Abstract
Municipal wastewater has been considered as one of the largest contributors and carriers of microplastics to the aquatic environment. However, the various residential activities that generate municipal wastewater are equally significant whenever the source of microplastics in aquatic system is accounted. However, so far, only municipal wastewater has received wide attention in previous review articles. Hence, this review article is written to address this gap by highlighting, firstly, the chances of microplastics arising from the usage of personal care products (PCPs), laundry washing, face masks, and other potential sources. Thereafter, the various factors influencing the generation and intensity of indoor microplastic pollution and the evidence available on the possibility of microplastic inhalation by humans and pet animals are explained. Followed by that, the removal efficiency of microplastics observed in wastewater treatment plants, the fate of microplastics present in the effluent and biosolids, and their impact on aquatic and soil environment are explored. Furthermore, the impact of aging on the characteristics of microsized plastics has been explored. Finally, the influence of age and size of microplastics on the toxicity effects and the factors impacting the retention and accumulation of microplastics in aquatic species are reviewed. Furthermore, the prominent pathway of microplastics into the human body and the studies available on the toxicity effects observed in human cells upon exposure to microplastics of different characteristics are explored.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Bakelite is the first synthetic polymer, discovered in the year 1907. The interesting properties of plastics such as light weight, corrosive resistance, and low cost have resulted in their widespread application and enormous production (Frias and Nash 2019). However, owing to the mismanagement of plastic waste, plastic has become an environmental threat. Based on the estimate of plastic production recorded in 2010 for a total of 192 coastal countries (275 million metric tons (MT)), it is projected that 4.88 to 12.7 million MT of plastic enter the ocean annually. Under improper waste management infrastructure, this estimate is projected to increase further by an order of magnitude by 2025 (Jambeck et al. 2015). On the other hand, based on the estimate of 2010, the overall weight of plastics in oceans is expected to exceed the amount of fish present in the sea by 2050 (Picó and Barceló 2019). As of 2020, the estimate of global plastic production is 367 million MT which is excluding the production of common synthetic fibres (polyethylene terephthalate (PET), polyamide (PA) and polyacrylate) (Plastics 2021). Therefore, the projected estimate of plastic waste reaching the ocean would be more when the production of those synthetic fibres is taken into account.
These plastic wastes, upon reaching the environment, get exposed to various physical, photoradiative, and biological actions, which result in their breakdown and the eventual formation of microplastics (Meng et al. 2021). Microplastics (< 5 mm in size) are the most significant pollutant of concern in recent years owing to their accumulative and toxic potential (Ma et al. 2020). Among the various sources that are polluting the environment with microplastics, municipal wastewater is reportedly one of the biggest sources (Browne et al. 2011). With respect to the constituents of municipal wastewater, some of the common household activities that generate municipal wastewater include vessel washing, laundry washing, bathing, and toilet usage (Delhiraja and Philip 2020). When all these activities are considered individually, each activity involves the usage of either plastics or primary microplastics. For instance, dish washing predominantly involves the usage of a plastic-based scouring pad. The softer side of the dishwashing sponge is made of polyurethane (PU) (Zimmermann et al. 2020), and the mesh attached to it to enhance the scouring action is made of polyethylene (PE) (Institute of Making (UC London) 2013). Over time, the visible reduction in the volume and size of the sponge indicates the loss of plastic material, which otherwise can be considered as generation of secondary microplastics. On the other hand, laundry washing releases a humongous number of microfibres into the wastewater (Henry et al. 2019). Furthermore, bathing and hygienic activities include the usage of PCPs like shower gels, face cleansers, and toothpaste that contain microbeads made of synthetic polymers (Bashir et al. 2021). Usage of flushable and non-flushable wipes in toilets was observed to result in microplastic pollution as well (Ó Briain et al. 2020).
During wastewater treatment, a significant proportion of microplastics present in the wastewater are being translocated from water to the solid phase (biosolids). However, owing to their small size, a large number of microplastics escape the treatment stages and remain in the treated effluent, eventually entering into the aquatic system upon discharge (W. Liu et al. 2021). As they stay in the environment, owing to their larger surface area and hydrophobicity, they tend to adsorb other co-existing pollutants (Fu et al. 2021). In addition to adsorption, toxic compounds are reported to leach out from the microplastics over time, thereby intensifying the impact of pollution (Meng et al. 2021). On the other hand, the treated sludge called biosolids are widely applied as a soil amendment in the agricultural fields, and such practise is notably disseminating the microplastics into the soil environment (Liu et al. 2021). The translocated microplastics further contribute to toxicity in aquatic species (Paul-Pont et al. 2016) and affect plant growth and the soil microbe community (Ren et al. 2021). Apart from the presence of microplastics in domestic wastewater, the primary and secondary microplastics generated from household plastic products and activities also result in the contamination of the air (Soltani et al. 2021). Therefore, it is clear that residential houses are the most significant source for microplastic pollution. However, to the best of our knowledge, the authors could not find any review article that cumulatively discusses the various activities and products of residential buildings that act as prime sources of microplastics.Therefore, this article will give the readers a glimpse of different prominent and unnoticed sources of microplastics associated with residential buildings, followed by their fate, transport, transformation, and toxicity effects in aquatic organisms and human beings.
Various sources of microplastics originating from residential households
Personal care products and cosmetics
Microplastics are an essential part of PCPs and cosmetics formulation. The primary microplastics of different sizes, shapes, and colour that are deliberately used as ingredients in the formulation of PCPs are termed as microbeads. It is to be noted that microbeads need not be spherical in shape but can be of any other shape (Anagnosti et al. 2021). Microbeads are widely used for cleansing and exfoliation purposes. Besides, the manufacturers use microbeads to increase product volume and aesthetics and to control viscosity. They find their use widely in PCPs such as face wash, shower gels, eyeliners, nail polish, sunscreen, hairspray, and toothpaste (Anagnosti et al. 2021). They are manufactured using polymers such as polypropylene (PP), PET, polymethyl methacrylate, nylons, PU, and PE, which is the most common one (Bashir et al. 2021). Microbeads have been identified in the PCPs available in the markets of different countries. However, comparative analysis of the size distribution reported in various studies would not be possible as the minimum detectable size is limited by the specifications of each microscope used in different studies. Concerning the other characteristics of those microbeads, it was observed that the colour and polymer type of microplastics present in various PCPs are not noted to be significantly different with respect to different countries. In a study conducted in Macao, China, Bashir et al. (2021) have observed that 69% of the 144 PCPs (facial care, body care and cosmetics) chosen for the study contained at least one type of microplastics. Among the samples containing microplastics, 76% had PE as the primary ingredient. Though packaging materials are reportedly the biggest source of PE-based microplastics (Jessieleena and Nambi 2023), from this review, it is realized that PCPs could also be another biggest source of PE-based microplastics. The size distribution was made for the three top-selling products (facial cleanser and scrubs), and the size was noted to vary between 11 and 968 μm. The colour of microbeads observed in those three products are white, colourless (white-transparent), green, and brown (Bashir et al. 2021). Most of the packaging materials are also white or transparent, and this further tells that the sources of PE could sometimes be mistaken as packaging material for the actual source is PCP especially during cases when the microbeads have lost its spherical shape due to abrasion occurred during their use. Another survey was conducted in the supermarkets in Beijing, China, and it was observed that approximately 7.1% of the facial cleansers (n = 126) and 2.2% of shower gel products (n = 136) under survey contained microplastics in their ingredients. The average size of microbeads noted in facial and shower gels was 313 and 422 µm, respectively, and the microbeads were predominantly white or colourless. Spectroscopic analysis has confirmed PE as the widely present polymer type in the microplastics identified (Lei et al. 2017). The results of this survey were similar to that of the previous study conducted in China; however, the proportion of products containing microplastics is largely different between both studies. Praveena et al. (2018) have explored the presence of microplastics in the top 10 PCPs used in Malaysia and have identified low density PE and PP as the common polymer used in facial scrubs and low density PE in case of toothpaste. The size was noted to be in the range of 10 to 178 μm in facial scrub and 3 to 145 μm in toothpaste, and colourless microbeads were observed in 9 out of the 10 PCPs. In this study, in addition to PE, PP was also identified and, hence, PCPs could be another source of PP-based microplastics which are also considered to be originating from packaging materials (Kashfi et al. 2022). Microplastics were also identified in the commercially available toothpastes of India. The size of the microplastics identified varied from 3.5 µm and went beyond 400 µm. Of the microplastics identified, around 33 to 83% of the microplastics are observed to be colourless. PP, polyvinyl chloride (PVC), PA, and cellophane are the commonly observed polymer types in the microplastics identified (Madhumitha et al. 2022). In the top American skincare products, the size range of microbeads was identified to be 60 to 800 µm (Chang 2015). A survey conducted in Spain found out that microplastics were present in all four categories of products considered in this study, namely bath gel, body, foot, and facial scrubs. In some products, the microplastics accounted for 6 to 7% of the total product weight. The lowest and highest concentrations reported were 123 and 3730 particles/g of the product under study, and the size was noted to vary between 8 µm and 2 mm. Most of the microplastics identified in these samples match with PE spectrum of FTIR analysis and are predominantly colourless (Godoy et al. 2019). Similar to the previous observation, products such as facial scrubs and bath gel are found to have only one type of microplastics and it could be possibly from the beads added for abrasive action. However with respect to toothpastes, it is a variety of microplastics. This draws an other observation that the microplastics identified in the toothpastes could be largely coming from the wear/tear of the multilayer based plastic cover (present at the tube mouth before use) rather than any microbeads that are deliberately added in the toothpastes. Also, from the above-described studies, it is understood that white/colourless microbeads are the dominant ones, and upon their entry into the aquatic environment, the colourless microplastics pose a higher risk of consumption by aquatic species than other coloured microplastics due to difference in visibility.
