Abstract
Purpose
Soil is a major pool of anthropogenically emitted mercury (Hg) and will in turn be a source of atmospheric Hg contamination. It was hypothesized that accumulation of anthropogenic Hg in the soil at a regional scale is associated with the amount of Hg emitted to the atmosphere and soil type.
Materials and methods
A total of 448 horizon samples were collected from the typical soil types of the Yangtze River Delta and Pearl River Delta. Titanium (Ti) was selected as a reference element to discriminate anthropogenic Hg from the Hg associated with pedogenic processes. Soil pools of anthropogenic Hg were quantified to show the difference in Hg sequestration among soil types and regions.
Results and discussion
Soil pools of anthropogenic Hg ranged from 0.70 to 6971 g/ha in the Yangtze River Delta (YRD) and from 35.81 to 13098.71 g/ha in the Pearl River Delta (PRD). The estimated soil Hg pools were well correlated with the amounts of Hg emitted in these two regions. Soil properties, namely organic matter content, amorphous iron oxides, and cation exchange capacity, were significantly (p < 0.01) and positively correlated with soil Hg accumulation. Meanwhile, soil organic matter was the most important factor to determine the depth distribution of Hg in soil profile of the majority of soils. Perudic Cambisols and Stagnic Anthrosols which contained higher content of amorphous iron oxides had the highest soil Hg pools, indicating the importance of amorphous iron oxides other than organic matter to the Hg retention in soil. Soil and soil horizons with low pH and high salinity did show a low accumulation of Hg probably due to the remobilization of Hg under such conditions.
Conclusions
In addition to the amount of Hg emitted, soil types may be a major factor in controlling the sequestration of anthropogenic Hg in both regions. Stagnic Anthrosols are an important pool for retention of anthropogenic Hg.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Mercury (Hg) is a globally toxic pollutant and has been of wide public health concern during the past decades (Feng and Qiu 2008; Hammond 1971; Lin et al. 1997; Lin et al. 2012; Zhang and Wong 2007). Anthropogenic emissions are considered to be the main source of global Hg contamination and have been estimated to account for 2320 t annually (Pirrone et al. 2010). China is currently the country with the highest anthropogenic Hg emissions (Lin et al. 2012). Streets et al. (2005) estimate that total Chinese emissions are 536 (±236) t of total mercury, slightly lower than estimated by Wu et al. (2006), 696 (±307) t annually. However, both studies indicate that smelting of nonferrous metals and coal combustion, the two dominant anthropogenic sources, together account for 80 % of total Hg emissions. Wu et al. (2006) also report total Hg emissions in individual provinces for 1995 and 2003 and show that provinces located in southeast China have dramatically higher Hg emissions that are related to growing industrialization coal combustion with the rapid economic development in this region (Jiang et al. 2006).
Anthropogenic Hg can be introduced to and accumulate in soil by various pathways such as dry and wet deposition from the atmosphere, release from waste products, and application of fertilizers and pesticides containing Hg. Atmospheric deposition is assumed to be the most important contribution. Luo et al. (2009) estimate that atmospheric sources contribute more than 60 % of total Hg inputs to agricultural soils in China followed by fertilizers which account for 30.4 %. Besides being affected by anthropogenic sources, accumulation of Hg in soil also depends on soil properties such as soil organic matter (SOM) content, pH, and cation exchange capacity (CEC) (Huang et al. 2011; Smith-Downey et al. 2010; Yang et al. 2007). It has been well known that Hg has a high affinity for organic matter in soil and sediment (Ravichandran 2004; Skyllberg et al. 2006; Wallschlager et al. 1998). Mercury therefore tends to be retained in soils although losses do occur by leaching and volatilization (Grigal 2002; Scholtz et al. 2003). However, loss rates are assumed to be fairly small, typically less than 1 % of the soil pool per annum (Tipping et al. 2011b). Hence, the soil pool of anthropogenic Hg, expressed as mass per unit area to a chosen soil depth, provides a measure of cumulative pollutant inputs. Tipping et al. (2011a) have calculated the soil Hg pool to a depth of 15 cm in the UK, and the results suggest that local deposition plays an important role in determining the Hg concentrations of UK soils. Yin et al. (2009) estimate that a cumulative input of anthropogenic Hg reaches 317 to 515 g/ha in the surrounding topsoil of secondary copper smelters, equivalent to the soil Hg pool of urban and industrial soils in the UK. The main objective of the present study was therefore to investigate the major factors that contribute to the cumulative content of anthropogenic Hg in the different soils of the Yangtze River Delta and Pearl River Delta regions. The two regions are the most economically developed areas on the southeast coast of China and have different climatic conditions and dominant soil types.
