Abstract
The iron ore tailings released into the Rio Doce basin after the Fundão dam collapse (Brazil), suppressed a large extent of local vegetation. The use of native species and appropriate fertilization techniques, with less economic and environmental impact, must be considered in the process for the restoration of affected areas by the tailings. For this purpose, six native tree species, pioneer (Anadenanthera colubrina, Bixa orellana, and Peltophorum dubium) and secondary (Cedrela fissilis, Handroanthus impetiginosus, and Handroanthus serratifolius), were selected. We used different conditions of fertilization: (1) inorganic fertilization, (2) inoculation with arbuscular mycorrhizal fungi and plant growth-promoting rhizobacteria, (3) combined treatment (fertilizer + inoculum), to evaluate leaf nutrient concentrations, photosynthetic capacity [chlorophyll index, maximum quantum efficiency of photosystem II (Fv/Fm) and gas exchange variables], and oxidative metabolism (H2O2, MDA, and antioxidant enzymes). Inoculation resulted in higher concentrations of foliar nitrogen, especially in pioneer species. In all treatments, the secondary species exhibited iron values considered phytotoxic, but showed reduced photosynthetic capacity only when inoculated. The highest concentrations of MDA were observed in inoculated plants of both successional groups. The antioxidant system proved to be effective in preventing oxidative damage for most of the species. These results showed that the use of inoculum can be considered an ecological alternative to inorganic additives in the area affected by iron ore tailings. Despite presenting different photosynthetic and antioxidant strategies, the evaluated species demonstrated potential for use in tailings revegetation projects.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
A large extension of the Rio Doce basin is inserted in the Atlantic Forest, one of the most degraded biomes on the planet and a global hotspot for biodiversity conservation (Myers et al. 2000). The development of unsustainable human activities (mining, smelting, pasture, extensive agriculture, among others) in this basin has led to the removal of significant amounts of vegetation and successive depositions of potentially toxic metals in the environment over time (Santolin 2015). The large input of metals as a result of the intense extraction and processing of ore and the indiscriminate use of fertilizers in agricultural areas has generated chronic contamination of the waters and soils of the region (Hora et al. 2012). Added to the extensive history of degradation was the environmental disaster resulting from the failure of the Fundão dam in Mariana (Minas Gerais, Brazil) in November 2015. The failure resulted in the dumping of a large amount of iron ore tailings, which suppressed part of the vegetation and caused changes in the chemical and physical composition of soils in the middle Rio Doce basin (Hatje et al. 2017). Natural regeneration of local vegetation could be compromised due to the chemical composition of the tailings, with a predominance of low availability of organic matter and essential nutrients for plant development, as well as high concentrations of trace metals such as iron and manganese (Segura et al. 2016; Andrade et al. 2018; Cruz et al. 2020).
When excessively absorbed, trace metals tend to accumulate in cells, affecting the metabolism and causing symptoms of toxicity in plants (Sharma and Agrawal 2005). Plants under stress by trace metals experience an exacerbated production of reactive oxygen species (ROS) due to the direct transfer of electrons between free O2, resulting from photochemical reactions, and transition metals such as Mn and Fe (Hajiboland 2014). Excessive production of ROS can generate a significant increase in lipid peroxidation by-products, such as malonaldehyde (MDA), and impact photosynthetic activity (Rai et al. 2016) through energy imbalance and reduced assimilation and/or fixation of carbon (Takahashi and Murata 2008). On the other hand, ROS overproduction stimulates biosynthesis and activation of the antioxidant system, which reduces the incidence of cellular damage by oxidative stress (Bailly et al. 2008). When efficient in maintaining ROS levels, the antioxidant system gives plants greater cellular protection and tolerance to survive in substrates contaminated by trace metals (Gomes et al. 2014a).
Studies have shown that the development of seedlings of both native tree species and agricultural species in this sediment was limited by the low concentration of nutrients (Segura et al. 2016; Andrade et al. 2018; Cruz et al. 2020, 2021; Almeida et al. 2022a, 2022b). Fertilization is necessary for the planting of plant species in degraded and/or contaminated soils, and nutrient addition is generally carried out with nitrogen and phosphate chemical fertilizers (Tian et al. 2003). However, cleaner revegetation techniques, from an ecological point of view, have been developed. One such technique is the cultivation of plants inoculated with arbuscular mycorrhizal fungi (AMF) and plant growth-promoting rhizobacteria (PGPR) (Burity et al. 2000). In addition to reducing the high costs of chemical fertilization, these types of mutualistic associations also represent an alternative for sustainable fertilization capable of giving plants greater capacity for the absorption of essential elements (mainly phosphorus and nitrogen), with direct implications for the performance of plants in the field (Tian et al. 2003).
Based on these premises, this study aimed to investigate the effects of inorganic fertilization, inoculation with AMF and PGPR and combined treatment (inorganic fertilization + inoculation) on leaf nutritional status, photosynthetic performance and oxidative metabolism of six native pioneer and secondary tree species growing in an area impacted by the iron ore tailings extravasated from the Fundão dam. The study aimed to answer the following questions: (1) are the different fertilization conditions equally efficient at promoting the nutritional status required for the maintenance of the essential physiological processes of native tree species? (2) under field conditions, do plants of different successional groups also exhibit different physiological strategies to develop in the tailings with high concentrations of iron and manganese? (3) does physiological response to fertilization treatments differ between species of the same successional groups?
Material and methods
Characterization of the experimental area
Field experiments were carried out in approximately 1.5 hectares of an area affected by iron ore tailings from the failure of the Fundão dam, located at Fazenda Paracatu in the district of Paracatu de Baixo (20°17′52″S 43°14′3″W), municipality of Mariana, Minas Gerais, Brazil. The macroclimate of the region is warm temperate with seasonal water deficit (Cwa by the Köppen System). Monthly averages for air temperature and rainfall for the period between planting and carrying out physiological analyses in the field are shown in Fig. 1.
Tailing nutrient concentrations and pH were evaluated using nine replicates of substrate samples collected from the planting hole, at a depth of no more than 10 cm. Concentrations of the nutrients P, K, Ca, Mg, S, Na, Cu, Ni, Zn, Fe, and Mn were determined in 1-g aliquots of tailings, prepared and extracted according to Rauret and López-Sánchez (2001). Analyses were performed using an inductively coupled plasma optical emission spectrometer (ICP-OES, Agilent 725). The pH was determined in the proportion 1:2.5 (v/v) of tailings: KCl (Teixeira et al. 2017). The chemical characterization of the formed technosoil is shown in Table 1.
