Abstract
Purpose
Sediment internal nutrient loading plays an important role in algal blooms. Thus, understanding the specific characteristics of nutrient (nitrogen (N) and phosphorus (P)) recycling from sediment is important for clarifying the processes and mechanisms of cyanobacterial growth and succession.
Materials and methods
Sediments and water samples were collected monthly from five sampling sites in Lake Chaohu for 1 year. N and P concentrations in surface and interstitial water were determined, and fractions of sediment organic matter, P, and iron were quantified. P adsorption characteristics were modeled with adsorption isotherms.
Results and discussion
Dolichospermum was the dominant bloom species in the western lake in April, followed by Microcystis from May to September in the whole lake. The impulsive regeneration and release mode from iron-bound P and ammonium (NH4+-N) regeneration from sediment in spring triggered the Dolichospermum bloom. In early summer, continuous P release potential as well as nitrate accumulation and NH4+-N deficiency due to NH4+-N prior assimilation by Dolichospermum jointly drove the transition from Dolichospermum to Microcystis due to the energy saving and competitive advantage of rapid uptake and storage of inorganic P as well as a wide range of N utilization forms for Microcystis. All these facts put the Dolichospermum towards common N and P stress. Furthermore, in summer, NH4+-N rapid regeneration from organic algal detritus remineralization due to the decline of Dolichospermum developed Microcystis blooms.
Conclusions
The specific mode and pathway of N and P recycling from sediment determined the dominant algal species based on the particular N and P utilization strategies of the algae. Pulsed and a vast amount of P release facilitated the Dolichospermum growth, while continuous and a small quantity of P release was in favor of Microcystis growth. Organic N hydrolysis from Dolichospermum detritus further supported the development of a Microcystis bloom.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Cyanobacterial blooms in eutrophic lakes are severe environmental problems worldwide and of global concern due to the multiple harmful risks they pose towards aquatic ecosystem and human health (Jiang et al. 2017; Jia et al. 2019). Sediment internal loading plays an important role in the eutrophication of Lake Chaohu (Yang et al. 2020) and represents a significant supply for the growth of cyanobacteria (Cao et al. 2016). Phosphorus (P) migration and transformation caused by the microbes in sediments during cyanobacterial resuscitation phase may lead to the release of phosphorus from sediments and accelerate algae growth (Tu et al. 2022). In eutrophic Lake Dianchi, up to 70.2% of the released P could be absorbed by cyanobacteria (Cao et al. 2016). In Lake Taihu, cyanobacteria relied extensively on regenerated ammonium (NH4+-N) to sustain the bloom (Hampel et al. 2018). NH4+-N recycling supported toxic Planktothrix blooms in Sandusky Bay, Lake Erie (Hampel et al. 2019). Hence, algal bloom was mainly controlled by nitrogen (N) and P fluxes from sediment.
Nitrogen and P diffusive fluxes at the sediment–water interface in Erhai Lake showed different temporal and spatial variations (Zhao et al. 2018). The content of total P and N and their forms in water and sediment were rather dynamic during the year-long field investigation in the most polluted area of a shallow eutrophic lake (Lake Chaohu, China), the phosphate and NH4+-N fluxes showed evident seasonal variation, and higher fluxes were observed in warmer seasons especially during the period of algal bloom with high sedimentation (Yang et al. 2020). Meanwhile, the forms and content of P and N in water influenced the dominance of cyanobacteria (Ma et al. 2015). Green algal dominance quickly switched to cyanobacterial dominance after N and P enrichment in a greenhouse with elevated temperature (Wang et al. 2015). The response to nutrient enrichment differs among cyanobacterial species, which showed differential growth in relation to N and P concentrations (Loza et al. 2014).
Cyanobacterial dominance and composition of cyanobacterial blooms depended on the total N and total P levels as well as N to P ratios (Gonzlez-Madina et al. 2019). In Missisquoi Bay, Lake Champlain, Dolichospermum was the major bloom-forming cyanobacterium during summer, followed by a second intense bloom event of Microcystis in the fall; and the variation in the cyanobacterial population was strongly associated with inorganic and readily available fractions of N and P such as nitrites (NO2−-N) and nitrates (NO3−-N), NH4+-N, and dissolved organic P (Celikkol et al. 2021). The growth of Microcystis was influenced by the synergistic uptake and assimilation of nitrogen and phosphorus (Cai and Tang 2021). Furthermore, high concentrations of dissolved inorganic N and dissolved reactive P in Lake Taihu sediments potentially stimulated the initiation and maintenance of cyanobacterial blooms (Fan et al. 2022). The shallow, polymictic Ornamental Lake in Australia had suffered significant blooms of toxic Anabaena then Microcystis species every summer over the last decade; the prolific algal growth was controlled by the springtime P fluxes from the sediment, which was caused by diel stratification, combined with high oxygen consumption associated with organic carbon loading (Grace et al. 2010). Therefore, the temporal and spatial variation of P and N fluxes from sediment stimulated the initiation and transition of cyanobacterial blooms, which may be attributed to the different characteristics of sediment N and P recycling.