2 Materials and methods
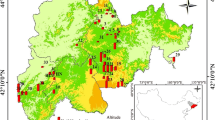
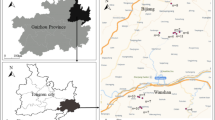
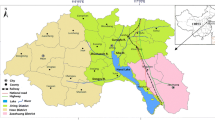
2.1 Description of the study areas
The Yangtze River Delta (YRD) and the Pearl River Delta (PRD) are the two most economically prosperous regions in southeast China, with annual rates of increase of gross domestic product (GDP) of 13.5 and 15.8 %, respectively, in 2006. These are higher than the 9.6 % national average GDP growth rate.
The YRD lies in the northern part of the subtropical zone with an annual average temperature of 17 °C and annual average rainfall of 1072 mm. The total area of the region is around 100,000 km2 and is mostly plain with only a small proportion of the upland in the west of the region. Udic Luvisols, Stagnic Anthrosols, and Aquic Cambisols are the dominant soil types in this region based on the FAO World Reference Base for Soil Resources (IUSS Working Group WRB 2006). Udic Luvisols are locally dominant in the uplands, and Stagnic Anthrosols have developed with long-term rice cultivation in the plain. Aquic Cambisols are distributed mainly in coastal areas and both sides of the Yangtze River and are primarily used for nonflooded crops such as vegetables and cotton. Other soil types of concern in this study are Udic Ferrisols and Perudic Cambisols.
The PRD is located on the southeast coast of China at the southern fringe of the subtropical zone. With a total land area of 25,000 km2 and a population of 42 million, it is the second most populous area in China and one of the most densely populated areas of the world. The climate is characterized by high humidity and temperatures, with an annual average precipitation of 1800 mm and annual average temperature of 22 °C. The soil is highly weathered in highland areas and waterlogged in flat terrain. Udic Ferralsols, Stagnic Anthrosols, and Orthic Halosols are the dominant soil types (IUSS Working Group WRB 2006). Soil types of concern in this study are Anthric Entisols, Orthic Halosols, and Sandic Entisols. Soils in this region are acid to neutral, and the soil organic matter content is high in paddy fields and orchards but comparatively low in uncultivated land.
