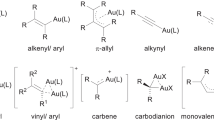
Abstract
Diazo compounds are generally used as carbene precursors. Traditionally, dirhodium, copper and iron catalysts were used to decompose diazo compounds to form the key metal carbene intermediates. Recently, the gold catalysts have been developed as a unique type of metal catalyst to decompose diazo compounds. The derived gold carbene showed much different characters comparing with other transition metal carbenes. They could go through a series of cycloaddition, insertion and coupling reactions. Here, the recent progress of the gold carbene chemistry from diazo compounds was reviewed, including the scope of reactions, mechanism and synthetic applications.
















Similar content being viewed by others
References
Fürstner A, Davies PW (2007) Catalytic carbophilic activation: catalysis by platinum and gold π acid. Angew Chem Int Ed 46:3410–3449
Jiménez-Núnez E, Echavarren AM (2008) Gold-catalyzed cycloisomerizations of enynes: a mechanistic perspective. Chem Rev 108:3326–3350
Gorin DJ, Sherry BD, Toste FD (2008) Ligand effects in homogeneous Au catalysis. Chem Rev 108:3351–3378
Rudolph M, Hashmi ASK (2012) Gold catalysis in total synthesis—an update. Chem Soc Rev 41:2448–2462
Ji KG, Zhang LM (2014) A non-diazo strategy to cyclopropanation via oxidatively generated gold carbene: the benefit of a conformationally rigid P,N-bidentate ligand. Org Chem Front 1:34–38
Qian DY, Zhang JL (2015) Gold-catalyzed cyclopropanation reaction using a carbenoid precursor toolbox. Chem Soc Rev 44:677–698
Zhou YB, Trewyn BG, Angelici RJ et al (2009) Catalytic reaction of carbene precursors on bulk gold metal. J Am Chem Soc 131:11734–11743
Obradors C, Echavarren AM (2013) Gold-catalyzed rearrangements and beyond. Acc Chem Res 47:902–912
López S, Herrero-Gómez E, Pérez-Galán P et al (2006) Gold(I)-catalyzed intermolecular cyclopropanation of enynes with alkenes: trapping of two different gold carbenes. Angew Chem Int Ed 45:6029–6032
Taduri BP, Sohel SMA, Cheng HM et al (2007) Pt- and Au-catalyzed oxidative cyclization of 2-ethenyl-1-(prop-29-yn-19-ol)benzenes to naphthyl aldehydes and ketones: catalytic oxidation of metal-alkylidene intermediates using H2O and H2O2. Chem Commun 19:2530–2532
Brooner REM, Brown TJ, Widenhoefer RA (2013) Direct observation of a cationic gold(I)-bicyclo[3.2.0]hept-1(7)-ene complex generated in the cycloisomerization of a 7-phenyl-1,6-enyne. Angew Chem Int Ed 52:6259–6261
Shiroodi RK, Gevorgyan V (2013) Metal-catalyzed double migratory cascade reactions of propargylic esters and phosphates. Chem Soc Rev 42:4991–5001
Li GT, Zhang GZ, Zhang LM (2008) Au-catalyzed synthesis of (1Z,3E)-2-pivaloxy-1,3-dienes from propargylic pivalates. J Am Chem Soc 130:3740–3741
Amijs CHM, López-Carrillo V, Echavarren AM (2007) Gold-catalyzed addition of carbon nucleophiles to propargyl carboxylates. Org Lett 9:4021–4024
Shapiro ND, Shi Y, Toste FD (2009) Gold-catalyzed [3 + 3]-annulation of azomethine imines with propargyl esters. J Am Chem Soc 131:11654–11655
Gorin DJ, Dubé P, Toste FD (2006) Synthesis of benzonorcaradienes by gold(I)-catalyzed [4 + 3] annulation. J Am Chem Soc 128:14480–14481
Zhang LM (2014) A non-diazo approach to α-oxo gold carbenes via gold-catalyzed alkyne oxidation. Acc Chem Res 47:877–888
Wang KB, Ran RQ, Xiu SD et al (2013) Synthesis of 3-aza-bicyclo[3.1.0]hexan-2-one derivative via gold-catalyzed oxidative cyclopropanation of N-allylynamides. Org Lett 15:2374–2377
