Abstract
The pollution of the marine environment by treated and untreated effluents has increased due to human activities. Monitoring the marine ecosystem is nowadays a global concern. In this work, we evaluated the effect of contaminated and uncontaminated seawater, from different Tunisian coastal areas, on the fertilization, gastrulation, and embryo-larval development events of sea urchins (Paracentrotus lividus). The station of Salakta (SA) is considered as a control station, while the stations of Hamdoun Wadi (HW), Port of Monastir (PM), Karaia Monastir (KM), Teboulba (TE), and Khniss Lagoon (KL) are considered to be contaminated stations. The analysis of seawater physicochemical characteristics showed that levels of the total suspended matter (TSM), chemical oxygen demand (COD), biochemical oxygen demand (BOD), total organic carbon (TOC), and nitrate (NO3−) were lower in the seawater of the reference site Salakta (SA) when compared to those of the contaminated seawater sites. In addition, a very strong variation in the levels of trace metals in seawaters sampled in the studied sites was noted. In fact, the highest concentrations of Pb and Cu were observed in Hamdoun Wadi (HW), port of Monastir (PM), and Karaia Monastir (KM), while the highest concentration of Zn was noted in the Teboulba lagoon (TE) and Khniss (LK). Alterations in physicochemical characteristics as well as elevated trace metal levels in the studied seawater samples were correlated with reduced fertility rate, gastrulation rate, and the frequency of normal sea urchin larvae. The total absence of normal sea urchin pluteus larvae in the sea waters of heavily polluted sites proves the great sensitivity of the larval frequency to mixed pollution. This work recommends the utility of urchin fertilization and gastrulation rates and normal pluteus larval frequencies as useful bioassays to monitor the exposure of marine ecosystems to mixed pollution.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Several pollutants enter the marine ecosystems as a result of anthropogenic activities such as agriculture, industries, tourism, and shipping, and as consequence, these pollutants are rich in higher concentrations in different compartments of the natural ecosystem depending of type of contamination, acute or chronic exposure release (days, month, or years…), and concentrations. Due to their toxicity, persistence, and ability to accumulate in marine organisms and sediments, chemical compounds are widely called persistent pollutants. This group includes polycyclic aromatic hydrocarbons (PAHs), polychlorinated biphenyls (PCBs), and heavy metals. However, other compounds that have not been considered as contaminants have appeared recently. These are called emerging pollutants, which involve a long list of compounds including drugs, hormones, antimicrobials, and plasticizers
(Parra Luna et al. 2020). The increasing toxic discharges into the marine environment led to a decline in species diversity and negative consequences for human health when consume contaminated seafood (e.g., natural toxins, metals, organic pollutants). For this reason, the monitoring of this ecosystem contamination is an urgent necessity. Furthermore, there is growing concern about how to assess the marine environmental pollution. The monitoring marine ecosystem contamination is based on the combination of the two approaches; the first is the chemicals quantifications by analytical tools which includes high-performance liquid chromatography associated to mass spectroscopy (HPLC-MS) and gas chromatography coupled to the mass spectroscopy (GC-MS). The second approach is based on the assessment of biological effects of pollutants in the particular animal living in contaminated ecosystem. This approach is called biomonitoring, which aims to assess the ecotoxicological risks of pollution.
Bioindicators are marine organisms that able to provide qualitative and quantitative information about the quality of its environment and has a high degree of sensitivity or tolerance to many types of contamination or its effects (Vallaeys et al. 2017; Bonanno and Orlando-Bonaca 2018). Sea urchins are considered as preferred model organisms in ecotoxicology (Amri et al. 2017). Besides their key role in structuring benthic communities and whole marine ecosystems through their grazing activity, sea urchins are important bioindicators of marine pollution for their sedentary and benthic lifestyle, its wide geographical distribution, its year-round availability, its easy sampling, its well-known biology, its high bioaccumulation of chemical contaminants, and subsequent ability to reflect the health of the surrounding environment (Milito et al. 2020).
This species is sensitive to pollutants including trace metals (Gharred et al. 2016; Guendouzi et al. 2017; Bonaventura et al. 2018), pesticides (Levert et al. 2018), endocrine disruptors (Bošnjak et al. 2014; Tato et al. 2018), antibiotics (Gharred et al. 2016), and polycyclic aromatic hydrocarbons (Bellas et al. 2008; Rocha et al. 2018), as well as physical agents (X-ray, UV ...) (Matranga et al. 2010; Bonaventura et al. 2011).
The assessment and monitoring marine pollution by larvae bioassay and chemical determinations are essential for the marine ecosystem management and the protection. Several invertebrates’ larvae bioassays have been developed and validated its usefulness in many in field ecotoxicology exercises (His et al. 1997; Beiras et al. 2003; Van Dam et al. 2016). The sea urchin embryo-larval bioassays are the old biological tests, frequently used in the determination of the availability of chemicals and assess its toxicity (Bougis 1959; Kobayashi 1971. Several authors reported the sensibility of the sea urchin larvae abnormalities frequency response to pollutants exposure in controlled conditions of laboratory or in natural marine-polluted ecosystems (Nacci et al. 1986; Saco-Álvarez et al. 2010; Beiras et al. 2012; Gharred et al. 2015, 2016). The sea urchin embryo-larval bioassay low cost, ease of realization, and particularly sensitivity to pollutants exposures encouraged ecotoxicologists to standardize this test for its future use in the biomonitoring of marine pollution (Kobayashi et al. 1994; ASTM 1995).
This study aims to assess the usefulness of the sea urchin P. lividus embryo-larval malformations as bioassay test for the biomonitoring of Tunisian marine waters contamination. We are particularly interested first, in the assessment of the seawater physicochemical proprieties which include TSM, BOD, COD, NO3, and TOC and in the trace metal levels such as Pb, Cd, Cu, Ca, Mg, and Zn in the seawaters collected from six studied sites on the eastern coast of central Tunisia. Secondly, we evaluated the sensitivity of the embryo-larval malformations to the contamination level of the Tunisian coast area. This study hypothesize that embryo-larval malformations can be easy, fast, and sensitive procedure for biomonitoring of Tunisian marine waters and others around the world exposed to various anthropogenic discharges.
