Abstract
Salt stress is a main abiotic stress that limits agricultural productivity in many parts of the world. To investigate whether poly-γ-glutamic acid (γ-PGA) can alleviate the negative effects of salt stress on wheat, a foliar application of 400 mg/L γ-PGA was applied to wheat seedlings, which were then subjected to 150 mM NaCl. Our results showed that after application of γ-PGA, the plant height, the plant weight, and the antioxidant enzymes including superoxide dismutase, peroxidase, and catalase were significantly increased compared with the treatment of 150 mM NaCl alone. Meanwhile, γ-PGA application also resulted in high accumulation of K+ and decreased storage of Na+ in wheat leaves. These results suggest that γ-PGA treatment may improve salt tolerance of wheat by diminishing ionic imbalances and enhancing antioxidant capacity. Our results indicate that exogenous γ-PGA could alleviate the damage caused by salt stress.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Wheat is the most important stable crop in the world and provides almost 20% of the total necessary calories for the world’s population (Hajihashemi et al. 2007). Salinity stress has been reported to disturb the integrity of cell membranes in wheat and barley and cause an increase in membrane permeability and decrease in lipid partiality (Mansour et al. 1993). Currently, salinity has become a widespread problem; presumably, by the year 2050, 50% of arable lands will be salinized (Wang et al. 2003). Salt stress is a major factor limiting growth and crop production in the world (Bohnert and Jensen 1996). High salinity induces ion and osmotic pressure imbalance (Xiong et al. 2002). The accumulation of toxic concentrations of salt in old leaves causes ion toxicity and accelerates the senescence of leaves and finally results in leaf death (Katerji et al. 2003). Osmotic stress restricts water availability and leaf growth in plants (Tuna et al. 2008). High salinity also induces several morphological, physiological, and metabolic responses, leading to gradual peroxidation of lipids and antioxidant enzyme inactivation (Garg and Manchanda 2009).
In order to reduce oxidative damage, plants have produced an enzymatic and non-enzymatic defense system to alleviate oxidative stress. The active oxygen-scavenging enzymes include superoxide dismutase (SOD), catalase (CAT), and peroxidase (POD) (Li et al. 2011; Ma et al. 2012). SOD is a major scavenger of toxic O2 · radicals and is subsequently converted to hydrogen peroxide (H2O2) by peroxidase (Mutlu et al. 2009; Triantaphylides and Havaux 2009). POD disintegrates H2O2 by oxidation of co-substrates such as phenolic compounds or antioxidants, whereas CAT decomposes H2O2 and O2 (Noctor and Foyer 1998). In contrast, non-enzyme antioxidants include glutathione, ascorbate, proline, and flavonoids (Li et al. 2011). It is therefore necessary to find methods for reducing the adverse effects of salt stress and increasing wheat productivity.
Poly-γ-glutamic acid (γ-PGA) is a water-soluble, biodegradable polypeptide (Xu et al. 2013); it is a biopolymer composed of D-glutamic acid and L-glutamic acid monomers connected by amide linkages via Bacillus subtilis fermentation (Birrer et al. 1994; Shih and Van 2001). γ-PGA is used in many fields, such as food, medicine, cosmetics, and agriculture (Shih and Van 2001). γ-PGA is able to hold as much water as hydrogels, allowing its use as a soil conditioner for crop fields in dry deserts. It is reported that γ-PGA can greatly raise the dry weight of the roots of both cucumber seedlings (Wang et al. 2008) and Chinese cabbage (Xu et al. 2014). γ-PGA application on wheat resulted in increased spike number, 1000 seed weight, seed number per spike, and dry matter production (Xu et al. 2013). γ-PGA treatment increased the chlorophyll content of tomato leaves during the flowering stage (Jiang et al. 2014).