2.2 Soil sampling and analytical methods
A total of 448 horizon samples were collected from the study areas in December of 2006. Based on pedogenic features, 262 horizon samples were taken from the 60 main soil profiles of the areas. A further 186 horizon samples were taken from the surface (0–20 cm) and subsurface (20–40 cm) depths of the 83 soil cores which were situated in the surrounding areas of the 60 main soil profiles. Detailed information on the sampling sites is shown in Table S1 (Electronic Supplementary Material). The samples were air-dried at room temperature for several days, screened through a 2-mm sieve, and then stored in glass bottles prior to analysis. In order to determine the concentration of Hg and other mineral elements in the samples, the soil samples were ground and passed through a 0.149-mm sieve before digestion. Total Hg in the soil was determined using the GB/T 17136–1997 method (SEPAC 1997). Briefly, 1.0 g of sieved soil was digested with 10-mL mixed concentrated acids (HNO3/H2SO4 = 1:1 (v/v)) and 10-mL 0.13 M KMnO4 solution on an electric hot plate at 90 °C for 1 h. The Hg concentration was determined using an atomic fluorescence spectrometer equipped with a flow injection system (AFS-930, Vital Co., Beijing). The total concentrations of Ti and Fe in the soil were determined by inductively coupled plasma atomic emission spectroscopy (ICP-AES; PerkinElmer 3300 DV) after 0.5-g sieved soil was digested with 1 mL 60 % perchloric acid, 8 mL 40 % hydrofluoric acid, and 17 mL concentrated nitric acid. Soil properties (pH, SOM, CEC, soil bulk density, humic acid, and iron oxides) were analyzed based on the methods of Lu (2000). Briefly, soil pH was measured by pH meter (LP115, Mettler-Toledo, Switzerland). SOM contents were measured by wet oxidation using K2Cr2O7. CEC was measured by the ammonium acetate method. Soil bulk density was measured by the gravimetric method after collecting undisturbed soil using a cutting ring. Humic acid was determined by a spectrophotometric method after isolating humic acid from the soil. Iron oxides were extracted using dithionite-citrate-bicarbonate (DCB) solution and acid (pH 3.0) ammonium oxalate extraction separately, representative of the crystal iron oxides and the amorphous iron oxides, respectively. The extracted iron was analyzed by ICP-AES. For quality control purposes, certified reference materials (CRMs, supplied by the National Research Center for CRMs, Beijing) were used as the external standards and analyzed with each batch of the samples in a proportion of 10 %. As a result, the geometric mean of the Hg concentration in the CRM soil (GBW07403) was determined to be 58.4 mg/kg (n = 18) with a range of 55–63 mg/kg, which is consistent with the certified value of 60.4 mg/kg.
2.3 Calculation of anthropogenic Hg concentration in the horizon soil samples
The measured Hg concentration in soil contains endogenous and exogenous Hg. Endogenous Hg is derived mainly from mineral weathering during the process of soil development, and exogenous Hg is mainly from the inputs of various anthropogenic sources. Calculation of anthropogenic Hg concentration in the horizon soil samples is mainly based on the methods of Zhang et al. (2007) and Zhang and Luo (2011). The anthropogenic Hg ([Hg]exo) was estimated by
where [Hg] total is the measured total Hg concentration (mg/kg) and [Hg] endo is the concentration (mg/kg) sourced from mineral weathering in the soil. The latter was estimated by
where [Ti] is the measured total Ti concentration (mg/kg) at each soil depth, 0.015 is the coefficient, and 0.004 is the constant; the standard error of the coefficient was 0.0048. All the coefficients were derived from the linear regression between Hg and Ti concentration values of the bottom soil layers. The linear regression function for the prediction of endogenous Hg was fitted at a significance level of 0.01.
2.4 Calculation of soil pools of anthropogenic Hg
The soil pools of anthropogenic Hg are the amounts of anthropogenic Hg sequestered by the soil. This is not restricted to the surface soil. Some of the anthropogenic Hg can migrate downward and accumulate in deeper layers or even the subsoil. Hence, the soil pool of anthropogenic Hg (P[Hg], g/ha) is the sum of anthropogenic Hg amounts in each pedogenic horizon (P i [Hg], g/ha) and can be calculated by
where ρ is the soil density (g/cm3) and h is the soil depth (cm).
2.5 Statistical analysis
Statistical analysis was performed using the SPSS for Windows v. 13.0 software package. Multiple comparison of the Hg concentration among the different genetic horizons of each soil type was conducted by one-way analysis of variance, and pairs of mean values were compared by least significant difference (LSD) at the 5 % level. Regression analysis between the Hg concentration and soil properties (CEC, soil organic matter, humic acid, and iron oxides) of the topsoil was performed to present the main factors contributing to the retention of anthropogenic Hg in the soils.