Concerning the presence of microbeads in realtime aquatic environment, a study solely conducted to analyse microbead pollution in 2015 in the marine system of Hongkong has confirmed the presence of blue microbeads. It was identified to have originated from facial scrubs and the size varied from 332 to 1015 µm. However, spectroscopic analysis has shown that they are made of wax and not synthetic polymers. But it was suggested that though the wax can be of natural origin, it has a tendency to adsorb other organic pollutants, similar to the behaviour of microplastics (Cheung and Fok 2016). However, this observation also suggests that chemical characterization of visually identified microplastics is very essential to avoid overestimation of microplastic pollution. Another study was conducted in the seawater of Hongkong between 2016 and 2017, and it was found that 3.6% of the overall microplastics identified was accounted for microbeads (So et al. 2018). Only 0.3- to 1-mm-sized microplastics were considered in this study, and microbeads in this size range were detected in approximately 60% of the samples. In contrast to the previous study (Cheung and Fok 2016), the spectroscopic analysis in this study has shown that all the 95 microbeads which are randomly chosen for the analysis were observed to be synthetic, with PE being the dominant (55%) followed by PP (27%), polystyrene (PS) (11%), and mixed polymers. Around 62% of the microbeads identified were observed to be transparent (colourless) (So et al. 2018). Based on the results of this study, a similarity could be observed in the characteristics of microbeads present in commercial products and real-time aquatic systems. Therefore, it is further confirmed that microbeads present in the PCPs of households are being transported into aquatic systems and polluting them. Nonetheless, it should be ensured that representative number of visually identified microplastics are being chemically characterized before arriving at a conclusion. Estimations suggest that 307 MT of microbeads get released into Mainland China yearly from using facial scrubs alone (Cheung and Fok 2017). On the other hand, shower gel products alone are reported to release 39 MT of microplastics into the environment of China (Lei et al. 2017). With respect to Malaysia, 0.199 trillion microplastics were estimated to be released annually into the marine environment due to the usage of personal care products and cosmetics (Praveena et al. 2018). The microplastics arising from toothpaste usage in India is estimated to release 1.4 billion g microplastics into the environment annually (Madhumitha et al. 2022). Similarly, Ustabasi and Baysal (2019) have estimated the release of 871 million g of microplastics annually from toothpaste usage in Istanbul, Turkey (Ustabasi and Baysal 2019). Owing to its persistent and toxic nature, many countries have either implemented or in the process of implementing a complete ban on plastic microbeads usage in PCPs. In the year 2018, the US government made the usage of microbeads illegal (5Gyres 2022). With respect to European countries, in the year 2021, the European Union has pledged to ban microbeads usage in several products like paints, medical products including detergents, and cosmetics. This has been proposed to prevent an enormous amount, i.e. 500,000 MT of microplastics from entering the environment (European Environmental Bureau (EEB) 2021). Countries like India and China have also taken initiatives to ban the usage of microbeads in rinse-off cosmetic products (Chak 2020). Therefore, a global ban of microbeads usage in PCPs is expected in the near future. However, through this review, it is noted that though there are numerous studies reporting the presence of microbeads in PCPs, a standardized method to report the concentration of microplastics is yet to be established. This is because currently it is reported in terms of number or weight concentration, and this non-uniformity in the unit of expression would make it difficult to frame policies on prevention and control measures.
Microplastics in laundry wastewater
One of the primary sources of microplastics originating from households is the synthetic fibers released during textile washing. Since these microplastics are fibrous, they are termed as microfibres. During washing, the fabrics undergo mechanical and chemical stresses in a laundry machine which releases microfibres. It was reported that the presence of shredded fibres in the laundry effluent has a substantial contribution towards microplastic pollution (Henry et al. 2019). Therefore, it is crucial to focus on the laundry process to reduce the overall microplastics’ impact on the environment. Galvão et al. (2020) reported that 18 million synthetic microfibres was released for a 6 kg wash load, i.e. 30 lakh microfibres/kg. In other studies, synthetic microfibre release rate ranged from 23,333 (Napper and Thompson 2016) to 35.4 × 105 microfibres/kg of washed clothes (De Falco et al. 2018). The vast difference noted in the case of the number concentration could be attributed to various factors such as temperature, age of fabric, type of washing machine, detergents, fabric type, and wash conditions. This is because it was reported that the microfiber emissions from top-load type washing machines were seven fold higher than front-load machines (Hartline et al. 2016). This could be because of the fact that top load washing machines are harsher in action than the front load ones, and this could be very well related to our experiences in our daily life dealing with these two types of washing machines. Another factor influencing the microplastic release from textiles is the type of synthetic fabric. In a study conducted by De Falco et al. (2018), it was observed that woven polyester released more microfibres when compared to woven PP and knitted polyester during machine wash. The lowest release of microfibres can be obtained by using softeners which has the ability to reduce friction among the fibres (De Falco et al. 2018). It has also been stated that powder detergent and higher water hardness increase the release of microfibres (O’Brien et al. 2020). According to Hartline et al. (2016), garment aging had a significant effect on microplastics release and it was reported that the older garments release more microfibres than the newer ones. It is to be noted that abrasion results in the release of microfibres, and since aged/old garments would have been subjected to more abrasion than new ones, old garments tend to release more microfibres than new ones. Washing at higher temperatures reportedly stimulates the release of microfibres (Cotton et al. 2020). The enormous amount of microfibres present in the laundry water (Galvão et al. 2020) could not be completely removed in the wastewater treatment processes (Liu et al. 2021). Thereby, it ultimately reaches the aquatic systems, including seas and oceans, through treated effluent or illegal sewage discharges. Hence, all these influencing variables should be considered while developing methods to reduce microplastic pollution. A filtering device namely XFiltra filter, which is installed in the washing machine’s outlet pipe, was observed to reduce the microfibre emission by 78% (Napper et al. 2020). Nevertheless, complete stoppage of microplastic emission from washing machines should be ensured in the future through technological innovations. Apart from the number of microplastics, their size is another key variable for assessing their effect on the environment. This is because smaller microplastics are more easily mistaken for prey by the aquatic species than the larger microplastics (Setälä et al. 2014). Besides, smaller microplastics also tend to escape wastewater treatment plant than larger ones (Liu et al. 2021). But, it is observed that though microfibres from laundry water constitute about 35% of the overall primary microplastics present in oceans (Boucher and Friot 2017), microfibres in laundry water has gained attention among the researchers only in the recent years and the year-wise publication data is given in Fig. 1. Among the available literature, the common methodology adapted to understand the occurrence of microfibres is to run washing machine with specific types of clothes and specific washing conditions so as to detailly investigate the impact of different parameters on the release of microfibres. However, it is also equally important in the future to conduct detailed investigation on the characteristics of microfibres present in the real-time laundry water coming out from a domestic household. This would signify the magnitude of microplastic pollution caused due to laundry water.
Other potential sources
Owing to the Covid-19 pandemic, the manufacturing and usage of face masks have increased tremendously. By the end of 2020, the annual use of masks has risen from 1.1 to 2 billion (Gereffi 2020). Over time, processes such as aquatic immersion, photodegradation, and other weathering processes are causing the fragmentation of macroplastics in the mask into micro sizes (Yang et al. 2020). Hence, single-use facemasks that end up in the landfills, dumpsites, oceans, and other public areas are a new source of microplastics. Currently, the research around microplastics and face mask is oriented towards understanding the release of microplastics from facemask and few studies are available on the toxicity effects on aquatic and terrestrial organism. However, above all these, human beings are the most susceptible ones and hence more research on the possibility of direct inhalation of microfibres by human beings using modified manikins is essential. Secondly, polyester, PE, and PA fabrics are known to be used for carpet manufacturing, along with polyacrylic as carpet thickener (Can 2007; Hari 2012; Du et al. 2016) and a study conducted by Soltani et al. (2021) confirmed the release of microfibres from carpets of similar polymer type. In the samples analysed, it was found that houses with carpeted floors have approximately twice the amount of polyester, PA, polyacrylic, and PE-based microfibres compared to non-carpeted houses. Scopus database with a search term ‘microplastics AND carpet’ has returned only 6 research articles (accessed on March 2023), and hence, more research needs to be carried out in this field as well since carpets are an integral part of indoor furnishings in majority of the regions. Shedding from other indoor fabrics, including indoor clothes and furnishings, is the other important but unnoticed source of microplastics (Vianello et al. 2019; Kashfi et al. 2022). Nextly, the glitters for artworks are found to be the biggest source of microplastics. Apart from its usage in craft works, glitters are commonly used in cosmetics and textile products as well. Most of the glitters are made up of metalized PET and they are one of the sources of primary microplastics. They are observed to pose more potential risk than microbeads since glitters have wider applications and a broader target audience, including children (Yurtsever 2019). Besides, scouring pads made of PE, nylon, and PU would also be expected to release microplastics over their usage during dishwashing and their consumption is huge and inevitable (Lassen et al. 2015; Zimmermann et al. 2020). For instance, 30 to 90 million scouring pads are consumed annually in Denmark. Due to the abrasion occurring over time, around 10 to 100 MT of microplastics are estimated to be released annually, of which 90 to 100% are expected to be released into sewage (Lassen et al. 2015). A recent study has indicated that synthetic grass sheets commonly made of recycled PE are also a probable source of microplastics (Mehmood and Peng 2022). These synthetic grass sheets are often used to improve the aesthetics of the balcony, and in some houses, the balcony is a place to have refreshing beverages and snacks. Therefore, the risk posed by these sources of microplastics is further increasing as the mode of exposure is both inhalation and ingestion. Recently, an experimental investigation conducted by Fang et al. (2022) has found that the exposure to microplastics through food contaminated with atmospheric microplastics is similar to that of inhalation and 2 to 3 times more than the exposure through food sources. Some of the other microplastics sources which has a direct impact on human beings include tea bags, beverage, and food containers. A study conducted by Hernandez et al. (2019) has reported that steeping a single plastic tea bag has released an average of 11.6 billion microplastic particles into the tea at brewing temperatures. On the other hand, four different beverages belonging to 27 different brands were selected for microplastic contamination studies in Mexico. Around 84% of the samples tested were contaminated with either microplastic fibres or fragments. Spectroscopic studies have confirmed the polymer type, and the cause of contamination was found to be synthetic textiles and packaging products. Since these microplastics present in beverages are expected to enter humans through ingestion, human faeces was reported to play a vital role in the dispersion and accumulation of microplastics in aquatic and terrestrial environments (Shruti et al. 2020). Fadare and Okoffo (2020) have explored the possibility of microplastics release from commonly used food packaging containers and disposable plastic cups (Fadare and Okoffo 2020). Ten millilitres of ultrapure water was added to these products and was mechanically shaken for approximately 6 min. The water was then analysed for microplastics, and it was found that 7.6 and 0.06 mg of microplastics were estimated to be released per plastic container and disposable cups, respectively. Another group of researchers has performed a similar study by simulating various eating conditions using food packaging containers. The simulation studies reported that approximately 3 to 29 particles were released per container (Du et al. 2020).
Apart from the above different usage of plastic products that are not commonly considered toxic to human beings, it is well known that smoking tobacco is unhealthy. But, the harmful effects are not limited to humans but extend to the environment since cigarette butts are one of the most found garbage in clean-up campaigns. Each cigarette filter is reported to contain more than 15,000 cellulose acetate fibre strands, along with plasticizers. The detachment rate of these fibres was found to be 100 microfibres/day of size less than 200 microns. A rough estimate of 0.3 million microfibres is found to have the potential to reach aquatic environments from this source alone. Belzagui et al. (2021) reported that abundant artificial polymers identified in the deep-sea sediments would have originated from cigarette butts. These microfibres are speculated to affect the atmospheric quality as well (Belzagui et al. 2021). Therefore, all these overlooked sources would add up to microplastic pollution and should be managed appropriately. Therefore, besides the commonly explored sources of microplastics like PCPs, textiles, and masks, more research needs to be carried out to investigate the behaviour of microplastics release from all the other above mentioned potential but less explored sources.