To explore the different characteristics of sediment N and P recycling during different dominated blooms and to test our hypothesis that the specific mode and pathway of sediment N and P recycling determines the pattern of algal succession, samples (including surface water and sediment) from five sampling sites in Lake Chaohu were collected monthly from January to December 2011 and were analyzed for chlorophyll a (Chl. a) in surface water, N and P species in surface water and interstitial water, labile organic matter, fractionation and sorption behaviors of phosphorus, extracellular enzymatic activities (EEA), and iron (Fe) in sediment. Through this study, we hope to (1) clarify the different modes and pathways of sediment N and P recycling and (2) illuminate the process that the different characteristics of sediment N and P recycling stimulated the initiation and transition of cyanobacterial blooms.
2 Materials and methods
2.1 Study sites and sample collection
Lake Chaohu, which is located in the middle of Anhui Province, China, is the fifth-largest shallow freshwater lake in China. The lake is commonly divided into three parts: western lake, central lake, and eastern lake. The western lake is evaluated as hypereutrophic status while the central and eastern lake stays in mesotrophic status (Shang and Shang 2007; Yu et al. 2011, 2014). In Lake Chaohu, cyanobacterial biomass showed high spatial–temporal changes, tending to increase in February and reaching a peak in April at some sites due to the growth of Dolichospermum; the second peak arrived in July and September and was caused by the rapid growth of Microcystis (Ren et al. 2021). Five sampling sites were established, and they were named with the initial letters of the rivers entering Lake Chaohu (to avoid confusion, sampling site X was named with the initial letter of another inflowing river, River Xiage). Sites X and T belong to eastern lake, site Z belongs to central lake, and sites S and N belong to western lake.
Surface water samples were taken by an organic glass hydrophore to analyze the N, P, and Chl. a concentration. Surface sediments samples were taken using a Peterson grab sampler to analyze P fractionation and sorption behavior, EEA, labile organic matter, and Fe. The interstitial water was separated from the sediment particles by centrifugation at 3000 r/min for 20 min. The supernatants were then filtered through a 0.45-μm membrane filter for analysis of N and P concentration. All samples were analyzed immediately after being back to the laboratory.
2.2 Nutrient forms in water samples
Chl. a was measured using the ethanol extraction method (Golterman et al. 1978). Soluble reactive phosphorus (SRP) concentration was detected by the molybdate blue method (Murphy and Riley 1962). Total P (TP) and dissolved total P (DTP) were determined following digestion by K2S2O8 according to GB11893-89 issued by State Environmental Protection Administration of China. Dissolved organic P (DOP) was calculated as DTP-SRP, and particulate P (PP) was calculated as TP-DTP. NH4+-N was determined by the indophenol-blue method described by Solórzano (1969) and Aminot et al. (1997), NO3−-N was determined by a UV-spectrophotometry determination method, and NO2−-N was determined by the α-naphthylamine method (Xu et al. 2005).
2.3 Nutrient forms in sediments
Sediment P fractionation was carried out according to Golterman (1996). This method groups sediment P into iron-bound P (Fe(OOH) ~ P), calcium-bound P (CaCO3 ~ P), acid-soluble organic P (ASOP), and hot NaOH-extractable organic P (Palk).
Ferrous iron (Fe2+) and total iron (TFe) were extracted from sediment by HCl and were determined according to the 1, 10 phenanthroline spectrometric method (Hauck et al. 2001). Before TFe was measured, all iron was reduced to the ferrous state with hydroxylamine hydrochloride (Stookey 1970). Ferric iron (Fe3+) was calculated as TFe-Fe2+.
Protein (PRT) analysis was conducted following extraction with NaOH (0.5 mol/L, 4 h) and was determined according to Hartree (1972) and modified by Rice (1982) to compensate for phenol interference and expressed as bovine serum albumin equivalents. Carbohydrates (CHO) were analyzed according to Gerchakov and Hatcher (1972) and expressed as glucose equivalents. Lipids were extracted by direct elution with chloroform–methanol according to Bligh and Dyer (1959) and Marsh and Weinstein (1966).