Material and methods
Animal sampling
Sexually mature sea urchins were sampled from reference site of the Tunisian littoral (Salakta site) (SA) (Fig. 1) from April to June 2019, using a landing net or submarine diving in the coastal region at depths less than 2 m. They were then transported to the laboratory in a cooler filled with seawater, where they have been kept in aquaria with clean, run, and aerated seawater until the start of tests, which will be initiated on the same day.
Study area and seawater sampling procedures
Seawater samples were taken approximately at the same depth (~3m) in all the studied sites to avoid the interference of salinity on the responses (false positives). The studied sites have been presented in (Figure 1). Salakta (SA) is a control site with low susceptibility to human influence and low levels of environmental contamination (Gharred et al. 2020). The other five sampling sites were chosen according to potential sources of industrial, harbor, and urban activities. Teboulba (TE), Khniss Lagoon (KL), and Hamdoun Wadi (HW) were characterized with chronic treated wastewater discharges (Jebali et al. 2011, 2013). Port of Monastir (PM), and Karia Monastir (KM) were characterized with the higher influence of harbor (Gharred et al. 2019).
The seawater was collected according to the standard sampling methods explained by Rodier et al. 1984, then, transported to the laboratory, and stored at −20°C until the analyses.
Chemical analysis of the seawater collected from the control and polluted sites
Seawater physicochemical proprieties determinations
The analysis of the chemical oxygen demand (COD), biochemical oxygen demand (BOD), total organic carbon (TOC), total suspended matter (TSM), and nitrate (NO3−) were determined in seawaters collected from control and polluted sites using a portable UV analyzer (Pastel UV, Secomam multimete, Alès, France) (Afsa et al. 2020)
The pH and the salinity of seawater of each site are measured by a pH meter and a refractometer respectively.
Trace metals analysis
Trace metals lead, copper, cadmium, calcium, magnesium, and zinc were quantified in seawater samples collected from reference and polluted sites (HW, PM, KM, KL, TE, and SA), by flame atomic absorption spectrometry (SP-AA 4000). (Aydin Urucu and Aydin 2015; Hernández et al. 2020).
Sea urchin embryo-larval test
Sea urchin bioassay was performed according to the methods described by many authors (McGibbon and Moldan 1986; Fernández and Beiras 2001; Bellas et al. 2005; Gharred et al. 2015, 2016). Mature sea urchins were placed individually, aboral side down on 50-ml beakers containing filtered seawater, collected from contaminated sites, Hamdoun Wadi (HW), Karaia Monastir (KM), Port of Monastir (PM), Khniss Lagoon (KL), Teboulba (TE) and from the reference site, Salakta (SA). Spawning is induced experimentally by injecting 1 ml of KCl (0.5 M) into the peristome, and the emission of the gametes occurs a few minutes later. Gamete emission and quality were visually inspected under light microscope at 200–×400 magnification to ensure the maturity of the gametes (mobile spermatozoa and regular spherical oocytes). Sperm suspension of one male and oocytes suspension of one female were mixed into 100 mL, using a sperm/oocyte ratio of 10/1. To follow the urchin embryo-larval development, three parameters were studied: the percentage of normal fertilization (% F), the percentage of normal gastrulas (% G), and the percentage of normal Pluteus larvae (% LP) determined after 20 min and 20 h and 48 h, respectively. These percentages were determined by counting at random a hundred oocytes, gastrulas, and larvae after their fixation with 100 μl of 40% formaldehyde. (Bellas et al. 2005; Manzo et al. 2013), and the calculation was done according to the following formulas:
% NF = 100 × ((number of fertilized eggs) / (total number of eggs counted))% NG = 100 × ((number of normal gastrulas) / (total number of gastrulas counted))% NLP = 100 × ((number of normal larvae) / (total number of larvae counted))
Values are shown as percentages ± standard deviation. Groups of 100 fertilized oocytes, gastrulas, or larvae were analyzed by optical microscopy and photographed using a digital camera. Three replicates per treatment were carried out.
The egg fertilization test was considered successful when the percentage of oocytes fertilization obtained in the seawater of the reference site was at least 90% (Nacci et al. 1986; Gharred et al. 2015, 2016). The percentage of abnormally fertilized eggs (those not surrounded fully or partially by a fertilization membrane), or failure rate, in 100 eggs was recorded in seawaters of the control and the contaminated sites. The frequencies of embryo-larval abnormalities were performed after the egg fertilization test. The gastrulation test was considered successful when the percentage of normally gastrula in the control was at least 90% (Gharred et al. 2015, 2016). The percentage of abnormal gastrula is characterized by the absence of the archenteron invagination and secondary mesenchymal cell detachment or failure rate. The larval test was considered successful when the percentage of normally developed pluteus in the control was at least 90% (Bellas et al. 2005; Gharred et al. 2015, 2016). The percentage of abnormally developed pluteus (those who do not have well-developed arms), or failure rate, was taken as an endpoint of contaminant toxicity according to the criteria of many previous works (Pesando et al. 2003, 2004; Carballeira et al. 2012a, b; Gambardella et al. 2013; Gharred et al. 2015, 2016).
Statistical analyses
The multiple comparisons of the seawater physicochemical parameters including TSM, COD, BOD, TOC, and NO3 and the embryo-larval development parameters %F, %G, and %LP between sites were conducted using SPSS software 20.0. The results were expressed as mean ± SD. Results were analyzed by one-way ANOVA followed by Duncan’s multiple range test. Differences were considered statistically significant at p<0.05.
The agglomerative hierarchical clustering (AHC), as multivariate analysis of the embryo-larval and physicochemical parameters, was conducted using the XLSTAT 2014 software. In this study, the embryo-larval and physicochemical parameters were used as variables and the six studied sites as observations. The similarity between sites was measured by Pearson correlation coefficient.
Results
Seawater physicochemical characteristics
The analysis of seawater physicochemical characteristics showed that levels of the total suspended matter (TSM), chemical oxygen demand (COD), biochemical oxygen demand (BOD), total organic carbon (TOC), and nitrate (NO3-) were lower in the seawater of the reference site Salakta (SA) when compared to those of the contaminated seawater sites. The highest levels of the parameters TSM, COD, TOC, NO3− were noted in the seawater of the Khniss Lagoon (KL) which could be related to the higher urban discharges rich in organic matter (Fig. 2).
