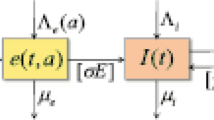Abstract
Demographic structure and latent phenomenon are two essential factors determining the rate of tuberculosis transmission. However, only a few mathematical models considered age structure coupling with disease stages of infectious individuals. This paper develops a system of delay partial differential equations to model tuberculosis transmission in a heterogeneous population. The system considers demographic structure coupling with the continuous development of disease stage, which is crucial for studying how aging affects tuberculosis dynamics and disease progression. Here, we determine the basic reproduction number, and several numerical simulations are used to investigate the influence of various progression rates on tuberculosis dynamics. Our results support that the aging effect on the disease progression rate contributes to tuberculosis permanence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Tuberculosis (TB), a disease caused by Mycobacterium tuberculosis (Mtb) (Flynn and Chan 2001), has long been considered as a cause of mortality and is one of the top 10 diseases of mortality worldwide (World Health Organization 2018). Around 1.3 million died because of tuberculosis, and 10.0 million developed tuberculosis in 2017 (World Health Organization 2018). Patients with smear-positive will generally infect ten individuals annually and over 20 people during the natural period of the disease until death (Styblo 1991). In Hong Kong, tuberculosis dynamics have declined in recent years, but it persists (Fig. 1a) and causes various mortality in different age groups (Fig. 1b). The transmission dynamics of tuberculosis is a complicated process which includes latent phenomenon (Parrish et al. 1998; Selwyn et al. 1989; Vynnycky and Fine 2000; Anderson and Trewhella 1985; Ma et al. 2017), aging effect (Marais et al. 2004; Chan-Yeung et al. 2002; Mossong et al. 2008; Castillo-Chavez and Feng 1998), exogenous reinfection issue (Feng et al. 2000; Vynnycky and Fine 1997; Cohen et al. 2006; Kasaie et al. 2014) and several stochastic factors (Liu et al. 2018).
Tuberculosis tendency in Hong Kong. a Notification and death rates of tuberculosis in Hong Kong, 1947–2016 (data from Department of Health of the Government of the Hong Kong (2018)). b Tuberculosis induced mortality in Hong Kong for various age groups [data from Department of Health of the Government of the Hong Kong (2018)]
Latent tuberculosis is the clinical syndrome that the immune system forces pathogens into a quiescent state after infection (Parrish et al. 1998; Barry et al. 2009; Houben and Dodd 2016). Through adapting the immune system of infected hosts, Mycobacterium tuberculosis-latent bacilli will break out several years later until encountering a favorable environment and further reactive to contribute tuberculosis break out (Cardona and Ruiz-Manzano 2004; Cardona 2016). Latent phenomenon contributes pressure in tuberculosis control (Selwyn et al. 1989), and the effect was studied in several susceptible-infected-infectious models (Castillo-Chavez and Feng 1998; Feng et al. 2000; Waaler et al. 1962; Jabbari et al. 2016; Kuniya 2011; Kapitanov 2015). This latent phenomenon was considered as the source of delay in tuberculosis control. Many factors (World Health Organization 2006; Cai et al. 2015) contribute to the delay phenomenon, and the longest total delay is larger than 120 days (Storla et al. 2008), which results in difficulty for tuberculosis diagnosis and treatment.
Demographic dynamics is another significant factor affecting tuberculosis transmission as individuals in various age groups may have different immune response levels to Mtb and various primary infection rates (Marais et al. 2004; Arregui et al. 2018). The probability of pulmonary disease for infants (less than one years old) is 30–40\(\%\), and then decreases (\(10\%\) for 1–2 years old, \(5\%\) for 2–5 years old, \(2\%\) for 5–10 years old), and increases for larger than ten years old (10–20\(\%\)) (Marais et al. 2004), which indirectly indicates the complicated relation between tuberculosis transmission and individuals’ age. In Hong Kong, longevity and the high rate of tuberculosis in the elderly are the significant factors for the persistence of tuberculosis, stated by Chan-Yeung et al. (2002). After infection by Mtb, extensive symptoms result in many concrete stages. In Aagaard et al. (2011), Aagaard et al. studied various vaccination methods for different stages to determine the efficient protection strategy before and after exposure to tuberculosis.
Mathematical and computational model can be used to study all these phenomenon with to provide new hypotheses and design therapeutic approaches to TB (Kirschner et al. 2017). Recently, Renardy and Kirschner (2020) considered a network-based model to study tuberculosis transmission in school, household and workplace. They obtained that the disease prevalence is sensitive to the contact weight assigned to transmissions between casual contacts. This result suggests a direction to prevent the spread of TB. Arregui et al. (2018) applied a data-driven model with demographic dynamics to demonstrate a greater burden level for those areas with heavy tuberculosis nowadays. The study in Okuonghae and Omosigho (2011) considered susceptible by two stages and infectious population by three stages to investigate how various stages affect tuberculosis transmission in the population.
