Abstract
Triclosan (TCS) is an antibacterial and antifungal agent widely used in personal care products, and it has been frequently detected in the aquatic environment. In the present study, the acute toxicity of TCS to Daphnia magna, Photobacterium phosphoreum, Danio rerio, and Limnodrilus hoffmeisteri was assessed under different pH conditions. Generally, TCS was more toxic to the four aquatic organisms in acidic medium. The LC50 values for D. magna and D. rerio were smaller among the selected species, suggesting that D. magna and D. rerio were more sensitive to TCS. In addition, the oxidative stress-inducing potential of TCS was evaluated in Carassius auratus at three pH values. Changes of superoxide dismutase (SOD) and catalase (CAT) activity, glutathione (GSH) level, and malondialdehyde (MDA) content were commonly observed in all TCS exposure groups, indicating the occurrence of oxidative stress in the liver of C. auratus. The integrated biomarker response (IBR) index revealed that a high concentration of TCS induced great oxidative stress in goldfish under acidic condition. This work supplements the presently available data on the toxicity data of TCS, which would provide some useful information for the environmental risk assessment of this compound.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Triclosan (2,4,4′-trichloro-2′-hydroxydiphenyl ether, TCS, CAS No. 3380-34-5) is a synthetic broad-spectrum and multipurpose antibacterial agent that is widely used in consumer products such as soaps, toothpaste, deodorants, household cleaners, skin care creams, textiles, and even in children’s toys (Hua et al. 2005; Von Der Ohe et al. 2012; Bedoux et al. 2012). Owing to its widespread use, TCS has been detected in lakes, sediments, aquatic organisms, and even in human breast milk (Adolfsson-Erici et al. 2002; Asimakopoulos et al. 2014). For example, Ramaswamy et al. (2011) reported that the maximum concentration of TCS was 5.16 μg/L in Tamiraparani River in India. Fair et al. (2009) detected a TCS concentration of 0.0075 μg/L in estuarine waters in the USA. Previous studies showed that the concentration of TCS was in the range of 0.07–20 μg/L in wastewater treatment plant (WWTP) inflows and in the range of 80–100 μg/g in surface sediments near the source of industrial wastewater (Jungclaus et al. 1978; Lopez-Avila and Hites 1980; McAvoy et al. 2002; Lindström et al. 2002). Furthermore, TCS has been found in samples of human breast milk at concentrations as high as 2100 μg/kg lipid (Dayan 2007).
Because of the widespread occurrence of TCS, people are deeply concerned about its potential environmental risk. To date, many researchers have investigated the toxicity of TCS to aquatic organisms. Orvos et al. (2002) found that the 48-h EC50 of TCS for Daphnia magna was 0.39 mg/L, and the 96-h LC50 for Pimephales promelas and Lepomis macrochirus was 0.26 and 0.37 mg/L, respectively. Tamura et al. (2012) pointed out that the 96-h LC50 of TCS for Oryzias latipes was 210 μg/L. Wang et al. (2013) reported that the 96-h LC50 of TCS to C. auratus (weight 4.00 ± 0.80 g) was 1.839 mg/L. Moreover, Gao et al. (2015) revealed that TCS can inhibit the growth of Tetrahymena thermophila (24 h EC50, 1063 μg/L) and evoke a statistically significant increase in DNA damage. In addition, there are some other studies concerning the effects of chronic exposure to TCS on the reproduction, growth, thyroid hormone action, and detoxification system of aquatic organisms (Hinther et al. 2011; Marlatt et al. 2013; Peng et al. 2013; Hwang et al. 2014).
TCS is a chlorinated phenoxyphenol compound, which has a high octanol-water partitioning coefficient (log-KOW = 4.76) and a low water solubility (10 mg/L in distilled water at 20 °C) (Ciba Specialty Chemicals 2001; Dhillon et al. 2015). Due to the presence of a phenolic hydroxyl group, its water solubility increases at higher pH (e.g., 20 mg/L at pH 10) (Wu et al. 2015). Moreover, TCS (pKa = 8.0) may exist in different forms in pH-fluctuating water (Fig. S1) (Rowett et al. 2016). Price et al. (2010) reported that about 50% of the total TCS was present in neutral form in English surface waters. Rendal et al. (2011) showed that exposure pH could greatly affect the toxicity and bioaccumulation of an ionizable compound. The normal pH of natural waters falls in 6.0–9.0, but low pH can be observed in acidic lakes and in acid mine drainage polluted waters. However, most current toxicity tests with TCS have been conducted within a narrow pH range. However, the toxicity of TCS may be different when the water pH changes. Therefore, it is important to assess the toxicological effects of TCS on aquatic organisms at different pH values.
Many xenobiotics may exert potentially adverse effects and induce oxidative stress in organisms due to the generation of reactive oxygen species (ROS: O2•−, H2O2, •OH, and 1O2) (Van der Oost et al. 2003; Lushchak 2011). Under normal physiological conditions, these reactive substances can be effectively scavenged by the antioxidant defense system that consists of antioxidant enzymes such as superoxide dismutase (SOD) and catalase (CAT) and low molecular weight nonenzymatic antioxidants such as reduced glutathione (GSH) (Song et al. 2006). If the generation of ROS overwhelms the antioxidant capacity, lipids, proteins, and DNA could be oxidized by these ROS (Mates 2000). Damage to membrane lipids can result in the formation of malondialdehyde (MDA) in a process termed as lipid peroxidation. These oxidation-related biomarkers, including both enzymatic and molecular parameters, are frequently used to evaluate the effects of pollutant exposure on organisms (Van der Oost et al. 2003; Qu et al. 2014; Li et al. 2016). Previously, Wang et al. (2014) showed that TCS could inhibit CAT and peroxidase (POD) activities and cause significant oxidative stress in the snail Achatina fulica. Lin et al. (2010) reported that exposure to TCS could induce oxidative stress in the earthworm Eisenia fetida. However, these studies mainly focus on the oxidative stress-inducing potential of TCS in terrestrial animals, and to date little attention has been paid to aquatic organisms.
The aim of this study was to explore the effect of TCS toxicity on diverse aquatic organisms under different pH levels. The acute toxicity tests were performed under five pH values (pH 5.0, 6.0, 7.0 8.0, and 9.0) using Daphnia magna, Photobacterium phosphoreum, Limnodrilus hoffmeisteri, and Danio rerio as the test organisms. Moreover, SOD and CAT activity, GSH level, and malondialdehyde MDA content were determined to evaluate the oxidative stress in Carassius auratus livers after exposure to TCS at three pH values (pH 6.0, 7.5, and 9.0). The integral influences of different exposure goldfish groups were compared through integrated biomarker response (IBR) indices, since the IBR has been successfully used to assess the potential toxicity of environmental contaminants to different organisms (Damiens et al. 2007; Kim et al. 2010b). This study may provide some valuable data for the toxicological risk assessment of this compound.