Microplastics from exposed household plastics
With respect to domestic households, any plastic product having long exposure to sunlight is susceptible to photooxidation. One such product of significance is the rainwater harvesting filter. Of late, manufacturers are coming up with compact rainwater harvesting filter systems of different sizes for rooftop areas from 1000 to even upto 10,000 sq.ft. However, all these systems are made of plastics like PVC and HDPE and are kept outside in the open atmosphere (Rainwayfilters 2022; Rainyfilters 2022). On the other hand, in the case of larger storage capacity, typical rainwater collection facilities include inspection well, storage tank, and pipeline. These are also made of HDPE, PP, and PVC plastics. To explore the leaching of microplastics from these facilitites, 45-day UV aging followed by 72 h hydraulic scouring experiment was conducted by Zhang et al. (2022). The release of microplastics was observed to increase with aging, and at the end of experiment, around 160 to 1905 microplastics were reportedly released per g of sample plastics that are obtained from the different rainwater collection facilities. Though the number of microplastics released were varied with the polymer type of plastics, all of the plastics did result in microplastic generation upon aging. Therefore, based on the results of Zhang et al. (2022), it is speculated that the compact rainwater harvesting filters available at residential houses would also leach out microplastics over time which ultimately would affect the health of the consumers. Therefore, the design of rainwater harvesting facilities using plastic materials should be reconsidered. Another interesting study was carried out by a group of researchers from South Australia to analyse the generation of microplastics from the commonly used nylon rope. The nylon rope used for hanging the swing and ladder in the natural outdoor environment for more than 10 years was taken as the sample. The difference in the dimensions between old and new rope was used to arrive at the conservative measure of the quantity of microplastics generated. It was estimated that approximately 6280 microplastics would have been released from the nylon rope in a span of 10 years. The number is expected to be even more as the assumed size of all the generated microplastics to arrive at the total quantity is 5 mm (Sobhani et al. 2022). The results of this study indirectly highlight the possible contamination of soil particles due to weathering. Apart from its usage in swings, households inevitably use nylon ropes for drying clothes and this could also be another potential contributor of microplastics.
Transport of microplastics originated from residential buildings
Microplastics in the indoor atmospheric environment
Studies on the presence of microplastics in indoor air are on the rise, and the details of various studies conducted to assess microplastic pollution in the indoors of residential houses are given in Table 1. Among the different geographical locations, China records significantly higher microplastic contamination than other countries. This was ascribed to the difference in anthropogenic activities, land use patterns, population density, and industrialization (Liao et al. 2021). Comparative studies based in China have also confirmed that the microplastic pollution observed in residential buildings are higher than in other indoor environments like offices, hotels, classrooms, laboratories, dormitories, etc. (Liao et al. 2021; Zhu et al. 2022). Another study based in Spain has also reported similar observations in the microplastic concentration reported between residential and working places (Torres-Agullo et al. 2022). These differences could be attributed to the variation in the type of activities performed in a building and the number of occupants (Bahrina et al. 2020; Liao et al. 2021). From previous studies, shedding of microfibres from indoor clothing and other indoor furnishings (curtain, sofa, chair), occurring naturally or due to human activities/interventions, is observed to be a major source of indoor microplastics based on previous studies (Table 1). Besides, line drying of clothes inside living spaces is another activity that supposedly releases microfibres into indoor air (Dris et al. 2017). Therefore, it could be said that the quantity and quality of textiles present inside the building largely affects the indoor microplastic concentration (Zhang et al. 2020b; Yao et al. 2022). This factor could be partially related to the lifestyle of people in the sampling region. Accordingly, it is found that urban indoors with more lavish interiors are more contaminated than rural indoors (Aslam et al. 2022). Furthermore, with respect to the flooring, carpeted houses have recorded double the concentration of synthetic microplastics like PE, PET, polyacrylic, and PA than non-carpeted houses. However, houses with wooden flooring have also been found to be contaminated, particularly with polyvinyl microplastics, and this was attributed to the wide application of polyvinyl in wooden finishings and linoleum floorings (Soltani et al. 2021). However, a study conducted by Uddin et al. (2022) in Kuwait is an exception as they had not observed any influence of carpets on the overall microplastic concentration. Therefore, it should be noted that though different sources release microplastics into the indoor spaces, factors like ventilation, airflow, resuspension of microplastics, and cleaning habits determine the number of microplastics eventually accumulating in the indoor environment (Liao et al. 2021; Yao et al. 2022). The influence of air flow on microplastics’ distribution has lead to an interesting discussion on whether carpets act more as a source or as retainer of microplastics by limiting the resuspension of particles which might not occur in non-carpeted floors (Dris et al. 2017). Then, in the case of air-conditioned rooms, owing to the non-occurrence of indoor-outdoor air exchange, there occurs an accumulation of microplastics, eventually resulting in increased microplastic concentration (Liao et al. 2021; Kashfi et al. 2022). Zhang et al. (2020b) claimed that air flow caused by air-conditioning systems would initiate the resuspension of microplastics, paving the way to its deposition onto uncovered food items, ultimately increasing the risk posed to human beings. Therefore, adequate ventilation would be necessary to reduce indoor microplastic pollution (Choi et al. 2022). Furthermore, external factors like population density and traffic load in the sampling region also play a role in the microplastic distribution. For instance, a case study in Iran has reported that a city with a higher population and traffic load has recorded higher microplastic pollution than the less populated city (Kashfi et al. 2022). From previous studies, it is noted that among the different shapes of indoor microplastics, microfibres are dominantly observed in many studies whereas very few studies have reported the predominance of non-fibrous microplastics (Vianello et al. 2019; Liao et al. 2021; Choi et al. 2022). Among the different polymer types, PET plastics are found to be the most common and dominant one in several previous studies. The main source of PET fibres is reported to be the shedding of fibres from textiles, fabrics in furniture, carpets, and curtains, whereas the presence of PET fragments is attributed to the degradation of household wares, plastic containers of cleaning products, and so on. For instance, UK-based homes are observed to contain 1414 microplastics/m2/day and around 90% of which are fibrous, with PET topping the list (Jenner et al. 2021). Jenner et al. (2021) reported that the influence of climate on the choice of people’s clothing will also affect the characteristics of indoor microplastics. Besides, the colour of indoor microplastics also reportedly varies with the colour of clothes used in different activities like drying, washing, or sewing (Aslam et al. 2022). Furthermore, the concentration of microplastics was noted to decrease with an increase in size (Jenner et al. 2021; Torres-Agullo et al. 2022; Uddin et al. 2022).
A UK-based study has concluded that human exposure to microplastics is 1–45 times higher in indoors than outdoors (Jenner et al. 2021). On the other hand, Zhang et al. (2019) have studied the exposure of pet dogs and cats to microplastics by analysing their food intake and faeces. It was found that their exposure to PET microplastics through diet constitutes only 3 and 4.9% of the overall microplastics identified in cats and dogs, respectively. In the case of PC microplastics, it was 14 and 31% in cats and dogs, respectively. This result has suggested that sources other than dietary intake constitute a significant exposure pathway and that source was claimed to be the indoor house dust being inhaled by the pets (Zhang et al. 2019). Apart from pet animals, humans are another living beings who spend a considerable amount of their time indoors than outdoors. Due to the Covid-19 pandemic and work-from-home culture, the time spent at home has increased lately. To simulate human being’s breathing behaviour, a study was conducted by Vianello et al. (2019) through the use of manikin in the indoors of three apartments. All the samples obtained in this study were found to be contaminated with microplastics, with polyester being identified predominantly (81%), followed by PE (5%), nylon (3%), and other non-specified polymers (11%). Microplastics have been identified even in the human placenta. Analysis of the pigments present in those microplastics revealed that some of those pigments are present in cosmetics, PCPs, paints, and finger paints (Ragusa et al. 2021). All these products are predominantly used in households, and it further confirms that domestic products and activities are considerably associated with atmospheric microplastic pollution.
Microplastics in wastewater treatment plants and its transport into aquatic and terrestrial environments
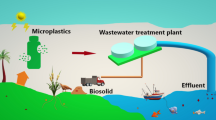
The wastewater generated from households enters either into the wastewater treatment plant before its discharge or directly into aquatic systems. The removal efficiency of microplastics from wastewater by existing wastewater treatment plants (WWTP) can go up to 98% (Lee and Kim 2018). Preliminary and primary units together contribute up to 72% removal efficiency of microplastics in WWTP (Iyare et al. 2020). In secondary treatment processes, membrane bioreactors show better microplastic removal than conventional activated sludge processes (Lares et al. 2018). Better removal efficiency in the former case may be ascribed to the synergistic effect of membrane filtration and biological process. In the case of filtration, the removal efficiency depends on the pore size of filters. The removal also occurs due to its adhesion to the sand particles that is driven by the presence of extracellular polymeric substances. A simple coagulation unit can also contribute to the better removal of microplastics. For instance, the addition of an optimum dose of polyaluminium chloride to wastewater was reported to have resulted in appreciable removal (81%) of microplastic along with other pollutants and the removal was noted to be driven by floc formation and precipitation mechanisms (Hidayaturrahman and Lee 2019). However, a very high coagulant dosage does not result in effective removal of microplastics (47–51%), which was ascribed to decreased zeta potential and electric charge inversion.
Despite observing appreciable but not 100% removal efficiency of microplastics in WWTP, the enormous release of microplastic into the aquatic environment is inevitable due to the high discharge volume of treated effluent (Ziajahromi et al. 2017b). Aquatic environments, including marine, surface, and groundwater bodies, are reported to contain microplastics. One of the major sources of microplastics in aquatic systems is reported to be the discharge of sewage and wastewater treatment plant effluent (Liu et al. 2021). The pollution does not end there but gets ingested by both aquatic and terrestrial animals such as penguins, turtles, mussels, sea birds, fishes, and several other organisms (Prata et al. 2020a). In a recent observation, the death of a penguin was solely attributed to the ingestion of a face mask, detected in its stomach as per necropsy results (Gallo Neto et al. 2021). Besides, sea birds ingest microplastics and act as a carrier of microplastics to different places via guano deposition. It was reported that migratory sea birds play a significant role in the fate and transport of microplastics in the Arctic ecosystem (Hamilton et al. 2021).