Plant material and experimental design
Three pioneer species [Anadenanthera colubrina (Vell.) Brenan (Fabaceae), Bixa orellana L. (Bixaceae), Peltophorum dubium (Spreng.) Taub. (Fabaceae)] and three secondary species [Cedrela fissilis Vell. (Meliaceae), Handroanthus impetiginosus (Mart. ex DC.) Mattos (Bignoniaceae), and Handroanthus serratifolius (Vahl) S.O.Grose (Bignoniaceae)] were selected based on their natural occurrence in the Rio Doce basin (Lombardi and Gonçalves 2000) and on the results obtained by Cruz (2018). Seedlings were produced directly in PVC tubes containing substrate fertilized with mixed mineral fertilizer “Osmocote Plus 15–9-12” (15% N, 9% P2O5, 12% K2O, 1.3% Mg, 6% S, 0.05% Cu, 0.46% Fe, 0.06% Mn, and 0.02% Mo; Dublin, US). In the nursery, 90 days before planting, inoculum based on microorganisms, obtained by the on farm technique (Czerniak and Stümer 2014), containing AMF and PGPR was added to the seedling substrate intended for inoculation treatments. The inoculum was provided by the micro-company Cogumê Biotecnologia in partnership with the Laboratório de Associações Micorrízicas of the Universidade Federal de Viçosa (Brazil).
Three treatments were used in field planting: (1) fertilization (F), in which plots received only inorganic fertilizer (100% of the dose of Osmocote Plus 15–9-12 recommended for native trees; 120 g of fertilizer per planting hole); (2) combined fertilization (FI), in which plots received half the dose of both the fertilizer and the inoculum based on AMF and PGPR; and (3) inoculation (I), in which the plots received only inoculum (150 g of compost per planting hole). Three plots with similar topography were used for each treatment for comparative purposes. Each plot received 20 seedlings of each one of the six plant species. In situ planting was carried out in the first week of January 2020 (rainy season). The seedlings, with about 30 cm in height, were arranged linearly, with a spacing of 3 m × 3 m, alternating the species of the successional groups. Planting was monitored and invasive species were controlled by manual mowing the plots.
Leaf nutrient concentrations, photosynthetic parameters, and markers of oxidative metabolism were evaluated 5 months after planting using fully expanded and visually healthy leaves of nine individuals (replications) per treatment, per species.
Leaf nutrient concentrations
The concentrations of N, P, K, Ca, Mg, S, Zn, Fe, and Mn were determined after the leaves were cleaned in running water, dried at 65 ºC in a forced circulation oven (FANEM 320-SE) until constant weight and macerated in a vibrating mill (Retsch MM 400). Samples (200 mg of ground plant tissue) were digested in nitro-perchloric solution (3:1) and submitted to mineral content quantification by atomic absorption spectrophotometry (Malavolta et al. 1997).
Photosynthetic parameters
Photosynthetic variables (total chlorophyll, chlorophyll a fluorescence, and gas exchange) were evaluated in the second pair of leaves from the shoot apex. Total chlorophyll content was determined from three measurements carried out in the middle portion of the leaf, using a portable chlorophyll meter ClorofiLOG (CFL1030, Falker, Brasil). Gas exchange analyses were performed using a Li 6400XT infrared gas analyzer Li 6400XT (Li-Cor Inc., Lincoln, NE, USA). Light intensity (1500 µmol m−2 s−1) was maintained by a LED light source (6400-02B, Li-Cor Inc.). Measurements were made using a CO2 control system (6400–01, Li-Cor Inc.) at 400 µmol mol−1 CO2. During evaluation days, the average air temperature fluctuated between 29 and 30 °C and air relative humidity between 49 and 51%. The obtained data were used to determine net photosynthesis (A, µmol m−2 s−1), stomatal conductance (gs, mol m−2 s−1), transpiration (E, mmol m−2 s−1), internal and external CO2 concentration ratio (Ci/Ca, µmol mol−1), and instantaneous water use efficiency (WUEi = A/E). Chlorophyll a fluorescence was evaluated in the same leaves, in parallel with gas exchange measurements, using a Mini-PAM fluorometer (Heinz Walz, Effeltrich, Germany). Minimum fluorescence (F0) and maximum fluorescence (Fm) were determined after acclimating leaves in the dark for at least 30 min. The maximum quantum efficiency of photosystem II (PSII; Fv/Fm) was calculated based on the values of F0 and Fm (Fm – F0) / Fm (Genty et al. 1989).
After photosynthetic evaluations, the leaves were collected, weighed, and stored at − 80 °C until the time of biochemical evaluations.
Quantification of H2O2 and MDA
Quantification of H2O2 was performed with leaf tissue samples (0.1 g) macerated in liquid nitrogen, homogenized with 0.1% trichloroacetic acid (TCA), and centrifuged at 12,000 g for 15 min. The extract then received 10 mM potassium phosphate buffer, pH 7.0 and 1 M potassium iodide (KI). The absorbance was read at 390 nm and the H2O2 content determined by a standard curve, according to Velikova et al. (2000).
Lipid peroxidation was estimated for leaf tissue samples (0.05 g) macerated in liquid nitrogen and homogenized in 1 mL of 80% ethanol. After three extractions, 1 mL of the final volume was used for reaction with 1 mL of 20% TCA solution, 0.65% thiobarbituric acid (TBA), and 0.01% butylhydroxytoluene (BHT) as a positive control. The extent of lipid peroxidation, according to MDA levels, was determined spectrophotometrically according to Du and Bramlage (1992).
Enzymatic antioxidant system
Extract for quantification of antioxidant enzymes was prepared according to Gomes et al. (2014b). Leaf tissue samples (0.1 g) were macerated in liquid nitrogen and homogenized with 1 mL of potassium phosphate buffer (100 mM at pH 7.8), with 100 mM of EDTA, 1 mM of ascorbic acid, and 5% of PVP. The extract was centrifuged at 12,000 g (4 °C) and used to determine enzyme activity and protein content. Protein dosage was performed by the method of Bradford (1976).
Catalase activity (CAT; EC1.11.1.6) was determined according to Aebi (1984); ascorbate peroxidase (APX; EC 1.11.1.11) according to Nakano and Asada (1981); superoxide dismutase (SOD; EC 1.15.1.1) according to Giannopolitis and Ries (1977); and glutathione reductase (GR; EC 1.6.4.2) according to Foyer and Halliwell (1976).
Statistical analysis
Data were previously submitted to tests for normality of residuals (Shapiro–Wilk) and homogeneity of variances (Levene’s test). Normal and homoscedastic or successfully transformed data (log + 1) were submitted to analysis of variance by ANOVA, followed by comparison of means (Tukey’s test) with a significance level of p < 0.05. Transformed data that did not meet ANOVA assumptions were submitted to the non-parametric Kruskal Wallis test, using R software version 4.0.2 (R Development Core Team 2020). The dataset was also used to create a Principal Component Analysis (PCA) correlation matrix using factoextra and readxl packages of R software version 4.0.2.