The most acidic pH and the highest salinity were noted in the seawater of (KL) (Table 1).
Seawater trace metals levels
The results of trace metals concentrations in seawaters of the reference and contaminated sites are presented in (Table 1). There is a very strong variation in the trace metals concentrations found according to the sampling stations. The results clearly show that the high Pb and Cu seawaters concentrations were observed in Hamdoun Wadi (HW), port of Monastir (PM), and Karaia Monastir (KM), while the high Zn seawater concentrations were noted in Teboulba (TE) and Khniss Lagoon (KL) sites. Indeed, the highest Cd seawater concentration was marked in Hamdoun Wadi (HW) site (Table 1).
Assessment of embryo-larval development of P. lividus in seawaters of reference and contaminated sites
The embryo larval development of P. lividus is characterized by crucial successive stages from fertilization to organogenesis. The success of each stage was evaluated by the percentage of the fertilized eggs, normal gastrulas, and the normal larva frequencies.
The percentage of oocyte fertilization success (%F) in seawater from each study site is given in (Fig. 3). The results clearly show that a distinguished higher fertilization rate was observed in the seawater of the reference site Salakta (SA) with 96 ± 2.3%, and very low percentages of oocyte fertilization (%F) were noted in the seawaters of the contaminated sites Khniss lagoon (KL), Port of Monastir (PM), Hamdoun Wadi (HW), and Karaia Monastir (KM). The lowest percentage of oocyte fertilization (%F) was in seawater of PM site with 53 ± 2.15% indicting a higher contamination level of this site.
(1) Percentage of fertilization, (2) percentages of normal gastrulas, and (3) percentages of normal pluteus larvae of oocytes in seawater collected from contaminated sites: Hamdoun Wadi (HW), Karaia Monastir (KM), Port of Monastir (PM), Khniss Lagoon (KL), Teboulba (TE), and Salakta (SA) reference site. Results are expressed as mean standard deviation (M ± SD). The letters a, b, c, d, and e indicate a significant difference between the sites
The gastrula stage of the sea urchin was obtained 20-h post-fertilization and by adding buffered formaldehyde to stop embryo larval development. The normal sea urchin gastrula is characterized by the archenteron invagination and by the secondary mesenchyme cells detachment. The percentages of normal gastrulas observed in the marine waters collected from several sites Tunisian littoral areas are given in (Fig. 3). The results show a higher rate of normal gastrulas in seawater of the reference site and low percentages that did not exceed 60% in contaminated sites. The normal gastrula rates significantly differed among the studied sites and were 54 ± 1.75%, 52 ± 3.15%, 52 ± 1.5%, 52± 2.5%, and 43 ± 2.8% for Teboulba (TE), Hamdoun Wadi (HW), Karaia Monastir (KM), Port of Monastir (PM), and Khniss Lagoon (KL) respectively.
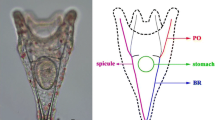
The larval stage of the sea urchin is obtained 48-h post-fertilization and by adding buffered formaldehyde to the medium to stop embryo-larval development. Organogenesis of the sea urchin gastrulas begins with the opening of the anus, then the mouth on the opposite side, then the formation of spicules and the four oral and anal arms of the pluteus larvae. The rates of normal larvae in seawaters from the control and contaminated sites are shown in (Fig. 3). A higher rate of normal larvae was noted in seawater of the control site, a low percentage of normal larvae seawater from TE, and dramatically diminished rates in seawaters of the Karaia Monastir (KM) and Hamdoun Wadi (HW) sites with percentages of 18 ± 1.78% and 12 ± 2.8%, respectively. Interestingly, total absence of larvae was in the seawaters of the Port of Monastir (PM) and Khniss Lagoon (KL) sites.
Evaluation of the embryo-larval development of P. lividus in diluted seawaters of highly contaminated sites
The total absence of larvae in the waters of the Khniss Lagoon site (KL) and port of Monastir (PM) encouraged us to investigate the effect of diluted seawaters on the fertilization success, gastrulation, and embryo-larvae of P. lividus. Successive dilutions of ½ and 1/3 of the contaminated sites seawater: Khniss Lagoon (KL) and port of Monastir (PM) with control seawater were adopted.
The percentages of fertilization success, gastrulation, and normal larvae are given in (Fig. 4). The obtained results show that the ½ and 1/3 dilutions of Khniss Lagoon (KL) seawaters improved the percentages of fertilization success at 61 ± 1.75% and 72 ± 2.5% respectively. The gastrulation rate ameliorated up to 55 ± 1.75% in ½ diluted contaminated seawater and increased to 63 ± 1% in 1/3 diluted contaminated seawater. The normal larvae frequency in ½ diluted contaminated seawater was 10 ± 2% and 28 ± 3.1% in 1/3 diluted contaminated seawater. The same for the case of port of Monastir (PM), the dilution of ½ and 1/3 shows a clear improvement in the percentages of fertilization success at 63 ± 1.75% and 67 ± 1.5% respectively. The gastrulation rate ameliorated up to 56 ± 1.75% in ½ dilution and increased to 60 ± 1% in 1/3 dilution. The normal larvae frequency was 11, 2 ± 2% in the ½ diluted contaminated seawater and 20 ± 3.1% in the 1/3 diluted contaminated seawater.
Percentages of fertilization, gastrulation, and normal larvae determined in seawater. A From the reference site Salakta (SA) and Khniss Lagoon (KL). B From the reference site Salakta (SA) and Port of Monastir (PM), diluted to ½ and 1/3. The results are expressed as an average of standard deviation (M ± DS). The letters A, B, C, D, a, b, c, d, (a), (b), (c), and (d) indicate a significant difference between the sites
The improved fertilization, gastrulation, and organogenesis success of P. lividus confirmed the sensitivity of these embryonic-larval developmental events to mixed pollution exposure and proved its utility in the program of biomonitoring of seawater contamination assessment.