Infection age is often utilized in mathematical systems for tuberculosis transmission (Castillo-Chavez and Feng 1998; Feng et al. 2002), but only a few mathematical models consider age structure coupling with disease stages of infectious individuals. Instead of using latent state in mathematical model (Feng et al. 2001), we utilize distributed delay to reflect latent phenomenon after infection as the distributed delay can capture the delay difference among individuals. In this paper, we develop a mathematical model for tuberculosis transmission, which involves age structure for all individuals and age-specific disease progression for infectious populations. Our model considers demographic structure coupling with the continuous development of disease stage, which is a crucial component for studying tuberculosis transmission. In this paper, we apply the local stability analysis to determine the basic reproduction number, demonstrating that the age-specific progression rate may affect tuberculosis permanence. Several numerical simulations are applied to investigate the influence of various progression rates on tuberculosis dynamics.
2 Mathematical Model
In our model, we differentiate the population into two groups, susceptible and infectious individuals, with demographic structure and multiple disease stages, and a delay term applied to represent the latent state.
Here, we let a and s be the individual’s age and the disease stage, respectively. To represent two types of population distribution at time t, we define the following two variables: S(t, a) for susceptible individuals, and I(t, a, s) for infectious individuals. We also consider several age-dependent parameters for modeling tuberculosis transmission as individuals of different ages may have different immune response levels and various transmission patterns after infection (Arregui et al. 2018; Makinodan and Kay 1980). The mathematical model for tuberculosis transmission in the population is based on the schematic diagram shown in Fig. 2.
Schematic diagram representing how tuberculosis are transmitted within the population. Population in the system is recruited through immigration and natural birth. Susceptible individuals (S(t, a)) are infected by infectious individuals (I(t, a, s)) and become new infectious individuals and this process exists with delay phenomenon (Makinodan and Kay 1980; World Health Organization 2006; Storla et al. 2008). Infectious individuals in I(t, a, s) will move back to susceptible state S(t, a) after effective treatment and may be reinfected later (Kasaie et al. 2014). The individuals in the population have natural mortality and tuberculosis-induced mortality (World Health Organization 2018)
2.1 Susceptible Population
Here, we consider the interaction between susceptible individuals and infectious individuals to explain the susceptible population dynamics. Newly infected individuals in the population at time t depend on the number of effective interactions between susceptible and infectious individuals (Mossong et al. 2008) modeled by the following term
where \(a_{\max }\) and \(s_{\max }\) are the maximum survival age and stage, respectively. If an individual’s age is larger than \(a_{\max }\) or the disease stage is over \(s_{\max }\), we consider that the individual is dead or removed from the system. The term \(T(a,a',s)\) is the infection rate for a susceptible individual with age a infected by an infectious individual with age \(a'\) and stage s at time t. After successful treatment, an infectious individual will be recovered back to susceptible state (Feng et al. 2001; Castillo-Chavez and Song 2004) with the recovery rate \(\gamma (a,s)\) for an individual with age a and stage s and the process is modeled by the term
In addition, immigration, natural birth and death will be considered with rates \(\beta (t,a)\), \(\beta _0(t)\) and \(\delta _S(a)S(t,a)\), respectively. Hence, the following equation can be utilized to describe the dynamics of S(t, a):
with the boundary condition \(S(t,0) = \beta _0(t)\) to represent the number of newborn in the population.
2.2 Infectious Population
After tuberculosis infection, individuals will either enter into a state that has no clinic symptom and noninfectious (World Health Organization 2018; Parrish et al. 1998; Blower et al. 1995) or directly become infectious (direct progression) (Okuonghae and Omosigho 2011; Blower et al. 1995) represented by I(t, a, s) with stage s. Reactivation (Cohen et al. 2006) and exogenous refection (Cohen et al. 2006; Kasaie et al. 2014) will transform quiescent individuals into infectious group again after several years, which also contributes to difficulty in tuberculosis control (World Health Organization 2018). Here, we utilize the delay phenomenon (World Health Organization 2006; Storla et al. 2008; Cai et al. 2015) to reflect latent phenomenon since various individuals may have different immune response levels after infection. Hence, the number of total new infectious individuals is modeled by the term
In the delay period, individuals are still under the mortality process from \(a-\tau \) to a, which implies the term \(\mathrm {e}^{-\int ^a_{a-\tau }\delta _S(\mu )\mathrm {d}\mu }\). Moreover, we introduce the delay kernel function K(s) to describe the distribution of the influence under delay, which satisfies
Here, \(\bar{\tau }\) is known as the average time delay for the kernel.