Results
Leaf nutrient concentrations
Leaf tissue nutrient concentrations were partially affected by treatment, depending on the species (Table 2). The species A. colubrina and H. impetiginosus had higher N concentrations when submitted to both inoculation treatments (FI and I) (p < 0.05). The species B. orellana, P. dubium and C. fissilis had higher N when exposed to inoculation and H. serratifolius when growing in the combined treatment (p < 0.05). No significant differences were found for leaf P concentration among treatments for all species, with the exception of H. serratifolius, which had higher concentrations in fertilization and combined treatments (p < 0.05). There were no significant differences in concentrations of K, Mg, Ca, S, and Zn among treatments for all species evaluated (p > 0.05). Higher concentrations of Mn were found for the pioneer species A. colubrina, B. orellana, and P. dubium when treated with fertilization (F and/or FI) (p < 0.05). Higher concentrations of Mn were also observed for the secondary species C. fissilis and H. impetiginosus when exposed to fertilization and for H. serratifolius when growing in inoculation treatment (p < 0.05). The species A. colubrina, B. orellana, P. dubium, and H. impetiginosus had higher Fe when subjected to fertilization treatments (F and FI), as did C. fissilis when exposed to only inorganic fertilizer (F) and H. serratifolius when exposed to the combined treatment (p < 0.05; Table 2).
Photosynthetic parameters
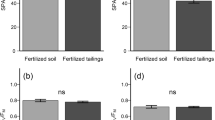
Chlorophyll content did not vary significantly among treatments for B. orellana, P. dubium and H. impetiginosus (p > 0.05). However, A. colubrina had lower chlorophyll values when subjected to fertilization and C. fissilis and H. serratifolius when subjected to inoculation, compared to the other treatments (p < 0.05). The only difference observed for maximum efficiency of PSII (Fv/Fm) was a lower value for C. fissilis in the inoculation treatment (I) (p < 0.05; Fig. 2).
Total chlorophyll content (index of chlorophyll Falker – ICF), maximum quantum yield of PSII (Fv/Fm), CO2 assimilation rate (A), stomatal conductance (gs), transpiration rate (E), ratio of internal to external CO2 (Ci/Ca), instantaneous water use efficiency (WUEi) of tree species submitted to inorganic fertilization (F), combined treatments (FI), and inoculation (I). The bars represent the means ± SD (n = 9). Different letters indicate significant differences between treatments (P < 0.05). ns = non-significant difference
No significant differences were observed among treatments for net photosynthetic rate (A), with the exception of H. impetiginosus and H. serratifolius, for which the lowest values were for the inoculation treatment (p < 0.05). The highest values for stomatal conductance (gs) were observed for P. dubium, H. impetiginosus, and H. serratifolius of fertilization treatment and for A. colubrina subjected to combined treatment (p < 0.05). There was no significant difference in gs among treatments for B. orellana and C. fissilis (p > 0.05). A higher transpiration rate (E) was observed for B. orellana, P. dubium, and H. impetiginosus of the fertilization treatment (F), for A. colubrina exposed to the combined fertilization treatment (FI), and for H. serratifolius of treatments F and FI (p < 0.05). Differences in the internal and external carbon ratio (Ci/Ca) were detected only for A. colubrina and P. dubium, with the highest values of Ci/Ca for both fertilization treatments (p < 0.05). The species B. orellana and P. dubium exhibited higher instantaneous water use efficiency (WUEi) for combined treatment and lower for fertilization treatment (p < 0.05). The species H. impetiginosus and H. serratifolius had higher WUEi values with the fertilization treatments (F and FI) (p < 0.05; Fig. 2).
Quantification of MDA and H2O2
Leaf concentrations of MDA and H2O2 were affected by treatment for most of the investigated species (p < 0.05; Fig. 3). Leaf tissue MDA concentration was higher for plants submitted to the inoculation treatment, than for those in the other treatments, for all species studied (p < 0.05). The species A. colubrina, B. orellana, C. fissilis, and H. impetiginosus had their highest H2O2 values when submitted to treatments containing inoculum, while P. dubium had a higher H2O2 concentration when submitted to inorganic fertilized treatments (p < 0.05). There was no difference in H2O2 concentration among treatments for H. serratifolius (p > 0.05).
Concentrations of malondialdehyde (MDA), hydrogen peroxide (H2O2), and antioxidant enzymes activity (CAT, APX, GR e SOD) of tree species submitted to inorganic fertilization (F), combined treatments (FI), and inoculation (I). The bars represent the means ± SD (n = 9). Different letters indicate significant differences between treatments (P < 0.05). ns = non-significant difference
Enzymatic antioxidant system
Antioxidant system enzyme activity is shown in Fig. 3. Most species, except C. fissilis, had higher CAT activity when treated with inoculation (FI and/or I), in relation to the fertilization treatment (F) (p < 0.05). The pioneer species A. colubrina and P. dubium exhibited higher APX activity in the combined treatment, and the secondary species C. fissilis in both treatments involving fertilization (F and FI; p < 0.05). The three pioneer species (A. colubrina, B. orellana, and P. dubium) showed higher GR activity when treated with fertilization (F and FI; p < 0.05). For half of the investigated species, the SOD activity was affected by treatments (P < 0,05). A. colubrina exhibited higher SOD activity when treated with inoculation (I), while for P. dubium and C. fissilis, the higher SOD values were observed in plants grown in both fertilization treatments (F and FI). In addition, the maximum value of SOD activity observed in the pioneer species was 0.03 ± 0.01 U min−1 g protein−1, while for the secondary species the values reached 0.06 ± 0.01 U min−1 g protein−1 (Fig. 3).
Principal component analysis
The principal component analysis (PCA) shows the relationship between the physiological parameters measured for the native tree species during initial growth in the tailings and the different fertilization conditions promoted in the field. The principal components generated by the PCA explained 43.7% of the variability in the data, with 23.2% of this total by dimension 1 (Dim 1), and 20.5% by dimension 2 (Dim 2) (Fig. 4). The PCA shows a clear separation of species according to ecological functional groups, with opposite ordering between the pioneer species A. colubrina, B. orellana, and P. dubium and the secondary species C. fissilis, H. impetiginosus, and H. serratifolius. The PCA also correlates the studied species with strategies that favored plant tolerance in the field. The responses of pioneer species (A. colubrina, B. orellana, and P. dubium) were characterized by the highest concentration of N, greater water use efficiency, and the maintenance of photosynthetic assimilation, transpiration capacity, and antioxidant enzyme activity. In contrast, the physiological responses of late secondary species to tailings with different fertilization conditions were modulated by foliar concentrations of the nutrients iron and manganese, total chlorophyll, and parameters indicative of oxidative stress (MDA and H2O2) (Fig. 4).
Principal component analysis (PCA) for the relationship between the nutrients concentration and physiological parameters evaluated in plants grown in iron ore tailing from the Fundão dam, Mariana, Minas Gerais (Brazil) and subject to treatments of inorganic fertilization (F), combined treatments (FI), and inoculation (I)
Discussion
The different fertilization conditions in the field promoted the development of native trees and allowed inferences to be made about possible ecological strategies that contributed to the establishment of plants in the tailings. In general, the treatments used were equally effective in maintaining the nutritional and physiological performance of the species evaluated in this study.