2.4 Phosphorus sorption in sediments
Phosphorus sorption characteristics of sediments were studied by batch incubation experiments (Li et al. 2014). Batch P sorption isotherm experiments were conducted in triplicate for sediment homogenates under reducing condition, using 0.01 mol L−1 KCl solution containing 0, 0.1, 0.2, 1, 2, 5, 8, 10, 15, 20, 25, 30, 40, and 50 mg P L−1 KH2PO4 as sorption solution matrices. The sealed centrifuge tubes with the mixed solution were shaken on a reciprocal shaker at a speed of 200 cycles min−1 for 24 h at 25 ± 3 °C. The suspension was centrifuged at 3500 rpm for 20 min. The supernatants were filtered through a 0.22-µm mixed cellulose ester membrane and determined for SRP. Phosphorus sorption parameters of sediments were simulated by the Langmuir and Freundlich isothermal model:
-
Langmuir equation: Q = QmaxKLC/(1 + KLC) − Q0L
-
Freundlich equation: Q = KFCn – Q0F
-
Q: amount of P sorbed by the solid phase after 24-h equilibrium (mg kg−1)
-
C: P concentration in solution after 24 h equilibrium (mg L−1)
-
Qmax: the maximum P sorption (mg kg−1)
-
KL: Langmuir adsorption energy parameter (L mg−1)
-
KF: Freundlich adsorption energy parameter (mg kg−1)
-
Q0F and Q0L: amount of P sorbed by the solid phase before the P sorption experiments (mg kg−1)
-
n: content (L kg−1)
Equilibrium P concentration (EPC0) was measured by the Freundlich equation, where no P sorption or desorption occurs. The P sorption maximum (Qmax) was measured by the Langmuir equation.
2.5 Extracellular enzymatic activity in sediments
Heterotrophic microorganisms in sediments produce extracellular enzymes to hydrolyze organic macromolecules, so their products can be transported inside the cell and used for energy and growth (Schmidt et al. 2021). Extracellular enzymatic activity is typically measured by addition of a fluorescently labeled substrate to an environmental sample, and hydrolysis is detected either as an increase in fluorescence as a fluorophore is cleaved (Hoppe 1983) or as a change in molecular weight distribution as a fluorescent substrate is hydrolyzed into lower molecular weight products (Arnosti 1996, 2003). Leucine aminopeptidase activity (LAP) and β-D-glucosidase activity (GLU) were measured fluorometrically according to Boetius and Lochte (1994), using the methylumbelliferone (MUF)–labeled substrates MCA-leu (L-leucine-4-methylcoumarinyl-7-amid HCl) and MUF-glu (MUG-Beta-D-glucopyranoside), respectively. Alkaline phosphatase (AP) was analyzed spectrophotometrically as p-nitrophenol (p-NP) resulting from the cleavage of phosphate from p-nitrophenylphosphate (p-NPP) (Sayler et al. 1979). Dehydrogenase activity (DHA) was determined by UV spectrophotometry according to Neto et al. (2007), with triphenyltetrazolium chloride as substrates.
2.6 Statistical analysis
All samples were analyzed in triplicate and the data are expressed as the average. Independent-sample T test was performed using the SPSS 18.0 package (SPSS, Chicago, IL), with a value of 0.05 or 0.01 selected for significance Fig. 1.
3 Results and discussion
3.1 Phosphorus recycling patterns
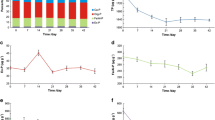
The significantly higher Chl. a concentration in western lake (sites N and S) in April (P < 0.01, Fig. 2a) and in the whole lake from May to September (Fig. 2a) indicated that severe algal bloom occurred in western lake in April and in whole lake from May to September. Dolichospermum was the dominant bloom species in western lake in April, followed by Microcystis from May to September in the whole lake. However, in April, the SRP concentration of surface water in western lake did not obviously decrease as algal blooms occurred and P was absorbed by Dolichospermum (Fig. 2b), indicating continuous P replenishment from sediment evidenced by significantly higher Fe(OOH) ~ P in sediments of western lake (sites N and S) (P < 0.01, Fig. 4a). Moreover, in April, the decrease of Fe3+ and the increase of Fe2+ as well as the increase of SRP in interstitial water in western lake (sites N and S) supported the above conclusion of the desorption of Fe(OOH) ~ P and P release from sediment to the interstitial water (Figs. 3a and 4c); that is to say, ferric iron was reduced to ferrous iron, finally leading to the release into interstitial water of inorganic P as SRP from Fe(OOH) ~ P. The re-dissolution of solid Fe(OOH) ~ P pools is the most important source of labile P and aggravates the P budget in lake water via anaerobic intervals (Yuan et al. 2019). Hence, desorption of Fe(OOH) ~ P was mainly responsible for P recycling in western lake. In April, the AP activity showed the higher levels than the other months in western lake (sites N and S) (Fig. 5a), suggesting that the hydrolysis of organic P may be another P source for the growth of Dolichospermum. The Dolichospermum culture experiments in different eutrophication scenarios showed that the expression of genes involved in P uptake, e.g., those involved in P-transport and the hydrolysis of phosphomonoesters, was upregulated in P-deficient cultures (Wang et al. 2018). AP was regulated by P availability in monocultures of Dolichospermum, but no cell bound extracellular phosphatase was found on Microcystis even in the culture without P supply (Wan et al. 2019). Therefore, the P sources required by Dolichospermum in western lake mainly derived from the impulsive release of Fe(OOH) ~ P and the hydrolysis of organic P, of which the former is more important.