In biological models, the delay kernel with Gamma distribution is often utilized as follows (Macdonald 1978)
where \(\alpha >0\) is known as exponentially fading memory, and the integer \(n\ge 0\). Here, we consider the weak kernel function (\(n=0\) in the above formula), that is,
After infected by Mtb, individuals will have an extensive spectrum of symptoms (World Health Organization 2018) since the antigens, such as Rv3407, are expressed in various states of tuberculosis infection (Ma et al. 2017). We apply the following term to represent the progression of disease stage:
where v(a) is the age-dependent progression rate of disease stage. We assume that the progression rate v(a) is a non-decreasing function. Without loss of generality, we further assume that \(0\le v(a)\le 1\). Individuals flowed out from S(t, a) will be counted as the inflow of I(t, a, 0) at stage 0 and all newborn will be susceptible. Hence, the boundary conditions are
There are two ways of outflow from an infectious state: recovery after successful treatment and mortality. Patients with various symptoms need different treatment methods (Nahid and Hopewell 2017), and hence we use the following terms to model recovery and mortality processes, respectively,
where \(\gamma (a,s)\) and \(\delta _I(a,s)\) depends on age and disease stage. Therefore, we have the following equation for modeling the dynamics of infectious individuals:
2.3 Governing Equations
On the basis of the discussion above and combining all the equations, we have
for \(a\in (0,a_{\max })\) and \(s\in (0,s_{\max })\). Initial and boundary conditions are as follows
Here, \(S_0(a)\) and \(I_0(a,s)\) are the initial conditions for susceptible and infectious classes, respectively. In our simulations, the values of the initial conditions are based on the data obtained in Hong Kong (Department of Health of the Government of the Hong Kong 2018; Census and Statistics Department of the Government of the Hong Kong SAR 2018; Census and Statistics Department 2018). The biological meanings of the parameters are summarized in Table 1.
3 Infection-free Equilibrium
For studying the equilibria, we assume that there exist \(\hat{\beta }(a)\) and \(\hat{\beta }_0\) such that
that is, the immigration rate \(\beta (t,a)\) only depends on age a and the birth rate \(\beta _0(t)\) is a constant when time approaches infinity. The equilibria of system (1) satisfy following equations
and
For the infection-free equilibrium \(E_0=(S(a),0)\), we have the following equation
then we obtain that
for \(a\ge 0\). Without loss of generality, we assume \(\hat{S}(a)=0\) when \(a<0\). In the following subsection, we will discuss the local stability of \(E_0\).
3.1 Stability Analysis
To investigate the local stability of \(E_0 = (\hat{S}(a),0)\), we linearized model (1) at the equilibrium first. Consider the small perturbations around equilibrium as follows
where the perturbation amplitude \(\epsilon \ll 1\). With the assumption \(T(a,a',s) = T_1(a)T_2(a',s)\), we obtain the following linearized system at \(E_0 = (\hat{S}(a),0)\):
and
where
We first calculate the second equation in (5), and rewrite this equation into following form
For the partial differential equation above, the characteristic curve passing through \((a_0,0)\) is
where \(C=f(a_0)\). Since v is a positive function, f is increasing and the curve and the inverse of f are well defined. By the boundary condition \(\bar{I}(0,s) = 0\) and integrating along the characteristic curves for \(C<0\), we obtain
if \(C\ge 0\), that is, \(f(a)\ge s>0\), we have
where
Equation (6) and the expression of \(\bar{I}(a,s)\) with the boundary conditions imply that
which yields the following equation
Equation (8) is the characteristic equation of the system (1) at the infection-free equilibrium \(E_0\). Through investigating the behaviors of \(g(\lambda )\), we can obtain the condition of local stability for the boundary equilibrium \(E_0\) as the following theorem.
Theorem 1
Define
where \(x(a',s) = f^{-1}(f(a')-s)\) which satisfies \( x=f^{-1}(f(a')-s)<a'\) as f is an increasing function; \(\mathscr {F}(\eta , a', s)\) is defined as (7). The infection-free equilibrium \(E_0 = (\hat{S}(a),0)\) for system (1) is locally asymptotically stable if \(\mathcal {R}_0<1\) and unstable if \(\mathcal {R}_0>1\).