The highest nitrogen values found in the leaf tissue of plants of the inoculation treatments (FI and/or I) represented an increase of about 23% in the pioneer species and from 19 to 35% in the secondary species, in comparison with the fertilized plants (F). All species were able to maintain satisfactory phosphorus levels for initial development, around 0.2 dag/kg−1 according to Marschner (2012), in all treatments, even those of H. serratifolius, which had a lower concentration of this nutrient in the inoculation treatment (I). Both nitrogen and phosphorus are macronutrients that play fundamental roles in the photosynthetic process (Harper 1994), being constituents of essential biomolecules for this metabolism, such as chlorophylls and ATP (Hawkesford et al. 2012). The use of a microorganism-based inoculum (such as AMF and RBCP) is highly recommended in projects to restore impacted areas (Berruti et al. 2016), including mining tailings (Gamalero et al. 2009), as they help the revegetation process by supplying fixed nitrogen and making phosphorus bioavailable to plants, in exchange for photoassimilates (Sheoran et al. 2010). The inoculation technique is commonly performed with plants of the family Fabaceae, due to the well-established relationship between its species and nitrogen-fixing rhizobacteria (Neuenkamp et al. 2019). However, the present study suggested that, in addition to those of Fabaceae, plants of species of the families Bignoniaceae, Bixaceae, and Meliaceae may respond positively to inoculation with greater accumulation of nitrogen in their leaves in this treatment. The increased absorption of phosphorus by plant species in symbiosis has been consistently reported in the literature (Matias et al. 2009; Prates Júnior et al. 2021). However, in the present study, a higher concentration of phosphorus was not observed in plants submitted to inoculation treatments, compared to fertilization treatment.
Although they are essential nutrients for proper plant growth, when in excess, manganese and iron can become toxic to plants (Hänsch and Mendel 2009). Manganese acts as a cofactor in the water oxidation process of photosystem II for the generation of O2 and also as a ligand for enzymes involved in antioxidant defense (e.g., SOD-Mn) and nitrogen metabolism (glutamine synthetase and arginase) (Hänsch and Mendel 2009). Reference values for the toxicity of this nutrient are not clear for many plant species (El-Jaoual and Cox 1998), however, foliar concentrations above 50 mg kg−1 dry weight are considered satisfactory for plant growth (Marschner 2012). The concentration of manganese in leaves found here (between 42.9 and 316.9 mg kg−1) is within the range expected for terrestrial plants (5–2000 mg kg−1 of leaf dry matter), according to Sheoran et al. (2010). Symptoms of manganese toxicity were not observed in plants studied here. Further, they were similar to the concentrations observed by Silva et al. (2021) for P. dubium in hydroponic cultivation and significantly lower than the values recorded by Cruz et al. (2020) and Cruz et al. (2021) for tropical trees growing on fertilized tailings in a greenhouse.
Iron concentrations considered phytotoxic (above 500 mg kg−1 of dry leaf mass) (Pugh et al. 2002) were found for the species A. colubrina in treatments F and FI and for C. fissilis, H. impetiginosus, and H. serratifolius, regardless of treatment. Similarly, phytotoxic concentrations of iron were found in leaves of herbaceous plants (Zago et al. 2019; Rios et al. 2021), trees (Cruz et al. 2020, 2021; Matos et al. 2020), and plants of agricultural interest (Almeida et al. 2022a) when grown in a substrate rich in iron ore residues. Iron is an essential metal in several physiological processes, such as the biosynthesis of DNA and plant hormones, nitrogen metabolism, respiration, and photosynthesis (Hänsch and Mendel 2009); is required as a cofactor of enzymes such as SOD-Fe, catalase, and ascorbate peroxidase; and has a central role as a catalyst for redox reactions (Becana et al. 1998). When present in high concentrations in leaf tissue, this transition metal induces overproduction of reactive oxygen species (ROS) via the Fenton reaction, which can cause irreversible damage to cell membranes (Becana et al. 1998; Kobayashi and Nishizawa 2012).
Interestingly, plants of the inoculated treatment showed up to three times lower iron content than plants from the fertilized treatments, with the exception of those of H. serratifolius. Plants subjected to high concentrations of trace metals in the soil developed strategies that result in the maintenance of ion homeostasis in cellular compartments and relief from phytotoxicity (Clemens 2001; Petrisor et al. 2004; Tank and Saraf 2009). Association with symbiotic microorganisms is a way to guarantee greater plant tolerance in environments contaminated by trace metals (Wu et al. 2007; Gamalero et al. 2009; Berruti et al. 2016). Plants in symbiosis with microorganisms may show reduced iron absorption and less accumulation of this metal in leaf tissues compared to fertilized plants, as observed by Petrisor et al. (2004), and corroborated by the results of the present study. This lower accumulation of iron in the leaves of inoculated plants may be related to the ability of microorganisms to immobilize potentially toxic compounds in their cell wall, vacuole, or cytoplasm (Punamiya et al. 2010), before they are absorbed by the plant (Begum et al. 2019). PGPR has a recognized role in iron chelation, via the production and activity of siderophores (Crowley et al. 1991), which, by incorporating it into their cellular metabolism, make it less available in the rhizosphere and, consequently, for plant uptake (Tank and Saraf 2009). Despite this, further investigation is required to clarify the role of microorganisms in the resistance of plants grown on substrates rich in iron ore.
The lower chlorophyll content observed in A. colubrina and H. serratifolius of treatments F and I, respectively, may be a result of the lower nitrogen concentration and higher iron concentration in the leaves of these species. Lower chlorophyll values in leaves of C. fissilis of the inoculation treatment (I) may be associated with the effects of iron phytotoxicity. Iron is required in chloroplasts as an essential mineral element for the synthesis of chlorophylls and as a cofactor involved in redox reactions in the electron transport chain (Hell and Stephan 2003; Broadley et al. 2012). However, when iron is in excess in the mesophyll, damage to pigments and other components of the photosynthetic chain can be triggered as a result of increased ROS and consequent oxidative stress (Kobayashi and Nishizawa 2012). Decreased chlorophyll concentration is one of the negative effects caused by ROS and has been regularly described for plants growing in substrate rich in iron ore residues (Pereira et al. 2013; Rios et al. 2021). Decreased chlorophyll content is reflected in photosystem II (PSII) performance (Baker 2008). Lower activity of PSII is observed in plants under stress, indicating less efficiency in the use of light energy for photosynthesis and disturbance or damage to the photosynthetic apparatus (Roosta et al. 2018). Studies show that herbaceous and tree plants growing in the fertilized tailings from the Fundão dam, despite having potentially toxic concentrations of iron in leaf tissue, did not change the Fv/Fm ratio (Esteves et al. 2020; Cruz et al. 2020, 2021), as corroborated by the present results, except for C. fissilis.