Many studies have reported that γ-PGA plays an important role in plant growth and regulation. However, there is meager information regarding the effects of γ-PGA on seedling growth under salt stress. It was reported that γ-PGA could enhance the tolerance of Brassica napus L. by promoting proline accumulation under salt stress (Lei et al. 2016). In this study, wheat seedlings were used to assess the effect of foliar application of γ-PGA on the growth of seedlings subjected to salt stress. This study shows that foliar application of γ-PGA alleviated the negative effects of salt stress on wheat by modulating ion balance and the antioxidant system. These results provide an insight into the role of γ-PGA in the alleviation of salt stress in wheat seedlings and information on the potential use of γ-PGA to regulate plant growth in salinity soil.
Materials and methods
Plant material, growth, and treatment conditions
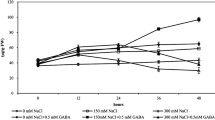
Wheat seeds were disinfected with 70% ethanol for 1 min and then rinsed with distilled water for several times. The seeds were swamped in water for 12 h and moved to wet filter paper to germinate in the darkness in a temperature-controlled chamber at 30 °C for 24 h. The germinated seeds were transferred to a vermiculite culture with standard Hoagland solution. The solution was changed every 3 days. The seedlings were kept under a 16/8-h day/night cycle with day/night temperatures of 24/20 °C. After growing 7 days, wheat seedlings with two leaves were randomly divided into four treatment groups (Shalata and Neumann 2001; Wang 2017; Xie et al. 2015): (1) seedlings sprayed with distilled water alone, (2) seedlings sprayed with 400 mg/L γ-PGA, (3) seedlings sprayed with γ-PGA for 12 h before addition of 150 mM NaCl, and (4) seedlings treated with 150 mM NaCl. Each pot contained 12 seedlings, and three pots were analyzed for each treatment at each time point. Seedlings were sprayed until leaves were completely wetted (about 30 mL for each pot). All the experiments were repeated three times. Based on our pre-experiments, the concentrations of 150 mM NaCl and 400 mg/L γ-PGA were selected for our experiments (Figs. S1 and S2).
Biomass assay
After 7 days of NaCl treatment, the seedlings had three leaves, and the shoot of wheat seedlings were collected for determination of fresh and dry weight. Dry weight was weighted after drying the seedlings at 105 °C for 5 min and then at 80 °C for 48 h.
Measurement of relative conductivity, chlorophyll content, and lipid peroxidation (MDA)
The method described by Barcia et al. (2014) was used to analyze relative conductivity. Chlorophyll was extracted using 80% acetone under the darkness for 24 h, and the chlorophyll concentration was calculated according to Arnon (1949). The MDA content was measured using the method of Madhava Rao and Sresty (2000).
Antioxidant enzyme extraction and analysis
Fresh leaves (0.2 g) were finely grounded in 2 mL extraction buffer (50 mM phosphate buffer (pH = 7.8)) containing 1 mM EDTA and 2% (w/v) PVP at chilled condition. After centrifugation at 10,000×g for 20 min at 4 °C, the crude extraction was transferred to another tube and used for enzyme determination. The SOD activity was determined following the method of Giannopolitis and Ries (1977), and POD and CAT were analyzed basing on the produce described by Cakmak and Marschner (1992) and Bergmeyer (1970), respectively. The activity of antioxidant enzymes was determined on fresh weight basis.
Assay of H2O2 content
H2O2 content was analyzed according to Chang and Kao (1998) with some modifications. In our experiment, we changed the concentration of sulfuric titanium from 2% (w/v) to 20% (w/v).
Histochemical staining using diaminobenzidine tetrahydrochloride and nitroblue tetrazolium
Diaminobenzidine tetrahydrochloride (DAB) staining was performed using wheat leaves placed in 1 mg/mL DAB (pH = 3.8) solution for 8 h in the darkness. Chlorophyll was removed by boiling in 95% ethanol for 15 min. Observations were made under a light microscope (Thordal-Christensen et al. 1997).
Nitroblue tetrazolium (NBT) staining was used to determine superoxide anion content. Wheat leaves were submerged in 10 mM PBS buffer (pH = 7.8) containing 1 mg/mL NBT; the mixture was placed under a vacuum for 20 min, and chlorophyll was removed by boiling in 95% ethanol (Garrett et al. 1990). The leaves were then preserved in 50% glycerinum for observation.