On the other hand, with respect to the fate of removed microplastics, the microplastics present in the wastewater are transferred into sludge/biosolids in the removal process. More than 90% of microplastics entering WWTP are reported to end up in primary and secondary sludge (Gies et al. 2018). The dominant microplastics detected in WWTP’s influent and treated effluent are synthetic textile fibers from laundry wastewater (Talvitie et al. 2017; Ziajahromi et al. 2017b) and that has reflected in the composition of microplastics present in the sludge. For instance, Gies et al. (2018) reported that the primary sludge had 9.7 ± 3.7 fibres/g and 5.2 ± 2.9 other particle types/g. Edo et al. (2020) revealed that sludge tends to release 8 × 1011 plastic particles annually, and their usage as soil amendments would disseminate 1013 microplastics into the soil. The soil microplastics are reported to affect the soil characteristics, soil microbial community, photosynthetic rate, and plant growth (Ren et al. 2021). Tympa et al. (2021), in fact, verified the uptake of acrylonitrile–butadiene–styrene (0.3 to 100 µm)-based microplastics by the roots of radish plant (Raphanus sativus). The result hence confirms the possibility of microplastics entering into human beings through food consumption (Tympa et al. 2021). Apart from biosolids, applying treated muncipal wastewater into the agricultural fields of arid regions would be another biggest source of soil microplastics. A study based in Spain has evaluated the microplastic pollution present in the agricultural fields which receive treated wastewater for irrigation. Around 159 ± 338 microplastic particles/kg of soil was identified in the analysis in red, black, blue, and tranaparent colours (Pérez-Reverón et al. 2022). Therefore, equal or may be more attention needs to be paid to microplastic pollution in the soil as that of water and other environments. A comprehensive picture interlinking the different sources of microplastics originating from domestic households and their transport pathways is given in Fig. 2.
Environmental fate of microplastics generated from domestic households
As they stay in the environment, the microplastics are subjected to weathering due to biotic, UV radiation, thermal and other actions (Meng et al. 2021) (Fig. 3). In the case of photo aging, the unsaturated bonds or chromophores present in the polymers are noted to absorb the UV-radiation, thereby initiating the overall degradation process (Liu et al. 2019b). As discussed earlier, wastewater generated from households is known to contain a larger proportion of microplastics Therefore, Li et al. (2021) has explored the physicochemical changes occurring in microplastics as they pass through different segments of a wastewater treatment plant, namely pipeline, grit chamber, and aeration tanks. Simulative lab scale studies have reported that after passing through different stages, the microplastics were observed to have developed various physicochemical modifications, which confirm the occurrence of chain scission and oxidation of microplastics (Li et al. 2021). However, the after-effects of aging need not be the same in all cases. For instance, the intensity of microplastic aging was observed to vary with the characteristics of the aging medium and type of radiation exposure. In the case of seawater, the chlorine ions present in it are found to inhibit aging by preventing the formation of superoxide radicals that aids in photodegradation. It was established that the aging of PP microplastics observed in seawater was lower than that of ultrapure water (Wu et al. 2021). In another interesting study, Ouyang et al. (2022) explored the degradation mechanism of PVC microplastics under three different UV radiation bands. UVC radiation reportedly has more influence on aging than UVA and UVB owing to its ionising property. With respect to the effect of photoaging, the process has been found to notably influence the mobility of microplastics. Photoaged PVC microplastics were observed to be more dispersed and less settleable than pristine microplastics. This behaviour was observed to be driven by the formation of reactive oxygen species (ROS) on the surface of microplastics (Zhu et al. 2020).
Owing to the larger specific surface area, environmental microplastics tend to adsorb other coexisting environmental contaminants (Fu et al. 2021). The microplastics collected from the beach sands of the Canary Islands in Spain was reportedly having ten different UV filters/stabilisers adsorbed on it with a concentration varying between 1 and 4031 ng/g (Santana-Viera et al. 2021). Aging can further aggravate the problem by resulting in increased specific surface area that will inturn alter its adsorption property. The UV filters that are commonly used in cosmetic products have been tested in a laboratory scale for their adsorption on virgin and photoaged HDPE microplastics in millipore and river water, respectively. The adsorption equilibrium was attained eight times faster in the case of aged microplastics than pristine microplastics in the respective matrices (Kapelewska et al. 2021). On the other side, studies carried out to find the effect of biofilm formation on microplastics have widely reported its influence on the adsorption of heavy metals upon microplastics. Polylactic acid (PLA) and low-density PE microplastics were tested for the influence of biofilm growth on heavy metal accumulation on its surface. Experimental and statistical studies have suggested that the concentration of heavy metals such as Fe, Ni, Ba, Ga, Cs, and Rb accumulated on the surface of microplastics showed a positive correlation with the amount of biofilm growth (Richard et al. 2019). A study conducted by Qiongjie et al. (2022) has demonstrated that biofilm formation has enhanced the adsorption capacity of microplastics for copper and lead even more than photoaging. The heavy metal adsorption had also impacted the microbial community distribution in the biofilm, especially in the case of lead. Apart from heavy metal accumulation on biofilm, adsorption of organic pollutants such as antibiotics, polychlorinated biphenyls, and polycyclic aromatic hydrocarbons has also been reported (Wang et al. 2021). Therefore, in the event of consumption of these microplastics by aquatic species, the adsorbed compounds will leach into the aquatic species, which in turn will translocate into species of higher trophic levels. Also, it is to be noted that these pollutants such as UV filters, heavy metals, and antibiotics are coexisting with microplastics in the domestic wastewater and, hence, there is always a higher chance for their interactions.
On the other hand, apart from absorbing/adsorbing other environmental pollutants, microplastics on their own are hazardous. This is because of the additives that are added to the plastics during their manufacturing to improve its property. The physicochemical changes caused by weathering actions are observed to result in the release of such additives like trace metals. For instance, Meng et al. (2021) reported the release of toxic metal additives from commercial PVC clapboard fragments upon the natural aging process in paddy soil. Another study by Feng et al. (2022) investigated the release of metals from three plastics namely PP, PVC, and PET plastics. It was found that just after 70 days of artificial UV radiation, the release of metal was observed. Notably, the release of lead from PP was observed to be 78.89% of the total concentration present originally. Therefore, it is clear that different prominent modes of aging, namely photo, physical, and biological aging, have their own kind of impacts. However, in most scenarios, different types of aging have been observed to intensify the environmental damage caused due to microplastics.
Toxicity of microplastics
Toxicity in aquatic organisms
Several factors have been found to influence the toxicity effects of microplastics in aquatic organisms. The factors include bioavailability, physical and chemical properties, size, colour, density, functional groups, type, surface charge, stability, crystallinity, the spatial and temporal distribution of microplastics in aquatic environment, and presence of other anthropogenic compounds (Paul-Pont et al. 2016; de Sá et al. 2018; Ma et al. 2020). Ingestion and accumulation of microplastics in aquatic organisms have adverse effects ranging from internal damage to increased mortality. Table 2 summarizes the toxicity effects of microplastics on different aquatic organisms. Accumulation of microplastics arising from households is widely reported in planktons, vertebrate and invertebrate animals including humans (Prata et al. 2020c). Microplastics of different polymer types that could originate from households and end up in different environmental compartments have been studied for their toxic effects on several aquatic organisms. For instance, Lei et al. (2018) investigated the toxic effects of five common microplastics found in the environment, viz, PA, PS, PVC, PP, and PE on zebrafish Danio rerio and nematode Caenorhabditis elegans and established the relationship between survival rate, the extent of internal damages, and size of microplastics. A reduction of survival rate up to 27.1% was observed in D. rerio after 10-day exposure, whereas intestinal damage was up to 86.7%. In the case of C. elegans, 2-day exposure to microplastics affected reproduction, body length, and survival rate. Exposure to PA, PVC, PP, and PE microplastics of size 1.0 μm affected the survival rate significantly when compared to the sizes 0.1 and 5.0 μm in dose-dependent manner (Lei et al. 2018).
Apart from the size, the age of the microplastics also seems to play an important role in the ingestion and toxicity of microplastics in aquatic organisms. Bråte et al. (2018) studied the impact of weathering and uptake of PE microbeads (< 50 μm) that are commonly present in the toothpaste in M. galloprovincialis (Mediterranean mussels). The PE beads used in the study were reportedly recovered from commercially available Colgate® Max Fresh® toothpaste. Weathered polyethylene (WPE) was evidently ingested more relative to virgin PE. Increased ingestion of WPE is influenced by a number of individual and combination of factors viz; density, hydrophilicity, and biofilm formation ability of the microplastics. Increased biofilm formation on microplastics results in more organic matter accumulation over its surface, thus increasing the nutrition value that promotes its ingestion by mussels (Bråte et al. 2018). Similar observations were made by Jovanović et al. (2018) who reported that virgin microplastics does not cause imminent harm in Sparus aurata when ingested.
Retention and accumulation of microplastics are observed to cause chronic toxicity in aquatic organisms. McGoran et al. (2020) studied the retention of microplastics in Thames estuary crabs, namely Eriocheir sinensis and Carcinus maenas. Microplastics in the form of fragments, fibres, a tangle of fibres, and films were reported in the gill chamber, gastrointestinal tract, and gastric mill. Fibre tangles of size more than 100 μm were found to be accumulated in the gastric gill, which results in retention due to the larger size of microplastics (McGoran et al. 2020). Retention and accumulation of microplastics reportedly vary not only with the size of microplastics but also with type of organisms and feeding habits. Van Cauwenberghe et al. (2015) observed variation in retention of microplastics within the tissues of Mytilus edulis (blue mussels) and Arenicola marina (lungworms). In the case of blue mussels, smaller size microplastics (10 μm) were retained more relative to that of larger-sized microplastics. However, this was not the case with lungworm, where particles of sizes both 10 μm and 30 μm are detected. This difference is attributed to the individual feeding habits of the aquatic organisms M. edulis and A. marina. The former is a filter feeder; hence, feeds only on algae and particles of selective size, whereas the latter is a deposit feeder and preys on diatoms and bacteria in the sediments (Van Cauwenberghe et al. 2015). Similar observations of specific intake of microplastics of size 6 μm were also reported in Pacific oysters (Sussarellu et al. 2016). The presence and ingestion of other pollutants such as fluoranthene along with microplastics were reported to increase hemolytic death, cellular oxidation, and toxicity in Mussels spp. due to the high accumulation of microplastics during the 7-day exposure study. Increased cellular oxidation evidently increased ROS in the hemocytes, which further worsened due to the presence of secondary pollutants (Paul-Pont et al. 2016). Interestingly, the presence of more than one type of microplastics increased the toxicity risk in aquatic organisms. Ziajahromi et al. (2017a) studied the acute (2-day exposure) and chronic (8-day exposure) effects of PS beads and polyester fibres on C. dubia, a freshwater zooplankton. Ingestion or accumulation of fibre or beads was not observed even though the size of the beads was in the range of their food size. However, exposure to both fibres and beads led to 40% mortality, reduction in body size, and reproductive ability, whereas exposure to fibres also led to deformities in the carapace and antenna and abnormal swimming behaviour in C. dubia, indicating that fibres pose comparatively more risk than microbeads (Ziajahromi et al. 2017a). This observation is alarming, given the abundant presence of microfibres in the wastewater generated from domestic households (Galvão et al. 2020).