Proof
From (8), it is easy to show that \(g(\lambda )\) is a continuous function which is monotone decreasing. In addition,
and
If \(\mathcal {R}_0=g(0)<1\), the characteristic equation \(g(\lambda ) = 1\) exists at least one negative solution. Furthermore, let \(\hat{\lambda }\) be a negative solution of \(g(\lambda ) = 1\), and for any complex solution \(\lambda = z_1+iz_2\ (z_1, z_2\in R)\) of \(g(\lambda ) = 1\), we can check that
which leads to \(0>\hat{\lambda }\ge z_1\) as g is a decreasing function. Therefore, we proved that the infection-free equilibrium \(E_0\) is locally asymptotically stable if \(\mathcal {R}_0<1\). On the contrary, if \(\mathcal {R}_0 = g(0)>1\), there exists a positive real solution for equation (8), and it implies that the infection-free equilibrium \(E_0\) is unstable. \(\square \)
3.2 Biological Meaning of \(\mathcal {R}_0\)
We can rewrite equation (9) into the following expression
In the above equation, the term \(\exp \left( -\int ^{a'}_x\mathscr {F}(\eta ,a',s)\mathrm {d} \eta \right) \) represents the probability that one individual who is infectious at age x and still stays in infectious state at age \(a'\). Hence,
is the number of contacts for an infectious individual who survives from age x to age \(a'\) and stays in stage s. Meanwhile, \(\hat{S}(x-\tau )\) is the total susceptible individual with age \(x-\tau \), and \(\exp \left( -\int ^{x}_{x-\tau }\delta _S(\mu )\mathrm {d}\mu \right) \) stands for the probability that one susceptible individual survives at age \(x-\tau \) and is still survived at age x. Therefore,
represents the number of susceptible individuals contacting one infectious individual with age \(x-\tau \) and surviving to age x. Here, \(T_1(x-\tau )\) is the infectiousness rate for susceptible at age \(x-\tau \). On the basis of the above discussion, the term
is the total number of new contact individuals between susceptible with age \(x-\tau \) and infectious with age \(a'\) and stage s. These individuals will become infectious after time period \(\tau \in (0,\infty )\), and \(\alpha \mathrm {e}^{-\alpha \tau }\) is the influence for every point \(\tau \) to current time t. Overall, we showed that \(\mathcal {R}_0\) represents the basic reproduction number for system (1).
4 Numerical Simulations
The disease progression rate is critical for altering the transmission dynamics of tuberculosis in populations since the patients with severe symptoms may easily transmit disease to others. In this section, we will use numerical simulations to verify this observation. First, we will estimate the parameters for the simulations. Then, we will apply two types of functions to model disease progression rate, constant and age-specific linear functions. The latter one can capture the aging effect for tuberculosis transmission. The detailed numerical scheme can be found in Appendix.
4.1 Parameter Estimation
The natural mortality rate in system (1) is referred from Væth et al. (2018) and we utilize the following form
Moreover, for the tuberculosis induced death rate in system (1), we can fit it based on the information and data from the Census and Statistics Department of Hong Kong and Department of Health of Hong Kong (Department of Health of the Government of the Hong Kong 2018) and obtained the age-specific function for \(\delta _{I1}(a)\) listed in Table 2.
Here, we further suppose that the mortality rate \(\delta _I(a,s) = \delta _{I1}(a)\times \delta _{I2}(s)\), where \(\delta _{I1}(a)\) is represented in Table 2. We assume that the mortality rate may increase when the disease stage s is increasing, so \(\delta _{I2}(s)\) is assumed to be an increasing function, defined as
where the maximum stage \(s_{\max }\) is 1. To match the results we observed in the data, we take \(s_1= 0.1\), and \(s_2=0.7\).
According to Census and Statistics Department of Hong Kong (Census and Statistics Department 2001, 2018) from 2001 to 2016, we have the data of immigration and birth. We use these data to estimate the birth rate and immigration rate and rescale them to make the steady state (4) to be around 100. The stage factor s in the infection rate matrix \(T(a,a',s)\) is assumed with the following function:
The function \(T_1(a)\) will be estimated based on the contact rates in Arregui et al. (2018) for the numerical simulations later. We assume that the function \(T_s(s)\) is an increasing function as
We assume that the age- and stage-specific recovery rate \(\gamma (a,s)=\gamma _a(a)\gamma _s(s)\). The function \(\gamma _a(a)\) will be estimated based on the data from the Department of Health of Hong Kong Department of Health of the Government of the Hong Kong (2018), and the function \(\gamma _s(s)\) will be defined as
which is increasing when \(0<s<s_1\) and decreasing when \(s_1<s<s_2\).
The progression rate v(a) in system (1) plays an important role on the dynamics of infected population. The rate of disease progression is a key component for studying the disease transmission in the system because the patients with serious symptoms may easily transmit disease than the patients with light symptoms. In the next section, we will apply different kinds of functions to model the effect of progression rate v(a).
For the weak delay kernel function \(K(s) = \alpha \mathrm {e}^{-\alpha s}\), we have the average delay as follows
which implies that \(\alpha \) determines the average delay time from infected to infectious individuals. When the value of \(\alpha \) is small, the average delay time \(\frac{1}{\alpha }\) will be large, which reflects a long period from infected individuals to infectious because of latent phenomenon (World Health Organization 2018; Parrish et al. 1998; Blower et al. 1995) or self-immune system (Makinodan and Kay 1980). If the value of \(\alpha \) is large, then the average delay time will be small, which means that the susceptible individuals will immediately become infectious after contacting infectious individuals through direct progression (Okuonghae and Omosigho 2011; Blower et al. 1995). Consequently, the larger value of \(\alpha \) has a positive effect on tuberculosis transmission because of the short average delay period. World health organization (World Health Organization 2006) stated that the average delay period from one and half months to four months. Storla et al. (2008) also pointed out that the average delay time within the range from 60 to 90 days and the longest delay time is larger than 120 days (126 in high endemic countries). Therefore, the average delay time here is assumed as around 120 days. In the following numerical simulations, we set \(\alpha \approx 365/120\) per year.