Materials and methods
Chemicals and reagents
TCS (purity 97%) and 3-(N-morpholino)propanesulfonic acid (MOPS, purity 99.5%) were purchased from Aladdin® Reagent (Shanghai, China). The kits from Nanjing Jiancheng Bioengineering Institute (Nanjing, China) were used to analyze the oxidative stress biomarkers. The TCS stock solution was prepared in sodium hydroxide solution, and the concentration was measured as 1003.06 mg/L by a HPLC system (Agilent 1200) equipped with a variable-wavelength UV absorbance detector; 20 μL of sample was injected into an Agilent Zorbax 300SB-C18 column (4.6 × 150 mm, 5 μm) at 30 °C. The mobile phase consisting of methanol (80%) and water (20%) was eluted at 1.0 mL/min, and the detection wavelength was 230 nm. Information about quality control is detailed in Text S1.
Test organisms
D. magna were purchased from the Research Center for Eco-Environmental Sciences, Chinese Academy of Sciences (Beijing, China). They were cultured at 23 ± 1 °C under 16 h light:8 h dark photoperiod and fed with green algae (Scenedesumus obliquus) twice a day. Tap water that was filtered through activated carbon and aerated for more than 24 h was used as the culture medium, which was refreshed every 2 days. D. magna neonates (6–24 h old) after three generations of parthenogenesis were employed in the experiments. In order to ensure the accuracy of the results, a sensitivity test was conducted with potassium dichromate as a reference substance (OECD 2004).
The lyophilized luminescent bacteria P. phosphoreum was supplied by the Institute of Soil Science, Chinese Academy of Sciences (Nanjing, China); 0.5 g of lyophilized powder was reactivated in 0.5 mL of cold sterilized 2% NaCl solution and agitated thoroughly by a vortex stirrer. The working suspensions for subsequent experiments were achieved by adding 10 μL of the revived bacterial liquid into 2 mL of 3% NaCl solution at 25 °C.
L. hoffmeisteri (length 3–4 cm) were purchased from a commercial aquatic market (Jinan, China), and they were maintained at 23 ± 1 °C in an aquarium containing dechlorinated and aerated water with a 16-h:8-h light:dark cycle. After an acclimation period of 1 week, uniform-sized healthy and intact worms were chosen for the subsequent toxicity tests.
Adult D. rerio were purchased from a local supplier (Nanjing, China). They were kept in a flow-through system with a 14-h light/10-h dark cycle at 23 ± 0.5 °C. The fish were acclimatized in the laboratory for 10 days. They were fed with commercial food (main component: freeze-dried bloodworms) twice a day but were not fed for 24 h before the test. During acclimation, the mortality rate was < 0.05%.
C. auratus (body mass 30.64 ± 4.13 g) were obtained from a local aquatic market. Before the experiments, they were acclimatized for 10 days in aquaria with 150 L dechlorinated and aerated freshwater. The fish were fed daily with commercial fish pellets containing protein, carbohydrates, fat, inorganic salts, vitamins, etc., and food residues were removed within an hour after feeding. Prior to the experiments, they were starved for 24 h. The fish mortality was zero during the acclimatization. The quality parameters of water used for acclimation of all test organisms are listed in Table S1.
General procedure for pH adjustment in acute toxicity test methods
The acute toxicity of TCS to four aquatic organisms (D. magna, P. phosphoreum, D. rerio, and L. hoffmeisteri) were assessed at five pH values (5.0, 6.0, 7.0, 8.0, and 9.0). The pH of the exposure solutions was adjusted by HCl and NaOH solutions and stabilized by the buffering agent MOPS at a concentration of 3.58 mM (De Schamphelaere et al. 2004).
Acute immobilization test with D. magna
The acute toxicity test was performed according to Chinese national standard method (Ministry of Environmental Protection of the People’s Republic of China 1991a, b). Ten neonates were exposed to 20 mL test solutions in 50 mL beakers, and the treatments were replicated three times. Based on the preliminary experiments, a series of exposure concentrations (Table S2) were prepared to determine LC50 values at five pH points (pH 5.0, 6.0, 7.0, 8.0, and 9.0). During the experiment, daphnids were not fed. The number of immobilized daphnids was recorded after 24 h exposure. Prior to the experiments, preliminary tests were conducted to investigate the effect of pH-adjusted experimental water on D. magna. Results showed that the activities of the organisms were not changed after 24 h exposure to the water at each pH, suggesting that there was no significant adverse effect in this pH range.
Bioluminescence inhibition assay with P. phosphoreum
The acute toxicity test was conducted according to Chinese national standard method (Ministry of Environmental Protection of the People’s Republic of China 1995). Toxicity response of P. phosphoreum was assessed in terms of EC50, and 11 gradient concentrations were used to determine the EC50 values at each pH. The concentration series (Table S3), which were replicated seven times, were arranged in a 96-well (8 rows × 12 columns) black flat-bottom microplate (GRE, USA). In each test, 20 μL of bacterial suspension was added into the wells containing 180 μL of test solutions. After 15 min exposure at 25 °C, the bioluminescence of each test groups was measured by an multimode microplate reader (Infinite® M200 PRO, Tecan, Switzerland), and the detection wavelengths were in the range of 280–850 nm.
Acute toxicity test with D. rerio
The acute toxicity test was performed according to the National Standard Method of China (Ministry of Environmental Protection of the People’s Republic of China 1991a, b). Seven adult D. rerio were randomly selected and exposed to 1 L test solutions at each pH. The test solutions were obtained by diluting the stock with aerated water, and the pH was monitored and adjusted every 2 h. The mortality of D. rerio was calculated at 96 h. Fish were considered as dead if no visible breathing or no moving was observed when the tail was touched. During the experiment, the fish were not fed and dead individuals were immediately removed. The medium was renewed every day to maintain the concentration of TCS. Each experiment was replicated three times and the exposure concentrations (mg/L) of TCS are listed in Table S4.
Acute toxicity of TCS to L. hoffmeisteri
The acute toxicity of TCS to L. hoffmeisteri was assessed at five pH values. According to the preliminary experiment, the activities of L. hoffmeisteri were normal after 24 h exposure to the pH-adjusted experimental water. Three replicates of ten individuals were randomly selected for exposure to each 20 mL test solutions containing different concentrations of TCS (Table S5) at each pH value. The mortality of L. hoffmeisteri was calculated after 24 h. Worms were considered dead if the body whitened and no moving was observed when slightly touching it.
Oxidative stress in C. auratus after TCS exposure at different pH values
The exposure experiments were conducted in nine glass tanks, and each tank contained 30 L of test solution or dechlorinated tap water. Eight randomly selected fish in each group were exposed to 0, 0.05, and 0.5 mg/L of TCS at three typical pH values (6.0, 7.5, and 9.0) for 3 and 15 days. In the experiment, the fish were fed daily and were starved a day before biochemical analysis. Food residue was removed timely, and 50% water in the tanks was renewed every day to maintain the concentration of TCS. During the exposure period, the fish mortality was zero in all exposure groups; 2.0 mol/L HCl or 3.0 mol/L NaOH solution was used to achieve the final pH of 6.0 and 9.0, which was monitored and adjusted every 2 h during the course of the experiment.