Hydrolysis of polymers through extracellular enzymes is the first and acknowledged rate-limiting step in the decomposition of organic matter in aquatic environments (Cunningham and Wetzel 1989; Chróst 1992). GLU, LAP, and AP are extracellular enzymes hydrolyzing carbohydrates, organic N, and organic P and mediating C, N, and P cycling, respectively. In western lake, after the decline of Dolichospermum, with the increase of algae detritus, the sediment labile organic matter (PRT, CHO, and lipid) did not obviously increase from May to September (Fig. 4b), and extracellular enzymatic activities maintained a constantly high level during this period (Fig. 5a–c), indicating that the quick organic matter decomposition led to the rapid consumption of labile organic matter. The decomposition of organic carbon is always accompanied by a shift of redox status expressed by DHA (potential of oxidation and dehydrogenation of organic compounds) (Simčič 2005), which measures microbial metabolic activity as respiratory electron transport and has proved to be a good tool for estimating the respiratory potential of sediment (Broberg 1984; Blenkinsopp and Lock 1990). High respiratory potential (DHA) can trigger the change of redox potential and the formation of anaerobic status (Li et al. 2016). In this process, the bottom anaerobic status was formed expressed as high DHA (Fig. 5d); SRP was continually regenerated through the desorption of Fe(OOH) ~ P and released into the surface water based on the data of the continuous decrease of Fe(OOH) ~ P and Fe3+, the increase of Fe2+, and a constantly higher SRP concentration in surface water from May to August (Figs. 2b and 4a, c). The AP activity did not obviously increase from May to September (Fig. 5a), suggesting that extracellular alkaline phosphatase did not play a key role in P release during Microcystis bloom. At the same time, EPC0 could be used to estimate the function for sediment to act as sink or source of P and has been proved as a key index to indicate the P release potential and flux rate from sediments to water (Dong et al. 2011; Fu et al. 2022). In this study, compared to central and eastern lake (sites X, T, and Z), EPC0 in sediments of western lake maintained a higher level from April to August (P < 0.01, Fig. 6a), and the Qmax was similar (Fig. 6b), indicating that the decline of Dolichospermum and decomposition of labile organic matter lead to significantly higher P release potential to water column in western lake. The released P was quickly utilized by Microcystis, in terms of continuous higher SRP and PP in surface water in western lake from May to August (Fig. 2b). Therefore, impulsive regeneration and release from Fe(OOH) ~ P triggered the Dolichospermum bloom, and then, continuous and rapid P release potential drove the transition from Dolichospermum to Microcystis due to the energy saving and competitive advantage for Microcystis to rapidly uptake and store inorganic P, which put the coexisting Dolichospermum into P stress.
3.2 Nitrogen recycling patterns
As mentioned above, in April, the Chl. a concentration in western lake (sites N and S) was significantly higher than that of central and eastern lake (sites X, T, and Z) (P < 0.01, Fig. 2a). Nevertheless, the NH4+-N and NO3−-N in surface water, as well as NH4+-N in interstitial water, did not decrease but increase in this period (Figs. 2c and 3b). On the one hand, in March and April, higher LAP activity in western lake (sites N and S) (Fig. 5b) fueled the NH4+-N production in interstitial water by ammonification. Subsequently, NH4+-N was released into water column and simultaneously transformed partially into NO3−-N in surface water due to nitrification. On the other hand, Dolichospermum was the dominant bloom species in western lake in April. Previous research showed that growth rates of Dolichospermum was significantly higher when grown on NH4+-N relative to NO3−-N, and the existence of NH4+-N and urea significantly lowered N2 fixation of Dolichospermum (Kramer et al. 2022). Thus, Dolichospermum growth gave priority to NH4+-N, which inevitably led to the decrease of NH4+-N and the accumulation of NO3−-N, in terms of the lower NH4+-N and higher NO3−-N in surface water in May (Fig. 2c). Besides, the decline of Dolichospermum produced abundance of organic N, which might provide enough N source for the breakout of Microcystis bloom.
