltricia cinnamomea
23* Mature pilei < 2 cm in diam; basidiospores 5.5–6.2 × 4.5–5.8 μm ..........................................Coltricia velutina
24 Basidiocarps resupinate to effused-reflexed ...............25
24* Basidiocarps stipitate ................................................26
25 Hyphae at dissepiment edge mostly moniliform; cystidioles present .....................Coltriciella subglobosa
25* Hyphae at dissepiment edge uniform; cystidioles absent ......................................Coltriciella baoshanensis
26 Basidiocarps pendent ..................................................27
26* Basidiocarps erect .....................................................28
27 Basidiospores 6–9 × 4–5.5 μm ...Coltriciella dependens
27* Basidiospores 9–11.8 × 5–6.2 μm ..........................................Coltriciella pseudodependens
28 Basidiocarps laterally stipitate, pilei fan-shaped ...........................................................Coltriciella pusilla
28* Basidiocarps centrally stipitate, pilei more or less ........................................................................circular 29
29 Basidiospores ellipsoid ...............................................30
29* Basidiospores naviculate or subglobose to globose ......................................................................................31
30 Pores 3–4 per mm; basidiospores 6.5–7.8 × 4.8–5.5 μm ........................................................Coltriciella subpicta
30* Pores 1–2 per mm; basidiospores 8.5–10.2 × 5–6 μm ...................................................Coltriciella oblectabilis
31 Basidiospores naviculate, 8–11 × 5–6.2 μm ...............................................Coltriciella naviculiformis
31* Basidiospores subglobose to globose, 6–7 × 5.8–7 μm .........................................................Coltriciella globosa
References
Aime MC, Henkel TW, Ryvarden L (2003) Studies in neotropical polypores 15: new and interesting species from Guyana. Mycologia 95:614–619. https://doi.org/10.2307/3761937
Baltazar JM, Ryvarden L, Gibertoni TB (2010) The genus Coltricia in Brazil: new records and two new species. Mycologia 102:1253–1262. https://doi.org/10.3852/09-227
Bian LS, Dai YC (2015) Coltriciella globosa and C. pseudodependens spp. nov. (Hymenochaetales) from southern China based on morphological and molecular characters. Mycoscience 56:190–197. https://doi.org/10.1016/j.myc.2014.06.001
Bian LS, Dai YC (2017) Morphological and molecular evidence for three new species of Coltricia (Hymenochaetaceae, Basidiomycota) from southern China. Mycologia 109:64–74. https://doi.org/10.1080/00275514.2017.1286571
Bian LS, Wu F, Dai YC (2016) Two new species of Coltricia (Hymenochaetaceae, Basidiomycota) from southern China based on the evidence from morphology and DNA sequence data. Mycol Prog 15:27. https://doi.org/10.1007/s11557-016-1173-0
Burdsall HH (1969) A new polypore from the Eastern United States. Mycologia 61:647–651. https://doi.org/10.2307/3757257
Chen JJ, Cui BK, Dai YC (2016) Global diversity and molecular systematics of Wrightoporia s.l. (Russulales, Basidiomycota). Persoonia 37:21–36. https://doi.org/10.3767/003158516x689666
Corner EJH (1991) Ad Polyporaceas 7. Nova Hedwigia 101:1–175
Dai YC (2010) Hymenochaetaceae (Basidiomycota) in China. Fungal Divers 45:131–343. https://doi.org/10.1007/s13225-010-0066-9
Dai YC, Cui BK (2006) Two new species of Hymenochaetaceae from eastern China. Mycotaxon 94:341–347. https://doi.org/10.1016/S0014-5793(01)02483-8
Dai YC, Li HJ (2012) Type studies on Coltricia and Coltriciella described by E. J H Corner from Southeast Asia Mycoscience 53:337–346. https://doi.org/10.1007/s10267-011-0174-8
Dai YC, Yuan HS, Cui BK (2010) Coltricia (Basidiomycota, Hymenochaetaceae) in China. Sydowia 62:11–21
Decock C (2013) Coltricia oboensis sp. nov. from the high elevation cloud forest of São Tomé. Cryptogam Mycol 34:175–181. https://doi.org/10.7872/crym.v34.iss2.2013.175
Felsenstein J (1985) Confidence intervals on phylogenetics: an approach using bootstrap. Evolution 39:783–791
Gilbertson RL, Ryvarden L (1986) North American polypores 1. Abortiporus–Lindtneria, Fungiflora, Oslo
Gomes-Silva AC, Ryvarden L, Gibertoni TB (2009) New and interesting species of Hymenochaetaceae from the Brazilian Amazonia. Mycol Prog 8:273–279. https://doi.org/10.1007/s11557-009-0606-4
Hall TA (1999) Bioedit: a user-friendly biological sequence alignment editor and analysis program for windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Hillis DM, Bull JJ (1993) An empirical test of bootstrapping as a method for assessing confidence in phylogenetic analysis. Syst Biol 42:182–192. https://doi.org/10.2307/2992540