At days 3 and 15, four fish in each treatment group were randomly sampled, sacrificed, and immediately dissected to obtain liver samples that were kept on ice. The liver tissues were rinsed with cold physiological saline (0.9% NaCl) to remove extraneous tissues and adherent blood, and dried on filter paper and weighed. Then, an Ultra Turrax homogenizer (IKA, Germany) was used to homogenize the liver in cold physiological saline (1:10, w/v). The homogenates were centrifuged (Eppendorf, Germany) at 4000×g for 15 min at 4 °C, and collected supernatants were used to analyze biochemical parameters. The protocols for fish maintenance, experimentation, and sacrifice were approved by the Ethics Committee of Nanjing University.
The protein concentration, SOD and CAT activities (unit, U/mg protein), GSH level (unit, μmol/g protein), and MDA content (unit, nmol/mg protein) in supernatants were analyzed using the diagnostic reagent kits. Protein was evaluated by the Coomassie Brilliant Blue dyeing technology according to the method described by Bradford (1976). The absorbance was measured at 595 nm with a UV-Vis spectrophotometer (Pucci, China). SOD activity was measured at 550 nm based on the inhibition of the reduction rate of cytochrome c by the superoxide radical (Flohe and Otting 1984). CAT activity was determined by monitoring the residual of H2O2 at 405 nm (Claiborne 1985). GSH level was measured at 412 nm following the method of Jollow et al. (1974). MDA content was estimated at 532 nm according to the method described by Luo et al. (2006), which was based on the reaction of the generated substrate and the thiobarbituric acid.
Calculations and statistics analyses
Acute toxicity data were expressed as LC50 or EC50 values, which were calculated by probit analysis with 95% confidence intervals. Data were presented as mean ± standard deviation (SD). Kolmogorov-Smirnov test and Levene test were used to examine the normality and homogeneity of variance of the data, respectively. One-way ANOVA (Duncan’s test) was applied to analyze the significant differences between the control and exposure groups at p < 0.05.
IBR is a method to summarize the biomarker responses into one general “stress index” (Beliaeff and Burgeot 2002). It was applied to assess the potential toxicity of TCS to goldfish in different exposure groups. The procedure of IBR calculation is described here briefly: (1) Data were calculated for mean and SD. (2) Data were standardized according to the formula Y = (X − m)/S, where Y is the standardized data, X is the data of each biomarker response, m is the mean data of the biomarker, and S is the standard deviation of the biomarker. (3) Z was calculated as Z = Y in the case of activation or Z = − Y in the case of inhibition. The minimum value (Min) was obtained. (4) S was calculated as S = Z + |Min|, where S > 0 and |Min| is the absolute value. (5) Calculation of star plot areas (A i ) by the formula \( {A}_i=\frac{S_i}{2}\sin \beta \left({S}_i\cos \beta +{S}_{i+1}\sin \beta \right) \), where \( \beta =\mathrm{arc}\ \tan \left(\frac{S_{i+1}\sin \alpha }{S_i-{S}_{i+1}\cos \alpha}\right) \), α is 2π/n radians, and S i is the obtained value of each biomarker. (6) The sum of the area A i gives the corresponding IBR value \( \mathrm{IBR}={\sum}_{i=1}^n{A}_i \), and n is the number of the biomarkers.
Results
The acute toxicity of TCS to the four aquatic species
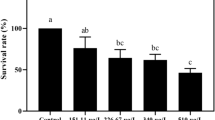
pH has a significant effect on the acute toxicity of TCS to the four aquatic species tested (Fig. 1); 24-h LC50 values of TCS to D. magna increased with pH in the studied pH range (Fig. 1a). When the pH increased from 5.0 to 6.0, the LC50 values increased slightly from 0.15 to 0.17 mg/L. At pH values higher than 6.0, there were significant increases (p < 0.05) in LC50 values, with maximum values to be observed at pH 9.0 (0.56 mg/L). The equation for the relationship between pH and LC50 was lgLC50 = − 0.1461 pH + 1.6031 (R2 = 0.9596), showing a positive relationship.
Similarly, the 15-min EC50 values obtained with P. phosphoreum increased with increasing pH (Fig. 1b). Significant differences (p < 0.05) in EC50 values were observed in the pH range of 5.0–8.0. Additionally, a positive relationship was found between EC50 data and pH values (EC50 = 0.1569 pH + 0.0592; R2 = 0.9838). The large regression coefficients demonstrated a strong correlation of the EC50 with pH.
In general, 96-h LC50 (mg/L) values of TCS to D. rerio increased with increasing pH (Fig. 1c). The three LC50 values were not significantly different in the pH region 7.0–8.0, but they showed a trend toward higher values than the data at pH 5.0 and 6.0. Additionally, a linear relationship was observed between the LC50 values and pH (LC50 = 0.1283 pH − 0.3558; R2 = 0.9889).
As for L. hoffmeisteri, a general increasing trend in 24-h LC50 values was observed with pH increasing from 5.0 to 9.0 (Fig. 1d). The LC50 was 1.57 mg/L at pH 5.0, which was significantly different from the value (1.86 mg/L) at pH 9.0. Additionally, the linear regression equation was determined as LC50 = 0.0686 pH + 1.2343 (R2 = 0.9837).
Oxidative stress in C. auratus
After 3 days of exposure, the groups of pH(6.0)-TCS(0.05), pH(6.0)-TCS(0.5) and pH(7.5)-TCS(0.5) showed a significant decrease (p < 0.05) in SOD activity as compared to the control group (Fig. 2a). After 15 days of exposure, significantly decreased SOD activity was observed in most of the treatment groups, i.e., pH(6.0)-TCS(0.05), pH(6.0)-TCS(0.5), pH(7.5)-TCS(0.05), pH(7.5)-TCS(0.5), pH(9.0)-TCS(0.05), and pH(9.0)-TCS(0.5) groups. However, the response at 15 days was less marked than at 3 days.
Compared with the control, only the pH(6.0)-TCS(0.5) group showed significantly increased CAT activity (p < 0.05) after 3 days of exposure (Fig. 2b). After 15 days, statistically significant differences from control (p < 0.05) were observed in the pH(6.0)-TCS(0.05), pH(6.0)-TCS(0.5), and pH(9.0)-TCS(0.5) groups.
After 3 days of exposure, there was no significant difference in GSH levels in fish liver after exposure to TCS among all exposure groups and the control (p < 0.05) (Fig. 2c). In contrast, the GSH level in the three treatment groups exposed to the high TCS concentration, i.e., pH(6.0)-TCS(0.5), pH(7.5)-TCS(0.5), and pH(9.0)-TCS(0.5), presented a highly significant increase by approximately 2-fold (p < 0.05) at 15 days.
