Abstract
The beneficial effect of compost and compost tea on plant growth and protection is mainly associated with the microbial diversity and the presence of bacteria with plant growth–promoting effect. PGPR are considered as eco-friendly bio-fertilizers that may reduce the use of chemical pesticides and fertilizers. Three composts (AT, A10, and A30) were previously prepared from industrial wastes (olive mill wastewater, olive pomace, coffee ground, and phosphogypsum). In the present study, we isolated three bacterial strains from the compost teas. The phylogenetic identification of these bacterial strains (B.AT, B.A10, and B.A30) showed that they correspond to Serratia liquefaciens (B.AT and B.A10) and Achromobacter spanius (B.A30) species. A further characterization of the PGPR traits of these bacteria showed that they produce siderophore, exopolysaccharides, and IAA. Their effect on potato plant growth, yields, and tuber quality was performed under field culture conditions. Results showed that these strains can be characterized as PGPR, the best effect on potato plant growth was observed with Serratia liquefaciens (B.AT), the best yield and tuber quality was observed with Serratia liquefaciens (B.A10) while bacterial treatment with Achromobacter spanius (B.A30) is a Cd-tolerant PGPR.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Compost teas (CTs) are the liquid organic product obtained from mixing compost with tap water for a known ratio and incubation period (Morales-Corts et al. 2018). The CT quality is related to several factors, such as compost to water ratio, compost type, and aeration, which determine the development of specific groups of microorganisms (Ingham 1999; Mengesha et al. 2017; De Corato 2020). Furthermore, CTs microbial population and soluble nutrients play an important role in suppressing disease and promoting plant growth (De Corato 2020; Castano et al. 2011).
Plant growth–promoting rhizobacteria (PGPR) are the most abundant microorganisms in compost teas. They promote plant growth through different mechanisms that make plants more resistant to different biotic and abiotic stresses (Hamid et al. 2021). From these mechanisms, we can mention nitrogen fixation (Beijerinck 1901), phosphorus, and potassium solubilization by producing organic acids. The application of these PGPR as bio-fertilizers can reduce the use of agrochemicals (Setiawati and Mutmainnah 2016). Field trials in India have shown that the use of phosphate-solubilizing microorganisms (PSM) can increase yields of tomatoes, lettuce, potatoes, and rice (Rodrìguez and Fraga 1999).
Bacterial siderophores also play a significant role in enhancing plant growth and protection (Aznar and Dellagi 2015; Khan et al. 2016). Indeed, given their ability to sequester iron, they are able to deprive pathogenic fungi of this essential element since fungal siderophores have a lower affinity than PGPR (Kumar et al. 2017; Meena et al. 2017).
The production of phytohormones is considered as one of the most important mechanisms, underlying the beneficial effect of PGPRs on plant growth and nutrition. It has been reported that they stimulate plant growth and improve their stress response (Ma et al. 2011; Etesami et al. 2015; Ullah et al. 2015). The main plant hormone produced by PGPR is indole acetic acid (IAA) (Notununu et al. 2022).
The role of PGPRs to alleviate plant abiotic stress has been confirmed in many reviews (Yang et al. 2009; Sessitsch et al. 2013; Meena et al. 2017; Backer et al. 2018; Ilangumaran and smith 2017). Resistance of PGPRs to heavy metals is associated with several mechanisms such as biosorption, bioaccumulation, precipitation, complexation, and enzymatic transformation of heavy metals, thus reducing their toxicity towards the plant (Rajkumar et al. 2012; Ma et al. 2016). Many PGPRs play also an important role in improving plant-water relations, ion homeostasis, and photosynthetic efficiency in plants under salt stress (Sati et al. 2022).
Such beneficial microorganisms can act also as pathogen antagonists by several mechanisms such as competition (Hoitink and Changa 2004; Diánez et al. 2005), hydrolytic enzyme production (Goswami et al. 2016), secondary metabolites production like hydrogen cyanide (HCN) (Voisard et al. 1989), and aminocyclopropane-carboxylic acid (ACC) deaminase production which reduce the level of ethylene (Jacobson et al. 1994; Glick 2010). These mechanisms protect plants against biotic stress (Glick 2010; Ma et al. 2011; Rajkumar et al. 2012).
PGPRs can elicit molecular and biochemical defense responses within the plant (Lugtenberg and Kamilova 2009). Indeed, to prime plant resistance against pathogen attack, PGPR trigger the induced systemic resistance (ISR) and activate pathogenesis-related genes expression (Pieterse et al. 2014).
The inoculation of plants with PGPRs by coating seeds, roots, or tubers is a very old practice used to improve plant growth (Brown 1974; Gaskins et al. 1985), mainly in legumes and cereals (Sessitsch and Mitter 2015).
The aim of the present study is firstly the identification and the characterization of bacterial strains isolated from three different compost teas by determining their in vitro PGPR proprieties and secondly to study their effect on potato plant growth yield and tuber quality in field condition. These compost teas, previously prepared (Samet et al. 2018), showed a positive effect on potato plant growth in greenhouse condition and their microbial characterization showed that they contain several plant growth–promoting bacterial strains (Samet et al 2018, 2019).
Material and methods
Isolation of bacterial strains from compost teas
Three different Composts (AT, A10, and A30) were previously prepared by mixing olive mill waste water (OMW) olive pomace (P), coffee grounds (G), and phosphogypsum (PG) (Samet et al. 2018; 2019).
Three bacteria were isolated from AT, A10, and A30 compost teas. Isolation was performed by the multiple streak method, sample/inoculum is diluted by streaking it across the surface of the agar plate, isolated colonies were picked, and re-streaked on fresh agar plates. Nutrient agar medium containing 0.025 g/l triclosan was used. Triclosan is a broad-spectrum antibiotic inactive against several gram-negative PGPR genera such as Pseudomonas, Serratia, and Achromobacter (Welsch and Gillock 2011).
Phylogenetic identification of the bacterial strains
-
a)
Genomic DNA extraction
Genomic DNA extraction was performed from pure strains already isolated and cultivated on LB medium using the Wizard Genomic DNA Purification Kit (Promega) from 3 ml of culture, following the protocol provided by the manufacturer.
-
b)
Amplification of the 16S rDNA
The DNA sequence (1.5 kb) that encodes for 16S rRNA was amplified by PCR using, Taq DNA polymerase (Fermentas) and specific primers, FD1 (sequence: AGAGTTTGATCCTGGCTCAG; Weisburg et al. 1991), and 1492r (sequence: GGTTACCTTGTTACGACTT; Lane 1991). The amplification was carried out as follow: a denaturation phase (5 min at 94 °C) followed by 30 cycles of: denaturation (45 s at 94 °C), hybridization (45 s at 55 °C) and elongation (1 min 45 s at 72 °C). For each test, a negative control was added comprising all the components except DNA which was replaced by ultra-pure water.
-
c)
Sequencing and phylogenetic analysis
The sequencing of the purified PCR products was performed using the primers FD1 and 1492R in an automatic sequencer of the ABI PRISM 3100-Avant Genetic Analyzer type (Applied Biosystems) using the BigDye® Terminator v3.1 Cycle Sequencing kit. From the results, the phylogenetic analysis of the strains was carried out.