3 Results and discussion
3.1 Total Hg concentration in the different soil types
The soil Hg concentration in the different soil types of the two regions is shown in Table 1. The highest average concentration of Hg was observed in the Orthic Halosols, followed by Anthric Entisols, both of which were sampled from the Pearl River Delta. In addition, the Stagnic Anthrosols that are widely distributed as paddy soils in both study regions also showed relatively high Hg concentrations. As shown in Tables 1 and 2, Hg concentrations in the topsoil of the Stagnic Anthrosols were similar in both regions although the average Hg concentration of the topsoil of the whole region was slightly higher in the Pearl River Delta. Aquic Cambisols developed from alluvial deposits of the Yangtze River and usually used for vegetable production had lower Hg concentrations than the Stagnic Anthrosols. Compared to the soils in the plain regions, soil types developed in the uplands such as Udic Ferrisols, Udic Luvisols, and Udic Ferralsols all had lower concentrations of soil Hg. The lowest average Hg concentration was observed in Sandic Entisols which is located in the estuary of the PRD region and influenced by marine water frequently. Therefore, in addition to the low content of soil organic matter in the Sandic Entisols (Table 1), the low concentration of Hg in the Sandic Entisols might be partially owing to the high soil salinity (data not shown) which enhance the solubility of Hg by forming Hg(II)-chloride complexes (HgCl2).
The measured topsoil Hg concentrations in both regions were generally higher than other reported values. As shown in Table 2, the average soil Hg concentration in the Yangtze River Delta was 0.261 mg/kg, over twice the values for Jiangsu and Zhejiang provinces reported by Shi et al. (2013). The average soil Hg concentration of the Pearl River Delta was 0.311 mg/kg, slightly higher than that reported by Chen et al. (2012) although the highest concentration was lower in the present study. The soil Hg concentrations of the two regions were also higher than those reported in the UK (Tipping et al. 2011a) and the conterminous USA (Burt et al. 2003). When using the Chinese soil standard as a benchmark (SEPAC 1995), the average Hg concentration in the Yangtze River Delta was higher than the background level, but the average Hg concentration in the Pearl River Delta was even higher than the second level of the standard, indicating potential risks to the local soil ecosystem.
3.2 Depth distribution of Hg in typical soil types
Some typical soil profiles were selected to present the depth distribution of soil Hg concentration (Fig. 1). The depth distributions of soil Hg concentration varied greatly between the two regions and among the soil types. In the case of Stagnic Anthrosols which was distributed widely in both regions, a dramatic decrease in soil Hg concentration occurred from the subsoil to bottom layer in the YRD region, while slight change occurred from topsoil to bottom layer in the same soil type in the PRD region. This trend is also supported by average horizontal concentration of the soil type in Table 1. The Hg concentration ratio of A/B/C (A, B, and C indicates topsoil, subsoil, and bottom layer, respectively) in the Stagnic Anthrosols of the Yangtze River Delta was 6.0:3.6:1 based on the data of Table 1 and was 2.9:2.3:1 for the Pearl River Delta. Multiple comparisons among the pedogenic horizons of the Stagnic Anthrosols also show that the Hg concentrations in the topsoil and subsoil were significantly (p < 0.05) higher than those in the bottom layers in the Yangtze River Delta, but there was no significant difference among the horizons in the Stagnic Anthrosols of the Pearl River Delta. Correspondingly, the depth distribution of SOM content in the YRD soil was much different to the PRD soil as shown in Table 1. The highest SOM content occurred in the topsoil and decreased dramatically with depth in the YRD soil; in contrast, no significant difference could be observed among the different horizons of the PRD soil. This implied that the depth distribution of Hg in the Stagnic Anthrosols was mainly determined by soil organic matter content. Meanwhile, lower soil pH and higher rainfall in the PRD region tended to enhance the mobilization of Hg from topsoil to bottom layer (Huang et al. 2011). In addition to the Stagnic Anthrosols, depth distributions of Hg in Aquic Cambisols, Perudic Cambisols, Udic Ferrisols, Udic Luvisols, Anthric Entisols, and Sandic Entisols were all associated with SOM content, although small variation of Hg concentration in the profile of these soils was observed.