We set the initial values for susceptible individuals (S(0, a)) as the steady state solution (4) and the infectious individuals (I(0, a, s)) is \(S(0,a)\times 10^{-4}\) for \(s=0\) and zero for \(s>0\).
4.2 Constant Progression Rate
Here, we simulate tuberculosis dynamics with the constant progression rate \(v(a) = v_0\). Based on the data in Okuonghae and Omosigho (2011); Blower et al. (1995), we set \(v_0 = 4.5\times 10^{-4}(a_{\max }/2)\), where \(a_{\max } = 100\). From (9), we obtain the value of reproduction number \(\mathcal {R}_0\approx 0.42<1\) and the solution of system A.10 will tend to the infection-free equilibrium, which means that disease will be eradicated at the end.
Figure 3 shows the dynamics of susceptible individuals \(\mathbf {S}(t)\) and infectious individuals \(\mathbf {I}(t)\) as time t increases, where
In Fig. 3, the susceptible individuals \(\mathbf {S}(t)\) slightly decreases and the number of infectious individuals \(\mathbf {I}(t)\) increases at the beginning. But, after certain time, \(\mathbf {I}(t)\) declines to zero and \(\mathbf {S}(t)\) increases back to the steady state. The simulation demonstrates the extinction of tuberculosis in the population if \(\mathcal {R}_0<1\). In this case, a system with a relatively low and age-independent progression rate does not have many infectious individuals with severe symptoms who transmit disease efficiently in the population. Combined with the effect of immune response to Mycobacterium tuberculosis, the disease will be eradicated with effective control strategies in this case.
The simulations in Fig. 4 display the population distribution of the infectious individuals I(t, a, s) at different times. Figure 4\((i)\) is the population distribution of infectious individuals at the initial time \(t=0\), and infectious individuals distribute in around all age groups with \(s=0\). From the result \(\mathcal {R}_0\approx 0.42<1\), we predict that the number of infectious individuals will decrease [Figs. 3 and 4(\(iii\))] to zero as time increases. Simulations in Figs. 3 and 4(\(iii\), \(iv\)) show that tuberculosis in the population will be controlled at the end, and Fig. 4(\(iv\)) is the population distribution of the infectious individuals under successful treatment at the end of the longtime simulation. The number of infectious individuals in the elderly age groups is much more than the number of infectious individuals in the other age groups since the elderly individuals have weaker immune systems compared with the other age groups (Makinodan and Kay 1980).
Now we consider a larger progression rate \(v_0 = 9\times 10^{-4}(a_{\max }/2)\) and simulate the corresponding disease dynamics. From (9), we obtain that \(\mathcal {R}_0\approx 1.30>1\), which implies that an outbreak of tuberculosis happens in the population. The numerical simulations in Fig. 5 show that the number of infectious individuals \(\mathbf {I}(t)\) increases again after a drop. This result is different from the simulation in the former example. Figure 6 displays the population distribution of infectious individuals I(t, a, s). The number of infectious individuals with elderly age groups keeps increasing (Figs. 6(\(i\))–(\(iii\))), which leads to the persistence of disease in the population. Compared with Fig. 4(\(iv\)), Fig. 6(\(iv\)) shows that Infections in the older population are in the region with large value of s. This observation is consistent with the results in Chan-Yeung et al. (2002); Department of Health of the Government of the Hong Kong (2018) that the high notification rate of tuberculosis in the elderly age groups contributes to the persistence of tuberculosis in the population.
4.3 Age-specific Progression Rate
In this subsection, we introduce the aging factor to the disease progression rate and simulate the tuberculosis transmission with an age-specific progression rate \(v(a) = ca\). First, we take \(c = 4.5\times 10^{-4}\). In this case, we obtain that \(\mathcal {R}_0\approx 0.84<1\), which implies that the infection-free equilibrium is stable based on Theorem 1. Figures 7 and 8 are the numerical simulations of disease dynamics for this case.
Figure 7 displays that the number of infectious individuals \(\mathbf {I}(t)\) decreases to zero as time t increases, which demonstrates that the disease will die out at the end. The temporal dynamics of \(\mathbf {S}(t)\) and \(\mathbf {I}(t)\) in Fig. 7 are similar with that in Fig. 3 but the speed of the decrease of \(\mathbf {I}(t)\) is slower in Fig. 7.
Figure 8 is the population distribution of the infectious individuals for various time t. As time t increases, the number of infectious individuals tends to zero with a low level of \(10^{-6}\) at the end [Figure 8(\(iv\))]. It reflects the extinction of disease in the population. Compared with Fig. 4, Fig. 8 shows that the elderly age group is in an area with larger s due to the aging effect in the disease progression rate (Fig. 9).