The agglomerative hierarchical clustering (AHC) of biological and physicochemical parameters
The agglomerative hierarchical clustering (AHC) was used to investigate the possible correlation between the embryo-larvae development and the physicochemical parameters of the control and contaminated site seawaters. The AHC is the most common type of hierarchical clustering used to group objects (sites in this study) in clusters based on their similarity. Thus, sites with similar biological and chemical data will be in the same cluster. The full dendrogram (Fig. 5) displays the progressive clustering of sites. The level of truncation marked by a broken line shows three clusters. The first cluster contained SA considered as reference and TE considered as the less polluted sites. The second cluster of HW and KM was considered as polluted sites, and the third cluster contained highly polluted sites PM and KL.
Discussion
Mediterranean coastal areas, including Tunisian area, are gradually affected by the intensification of anthropogenic activities which increase pollution, eutrophication, and habitat destruction that negatively affect coastal marine ecosystems (Jebali et al. 2013; Gharred et al. 2019; Parra Luna et al. 2020). The anthropogenic activities cause growing discharges of pollutants of different nature into the marine environment that threat aquatic organisms. Thus, development and validation of the monitoring approach of marine pollution based on the chemical and biological tools would be of great interest in Tunisia and in other countries around the world. This study aimed to assess the usefulness of the sea urchin P. lividus embryo-larval malformations as bioassay test for the biomonitoring of Tunisian marine water contaminations. This study hypothesizes that embryo-larval malformations can be easy, fast, and sensitive biological approach for biomonitoring of marine environment contamination. Sea urchins are a widely used model system for toxicological studies in controlled and in-field conditions (Warnau et al. 2006; Pétinay et al. 2009; Martin et al. 2011; Amaroli et al. 2013; Gharred et al. 2015, 2016; Martino et al. 2018; Ruocco et al. 2020). Indeed, as key species well represented both in planktonic and benthic marine ecosystem at larval and adult stages, respectively, and being highly sensitive to ambient condition variations, sea urchins can be used to assess the biological effects of marine pollution on the early embryo-larval development disruption.
It is well known that the pollutants arriving in the marine environment are distributed in the different compartments of sediments, seawater, and living organisms. However, the sediments are the main compartments of pollutants storage and are in continuous exchange of pollutants with the other compartments in which directly linked seawater and organisms (Jebali et al. 2012). In this work, we decided to assess the impact of the polluted seawaters of polluted sites of the Tunisian coastal areas on the embryo-larval developmental stages of sea urchin (P. lividus). Several arguments encourage us to select the seawaters of polluted sites to assess the responses of this sea urchin bioassay test to seawaters pollutants; the first, this animal occupied the rocky littoral zones and in direct contact with seawater and secondly, the embryo-larval development accrued in seawaters.
The results indicated that fertilization, gastrulation, and normal pluteus larvae frequencies were highly diminished in the contaminated sites and were correlated to the measured physicochemical parameters and traces metals. The alteration of the seawater physico-chemical parameters such as TSM, BOD, COD, NO3, TOC, and turbidity could be indicators of eutrophication phenomenon when dissolved oxygen highly decreased and negatively affect on the sea urchin embryo-larvae development and animal growth (Ternengo et al. 2018). The low seawater pH of khniss lagoon (KL; pH = 6.22) affected the normal fertilization, gastrulation, and organogenesis processes of sea urchin (Paracentrotus lividus). These results are in agreement with those found by Limatola et al. (2020). In addition, Dorey et al. (2018) clearly demonstrated that a reduction in the seawater pH by one unit (from pH 8.1 to 7.1) increased the incorporation kinetics of six trace metals (Mn, Co, Zn, Se, Ag, Cd, Cs) in the sea urchin larvae.
Interestingly, a total absence of pluteus larvae was noted in the seawaters of the highly polluted sites: Port of Monastir (PM) and Khniss Lagoon (KL) sites. Zúñiga et al. (1995) reported strong effects on fertilization after exposure of the sperm cell of the sea urchin Arbacia spatuligira to the seawaters from two coastal areas with different sources of pollution; the first site characterized with high organic-waste discharges (Rocuant site) and the second site received chemical discharges.
The agglomerative hierarchical clustering (AHC) analysis of the chemical and embryo-larval results showed a dendrogram of three distinct site groups.
The first cluster formed by SA as reference and TE as the less polluted sites. Indeed, the site of Salakta (SA) is far from any source of contamination because its coast forms a cape sheltered from any discharge of wastewater and is characterized by constant agitation and coastal currents promoting good water renewal (Gharred et al. 2019, 2020). This explains why Salakta (SA) site reflects the highest percentages of embryonic development success.
The Teboulba (TE) is a moderately polluted site with trace metals. The presence of these elements could be linked to road traffic, maritime activities, the fishing port, and human activity marked by the drainage of wastewater to the sea. This explains the highest percentages of embryonic development success among all polluted sites.
The highly polluted cluster is formed by the Khniss Lagoon site (KL) and the Port of Monastir. The Khniss Lagoon site (KL) is affected by the presence of multiple sources of pollution from urban, industrial, and aquaculture activities of the limitroph agglomerations. The anthropogenic activities result in significant increase in several chemical concentrations in different compartments of the littoral area (Damak et al. 2019a, b). In an example, a recent study of Amor et al. (2020) shows that the coastal area of Khniss to Sayada is contaminated particularly by trace metals (Zn, Cu…) and with higher contents of TOC. The discharges cause also eutrophication phenomenon altering the seawater quality (Nouira et al. 2013). Recently, Khiari et al. (2021) reported that Monastir lagoon sediments have been polluted for a long time, with hydrocarbons from varied anthropogenic sources and fifteen total polyaromatic hydrocarbon (15 priority pollutants) were found at different levels ranged from 222 to 2992 μg kg−1 of core sediments.
The chronic pollution of the Port of Monastir (PM) comes from hydrocarbons resulting from normal maritime and port activities or from involuntary or voluntary leaks and discharges of fuels and used oils.
The mixture contaminations of these two sites explained the highest rates of failure of the sea urchin embryonic development noted in this work.
The second cluster gathers the two polluted sites: Hamdoun Wadi (HW) and Karaia Monastir (KM).