Depth distribution of Hg in the Orthic Halosols was very different from the other soil types. The typical soil profile of the Orthic Halosols was collected from the land where there was a saltmarsh prior to cultivation in the PRD. The topsoil of the Orthic Halosols had values up to 56 mg/kg and changed gradually from the topsoil to the third layer. A sharp decrease occurred from the third layer to the fourth layer as shown in Fig. 1. The depth distribution of Hg concentration in this soil type was associated with the pH value rather than SOM content, especially the extremely low concentration of Hg corresponding to the extremely low pH (pH <3) in the bottom layers. This was probably due to the increase of solubility of the Hg organic complex under low pH condition. Ravichandran (2004) has summarized the conditional stability constant (K) of Hg organic complexes from the literature, in which the K value is usually lower than 1010 at low pH (pH <5), while the K value is as high as 1028.5 at pH of 8.
In contrast to the decrease of Hg with depth in most soils, the depth distribution of Hg increased with depth in the Udic Ferralsols although SOM content of the soil decreased from topsoil to bottom layer (Fig. 1 and Table 1). Mercury remobilization could explain the lower concentration of Hg in the topsoil of the Udic Ferralsols. Hg remobilization can occur after the dissolution or erosion of Hg-bearing phases (such as iron oxides) under temporal reducing condition (Grimaldi et al. 2008).
3.3 Soil pools of anthropogenic Hg
Soil pools of anthropogenic Hg at a depth of 100 cm ranged from 0.70 to 6971.36 g/ha in the Yangtze River Delta, lower than that in the Pearl River Delta which ranged from 35.8 to 13,098.7 g/ha. The soil Hg pool was further illustrated by cumulative frequency distribution curves. As shown in Fig. 2, the inflection point was determined at 838.4 and 925.3 g/ha for the soil Hg pool to a depth of 100 cm in the Yangtze and Pearl River Deltas, respectively. These two values did reflect the 79 and 75 % percentiles, respectively. For the soil Hg pool to a depth of 40 cm, the inflection point was 670.9 and 839.2 g/ha, reflecting the 76 and 71 % percentiles, respectively. The value beyond the inflection point usually implies that the sampling sites might be contaminated by some specific sources in addition to atmospheric precipitation. Therefore, these data were not included in the calculation of the average pool of the whole region (Zhang et al. 2007).
Cumulative frequency distribution curves showing the data distribution of the calculated soil Hg pool for depths of 40 and 100 cm. YRD_40 and PRD_40 indicate the soil Hg pool in the top 40-cm soil of the two regions, respectively; YRD_100 and PRD_100 indicate the soil Hg pool in the whole soil profile of the Yangtze River Delta (YRD) and Pearl River Delta (PRD), respectively
The differences in soil pools of anthropogenic Hg between the two regions were correlated to their Hg emissions as estimated by Cheng et al. (2009) and Zheng et al. (2011). The annual emission of Hg was estimated at 3.38 g/ha in the Yangtze River Delta and 4.15 g/ha in the Pearl River Delta (Table 3). In terms of the average soil Hg pool of the whole region, the emission ratio (0.81) between the Yangtze and Pearl River Deltas was quite similar to the ratio of the soil Hg pool (0.71). This implies that atmospheric deposition from localized sources did account for most of the soil Hg pool. The soil pool of anthropogenic Hg was very different among the different soil types. As shown in Table 3, Perudic Cambisols of the Yangtze River Delta had the highest soil Hg pool followed by Stagnic Anthrosols of the Pearl River Delta. The former developed in high mountains with high humidity and soil organic matter contents (Table 1), but the latter developed from long-term flooded rice cultivation. The humid and reducing conditions would facilitate the sequestration of anthropogenic Hg in the soils (Rothenberg and Feng 2012; Schuster 1991).