Organic N produced by Dolichospermum bloom was rapidly mineralized into NH4+-N with Dolichospermum collapse from spring to summer based on the data of continuous lower PRT content and higher LAP activity from May to September (Figs. 4b and 5b). As mentioned above, organic carbon, N, and P decomposition increased from May to September in terms of low sediment labile organic matter (PRT, CHO, and lipid) content (Fig. 4b) and a constantly high AP, LAP, and GLU in this time (Fig. 5a–c). And NH4+-N was partially released into water column and simultaneously transformed into NO3−-N by nitrification in surface water (Li et al. 2021), in terms of the higher NH4+-N in interstitial water and higher NO3−-N in surface water before May (Figs. 2c and 3b). But from June to October, both NH4+-N and NO3−-N in surface water were very low (Fig. 2c), which may be due to rapid assimilation by Microcystis. Microcystis can utilize different forms of inorganic and organic N, with the greatest capacity for NH4+-N uptake and the least for glutamic acid uptake (Lee et al. 2015). High concentration of NO3−-N facilitated the dominance of Microcystis, and Microcystis displayed faster growth rates in NH4+-N than in NO3−-N (Tan et al. 2019). Moreover, in the presence of SRP enrichment, the maximal uptake velocity of NH4+-N by Microcystis substantially increased without evidence of saturation (Yang et al. 2017). Additionally, in this study, anaerobic condition limited the nitrification, resulting in the NH4+-N accumulation in interstitial water in summer (Fig. 3b), which could provide a continuous N source for Microcystis blooms. Thus, we may conclude that in spring, NO3−-N accumulation due to NH4+-N prior assimilation of Dolichospermum initiated the transition from Dolichospermum to Microcystis, and in summer, NH4+-N rapid regeneration from organic algal detritus remineralization due to the decline of Dolichospermum developed Microcystis blooms.
Finally, the decline of Microcystis blooms led to accumulation of sediment labile organic matter (PRT, CHO, and lipid), and extracellular enzyme activity decreased due to low temperature based on the data of remarkable increasing PRT, CHO, and lipid content from October to December and decreasing AP, LAP, and GLU in November and December (Figs. 4b and 5a–c), which suggested that labile organic matter was stored in sediment at the end of algal bloom and can provide abundant N and P for algal bloom the next year.
4 Conclusions
Dolichospermum was the dominant bloom species in western lake in April, followed by Microcystis from May to September in the whole lake. The impulsive regeneration and release mode from Fe(OOH) ~ P and NH4+-N regeneration from the sediment in spring triggered the Dolichospermum bloom. In early summer, the continuous and rapid P release potential as well as NO3−-N accumulation and NH4+-N deficiency due to NH4+-N prior assimilation by Dolichospermum jointly drove the switch from Dolichospermum to Microcystis due to the energy saving and competitive advantage of rapidly uptake and store inorganic P as well as a wide range of N utilization forms for Microcystis. All these facts put the Dolichospermum towards N and P common stress. Furthermore, in summer, NH4+-N rapid regeneration from organic algal detritus remineralization due to the decline of Dolichospermum developed Microcystis blooms. Hence, the mode and pathway of P regeneration and release from sediment determined algal bloom species based on algal own special P utilization strategies. In detail, pulsed and a vast amount of P release facilitated the Dolichospermum growth, while continuous and a small quantity of P release was in favor of Microcystis growth. Organic N hydrolysis from Dolichospermum detritus further supported the development of Microcystis bloom.
Data availability
The data that support the findings of this study are openly available in https://pan.baidu.com/s/16IluhwQgalYtn4p--kqF8g.
References
Aminot A, Kirkwood DS, Kérouel R (1997) Determination of ammonia in seawater by the indophenol-blue method: Evaluation of the ICES NUTS I/C 5 questionnaire. Mar Chem 56(1):59–75. https://doi.org/10.1016/S0304-4203(96)00080-1
Arnosti C (1996) A new method for measuring polysaccharide hydrolysis rates in marine environments. Org Geochem 25:105–115. https://doi.org/10.1016/S0146-6380(96)00112-X
Arnosti C (2003) Fluorescent derivatization of polysaccharides and carbohydrate-containing biopolymers for measurement of enzyme activities in complex media. J Chromatogr B 793(1):181–191. https://doi.org/10.1016/S1570-0232(03)00375-1