Similarity search was performed to find the closest sequences using NCBI’s Blast program. The phylogenetic trees were constructed according to the method of the nearest neighbor-joining method (Saitou and Nei 1987) using the software Mega 7 (Tamura et al. 2013). The robustness of the tree was tested by a bootstrap analysis obtained on 1000 replicas. The obtained sequences were deposited in GenBank under accession numbers ON210808 (B.AT), ON210809 (B.A10), and ON210810 (B.A30).
In vitro determination of PGPRs traits
-
1.
Growth of bacterial strains in the presence of salt or cadmium
Each bacterial strain was inoculated as a surface streak on solid LB medium in the presence of salt (NaCl) at different concentrations (5, 15, 35, 40, 50 g/L) or of cadmium (CdCl2) at 10, 40, 200, 300, and 400 mg/L and incubated at 30 °C for 24 h.
-
2.
Phosphate solubilization
The solubilization of the phosphate was qualitatively evaluated on Pikovskaya medium, according to the method described by Mehta and Nautiyal (2001). A colony of each bacterial strain was cultivated on the surface of the medium at 30 °C for 7 days. Phosphate solubilization is indicated by the formation of a clear zone around the colonies.
-
3.
Production of siderophores
The bacteria were cultured on the surface of the Chrome Azurol S (CAS) medium, for 1 to 3 days at 30° C. The production of siderophores is indicated by the appearance of a yellow-orange halo around the colonies (Husen 2003).
-
4.
Exopolysaccharids EPS production
The EPS production was determined quantitatively. The extraction was carried on by adding ethanol to the supernatant of cellular culture (3:1). After 48 h of precipitation at 4 °C, the precipitated EPS were washed three times, lyophilized, and weighted (Meneses et al. 2011).
-
5.
Production of indole 3 acetic acid (IAA)
The bacterial isolates were cultured in liquid LB medium supplemented with L-tryptophan (100 mg/l), with stirring at 200 rpm, at 30 °C for 24 h. The supernatants were obtained by centrifuging the bacterial cultures at 6000 rpm for 10 min. Colorimetric detection of IAA was performed according to the method of Bric et al. (1991). Two millimeters of the supernatant were mixed with 2 ml of Salkowski’s reagent and 2 drops of O-phosphoric acid. After incubation in the dark at room temperature for 30 min, the development of a pink color is indicative of IAA production (Tarnawski et al. 2006; Ahmad et al. 2008). The absorbance at 530 nm was measured to determine the intensity of the resulting coloration. The concentration of IAA (µg/ml) was determined by comparison to the standard curve, established from a standard range of IAA (Sigma-Aldrich) (0, 5, 10, 25, 50, and 100 μg/ml).
-
6.
Production of hydrogen cyanide (HCN)
The bacteria were cultured on solid LB medium supplemented with glycine (4.4 g/l). A Whatman paper saturated with alkaline picrate was placed in the lid of each box. The dishes were sealed with parafilm and incubated at 30° C for 4 days (Ahmad et al. 2008). The appearance of a red–orange color confirms the production of HCN.
-
7.
Nitrogen fixation
Atmospheric nitrogen fixation was tested on a solid nitrogen-free medium (NFM). This medium was inoculated with streaks from the bacterial culture and then incubated at 30 °C for 48 h. Any growth on this medium reflects the bacteria ability to fix nitrogen (Ding et al. 2005).
-
8.
Production of hydrolytic enzymes
The capacity of each strains to produce hydrolytic enzymes was qualitatively evaluated separately on solid medium containing the appropriate substrate for each hydrolytic enzyme activity: cellulase (Verma et al. 2007), glucanases, chitinases (Naik & Sakthivel 2006), pectinases (Mefteh et al. 2017), amylases (Saleem and Ebrahim 2014), laccases (Mefteh et al. 2017,) and proteases (Naik & Sakthivel 2006). The dishes were incubated at 30° C for 7 days. The presence of the enzyme activity appeared as halos around the colonies.
Plant growth parameters
Bacterial inoculum was prepared as follow: BAT (103 cfu/ml and 107 cfu/ml); BA10 (103 cfu/ml and 107 cfu/ml); BA30 (103 cfu/ml and 107 cfu/ml).
Thirteen potato seed tubers of the Spunta variety were soaked in the bacterial suspensions for 15 min before plantation in soil (without fertilizer). Control tubers were soaked in sterile distilled water. During the period of potato plants cultivation, the stem elongation, leaf, stem, and root fresh weight and leaf number were measured after 45 and 95 days of plantation.
-
1.
Determination of leaf chlorophyll and carotenoid content
The chlorophyll a and b and carotenoid contents were extracted according to the method of Arnon (1949). About 100 mg of fresh leaves were weighed and crushed in a mortar in the presence of 500 μl of pure acetone, then 1 ml of 80% acetone was added. The extract was centrifuged at 4 °C for 10 min at 12,000 rpm, and the supernatant was adjusted to a volume of 2 ml with 80% acetone. The absorbance at 663 and 645 nm was measured. The chlorophyll a and b contents were calculated in μg/g FW as follows:
$$\mathrm{Ca}\;(\mathrm{\mu g}/\mathrm g\;\mathrm{FW})\:=\:\mathrm{Chlorophyll}\;\mathrm a\:\:=\:(12,7\:\times\:\mathrm{OD}663\;\mathrm{nm})-(2,69\:\times\:\mathrm{OD}645\;\mathrm{nm})\:\times\:\mathrm V/\mathrm{FW}$$$$\mathrm{Cb}\;(\mathrm{\mu g}/\mathrm g\;\mathrm{FW})\:=\:\mathrm{Chlorophyll}\;\mathrm b\:=\:(22,9\:\times\:\mathrm{OD}645\;\mathrm{nm})-(4.68\:\times\:\mathrm{OD}663\;\mathrm{nm})\:\times\:\mathrm V/\mathrm{FW}$$where V = volume of adjustment acetone added to the supernatant; FW = fresh weight.
Carotenoid concentration was determined by spectrophotometry at 450 nm using a molar extinction coefficient of 2500 mol−1 L−1 cm−1 as reported by Morris et al. (2004). Results were expressed in terms of µg/g FW.
-
2.
Stomatal activity
The conductance of the stomata was measured with a Leaf Porometer (Model SC-1; DecaGon Devices).
-
3.
Chemical characterization of tubers
-
a.
Determination of dry weight (DW)
The percentage of dry weight was determined after drying the fresh sample at 105 °C for 48 h (AFNOR 1991).
-
b.
Determination of reducing sugars
The dry sample (1 g) was dispersed in distilled water (10 ml). The mixture was boiled for 10 min then cooled. The solution (1 ml) was defecated by adding 0.1 ml of potassium ferrocyanide (15%) and 0.2 ml of zinc acetate (30%). The mixture was centrifuged at 1000 rpm for 5 min. The reducing sugars recovered in the supernatant were then determined using dinitrosalycilic acid (DNS): A volume of 0.1 ml of supernatant diluted in 0.9 ml of distilled water was mixed with 3 ml of DNS and boiled for 10 min. The absorbance was then determined at 550 nm. The sugar content reducing agents was determined based on a standard range of glucose (0, 0.1, 0.2, 0.4, 0.8, 1.2, 1.6, 2 g/l); Miller (1959).