Next, we increase the coefficient to \(c = 9\times 10^{-4}\). In this case, the value of reproduction number will increase to \(\mathcal {R}_0\approx 1.99>1\), which means that the infection-free equilibrium is unstable. Figure 10 shows that the number of infectious individuals \(\mathbf {I}(t)\) increases first and declines later. When \(t>15\), \(\mathbf {I}(t)\) increases rapidly as shown in Fig. 10.
Figure 11 is the population distribution of the infectious individuals at various times. The simulations in Fig. 11 display that the number of infectious individuals in the elderly age groups does not tend to zero and infectious individuals are mainly with high levels of a and s. This result is similar to Fig. 6 but the increased speed of I(t) is faster in Fig. 11.
The disease transmission may be underestimated from our simulations when we assume that the progression rate is independent of age. Now we compare the simulations of the models with the constant progression rate and age-specific progression rate. We consider the constant progression rate \(v(a) = v_0= 6\times 10^{-4}(a_{\max }/2)\) and the age-specific progression rate \(v(a) = 6\times 10^{-4}a\), which have same average value. Figure 12 displays the numbers of susceptible individuals and infectious individuals under two types of progression rates. In Fig. 12, the number of susceptible individuals with the constant progression rate is slightly more than the number of susceptible individuals with the age-specific progression rate. On the contrary, Fig. 12 shows that the number of infectious individuals with the constant progression rate is less than that with the age-specific progression rate after \(t>5\). We further observe that the number of patients under the constant progression rate is six times less than the number of patients with the age-specific progression rate after \(t>40\). As shown in the former simulations and Chan-Yeung et al. (2002); Department of Health of the Government of the Hong Kong (2018), the elderly age groups are the major population that contributes to the permanence of disease in the population. Using the constant progression rate may not capture this point, and the age-specific progression rate can accurately model the aging effect in disease transmission. Consequently, considering the progression rate with the aging factor will be more feasible for simulating the transmission dynamics of tuberculosis as it will avoid the underestimation in the number of infected population.
Figure 13 validates the increase of the basic reproduction number \(\mathcal {R}_0\) for the age-specific progression rate, \(3c\times 10^{-4}a\), and the constant progression rate, \(3c\times 10^{-4} \frac{a_{\max }}{2}\). The value of \(\mathcal {R}_0\) with the age-specific progression rate is greater than the case with constant progression rate as shown in Fig. 13. Consequently, as the value of c increases, tuberculosis may transmit faster with the age-specific progression rate.
5 Conclusion
In this paper, we have developed a tuberculosis system with demographic structure and delay effect after infection. The model involved a continuous disease progression process with an age-specific progression rate. In this work, stability analysis for the infection-free equilibrium was applied to determine the formula of the basic reproduction number, which is essential to study the condition of disease outbreak. During the transmission process of tuberculosis, the disease progression may be enhanced by aging. The formula of the basic reproduction number (9) provides a tool to understand how the age-specific progression affects the disease transmission dynamics through changing the stability of infection-free equilibrium. In our simulations, we observed that the age-specific progression rate would accelerate tuberculosis transmission. In fact, if the progression rate rapidly increases in the elderly population, the number of infectious individuals with severe symptoms will increase at the same time, which further contributes to more new patients in the population.
The system with a constant disease progression rate cannot capture the aging effect on the disease progression. Consequently, considering the disease progression rate with aging effect will avoid underestimating the number of infected population during prediction since infectious elderlies may quickly become patients with severe symptoms. Individuals with serious symptoms can easily infect susceptible individuals, which leads to a higher value of \(\mathcal {R}_0\) and further leads to more infectious individuals in the whole population. Therefore, to control tuberculosis, the government should take some strategies for elderly patients, such as regular checks and immediate treatment for elderly patients, to further control tuberculosis in the future.
The expression of \(\mathcal {R}_0\) obtained here depends on some functions, such as delay and disease progression rate. Our study can be generalized to study the basic reproduction number with other types of delay and disease progression functions. In addition, random contact in society may influence TB transmission dynamics. Some stochastic factors can be included to our model for understanding the transmission process of tuberculosis under random effects.