Katoh K, Toh H (2008) Improved accuracy of multiple ncRNA alignment by incorporating structural information into a MAFFT-based framework. BMC Bioinformatics 9:212. https://doi.org/10.1186/1471-2105-9-212
Kornerup A, Wanscher JH (1978) Methuen handbook of colour, 3rd edn. Eyre Methuen, London
Larsson K-H, Parmasto E, Fischer M, Langer E, Nakasone KK, Redhead SA (2006) Hymenochaetales: a molecular phylogeny for the hymenochaetoid clade. Mycologia 98:926–936. https://doi.org/10.1080/15572536.2006.11832622
Masuka A, Ryvarden L (1993) Two new polypores from Malawi. Mycologia Helvetica 5:143–148
Matheny PB (2005) Improving phylogenetic inference of mushrooms with RPBl and RPB2 nucleotide sequences. Mol Phylogen Evol 35:1–20. https://doi.org/10.1016/j.ympev.2004.11.014
Murrill WA (1904) The Polyporaceae of North America: VII. The genera Hexagona, Grifola, Romellia, Coltricia and Coltriciella. Bull Torrey B Club 31:325–348. https://doi.org/10.2307/2478798
Murrill WA (1920) Corrections and additions to the polypores of temperate North America. Mycologia 12:6–24. https://doi.org/10.2307/3753482
Niemelä T (2005) Polypores, lignicolous fungi. Norrlinia 13:1–320
Núñez M, Ryvarden L (2000) East Asian polypores 1. Ganodermataceae and Hymenochaetaceae. Synop Fungorum 13:1–168
Posada D, Crandall KA (1998) Modeltest: testing the model of DNA substitution. Bioinformatics 14:817–818. https://doi.org/10.1093/bioinformatics/14.9.817
Rajchenberg M, Wright JE (1998) Two interesting polypore species (Hymenochaetales) from Argentina. Folia Cryptog Estonica 33:119–122
Ronquist F, Huelsenbeck JP (2003) MRBAYES 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574. https://doi.org/10.1093/bioinformatics/btg180
Ryvarden L (1991) Genera of polypores. Nomenclature and taxonomy. Synop Fungorum 5:1–363
Ryvarden L (2007) Studies in Neotropical polypores 23. New and interesting wood-inhabiting fungi from Belize. Synop Fungorum 23:32–50
Ryvarden L, Melo I (2014) Poroid fungi of Europe. Synop Fungorum 31:1–455
Ryvarden L, Krisai-Greilhuber I, Hausknecht A (2006) Coltricia grandispora and Tyromyces vitellinus, two new polypores. Österr Z Pilzk 15:143–147
Stamatakis A (2006) RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 22:2688–2690. https://doi.org/10.1093/bioinformatics/btl446
Swofford DL (2002) PAUP*: phylogenetic analysis using parsimony (*and other methods), version 4.0b10. Sinauer Associates, Sunderland, Massachusetts
Tedersoo L, Suvi T, Beaver K, Saar I (2007) Ectomycorrhizas of Coltricia and Coltriciella (Hymenochaetales, Basidiomycota) on Caesalpiniaceae, Dipterocarpaceae and Myrtaceae in Seychelles. Mycol Prog 6:101–107. https://doi.org/10.1007/s11557-007-0530-4
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The Clustal_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882. https://doi.org/10.1093/nar/25.24.4876
Valenzuela R, Raymundo T, Cifuentes J, Esqueda M, Amalfi M, Decock C (2012) Coltriciella sonorensis sp. nov. (Basidiomycota, Hymenochaetales) from Mexico: evidence from morphology and DNA sequence data. Mycol Prog 11:181–189. https://doi.org/10.1007/s11557-011-0740-7
Vasco-Palacios AM (2016) Ectomycorrhizal fungi in Amazonian tropical forests in Colombia. geboren op 13 januari te Bogotá, Colombia
Vilgalys R, Hester M (1990) Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J Bacteriol 172:4238–4246. https://doi.org/10.1128/jb.172.8.4238-4246.1990
Wagner T, Fischer M (2002) Proceedings towards a natural classification of the worldwide taxa Phellinus s.l. and Inonotus s.l., and phylogenetic relationships of allied genera. Mycologia 94:998–1016. https://doi.org/10.2307/3761866
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press, San Diego
Zhou LW, Tedersoo L (2012) Coltricia australica sp. nov. (Hymenochaetales, Basidiomycota) from Australia. Mycotaxon 122:123–128. https://doi.org/10.5248/122.123
Funding
The research was supported by the National Natural Science Foundation of China (Project No. 31800018).
Author information
Authors and Affiliations
Corresponding author
Additional information
Section Editor: Marc Stadler
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bian, LS., Dai, YC. Molecular phylogeny and morphology reveal two new species of Coltricia (Hymenochaetaceae Basidiomycota) from China. Mycol Progress 19, 657–666 (2020). https://doi.org/10.1007/s11557-020-01583-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11557-020-01583-7