Toxicity in humans
Humans are continuously exposed to microplastics in one of the routes of exposure, viz, ingestion, inhalation, and dermal contact. Microplastics have been widely reported in salt (Karami et al. 2017), sugar (Kwon et al. 2020), packed drinking water, food packed in plastic containers (Fadare et al. 2020), and seafood such as fish (Neves et al. 2015), crabs (Zhang et al. 2021), shrimps (Curren et al. 2020), and mussels (Li et al. 2016). Consumption of these contaminated food items exposes human beings to microplastic ingestion. In the American diet, it is estimated that about 39,000–52,000 microplastic particles are consumed annually. However, this number reportedly increased to 74,000 and 121,000 when inhalation of microplastics is accounted (Cox et al. 2019). Outdoor and indoor exposures to suspended microplastics, those arising from textile and abrasion/aging of plastics, are common inhalation routes (Prata et al. 2020c). The dermal route of microplastic exposure in humans is less significant because the skin barrier prevents microplastics from penetrating into skin layers and the circulatory system (Revel et al. 2018).
Schirinzi et al. (2017) studied the oxidative stress and cytotoxic effects of microplastics on cerebral and epithelial cells. Two cell lines of cerebral (T98G) and epithelial (HeLa) origins were exposed to PS- and PE-based microplastics of concentration 10 ng/mL to 10 µg/mL for 24- to 48-h period. It has resulted in oxidative stress causing generation of ROS. However, no significant reduction of viable cells was observed. Of the two microplastics, cells exposed to PS generated high ROS compared to PE, which was attributed to the smaller size of PS. ROS generation due to microplastics could be dangerous since high ROS generation could lead to the growth and proliferation of cancer cells (Schirinzi et al. 2017). Exposure of cells to microplastics also triggers immune response. Hwang et al. (2019) studied the effect of PP microplastics of two categories, namely size ~ 20 μm and 25–200 μm on a variety of human-derived cells viz; peripheral blood mononuclear cells (PBMC), human dermal fibroblasts (HDF), the human mast cell line-1 (HMC-1), basophilic leukaemia cells secreting histamine and cytokines (RBL-2H3), and Raw 264.7 cells. PP microplastics of size ~ 20 μm were reported to be cytotoxic to PBMCs and Raw 264.7 cells. Cytotoxicity was supposedly due to ROS generated in the cells exposed to microplastics. An increase in cytokines concentration, namely interleukin-6 (IL-6) and tumour necrosis factor-α (TNF-α), was also observed in Raw 264.7, PBMCs, and HMC-1 cells, which are responsible for inflammation. In HMC-1 cells, an increase in histamine secretion was observed, which are responsible for allergic reactions in human (Hwang et al. 2019). In a similar study with PS microplastics, particles of size 0.46 and 1 μm were reported to be toxic to red blood cells. An increase in IL- 6 and TNF-α in response to PS microplastics was also reported (Hwang et al. 2020). In another interesting study, Stock et al. (2021) have compared the toxicity effects on Caco-2, HepG2, and HepaRG cells with four different microplastics belonging to PE, PP, PET, and PVC categories. The investigation has found that PE particles could transport more through the intestinal barrier than PS particles of similar size and shape as that of PE. Besides, Choi et al. (2020a) have highlighted the significance of physical and chemical characteristics of PS microplastic fragments while exploring the toxicity effects on peripheral blood mononuclear cells, KATO III cells, HeLa cells, and human dermal fibroblasts. It was reported that microplastics with higher roughness and sharpness have resulted in intense cell lysis than smoother microplastics. It was also observed that the chemical reagents/plasticizers released from microplastics would enhance the toxicity effect. Likewise, Yu et al. (2022) have reported that photoaged PS microspheres showed enhanced toxicity to Caco-2 cells. Therefore, it is evident that microplastics of different physical and chemical properties would pose varying toxicity effects. However, from Fig. 4a–c, it could be seen that except very few studies which have utilized irregularly shaped microplastics synthesized from waste plastics and household products (Han et al. 2020; Bengalli et al. 2022; Weber et al. 2022); all the other remaining studies have utilized commercially purchased PS microspheres in their investigation. Besides, from the existing literature database, it could be seen that no research has been done so far to understand the toxicity effect of fibrous microplastics on human cell lines (Fig. 4a). This is one of the major research gaps identified through this review, and it very necessary to conduct toxicity research with fibrous microplastics as they are reportedly the prominent form of microplastics originating from domestic households where the human beings are living. With respect to the types of toxicity studies, though studies on neurotoxicity, bioaccumulation, reproductive toxicity, and translocation due to microplastics in human cells are limited, studies in mice models with PS microplastics and nanoplastic particles have been reported to affect neurotransmission, causing neurotoxicity and enzyme imbalance due to bioaccumulation and translocation of microplastics after uptake (Murali et al. 2015; Deng et al. 2017). In a study conducted by Hu et al. (2021), PS microspheres were evaluated for their toxicity on immune response and reproductivity in pregnant mice (C57BL/6-mated BALB/c mice). It was observed that exposure to PS microplastics leads to foetal losses due to excessive immune response triggered by microplastics. Hence, it is speculated that microplastics could pose a similar risk in humans when ingested. Therefore, it is very essential to work with real time environmental microplastics in order to explore the significance of toxicity effects to human health.
Details of existing literature on the toxicity effects of micro/nanoplastics exposure to human cells — a various shapes of microplastics that are so far investigated; b polymer types of microplastics that are so far utilized to conduct the toxicity studies: PE, polyethylene; PP,polypropylene; PMMA, poly (methyl methacrylate); ABS, acrylonitrile butadiene styrene; PVC, polyvinyl chloride; PET, polyethylene terephthalate; PS, polystyrene; c different sources of microplastics that have been used in this area of toxicity research so far
Conclusions
In this article, the various sources of microplastics originating from residential household activities and products are explored, followed by their transport, transformation, and toxicity. Even basic household products like tea bags, scouring pads, plastic food packaging containers, and glitters are found to be the sources of microplastic pollution in addition to the commonly reported sources such as PCPs, synthetic textiles, and face masks. Other household plastic products like rainwater harvesting system and nylon rope, which is present outdoor and has long-term exposure to sunlight, are also resulting in microplastics formation as they undergo weathering. With respect to the fate of these microplastics that are entering wastewater streams, though it is claimed that WWTP is capable of removing microplastics from the municipal wastewater, complete removal has not been achieved. Therefore, the effluent contaminates the surface water bodies upon its discharge, and the microplastics further enter the body of aquatic species, ultimately resulting in bioaccumulation and biomagnification. In addition, aging factors are reported to intensify the toxicity of microplastics by altering their adsorption affinity towards other coexisting environmental contaminants like heavy metals, pharmaceuticals, and other organic contaminants. On the other hand, with respect to the microplastics removed from the wastewater treatment plant, there has occurred only phase transfer of pollutants. The microplastics that are claimed to have been removed ends up in primary and secondary sludge. Currently, the treated sludge called biosolids is widely applied in agricultural fields in many countries, including developed nations like the United States. Before wastewater treatment, the pollution was restricted only to the water environment, but after treatment, the pollution has extended to one more environmental compartment, i.e. soil. This has made researchers to largely focus on the interaction of microplastics with the soil environment in recent times. The microplastic pollution in the soil is reported to be significantly more than that in the aquatic environment. Therefore, a proper strategical plan is required in the management of sludge to prevent future haphazard-like insufficiency of food grains as the soil microplastics are reportedly affecting the plant growth and soil microorganisms. Besides contaminating the aquatic and terrestrial compartments, the microplastics shedding from the synthetic fabrics of indoor clothing and furnishings are observed to result in intense indoor air contamination. The contamination level of indoor is apparently many folds higher than outdoor environment. Therefore, pet animals and especially humans due to the Covid-19 caused work from home culture are excessively subjected to inhaling as well as ingesting these indoor microplastics in the event of airborne microplastic deposition on uncovered food items. Hence, a detailed study needs to be undertaken to explore the impact of increased indoor time on human health and well-being. With respect to toxicity effects of microplastics, several studies have reported individual effects of microplastics on organisms, whereas more studies should focus on the combined effects of different microplastics and other anthropogenic pollutants on aquatic organisms at environmentally relevant microplastic concentrations. Toxicity studies carried out in human models are limited and have also utilized commercially purchased microplastics which are not representative of the real time environmental microplastics. Therefore, more detailed research with real time environmental microplastics and microfibres is essential to ascertain the myths and facts related to the risk associated with exposure to microplastics in humans. Conclusively, source reduction should be of vital consideration in combating microplastic pollution. It includes replacement of synthetic microbeads in PCPs with natural exfoliants, reduction in the procurement of synthetic textile products, shifting to consumption of biodegradable products like biodegradable glitters (Droguet et al. 2022), reduction in the usage of plastic-based scouring pads and tea bags, and installation of high efficient filters in laundry machines. With Covid-19 preventive measures in place, the usage of face masks, gloves, and other personal protective equipment has leaped. SAGES (2020) has suggested that mask rotation can be adopted as a reuse strategy after allowing them to dry for long enough periods. Reuse of masks after appropriate processes like application of heat and UV treatment can be taken as one of the alternatives for reducing the consumption of masks. The usage of biodegradable masks would be a more sustainable solution.
Data availability
Not applicable.