References
Aagaard C, Hoang T, Dietrich J, Cardona P, Izzo A, Dolganov G, Schoolnik G, Cassidy J, Billeskov R, Andersen P (2011) A multistage tuberculosis vaccine that confers efficient protection before and after exposure. Nat Med 17(2):189
Anderson R, Trewhella W (1985) Population dynamics of the badger (Meles meles) and the epidemiology of bovine tuberculosis (Mycobacterium bovis). Philos Trans RSoc B Biol Sci 310(1145):327–381
Arregui S, Iglesias M, Samper S, Marinova D, Martin C, Sanz J, Moreno Y (2018) Data-driven model for the assessment of mycobacterium tuberculosis transmission in evolving demographic structures. P Natl Acad Sci USA 115(14):E3238–E3245
Barry C 3rd, Boshoff H, Dartois V, Dick T, Ehrt S, Flynn J, Schnappinger D, Wilkinson R, Young D (2009) The spectrum of latent tuberculosis: rethinking the biology and intervention strategies. Nat Rev Microbiol 7(12):845
Blower S, Mclean A, Porco T, Small P, Hopewell P, Sanchez M, Moss A (1995) The intrinsic transmission dynamics of tuberculosis epidemics. Nat Med 1(8):815
Cai J, Wang X, Ma A, Wang Q, Han X, Li Y (2015) Factors associated with patient and provider delays for tuberculosis diagnosis and treatment in asia: a systematic review and meta-analysis. PloS one 10(3):e0120088
Cardona P (2016) Reactivation or reinfection in adult tuberculosis: Is that the question? Int J Mycobacteriol 5(4):400–407
Cardona P, Ruiz-Manzano J (2004) On the nature of mycobacterium tuberculosis-latent bacilli. Eur Respir J 24(6):1044–1051
Castillo-Chavez C, Feng Z (1998) Global stability of an age-structure model for TB and its applications to optimal vaccination strategies. Math Biosci 151(2):135–154
Castillo-Chavez C, Song B (2004) Dynamical models of tuberculosis and their applications. Math Biosci Eng 1(2):361–404
Census and Statistics Department (2001) 2001 population by-census main report: volume I. Hong Kong Census and Statistics Department
Census and Statistics Department of the Government of the Hong Kong SAR (2018) Fertility trend in hong kong, 1981 to 2017. https://www.censtatd.gov.hk/hkstat/sub/sp160.jsp?productCode=FA100090
Census and Statistics Department of the Government of the Hong Kong SAR: Statistics on population (2018) https://www.censtatd.gov.hk/hkstat/sub/so20.jsp
Chan-Yeung M, Noertjojo K, Tan J, Chan S, Tam C (2002) Tuberculosis in the elderly in hong kong. Int J Tuberc Lung D 6(9):771–779
Cohen T, Colijn C, Finklea B, Murray M (2006) Exogenous re-infection and the dynamics of tuberculosis epidemics: local effects in a network model of transmission. J R Soc Interface 4(14):523–531
Department of Health of the Government of the Hong Kong SAR: Statistics on tuberculosis (1995-2018). https://www.info.gov.hk/tb_chest/en/index_2.htm (2018)
Feng Z, Castillo-Chavez C, Capurro A (2000) A model for tuberculosis with exogenous reinfection. Theor Popul Biol 57(3):235–247
Feng Z, Huang W, Castillo-Chavez C (2001) On the role of variable latent periods in mathematical models for tuberculosis. J Dyn Differ Equ 13(2):425–452
Feng Z, Iannelli M, Milner F (2002) A two-strain tuberculosis model with age of infection. SIAM J Appl Math 62(5):1634–1656
Flynn J, Chan J (2001) Tuberculosis: latency and reactivation. Infec Immun 69(7):4195–4201
Houben R, Dodd P (2016) The global burden of latent tuberculosis infection: a re-estimation using mathematical modelling. PLoS Med 13(10):e1002152
Jabbari A, Castillo-Chavez C, Nazari F, Song B, Kheiri H (2016) A two-strain tb model with multiple latent stages. Math Biosci Eng 13:741–785
Kapitanov G (2015) A double age-structured model of the co-infection of tuberculosis and HIV. Math Biosci Eng 12(1):23–40
Kasaie P, Andrews J, Kelton W, Dowdy D (2014) Timing of tuberculosis transmission and the impact of household contact tracing. An agent-based simulation model. Am J Resp Crit Care 189(7):845–852
Kirschner D, Pienaar E, Marino S, Linderman J (2017) A review of computational and mathematical modeling contributions to our understanding of mycobacterium tuberculosis within-host infection and treatment. Curr Opin Syst Biol 3:170–185
Kuniya T (2011) Global stability analysis with a discretization approach for an age-structured multigroup SIR epidemic model. Nonlinear Anal-Real 12(5):2640–2655
Liu Q, Jiang D, Hayat T, Alsaedi A (2018) Dynamics of a stochastic tuberculosis model with antibiotic resistance. Chaos Soliton Fract 109:223–230
Ma J, Teng X, Wang X, Fan X, Wu Y, Tian M, Zhou Z, Li L (2017) A multistage subunit vaccine effectively protects mice against primary progressive tuberculosis, latency and reactivation. EBioMedicine 22:143–154
Macdonald N (1978) Time lags in biological models. In: Lecture notes in biomathmatics, Springer-Verlag, Heidelberg
Makinodan T, Kay M (1980) Age influence on the immune system. Adv Immunol 29:287–330