The lowest values of A, gs, E, and WUEi found for H. impetiginosus and H. serratifolius in treatments that involved inoculation (FI and/or I) corroborate the findings of Pereira et al. (2013) for rice plants, in which reduced photosynthesis was positively correlated with stomatal limitation developed in leaves with phytotoxic concentrations of iron. In this scenario, the reduction in gas exchange responses limits the transport of iron to the aerial part of the plant and thus minimizes oxidative damage due to high concentrations of it in leaf tissue (Kobayashi and Nishizawa 2012; Pereira et al. 2013). No reduction in photosynthesis or stomatal limitation was observed for the pioneer species studied here. The species B. orellana and P. dubium responded to the combined (FI) and inoculation (I) treatments with greater water efficiency, although the WUEi parameter was not positively correlated with the highest Ci/Ca ratio or the highest gs. For both species, greater water efficiency seems to have been favored by the insertion of symbiotic microorganisms in the planting substrate. The use of symbiotic microorganisms in association with plant species is recognized as favoring plant tolerance of water stress, as they help plants actively explore greater volumes of soil through the extension of their mycelia and, thus, reach new sources of water in the substrate (Saggin Junior and Silva 2006; Li et al. 2019).
Plants of all the investigated species exhibited higher concentrations of MDA in the inoculation treatment compared to the fertilization treatment. For most species, higher concentrations of H2O2 were observed in the inoculation treatment (I.) There was a reduction in photosynthetic activity and efficiency in the secondary species, however, the results of the present study suggest damage by oxidative stress only for C. fissilis. Maintaining phytotoxic concentrations of trace metals in leaves favors an exacerbated production of ROS, such as H2O2, and potentiates the occurrence of oxidative damage to cellular structures (Becana et al. 1998; Kobayashi and Nishizawa 2012; Gomes and Garcia 2013; Gomes et al. 2014a). ROS react with lipids, proteins, pigments, and nucleic acids, causing peroxidation of cellular components and membranes (Hajiboland 2014). In chloroplasts, oxidative damage limits photosynthetic efficiency, carbon assimilation, and energy yield and can culminate in cell death (Takahashi and Murata 2008; Hajiboland 2014). On the other hand, non-toxic concentrations of H2O2 promote intracellular signaling (Henzler and Steudle 2000). Characteristics, such as moderate reactivity, long cellular half-life, and ability to freely diffuse across membranes and over long distances, involve H2O2 in the control of different physiological responses to biotic and abiotic factors (Foyer and Noctor 2005). Under stress conditions, stomatal closure, activation of heat shock proteins, and regulation of hormone cascades, such as those of abscisic acid and salicylic acid, are examples of H2O2-mediated responses that impact plant tolerance in adverse habitats (Tuteja 2007; Pandey et al. 2021). Similarly, MDA, a molecule of low chemical reactivity, also plays a signaling role in plants under stress, as it stimulates the expression of genes for enzymes and antioxidant molecules (Morales and Munné-Bosch 2019), which modulate concentrations of ROS and reduce the chances of oxidative stress occurring (Gajić et al. 2020).
In general, the enzymes of the antioxidant system were effective in controlling H2O2 in leaves of the species investigated here. There was an increase in CAT concentration for plants of the inoculation treatment, regardless of the ecological group, compared to plants submitted to fertilization. Pioneer species treated with fertilization showed higher GR activity, while secondary species, regardless of treatment, exhibited higher SOD concentrations in leaf tissue. The proper balance between production and neutralization of H2O2 is essential for the survival of plants in degraded ecosystems and involves the antioxidant defense system (Foyer and Noctor 2005). CAT, together with APX, contributes to the reduction of oxidative damage by converting H2O2 into water and oxygen (Sharma and Ahmad 2014). The action of GR contributes, via maintenance of the reduced glutathione pool (GSH), to the regeneration of ascorbic acid — an indispensable substrate for the functioning of APX (Noctor and Foyer 1998). SOD acts by promoting the detoxification of O2•− and, thus, modulates the levels of H2O2 to be neutralized in leaf tissue (Hajiboland 2014). Symbiotic microorganisms also contribute to plant resistance in habitats contaminated with trace metals (Vilela et al. 2018). Studies have shown the effective action of beneficial microorganisms in promoting the antioxidant defense of their host plants. According to Wu et al. (2007), the accumulation of H2O2 in AMF structures, such as arbuscules, hyphae, cell walls, mycelia and spores, contributes to the maintenance of non-toxic levels of this ROS in plant tissues. Li et al. (2019) observed that the improvement in the detoxification capacity of inoculated plants, compared to non-inoculated plants, is due to the increase in the concentration of antioxidant enzymes promoted by symbiotic microorganisms, as the present results suggest.
The results of the present study indicate that, in general, the nutritional quality of the studied plants was adequately supplied by the treatments. Thus, both inoculation and fertilization treatments favored the performance of tree species growing in an area affected by iron ore tailings from the Fundão dam. Phytotoxic concentrations of Fe were observed in four of the six investigated species in the fertilization treatment. Only secondary species showed reduced photosynthetic capacity in the inoculation treatment, in relation to the fertilization treatment. Higher concentrations of MDA and H2O2 were associated with the inoculation treatment for species from both ecological groups. Although there was a limitation of photosynthetic activity in secondary species, the antioxidant system proved to be effective in preventing oxidative damage for the evaluated species, with the exception of C. fissilis. In conclusion, the use of inoculum can be considered an ecological alternative to inorganic additives in the area affected by tailings from the Fundão dam and, despite presenting different photosynthetic and antioxidant strategies, the evaluated species can be recommended for use in revegetation projects in areas contaminated with iron ore tailings. Further studies should follow the development of plants in this area to assess whether the high concentration of iron in plant tissues can induce morphophysiological changes and late phytotoxic responses and, thus, affect the resilience of native species in the field, as well as the successional processes in the long term.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Aebi H (1984) Catalase in vitro. methods in enzymology 105:121-126. https://doi.org/10.1016/S0076-6879(84)05016-3