The Hamdoun Wadi (HW) site is subjected to metal contaminations which include Pb and Cu and other organic chemicals such as textile disperse red 1 and the disperse yellow 3, which were detected in the Hamdoun River with concentrations of 3.873 μg/L and 1895 μg/L, respectively (Methneni et al. 2021).
For the Karaia Monastir (KM) site (swimming area), the presence of the contamination elements could be linked to road traffic and maritime and tourist activities.
Metallic pollution by Cd, Pb, Zn, Mg, and Cu of marine environment has been considered as serious health hazard for living organisms due to their persistence, ability to accumulate in marine organisms, and toxicity (Guendouzi et al. 2017). There are many studies reported the effects of Cd on the development of sea urchin embryos and morphological effects on larvae. In the laboratory conditions, Gharred et al. (2016) showed that concentrations of Cd and Cu superior to 338 μg/L and 0.56 μg/L respectively caused significant larval malformations in P. lividus. In this work, we noted that the seawater-contaminated sites by Cd and Cu (HW) have a minimal normal larval development and maximum larval anomalies. It has been shown that high concentrations of Cu and Pb caused larval malformations and blockage at different stages of P. lividus embryo-larval development (Fernández and Beiras 2001; Lewis and Ford 2012). That may explain our results where sites with high level of Pb (PM and KM) presented respectively 100% ± 2.7 and 82% ± 1.75 of larval malformation.
According to Casas (2005), the presence of Zn in sea water at high concentrations causes disturbances in sea urchin fertilization and segmentation (Lallier 1965) and inhibits the sea urchin embryonic development through the delay of the fertilization membrane elevation (Kobayashi and Okamura 2005).
The comparison of the sensitivity of the analyzed embryo-larval events to control and contaminated seawater’s exposure showed that the sea urchin larval plutei were strongly affected than the fecundity and gastrula frequencies. Thus, sea urchin larval plutei anomalies could be a useful bioassay to assess mixture pollutions of marine ecosystem and involved in the biomonitoring program.
The total absence of larvae in the waters of the Khniss Lagoon site (KL) and port of Monastir (PM) encouraged us to investigate the effect of polluted seawater dilutions on the fertilization success, gastrulation, and embryo-larvae of P. lividus. The diluted polluted seawaters highly improved the sea urchin embryo-larval development. Thus, the obtained results proved the sensitivity of the fertilization, gastrulation, and particularly the embryo-larvae anomalies to polluted seawaters. Previous work showed the exposure of spermatozoids to copper concentrations superior to 50 μg/l for 30 min inhibits fertilization, and lower concentrations have a severe toxic effect concentration-dependent on the acceleration of kinetics blastomere divisions, reduction of pluteus larvae size, general larva shape alteration, and increase in the embryo anomalies (Gharred et al. 2015). Moreover, the sea urchin P. lividus embryo-larval malformation is a very sensitive bioassay to a variety of effluents in coastal areas including the effluents from pisciculture farms (Carballeira et al. 2012a) and industrial and urban agglomeration (Meriç et al. 2005).
Conclusions
This study confirmed the different contamination levels of Tunisian coast areas via the trace metals and seawater’s physic-chemical parameter alterations. The high trace metals were correlated with the reduction of fecundity, gastrulation, and normal sea urchin pluteus larval frequencies. A total absence of the sea urchin pluteus larval in the highly polluted seawaters proved the high sensitivity of the larval anomalies frequency to the mixture of pollution. All the results recommended the usefulness of the sea urchin Paracentrotus lividus as appropriate indicator of the marine environment quality and can be integrated in the biomonitoring program of the marine pollution.
Data availability
The datasets used during the current study are available from the corresponding author on reasonable request.
References
Afsa S, Hamden K, Martin PAL, Mansour HB (2020) Occurrence of 40 pharmaceutically active compounds in hospital and urban wastewaters and their contribution to Mahdia coastal seawater contamination. Environ Sci Pollut Res 27(2):1941–1955. https://doi.org/10.1007/s11356-019-06866-5
Amaroli A, Grazia M, Falugi C, Giovanna M (2013) Effects of the neurotoxic thiophosphate pesticide chlorpyrifos on differentiating alternative models. Chemosphere. 90:2115–2122. https://doi.org/10.1016/j.chemosphere.2012.11.005
Amor RB, Jerbi H, Abidi M, Gueddari M (2020) Assessment of trace metal contamination, total organic carbon and nutrient accumulation in surface sediments of Monastir Bay (Eastern Tunisia, Mediterranean Sea). Reg Stud Mar Sci 34:101089. https://doi.org/10.1016/j.rsma.2020.101089
Amri S, Samar MF, Sellem F, Ouali K (2017) Seasonal antioxidant responses in the sea urchin Paracentrotus lividus (Lamarck 1816) used as a bioindicator of the environmental contamination in the South-East Mediterranean. Mar Pollut Bull 122:392–402. https://doi.org/10.1016/j.marpolbul.2017.06.079
ASTM (1995) Standard guide for conducting static acute toxicity tests with echinoid embryos. American Society for Testing and Materials E1563-95:962–980
Aydin Urucu O, Aydin A (2015) Coprecipitation for the determination of copper (II), zinc (II), and lead (II) in seawater by flame atomic absorption spectrometry. Anal Lett 48(11):1767–1776. https://doi.org/10.1080/00032719.2014.999275