MDA content in all exposure groups presented no statistically significant difference from control (p < 0.05) after 3 days of exposure (Fig. 2d). Following 15 days of exposure, MDA content was significantly increased (p < 0.05) in the high treatment groups of pH(6.0)-TCS(0.05), pH(6.0)-TCS(0.5), pH(7.5)-TCS(0.5), and pH(9.0)-TCS(0.5) as compared to the control group.
Integrated biomarker response
IBR values after 15 days of exposure were generally higher than the corresponding values after 3 days of exposure (Fig. 3). According to the IBR values, the most affected group could be ranked as follows: pH(6.0)-TCS(0.5) > pH(6.0)-TCS(0.05) > pH(9.0)-TCS(0.5) > pH(9.0)-TCS(0.05) > pH(7.5)-TCS(0.05) > pH(7.5)-TCS(0.5) > pH 6.0 > pH 9.0 > control(pH 7.5) for 3 days; pH(6.0)-TCS(0.5) > pH(9.0)-TCS(0.5) > pH(7.5)-TCS(0.5) > pH(6.0)-TCS(0.05) > pH(7.5)-TCS(0.05) > pH(9.0)-TCS(0.05) > pH 6.0 > pH 9.0 > control(pH 7.5) for 15 days.
Discussion
In general, the neutral and ionized forms of TCS have been shown to exhibit different toxic potencies to Ceriodaphnia dubia (Orvos et al. 2002). Rendal et al. (2011) reported that exposure pH could greatly affect the toxicity and bioaccumulation of ionizable compounds. In this work, TCS was more toxic to the four aquatic species (D. magna, P. phosphoreum, D. rerio, and L. hoffmeisteri) under low pH conditions (i.e., pH 5.0 and 6.0) than under high pH conditions, suggesting that pH can have a significant effect on the acute toxicity of TCS. The phenolic hydroxyl in TCS allows it to be present in two different forms, including the neutral form (TCS), which mainly exists at pH values < 8.0, and the ionized form (TCS−) mainly existing at pH values > 8.0 (Dhillon et al. 2015). Therefore, the solubility and existing form of TCS can be significantly changed along pH gradients (Orvos et al. 2002). The proportion of unionized form was greater than 90% when the solution pH is lower than 7 (Fig. S1). TCS is a highly lipid soluble compound, and its unionized form can readily pass through phospholipid membranes (Orvos et al. 2002; Roberts et al. 2014). Thus, TCS toxicity is mainly derived from the unionized form due to its greater bioavailability (Lyndall et al. 2010; Rowett et al. 2016). Currently, acidic pH (3.5~5.6) is most frequently reported in an acid rain area of south China (Dai et al. 2013). In these acid rain areas, TCS was present at different concentrations in surface waters (up to 0.478 μg/L) and sediments (up to 1.329 μg/L) (Zhao et al. 2013). Furthermore, Ramaswamy et al. (2011) reported that the concentration of TCS reached 5.16 μg/L in Tamiraparani River in India. Given this information, we should pay great attention to the potential environment risk of TCS at these locations. Overall, pH fluctuations in the natural environment will affect the acute toxicity of TCS to aquatic organisms (Peng et al. 2013).
Among the four test organisms, the LC50 values for D. magna were always the smallest at each pH, indicating that D. magna was the most sensitive to TCS. The LC50 values increased by almost 4-fold as the pH changed from 5.0 to 9.0, suggesting that pH can significantly affect the toxicity of TCS. Some researchers have found that the undissociated species of organic chemicals are more likely to pass through phospholipid membranes and increase the bioavailability or bioaccumulation potential (Orvos et al. 2002, Kim et al. 2010a). Orvos et al. (2002) assumed that only the unionized form was responsible for the toxicity of TCS to daphnids. These findings can explain our results well. Similar to D. magna, the toxicity of TCS to other organisms was also decreased as the pH of solution increased. As a single-cell organism, P. phosphoreum shows a higher tolerance for TCS. This was consistent with our previous findings that D. magna was more sensitive than P. phosphoreum to the toxicity of Cd and most of the benzophenone-type UV filters (Qu et al. 2013; Liu et al. 2015). The LC50 for D. rerio was slightly larger than D. magna, indicating that they were not as sensitive as D. magna. At the studies pH range, the LC50 was measured as 0.28–0.81 mg/L for D. rerio. These values agree well with the reported data of other researchers, which reported that the 96-h LC50 values of TCS to fish were 0.37, 0.26, and 0.34 mg/L for Lepomis macrochirus, Pimephales promelas, and D. rerio, respectively (Orvos et al. 2002; Oliveira et al. 2009). The 24-h LC50 values of TCS to L. hoffmeisteri were the largest among the test species, showing that L. hoffmeisteri was more resistant to TCS toxicity. The freshwater oligochaete L. hoffmeisteri have a high tolerance to harsh environmental conditions and organic or inorganic pollutants and can thus survive in a contaminated environment (Qu et al. 2016). Unlike the other three organisms, the LC50 values of L. hoffmeisteri changed slowly in the pH range of 5.0–9.0. This may be due to the fact that the organisms have the capacity to keep the internal pH relatively stable; thus, the amount of unionized TCS will not change significantly, leading to small variations in LC50 values. Overall, the toxicity of TCS was largely dependent on environmental pH and exposed species. The high sensitivity of D. magna to TCS may guarantee its applicability as the first screening method in assessing environmental samples.
The pH of aquatic systems can be increased or decreased as a result of various anthropogenic activities, including agriculture, urbanization, industry, and mining. According to Wood (2001), low and high water pH per se are potentially toxic to aquatic organisms. In this work, C. auratus showed high tolerance to TCS in slightly acidic or alkaline water environments as no fish died during the experiment. As suggested by Rodriguez-Ariza et al. (1993), chronic exposure to low concentrations of contaminants is often more likely to produce sublethal responses (e.g., oxidative stress) than acute lethality. A variety of environmental contaminants can trigger the generation of ROS in exposed organisms, including TCS (Lin et al. 2010; Wang et al. 2014). The inducible antioxidant defense system that consists of antioxidant enzymes (e.g., SOD and CAT) and numerous nonenzymatic antioxidants (e.g., GSH) becomes vital for scavenging ROS and reducing oxidative stress. These antioxidants can be induced as a compensatory response to a mild oxidative stress. However, excess ROS produced by xenobiotics may overwhelm the detoxifying or antioxidant mechanism, leading to the suppression of antioxidant enzyme activities (Zhang et al. 2004). The experimental results showed that both enzymatic and nonenzymatic antioxidants in C. auratus responded rapidly to TCS-mediated stress, which was related to the concentration of pollutant and the pH of exposure water. Some significant changes in oxidative stress biomarkers were observed as the pH of the test medium increased from 6.0 to 9.0, indicating that the antioxidant system was sensitive to pH fluctuations.