3.4 Influence of soil properties
Linear regression analysis was performed to show the relationships between soil Hg concentrations and soil properties (Fig. 3). The data were selected based on Hg concentrations < 0.4 mg/kg in the topsoil of the two regions. Hence, all the selected data reflected the absence of severe contamination at the sites in addition to atmospheric deposition. As shown in Fig. 3, significant (p < 0.01) positive correlations were found between the soil Hg concentrations and soil properties (SOM, humic acid, CEC, and amorphous iron oxides).
The amount of Hg binding in the soil depended on the sequestration of anthropogenic Hg in soil. There are several mechanisms to govern the binding of Hg in the soils, including nonspecifically adsorbed, specifically adsorbed, chelated, and precipitated (Schuster 1991). The significant correlation between Hg concentration and SOM, humic acids, CEC, and amorphous iron oxides suggested that chelated with humic substance and specifically adsorbed to the hydrous ferric oxides (HFO) might be the dominant mechanisms of Hg sequestration in soils of the YRD and the PRD. Mercury exhibits a great affinity for organic matter in soils by forming complexes with reduced sulfur groups (Skyllberg et al. 2006; Wallschlager et al. 1998). Humic acids (HAs) are major constituents of soil organic matter, containing complex mixtures of many different acids including carboxyl and phenolate groups. Therefore, the humic acids could form stable complexes with Hg in soil. Zhang et al. (2009) have reported a higher adsorption capacity and lower desorption of Hg2+ by humic acids than by fulvic acids. The reason might be attributed to the chemisorption capacity of humic acids, but not to fulvic acids (Wallschlager et al. 1996). Gu et al. (2011) reported that strong interactions can be observed between reduced HAs and Hg(0) under anoxic environment, and Hg(0) is the major speciation of atmospheric Hg which accumulates in topsoil through dry and wet deposition.
The specific adsorption of Hg on the hydrous ferric oxides could be explained by the concept of surface complexation (Schuster 1991). Liang et al. (2013) have compared the adsorption capacity for Hg2+ between HFO and goethite and found that the maximum Hg2+ adsorption (S max) was higher for HFO due to its higher specific surface area. The mechanisms for the adsorption of Hg by HFO include the formation of ternary surface complexes and surface precipitation at high sorbate/sorbent ratio (Tiffreau et al. 1995). Therefore, the Perudic Cambisols and Stagnic Anthrosols which contained a relatively higher amorphous iron oxides content in the soils had a relatively higher soil Hg pools than other soil 3).
4 Conclusions
The measured Hg concentrations in the topsoils of the Yangtze River Delta and Pearl River Delta ranged from 0.054 to 1.78 mg/kg and from 0.028 to 1.91 mg/kg, respectively. The difference in soil Hg between the two regions was associated with the amount emitted as well as soil properties. In the case of soil properties, soil organic matter, in particular of humic acid, was supposed to be the dominant factor to influence the spatial distribution and depth distribution of soil Hg. Meanwhile, amorphous iron oxides (e.g., HFO) enhanced retention of Hg in the soils by forming surface complexes. This could be reflected from the higher soil Hg pools of Stagnic Anthrosols and Perudic Cambisols which had higher content of amorphous iron oxides as well. In addition, the soil that is widely distributed in the coastal zone of the PRD and characterized by low pH and high salinity had a low soil Hg pool in general as a result of high remobilization of soil Hg under such conditions.