-
c.
Starch dosage
The starch content was determined based on the enzymatic method described by Khabou et al. (1996). The sample, dried at 50 °C and ground into a powder (1 g) was dispersed in 10 ml of distilled water. The mixture was supplemented with 10 μl of α-amylase (Termamyl 120L, Novozyme) and incubated for 2 h at 90 °C. After cooling, 20 μl of amyloglycosidase (AMG 300, Novozyme) were added and the solution was incubated for 6 h at 60 °C, then centrifuged at 1000 rpm for 5 min. The reducing sugars released in the supernatant were determined with dinitrosalycilic acid (DNS): 20 μl of the supernatant diluted in 980 μl of distilled water were treated with 3 ml of DNS, and boiled for 10 min. The absorbance was finally measured at 550 nm. A control was prepared by following the same steps but without adding enzymes. The glucose content of the sample was determined based on a standard glucose range (0.2, 0.4, 0.6, 0.8, 1, 1.2, 1.4, 1.6, 1.8, 2 g/l).
Statistical analyses
All data are expressed as the mean ± the standard deviation, from three independent biological replicates. Statistical analyzes of variance (ANOVA) were carried out with the statistical software SPSS (version 17.0, SPSS Inc., Chicago, IL, USA). Significant means were separated using the Duncan procedure. Significant differences between means were determined at P value < 0.05.
Results and discussion
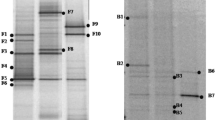
In a previous work, we prepared three composts by mixing olive mill waste water (OMW), olive pomace, coffee grounds, and phosphogypsum (0, 10, and 30%). Their derived compost teas showed a beneficial effect on potato plant growth and protection against Fusarium solani infection (Samet et al 2018). The characterization of these compost teas showed that they harbor plant growth–promoting bacterial strains such as Pseudomonas (Samet et al. 2019). In this study, we isolated one bacterial strain from each compost tea using a selective medium. The microscopic observation of the different isolated bacteria showed that they were motile gram-negative bacilli. These bacteria were also identified by 16 s rDNA sequencing and were affiliated with Serratia liquefaciens (B.AT and B.A10) and Achromobacter spanius (B.A30) species (Fig. 1). A characterization of the PGPR traits of these three bacteria was carried out and the study of their effect on potato plant growth and yields under field culture conditions were investigated.
Evaluation of PGPR traits of the different isolated strains
The biological properties of the isolated strains were analyzed in order to determine the various secondary metabolites and enzymatic activities related to a plant growth promotion they may exhibit.
-
1.
Nitrogen fixation
Nitrogen-free medium was used to evaluate nitrogen fixation ability. Results showed that B.AT and B.A10 strains can fix atmospheric nitrogen. Nitrogen-fixing bacteria associated with the rhizosphere are increasingly used on non-leguminous plants such as sugar beet, sugar cane, rice, corn, and wheat (Basu et al. 2021). Similar results were reported by Zelaya-Molina et al. (2016) who showed that the Serratia liquefaciens bacteria are able to fix atmospheric nitrogen.
-
2.
Phosphorus solubilization
The isolated bacteria were capable to dissolve tricalcium phosphate (Ca3 PO4) as reported by Zelaya-Molina et al., (2016) who showed that the bacteria Serratia liquefaciens was phosphate solubilizer. Likewise, Santos and Rigobelo (2021) have shown that the bacterium Achromobacter spanius was able to solubilize phosphate.
Successful applications of phosphate-solubilizing bacteria were carried out for many important crops such as wheat (Kumar et al. 2014), mung bean (Biswas et al. 2018), rapeseed (Valetti et al. 2018), tomatoes (Nassal et al. 2018), and potato (Aloo et al. 2020).
-
3.
IAA production
The production of IAA was noticed for all strains (B.AT, B.A10, and B.A30). The quantification of these compounds suggested that the B.A10 strain is the most productive (Table 1). Similarly, Aloo et al. (2020) showed that S. liquefaciens can produce IAA and Ahmad et al. (2008) showed the ability of Achromobacter spanius bacteria to produce IAA.
-
4.
Enzymatic and antifungal activities
Bacteria isolated from the different compost teas didn’t show any amylase, pectinase, glucanase, cellulase, laccase, or chitinase activity in vitro. This may explain the lack of antifungal activity against Botrytis cinerea and Fusarium solani (data not shown). However, both B.A10 and B.AT strains showed proteolytic activity. The higher activity noticed for B.AT strain with an inhibitory halo of approximately 4 cm in diameter, while that of the B.A10 strain was of 1.8 cm. Previous studies described such protease production for S. liquefaciens (Baglinière et al. 2017).
-
5.
HCN production
Although the production of hydrogen cyanide (HCN) measured in a number of PGPR bacteria, none of the strains studied here (B.AT, B.A10, and B.A30) showed such production. Cyanide hydrogen production can limit the growth of plant pathogens in the soil, but it can also affect the growth of beneficial microorganisms in the rhizosphere and therefore indirectly plant growth (Zdor 2015).
-
6.
Siderophores production
The B.AT and B.A10 bacteria of Serratia genus showed a distinct yellow-orange halo on the O-CAS medium, indicating the production of hydroxamate-type siderophores (Schwyn and Neilands 1987) while B.A30 strain did not give any response. These results are in agreement with those of Zelaya-Molina et al. (2016) who have shown that the S. liquefaciens bacteria are producers of siderophores Enterobacter is the main Enterobacteriaceae siderophore. Two of the four enterobacterin synthase genes (entE and entF) are annotated in the complete genome of S. liquefaciens ATCC 27,592 (Zelaya-Molina et al. 2016). In contrast, Abo et al. (2019) showed that Achromobacter spanius does not produce siderophores which corroborated our results.
-
7.
Exopolysaccharide production
The synthesis of exopolysaccharides was evaluated by precipitation with ethanol. Results showed that all the bacterial strains were able to produce exopolysaccharides (EPS) especially the B.AT strain (Table 1). Similarly, Abaid-ullah et al. (2015) showed that S. liquefaciens can produce EPS. The exopolysaccharide contains many negative charges that can efficiently sequester metal cations (van Hullebush et al. 2003).
-
8.
Assessment of the tolerance of bacteria to NaCl and Cd
The tolerance of the different strains (B.AT, B.A10, and B.A30) to Cd and NaCl was studied by culture on LB medium containing different concentrations of CdCl2 and NaCl. All isolated strains were capable to grow in media supplemented with NaCl (15 g/l) and CdCl2 (400 mg/l). B.A10 seemed to be more tolerant to NaCl (40 g/l) than the others, while the B.A30 exhibited higher tolerance to Cd (Table 2). In fact, bacteria of the Serratia genus (B.AT and B.A10) continue to grow in the presence of 35 g/l NaCl. In contrast, Achromobacter (B.A30) were capable to grow in a medium supplemented with 200 mg/l CdCl2. These results corroborate several studies which have shown that bacteria of the genus Serratia and Achromobacter are tolerant to salinity and heavy metals (Ma et al. 2011; Barra et al. 2016).