SOD and CAT play a vital role in scavenging ROS, and they are usually considered the first line of defense against oxidative stress (Pandey et al. 2003). SOD can catalyze the dismutation of superoxide to H2O and H2O2, while CAT is responsible for decomposing H2O2 to H2O and O2 (Peng et al. 2013). In this study, SOD activity presented significant decreases in most TCS-treated groups after 3 and 15 days of exposure to TCS. In a previous study, we found that the excessive ROS production overwhelmed the capacity of the antioxidant system, leading to an obvious reduction in SOD activity in C. auratus exposed to polyfluorinated dibenzo-p-dioxins (PFDDs) (Li et al. 2016). The inhibitive response of SOD activity may suggest that high concentration of TCS could inhibit detoxification of O2− by Microcystis aeruginosa cells (Huang et al. 2016). Therefore, in the present study, the decreased SOD activity might be attributed to the excessive production of ROS after TCS exposure and the consequent accumulation of oxidative substances in the cells. By contrast, CAT activity was significantly increased in some TCS-treated groups. Similar to this work, some previous studies (Lin et al. 2010; Wang et al. 2014) have also reported that the activity of CAT in organisms may be induced by low concentrations of TCS. The increase in CAT activity after exposure to low concentrations of TCS might result from increased synthesis of the enzyme to eliminate ROS, which can be regarded as an adaptive response to counteract the impact of increased ROS generation provoked by toxicants (McCord and Fridovich 1969).
GSH, one primary cytosolic nonprotein thiol, can eliminate free radicals by the sulfhydryl group (Van der Oost et al. 2003). In addition to directly scavenging ROS-like oxyradicals, GSH can work as a cofactor for other antioxidant enzymes including glutathione transferase, which contributes to the removal of certain chemicals and other reactive species from cells (Zhang et al. 2008). Therefore, an increase in GSH levels is considered an indicator of detoxification activity of goldfish (Dautremepuits et al. 2009). In the present study, a significant increase in GSH levels was observed in C. auratus after exposure to TCS. Consistent with this work, Huang et al. (2016) showed that TCS exposure (500 ng/L) significantly increased GSH levels in M. aeruginosa in a dose-dependent manner. He et al. (2015) also observed a significant increase in GSH level in the livers of C. auratus exposed to tetrabromobisphenol A (TBBPA). The significant increase in GSH in our study suggested the upregulation of enzymes participating in GSH synthesis (Stephensen et al. 2002), and the availability of GSH is very important for C. auratus in defending against the oxidative stress caused by TCS (Peng et al. 2013).
MDA content is one of the products of cell membrane lipid peroxidation. The oxidative reaction between ROS and membrane lipids may lead to lipid peroxidation of membrane and damage in cell function. Moreover, the reactive aldehydes produced during lipid peroxidation can cause serious damage to intra- and extracellular targets by diffusing from the original site of radical production (Esterbauer et al. 1990). Thus, MDA is often used as an indicator to show the extent of cell membrane damage (Livingstone et al. 1990). In the present study, significantly increased MDA content was observed after TCS exposure. According to Lin et al. (2010), increased MDA content indicated the occurrence of oxidative damage in TCS-exposed Eisenia fetida. Wang et al. (2014) also reported that the increased MDA content in Achatina fulica was due to oxidative stress induced by TCS exposure. Therefore, we considered that exposure to TCS caused lipid peroxidation and oxidative damage in fish.
In the present study, the changes of oxidation-related biomarkers mentioned above confirmed the oxidative stress effects of TCS. Although cellular mechanisms responsible for TCS-induced oxidative stress are seldom reported (Ciniglia et al., 2005), several possible mechanisms have been proposed before. TCS exposure could destroy structures of cell membranes, resulting in damaged functional integrity (Villalaín et al. 2001); the mitochondria, the activity of which depends on membrane integrity, would be potentially an important target of this effect (Brookes et al. 2004). Another explanation involves the interaction with a wide range of proteins, including proteins belonging to the stress response and to calcium binding (Riva et al. 2012). Moreover, several studies have suggested that TCS is able to alter/elevate intracellular free Ca2+ levels and induce ROS formation in animal and human cell lines (Ahn et al. 2008; Cherednichenko et al. 2012; Palmer et al. 2012), which may also account for the observed oxidative stress responses.
Because different exposure protocols presented completely different biomarker responses, IBR was employed to process four biomarker responses into one general stress index. It can provide a simple method to evaluate the potential effect of TCS to aquatic organisms. Generally, the higher the IBR value is, the more stressful the environment is. From the IBR calculation results, we can conclude the following points. Firstly, the IBR for an exposure group changed with pH, suggesting that pH has an influence on the toxicity of TCS. Specifically, TCS caused the largest damage to the antioxidant defense system in acidic medium. Secondly, at a given pH, the IBR value for low concentration treatment groups was always less than that for high concentration treatment groups, implying oxidative stress became more serious with the increasing TCS concentrations. Therefore, oxidative stress becomes a greater risk factor at high concentrations of TCS under acidic condition in goldfish.
Conclusions
The present study revealed that TCS exposure could cause acute lethality and oxidative stress in aquatic organisms under different pH conditions. Acute toxicity results demonstrated that TCS was more toxic to L. hoffmeisteri, P. phosphoreum, D. rerio, and D. magna under acidic conditions. The LC50 values of D. magna and D. rerio were smaller among the four organisms, indicating that D. magna and D. rerio were more sensitive to TCS. The oxidative stress study showed that SOD and CAT activity, GSH level, and MDA content in C. auratus were altered by TCS exposure, indicating that TCS enhanced the production of ROS and induced oxidative stress in the fish. Moreover, TCS was more toxic to the goldfish in an acidic environment. Overall, this work supplements the presently available data on the toxicity data of TCS, which would provide some useful information for the environmental risk assessment of this compound.