Li L, Shu C, Zhou B et al (2014) Generation of gold carbenes in water: efficient intermolecular trapping of the α-oxo gold carbenoids by indoles and anilines. Chem Sci 5:4057–4064
Ye LW, He WM, Zhang LM (2011) A flexible and stereoselective synthesis of azetidin-3-ones through gold-catalyzed intermolecular oxidation of alkynes. Angew Chem Int Ed 50:3236–3239
Noey EL, Luo YD, Zhang LM et al (2012) Mechanism of gold(I)-catalyzed rearrangements of acetylenic amine-N-oxides: computational investigations lead to a new mechanism confirmed by experiment. J Am Chem Soc 134:1078–1084
Solorio-Alvarado C, Wang YH, Echavarren AM (2011) Cyclopropanation with gold(I) carbenes by retro-Buchner reaction from cycloheptatrienes. J Am Chem Soc 133:11952–11955
Wang YH, McGoniga PR, Herlé B et al (2014) Gold(I) carbenes by retro-Buchner reaction: generation and fate. J Am Chem Soc 136:801–809
Wang YH, Muratore ME, Rong Z et al (2014) Formal (4 + 1) cycloaddition of methylenecyclopropanes with 7-ary-1,3,5-cycloheptatrienes by triple gold(I) catalysis. Angew Chem Int Ed 53:14022–14026
Komine N, Flores JA, Pal K et al (2013) Buchner reaction catalyzed by a silver(I) pyridylpyrrolide: understanding arene C=C insertion selectivity. Organometallics 32:3185–3191
Davies HML, Beckwith REJ (2003) Catalytic enantioselective C–H activation by means of metal–carbenoid-induced C–H insertion. Chem Rev 103:2861–2903
Doyle MP, Duffy R, Ratnikov M et al (2010) Catalytic carbene insertion into C–H bonds. Chem Rev 110:704–724
Gillingham D, Fei N (2013) Catalytic X–H insertion reactions based on carbenoids. Chem Soc Rev 42:4918–4931
Sun XL, Tang Y (2008) Ylide-initiated Michael addition–cyclization reactions beyond cyclopropanes. Acc Chem Res 41:937–948
Zhou J, Tang Y (2005) The development and application of chiral trisoxazolines in asymmetric catalysis and molecular recognition. Chem Soc Rev 34:664–676
Lebel H, Marcoux JF, Molinaro C et al (2003) Stereoselective cyclopropanation reactions. Chem Rev 103:977–1050
Xia Y, Feng S, Liu Z et al (2015) Rhodium(I)-catalyzed sequential C(sp)–C(sp3) and C(sp3)–C(sp3) bond formation through migratory carbene insertion. Angew Chem Int Ed 54:7891–7894
Hu WH, Xu XF, Zhou J et al (2008) Cooperative catalysis with chiral brønsted acid-Rh2(OAc)4: highly enantioselective three-component reactions of diazo compounds with alcohols and imines. J Am Chem Soc 130:7782–7783
Cheng QQ, Zhu SF, Zhang YZ et al (2013) Copper-catalyzed B–H bond insertion reaction: a highly efficient and enantioselective C–B bond-forming reaction with amine-borane and phosphine–borane adducts. J Am Chem Soc 135:14094–14097
Sheng JJ, Zhu SF, Cai Y et al (2014) Enantioselective iron-catalyzed intramolecular cyclopropanation reactions. Angew Chem Int Ed 53:13188–13191
Li J, Liao SH, Xiong H et al (2012) Highly diastereo- and enantioselective cyclopropanation of 1,2-disubstituted alkenes. Angew Chem Int Ed 51:8838–8841
Zhou JL, Wang LJ, Xu H et al (2013) Highly enantioselective synthesis of multifunctionalized dihydrofurans by copper-catalyzed asymmetric [4 + 1] cycloadditions of α-benzylidene-β-ketoester with diazo compound. ACS Catal 3:685–688
Zhou JL, Liang Y, Deng C et al (2011) Tunable carbonyl ylide reactions: selective synthesis of dihydrofurans and dihydrobenzoxepines. Angew Chem Int Ed 50:7874–7878