The presence of Achromobacter bacteria in A30 compost tea may be associated with the presence of phosphogypsum with high Cd concentration (15 ppm) as reported by Kammoun et al. (2017). The Cd content of the phosphogypsum seems to affect the microbial biodiversity in the A30 compost tea (Chen et al. 2015; Sarathambal et al. 2017). Cd tolerance and PGPR traits of these bacterial strain may improve plant growth under Cd stress (Sinha and Mukherjee 2008; Sarathambal et al. 2017; Pramanik et al. 2017).
The capacity of Achromobacter (B.A30) to solubilize phosphate and its tolerance to Cd may minimize Cd mobilization to plants by increasing soluble phosphorus in the soil, thereby promoting plant growth under stress conditions (Pramanik et al. 2018). In addition, the polysaccharide coating of this bacteria can provide numerous sites for adsorption and scavenging of toxic metals from the soil (Bruins et al. 2000; Rajkumar et al. 2010).
Effect of the isolated bacterial strains on the growth and yields of potato
Potato tubers of the Spunta variety were soaked for 15 min in B.AT, B.A10, or B. A30 bacterial suspensions at 103 CFU/ml or 107 CFU/ml. Control tubers were soaked in sterile distilled water. The treated and control tubers were then planted in the soil and irrigated regularly with tap water.
-
1.
Effect of bacterial inoculation on potato plant growth
Regular observations of potato plants from bacteria treated tubers showed the positive effect of the inoculation on plant vigor in comparison to the control plants (Fig. 2). Plant growth parameters (Fig. 3) showed that tuber inoculation with the strains at a concentration of 103 cfu/ml had no significant effect on the elongation of the main stem. However, the treatment with 107 cfu/ml with B.AT and B.A10 promoted the elongation of plant stem after 45 days of culture compared to the control plants. Inoculation with the B.A30 strain at the 107 concentration resulted in a better elongation after 95 days of culture in comparison to control (Vejan et al. 2016).
Significantly, higher leaf number and area were observed in the inoculated plants compared to the control ones (Fig. 4). Determination of plants fresh weight confirmed these results. Indeed, after 45 days of cultivation plants from tubers treated with 107 cfu/ml bacterial suspension displayed higher leaf fresh weight than the others. The roots were more developed after 45 days of culture in the plants from tubers inoculated by 103 cfu/ml of the different strains. The best root fresh weight was obtained with B.A30 treatment. Plants inoculated with B.AT showed higher leaf and stem fresh weight than control. Similar, stem FW of plants inoculated with B.A10 and B.A30 and of the control plants were noticed (Fig. 5).
These results confirm that all the tested strains isolated from compost tea had a beneficial effect on potato plant vigor in term of leaf size and number.
-
a)
Chlorophyll and carotenoid contents
The pre-treatment of the tubers with the bacterial strains resulted in a chlorophyll gain in the potato plant leaves in comparison to control ones (Fig. 6). Plants from tubers treated with B.AT strain showed the best chlorophyll content. Such increase of chlorophyll content in plants obtained from the treated tubers suggests a more important photosynthetic activity than that of the control plants (Fig. 6).
The evaluation of the carotenoid content in leaves showed higher level in plants inoculated by B.AT strain in comparison to the others. B.A10 and B.A30 strains seem to have a less marked effect since the carotenoids contents in the leaves increased slightly after 95 days of cultivation (Fig. 7).
All these results confirm that Serratia and Achromobacter can be used as PGPRs to improve plant growth as reported by other reports (Zhang et al. 2002; Belimov et al. 2009; Jha and Kumar 2009; Zahir et al. 2011). Moreover, El-Esawi et al. (2018) showed that Serratia liquefaciens could enhance maize growth under normal and high salinity conditions. Aloo et al. (2020) also showed that S. liquefaciens isolated from potato rhizosphere is a PGPR which can be exploited as biofertilizer. Likewise, Achromobacter spanius inoculation was able to increase the growth of sugarcane plants under greenhouse conditions (Santos and Rigobelo 2021).
A reduction of stomatal activity was observed in leaves of plants coming from B.AT-, B.A10-, and B.A30-treated tubers, after 45 days of culture in comparison to control plants (Fig. 8). Stomatal closure following inoculation with PGPRs represents a structural barrier that may delay disease progression (Rudrappa et al. 2008; Pieterse et al. 2014). After 95 days of culture, at the end of the vegetative potato cycle, an increase of stomatal activity was observed in B.A10- and B.A30-treated plants (107 cfu/ml) (Fig. 8).
-
b)
Effect of bacterial inoculation on plant nutrient absorption
Analysis of the mineral content in the leaves and roots of plants after 45 days of cultivation showed a significant increase in the K, Mg, and Zn contents in leaves of plants from tubers treated with B.AT (107) (Table 3). The P accumulation increased significantly in leaves of all plants treated by bacterial strains. An increase in Fe uptake was also observed in leaves of plants treated by B.A10 (107 cfu/ml) and B.A30 (103 and 107 cfu/ml). Leaves of plants treated with B.A30 (103 cfu/ml) showed a significant increase of Zn accumulation in addition to the improved uptake of Fe for 103 and 107 cfu/ml bacterial treatment in comparison to control. Roots from B.AT- and B.A10-treated plants exhibited higher K, Mg, and Ca accumulation than control. The Fe content increase significantly in roots of plants treated by B.A10 (103 and 107 cfu/ml) and B.A30 (103 cfu/ml), while Zn content increased in roots of plants treated by B.AT (103 cfu/ml), B.A10 (103 and 107 cfu/ml), and B.A30 (103 cfu/ml) (Table 3).
The nutrient uptake increase can be attributed to the production of growth regulators by the bacteria, which stimulate root development and allows a better uptake of water and plant nutrients (Lifshitz et al. 1987; Höflich et al. 1997). Even though a decrease of Mg, Zn, and Ca contents was observed in leaves of plants treated by 103 cfu/ml B.A10 and P contents in roots of plants treated with B.A30 (103 cfu/ml) (Table 3), these values remain within the range of concentrations found by Walworth and Muniz (1993) who studied the variations in the concentrations of mineral elements in potato plant tissues during the vegetative growth period.
El-Esawi et al. (2018) showed that maize plants inoculated with Serratia liquefaciens KM4 exhibited significant improvements in K + and Ca2 + contents as compared with non-inoculated plants. Several studies reported that the inoculation with PGPRs increases the bioavailability of nutrients for several crops such as chickpea (Elkoca et al. 2008), barley (Cakmakc et al. 2007), tomato (Adesemoye et al. 2010), strawberries (Günes et al. 2009), and broccoli (Yildirim et al. 2011). Other reports suggested that improved nutrient uptake by PGPRs is attributed to the increased water uptake by plants (Dey et al. 2004). Other studies suggested that increased nutrient uptake by plants is related to increased root area (Adesemoye et al. 2008; Yildirim et al. 2011; Ndakidemi et al. 2011).