Beiras R, Fernández N, Bellas J, Besada V, González-Quijano A, Nunes T (2003) Integrative assessment of marine pollution in Galician estuaries using sediment chemistry, mussel bioaccumulation, and embryo-larval toxicity bioassays. Chemosphere 52(7):1209–1224. https://doi.org/10.1016/s0045-6535(03)00364-3
Beiras R, Duran I, Bellas J, Sánchez-Marín P (2012) Biological effects of contaminants: Paracentrotus lividus sea urchin embryo test with marine sediment elutriates. ICES Techniques in Marine Environmental Sciences No 51:13
Bellas J, Beiras R, O-Balsa JCM, Fernandez N (2005) Toxicity of organic compounds to marine invertebrate embryos and larvae: a comparison between the sea urchin embryogenesis bioassay and alternative test species. Ecotoxicology 14:337–353
Bellas J, Saco-Álvarez L, Nieto O, Beiras R (2008) Ecotoxicological evaluation of polycyclic aromatic hydrocarbons using marine invertebrate embryo-larval bioassays. Mar Pollut Bull 57:493–502. https://doi.org/10.1016/j.marpolbul.2008.02.039
Bonanno G, Orlando-Bonaca M (2018) Perspectives on using marine species as bioindicators of plastic pollution. Mar Pollut Bull 137:209–221. https://doi.org/10.1016/j.marpolbul.2018.10.018
Bonaventura R, Zito F, Costa C, Giarrusso S, Celi F, Matranga V (2011) Stress response gene activation protects sea urchin embryos exposed to X-rays. Cell Stress Chaperones 16:681–687. https://doi.org/10.1007/s12192-011-0277-3
Bonaventura R, Zito F, Chiaramonte M, Costa C, Russo R (2018) Nickel toxicity in P. lividus embryos: dose dependent effects and gene expression analysis. Mar Environ Res 139:113–121. https://doi.org/10.1016/j.marenvres.2018.05.002
Bošnjak I, Borra M, Iamunno F, Benvenuto G, Ujević I, Bušelić I, Roje-Busatto R, Mladineo I (2014) Effect of bisphenol A on P-glycoprotein-mediated efflux and ultrastructure of the sea urchin embryo. Aquat Toxicol 156:21–29. https://doi.org/10.1016/j.aquatox.2014.07.018
Bougis P (1959) Sur l’effet biologique du cuivre en eau de mer. C.R.A cad. Sci. Paris 249(12):326–328
Carballeira C, Ramos-Gómez J, Martín-Díaz L, DelValls TA (2012a) Identification of specific malformations of sea urchin larvae for toxicity assessment: application to marine pisciculture effluents. Mar Environ Res 77:12–22. https://doi.org/10.1016/j.marenvres.2012.01.001
Carballeira C, De Orte M, Viana R, DelValls IG, Carballeira TA (2012b) Assessing the toxicity of chemical compounds associated with land-based marine fish farms: the sea urchin embryo bioassay with Paracentrotus lividus and Arabica lixula. Arch Environ ContamToxicol 63:249–261
Casas, S. 2005 Modélisation de la bioaccumulation de métaux traces (Hg, Cd, Pb, Cu et Zn) chez la moule, Mytilus galloprovincialis, en milieu méditerranéen. Doctorat de l’université du Sud Toulon Var. Spécialité: Océanologie biologique, Environnement marin, France pp. 363
Damak M, Frontalini F, Elleuch B, Kallel M (2019a) Benthic foraminiferal assemblages as pollution proxies along the coastal fringe of the Monastir Bay (Tunisia). J Afr Earth Sci 150:379–388. https://doi.org/10.1016/j.jafrearsci.2018.11.013
Damak M, Fourati R, Ellech B, Kallel M (2019b) Assessment of organic and metallic contamination in the surface sediment of Monastir Bay (Eastern Tunisia): Spatial distribution, potential sources, and ecological risk assessment. Mar Pollut Bull 149:110500. https://doi.org/10.1016/j.marpolbul.2019.110500
Dorey N, Martin S, Oberhänsli F, Teyssié JL, Jeffree R, Lacoue-Labarthe T (2018) Ocean acidification modulates the incorporation of radio-labeled heavy metals in the larvae of the Mediterranean sea urchin Paracentrotus lividus. J Environ Radioact 190:20–30. https://doi.org/10.1016/J.JENVRAD.2018.04.017
Fernández N, Beiras R (2001) Combined toxicity of dissolved mercury with copper, lead and cadmium on embryogenesis and early larval growth of the Paracentrotus lividus sea-urchin. Ecotoxicology. 10:263–271. https://doi.org/10.1023/A:1016703116830
Gambardella C, Aluigia MG, Ferrandoa S, Gallusa L, Ramoinoa P, Gattib AM, Rottignia M, Falugi C (2013) Developmental abnormalities and changes in cholinesterase activity in sea urchin embryos and larvae from sperm exposed to engineered nanoparticles. Aquat Toxicol 130–131:77–85
Gharred T, Kawther Ezzine I, Naija A, Bouali RR, Jebali J (2015) Assessment of toxic interactions between deltamethrin and copper on the fertility and developmental events in the Mediterranean sea urchin, Paracentrotus lividus. Environ Monit Assess 187(4):193. https://doi.org/10.1007/s10661-015-4407-8
Gharred T, Jebali J, Belgacem M, Mannai R, Achour S (2016) Assessment of the individual and mixture toxicity of cadmium, copper, and oxytetracycline, on the embryo-larval development of the sea urchin Paracentrotus lividus. Environ Sci Pollut Res 23:18064–18072. https://doi.org/10.1007/s11356-016-6988-3
Gharred T, Helaoui A, Mannai R, Jebali J (2019) Effect of multiple pollution on the cholinesterase activity, morphometry and reproduction performance of Patella caerulea collected from eastern Tunisian coasts. Cah Biol Mar 60:11–20. https://doi.org/10.21411/CBM.A.132317A6
Gharred T, Mannai R, Belgacem M, Jebali J (2020) Incidence of morphometry variation, growth alteration, and reproduction performance of the annular sea bream (Diplodus annularis) as effective tools to assess marine contamination: how useful is a multi-biotimarkers approach? Environ Sci Pollut Res 27(4):4075–4088. https://doi.org/10.1007/s11356-019-07014-9