References
Burt R, Wilson MA, Mays MD, Lee CW (2003) Major and trace elements of selected pedons in the USA. J Environ Qual 32(6):2109–2121
Chen LG, Xu ZC, Ding XY, Zhang WD, Huang YM, Fan RF, Sun JR, Liu M, Qian DL, Feng YB (2012) Spatial trend and pollution assessment of total mercury and methylmercury pollution in the Pearl River Delta soil, South China. Chemosphere 88(5):612–619
Cheng K, Xue ZG, Zhang ZQ, Li H, Yi P, Wang YH, Xu YX (2009) Emission and control of air pollutants in major industries of Yangtze Delta. Environ Sci Technol (China) 32(9):120–123
Feng XB, Qiu GL (2008) Mercury pollution in Guizhou, Southwestern China—an overview. Sci Total Environ 400(1–3):227–237
Grigal DF (2002) Inputs and outputs of mercury from terrestrial watersheds: a review. Environ Rev 10(1):1–39
Grimaldi C, Grimaldi M, Guedron S (2008) Mercury distribution in tropical soil profiles related to origin of mercury and soil processes. Sci Total Environ 401(1–3):121–129
Gu BH, Bian YR, Miller CL, Dong WM, Jiang X, Liang LY (2011) Mercury reduction and complexation by natural organic matter in anoxic environments. P Natl Acad Sci USA 108(4):1479–1483
Hammond AL (1971) Mercury in environment—natural and human factors. Science 171(3973):788–789
Huang BA, Wang M, Yan LX, Sun WX, Zhao YC, Shi XZ, Weindorf DC (2011) Accumulation, transfer, and environmental risk of soil mercury in a rapidly industrializing region of the Yangtze River Delta, China. J Soils Sediments 11(4):607–618
IUSS Working Group WRB (2006) World Reference Base for Soil Resouces. 2nd edition. World Soil Resources Reports No. 103. FAO, Rome, 128p
Jiang GB, Shi JB, Feng XB (2006) Mercury pollution in China. Environ Sci Technol 40(12):3672–3678
Liang P, Li YC, Zhang C, Wu SC, Cui HJ, Yu S, Wong MH (2013) Effects of salinity and humic acid on the sorption of Hg on Fe and Mn hydroxides. J Hazard Mater 244:322–328
Lin YH, Guo MX, Gan WM (1997) Mercury pollution from small gold mines in China. Water Air Soil Poll 97(3–4):233–239
Lin Y, Vogt R, Larssen T (2012) Environmental mercury in China: a review. Environ Toxicol Chem 31(11):2431–2444
Lu R (2000) Analytical methods for soil and agricultural chemistry (In Chinese). China Agricultural Science and Technology Press, Beijing
Luo L, Ma Y, Zhang S, Wei D, Zhu YG (2009) An inventory of trace element inputs to agricultural soils in China. J Environ Manage 90(8):2524–2530
Pirrone N, Cinnirella S, Feng X, Finkelman RB, Friedli HR, Leaner J, Mason R, Mukherjee AB, Stracher GB, Streets DG, Telmer K (2010) Global mercury emissions to the atmosphere from anthropogenic and natural sources. Atmos Chem Phys 10(13):5951–5964
Qiu GL, Feng XB, Wang SF, Shang LH (2006) Environmental contamination of mercury from Hg-mining areas in Wuchuan, northeastern Guizhou, China. Environ Pollut 142:549–558
Ravichandran M (2004) Interactions between mercury and dissolved organic matter—a review. Chemosphere 55:319–331
Rothenberg SE, Feng XB (2012) Mercury cycling in a flooded rice paddy. J Geophys Res-Biogeo 117, G03003
Scholtz MT, Van Heyst BJ, Schroeder W (2003) Modelling of mercury emissions from background soils. Sci Total Environ 304(1–3):185–207
Schuster E (1991) The behavior of mercury in the soil with special emphasis on complexation and adsorption processes—a review of the literature. Water Air Soil Pollut 56:667–680
Shi JB, Meng M, Shao JJ, Zhang KG, Zhang QH, Jiang GB (2013) Spatial distribution of mercury in topsoil from five regions of China. Environ Sci Pollut Res 20(3):1756–1761
Skyllberg U, Bloom PR, Qian J, Lin CM, Bleam WF (2006) Complexation of mercury(II) in soil organic matter: EXAFS evidence for linear two-coordination with reduced sulfur groups. Environ Sci Technol 40(13):4174–4180