References
5Gyres (2022) Plastic microbeads. In: 5Gyres. https://www.5gyres.org/microbeads. Accessed 23 Mar 2022
Anagnosti L, Varvaresou A, Pavlou P et al (2021) Worldwide actions against plastic pollution from microbeads and microplastics in cosmetics focusing on European policies. Has the issue been handled effectively? Mar Pollut Bull 162:111883. https://doi.org/10.1016/j.marpolbul.2020.111883
Aslam I, Qadir A, Ahmad SR (2022) A preliminary assessment of microplastics in indoor dust of a developing country in South Asia. Environ Monit Assess 194:340. https://doi.org/10.1007/s10661-022-09928-3
Bahrina I, Syafei AD, Satoto R et al (2020) An occupant-based overview of microplastics in indoor environments in the City of Surabaya, Indonesia. J Ecol Eng 21:236–242. https://doi.org/10.12911/22998993/126876
Bashir SM, Kimiko S, Mak CW et al (2021) Personal care and cosmetic products as a potential source of environmental contamination by microplastics in a densely populated Asian city. Front Mar Sci 8:1–11. https://doi.org/10.3389/fmars.2021.683482
Belzagui F, Buscio V, Gutiérrez-Bouzán C, Vilaseca M (2021) Cigarette butts as a microfiber source with a microplastic level of concern. Sci Total Environ 762:144165. https://doi.org/10.1016/j.scitotenv.2020.144165
Bengalli R, Zerboni A, Bonfanti P et al (2022) Characterization of microparticles derived from waste plastics and their bio-interaction with human lung A549 cells. J Appl Toxicol 42:2030–2044. https://doi.org/10.1002/jat.4372
Boucher J, Friot D (2017) Primary microplastics in the oceans: a global evaluation of sources. IUCN, Gland Switz. https://doi.org/10.2305/iucn.ch.2017.01.en
Bråte ILN, Blázquez M, Brooks SJ, Thomas KV (2018) Weathering impacts the uptake of polyethylene microparticles from toothpaste in Mediterranean mussels (M. galloprovincialis). Sci Total Environ 626:1310–1318. https://doi.org/10.1016/j.scitotenv.2018.01.141
Browne MA, Crump P, Niven SJ et al (2011) Accumulation of microplastic on shorelines woldwide: sources and sinks. Environ Sci Technol 45:9175–9179. https://doi.org/10.1021/es201811s
Can B (2007) Manufacturing of environment-friendly floor carpets. Pak Text J 56:73
Chak AK (2020) Microplastics, microbeads: what you did not know about self-care products. DownToEarth
Chang M (2015) Reducing microplastics from facial exfoliating cleansers in wastewater through treatment versus consumer product decisions. Mar Pollut Bull 101:330–333. https://doi.org/10.1016/j.marpolbul.2015.10.074
Cheung PK, Fok L (2016) Evidence of microbeads from personal care product contaminating the sea. Mar Pollut Bull 109:582–585. https://doi.org/10.1016/j.marpolbul.2016.05.046
Cheung PK, Fok L (2017) Characterisation of plastic microbeads in facial scrubs and their estimated emissions in Mainland China. Water Res 122:53–61. https://doi.org/10.1016/j.watres.2017.05.053
Choi D, Bang J, Kim T et al (2020a) In vitro chemical and physical toxicities of polystyrene microfragments in human-derived cells. J Hazard Mater 400:123308. https://doi.org/10.1016/j.jhazmat.2020.123308
Choi JS, Hong SH, Park JW (2020b) Evaluation of microplastic toxicity in accordance with different sizes and exposure times in the marine copepod Tigriopus japonicus. Mar Environ Res 153:104838. https://doi.org/10.1016/j.marenvres.2019.104838
Choi H, Lee I, Kim H et al (2022) Comparison of microplastic characteristics in the indoor and outdoor air of urban areas of South Korea. Water, Air, Soil Pollut 233:1–10. https://doi.org/10.1007/s11270-022-05650-5
Cotton L, Hayward AS, Lant NJ, Blackburn RS (2020) Improved garment longevity and reduced microfibre release are important sustainability benefits of laundering in colder and quicker washing machine cycles. Dye Pigment 177:108120. https://doi.org/10.1016/j.dyepig.2019.108120
Cox KD, Covernton GA, Davies HL et al (2019) Human consumption of microplastics. Environ Sci Technol 53:7068–7074. https://doi.org/10.1021/acs.est.9b01517
Curren E, Leaw CP, Lim PT, Leong SCY (2020) Evidence of marine microplastics in commercially harvested seafood. Front Bioeng Biotechnol 8:1–9. https://doi.org/10.3389/fbioe.2020.562760
De Falco F, Gullo MP, Gentile G et al (2018) Evaluation of microplastic release caused by textile washing processes of synthetic fabrics. Environ Pollut 236:916–925. https://doi.org/10.1016/j.envpol.2017.10.057
de Sá LC, Oliveira M, Ribeiro F et al (2018) Studies of the effects of microplastics on aquatic organisms: what do we know and where should we focus our efforts in the future? Sci Total Environ 645:1029–1039. https://doi.org/10.1016/j.scitotenv.2018.07.207
Delhiraja K, Philip L (2020) Characterization of segregated greywater from Indian households: part A—physico-chemical and microbial parameters. Environ Monit Assess 192:428. https://doi.org/10.1007/s10661-020-08369-0
Deng Y, Zhang Y, Lemos B, Ren H (2017) Tissue accumulation of microplastics in mice and biomarker responses suggest widespread health risks of exposure. Sci Rep 7:1–10. https://doi.org/10.1038/srep46687
Dris R, Gasperi J, Mirande C et al (2017) A first overview of textile fibers, including microplastics, in indoor and outdoor environments. Environ Pollut 221:453–458. https://doi.org/10.1016/j.envpol.2016.12.013
Droguet BE, Liang HL, Frka-Petesic B et al (2022) Large-scale fabrication of structurally coloured cellulose nanocrystal films and effect pigments. Nat Mat 21:352–358. https://doi.org/10.1038/s41563-021-01135-8
Du F, Cai H, Zhang Q et al (2020) Microplastics in take-out food containers. J Hazard Mat 399:122969. https://doi.org/10.1016/j.jhazmat.2020.122969
Du S, Valla JA, Parnas RS, Bollas GM (2016) Conversion of polyethylene terephthalate based waste carpet to benzene-rich oils through thermal, catalytic, and catalytic steam pyrolysis. ACS Sustain Chem Eng 4:2852–2860. https://doi.org/10.1021/acssuschemeng.6b00450
Edo C, González-Pleiter M, Leganés F et al (2020) Fate of microplastics in wastewater treatment plants and their environmental dispersion with effluent and sludge. Environ Pollut 259:113837. https://doi.org/10.1016/j.envpol.2019.113837
Elizalde-Velázquez A, Carcano AM, Crago J, et al (2020) Translocation, trophic transfer, accumulation and depuration of polystyrene microplastics in Daphnia magna and Pimephales promelas. Environ Pollut 259:. https://doi.org/10.1016/j.envpol.2020.113937
European Environmental Bureau (EEB) (2021) EU microplastic ban just got a step closer, but has major loopholes. Recycl. Mag
Fadare OO, Okoffo ED (2020) Covid-19 face masks: a potential source of microplastic fibers in the environment. Sci Total Environ 737:140279. https://doi.org/10.1016/j.scitotenv.2020.140279
Fadare OO, Wan B, Guo LH, Zhao L (2020) Microplastics from consumer plastic food containers: are we consuming it? Chemosphere 253:126787. https://doi.org/10.1016/j.chemosphere.2020.126787
Fang M, Liao Z, Ji X et al (2022) Microplastic ingestion from atmospheric deposition during dining/drinking activities. J Hazard Mat 432:128674. https://doi.org/10.1016/j.jhazmat.2022.128674
Feng W, Huang C, Tan X, et al (2022) Release of the additive metals from 3 commonly used plastics during the degradation under the treatment of UV irradiation. Ecotoxicol 75–84. https://doi.org/10.1007/s10646-021-02467-6
Frias JPGL, Nash R (2019) Microplastics: finding a consensus on the definition. Mar Pollut Bull 138:145–147. https://doi.org/10.1016/j.marpolbul.2018.11.022
Fu L, Li J, Wang G et al (2021) Adsorption behavior of organic pollutants on microplastics. Ecotoxicol Environ Saf 217:112207. https://doi.org/10.1016/j.ecoenv.2021.112207
Gallo Neto H, Gomes Bantel C, Browning J et al (2021) Mortality of a juvenile Magellanic penguin (Spheniscus magellanicus, Spheniscidae) associated with the ingestion of a PFF-2 protective mask during the Covid-19 pandemic. Mar Pollut Bull 166:112232. https://doi.org/10.1016/j.marpolbul.2021.112232
Galvão A, Aleixo M, De Pablo H et al (2020) Microplastics in wastewater: microfiber emissions from common household laundry. Environ Sci Pollut Res 27:26643–26649. https://doi.org/10.1007/s11356-020-08765-6
Gambardella C, Morgana S, Ferrando S et al (2017) Effects of polystyrene microbeads in marine planktonic crustaceans. Ecotoxicol Environ Saf 145:250–257. https://doi.org/10.1016/j.ecoenv.2017.07.036
Gereffi G (2020) What does the COVID-19 pandemic teach us about global value chains? The case of medical supplies. J Int Bus Policy 3:287–301. https://doi.org/10.1057/s42214-020-00062-w
Gies EA, LeNoble JL, Noël M et al (2018) Retention of microplastics in a major secondary wastewater treatment plant in Vancouver, Canada. Mar Pollut Bull 133:553–561. https://doi.org/10.1016/j.marpolbul.2018.06.006
Godoy V, Martín-Lara MA, Calero M, Blázquez G (2019) Physical-chemical characterization of microplastics present in some exfoliating products from Spain. Mar Pollut Bull 139:91–99. https://doi.org/10.1016/j.marpolbul.2018.12.026
Hamilton BM, Bourdages MPT, Geoffroy C et al (2021) Microplastics around an Arctic seabird colony: particle community composition varies across environmental matrices. Sci Total Environ 773:145536. https://doi.org/10.1016/j.scitotenv.2021.145536
Han S, Bang J, Choi D et al (2020) Surface pattern analysis of microplastics and their impact on human-derived cells. ACS Appl Polym Mater 2:4541–4550. https://doi.org/10.1021/acsapm.0c00645
Hari PK (2012) Types and properties of fibres and yarns used in weaving Pramod. Woodhead Publishing Limited
Hartline NL, Bruce NJ, Karba SN et al (2016) Microfiber masses recovered from conventional machine washing of new or aged garments. Environ Sci Technol 50:11532–11538. https://doi.org/10.1021/acs.est.6b03045
Henry B, Laitala K, Klepp IG (2019) Microfibres from apparel and home textiles: prospects for including microplastics in environmental sustainability assessment. Sci Total Environ 652:483–494. https://doi.org/10.1016/j.scitotenv.2018.10.166
Hernandez LM, Xu EG, Larsson HCE et al (2019) Plastic teabags release billions of microparticles and nanoparticles into tea. Environ Sci Technol 53:12300–12310. https://doi.org/10.1021/acs.est.9b02540
Hidayaturrahman H, Lee TG (2019) A study on characteristics of microplastic in wastewater of South Korea: identification, quantification, and fate of microplastics during treatment process. Mar Pollut Bull 146:696–702. https://doi.org/10.1016/j.marpolbul.2019.06.071