Guendouzi Y, Boulahdid M, Boudjenoun M, Mezali K, Soualili DL (2017) Seasonal variation in bioavailability of trace metals in the echinoid Paracentrotus lividus (Lamarck, 1816) from Algerian coastal waters: effect of physiological indices. Reg Stud Mar Sci 14:112–117. https://doi.org/10.1016/j.rsma.2017.05.010
Hernández MA, Portillo R, Salgado MA, Hernández GI, Zagoya J, De los Ángeles Velasco M, Rivera A, Romero O (2020) Textural and morphological analysis of the Popocatépetl volcano ashes. Mex J Mat Sci Eng 7:1–8
His E, Seaman MNL, Beiras R (1997) A simplification of the bivalve embryogenesis and larval development bioassay method for water quality assessment. Water Res 31(2):351–355. https://doi.org/10.1016/s0043-1354(96)00244-8
Jebali J, Ben-Khedher S, Ghedira J, Kamel N, Boussetta H (2011) Integrated assessment of biochemical biomarker responses in Mediterranean crab (Carcinus maenas) collected from Monastir Bay (Tunisia). J Environ Scie, (2011) 23(10):1714–1720. https://doi.org/10.1016/S1001-0742(10)60617-1
Jebali J, Banni M, Boussetta H (2012) Biochemical biomarkers in aquatic ecotoxicology: fundamental mechanisms, application and perspectives. In: Daniel JA (ed) Advance in environmental research, vol 23–323. Nova Science Publisher, New York, pp 143–168
Jebali J, Sabbagh M, Banni M, Kamel N, Ben-Khedher S, M’hamdi N, Boussetta H (2013) Multiple biomarkers of pollution effects in Solea solea fish on the Tunisia coastline. Environmental Science and Pollution Research 4 20(6):3812–3821. https://doi.org/10.1007/s11356-012-1321-2
Khiari N, Charef A, Atoui A, Azouzi R, Khalil N, Khadhar S (2021) Southern Mediterranean coast pollution: long-term assessment and evolution of PAH pollutants in Monastir Bay (Tunisia). Mar Pollut Bull 167:112268. https://doi.org/10.1016/j.marpolbul.2021.112268
Kobayashi N (1971) Fertilized sea urchin eggs as an indicatory material for marine pollution bioassay, preliminary experiments. Publ Seto Mar Biol Lab 18(6):379–406
Kobayashi N, Okamura H (2005) Effects of heavy metals on sea urchin embryo development. Part 2. Interactive toxic effects of heavy metals in synthetic mine effluents. Chemosphere. 61:1198–1203. https://doi.org/10.1016/j.chemosphere.2005.02.071
Kobayashi N, Naidenko TKH, Vashchenko MA (1994) Standardization of a bioassay using sea-urchin embryos. Russ J Mar Biol 20(6):351–357
Lallier R (1965) Effets du 5-fluoro-uracile et de la 6-méthylpurine sur le développement de l’œuf de l’oursin Paracentrotus lividus. Development. 14:181–189. https://doi.org/10.1242/dev.14.2.181
Levert A, Alvariño R, Bornancin L, Mansour EA, Burja AM, Genevière AM, Bonnard I, Alonso E, Botana L, Banaigs B (2018) Structures and activities of Tiahuramides A-C, cyclic depsipeptides from a Tahitian collection of the marine Cyanobacterium Lyngbya majuscule. J Nat Prod 81:1301–1310. https://doi.org/10.1021/acs.jnatprod.7b00751
Lewis C, Ford AT (2012) Infertility in male aquatic invertebrates: a review. Aquat Toxicol 120:79–89. https://doi.org/10.1016/j.aquatox.2012.05.002
Limatola N, Chun JT, Santella L (2020) Effects of salinity and pH of seawater on the reproduction of the sea urchin Paracentrotus lividus. Biol Bull 239(1):13–23. https://doi.org/10.1086/710126
Manzo S, Miglietta ML, Rametta G, Buono S, Di Francia G (2013) Embryotoxicity and spermiotoxicity of nanosized ZnO for Mediterranean sea urchin Paracentrotus lividus. J Hazard Mater 254:1–7. https://doi.org/10.1016/j.jhazmat.2013.03.027
Martin S, Richier S, Pedrotti M, Dupont S, Castejon C, Gerakis Y, Kerros M, Oberhänsli F, Teyssié J, Jeffree R, Gattuso JP (2011) Early development and molecular plasticity in the Mediterranean sea urchin Paracentrotus lividus exposed to CO2-driven acidification. J Exp Biol 214:1357–1368. https://doi.org/10.1242/jeb.051169
Martino C, Costa CR, Maria C, Koop D, Scudiero R, Byrne M (2018) Gadolinium perturbs the expression of skeletogenic genes, calcium uptake, and larval development in phylogenetically distant sea urchin species. Aquat Toxicol 194:57–66. https://doi.org/10.1016/J.AQUATOX.2017.11.004
Matranga V, Zito F, Costa C, Bonaventura R, Giarrusso S, Celi F (2010) Embryonic development and skeletogenic gene expression affected by X-rays in the Mediterranean sea urchin Paracentrotus lividus. Ecotoxicology. 19:530–537. https://doi.org/10.1007/s10646-009-0444-9
McGibbon S, Moldan AGS (1986) Routine toxicity testing of toxicants using a sea urchin gamete bioassay. Mar Pollut Bull 17(2):68–72
Meriç S, De Nicola E, Iaccarino M, Gallo M, Di Gennaro A, Morrone G, Warnau M, Belgiorna V, Pagano G (2005) Toxicity of leather tanning wastewater effluents in sea urchin early development and in marine microalgae. Chemosphere 61(2):208–217. https://doi.org/10.1016/j.chemosphere.2005.02.037
Methneni N, González JAM, Van Loco J, Anthonissen R, de Maele JV, Verschaeve L, Fernandez-Serrano M, Mansour HB (2021) Ecotoxicity profile of heavily contaminated surface water of two rivers in Tunisia. Environ Toxicol Pharmacol 82:103550. https://doi.org/10.1016/j.etap.2020.103550
Milito A, Murano C, Castellano I, Romano G, Palumbo A (2020) Antioxidant and immune response of the sea urchin Paracentrotus lividus to different re-suspension patterns of highly polluted marine sediments. Mar Environ Res 160:104978. https://doi.org/10.1016/j.marenvres.2020.104978
Nacci D, Jackim E, Walsh R (1986) Comparative evaluation of three rapid marine toxicity tests: sea urchin early embryo growth test, sea urchin sperm cell toxicity test and microtox. Environ Toxicol Chem 5:521–525
Nouira T, Risso C, Chouba L, Budzinski H, Boussetta H (2013) Polychlorinated biphenyls (PCBs) and polybrominated diphenyl ethers (PBDEs) in surface sediments from Monastir Bay (Tunisia, Central Mediterranean): occurrence, distribution and seasonal variations. Chemosphere 93(3):487–493. https://doi.org/10.1016/j.chemosphere.2013.06.017