-
c)
Bacterial concentration in plants tissues
The determination of bacterial concentration in leaves, stems and roots showed that plants treated with the B.AT, B.A10, and B.A30 bacteria (103 and 107 cfu/ml) exhibited high bacterial concentration in roots. Similarly, high bacterial concentrations were measured in leaves and stems of plants inoculated with B.AT. B.A30 bacteria (107 cfu/ml) led to bacterial accumulation in leaves and stems. For plants inoculated with B.A10 strain (107 cfu/ml), high bacterial concentration was detected in the stems (Table 4). These results are in agreement with those of Devi et al. (2016) and Mukherjee et al. (2017) who showed that Serratia and Achromobacter bacteria are endophytes that colonize the plant tissues without causing symptoms while improving their growth.
-
2.
Effect of inoculation on tuber yield and quality
Plants yields were determined in term of weight and number of tubers per plant. Tuber size were also measured (Table 5). The results showed that all the inoculations with B.AT, B.A10, and B.A30 increased significantly the plant yield in term of number of tuber/plant by 20.81%, 38.39%, and 30.6% respectively. A significant increase in tuber size with 53.488% and 46.511% was also observed in B.A10- and B.A30-treated plants, respectively. (Table 5, Fig. 9). These results are in agreement with those of El-Esawi et al. (2018) who showed that maize seed inoculation by Serratia liquefaciens KM4 significantly improved maize plant growth and yield biomass. Abdel-Rahman et al. (2017) also showed that Achromobacter sp. bacteria increased the yield of inoculated tomato plants.
The evaluation of the bacterial treatment on tuber quality was carried out by measuring dry matter, starch, reducing sugars, and minerals content. The results (Table 6) showed that with B.AT (107 cfu/ml) and B.A10 (103 cfu/ml) increased tuber DW and starch contents and reduced reducing sugars levels compared to the tubers of control plants. In addition, the P, K, and Fe contents increased in almost all tubers from treated plants.
Tubers of plants treated with B.A30 showed a significant increase in the Ca and Mg contents. Similar Zn contents were measured in the tubers obtained from treated or control plants except for those obtained from plants treated with the B.A10 (107 cfu/ml) where the Zn contents decreased significantly.
These results are in agreement with several studies which have shown that inoculation with PGPRs increased the nutritional quality of plant products such as broccoli, tomato, and beans (Yildirim et al 2011; Sirichaiwetchakul et al. 2011).
Conclusion
This study showed that the bacterial strains isolated from AT, A10, and A30 compost teas can be characterized as PGPR. Serratia liquefaciens strains isolated from AT and A10 compost teas exhibited higher PGPR traits (nitrogen fixation, solubilization of P, production of AIA, siderophores, and exopolysaccharides production) than Achromobacter spanius (B.A30). However, this latter species showed higher Cd tolerance than S. liquefaciens allowing using it in contaminated soils. Field trial results showed that all the isolated strains improved potato plant growth by increasing stem elongation, leaves (at 107 CFU/ml) and roots (at103 CFU/ml) fresh weight, and chlorophyll and carotenoids content. The best plant growth parameters were observed in S. liquefaciens (B.AT)-inoculated plants. These results can be related to by the best nutrient (K and P) uptake observed in these plants. The CFU counting in plant tissues show also that S. liquefaciens (B.AT) can colonize all plant tissues. Moreover, the use of treatment 107 cfu/ml S. liquefaciens (B.A10) as inoculum seems to be more efficient in increasing tuber yield and quality than the other treatments. Therefore, we can conclude that S. liquefaciens (BAT and BA10) acts as PGPR for potato plants under standard conditions and it can be used as biofertilizer.
References
Abaid-Ullah M, Hassan MN, Jamil M, Brader G, Shah MKN, Sessitsch A et al (2015) Plant growth promoting rhizobacteria: an alternate way to improve yield and quality of wheat (Triticum aestivum). Int J Agric Biol 17:51–60
Abdel-Rahman HM, Salem AA, Moustafa MMA, El-Garhy HAS (2017) A novice Achromobacter sp. EMCC1936 strain acts as a plant-growth-promoting agent. Acta Physiol Plant 39:61. https://doi.org/10.1007/s11738-017-2360-6
Abo É, Laslo E, Szentes S, Lányi S, Mara G (2019) Plant growth-promoting bacteria: strategies to improve wheat growth and development under sustainable agriculture, plant growth promoting rhizobacteria for agricultural sustainability. Springer, 1–17
Adesemoye AO, Torbert HA, Kloepper JW (2008) Enhanced plant nutrient use efficiency with PGPR and AMF in an integrated nutrient management system. Can J Microbiol 54:876–886
Adesemoye AO, Torbert HA, Kloepper JW (2010) Increased plant uptake of nitrogen from 15N-depleted fertilizer using plant growth promoting rhizobacteria. Appl Soil Ecol 46:54–58
AFNOR (1991) Matières fertilisantes et supports de culture, Recueil des normes Françaises. AFNOR, Paris, p 713
Ahmad F, Ahmad I, Khan MS (2008) Screening of free-living rhizospheric bacteria for their multiple plant growth promoting activities. Microbiol Res 163:173–181
Aloo BN, Mbega ER, Makumba BA, Hertel R, Danel R (2020) Molecular identification and in vitro plant growth-promoting activities of culturable Potato (Solanum tuberosum L.) rhizobacteria in Tanzania. Potato Res. https://doi.org/10.1007/s11540-020-09465-x.