Hu J, Qin X, Zhang J et al (2021) Polystyrene microplastics disturb maternal-fetal immune balance and cause reproductive toxicity in pregnant mice. Reprod Toxicol 106:42–50. https://doi.org/10.1016/j.reprotox.2021.10.002
Hwang J, Choi D, Han S et al (2019) An assessment of the toxicity of polypropylene microplastics in human derived cells. Sci Total Environ 684:657–669. https://doi.org/10.1016/j.scitotenv.2019.05.071
Hwang J, Choi D, Han S et al (2020) Potential toxicity of polystyrene microplastic particles. Sci Rep 10:1–12. https://doi.org/10.1038/s41598-020-64464-9
Institute of Making (UC London) (2013) Materials - dish sponge. https://www.instituteofmaking.org.uk. Accessed 20 Mar 2022
Iyare PU, Ouki SK, Bond T (2020) Microplastics removal in wastewater treatment plants: a critical review. Environ Sci Water ResTechnol 6:2664–2675. https://doi.org/10.1039/d0ew00397b
Jambeck J, Geyer R, Wilcox C et al (2015) Plastic waste inputs from land into the ocean. Science 347:768–771
Jenner LC, Sadofsky LR, Danopoulos E, Rotchell JM (2021) Household indoor microplastics within the Humber region (United Kingdom): quantification and chemical characterisation of particles present. Atmos Environ 259:118512. https://doi.org/10.1016/j.atmosenv.2021.118512
Jeong CB, Won EJ, Kang HM et al (2016) Microplastic size-dependent toxicity, oxidative stress induction, and p-JNK and p-p38 activation in the Monogonont rotifer (Brachionus koreanus). Environ Sci Technol 50:8849–8857. https://doi.org/10.1021/acs.est.6b01441
Jessieleena AA, Nambi IM (2023) Distribution of microplastics in the catchment region of Pallikaranai marshland, a Ramsar site in Chennai India. Environ Pollut 318:120890. https://doi.org/10.1016/j.envpol.2022.120890
Jin Y, Xia J, Pan Z et al (2018) Polystyrene microplastics induce microbiota dysbiosis and inflammation in the gut of adult zebrafish. Environ Pollut 235:322–329. https://doi.org/10.1016/j.envpol.2017.12.088
Jovanović B, Gökdağ K, Güven O et al (2018) Virgin microplastics are not causing imminent harm to fish after dietary exposure. Mar Pollut Bull 130:123–131. https://doi.org/10.1016/j.marpolbul.2018.03.016
Kapelewska J, Klekotka U, Ewa Ż, Karpi J (2021) Simultaneous sorption behaviors of UV filters on the virgin and aged micro-high-density polyethylene under environmental conditions. Sci Total Environ 789:147979. https://doi.org/10.1016/j.scitotenv.2021.147979
Karami A, Golieskardi A, Keong Choo C et al (2017) The presence of microplastics in commercial salts from different countries. Sci Rep 7:1–11. https://doi.org/10.1038/srep46173
Kashfi FS, Ramavandi B, Arfaeinia H et al (2022) Occurrence and exposure assessment of microplastics in indoor dusts of buildings with different applications in Bushehr and Shiraz cities. Iran. Sci Total Environ 829:154651. https://doi.org/10.1016/j.scitotenv.2022.154651
Kwon JH, Kim JW, Pham TD et al (2020) Microplastics in food: a review on analytical methods and challenges. Int J Environ Res Public Heal 17:1–23. https://doi.org/10.3390/ijerph17186710
Lares M, Ncibi MC, Sillanpää M, Sillanpää M (2018) Occurrence, identification and removal of microplastic particles and fibers in conventional activated sludge process and advanced MBR technology. Water Res 133:236–246. https://doi.org/10.1016/j.watres.2018.01.049
Lassen C, Hansen SF, Magnusson K, et al (2015) Microplastics : occurrence, effects and sources of releases to the environment in Denmark. In: Danish Environ. Prot. Agency. https://www.orbit.dtu.dk. Accessed 20 Mar 2022
Lee H, Kim Y (2018) Treatment characteristics of microplastics at biological sewage treatment facilities in Korea. Mar Pollut Bull 137:1–8. https://doi.org/10.1016/j.marpolbul.2018.09.050
Lei K, Qiao F, Liu Q et al (2017) Microplastics releasing from personal care and cosmetic products in China. Mar Pollut Bull 123:122–126. https://doi.org/10.1016/j.marpolbul.2017.09.016
Lei L, Wu S, Lu S et al (2018) Microplastic particles cause intestinal damage and other adverse effects in zebrafish Danio rerio and nematode Caenorhabditis elegans. Sci Total Environ 619–620:1–8. https://doi.org/10.1016/j.scitotenv.2017.11.103
Li J, Qu X, Su L et al (2016) Microplastics in mussels along the coastal waters of China. Environ Pollut 214:177–184. https://doi.org/10.1016/j.envpol.2016.04.012
Li X, Li M, Mei Q et al (2021) Aging microplastics in wastewater pipeline networks and treatment processes: physicochemical characteristics and Cd adsorption. Sci Total Environ 797:148940. https://doi.org/10.1016/j.scitotenv.2021.148940
Liao Z, Ji X, Ma Y et al (2021) Airborne microplastics in indoor and outdoor environments of a coastal city in Eastern China. J Hazard Mat 417:126007. https://doi.org/10.1016/j.jhazmat.2021.126007
Liu C, Li J, Zhang Y et al (2019a) Widespread distribution of PET and PC microplastics in dust in urban China and their estimated human exposure. Environ Int 128:116–124. https://doi.org/10.1016/j.envint.2019.04.024
Liu P, Qian L, Wang H et al (2019b) New insights into the aging behavior of microplastics accelerated by advanced oxidation processes. Environ Sci Technol 53:3579–3588. https://doi.org/10.1021/acs.est.9b00493
Liu W, Zhang J, Liu H et al (2021) A review of the removal of microplastics in global wastewater treatment plants: characteristics and mechanisms. Environ Int 146:106277. https://doi.org/10.1016/j.envint.2020.106277
Lu Y, Zhang Y, Deng Y et al (2016) Uptake and accumulation of polystyrene microplastics in zebrafish (Danio rerio) and toxic effects in liver. Environ Sci Technol 50:4054–4060. https://doi.org/10.1021/acs.est.6b00183
Ma H, Pu S, Liu S et al (2020) Microplastics in aquatic environments: toxicity to trigger ecological consequences. Environ Pollut 261:114089. https://doi.org/10.1016/j.envpol.2020.114089
Madhumitha CT, Karmegam N, Biruntha M et al (2022) Extraction, identification, and environmental risk assessment of microplastics in commercial toothpaste. Chemosphere 296:133976. https://doi.org/10.1016/j.chemosphere.2022.133976
McGoran AR, Clark PF, Smith BD, Morritt D (2020) High prevalence of plastic ingestion by Eriocheir sinensis and Carcinus maenas (Crustacea: Decapoda: Brachyura) in the Thames Estuary. Environ Pollut 265:114972. https://doi.org/10.1016/j.envpol.2020.114972
Mehmood T, Peng L (2022) Polyethylene scaffold net and synthetic grass fragmentation: a source of microplastics in the atmosphere? J Hazard Mat 429:128391. https://doi.org/10.1016/j.jhazmat.2022.128391
Meng J, Xu B, Liu F et al (2021) Effects of chemical and natural ageing on the release of potentially toxic metal additives in commercial PVC microplastics. Chemosphere 283:131274. https://doi.org/10.1016/j.chemosphere.2021.131274
Murali K, Kenesei K, Li Y et al (2015) Uptake and bio-reactivity of polystyrene nanoparticles is affected by surface modifications, ageing and LPS adsorption: in vitro studies on neural tissue cells. Nanoscale 7:4199–4210. https://doi.org/10.1039/c4nr06849a
Napper IE, Barrett AC, Thompson RC (2020) The efficiency of devices intended to reduce microfibre release during clothes washing. Sci Total Environ 738:140412. https://doi.org/10.1016/j.scitotenv.2020.140412
Napper IE, Thompson RC (2016) Release of synthetic microplastic plastic fibres from domestic washing machines: effects of fabric type and washing conditions. Mar Pollut Bull 112:39–45. https://doi.org/10.1016/j.marpolbul.2016.09.025
Neves D, Sobral P, Ferreira JL, Pereira T (2015) Ingestion of microplastics by commercial fish off the Portuguese coast. Mar Pollut Bull 101:119–126. https://doi.org/10.1016/j.marpolbul.2015.11.008
O’Brien S, Okoffo ED, O’Brien JW et al (2020) Airborne emissions of microplastic fibres from domestic laundry dryers. Sci Total Environ 747:141175. https://doi.org/10.1016/j.scitotenv.2020.141175
Ó Briain O, Marques Mendes AR, McCarron S et al (2020) The role of wet wipes and sanitary towels as a source of white microplastic fibres in the marine environment. Water Res 182:116021. https://doi.org/10.1016/j.watres.2020.116021
Ouyang Z, Zhang Z, Jing Y et al (2022) The photo-aging of polyvinyl chloride microplastics under different UV irradiations. Gondwana Res 108:72–80. https://doi.org/10.1016/j.gr.2021.07.010
Paul-Pont I, Lacroix C, González Fernández C et al (2016) Exposure of marine mussels Mytilus spp. to polystyrene microplastics: toxicity and influence on fluoranthene bioaccumulation. Environ Pollut 216:724–737. https://doi.org/10.1016/j.envpol.2016.06.039
Pedà C, Caccamo L, Fossi MC et al (2016) Intestinal alterations in European sea bass Dicentrarchus labrax (Linnaeus, 1758) exposed to microplastics: preliminary results. Environ Pollut 212:251–256. https://doi.org/10.1016/j.envpol.2016.01.083
Pérez-Reverón R, González-Sálamo J, Hernández-Sánchez C et al (2022) Recycled wastewater as a potential source of microplastics in irrigated soils from an arid-insular territory (Fuerteventura, Spain). Sci Total Environ 817:152830. https://doi.org/10.1016/j.scitotenv.2021.152830
Piccardo M, Provenza F, Grazioli E et al (2020) PET microplastics toxicity on marine key species is influenced by pH, particle size and food variations. Sci Total Environ 715:136947. https://doi.org/10.1016/j.scitotenv.2020.136947
Picó Y, Barceló D (2019) Analysis and prevention of microplastics pollution in water: current perspectives and future directions. ACS Omega 4:6709–6719. https://doi.org/10.1021/acsomega.9b00222
Plastics E (2021) Plastics - the Facts 2021. An analysis of European plastics production, demand and waste data
Prata JC, da Costa JP, Lopes I et al (2020a) Environmental status of (micro)plastics contamination in Portugal. Ecotoxicol Environ Saf 200:110753. https://doi.org/10.1016/j.ecoenv.2020.110753
Prata JC, da Costa JP, Lopes I et al (2020b) Environmental exposure to microplastics: an overview on possible human health effects. Sci Total Environ 702:134455. https://doi.org/10.1016/j.scitotenv.2019.134455
Prata JC, Castro JL, da Costa JP et al (2020c) The importance of contamination control in airborne fibers and microplastic sampling: experiences from indoor and outdoor air sampling in Aveiro. Portugal. Marine Pollut Bull 159:111522. https://doi.org/10.1016/j.marpolbul.2020.111522
Praveena SM, Shaifuddin SNM, Akizuki S (2018) Exploration of microplastics from personal care and cosmetic products and its estimated emissions to marine environment: an evidence from Malaysia. Mar Pollut Bull 136:135–140. https://doi.org/10.1016/j.marpolbul.2018.09.012