Liu CR, Zhu BH, Zheng JC et al (2011) Highly diastereroselective synthesis of dihydrofurans and dihydropyrroles via pyridine catalyzed formal [4 + 1] annulation. Chem Commun 47:1342–1344
Xu ZH, Zhu SN, Sun XL et al (2007) Sidearm effects in the enantioselective cyclopropanation of alkenes with aryldiazoacetates catalyzed by trisoxa zoline/Cu(I). Chem Commun 43:1960–1962
Fructos MR, Bedlderrain TR, de Frémont P et al (2005) A gold catalyst for carbene-transfer reactions from ethyl diazoacetate. Angew Chem Int Ed 44:5284–5288
Pérez PJ, Díaz-Requejo MM, Rivilla I (2011) Gold-catalyzed naphthalene functionalization. Beilstein J Org Chem 7:653–657
Prieto A, Fructos MR, Díaz-Requejo MM et al (2009) Gold-catalyzed olefin cyclopropanation. Tetrahedron 65:1790–1793
Lonzi G, López LA (2013) Regioselective synthesis of functionalized pyrroles via gold(I)-catalyzed [3 + 2] cycloaddition of stabilized vinyl diazo derivatives and nitriles. Adv Synth Catal 355:1948–1954
Neupane P, Xia LK, Lee YR (2014) One-pot synthesis of multi-substituted 4-pyrones by a gold-catalyzed cascade of Wolff rearrangement/[4 + 2] cycloaddition/elimination reaction. Adv Synth Catal 356:2566–2574
Cao ZY, Wang XM, Tan C et al (2013) Highly stereoselective olefin cyclopropanation of diazooxindoles catalyzed by a C2-symmetric spiroketal bisphosphine/Au(I) complex. J Am Chem Soc 135:8197–8200
Lou Y, Horikawa M, Kloster RA et al (2004) A new chiral Rh(II) catalyst for enantioselective [2 + 1]-cycloaddition. Mechanistic implications and applications. J Am Chem Soc 126:8916–8918
Davies HML, Lee GH (2004) Dirhodium(II) tetra(N-(dodecylbenzenesulfonyl)prolinate) catalyzed enantioselective cyclopropenation of alkynes. Org Lett 6:1233–1236
Briones JF, Hansen J, Hardcastle KI et al (2010) Highly enantioselective Rh2(S-DOSP)4-catalyzed cyclopropenation of alkynes with styryldiazoacetates. J Am Chem Soc 132:17211–17215
Goto T, Takeda K, Shimada N et al (2011) Highly enantioselective cyclopropenation reaction of 1-alkynes with α-alkyl-α-diazoesters catalyzed by dirhodium(II) carboxylates. Angew Chem Int Ed 50:6803–6808
Cui X, Xu X, Lu HJ et al (2011) Enantioselective cyclopropenation of alkynes with acceptor/acceptor-substituted diazo reagents via Co(II)-based metalloradical catalysis. J Am Chem Soc 133:3304–3307
Uehara M, Suematsu H, Yasutomi Y et al (2011) Enantioenriched synthesis of cyclopropenes with a quaternary stereocenter, versatile building blocks. J Am Chem Soc 133:170–171
Briones JF, Davies HML (2011) Silver triflate-catalyzed cyclopropenation of internal alkynes with donor-/acceptor-substituted diazo compounds. Org Lett 13:3984–3987
Briones JF, Davies HML (2012) Gold(I)-catalyzed asymmetric cyclopropenation of internal alkynes. J Am Chem Soc 134:11916–11919
Briones JF, Davies HML (2013) Enantioselective gold(I)-catalyzed vinylogous [3 + 2] cycloaddition between vinyldiazoacetates and enol ethers. J Am Chem Soc 135:13314–13317
Pagar VV, Jadhav AM, Liu RS (2011) Gold-catalyzed formal [3 + 3] and [4 + 2] cycloaddition reactions of nitrosobenzenes with alkenylgold carbenoids. J Am Chem Soc 133:20728–20731
Jadhav AM, Pagar VV, Liu RS (2012) Development of a Povarov reaction/carbene generation sequence for alkenyldiazocarbonyl compounds. Angew Chem Int Ed 51:11809–11813
Pawar SK, Wang CD, Bhunia S et al (2013) Gold-catalyzed formal cycloaddition of 2-ethynylbenzyl ethers with organic oxides and α-diazoesters. Angew Chem Int Ed 52:7559–7563