Arnon DL (1949) A copper enzyme is isolated chloroplast polyphenol oxidase in Beta vulgaries. Plant Physiol 24:1–15
Aznar A, Dellagi A (2015) New insights into the role of siderophores as triggers of plant immunity: what can we learn from animals? J Exp Bot 66:3001–3010
Backer R, Rokem JS, Ilangumaran G, Lamont J, Praslickova D, Ricci E (2018) Plant growth-promoting rhizobacteria: context, mechanisms of action, and roadmap to commercialization of biostimulants for sustainable agriculture. Front Plant Sci 9:1473
Baglinière F, Jardin J, Gaucheron F, de Carvalho AF, Vanetti MCD (2017) Proteolysis of casein micelles by heat-stable protease secreted by Serratia liquefaciens leads to the destabilisation of UHT milk during its storage. Int Dairy J 68:38–45
Barra PJ, Inostroza NG, Acuña JJ, Mora ML, Crowley DE, Jorquera MA (2016) Formulation of bacterial consortia from avocado (Persea americana Mill.) and their effect on growth, biomass and superoxide dismutase activity of wheat seedlings under salt stress. Appl Soil Ecol 102:8
Basu A, Prasad P, Das SN, Kalam S, Sayyed RZ, Reddy MS, El Enshasy H (2021) Plant growth promoting rhizobacteria (PGPR) as green bioinoculants: recent developments, constraints, and prospects. Sustainability 13:1140
Beijerinck MW (1901) Ueber Oligonitophile Mikroben, Zentralblattfiir Bakteriologie, Parasitenkunde, Infektionskrankheiten and Hygiene. Abteilung II 7:561–582
Belimov AA, Dodd IC, Hontzeas N, Theobald JC, Safronova VI, Davies WJ (2009) Rhizosphere bacteria containing 1-aminocyclopropane-1-carboxylate deaminase increase yield of plants grown in drying soil via both local and systemic hormone signalling. New Phytol 181:413–423
Bhardwaj D, Ansari MW, Sahoo RK, Tuteja N (2014) Biofertilizers function as key player in sustainable agriculture by improving soil fertility, plant tolerance and crop productivity. Microb Cell Fact 13:1–10
Biswas JK, Banerjee A, Rai M, Naidu R, Biswas B, Vilthanaje M, Dash MC, Sarkar SK, Meers E (2018) Potential application of selected metal resistant phosphate solubilizing bacteria isolated from the gut of earthworm (Metaphire posthuma) in plant growth promotion. Geoderma 330:117–124
Bric JM, Bostock RM, Silverstone SE (1991) Rapid in situ assay for indoleacetic acid production by bacteria immobilized on nitrocellulose membrane. Appl Environ Microbiol 57:535–538
Brown ME (1974) Seed and root bacterization. Annu Rev Phytopatol 12:181–197
Bruins MR, Kapil S, Oehme FW (2000) Microbial resistance to metals in the environment. Ecotoxicol Environ Saf 45:198–207
Cakmakc R, Donmez MF, Erdogan U (2007) The effect of plant growth promoting rhizobacteria on barley seedling growth, nutrient uptake, some soil properties, and bacterial counts. Turk J Agric for 31(Suppl 3):189–199
Cardinale M, Ratering S, Suarez C, Zapata Montoya AM, Geissler-Plaum R, Schnell S (2015) Paradox of plant growth promotion potential of rhizobacteria and their actual promotion effect on growth of barley (Hordeum vulgare L.) under salt stress. Microbiol Res 181:22–32. https://doi.org/10.1016/j.micres.2015.08.002
Castano R, Borrero C, Aviles M (2011) Organic matter fractions by SP-MAS 13C NMR and microbial communities involved in the suppression of Fusarium wilt in organic growth media. Biol Control 58:286–293
Chen M, Xu P, Zeng G, Yang C, Huang D, Zhang J (2015) Bioremediation of soils contaminated with polycyclic aromatic hydrocarbons, petroleum, pesticides, chlorophenols and heavy metals by composting: applications, microbes and future research needs. Biotechnol Adv 33:745–755
De Corato U (2020) Agricultural waste recycling in horticultural intensive farming systems by on-farm composting and compost based tea application improves soil quality and plant health: a review under the perspective of a circular economy. Sci Total Environ 738:139840
De Vleesschauwer D, Höfte M (2009) Rhizobacteria-induced systemic resistance. Adv Bot Res 51:223–281
Devi KA, Pandey P, Sharma GD (2016) Plant growth-promoting endophyte serratia marcescens AL2-16 enhances the growth of achyranthes aspera L., a medicinal plant. HAYATI J Biosci 23:173–180. https://doi.org/10.1016/j.hjb.2016.12.006
Dey R, Pal K, Bhatt D, Chauhan S (2004) Growth promotion and yield enhancement of peanut (Arachis hypogaea L.) by application of plant growth promoting rhizobacteria. Microbiol Res 159:371–394
Diánez F, Santos M, Tello JC (2005) Suppresion of soilborne pathogens by compost, suppresive effects of grape marc compost on phytopathogenics oomycetes. Acta Hort 697:441–460
Dimkpa CO, Merten D, Svatoš A, Büchel G, Kothe E (2009) Siderophores mediate reduced and increased uptake of cadmium by Streptomyces tendae F4 and sunflower (Helianthus annuus), respectively. J Appl Microbiol 107:1687–1696
Ding Y, Wang J, Liu Y, Chen S (2005) Isolation and identification of nitrogen fixing bacilli from plant rhizospheres in Beijing region. J Appl Microbiol 99:1271–2128
Dutta S, Podile AR (2010) Plant growth promoting rhizobacteria (PGPR): the bugs to debug the root zone. Crit Rev Microbiol 36:232–244. https://doi.org/10.3109/10408411003766806
El-Esawi MA, Alaraidh IA, Alsahli AA, Alzahrani SM, Ali HM, Alayafi AA, Ahmad M (2018) Serratia liquefaciens KM4 improves salt stress tolerance in maize by regulating redox potential, ion homeostasis, leaf gas exchange and stress-related gene expression. Int J Mol Sci 19:3310
Elkoca E, Kantar F, Sahin F (2008) Influence of nitrogen fixing and phosphorus solubilising bacteria on the nodulation, plant growth and yield of chickpea. J Plant Nutr 31:157–171
Etesami H, Alikhani HA, Hosseini HM (2015) Indole-3-acetic acid and 1-aminocyclopropane-1-carboxylate deaminase: bacterial traits required in rhizosphere, rhizoplane and/or endophytic competence by beneficial bacteria. Bacterial metabolites in sustainable agroecosystem. Springer 183–258.
Gaskins MH, Albrecht SL, Hubbell DH (1985) Rhizosphere bacteria and their use to increase plant productivity: a review. Agric Ecosyst Environ 12:99–116
Glick BR (2010) Using soil bacteria to facilitate phytoremediation. Biotechnol Adv 28:367–374
Goswami D, Thakker JN, Dhandhukia PC (2016) Portraying mechanics of plant growth promoting rhizobacteria (PGPR): a review. Cogent food Agric. https://doi.org/10.1080/23311932.2015.1127500
Günes A, Atatoglu N, Turan M, Esitken A, Ketterings QM (2009) Effects of phosphate solubilizing microorganisms on strawberry yield and nutrient concentrations. J Plant Nutr Soil Sci 172:385–392. Yildirim et al. 2011
Guo LJ, Zhao B, An Q, Tian M (2016) Characteristics of a novel aerobic denitrifying bacterium, Enterobacter cloacae strain HNR. Appl Biochem Biotechnol 178:947–959
Hamid S, Ahmad I, Akhtar MJ, Iqbal MN, Shakir M, Tahir M, Rasool A et al (2021) Bacillus subtilis Y16 and biogas slurry enhanced potassium to sodium ratio and physiology of sunflower (Helianthus annuus L.) to mitigate salt stress. Environ Sci Pollut Res Int. https://doi.org/10.1007/s11356-021-14344-0
Höflich G, Tappe E, Kuhn G, Wiehe W (1997) EinfluX associativer Rhizosph.arenbakterien auf die N.ahrstoffaufnahme und den Ertrag von Mais. Archiv Fuer Acker- Und Pflanzenbau Und Bodenkunde 41:323–333
Hoitink HAJ, Changa CM (2004) Managing soil-borne pathogens. Acta Hortic 635:87–92
Husen E (2003) Screening of soil bacteria for plant growth promotion activities in vitro. Indones J Agric Sci 4:27–31
Ilangumaran G, Smith DL (2017) Plant growth promoting rhizobacteria in amelioration of salinity stress: a systems biology perspective. Front Plant Sci 8:1768
Ingham ER (1999) What is compost tea? Part 1. Biocycle 40:74–75
Jacobson CB, Pasternak JJ, Glick BR (1994) Partial purification and characterization of 1-aminocyclopropane-1-carboxylate deaminase from the plant growth promoting rhizobacterium Pseudomonas putida GR12-2. Can J Microbiol 40:1019–1025
Jha P, Kumar A (2009) Characterization of novel plant growth promoting endophytic bacterium Achromobacter xylosoxidans from wheat plant. Microb Ecol 58:179–188
Kammoun M, Ghorbel I, Charfeddine S, Kamoun L, Gargouri-Bouzid R, Nouri-Ellouz O (2017) The positive effect of phosphogypsumsupplemented composts on potato plant growth in the field and tuber yield. J Environ Manag 20:475–483
Khabou W, Trigui A, Ghorbel R, Bejar S (1996) L’amidon dans les rameaux d’olivier (Oleaeuropaea) Cv.“Chemlali de sfax” Etude comparative de deux methodes d ’hydrolyse. Olivea 61:57e61.