Marais B, Gie R, Schaaf H, Hesseling A, Obihara C, Starke J, Enarson D, Donald P, Beyers N (2004) The natural history of childhood intra-thoracic tuberculosis: a critical review of literature from the pre-chemotherapy era [state of the art]. Int J Tuberc Lung D 8(4):392–402
Mossong J, Hens N, Jit M, Beutels P, Auranen K, Mikolajczyk R, Massari M, Salmaso S, Tomba G, Wallinga J (2008) Social contacts and mixing patterns relevant to the spread of infectious diseases. PLoS Med 5(3):e74
Nahid P, Hopewell P (2017) Tuberculosis treatment. In: International encyclopedia of public health, 2nd edn. Academic Press, pp 267–276
Okuonghae D, Omosigho S (2011) Analysis of a mathematical model for tuberculosis: what could be done to increase case detection. J Theor Biol 269(1):31–45
Parrish N, Dick J, Bishai W (1998) Mechanisms of latency in mycobacterium tuberculosis. Trends Microbiol 6(3):107–112
Renardy M, Kirschner D (2020) A framework for network-based epidemiological modeling of tuberculosis dynamics using synthetic datasets. Bull Math Biol 82(6):1–20
Selwyn P, Hartel D, Lewis V, Schoenbaum E, Vermund S, Klein R, Walker A, Friedland G (1989) A prospective study of the risk of tuberculosis among intravenous drug users with human immunodeficiency virus infection. New Engl J Med 320(9):545–550
Storla D, Yimer S, Bjune G (2008) A systematic review of delay in the diagnosis and treatment of tuberculosis. BMC Public Health 8(1):15
Styblo K (1991) Epidemiology of tuberculosis, 2nd edn. Royal Netherlands Tuberculosis Association, The Hague
Væth M, Skriver M, Støvring H (2018) The impact of proportional changes in age-specific mortality on life expectancy when mortality is a log-linear function of age. Demogr Res 39:671–684
Vynnycky E, Fine P (1997) The natural history of tuberculosis: the implications of age-dependent risks of disease and the role of reinfection. Epidemiol Infect 119(2):183–201
Vynnycky E, Fine P (2000) Lifetime risks, incubation period, and serial interval of tuberculosis. Am J Epidemiol 152(3):247–263
Waaler H, Geser A, Andersen S (1962) The use of mathematical models in the study of the epidemiology of tuberculosis. Am J Public Health 52(6):1002–1013
World Health Organization (2006) Regional Office for the Eastern Mediterranean: diagnostic and treatment delay in tuberculosis. World Health Organization
World Health Organization (2018) Global tuberculosis report 2018. World Health Organization
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
A. Numerical Scheme
We first define a function Z(t, a) as
We rewrite system (1) into the following system
The initial and boundary conditions are
We first discretize the space of time, age and stage into mesh as followed
where \(\varDelta t, \varDelta a, \varDelta s\) are the step sizes of the time, age and stage, respectively. We suppose that all the age densities distribute in the interval \([0,a_{\max }]\) and all the stage densities distribute in the interval \([0,s_{\max }]\) and \(t\in [0,T]\), where T is the time span of the prediction period for the disease transmission. We have the following equalities for \(N, L_1, L_2\)
that is, \(N, L_1, L_2\) are the number of steps from 0 to T, \(a_{\max }\) and \(s_{\max }\) separately.
Here, we simulate the dynamics of system in the population with stationary structure and hence we suppose that the immigration rate and the birth rate are only related with age in the following simulations, that is, \(\beta (t,a) = \beta (a)\). Using i, j, k represent \(t_i, a_j,s_k\) for simplification, we have the following identities for the boundary conditions
where \(\beta _0\) is a positive constant representing the birth rate of the population. On the basis of the finite difference method for derivative and the trapezoidal rule for approximating integral, we discretize system (A.10) as follows.
Here,
For \(i = 0\), we utilize the initial epidemic data
For \(i = 1\), we do the following
-
1.
Calculating \(I^1_{j,0}\) for all j with
$$\begin{aligned} I^1_{j,0} = S^0_jG^0_j, \end{aligned}$$and
$$\begin{aligned} Z^1_j = I^1_{j,0}. \end{aligned}$$ -
2.
Calculating \(S^1_j\) for all j with formula (A.11) and \(I^1_{j,k}\) for all k with formula (A.12).
For \(2\le i\le N\), we do the following
-
1.
Calculating \(S^i_j\), \(I^i_{j,k}\) with the formula in (A.11) and (A.12) separately.
-
2.
Calculating \(Z^i_j\) with formula (A.13) and then replacing \(I^i_{j,0}\) with \(Z^i_j\), that is, \(I^i_{j,0} = Z^i_j\) for the boundary condition of infectious individuals.
In our numerical simulations, we set \(\varDelta t = 0.1\) and \(\varDelta a =\varDelta s = 0.01\).
Rights and permissions
About this article
Cite this article
Mu, Y., Chan, TL., Yuan, HY. et al. Transmission Dynamics of Tuberculosis with Age-specific Disease Progression. Bull Math Biol 84, 73 (2022). https://doi.org/10.1007/s11538-022-01032-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11538-022-01032-4