Measurement of Na+ and K+ concentration
Elemental analysis was according to the method described by Zarcinas et al. (1987).
Results
Effect of foliar application of γ-PGA on growth parameters and chlorophyll content in wheat seedlings under salt stress
To investigate the effects of exogenous γ-PGA in wheat, the biomass of wheat seedlings under salt stress was analyzed. The data suggested that salt decreased the fresh and dry weights by 21 and 20%, respectively, compared with the control. However, the fresh and dry weights only decreased by 7.7 and 5.4%, respectively, under γ-PGA treatment with salt stress (Fig. 1a,b). Salt stress also significantly reduced wheat height, and exogenous application of γ-PGA alleviated this effect. However, the chlorophyll content in wheat seedlings subjected to salt stress was not significantly different from that of the control (Fig. 1c).
Effect of exogenous γ-PGA (400 mg/L) on the regulation of fresh weight (a), dry weight (b), and chlorophyll content (c) in wheat seedlings under NaCl (150 mM) treatment for 7 days. The different treatments included distilled water (Control), 150 mM salt stress (S), 400 mg/L γ-PGA (P), and 150 mM salt stress + 400 mg/L γ-PGA (P + S). Bars are means ± standard error of three replicates. Different letters indicate significant differences among treatments at the 0.05 level based on Duncan’s test
Effect of exogenous γ-PGA on lipid peroxidation and relative conductivity in wheat seedlings under salt stress
The extent of lipid peroxidation is indicated by the MDA content. Salt stress induced MDA content, and this induction was significantly mitigated by treatment with 400 mg/L γ-PGA, although γ-PGA treatment alone did not have a significant effect compared with the control (Fig. 2b). The relative conductivity increased quickly under salt stress, and this effect was diminished by exogenous application of 400 mg/L γ-PGA (Fig. 2a).
Effect of exogenous γ-PGA (400 mg/L) on the regulation of relative conductivity (a) and MDA content (b) in wheat seedling under NaCl (150 mM) treatment for 7 days. The different treatments included distilled water (Control), 150 mM salt stress (S), 400 mg/L γ-PGA (P), and 150 mM salt stress + 400 mg/L γ-PGA (P + S). Bars are means ± standard error of three replicates. Different letters indicate significant differences among treatments at the 0.05 level based on Duncan’s test
Effect of γ-PGA on antioxidant enzyme activity in wheat seedlings under salt stress
The effect of exogenous γ-PGA on the activities of SOD, POD, and CAT in leaves is shown in Fig. 3. Salt stress significantly decreased SOD (Fig. 3a), POD (Fig. 3b), and CAT (Fig. 3c) activities in leaves compared with those of the control. In contrast, exogenous application of γ-PGA increased these activities under salt stress. As seen in Fig. 3, treatment with γ-PGA alone notably enhanced the activities of SOD, POD, and CAT compared with those of the control.
Effect of exogenous γ-PGA (400 mg/L) on the regulation of SOD activity (a), POD activity (b), and CAT activity (c) in wheat seedling under NaCl (150 mM) treatment for 7 days. The different treatments included distilled water (Control), 150 mM salt stress (S), 400 mg/L γ-PGA (P), and 150 mM salt stress + 400 mg/L γ-PGA (P + S). Bars are means ± standard error of three replicates. Different letters indicate significant differences among treatments at the 0.05 level based on Duncan’s test
H2O2 content and DAB and NBT staining
H2O2 content was significantly increased, rising sharply at 12 h, under salt stress; however, H2O2 content was decreased after application of γ-PGA. There was no significant difference between γ-PGA alone treatment and the control (Fig. 4). DAB and NBT staining showed the same results. The red staining represents DAB polymerization products, indicating H2O2 formation. The blue staining indicates NBT polymerization, revealing O2 − as the product. Little staining was observed in the control and γ-PGA-treated leaves, and stronger reddish brown and blue staining appeared in wheat leaves under salt stress, with more reddish and blue coloration seen in those treated with 150 mM NaCl alone.