Qiao R, Sheng C, Lu Y et al (2019) Microplastics induce intestinal inflammation, oxidative stress, and disorders of metabolome and microbiome in zebrafish. Sci Total Environ 662:246–253. https://doi.org/10.1016/j.scitotenv.2019.01.245
Qiongjie W, Yong Z, Yangyang Z et al (2022) Effects of biofilm on metal adsorption behavior and microbial community of microplastics. J Hazard Mat 424:127340. https://doi.org/10.1016/j.jhazmat.2021.127340
Ragusa A, Svelato A, Santacroce C et al (2021) Plasticenta: first evidence of microplastics in human placenta. Environ Int 146:106274. https://doi.org/10.1016/j.envint.2020.106274
Rainwayfilters (2022) Rainway filters. In: https://www.rainyfilters.com/products/rainy-filters. https://rainwayfilters.com/. Accessed 24 Mar 2022
Rainyfilters (2022) Rainy filters. In: Rainyfilters. https://www.rainyfilters.com. Accessed 24 Mar 2022
Ren X, Tang J, Wang L, Liu Q (2021) Microplastics in soil-plant system: effects of nano/microplastics on plant photosynthesis, rhizosphere microbes and soil properties in soil with different residues. Plant Soil 462:561–576. https://doi.org/10.1007/s11104-021-04869-1
Revel M, Châtel A, Mouneyrac C (2018) Micro(nano)plastics: a threat to human health? Curr Opin Environ Sci Heal 1:17–23. https://doi.org/10.1016/j.coesh.2017.10.003
Richard H, Carpenter EJ, Komada T et al (2019) Biofilm facilitates metal accumulation onto microplastics in estuarine waters. Sci Total Environ 683:600–608. https://doi.org/10.1016/j.scitotenv.2019.04.331
SAGES (2020) N95 mask re-use strategies. In: Soc. Am. Gastrointest. Endosc. Surg. https://www.sages.org. Accessed 15 Mar 2022
Santana-Viera S, Montesdeoca-Esponda S, Sosa-Ferrera Z, Santana-Rodríguez JJ (2021) UV filters and UV stabilisers adsorbed in microplastic debris from beach sand. Mar Pollut Bull 168:112434. https://doi.org/10.1016/j.marpolbul.2021.112434
Schirinzi GF, Pérez-Pomeda I, Sanchís J et al (2017) Cytotoxic effects of commonly used nanomaterials and microplastics on cerebral and epithelial human cells. Environ Res 159:579–587. https://doi.org/10.1016/j.envres.2017.08.043
Setälä O, Fleming-Lehtinen V, Lehtiniemi M (2014) Ingestion and transfer of microplastics in the planktonic food web. Environ Pollut 185:77–83. https://doi.org/10.1016/j.envpol.2013.10.013
Shruti VC, Pérez-Guevara F, Elizalde-Martínez I, Kutralam-Muniasamy G (2020) First study of its kind on the microplastic contamination of soft drinks, cold tea and energy drinks - future research and environmental considerations. Sci Total Environ 726:138580. https://doi.org/10.1016/j.scitotenv.2020.138580
So WK, Chan K, Not C (2018) Abundance of plastic microbeads in Hong Kong coastal water. Mar Pollut Bull 133:500–505. https://doi.org/10.1016/j.marpolbul.2018.05.066
Sobhani Z, Luo Y, Gibson CT et al (2022) Collecting microplastics in gardens: case study (ii) from ropes. Environ Technol Innov 26:102322. https://doi.org/10.1016/j.eti.2022.102322
Soltani NS, Taylor MP, Wilson SP (2021) Quantification and exposure assessment of microplastics in Australian indoor house dust. Environ Pollut 283:117064. https://doi.org/10.1016/j.envpol.2021.117064
Stock V, Laurisch C, Franke J et al (2021) Uptake and cellular effects of PE, PP, PET and PVC microplastic particles. Toxicol Vitr 70:105021. https://doi.org/10.1016/j.tiv.2020.105021
Sussarellu R, Suquet M, Thomas Y et al (2016) Oyster reproduction is affected by exposure to polystyrene microplastics. Proc Natl Acad Sci USA 113:2430–2435. https://doi.org/10.1073/pnas.1519019113
Talvitie J, Mikola A, Koistinen A, Setälä O (2017) Solutions to microplastic pollution – removal of microplastics from wastewater effluent with advanced wastewater treatment technologies. Water Res 123:401–407. https://doi.org/10.1016/j.watres.2017.07.005
Torres-Agullo A, Karanasiou A, Moreno T, Lacorte S (2022) Airborne microplastic particle concentrations and characterization in indoor urban microenvironments. Environ Pollut 308:119707. https://doi.org/10.2139/ssrn.4058106
Tympa LE, Katsara K, Moschou PN et al (2021) Do microplastics enter our food chain via root vegetables? A raman based spectroscopic study on raphanus sativus. Materials (basel) 14:1–11. https://doi.org/10.3390/ma14092329
Uddin S, Fowler SW, Habibi N et al (2022) Indoor aerosol — Kuwait’s baseline. Toxics 10:71. https://doi.org/10.3390/toxics10020071
Ustabasi GS, Baysal A (2019) Occurrence and risk assessment of microplastics from various toothpastes. Environ Monit Assess 191:438. https://doi.org/10.1007/s10661-019-7574-1
Van Cauwenberghe L, Claessens M, Vandegehuchte MB, Janssen CR (2015) Microplastics are taken up by mussels (Mytilus edulis) and lugworms (Arenicola marina) living in natural habitats. Environ Pollut 199:10–17. https://doi.org/10.1016/j.envpol.2015.01.008
Vianello A, Jensen RL, Liu L, Vollertsen J (2019) Simulating human exposure to indoor airborne microplastics using a breathing thermal manikin. Sci Rep 9:1–11. https://doi.org/10.1038/s41598-019-45054-w
Wang J, Guo X, Xue J (2021) Biofilm-developed microplastics as vectors of pollutants in aquatic environments. Environ Sci Technol 55:12780–12790. https://doi.org/10.1021/acs.est.1c04466
Watts AJR, Urbina MA, Corr S et al (2015) Ingestion of plastic microfibers by the crab Carcinus maenas and its effect on food consumption and energy balance. Environ Sci Technol 49:14597–14604. https://doi.org/10.1021/acs.est.5b04026
Watts AJR, Urbina MA, Goodhead R et al (2016) Effect of microplastic on the gills of the shore crab Carcinus maenas. Environ Sci Technol 50:5364–5369. https://doi.org/10.1021/acs.est.6b01187
Weber A, Schwiebs A, Solhaug H et al (2022) Nanoplastics affect the inflammatory cytokine release by primary human monocytes and dendritic cells. Environ Int 163:107173. https://doi.org/10.1016/j.envint.2022.107173
Wu X, Liu P, Wang H et al (2021) Photo aging of polypropylene microplastics in estuary water and coastal seawater: important role of chlorine ion. Water Res 202:117396. https://doi.org/10.1016/j.watres.2021.117396
Xumiao L, Prata JC, Alves JR, et al (2021) Airborne microplastics and fibers in indoor residential environments in Aveiro, Portugal. Environmental Advances 6:. https://doi.org/10.1016/j.envadv.2021.100134
Yang Y, Liu W, Zhang Z et al (2020) Microplastics provide new microbial niches in aquatic environments. Appl Microbiol Biotechnol 104:6501–6511. https://doi.org/10.1007/s00253-020-10704-x
Yao Y, Glamoclija M, Murphy A, Gao Y (2022) Characterization of microplastics in indoor and ambient air in northern New Jersey. Environ Res 207:112142. https://doi.org/10.1016/j.envres.2021.112142
Yu P, Liu Z, Wu D et al (2018) Accumulation of polystyrene microplastics in juvenile Eriocheir sinensis and oxidative stress effects in the liver. Aquat Toxicol 200:28–36. https://doi.org/10.1016/j.aquatox.2018.04.015
Yu X, Lang M, Huang D et al (2022) Photo-transformation of microplastics and its toxicity to Caco-2 cells. Sci Total Environ 806:150954. https://doi.org/10.1016/j.scitotenv.2021.150954
Yurtsever M (2019) Tiny, shiny, and colorful microplastics: are regular glitters a significant source of microplastics? Mar Pollut Bull 146:678–682. https://doi.org/10.1016/j.marpolbul.2019.07.009
Zhang J, Wang L, Kannan K (2019) Polyethylene terephthalate and polycarbonate microplastics in pet food and feces from the United States. Environ Sci Technol 53:12035–12042. https://doi.org/10.1021/acs.est.9b03912
Zhang J, Wang L, Kannan K (2020a) Microplastics in house dust from 12 countries and associated human exposure. Environ Int 134:105314. https://doi.org/10.1016/j.envint.2019.105314
Zhang Q, Zhao Y, Du F et al (2020b) Microplastic fallout in different indoor environments. Environ Sci Technol 54:6530–6539. https://doi.org/10.1021/acs.est.0c00087
Zhang T, Sun Y, Song K et al (2021) Microplastics in different tissues of wild crabs at three important fishing grounds in China. Chemosphere 271:129479. https://doi.org/10.1016/j.chemosphere.2020.129479
Zhang X, Liu C, Liu J et al (2022) Release of microplastics from typical rainwater facilities during aging process. Sci Total Environ 813:152674. https://doi.org/10.1016/j.scitotenv.2021.152674
Zhu J, Zhang X, Liao K et al (2022) Microplastics in dust from different indoor environments. Sci Total Environ 833:155256. https://doi.org/10.1016/j.scitotenv.2022.155256
Zhu K, Jia H, Sun Y et al (2020) Long-term phototransformation of microplastics under simulated sunlight irradiation in aquatic environments: roles of reactive oxygen species. Water Res 173:115564. https://doi.org/10.1016/j.watres.2020.115564
Ziajahromi S, Kumar A, Neale PA, Leusch FDL (2017a) Impact of microplastic beads and fibers on waterflea (Ceriodaphnia dubia) survival, growth, and reproduction: implications of single and mixture exposures. Environ Sci Technol 51:13397–13406. https://doi.org/10.1021/acs.est.7b03574
Ziajahromi S, Neale PA, Rintoul L, Leusch FDL (2017b) Wastewater treatment plants as a pathway for microplastics: development of a new approach to sample wastewater-based microplastics. Water Res 112:93–99. https://doi.org/10.1016/j.watres.2017.01.042
Zimmermann L, Göttlich S, Oehlmann J et al (2020) What are the drivers of microplastic toxicity? Comparing the toxicity of plastic chemicals and particles to Daphnia magna. Environ Pollut 267:115392. https://doi.org/10.1016/j.envpol.2020.115392
Author information
Authors and Affiliations
Contributions
Angel Jessieleena: conceptualization, problem definition and description, investigation, writing — part of the original draft, original draft compilation, review and editing and revision.
Sasikaladevi Rathinavelu: supervision, problem description, writing — part of the original draft.
Kiruthika Eswari Velmaiel: problem description, writing — part of the original draft.
Anju Anna John: problem description, writing — part of the original draft.
Indumathi M. Nambi: supervision, problem description, resources, project administration, writing — review and editing.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors have consented to publish this work.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Philippe Garrigues
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jessieleena, A., Rathinavelu, S., Velmaiel, K.E. et al. Residential houses — a major point source of microplastic pollution: insights on the various sources, their transport, transformation, and toxicity behaviour. Environ Sci Pollut Res 30, 67919–67940 (2023). https://doi.org/10.1007/s11356-023-26918-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-26918-1