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37(8):911–917. https://doi.org/10.1139/o59-099
Boetius A, Lochte K (1994) Regulation of microbial enzymatic degradation of OM in deep-sea sediments. Mar Ecol Prog Ser 104(3):299–307. https://doi.org/10.3354/meps104299
Broberg A (1984) A modified method for studies of electron transport system activity in freshwater sediments. Hydrobiologia 120(2):181–187. https://doi.org/10.1007/BF00032140
Blenkinsopp SA, Lock MA (1990) The measurement of electron transport system activity in river biofilms. Water Res 24(4):441–445. https://doi.org/10.1016/0043-1354(90)90226-V
Cai YY, Tang CY (2021) A kinetics model for predicting Microcystis growth based on the synergistic effect of nitrogen and phosphorus on the growth of Microcystis densa (Cyanobacteria). Pol J Environ Stud 30(2):1067–1077. https://doi.org/10.15244/pjoes/124756
Cao X, Wang YQ, He J, Luo XZ, Zheng Z (2016) Phosphorus mobility among sediments, water and cyanobacteria enhanced by cyanobacteria blooms in eutrophic Lake Dianchi. Environ Pollut 219(16):580–587. https://doi.org/10.1016/j.envpol.2016.06.017
Celikkol S, Fortin N, Tromas N, Andriananjamanantsoa H, Greer CW (2021) Bioavailable nutrients (N and P) and precipitation patterns drive cyanobacterial blooms in Missisquoi Bay. Lake Champlain Microorganisms 9(10):2097. https://doi.org/10.3390/microorganisms9102097
Chróst RJ (1992) Significance of bacterial ectoenzymes in aquatic environments. Hydrobiologia 243:61–70. https://doi.org/10.1007/BF00007020
Cunningham HW, Wetzel RG (1989) Kinetic-analysis of protein-degradation by a fresh-water wetland sediment community. Appl Environ Microbiol 55(8):1963–1967. https://doi.org/10.1128/aem.55.8.1963-1967.1989
Dong LM, Yang ZF, Liu XH (2011) Phosphorus fractions, sorption characteristics, and its release in the sediments of Baiyangdian Lake. China Environ Monit Assess 179(1–4):335–345. https://doi.org/10.1007/s10661-010-1740-9
Fan XF, Gao SS, Zhang Y, Qin BQ, Xu H, Ding SM (2022) Stimulation of high-concentration dissolved nitrogen and reactive phosphorus in Lake Taihu sediments on the initiation and maintenance of cyanobacterial blooms. Sci Total Environ 851:158088. https://doi.org/10.1016/j.scitotenv.2022.158088
Fu ZH, Hong ZJ, Wei JL, Liao YK, You SL, Wang YF, Lv JJ, Feng H, Kolenčík M, Chang XX, Qian Y (2022) Phosphorus fractionation and adsorption characteristics in drinking water reservoir inlet river sediments under human disturbance. J Soils Sediments 22(9):2530–2547. https://doi.org/10.1007/s11368-022-03257-1
Gerchakov SM, Hatcher PG (1972) Improved technique for analysis of carbohydrates in sediment. Limnol Oceanogr 17(6):938–943. https://doi.org/10.4319/lo.1972.17.6.0938
Golterman HL (1996) Fractionation of sediment phosphate with chelating compounds: a simplification, and comparison with other methods. Hydrobiologia 335(1):87–95. https://doi.org/10.1007/BF00013687
Golterman HL, Clymo RS, Ohmstad MAM (eds) (1978). Blackwell Scientific Publications, Oxford
González-Madina L, Pacheco JP, Yema L, de Tezanos P, Levrini P, Clemente J, Crisci C, Lagomarsino JJ, Mndez G, Fosalba C, Goyenola G, Mazzeo N (2019) Drivers of cyanobacteria dominance, composition and nitrogen fixing behavior in a shallow lake with alternative regimes in time and space, Laguna del Sauce (Maldonado, Uruguay). Hydrobiologia 829:61–76. https://doi.org/10.1007/s10750-018-3628-6
Grace MR, Scicluna TR, Vithana CL, Symes P, Lansdown KP (2010) Biogeochemistry and cyanobacterial blooms: investigating the relationship in a shallow, polymictic, temperate lake. Environ Chem 7(5):443–456. https://doi.org/10.1071/EN10042
Hampel JJ, McCarthy MJ, Gardner WS, Zhang L, Xu H, Zhu GW, Newell SE (2018) Nitrification and ammonium dynamics in Taihu Lake, China: seasonal competition for ammonium between nitrifiers and cyanobacteria. Biogeosciences 15(3):733–748. https://doi.org/10.5194/bg-15-733-2018
Hampel JJ, McCarthy MJ, Neudeck M, Bullerjahn GS, McKay RML, Newell SE (2019) Ammonium recycling supports toxic Planktothrix blooms in Sandusky Bay, Lake Erie: evidence from stable isotope and metatranscriptome data. Harmful Algae 81:42–52. https://doi.org/10.1016/j.hal.2018.11.011
Hartree EF (1972) Determination of protein: a modification of the Lowry method that gives a linear photometric response. Anal Biochem 48(2):422–427. https://doi.org/10.1016/0003-2697(72)90094-2
Hauck S, Benz M, Brune A, Schink B (2001) Ferrous iron oxidation by denitrifying bacteria in profundal sediments of a deep lake (Lake Constance). FEMS Microbiol Ecol 37(2):127–134. https://doi.org/10.1016/S0168-6496(01)00153-2