Parra Luna M, Martín Pozo L, Hidalgo F, Zafra Gómez A (2020) Common sea urchin (Paracentrotus lividus) and sea cucumber of the genus Holothuria as bioindicators of pollution in the study of chemical contaminants in aquatic media. A revision Ecol Indic 113:106185. https://doi.org/10.1016/j.ecolind.2020.106185
Pesando D, Huitorelb P, Dolcinia V, Angelinic C, Guidettid P, Falugic C (2003) Biological targets of neurotoxic pesticides analysed by alteration of developmental events in the Mediterranean Sea urchin, Paracentrotus lividus. Mar Environ Res 55:39–57
Pesando D, Robert S, Huitorel P, Gutknecht E, Pereira L, Girard JP, Ciapa B (2004) Effects of methoxychlor, dieldrin and lindane on sea urchin fertilization and early development. Aquat Toxicol 66:225–239
Pétinay S, Chataigner C, Basuyaux O (2009) Standardisation du développement larvaire de l’oursin, Paracentrotus lividus, pour l’évaluation de la qualité d’une eau de mer. C R Biol 332:1104–1114. https://doi.org/10.1016/j.crvi.2009.08.002
Rocha AC, Camacho C, Eljarrat E, Peris A, Aminot Y, Readman JW, Boti V, Nannou C, Marques A, Nunes ML, Almeida CM (2018) Bioaccumulation of persistent and emerging pollutants in wild sea urchin Paracentrotus lividus. Environ Res 161:354–363. https://doi.org/10.1016/j.envres.2017.11.029
Rodier J, Geoffray C, Rodi L (1984) L’analyse de l’eau: eaux naturelles, eaux résiduaires, eau de mer: chimie, physico-chimie, bactériologie, biologie (p. 1365). Dunod, Paris
Ruocco N, Bertocci I, Munari M, Musco L, Caraniello D, Danovaro R, Zupo V, Costantini M (2020) Morphological and molecular responses of the sea urchin Paracentrotus lividus to highly contaminated marine sediments: the case study of Bagnoli-Coroglio brownfield (Mediterranean Sea). Mar Environ Res 104865:104865. https://doi.org/10.1016/j.marenvres.2019.104865
Saco-Álvarez L, Durán I, Ignacio Lorenzo J, Beiras R (2010) Methodological basis for the optimization of a marine sea-urchin embryo test (SET) for the ecological assessment of coastal water quality. Ecotoxicol Environ Saf 73(4):491–499. https://doi.org/10.1016/j.ecoenv.2010.01.018
Tato T, Salgueiro-González N, León VM, González S, Beiras R (2018) Ecotoxicological evaluation of the risk posed by bisphenol A, triclosan, and 4-nonylphenol in coastal waters using early life stages of marine organisms (Isochrysis galbana, Mytilus galloprovincialis, Paracentrotus lividus, and Acartia clausi). Environ Pollut 232:173–182. https://doi.org/10.1016/j.envpol.2017.09.031
Ternengo S, Marengo M, El Idrissi O, Yepka J, Pasqualini V, Gobert S (2018) Spatial variations in trace element concentrations of the sea urchin, Para-centrotus lividus, a first reference study in the Mediterranean Sea. Mar PollutBull 129:293e298–293e298. https://doi.org/10.1016/j.marpolbul.2018.02.049
Vallaeys T, Klink SP, Fleouter E, Le Moing B, Lignot JH, Smith AJ (2017) Bioindicators of marine contaminations at the frontier of environmental monitoring and environmental genomics. Adv Biotechnol Microbiol 4:555629. https://doi.org/10.19080/AIBM.2017.04.555629
Van Dam JW, Trenfield MA, Harries SJ, Streten C, Harford AJ, Parry D, van Dam RA (2016) A novel bioassay using the barnacle Amphibalanus amphitrite to evaluate chronic effects of aluminium, gallium and molybdenum in tropical marine receiving environments. Mar Pollut Bull 112(1-2):427–435. https://doi.org/10.1016/j.marpolbul.2016.07.015
Warnau M, Dutrieux S, Ledent G, Rodriguez Y, Baena AM, Dúbois P (2006) Heavy metals in sea cucumber Holothuria tubulosa (Echinodermata) from the Mediterranean Posidonia oceanica ecosystem: body compartment, seasonal, geographical and bathymetric variations. Environ Bioindic 1:268–285. https://doi.org/10.1080/15555270601034388
Zúñiga M, Roa R, Larrain A (1995) Sperm cell bioassays with the sea urchin Arbacia spatuligera on samples from two polluted Chilean coastal sites. Mar Pollut Bull 30(5):313–319. https://doi.org/10.1016/0025-326X(94)00179-D
Acknowledgements
Authors acknowledge Pr. Hedi Ben Mansour, head of the Research Unit for Environmental Analysis and Processes (UR17ES32) (APAE) of the Higher Institute of Applied Sciences and Technology of Mahdia (ISSATM), for their help in the seawaters physicochemical parameters and trace metals analyses.
Funding
This work was supported by a fund from the Ministry of Scientific Research and Technology, University of Monastir, Tunisia (Research Laboratory Bioresources: Integrative Biology and Valorization, Higher Institute of Biotechnology of Monastir, Tunisia).
Author information
Authors and Affiliations
Contributions
CG: Methodology, Investigation, Formal analysis, Writing—original draft; MJ: Conceptualization, Methodology, Investigation; Formal analysis; ZB: Conceptualization, Methodology, Investigation; HG: Methodology, Investigation; JJ: Conceptualization, Methodology, Investigation; GT: Conceptualization, Methodology, Investigation. All authors participated in the final writing—review and editing of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable
Consent for publication
Not applicable
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Cinta Porte
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gharred, ., Jenzri, M., Bouraoui, Z. et al. Application of the Paracentrotus lividus sea-urchin embryo-larval bioassay to the marine pollution biomonitoring program in the Tunisian coast. Environ Sci Pollut Res 29, 5787–5797 (2022). https://doi.org/10.1007/s11356-021-16101-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-16101-9