Rivilla I, Gómez-Emeterio BP, Fructos M et al (2011) Exclusive aromatic vs aliphatic C–H bond functionalization by carbene insertion with gold-based catalysts. Organometallics 30:2855–2860
Zhou L, Liu YZ, Zhang Y et al (2011) Sequential Au(I)-catalyzed reaction of water with o-acetylenyl-substituted phenyldizaoacetates. Beilstein J Org Chem 7:631–637
Fructos MR, de Frémont P, Nolan SP et al (2006) Alkane carbon-hydrogen bond functionalization with (NHC)MCl precatalysts (M = Cu, Au; NHC = N-heterocyclic carbene). Organometallics 25:2237–2241
Wang YH, Muratore ME, Echavarren AM (2015) Gold carbene or carbenoid: is there a difference? Chem Eur J 21:7332–7339
Yu ZZ, Ma B, Chen MJ et al (2014) Highly site-selective direct C–H bond functionalization of phenols with α-aryl-α-diazoacetates and diazooxindoles via gold catalysis. J Am Chem Soc 136:6904–6907
Xi YM, Su YJ, Yu ZY et al (2014) Chemoselective carbophilic addition of α-diazoesters through ligand-controlled gold catalysis. Angew Chem Int Ed 53:9817–9821
Barluenga J, Lonzi G, Tomás M et al (2013) Reactivity of stabilized vinyl diazo derivatives toward unsaturated hydrocarbons: regioselective gold-catalyzed carbon–carbon bond formation. Chem Eur J 19:1573–1576
López E, Lonzi G, López LA (2014) Gold-catalyzed C–H bond functionalization of metallocenes: synthesis of densely functionalized ferrocene derivatives. Organometallics 33:5924–5927
Karad SN, Liu RS (2014) Gold-catalyzed 1,2-oxoarylations of nitriles with pyridine-derived oxides. Angew Chem Int Ed 53:5444–5448
Pagar VV, Jadhav AM, Liu RS (2013) Gold-catalyzed reactions between alkenyldiazo carbonyl species and acetals. J Org Chem 78:5711–5716
Pagar VV, Liu RS (2015) Gold-catalyzed α-furanylations of quinoline N-oxides with alkenyldiazo carbonyl species. Org Biomol Chem 13:6166–6169
Xia Y, Liu ZX, Xiao Q et al (2012) Rhodium(II)-catalyzed cyclization of bis(N-tosylhydrazone)s: an efficient approach towards polycyclic aromatic compounds. Angew Chem Int Ed 51:5714–5717
Zhang DM, Xu GY, Ding D et al (2014) Gold(I)-catalyzed diazo coupling: strategy towards alkene formation and tandem benzannulation. Angew Chem Int Ed 53:11070–11074
Xu GY, Zhu CH, Gu WJ et al (2015) Gold(I)-catalyzed diazo cross-coupling: a selective and ligand-controlled denitrogenation/cyclization cascade. Angew Chem Int Ed 54:883–887
Zhang S, Wei F, Song CL et al (2014) Recent advances of Au/acid combined catalysis. Chin J Chem 32:937–956
Zhang S, Xu ZL, Jia J et al (2014) Synthesis of spiroaminals by bimetallic Au/Sc relay catalysis: TMS as a traceless controlling group. Chem Commun 50:12084–12087
Wang XH, Yao ZL, Dong SL et al (2013) Synthesis of fused bicyclic aminals through sequential gold/Lewis acid catalysis. Org Lett 15:2234–2237
Wang XH, Dong SL, Yao ZL et al (2014) Synthesis of spiroaminals and spiroketals with bimetallic relay catalysis. Org Lett 16:22–25
Acknowledgments
This work was supported by the National Natural Science Foundation of China (21372144), China Postdoctoral Science Foundation, the Fundamental Research Funds of Shandong University (2014JC008) and the Subject Construction Funds of Shandong University (104.205.2.5).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
About this article
Cite this article
Wei, F., Song, C., Ma, Y. et al. Gold carbene chemistry from diazo compounds. Sci. Bull. 60, 1479–1492 (2015). https://doi.org/10.1007/s11434-015-0874-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-015-0874-0