Hoppe H (1983) Significance of exoenzymatic activities in the ecology of brackish water: measurements by means of methylumbelliferyl-substrates. Mar Ecol Prog Ser 11:299–308. https://doi.org/10.3354/meps011299
Jia YL, Chen QQ, Crawford SE, Song LR, Chen W, Hammers-Wirtz M, Strauss T, Seiler TB, Schäffer A, Hollert H (2019) Cyanobacterial blooms act as sink and source of endocrine disruptors in the third largest freshwater lake in China. Environ Pollut 245:408–418. https://doi.org/10.1016/j.envpol.2018.11.021
Jiang YG, Xiao P, Liu Y, Wang JX, Li RH (2017) Targeted deep sequencing reveals high diversity and variable dominance of bloom-forming cyanobacteria in eutrophic lakes. Harmful Algae 64:42–50. https://doi.org/10.1016/j.hal.2017.03.006
Kramer BJ, Jankowiak JG, Nanjappa D, Harke MJ, Gobler CJ (2022) Nitrogen and phosphorus significantly alter growth, nitrogen fixation, anatoxin-a content, and the transcriptome of the bloom-forming cyanobacterium, Dolichospermum. Front Microbiol 13:955032. https://doi.org/10.3389/fmicb.2022.955032
Lee J, Parker AE, Wilkerson FP, Dugdale RC (2015) Uptake and inhibition kinetics of nitrogen in Microcystis aeruginosa: results from cultures and field assemblages collected in the San Francisco Bay Delta, CA. Harmful Algae 47:126–140. https://doi.org/10.1016/j.hal.2015.06.002
Li H, Song CL, Cao XY, Zhou YY (2016) The phosphorus release pathways and their mechanisms driven by organic carbon and nitrogen in sediments of eutrophic shallow lakes. Sci Total Environ 572:280–288. https://doi.org/10.1016/j.scitotenv.2016.07.221
Li H, Song CL, Yang L, Qin HD, Cao XY, Zhou YY (2021) Nutrients regeneration pathway, release potential, transformation pattern and algal utilization strategies jointly drove cyanobacterial growth and their succession. J Environ Sci 103:255–267. https://doi.org/10.1016/j.jes.2020.11.010
Li H, Sun AH, Hou J, Chen X, Bai YQ, Cao XY, Song CL, Zhou YY (2014) Phosphorus fractions, sorption characteristics, and its release in sediments of a Chinese eutrophic river (Nanfei River). Fresen Environ Bull 23(5):1222–1231
Loza V, Perona E, Mateo P (2014) Specific responses to nitrogen and phosphorus enrichment in cyanobacteria: factors influencing changes in species dominance along eutrophic gradients. Water Res 48:622–631. https://doi.org/10.1016/j.watres.2013.10.014
Ma JR, Qin BQ, Paerl HW, Brookes JD, Wu P, Zhou J, Deng JM, Guo JS, Li Z (2015) Green algal over cyanobacterial dominance promoted with nitrogen and phosphorus additions in a mesocosm study at Lake Taihu. China Environ Sci Pollut Res 22(7):5041–5049. https://doi.org/10.1007/s11356-014-3930-4
Marsh JB, Weinstein DB (1966) Simple charring method for determination of lipids. J Lipid Res 7(4):574–576. https://doi.org/10.1016/S0022-2275(20)39274-9
Murphy J, Riley JP (1962) A modified single solution method for determination of phosphate in natural waters. Anal Chim Acta 26(1):31–36. https://doi.org/10.1016/S0003-2670(00)88444-5
Neto M, Ohannessian A, Delolme C, Bedell JP (2007) Towards an optimized protocol for measuring global dehydrogenase activity in storm-water sediments. J Soils Sediments 7(2):101–110. https://doi.org/10.1065/jss2007.02.206
Ren MD, Zhang M, Fan F, Yang JS, Yang Z, Chen KN, Li YC, Shi XZ (2021) Difference in temporal and spatial distribution pattern of cyanobacteria between the sediment and water column in Lake Chaohu. Environ Pollut 291:118163. https://doi.org/10.1016/j.envpol.2021.118163
Rice DL (1982) The detritus nitrogen problem: new observations and perspectives from organic geochemistry. Mar Ecol Prog Ser 9(2):153–162. https://doi.org/10.3354/meps009153
Sayler GS, Puziss M, Silver M (1979) Alkaline-phosphatase assay for freshwater sediments - application to perturbed sediment systems. Appl Environ Microb 38(5):922–927. https://doi.org/10.1128/aem.38.5.922-927.1979
Schmidt JM, Royalty TM, Lloyd KG, Steen AD (2021) Potential activities and long lifetimes of organic carbon-degrading extracellular enzymes in deep subsurface sediments of the Baltic Sea. Front Microbiol 12:702015. https://doi.org/10.3389/fmicb.2021.702015
Shang GP, Shang JC (2007) Spatial and temporal variations of eutrophication in western Chaohu Lake, China. Environ Monit Assess 130:99–109. https://doi.org/10.1007/s10661-006-9381-8
Simčič T (2005) The role of plankton, zoobenthos, and sediment in organic matter degradation in oligotrophic and eutrophic mountain lakes. Hydrobiologia 532(1–3):69–79. https://doi.org/10.1007/s10750-004-9015-5