Effect of exogenous γ-PGA (400 mg/L) on the regulation of H2O2 content (a) and DAB staining in different hours after treatment (left) and NBT staining in different hours after treatment (right) in wheat seedling under NaCl (150 mM) treatment for 7 days. The different treatments included distilled water (Control), 150 mM salt stress (S), 400 mg/L γ-PGA (P), and 150 mM salt stress + 400 mg/L γ-PGA (P + S). Bars are means ± standard error of three replicates
Effect of γ-PGA on Na+ and K+ contents in wheat leaves under salt stress
Salt stress promoted absorption of Na+ and decreased absorption of K+ in the leaves (Fig. 5a, b). Foliar application of γ-PGA inhibited the absorption of Na+ in the leaves of wheat seedlings. However, γ-PGA had no effect on the accumulation of K+. Salt stress resulted in an obvious decrease in the K+/Na+ ratio, which was enhanced by exogenous γ-PGA application in wheat seedlings (Fig. 5c).
Effect of exogenous γ-PGA (400 mg/L) on the regulation of Na+ content (a), K+ content (b), and K+/Na+ (c) in wheat seedling leaves under NaCl (150 mM) treatment for 7 days. The different treatments included distilled water (Control), 150 mM salt stress (S), 400 mg/L γ-PGA (P), and 150 mM salt stress + 400 mg/L γ-PGA (P + S). Bars are means ± standard error of three replicates. Different letters indicate significant differences among treatments at the 0.05 level based on Duncan’s test
Discussion
Salt stress decreases the metabolic activity of plant cells, indicated by inhibition of their growth. In this study, we found that the growth of wheat seedlings was significantly inhibited by 150 mM salt. However, exogenous application of γ-PGA decreased the side effects of salt stress on the growth of wheat seedlings. It has been reported that treatment with 500 mg/L γ-PGA protected rice seedlings from the adverse effects of Cd by enhancing shoot length (Kotabin et al. 2015). Chlorophyll content is widely used as an index of abiotic tolerance in plants. While our results showed that 7 days of salt stress did not affect chlorophyll content, Chen et al. (2014) reported that under salt treatment, the chlorophyll content in wheat seedlings was decreased sharply after 21 days. This discordance may be attributed to the different application durations of the salt treatments used in each study.
Plants under salt stress use the antioxidant system to resist the adverse effects of salt stress on their growth. MDA is one of the final products of lipid peroxidation in plant cell membranes and a marker of oxidative damage in plants exposed to stress (Meloni et al. 2003). Under cold stress, γ-PGA protected rape seedlings by reducing MDA content (Lei et al. 2015). In our study, a significant increase in lipid peroxidation was observed under salt stress. Nevertheless, exogenous application of γ-PGA prevented lipid peroxidation and protected cells from salt stress. Salt-induced inhibition on plant growth could also be attributed to accumulation of ROS, which can damage macromolecules and cellular structures. It was reported that elevated expression of antioxidant system components has been linked to the decreased oxidative damage under different stresses, because high antioxidase activity can eliminate ROS quickly and protect plants from stress damage (Wang et al. 2010). Our results suggested that under salt stress, SOD, POD, and CAT activities were decreased significantly, while γ-PGA alleviated the deleterious effects of the salt stress. Moriyama et al. (2015) revealed that cross-linked catechol-PGA nanoparticles have H2O2 scavenging activity. γ-PGA can also serve as a capping agent to scavenge intracellular ROS formed (Stevanović et al. 2011; Stevanović et al. 2012). It has also been reported that γ-PGA has potential for use in the food, cosmetic, and biomedical industries in the development of novel products with radical scavenging activity (Lee et al. 2014). This study revealed that H2O2 content was decreased in wheat seedlings when γ-PGA was applied under salt stress. Histochemical staining supported this result. After DAB and NBT staining, we found that H2O2 and O2 − were diminished after application of γ-PGA. We therefore presumed that γ-PGA acts as a H2O2 scavenging agent to alleviate salt stress by modulation of the antioxidant system.