Almeida HA, Silva JG, Custódio IG, Karam D, Garcia QS (2022a) Productivity and food safety of grain crops and forage species grown in iron ore tailings. J Food Compost Anal 105:104198. https://doi.org/10.1016/j.jfca.2021.104198
Almeida HA, Torre FD, Garcia QS (2022b) Cultivating vegetables in tailings from the Fundão damcollapse: metal accumulation and risks to food safety. Environ Monit Assess 194:410–418. https://doi.org/10.1007/s10661-022-10060-5
Andrade GF, Paniz FP, Martins AC Jr, Rocha BA, da Silva Lobato AK, Rodrigues JL, Cardoso-Gustavson P, Masuda HP, Batista BL (2018) Agricultural use of Samarco’s spilled mud assessed by rice cultivation: a promising residue use? Chemosphere 193:892–902. https://doi.org/10.1016/j.chemosphere.2017.11.099
Bailly C, El-Maarouf-Bouteau H, Corbineau F (2008) From intracellular signaling networks to cell death: the dual role of reactive oxygen species in seed physiology. CR Biol 331:806–814. https://doi.org/10.1016/j.crvi.2008.07.022
Baker NR (2008) Chlorophyll fluorescence: a probe of photosynthesis in vivo. Annu Rev Plant Biol 59:89–113. https://doi.org/10.1146/annurev.arplant.59.032607.092759
Becana M, Moran JF, Iturbe-Ormaetxe I (1998) Iron-dependent oxygen free radical generation in plants subjected to environmental stress: toxicity and antioxidant protection. Plant Soil 201:137–147. https://doi.org/10.1023/A:1004375732137
Begum N, Qin C, Ahanger MA, Raza S, Khan MI, Ashraf M, Ahmed N, Zhang L (2019) Role of arbuscular mycorrhizal fungi in plant growth regulation: implications in abiotic stress tolerance. Front Plant Sci 10:1068. https://doi.org/10.3389/fpls.2019.01068
Berruti A, Lumini E, Balestrini R, Bianciotto V (2016) Arbuscular mycorrhizal fungi as natural biofertilizers: let’s benefit from past successes. Front Microbiol 6:1559. https://doi.org/10.3389/fmicb.2015.01559
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Broadley M, Brown P, Cakmak I, Rengel Z, Zhao F (2012) Function of nutrients: micronutrients. In: Marschner P (ed) Marschner’s mineral nutrition of higher plants, 3rd edn. Academic Press, London, pp 191–248. https://doi.org/10.1016/B978-0-12-384905-2.00007-8
Burity HA, Lyra M, Souza ES, Santo Mergulhão ACE, Silva MLRB (2000) Efetividade da inoculação com rizóbio e fungos micorrízicos arbusculares em mudas de sabiá submetidas a diferentes níveis de fósforo. Pesq Agrop Brasileira 35:801–807. https://doi.org/10.1590/S0100-204X2000000400018
Clemens S (2001) Molecular mechanisms of plant metal tolerance and homeostasis. Planta 212:475–486. https://doi.org/10.1007/s004250000458
Crowley DE, Wang YC, Reid CPP, Szaniszlo PJ (1991) Mechanisms of iron acquisition from siderophores by microorganisms and plants. In: Chen Y, Hadar Y (eds) Iron nutrition and interactions in plants. Springer, Dordrecht, pp 213–232. https://doi.org/10.1007/978-94-011-3294-7_27
Cruz FV (2018) Desempenho de sementes e plântulas de espécies arbóreas no rejeito oriundo do rompimento da Barragem de Fundão (Mariana, MG), Master Thesis Universidade Federal de Minas Gerais. http://hdl.handle.net/1843/BUOS-B5PJKW
Cruz FV, Gomes MP, Bicalho EM, Della Torre F, Garcia QS (2020) Does Samarco’s spilled mud impair the growth of native trees of the Atlantic Rainforest? Ecotoxicol Environ Safe 189:110021. https://doi.org/10.1016/j.ecoenv.2019.110021
Cruz FVS, Gomes MP, Bicalho EM, Garcia QS (2021) Fertilization assures mineral nutrition but does not overcome the effects of Fe accumulation in plants grown in iron ore tailings. Environ Sci Pollut Res 29:18047–18062. https://doi.org/10.1007/s11356-021-16989-3
Czerniak MJ, Stümer SL (2014) Produção de inoculante micorrízico on farm utilizando resíduos da indústria florestal. Revista Brasileira De Ciências Do Solo 38:1712–1721. https://doi.org/10.1590/S0100-06832014000600006
Du Z, Bramlage WJ (1992) Modified thiobarbituric acid assay for measuring lipid oxidation in sugar-rich plant tissue extracts. J Agric Food Chem 40:1566–1570. https://doi.org/10.1021/jf00021a018
El-Jaoual T, Cox DA (1998) Manganese toxicity in plants. J Plant Nutr 21:353–386. https://doi.org/10.1080/01904169809365409
Esteves G, Bressanin LA, de Souza KRD, da Silva AB, Mantovani JR, Marques DM, Magalhães PC, Pasqual M, de Souza TC (2020) Do tailings from the Mariana, MG (Brazil), disaster affect the initial development of millet, maize, and sorghum? Environ Sci Pollut Res 27:38662–38673. https://doi.org/10.1007/s11356-020-10013-w
Foyer CH, Halliwell B (1976) The presence of glutathione and glutathione reductase in chloroplasts: A proposed role in ascorbic acid metabolism. Planta 133:21–25. https://doi.org/10.1007/BF00386001
Foyer CH, Noctor G (2005) Oxidant and antioxidant signalling in plants: a re-evaluation of the concept of oxidative stress in a physiological context. Plant, Cell Environ 28:1056–1071. https://doi.org/10.1111/j.1365-3040.2005.01327.x
Gajić G, Djurdjević L, Kostić O, Jarić S, Stevanović B, Mitrović M, Pavlović P (2020) Phytoremediation potential, photosynthetic and antioxidant response to arsenic-induced stress of Dactylis glomerata L. sown on fly ash deposits. Plants 9:657. https://doi.org/10.3390/plants9050657
Gamalero E, Lingua G, Berta G, Glick BR (2009) Beneficial role of plant growth promoting bacteria and arbuscular mycorrhizal fungi on plant responses to heavy metal stress. Can J Microbiol 55:501–514. https://doi.org/10.1139/W09-010
Genty B, Briantais JM, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta 990:87–92. https://doi.org/10.1016/S0304-4165(89)80016-9
Giannopolitis CN, Ries SK (1977) Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol 59:309–314. https://doi.org/10.1104/pp.59.2.309
Gomes MP, Garcia QS (2013) Reactive oxygen species and seed germination. Biologia 68:351–357. https://doi.org/10.2478/s11756-013-0161-y