Smith-Downey NV, Sunderland EM, Jacob DJ (2010) Anthropogenic impacts on global storage and emissions of mercury from terrestrial soils: insights from a new global model. J Geophys Res-Biogeo 115, G03008
State Environment Protection Administration of China (SEPAC) (1995) Environmental quality standards for soils (GB 5618-1995). Available at: http://english.mep.gov.cn/standards_reports/standards/Soil/Quality_Standard3/200710/W020070313485587994018.pdf
State Environment Protection Administration of China (SEPAC) (1997) Soil quality—determination of total mercury-cold atomic absorption spectrophotometer (GB/T 17136-1997)
Streets DG, Hao JM, Wu Y, Jiang JK, Chan M, Tian HZ, Feng XB (2005) Anthropogenic mercury emissions in China. Atmos Environ 39(40):7789–7806
Tiffreau C, Lutzenkirchen J, Behra P (1995) Modeling the adsorption of mercury(II) on (hydr)oxides.1. Amorphous iron-oxide and alpha-quartz. J Colloid Interface Sci 172:82–93
Tipping E, Poskitt JM, Lawlor AJ, Wadsworth RA, Norris DA, Hall JR (2011a) Mercury in United Kingdom topsoils; concentrations, pools, and critical limit exceedances. Environ Pollut 159(12):3721–3729
Tipping E, Wadsworth RA, Norris DA, Hall JR, Ilyin I (2011b) Long-term mercury dynamics in UK soils. Environ Pollut 159(12):3474–3483
Wallschlager D, Desai MVM, Wilken RD (1996) The role of humic substances in the aqueous mobilization of mercury from contaminated floodplain soils. Water Air Soil Pollut 90(3–4):507–520
Wallschlager D, Desai MVM, Spengler M, Windmoller CC, Wilken RD (1998) How humic substances dominate mercury geochemistry in contaminated floodplain soils and sediments. J Environ Qual 27(5):1044–1054
Wu Y, Wang SX, Streets DG, Hao JM, Chan M, Jiang JK (2006) Trends in anthropogenic mercury emissions in China from 1995 to 2003. Environ Sci Technol 40(17):5312–5318
Yang YK, Zhang C, Shi XJ, Lin T, Wang DY (2007) Effect of organic matter and pH on mercury release from soils. J Environ Sci-China 19(11):1349–1354
Yin XB, Yao CX, Song J, Li ZB, Zhang CB, Qian W, Bi D, Li CX, Teng Y, Wu LH, Wan HD, Luo YM (2009) Mercury contamination in vicinity of secondary copper smelters in Fuyang, Zhejiang Province, China: levels and contamination in topsoils. Environ Pollut 157(6):1787–1793
Zhang HB, Luo YM (2011) Endogenous and exogenous lead in soils of Yangtze River Delta region, China: identified by lead isotopic compositions and multi-elemental approaches. Environ Earth Sci 62(5):1109–1115
Zhang L, Wong MH (2007) Environmental mercury contamination in China: sources and impacts. Environ Int 33(1):108–121
Zhang HB, Luo YM, Wong MH, Zhao QG, Zhang GL (2007) Defining the geochemical baseline: a case of Hong Kong soils. Environ Geol 52(5):843–851
Zhang J, Dai JL, Wang RQ, Li FS, Wang WX (2009) Adsorption and desorption of divalent mercury (Hg2+) on humic acids and fulvic acids extracted from typical soils in China. Colloid Surface A 335(1–3):194–201
Zheng JY, Ou JM, Mo ZW, Yin SS (2011) Mercury emission inventory and its spatial characteristics in the Pearl River Delta region, China. Sci Total Environ 412:214–222
Acknowledgments
The authors are grateful for financial support from the Key Research Program of the Chinese Academy of Sciences (KZZD-EW-14) and the National Natural Science Foundation of China (NSFC) (41230858, 41371313).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Jean-Paul Schwitzguébel
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 26 kb)
Rights and permissions
About this article
Cite this article
Zhang, H., Li, Y., Luo, Y. et al. Anthropogenic mercury sequestration in different soil types on the southeast coast of China. J Soils Sediments 15, 962–971 (2015). https://doi.org/10.1007/s11368-015-1062-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-015-1062-1