Salt-induced inhibition of plant growth could also be attributed to ion toxicity. Plant cells under salt stress show increased toxic level of cellular Na+ and restricted absorption of macroelement K+. This causes a rapid reduction in the cytosolic K+/Na+ ratio and to salt sensitivity. Reducing the Na+ accumulate in leaves is thought as an important mechanism of salt stress (Tester and Davenport 2003). Our results showed that the Na+ concentration in wheat seedlings was increased markedly under 150 mM NaCl and that the K+ concentration was decreased. After foliar application of γ-PGA, the accumulation of Na+ was inhibited and K+ uptake stimulated in the leaves of salt-stressed wheat seedlings. This is consistent with reports that γ-PGA can slow down the decreasing rate of K+ uptake under stress and promote K+ accumulation. γ-PGA was also reported to be a sorbent for increasing Ca and N absorption (Tanimoto et al. 2001; Xu et al. 2013). These results suggest that γ-PGA alleviates ion-specific toxicity by decreasing cellular accumulation of Na+.
Conclusion
In conclusion, salt stress reduced the growth of wheat seedlings, and 400 mg/L γ-PGA alleviated salinity damage by increasing SOD, POD, and CAT activities and decreasing ion toxicity in wheat seedlings. These results indicate that exogenously applied γ-PGA can protect wheat seedlings from the adverse effects of salt stress.
References
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris, Plant Physiol 24:1
Barcia RA, Pena LB, Zawoznik MS, Benavides MP, Gallego SM (2014) Osmotic adjustment and maintenance of the redox balance in root tissue may be key points to overcome a mild water deficit during the early growth of wheat. Plant Growth Regul 74:107–117. doi:10.1007/S10725-014-9902-3
Bergmeyer HU (1970) Methoden der enzymatischen Analyse, vol 432. Verlag Chemie, Weinheim
Birrer GA, Cromwick AM, Gross RA (1994) Gamma-poly(glutamic acid) formation by Bacillus-Licheniformis 9945a—physiological and biochemical studies. Int J Biol Macromol 16:265–275. doi:10.1016/0141-8130(94)90032-9
Bohnert HJ, Jensen RG (1996) Metabolic engineering for increased salt tolerance—the next step—comment. Aust J Plant Physiol 23:661–666
Cakmak I, Marschner H (1992) Magnesium deficiency and high light intensity enhance activities of superoxide dismutase, ascorbate peroxidase, and glutathione reductase in bean leaves. Plant Physiol 98:1222–1227. doi:10.1104/Pp.98.4.1222
Chang CJ, Kao CH (1998) H2O2 metabolism during senescence of rice leaves: changes in enzyme activities in light and darkness. Plant Growth Regul 25:11–15. doi:10.1023/A:1005903403926
Chen D, Yin L, Deng X, Wang S (2014) Silicon increases salt tolerance by influencing the two-phase growth response to salinity in wheat (Triticum aestivum L.). Acta Physiol Plant 36:2531–2535
Garg N, Manchanda G (2009) ROS generation in plants: boon or bane? Plant Biosyst 143:81–96. doi:10.1080/11263500802633626
Garrett IR, Boyce BF, Oreffo ROC, Bonewald L, Poser J, Mundy GR (1990) Oxygen-derived free-radicals stimulate osteoclastic bone resorption in rodent bone in vitro and in vivo. J Clin Invest 85:632–639. doi:10.1172/Jci114485
Giannopolitis CN, Ries SK (1977) Superoxide dismutases I. Occurrence in higher plants. Plant Physiol 59:309–314
Hajihashemi S, Kiarostami K, Saboora A, Enteshari S (2007) Exogenously applied paclobutrazol modulates growth in salt-stressed wheat plants. Plant Growth Regul 53:117–128. doi:10.1007/S10725-007-9209-8