References
Adolfsson-Erici M, Pettersson M, Parkkonen J, Sturve J (2002) Triclosan, a commonly used bactericide found in human milk and in the aquatic environment in Sweden. Chemosphere 46(9–10):1485–1489. https://doi.org/10.1016/S0045-6535(01)00255-7
Ahn KC, Zhao B, Chen J, Cherednichenko G, Sanmarti E, Denison MS, Lasley B, Pessah IN, Kültz D, Chang DPY, Gee SJ, Hammock BD (2008) In vitro biologic activities of the antimicrobials triclocarban, its analogs, and triclosan in bioassay screens: receptor-based bioassay screens. Environ Health Perspect 116(9):1203–1210. https://doi.org/10.1289/ehp.11200
Asimakopoulos AG, Thomaidis NS, Kannan K (2014) Widespread occurrence of bisphenol A diglycidyl ethers, p-hydroxybenzoic acid esters (parabens), benzophenone type-UV filters, triclosan, and triclocarban in human urine from Athens, Greece. Sci Total Environ 470–471:1243–1249
Bedoux G, Roig B, Thomas O, Dupont V, Le Bot B (2012) Occurrence and toxicity of antimicrobial triclosan and by-products in the environment. Environ Sci Pollut Res Int 19(4):1044–1065
Beliaeff B, Burgeot T (2002) Integrated biomarker response: a useful tool for ecological risk assessment. Environ Toxicol Chem 21(6):1316–1322. https://doi.org/10.1002/etc.5620210629
Bradford MM (1976) A rapid and sensitive method for quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Brookes PS, Yoon Y, Robotham JL, Anders MW, Sheu SS (2004) Calcium, ATP, and ROS: a mitochondrial love-hate triangle. Am J Physiol Cell Physiol 287:817–833
Cherednichenko G, Zhang R, Bannister RA, Timofeyev V, Li N, Fritsch EB, Feng W, Barrientos GC, Schebb NH, Hammock BD, Beam KG, Chiamvimonvat N, Pessah IN (2012) Triclosan impairs excitation-contraction coupling and Ca2+ dynamics in striated muscle. Proc Natl Acad Sci U S A 109(35):14158–14163. https://doi.org/10.1073/pnas.1211314109
Ciba Specialty Chemicals (2001) General information on chemical, physical and microbiological properties of Irgasan DP300, Irgacare MP and Irgacide LP10. Brochure 2520. Publication AgB2520e.02. Basel, Switzerland
Ciniglia C, Cascone C, Giudice RL, Pinto G, Pollio A (2005) Application of methods for assessing the geno- and cytotoxicity of triclosan to C. ehrenbergii. J Hazard Mater 122:227–232
Claiborne A (1985) Catalase activity. In: Greenwald RA (ed) Handbook of methods for oxygen radical research. CRC, Boca Raton, pp 283–284
Dai ZM, Liu XM, Wu JJ, Xu JM (2013) Impacts of simulated acid rain on recalcitrance of two different soils. Environ Sci Pollut Res 20(6):4216–4224. https://doi.org/10.1007/s11356-012-1288-z
Damiens G, Gnassia-Barelli M, Loques F, Romeo M, Salbert V (2007) Integrated biomarker response index as a useful tool for environmental assessment evaluated using transplanted mussels. Chemosphere 66:574–583
Dautremepuits C, Marcogliese DJ, Gendron AD, Fournier M (2009) Gill and head kidney antioxidant processes and innate immune system responses of yellow perch (Perca flavescens) exposed to different contaminants in the St. Lawrence River, Canada. Sci Total Environ 407(3):1055–1064. https://doi.org/10.1016/j.scitotenv.2008.10.004
Dayan AD (2007) Risk assessment of triclosan [Irgasan®] in human breast milk. Food Chem Toxicol 45(1):125–129. https://doi.org/10.1016/j.fct.2006.08.009
De Schamphelaere KAC, Heijerick DG, Janssen CR (2004) Comparison of the effect of different pH buffering techniques on the toxicity of copper and zinc to Daphnia magna and Pseudokirchneriella subcapitata. Ecotoxicology 13(7):697–705. https://doi.org/10.1007/s10646-003-4429-9
Dhillon GS, Kaur S, Pulicharla R, Brar SK, Cledon M, Verma M, Surampalli RY (2015) Triclosan: current status, occurrence, environmental risks and bioaccumulation potential. Int J Envirn Res Public Health 12(5):5657–5684. https://doi.org/10.3390/ijerph120505657
Esterbauer H, Zollner H, Schaur RJ (1990) Aldehydes formed by lipid peroxidation: mechanisms of formation, occurrence, and determination. In: Vigo-Pelfrey C (ed) Membrane lipid oxidation. CRC, Boca Raton, pp 239–268
Fair PA, Lee HB, Adams J, Darling C, Pacepavicius G, Alaee M, Bossart GD, Henry N, Muir D (2009) Occurrence of triclosan in plasma of wild Atlantic bottlenose dolphins (Tursiops truncatus) and in their environment. Environ Pollut 157(8–9):2248–2254. https://doi.org/10.1016/j.envpol.2009.04.002
Flohe L, Otting F (1984) Superoxide dismutase assays. Method Enzym 105:93–104. https://doi.org/10.1016/S0076-6879(84)05013-8
Gao L, Yuan T, Cheng P, Bai QF, Zhou CQ, Ao JJ, Wang WH, Zhang HM (2015) Effects of triclosan and triclocarban on the growth inhibition, cell viability, genotoxicity and multixenobiotic resistance responses of Tetrahymena thermophila. Chemosphere 139:434–440. https://doi.org/10.1016/j.chemosphere.2015.07.059
He Q, Wang XH, Sun P, Wang ZY, Wang LS (2015) Acute and chronic toxicity of tetrabromobisphenol A to three aquaticspecies under different pH conditions. Aquat Toxicol 164:145–154. https://doi.org/10.1016/j.aquatox.2015.05.005
Hinther A, Bromba CM, Wulff JE, Helbing CC (2011) Effects of triclocarban, triclosan, and methyl triclosan on thyroid hormone action and stress in frog and mammalian culture systems. Environ Sci Technol 45(12):5395–5402. https://doi.org/10.1021/es1041942
Hua WY, Bennett ER, Letcher RJ (2005) Triclosan in waste and surface waters from the upper Detroit River by liquid chromatography–electrospray–tandem quadrupole mass spectrometry. Environ Int 31(5):621–630. https://doi.org/10.1016/j.envint.2004.10.019
Huang XL, Tu YN, Song CF, Li TC, Lin J, Wu YH, Liu JT, Wu CX (2016) Interactions between the antimicrobial agent triclosan and the bloom-forming cyanobacteria Microcystis aeruginosa. Aquat Toxicol 172:103–110. https://doi.org/10.1016/j.aquatox.2016.01.002
Hwang J, Suh SS, Chang M, Yun Park S, Ryu TK, Lee S, Lee TK (2014) Effects of triclosan on reproductive prarmeters and embryonic development of sea urchin, Strongylocentrotus nudus. Ecotoxicol Environ Saf 100:148–152. https://doi.org/10.1016/j.ecoenv.2013.10.029