Gomes MP, Carneiro MMLC, Garcia QS (2014a) Trace elements tolerance modulated by antioxidant system in plants. In: Ahmad P (ed) Oxidative Damage to Plants: antioxidant networks and signaling, 1st edn. Academic Press, San Diego, pp 523–540. https://doi.org/10.1016/B978-0-12-799963-0.00017-4
Gomes MP, Soares AM, Garcia QS (2014b) Phosphorous and sulfur nutrition modulate antioxidant defenses in Myracrodruom urundeuva plants exposed to arsenic. J Hazard Mater 276:97–104. https://doi.org/10.1016/j.jhazmat.2014.05.020
Hajiboland R (2014) Reactive oxygen species and photosynthesis. In: Ahmad P (ed) Oxidative Damage to Plants: antioxidant networks and signaling. Academic Press, San Diego, pp 1–63. https://doi.org/10.1016/B978-0-12-799963-0.00001-0
Hänsch R, Mendel RR (2009) Physiological functions of mineral micronutrients (cu, Zn, Mn, Fe, Ni, Mo, B, cl). Curr Opin Plant Biol 12:259–266. https://doi.org/10.1016/j.pbi.2009.05.006
Harper JE (1994) Nitrogen metabolism. In: Boote KJ, Bennett JM, Sinclair TR, Paulsen GM (eds) Physiology and determination of crop yield. ASA/CSSA/SSSA, Madison, pp 285–302. https://doi.org/10.2134/1994.physiologyanddetermination.c19
Hatje V, Pedreira RM, Rezende CE, Schettini CAF, Souza GC, Marin DC, Hackspacher PC (2017) The environmental impacts of one of the largest tailing dam failures worldwide. Sci Rep 7:10706. https://doi.org/10.1038/s41598-017-11143-x
Hawkesford M, Horst W, Kichey T, Lambers H, Schjoerring J, Møller IS, White P (2012) Functions of macronutrients. In: Marschner P (ed) Marschner’s mineral nutrition of higher plants. Academic Press, London, pp 135–189. https://doi.org/10.1016/B978-0-12-384905-2.00006-6
Hell R, Stephan UW (2003) Iron uptake, trafficking and homeostasis in plants. Planta 216:541–551. https://doi.org/10.1007/s00425-002-0920-4
Henzler T, Steudle E (2000) Transport and metabolic degradation of hydrogen peroxide in Chara corallina: model calculations and measurements with the pressure probe suggest transport of H2O2 across water channels. J Exp Bot 51:2053–2066. https://doi.org/10.1093/jexbot/51.353.2053
Hora AM, Dias CA, Guedes GR, Costa ASV, Júnior MJF (2012) Da exploração econômica da bacia hidrográfica do Rio Doce ao atual processo de degradação de seus recursos naturais. In: Guedes GR, Ojima R (eds) Território Mobilidade Populacional e Ambiente. Univale, Governador Valadares, pp 201–234
Kobayashi T, Nishizawa NK (2012) Iron uptake, translocation, and regulation in higher plants. Annu Rev Plant Biol 63:131–152. https://doi.org/10.1146/annurev-arplant-042811-105522
Li J, Meng B, Chai H, Yang X, Song W, Li S, Lu A, Zhang T, Sun W (2019) Arbuscular mycorrhizal fungi alleviate drought stress in C3 (Leymus chinensis) and C4 (Hemarthria altissima) grasses via altering antioxidant enzyme activities and photosynthesis. Front Plant Sci 10:499. https://doi.org/10.3389/fpls.2019.00499
Lombardi JA, Gonçalves M (2000) Composição florística de dois remanescentes de Mata Atlântica do sudeste de Minas Gerais, Brasil. Braz J Bot 23:255–282. https://doi.org/10.1590/S0100-84042000000300003
Malavolta E, Vitti GC, de Oliveira SA (1997) Evaluation of the nutritional state of plants: principles and applications (in Portuguese), 2nd edn. POTAFOS, Piracicaba
Marschner H (2012) Marschner’s mineral nutrition of higher plants, 3rd edn. Academic Press, London. https://doi.org/10.1016/C2009-0-63043-9
Matias SR, Pagano MC, Muzzi FC, Oliveira CA, Carneiro AA, Horta SN, Scotti MR (2009) effect of rhizobia, mycorrhizal fungi and phosphate-solubilizing microorganisms in the rhizosphere of native plants used to recover an iron ore area in Brazil. Eur J Soil Biol 45:259–266. https://doi.org/10.1016/j.ejsobi.2009.02.003
Matos LP, Andrade HM, Marinato CS, de Oliveira Prado IG, Coelho DG, Montoya SG, Kasuya MCM, de Oliveira JA (2020) Limitations to use of Cassia grandis L. in the revegetation of the areas impacted with mining tailings from Fundão Dam. Water Air Soil Pollut 231:1–9. https://doi.org/10.1007/s11270-020-04479-0
Morales M, Munné-Bosch S (2019) Malondialdehyde: facts and artifacts. Plant Physiol 180:1246–1250. https://doi.org/10.1104/pp.19.00405
Myers N, Mittermeier RA, Mittermeier CG, da Fonseca GA, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858. https://doi.org/10.1038/35002501
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Neuenkamp L, Prober SM, Price JN, Zobel M, Standish RJ (2019) Benefits of mycorrhizal inoculation to ecological restoration depend on plant functional type, restoration context and time. Fungal Ecol 40:140–149. https://doi.org/10.1016/j.funeco.2018.05.004
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Biol 49:249–279. https://doi.org/10.1146/annurev.arplant.49.1.249
Pandey A, Verma N, Singh S, Bashri G, Prasad SM (2021) Salicylic Acid (SA): Its interaction with different molecules in the stress tolerance signaling pathways. In: Aftab T, Yusuf M (eds) Jasmonates and Salicylates Signaling in Plants, 1st edn. Springer, Switzerland, pp 301–323. https://doi.org/10.1007/978-3-030-75805-9_15
Pereira EG, Oliva MA, Rosado-Souza L, Mendes GC, Colares DS, Stopato CH, Almeida AM (2013) Iron excess affects rice photosynthesis through stomatal and non-stomatal limitations. Plant Sci 201–202:81–92. https://doi.org/10.1016/j.plantsci.2012.12.003
Petrisor IG, Dobrota S, Komnitsas K, Lazar I, Kuperberg JM, Serban M (2004) Artificial inoculation—perspectives in tailings phytostabilization. Int J Phytoremediation 6:1–15. https://doi.org/10.1080/16226510490439918
Prates Júnior P, Moreira BC, Silva MCS, Diogo NV, Luz JMR, Jordão TC, Paiva HN, Kasuya MCM (2021) Mycorrhizal inoculation and phosphorus fertilization show contrasts on native species of the Brazilian Atlantic Forest and Cerrado. Revista Brasileira de Ciência do Solo, 45. https://doi.org/10.36783/18069657rbcs20210013
Pugh RE, Dick DG, Fredeen AL (2002) Heavy metal (Pb, Zn, Cd, Fe, and Cu) contents of plant foliage near the Anvil Range lead/zinc mine, Faro, Yukon Territory. Ecotoxicol Environ Safe 52:273–279. https://doi.org/10.1006/eesa.2002.2201
Punamiya P, Datta R, Sarkar D, Barber S, Patel M, Das P (2010) Symbiotic role of Glomus mosseae in phytoextraction of lead in vetiver grass [Chrysopogon zizanioides (L.)]. J Hazard Mater 177:465–474. https://doi.org/10.1016/j.jhazmat.2009.12.056
Rai R, Agrawal M, Agrawal SB (2016) Impact of heavy metals on physiological processes of plants: with special reference to photosynthetic system. In: Singh A, Prasad S, Singh R (eds) Plant responses to xenobiotics, 1st edn. Springer, Singapore, pp 127–140. https://doi.org/10.1007/978-981-10-2860-1_6