Jiang Y, Liu L, Luo G, Zhang Z, Davidmark J, Huang Y (2014) Effect of γ-PGA coated urea on N-release rate and tomato growth. Wuhan University Journal of Natural Sciences 19:335–340
Katerji N, van Hoorn JW, Hamdy A, Mastrorilli M (2003) Salinity effect on crop development and yield, analysis of salt tolerance according to several classification methods, Agr water. Manage 62:37–66. doi:10.1016/S0378-3774(03)00005-2
Kotabin N, Tahara Y, Issakul K, Chunhachart O (2015) Potential of γ-polyglutamic acid for cadmium toxicity alleviation in rice, World Academy of Science, Engineering and Technology, International Journal of Biological, Biomolecular, Agricultural, Food and Biotechnological. Engineering 9:915–919
Lee NR et al (2014) Improved production of poly-gamma-glutamic acid by Bacillus subtilis D7 isolated from Doenjang, a Korean traditional fermented food, and its antioxidant activity. Appl Biochem Biotech 173:918–932. doi:10.1007/S12010-014-0908-0
Lei P et al (2015) Effect of poly (γ-glutamic acid) on the physiological responses and calcium signaling of rape seedlings (Brassica napus L.) under cold stress. J Agric Food Chem 63:10399–10406
Lei P, ZQ X, Liang JF, Luo XH, Zhang YX, Feng XH, Xu H (2016) Poly(gamma-glutamic acid) enhanced tolerance to salt stress by promoting proline accumulation in Brassica napus L. Plant Growth Regul 78:233–241. doi:10.1007/s10725-015-0088-0
Li JT, Qiu ZB, Zhang XW, Wang LS (2011) Exogenous hydrogen peroxide can enhance tolerance of wheat seedlings to salt stress. Acta Physiol Plant 33:835–842. doi:10.1007/S11738-010-0608-5
Ma LJ et al (2012) Alleviation of exogenous oligochitosan on wheat seedlings growth under salt stress. Protoplasma 249:393–399. doi:10.1007/S00709-011-0290-5
Madhava Rao KV, Sresty TVS (2000) Antioxidative parameters in the seedlings of pigeonpea (<i> Cajanus cajan </i> (L.) Millspaugh) in response to Zn and Ni stresses. Plant Sci 157:113–128
Mansour MMF, Leestadelmann OY, Stadelmann EJ (1993) Salinity stress and cytoplasmic factors—a comparison of cell permeability and lipid partiality in salt-sensitive and salt-resistant cultivars and lines of Triticum aestivum and Hordeum vulgare. Physiol Plantarum 88:141–148. doi:10.1034/J.1399-3054.1993.880120.X
Meloni DA, Oliva MA, Martinez CA, Cambraia J (2003) Photosynthesis and activity of superoxide dismutase, peroxidase and glutathione reductase in cotton under salt stress. Environ Exp Bot 49:69–76. doi:10.1016/S0098-8472(02)00058-8
Moriyama M, Uyama H, van der Vlies AJ, Hasegawa U (2015) Cross-linked catechol-bearing poly (γ-glutamic acid) self-aggregates with antioxidant activity. Colloid Polym Sci 293:1245–1251
Mutlu S, Atici O, Nalbantoglu B (2009) Effects of salicylic acid and salinity on apoplastic antioxidant enzymes in two wheat cultivars differing in salt tolerance. Biol Plantarum 53:334–338. doi:10.1007/S10535-009-0061-8
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Phys 49:249–279. doi:10.1146/Annurev.Arplant.49.1.249
Shalata A, Neumann PM (2001) Exogenous ascorbic acid (vitamin C) increases resistance to salt stress and reduces lipid peroxidation. J Exp Bot 52:2207–2211
Shih IL, Van YT (2001) The production of poly-(gamma-glutamic acid) from microorganisms and its various applications. Bioresource Technol 79:207–225. doi:10.1016/S0960-8524(01)00074-8
Stevanović M, Kovačević B, Petković J, Filipič M, Uskoković D (2011) Effect of poly-α, γ, L-glutamic acid as a capping agent on morphology and oxidative stress-dependent toxicity of silver nanoparticles. Int J Nanomedicine 6:2837