Jollow DJ, Mitchell JR, Zampagilone N, Gilete JR (1974) Bromobenzene-induced liver necrosis: protective role of glutathione and evidence for 3,4-bromo benzene oxide as the hepatotoxic metabolite. Pharmacology 11(3):151–169. https://doi.org/10.1159/000136485
Jungclaus GA, Lopez-Avila V, Hites RA (1978) Organic compounds in an industrial wastewater: a case study of their environmental impact. Environ Sci Technol 12:88–96
Kim J, Park J, Kim PG, Lee C, Choi K, Choi K (2010a) Implication of global environmental changes on chemical toxicity effect of water temperature, pH, and ultraviolet B irradiation on acute toxicity of several pharmaceuticals in Daphnia magna. Ecotoxicology 19(4):662–669. https://doi.org/10.1007/s10646-009-0440-0
Kim WK, Lee SK, Jung J (2010b) Integrated assessment of biomarker responses in common carp (Cyprinus carpio) exposed to perfluorinated organic compounds. J Hazard Mater 180(1–3):395–400. https://doi.org/10.1016/j.jhazmat.2010.04.044
Li CG, Qin L, Qu RJ, Sun P, Wang ZY (2016) Responses of antioxidant defense system to polyfluorinated dibenzo-p-dioxins (PFDDs) exposure in liver of freshwater fish Carassius auratus. Ecotoxicol Environ Saf 126:170–176. https://doi.org/10.1016/j.ecoenv.2015.12.036
Lin DS, Zhou QX, Xie XJ, Liu Y (2010) Potential biochemical and genetic toxicity of triclosan as an emerging pollutant on earthworms (Eisenia fetida). Chemosphere 81(10):1328–1333. https://doi.org/10.1016/j.chemosphere.2010.08.027
Lindström A, Buerge IJ, Poiger T, Bergqvist PA, Müller MD, Buser HR (2002) Occurrence and environmental behavior of the bactericide triclosan and its methyl derivative in surface waters and in wastewater. Environ Sci Technol 36(11):2322–2329. https://doi.org/10.1021/es0114254
Liu H, Sun P, Liu HX, Yang SG, Wang LS, Wang ZY (2015) Acute toxicity of benzophenone-type UV filters for Photobacterium phosphoreum and Daphnia magna: QSAR analysis, interspecies relationship and integrated assessment. Chemosphere 135:182–188. https://doi.org/10.1016/j.chemosphere.2015.04.036
Livingstone DR, Martinez PG, Michel X, Narbonne JF, O’Hara S, Ribera D, Winston GW (1990) Oxyradical production as a pollution-mediated mechanism of toxicity in the common mussel, Mytilus edulis L., and other molluscs. Funct Ecol 4(3):415–424. https://doi.org/10.2307/2389604
Lopez-Avila V, Hites RA (1980) Organic compounds in an industrial wastewater. Their transport into sediments. Environ Sci Technol 14(11):1382–1390. https://doi.org/10.1021/es60171a007
Luo Y, Su Y, Lin RZ, Shi HH, Wang XR (2006) 2-Chlorophenol induced ROS generation in fish Carassius auratus based on the EPR method. Chemosphere 65(6):1064–1073. https://doi.org/10.1016/j.chemosphere.2006.02.054
Lushchak VI (2011) Environmentally induced oxidative stress in aquatic animals. Aquat Toxicol 101(1):13–30. https://doi.org/10.1016/j.aquatox.2010.10.006
Lyndall J, Fuchsman P, Bock M, Barber T, Lauren D, Leigh K, Perruchon E, Capdevielle M (2010) Probabilistic risk evaluation for triclosan in surface water, sediments, and aquatic biota tissues. Integr Environ Asses 6(3):419–440. https://doi.org/10.1897/IEAM_2009-072.1
Marlatt VL, Veldhoen N, Lo BP, Bakker D, Rehaume V, Vallee K, Haberl M, Shang D, Van Aggelen GC, Skirrow RC, Elphick JR, Helbing CC (2013) Triclosan exposure alters postembryonic development in a Pacific tree frog (Pseudacris regilla) amphibian metamorphosis assay (TREEMA). Aquat Toxicol 126:85–94. https://doi.org/10.1016/j.aquatox.2012.10.010
Mates JM (2000) Effects of antioxidant enzymes in the molecular control of reactive oxygen species toxicology. Toxicology 153(1–3):83–104. https://doi.org/10.1016/S0300-483X(00)00306-1
McAvoy DC, Schatowitz B, Jacob M, Hauck A, Eckhoff WS (2002) Measurement of triclosan in wastewater treatment systems. Environ Toxicol Chem 21(7):1323–1329
McCord JM, Fridovich I (1969) Superoxide dismutase an enzymic function for erythrocuprein (hemocuprein). J Biol Chem 244(22):6049–6055
Ministry of Environmental Protection of the People’s Republic of China (1991a) Water quality-determination of the acute toxicity of substances to Daphnia (D. magna straus). GB/T 13266-1991
Ministry of Environmental Protection of the People’s Republic of China (1991b) Water quality-determination of the acute toxicity of substances to a freshwater fish (Brachydanio rerio Hamilton-Buchanan). GB/T 13267-1991
Ministry of Environmental Protection of the People’s Republic of China (1995) Water quality—determination of the acute toxicity—luminescent bacteria test. GB/T 15441-1995
OECD (2004) OECD guideline for testing of chemicals—Daphnia sp., acute immobilisation test 202. In: Effects on biotic systems. OECD, Section 2
Oliveira R, Domingues I, Grisolia CK, Soares AMVM (2009) Effects of triclosan on zebrafish early-life stages and adults. Environ Sci Pollut Res 16(6):679–688. https://doi.org/10.1007/s11356-009-0119-3
Orvos DR, Versteeg DJ, Inauen J, Capdevielle M, Rosthenstein A, Cunningham V (2002) Aquatic toxicity of triclosan. Environ Toxicol Chem 21(7):1338–1349. https://doi.org/10.1002/etc.5620210703
Palmer RK, Hutchinson LM, Burpee BT, Tupper EJ, Pelletier JH, Kormendy Z, Hopke AR, Malay ET, Evans BL, Velez A, Gosse JA (2012) Antibacterial agent triclosan suppresses RBL-2H3 mast cell function. Toxicol Appl Pharmacol 258(1):99–108. https://doi.org/10.1016/j.taap.2011.10.012
Pandey S, Parvez S, Sayeed I, Haque R, Bin-Hafeez B, Raisuddin S (2003) Biomarkers of oxidative stress: a comparative study of river Yamuna fish Wallago attu (Bl. & Schn.) Sci Total Environ 309(1–3):105–115. https://doi.org/10.1016/S0048-9697(03)00006-8
Peng Y, Luo Y, Nie XP, Liao W, Yang YF, Ying GG (2013) Toxic effects of triclosan on the detoxification system and breeding of Daphnia magna. Ecotoxicology 22(9):1384–1394. https://doi.org/10.1007/s10646-013-1124-3
Price OR, Williams RJ, Egmond RV, Wilkinson MJ, Whelan MJ (2010) Predicting accurate and ecologically relevant regional scale concentrations of triclosan in rivers for use in higher-tier aquatic risk assessments. Environ Int 36(6):521–526. https://doi.org/10.1016/j.envint.2010.04.003