Solórzano L (1969) Determination of ammonia in natural waters by the phenolhypochlorite method. Limnol Oceanogr 14:700–801. https://doi.org/10.4319/lo.1969.14.5.0799
Stookey LL (1970) Ferrozine-a new spectrophotometric reagent for iron. Anal Chem 42(7):779–781. https://doi.org/10.1021/AC60289A016
Tan X, Gu HH, Ruan YL, Zhong JJ, Parajuli K, Hu JY (2019) Effects of nitrogen on interspecific competition between two cell-size cyanobacteria: Microcystis aeruginosa and Synechococcus sp. Harmful Algae 89:101661. https://doi.org/10.1016/j.hal.2019.101661
Tu CQ, Jin ZH, Che FF, Cao X, Song XS, Lu CY, Huang W (2022) Characterization of phosphorus sorption and microbial community in lake sediments during overwinter and recruitment periods of cyanobacteria. Chemosphere 307:135777. https://doi.org/10.1016/j.chemosphere.2022.135777
Wan LL, Chen XY, Deng QH, Yang L, Li XW, Zhang JY, Song CL, Zhou YY, Cao XY (2019) Phosphorus strategy in bloom-forming cyanobacteria (Dolichospermum and Microcystis) and its role in their succession. Harmful Algae 84:46–55. https://doi.org/10.1016/j.hal.2019.02.007
Wang SY, Xiao J, Wan LL, Zhou ZJ, Wang ZC, Song CL, Zhou YY, Cao XY (2018) Mutual dependence of nitrogen and phosphorus as key nutrient elements: one facilitates Dolichospermum flos-aquae to overcome the limitations of the other. Environ Sci Technol 52(10):5653–5661. https://doi.org/10.1021/acs.est.7b04992
Wang XD, Liu XG, Qin B, Gu ZJ, Wu ZF, Xu H, Zhu H, Cheng GF, Liu H (2015) Green algae dominance quickly switches to cyanobacteria dominance after nutrient enrichment in greenhouse with high temperature. J Ecol Environ 38(3):293–305. https://doi.org/10.5141/ecoenv.2015.030
Xu J, Xie P, Zhang M, Yang H (2005) Variation in stable isotope signatures of seston and a zooplanktivorous fish in a eutrophic Chinese lake. Hydrobiologia 541(1):215–220. https://doi.org/10.1007/s10750-004-5709-y
Yang CH, Yang P, Geng J, Yin HB, Chen KN (2020) Sediment internal nutrient loading in the most polluted area of a shallow eutrophic lake (Lake Chaohu, China) and its contribution to lake eutrophication. Environ Pollut 262:114292. https://doi.org/10.1016/j.envpol.2020.114292
Yang JJ, Gao H, Glibert PM, Wang Y, Tong MM (2017) Rates of nitrogen uptake by cyanobacterially-dominated assemblages in Lake Taihu, China, during late summer. Harmful Algae 65:71–84. https://doi.org/10.1016/j.hal.2017.04.001
Yu HB, Xi BD, Jiang JY, Heaphy MJ, Wang HL, Li DL (2011) Environmental heterogeneity analysis, assessment of trophic state and source identification in Chaohu Lake. China Environ Sci Pollut R 18(8):1333–1342. https://doi.org/10.1007/s11356-011-0490-8
Yu L, Kong FX, Zhang M, Yang Z, Shi XL, Du MY (2014) The dynamics of Microcystis genotypes and microcystin production and associations with environmental factors during blooms in Lake Chaohu. China Toxins 6(12):3238–3257. https://doi.org/10.3390/toxins6123238
Yuan HZ, Tai ZQ, Li Q, Liu EF (2019) In-situ, high-resolution evidence from water-sediment interface for significant role of iron bound phosphorus in eutrophic lake. Sci Total Environ 706:136040. https://doi.org/10.1016/j.scitotenv.2019.136040
Zhao HC, Zhang L, Wang SR, Jiao LX (2018) Features and influencing factors of nitrogen and phosphorus diffusive fluxes at the sediment-water interface of Erhai Lake. Environ Sci Pollut R 25(2):1933–1942. https://doi.org/10.1007/s11356-017-0556-3
Funding
This work was funded by the Strategic Priority Research Program of the Chinese Academy of Sciences (No. XDA23040403), National Natural Science Foundation of China (41701557; 41877381; 42077314), and Research Funding of Wuhan Polytechnic University (No. 2022RZ078; Grant 2019y12).
Author information
Authors and Affiliations
Contributions
The study conception and design were performed by Hui Li, Chunlei Song, Yiyong Zhou, and Xiuyun Cao. Material preparation and data collection were performed by Hui Li and Daizhong Huang, and data analysis was performed by Hui Li, Wenjuan Shen, Yun He, and Jianfen Li. The first draft of the manuscript was written by Hui Li and revised by Chunlei Song. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Responsible editor: Haihan Zhang
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, H., Song, C., Huang, D. et al. Different characteristics of sediment nitrogen and phosphorus recycling during cyanobacterial growth and their succession. J Soils Sediments 23, 2951–2961 (2023). https://doi.org/10.1007/s11368-023-03527-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-023-03527-6