Stevanović MM, Škapin SD, Bračko I, Milenković M, Petković J, Filipič M, Uskoković DP (2012) Poly (lactide-co-glycolide)/silver nanoparticles: synthesis, characterization, antimicrobial activity, cytotoxicity assessment and ROS-inducing potential. Polymer 53:2818–2828
Tanimoto H, Mori M, Motoki M, Torii K, Kadowaki M, Noguchi T (2001) Natto mucilage containing poly-γ-glutamic acid increases soluble calcium in the rat small intestine. Biosci Biotechnol Biochem 65:516–521
Tester M, Davenport R (2003) Na+ tolerance and Na+ transport in higher plants. Ann Bot-London 91:503–527. doi:10.1093/Aob/Mcg058
Thordal-Christensen H, Zhang Z, Wei Y, Collinge DB (1997) Subcellular localization of H2O2 in plants. H2O2 accumulation in papillae and hypersensitive response during the barley—powdery mildew interaction. Plant J 11:1187–1194
Triantaphylides C, Havaux M (2009) Singlet oxygen in plants: production, detoxification and signaling. Trends Plant Sci 14:219–228. doi:10.1016/J.Tplants.2009.01.008
Tuna AL, Kaya C, Higgs D, Murillo-Amador B, Aydemir S, Girgin AR (2008) Silicon improves salinity tolerance in wheat plants. Environ Exp Bot 62:10–16. doi:10.1016/J.Envexpbot.2007.06.006
Wang Z (2017) Cadmium stress tolerance in wheat seedlings induced by ascorbic acid was mediated by NO signaling pathways. Ecotoxicology & Environmental Safety 135:75–81
Wang WX, Vinocur B, Altman A (2003) Plant responses to drought, salinity and extreme temperatures: towards genetic engineering for stress tolerance. Planta 218:1–14. doi:10.1007/S00425-003-1105-5
Wang QJ, Chen SW, Zhang JB, Sun M, Liu ZD, ZI Y (2008) Co-producing lipopeptides and poly-gamma-glutamic acid by solid-state fermentation of Bacillus subtilis using soybean and sweet potato residues and its biocontrol and fertilizer synergistic effects. Bioresource Technol 99:3318–3323. doi:10.1016/J.Biortech.2007.05.052
Wang Y, Li JL, Wang JZ, Li ZK (2010) Exogenous H2O2 improves the chilling tolerance of manilagrass and mascarenegrass by activating the antioxidative system. Plant Growth Regul 61:195–204. doi:10.1007/S10725-010-9470-0
Xie YJ et al (2015) Heme-heme oxygenase 1 system is involved in ammonium tolerance by regulating antioxidant defence in Oryza sativa. Plant Cell Environ 38:129–143. doi:10.1111/pce.12380
Xiong LM, Schumaker KS, Zhu JK (2002) Cell signaling during cold, drought, and salt stress. Plant Cell 14:S165–S183. doi:10.1105/Tpc.000596
Xu Z, Wan C, Xu X, Feng X, Xu H (2013) Effect of poly (gamma-glutamic acid) on wheat productivity, nitrogen use efficiency and soil microbes. J Soil Sci Plant Nut 13:744–755. doi:10.4067/S0718-95162013005000071
Xu ZQ, Lei P, Feng XH, Xu XJ, Liang JF, Chi B, Xu H (2014) Calcium involved in the poly(gamma-glutamic acid)-mediated promotion of Chinese cabbage nitrogen metabolism. Plant Physiol Bioch 80:144–152. doi:10.1016/j.plaphy.2014.03.036
Zarcinas BA, Cartwright B, Spouncer LR (1987) Nitric acid digestion and multi-element analysis of plant material by inductively coupled plasma spectrometry. Communications in Soil Science & Plant Analysis 18:131–146
Acknowledgements
This work was funded by the National Natural Science Foundation of China (grant no. 31501236) and the Fundamental Research Funds for the Central Universities (KJQN201639).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Guo, Z., Yang, N., Zhu, C. et al. Exogenously applied poly-γ-glutamic acid alleviates salt stress in wheat seedlings by modulating ion balance and the antioxidant system. Environ Sci Pollut Res 24, 6592–6598 (2017). https://doi.org/10.1007/s11356-016-8295-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-8295-4