Khan H, Parmar N, Kahlon RS (2016) Pseudomonas-plant interactions I: plant growth promotion and defense-mediated mechanisms. In: Kahlon RS (ed) Pseudomonas: molecular and applied biology. Springer International Publishing, Switzerland
Kristin A, Miranda H (2013) The root microbiota—a fingerprint in the soil? Plant Soil 370:671–686. https://doi.org/10.1007/s11104-013-1647-7
Kumar A, Maurya BR, Raghuwanshi R (2014) Isolation and characterization of PGPR and their effect on growth, yield and nutrient content in wheat (Triticum aestivum L.). Biocatal Agric Biotechnol 3:121–128
Kumar V, Menon S, Agarwal H, Gopalakrishnan D (2017) Characterization and optimization of bacterium isolated from soil samples for the production of siderophores. Resource-Efficient Technologies 3(4):434–439
Lane DJ (1991) 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic acid techniques in bacterial systematics. John Wiley and Sons, Chichester, pp 115–175
Lifshitz R, Kloepper JM, Kozlowski M, Simonson C, Carlso J, Tipping EM, Zaleska I (1987) Growth promotion of canola (rapeseed) seedlings by a strain of Pseudomonas putida under gnotobiotic conditions. Can J Microbiol 33(5):390–395
Lugtenberg B, Kamilova F (2009) Plant-growth-promoting rhizobacteria. Annu Rev Microbiol 63:541–556
Ma Y, Prasad MNV, Rajkumar M, Freitas H (2011) Plant growth promoting rhizobacteria and endophytes accelerate phytoremediation of metalliferous soils. Biotechnol Adv 29:248–258
Ma Y, Rajkumar M, Zhang C, Freitas H (2016) Inoculation of Brassica oxyrrhina with plant growthpromoting bacteria for the improvement of heavy metal phytoremediation under drought conditions. J Hazard Mater 320:36–44
Martínez-Viveros O, Jorquera MA, Crowley DE, Gajardo G, Mora ML (2010) Mechanisms and practical considerations involved in plant growth promotion by Rhizobacteria. J Soil Sci Plant Nutr 10(3):293–319
Meena VS, Mishra PK, Bisht JK, Pattanayak A (2017) Agriculturally important microbes for sustainable agriculture, Volume 2: Applications in crop production and protection. Springer, Singapore. https://doi.org/10.1007/978- 981-10-5343-6
Mefteh FB, Daoud A, Chenari BA, Alenezi FN, Luptakova L, Rateb ME, Kadri A, Gharsallah N, Belbahri L (2017) Fungal root microbiome from healthy and brittle leaf diseased date palm trees (Phoenix dactylifera L.) reveals a hidden untapped arsenal of antibacterial and broad spectrum antifungal secondary metabolites. Front Microbiol 8:307
Mehta S, Nautiyal CS (2001) An efficient method for qualitative screening of phosphate-solubilizing bacteria. Curr Microbiol 43:51–56
Meneses CHSG, Rouws LFM, Simões-Araújo JL, Vidal MS, Baldani JI (2011) Exopolysaccharide production is required for biofilm formation and plant colonization by the nitrogen-fixing endophyte Gluconacetobacter diazotrophicus. Mol Plant-Microbe Interact 24:1448–1458. https://doi.org/10.1094/MPMI-05-11-0127
Mengesha WK, Powel SM, Evans KJ, Barry KM (2017) Diverse microbial communities in non-aerated compost teas suppress bacterial wilt. World J Microbiol Biotechnol 33:49
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination ofreducing sugar. Anal Chem 31:426–8
Mody B, Bindra M, Modi V (1989). Arch Microbial 153:3842
Morales-Corts MR, Pérez-Sánchez R, Gómez-Sánchez MA (2018) Efficiency of garden waste compost teas on tomato growth and its suppressiveness against soilborne pathogens. Sci Agric 75:400–409
Morris WL, Ducreux L, Griffiths DW, Stewart D, Davies HV, Taylor MA (2004) Carotenogenesis during tuber development and storage in potato. J Exp Bot 55:975–982
Mukherjee A, Bhattacharjee P, Das R, Pal A, Paul AK (2017) Endophytic bacteria with plant growth promotingabilities from Ophioglossum reticulatum L. AIMS Microbiol 3(3):596
Naik PR, Sakthivel N (2006) Functional characterization of a novel hydrocarbonoclastic Pseudomonas sp. strain PUP6 with plant-growth-promoting traits and antifungal potential. Res Microbiol 157:538–546
Nassal D, Spohn M, Eltbany N, Jacquiod S, Smalla K, Marhan S, Kandeler E (2018) Effects of phosphorus-mobilizing bacteria on tomato growth and soil microbial activity. Plant Soil 427:17–37
Ndakidemi PA, Bambara S, Makoi JHJR (2011) Micronutrient uptake in common bean (Phaseolus vulgaris L.) as affected by rhizobium inoculation, and the supply of molybdenum and lime. Plant Omics J 4(1):40–52
Notununu I, Moleleki L, Roopnarain A, Adeleke R (2022) Effects of plant growth-promoting rhizobacteria on the molecular responses of maize under drought and heat stresses: a review. Pedosphere 32(1):90–106
Pieterse CMJ, Zamioudis C, Berendsen RL, Weller DM, van Wees SCM, Bakker PA (2014) Induced systemic resistance by beneficial microbes. Annu Rev Phytopathol 52:347–375
Pramanik K, Mitra S, Sarkar A, Soren T, Maiti TK (2017) Characterization of cadmium resistant Klebsiella pneumoniae MCC 3091 promoted rice seedling growth by alleviating phytotoxicity of cadmium. Environ Sci Pollut Res 24:24419–24437
Pramanik K, Mitra S, Sarkar A, Maiti TK (2018) Alleviation of phytotoxic effects of cadmium on rice seedlings by cadmium resistant PGPR strain Enterobacter aerogenes MCC 3092. J Hazard Mater 351:317–329
Rajkumar M, Ae N, Prasad MNV, Freitas H (2010) Potential of siderophore-producing bacteria for improving heavy metal phytoextraction. Trends Biotechnol 28:142–149
Rajkumar M, Sandhya S, Prasad MNV, Freitas H (2012) Perspectives of plant associated microbes in heavy metal phytoremediation. Biotechnol Adv 30:1562–1574
Rodrìguez H, Fraga R (1999) Phosphate solubilizing bacteria and their role in plant growth promotion. Biotechnol Adv 17:319–339
Rudrappa T, Biedrzycki ML, Bais HP (2008) Causes and consequences of plant-associated biofilms. FEMS Microbiol Ecol 64:153–66
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Saleem A, Ebrahim MKH (2014) Production of amylase by fungi isolated from legume seeds collected in Almadinah Almunawwarah, Saudi Arabia. J Taibah Univ Sci 8:90–97
Samet M, Charfeddine M, Kamoun L, Nouri-Ellouze O, Gargouri-Bouzid R (2018) Effect of compost tea containing phosphogypsum on potato plant growth and protection against Fusarium solani infection. Environ Sci Pollut Res 25(19):18921–18937
Samet M, Karray F, Mhiri N, Kamoun L, Sayadi S, Gargouri-Bouzid R (2019) Effect of phosphogypsum addition in the composting process on the physico-chemical proprieties and the microbial diversity of the resulting compost tea. Environ Sci Pollut Res 26(21):21404–21415