Qu RJ, Wang XH, Feng MB, Li Y, Liu HX, Wang LS, Wang ZY (2013) The toxicity of cadmium to three aquatic organisms (Photobacterium phosphoreum, Daphnia magna and Carassius auratus) under different pH levels. Ecotoxicol Environ Saf 95:83–90. https://doi.org/10.1016/j.ecoenv.2013.05.020
Qu RJ, Feng MB, Wang XH, Qin L, Wang C, Wang ZY, Wang LS (2014) Metal accumulation and oxidative stress biomarkers in liver of freshwater fish Carassius auratus following in vivo exposure to waterborne zinc under different pH values. Aquat Toxicol 150:9–16. https://doi.org/10.1016/j.aquatox.2014.02.008
Qu RJ, Liu JQ, Wang LS, Wang ZY (2016) The toxic effect and bioaccumulation in aquatic oligochaete Limnodrilus hoffmeisteri after combined exposure to cadmium and perfluorooctane sulfonate at different pH values. Chemosphere 152:496–502. https://doi.org/10.1016/j.chemosphere.2016.03.024
Ramaswamy BR, Shanmugam G, Velu G, Rengarajan B, Larsson DG (2011) GC-MS analysis and ecotoxicological risk assessment of triclosan, carbamazepine and parabens in Indian rivers. J Hazard Mater 186:1586–1593
Rendal C, Kusk KO, Trapp S (2011) Optimal choice of pH for toxicity and bioaccumulation studies of ionizing organic chemicals. Environ Toxicol Chem 30(11):2395–2406. https://doi.org/10.1002/etc.641
Riva C, Cristoni S, Binelli A (2012) Effects of triclosan in the freshwater mussel Dreissena polymorpha: a proteomic investigation. Aqua Toxicol 118–119:62–71
Roberts J, Price OR, Bettles N, Rendal C, Egmond RV (2014) Accounting for dissociation and photolysis: a review of the algal toxicity of triclosan. Environ Toxicol Chem 33(11):2551–2559. https://doi.org/10.1002/etc.2710
Rodriguez-Ariza A, Peinado J, Pueyo C, Lopez-Barea J (1993) Biochemical indicators of oxidative stress in fish from polluted littoral areas. Can J Fish Aquat Sci 50(12):2568–2573. https://doi.org/10.1139/f93-280
Rowett CJ, Hutchinson TH, Comber SDW (2016) The impact of natural and anthropogenic dissolved organic carbon (DOC), and pH on the toxicity of triclosan to the crustacean Gammarus pulex (L.) Sci Total Environ 565:222–231. https://doi.org/10.1016/j.scitotenv.2016.04.170
Song SB, Xu Y, Zhou BS (2006) Effects of hexachlorobenzene on antioxidant status of liver and brain of common carp (Cyprinus carpio). Chemosphere 65(4):699–706. https://doi.org/10.1016/j.chemosphere.2006.01.033
Stephensen E, Sturve J, Főrlin L (2002) Effects of redox cycling compounds on glutathione content and activity of glutathione-related enzymes in rainbow trout liver. Comp Biochem Physiol 133C:435–442
Tamura I, Kagota KI, Yasuda Y, Yoneda S, Morita J, Nakada N, Kameda Y, Kimura K, Tatarazako N, Yamamoto H (2012) Ecotoxicity and screening level ecotoxicological risk assessment of five antimicrobial agents: triclosan, triclocarban, resorcinol, phenoxyethanol and p-thymol. J Appl Toxicol 33:1222–1229
Van der Oost R, Beyer J, Vermeulen NPE (2003) Fish bioaccumulation and biomarkers in environmental risk assessment: a review. Environ Toxicol Phar 13(2):57–149. https://doi.org/10.1016/S1382-6689(02)00126-6
Villalaín J, Mateo CR, Aranda FJ, Shapiro S, Micol V (2001) Membranotropic effects of the antibacterial agent triclosan. Arch Biochem Biophys 390(1):128–136. https://doi.org/10.1006/abbi.2001.2356
Von Der Ohe PC, Schmitt-Jansen M, Slobodnik J, Brack W (2012) Triclosan—the forgotten priority substance? Environ Sci Pollut Res Int 19(2):585–591. https://doi.org/10.1007/s11356-011-0580-7
Wang XN, Liu ZT, Yan ZG, Zhang C, Wang WL, Zhou JL, Pei SW (2013) Development of aquatic life criteria for triclosan and comparison of the sensitivity between native and non-native species. J Hazard Mater 260:1017–1022. https://doi.org/10.1016/j.jhazmat.2013.07.007
Wang XN, Liu ZT, Wang WH, Yan ZG, Zhang C, Wang WL, Chen LH (2014) Assessment of toxic effects of triclosan on the terrestrial snail (Achatina fulica). Chemosphere 108:225–230. https://doi.org/10.1016/j.chemosphere.2014.01.044
Wood CM (2001) Toxic responses of the gill. In: Schlenk DS, Benson WH (eds) Target organ toxicity in marine and freshwater teleosts, vol 1. Taylor and Francis, London, pp 1–89. https://doi.org/10.1201/9781315109244-2
Wu WJ, Hu YY, Guo Q, Yan J, Chen YC, Cheng JH (2015) Sorption/desorption behavior of triclosan in sediment–water–rhamnolipid systems: effects of pH, ionic strength, and DOM. J Hazard Mater 297:59–65. https://doi.org/10.1016/j.jhazmat.2015.04.078
Zhang JF, Shen H, Wang XR, Wu JC, Xue YQ (2004) Effects of chronic exposure of 2, 4-dichlorophenol on the antioxidant system in liver of freshwater fish Carassius auratus. Chemosphere 55(2):167–174. https://doi.org/10.1016/j.chemosphere.2003.10.048
Zhang X, Yang FX, Zhang XL, Xu Y, Liao T, Song SB, Wang JW (2008) Induction of hepatic enzymes and oxidative stress in Chinese rare minnow (Gobiocypris rarus) exposed to waterborne hexabromocyclododecane (HBCDD). Aquat Toxicol 86(1):4–11. https://doi.org/10.1016/j.aquatox.2007.07.002
Zhao JL, Zhang QQ, Chen F, Wang L, Ying GG, Liu YS, Yang B, Zhou LJ, Liu S, Su HC, Zhang RJ (2013) Evaluation of triclosan and triclocarban at river basin scale using monitoring and modeling tools: implications for controlling of urban domestic sewage discharge. Water Res 47(1):395–405. https://doi.org/10.1016/j.watres.2012.10.022
Funding
This research was financially supported by the National Natural Science Foundation of China (No. 21577063, 21377051) and the Major Science and Technology Program for Water Pollution Control and Treatment of China (No. 2012ZX07506-001).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Markus Hecker
Electronic supplementary material
ESM 1
(DOCX 512 kb)
Rights and permissions
About this article
Cite this article
Li, C., Qu, R., Chen, J. et al. The pH-dependent toxicity of triclosan to five aquatic organisms (Daphnia magna, Photobacterium phosphoreum, Danio rerio, Limnodrilus hoffmeisteri, and Carassius auratus). Environ Sci Pollut Res 25, 9636–9646 (2018). https://doi.org/10.1007/s11356-018-1284-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-1284-z