Rauret G, López-Sánchez JF (2001) New sediment and soil CRMs for extractable trace metal content. Int J Environ Anal Chem 79:81–95. https://doi.org/10.1080/03067310108034155
R Core Team (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, 4.0.2. http://www.r-project.org/index.html
Rios CO, Siqueira-Silva AI, Pereira EG (2021) How does drought affect native grasses’ photosynthesis on the revegetation of iron ore tailings? Environ Sci Pollut Res 28:14797–14811. https://doi.org/10.1007/s11356-020-11599-x
Roosta HR, Estaji A, Niknam F (2018) Effect of iron, zinc and manganese shortage-induced change on photosynthetic pigments, some osmoregulators and chlorophyll fluorescence parameters in lettuce. Photosynthetica 56:606–615. https://doi.org/10.1007/s11099-017-0696-1
Santolin CVA (2015) Quantificação e avaliação ambiental da contaminação por metais e arsênio em sedimentos da bacia do Rio Doce-MG. PhD Thesis Universidade Federal de Minas Gerais http://hdl.handle.net/1843/SFSA-A7EFLW
Segura FR, Nunes EA, Paniz FP, Paulelli ACC, Rodrigues GB, Braga GÚL, Pedreira Filho WR, Barbosa F Jr, Cerchiaro G, Silva FF, Batista BL (2016) Potential risks of the residue from Samarco’s mine dam burst (Bento Rodrigues, Brazil). Environ Pollut 218:813–825. https://doi.org/10.1016/j.envpol.2016.08.005
Sharma RK, Agrawal M (2005) Biological effects of heavy metals: an overview. J Environ Biol 26:301–313
Sharma I, Ahmad P (2014) Catalase: a versatile antioxidant in plants. Oxidative Damage to Plants: antioxidant networks and signaling, 1st edn. Academic Press, San Diego, pp 131–148. https://doi.org/10.1016/B978-0-12-799963-0.00004-6
Saggin Junior OJ, da Silva EMR (2006) Micorriza arbuscular–Papel, funcionamento e aplicação da simbiose. Miolo Biota 12:101–150. https://doi.org/10.1590/S0100-204X2006000100011
Sheoran V, Sheoran AS, Poonia P (2010) Soil reclamation of abandoned mine land by revegetation: a review. Int J Soil, Sediment Water 3:13
Silva JG, Gomes MP, Pereira EG, Bicalho EM, Garcia QS (2021) initial growth of peltophorum dubium is affected by nitrogen source and manganese concentration. J Soil Sci Plant Nutr 22:201–211. https://doi.org/10.1007/s42729-021-00644-4
Takahashi S, Murata N (2008) How do environmental stresses accelerate photoinhibition? Trends Plant Sci 13:178–182. https://doi.org/10.1016/j.tplants.2008.01.005
Tank N, Saraf M (2009) Enhancement of plant growth and decontamination of nickel-spiked soil using PGPR. J Basic Microbiol 49:195–204. https://doi.org/10.1002/jobm.200800090
Teixeira PC, Campos DVB, Saldanha MFC (2017) pH do solo. In Teixeira PC, Donagemma GK, Fontana A, Teixeira WG (eds), Manual de Métodos de Análise de Solo. Empresa Brasileira de Pesquisa Agropecuária (EMBRAPA), Brasília, pp 3–6. https://www.infoteca.cnptia.embrapa.br/infoteca/bitstream/doc/1087312/1/Pt2Cap1pHdosolo.pdf
Tian C, He X, Zhong Y, Chen J (2003) Effect of inoculation with ecto-and arbuscular mycorrhizae and rhizobium on the growth and nitrogen fixation by black locust, Robinia pseudoacacia. New for 25:125–131. https://doi.org/10.1023/A:1022675915627
Tuteja N (2007) Abscisic acid and abiotic stress signaling. Plant Signal Behav 2:135–138. https://doi.org/10.4161/psb.2.3.4156
Velikova V, Yordanov I, Edreva A (2000) Oxidative stress and some antioxidant systems in acid rain-treated bean plants: protective role of exogenous polyamines. Plant Sci 151:59–66. https://doi.org/10.1016/S0168-9452(99)00197-1
Vilela LA, Teixeira AF, Lourenço FM, Souza MD (2018) Symbiotic microorganisms enhance antioxidant defense in plants exposed to metal/metalloid-contaminated soils. In: Hasanuzzaman M, Nahar K, Fujita M (eds) Plants Under Metal and Metalloid Stress, 1st edn. Springer, Singapore, pp 337–366. https://doi.org/10.1007/978-981-13-2242-6_13
Wu FY, Ye ZH, Wu SC, Wong MH (2007) Metal accumulation and arbuscular mycorrhizal status in metallicolous and nonmetallicolous populations of Pteris vittata L. and Sedum alfredii Hance. Planta 226:1363–1378. https://doi.org/10.1007/s00425-007-0575-2
Zago VCP, das Dores NC, Watts BA (2019) Strategy for phytomanagement in an area affected by iron ore dam rupture: A study case in Minas Gerais State, Brazil. Environ Pollut 249:1029–1037. https://doi.org/10.1016/j.envpol.2019.03.060
Acknowledgements
We thank to owners of Paracatu Farm for conceding the planting area, in particular Pedro Fonseca de Souza and to Fundação Renova for their help in selection and fencing of the planting area. We thank to Instituto Espinhaço – Biodiversidade, Cultura e Desenvolvimento Socioambiental for their technical support during planting and maintenance of the experimental area and to Dr. Fábio Vieira and MSc Humberto Araújo Almeida for assistance with the planting and sample collect. The authors also thank to Prof. Dr. Maria Catarina Kasuya, Dr. Paulo Prates Junior, and Dr. Marliane de Cássia Soares da Silva for their help to obtain the inoculum and in plant inoculation procedures; to Paulo Antônio Pimentel, Tiago Tarcisio Silva, and Ana Kelly Oliveira for assistance with plant measurements and sample collection and to Gabriel Henrique Assunção for statistical assistance.
Funding
This work was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES)—Finance Code 001, and the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Brazil) through a strategic grant “REDES – Remediation of the Rio Doce Basin: potential of the aquatic and terrestrial biota” (grant number 88881.118082/2016–01). T.R.S. Santos and J.A.S. Santos received a PhD scholarship from CAPES, E.G. Pereira and Q.S. Garcia received a research productivity grant from CNPq.
Author information
Authors and Affiliations
Contributions
Santos, T.R.S.: methodology, data curation, formal analysis, investigation, writing—original draft, review & editing. Santos, J.A.S.: data curation, formal analysis, writing—review & editing. Pereira, E.G.: conceptualization, methodology, data curation, writing – review & editing. Garcia, Q.S.: conceptualization, funding acquisition, project administration, supervision, writing – review & editing.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Elena Maestri
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Santos, T.R.S., Santos, J.A.S., Pereira, E.G. et al. Revegetation of an area impacted by iron ore tailings: evaluating fertilization alternatives in native pioneer and secondary trees. Environ Sci Pollut Res 30, 3760–3773 (2023). https://doi.org/10.1007/s11356-022-22376-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-22376-3