Santos RMD, Rigobelo EC (2021) Growth-promoting potential of rhizobacteria isolated from sugarcane. Front Sustain Food Syst 5:105
Sarathambal C, Khankhane PJ, Gharde Y, Kumar B, Varun M, Arun S (2017) The effect of plant growth promoting rhizobacteria on the growth, physiology, and Cd uptake of Arundo donax L. Int J Phytoremed 19:360–370
Sati D, Pande V, Pandey SC et al (2022) Recent advances in PGPR and molecular mechanisms involved in drought stress resistance. J Soil Sci Plant Nutr. https://doi.org/10.1007/s42729-021-00724-5
Schwyn B, Neilands JB (1987) Universal chemical assay for the detection and determination of siderophores. Anal Biochem 160:47–56. https://doi.org/10.1016/0003-2697(87)90612-9
Sessitsch A, Mitter B (2015) 21st century agriculture: integration of plant microbiomes for improved crop production and food security. Microb Biotechnol 8:32–33. https://doi.org/10.1111/1751-7915.12180
Sessitsch A, Kuffner M, Kidd P, Vangronsveld J, Wenzel WW, Fallmann K, Puschenreiter M (2013) The role of plant-associated bacteria in the mobilization and phytoextraction of trace elements in contaminated soils. Soil Biol Biochem 60:182–194
Setiawati T, Mutmainnah L (2016) Solubilization of potassium containing mineral by microorganisms from sugar cane rhizosphere. Agric Sci Procedia 9:108–117
Sinha S, Mukherjee SK (2008) Cadmium-induced siderophore production by a high Cd-resistant bacterial strain relieved Cd toxicity in plants through root colonization. Curr Microbiol 56:55–60
Sirichaiwetchakul S, Sirithorn P, Manakasem Y (2011) Arbuscular mycorrhizal fungi on growth, fruit yield and quality of cherry tomato under glasshouse conditions. Suranaree J Sci Tech 18:273–280
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6:molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Tarnawski S, Hamelin J, Jossi M, Aragno M, Fromin N (2006) Phenotypic structure of Pseudomonas populations is altered under elevated pCO2 in the rhizosphere of perennial grasses. Soil Biol Biochem 38:1193–1201
Ullah A, Heng S, Munis MFH, Fahad S, Yang X (2015) Phytoremediation of heavy metal assisted by plant growth promoting (PGP) bacteria: a review. Environ Exp Bot 117:28–40
Ulloa-Ogaz AL, Muñoz-Castellanos LN, Nevárez-Moorillón GV (2015) Biocontrol of phytopathogens: antibiotic production as mechanism of control. In: Mendez-Vilas A (ed) The battle against microbial pathogens: basic science, technological advances and educational programs. Formatex Research Center, Spain, pp 305–309
Valetti L, Iriarte L, Farba A (2018) Growth promotion of rapeseed (Brassica napus) associated with the inoculation of phosphate solubilizing bacteria. Appl Soil Ecol 132:1–10
van Hullebush ED, Zandvoort MH, Lens PNL (2003) Metal immobilization by biofilms: mechanisms and analytical tools. Environ Sci Technol 2:9–33
Vejan P, Abdullah R, Khadiran T, Ismail S, Nasrulhaq Boyce A (2016) Role of plant growth promoting rhizobacteria in agricultural sustainability—a review. Molecules: 21–573.
Verma M, Satinder K, Brar RD, Tyagi RY, Surampalli J, Valero R (2007) Antagonistic fungi, Trichoderma spp. panoply of biological control. Biochem Eng J 37:1–20
Voisard C, Keel C, Haas D, Defago H (1989) Cyanide production by Pseudomonas fluorescens helps suppress black root of tobacco under gnotobiotic conditions. EMBO J 8:351–358
Walworth JL, Muniz JE (1993) A compendium of tissue nutrient concentration for field-grown potatoes. Am Potato J 70:570–597
Weisburg WG, Barns SM, Pelletier DA, Lane DJ (1991) 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 173:697–703
Welsch TT, Gillock ET (2011) Triclosan-resistant bacteria isolated from feedlot and residential soils. J Environ Sci Health Part A 46(4):436–440
Yang J, Kloepper JW, Ryu CM (2009) Rhizosphere bacteria help plants tolerate abiotic stress. Trends Plant Sci 14:1–4
Yildirim E, Karlidag H, Turan M, Dursun A, Goktepe F (2011) Growth, nutrient uptake, and yield promotion of broccoli by plant growth promoting rhizobacteria with manure. Hort Sci 46:932–936
Zahir ZA, Zafar-ul-Hye M, Sajjad S, Naveed M (2011) Comparative effectiveness of Pseudomonas and Serratia sp. containing ACC-deaminase for coinoculation with Rhizobium leguminosarum to improve growth, nodulation, and yield of lentil. Biol Fertil Soils 47:457–465
Zdor RE (2015) Bacterial cyanogenesis: impact on biotic interactions. J Appl Microbiol 118:267–274
Zelaya-Molina LX, Hernández-Soto LM, Guerra-Camacho JE, Monterrubio-López R, Patiño-Siciliano A, Villa-Tanaca L, Hernández-Rodríguez C (2016) Ammonia-oligotrophic and diazotrophic heavy metal-resistant Serratia liquefaciens strains from pioneer plants and mine tailings. Microb Ecol 72:324–346
Zhang S, Reddy M, Kloepper JW (2002) Development of assays for assessing induced 12 systemic resistance by plant growth-promoting rhizobacteria against blue mold of 13 tobacco.
Zhao B, Cheng D, Tan P, An Q, Guo JS (2018) Characterization of an aerobic denitrifier Pseudomonas stutzeri strain XL-2 to achieve efficient nitrate removal. Bioresour Techno 250:564–573
Funding
This work was financially supported by the Tunisian Ministry of High Education and Scientific Research.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Mariem Samet and Imen Ghazala. Dr. Fatma Karray carried out the phylogenetic analysis, Cyrine Abid contributed to the in vitro analysis of the PGPR proprieties, Nour Chiab and Oumèma Nouri-Ellouz contributed to the field trials, Sami Sayadi helped to draft the manuscript and Radhia Gargouri-Bouzid participated in the design of the study and its revision. The first draft of the manuscript was written by Mariem Samet and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent to Publish
The participant has consented to the submission of the case report to the journal.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Diane Purchase
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Samet, M., Ghazala, I., Karray, F. et al. Isolation of bacterial strains from compost teas and screening of their PGPR properties on potato plants. Environ Sci Pollut Res 29, 75365–75379 (2022). https://doi.org/10.1007/s11356-022-21046-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-21046-8