Abstract
Nutrient removal is among the most valuable ecosystem services provided by marshes and is often a stated goal of coastal restoration projects. However, the removal capacity of constructed marshes is potentially affected by several site-specific and design factors, such as marsh platform elevation, slope, sediment type, initial planting density and wave climate. Here, the main and interactive effects of these factors on the capacity of constructed marshes to remove nitrate from runoff was explored in field experiments at sites protected from and exposed to waves. At both sites, three experimental blocks were established, each with 24 treatment combinations of factors in experimental flumes: two platform elevations (high and low), two slopes (steep and shallow), two sediment types (coarse and fine grain), and three initial planting densities (0%, 50% and 100% cover). Nutrient rich (KNO3) groundwater solution was fed through the marsh rhizosphere using subsurface diffusers. The relative effects of treatment combinations were then assessed by analyzing porewater NOx concentrations with ANOVA models. None of the treatment combinations had any observable effect on porewater NOx concentrations at the exposed site. However, both sediment type and planting density were significant main effects at the protected site with the lowest NOx concentrations found in flumes with fine sediments and initially planted. These results confirm that design factors can have large implications on nutrient dynamics of constructed marshes in areas protected from waves and that wave energy can substantially reduce the influence of these design factors.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Alarming rates of coastal wetland loss and their associated natural benefits (i.e., ecosystem services; Mehvar et al. 2019) have been driving wetland restoration, conservation, and enhancement efforts. Constructed wetlands and their natural counterparts offer many ecosystem services including habitat provisioning for wildlife and fisheries (Gittman et al. 2016), storm surge protection (Barbier 2015; Gedan et al. 2011; Van Slobbe et al. 2013), attenuation of waves and subsequent erosion (Bilkovic et al. 2019; McConchie and Toleman 2003), nutrient removal via various processes (e.g., plant uptake or biologically mediated transformations; Fisher and Acreman 2004; Kleinhuizen and Mortazavi 2018; Sparks et al. 2015), and cultural benefits (Gupta and Foster 1973; Nassauer 2004). As such, one or several of these benefits are often stated goals of many restoration and conservation projects (Yozzo et al. 1996; Zedler 1996). Increased nutrient concentrations and its associated impacts (e.g., harmful algal blooms, hypoxia, etc.; Dodds 2006; Rabalais et al. 2002) have made runoff nutrient removal one of the top priorities for wetland projects in the Northern Gulf of Mexico.
Current best practices for designing marsh construction projects consider several environmental and material characteristics, including: platform elevation (i.e., position along an elevation gradient as it relates to inundation frequency; Morris et al. 2002) and slope (i.e., as it relates to water flow; e.g., Spieles and Mitsch 1999), sediment characteristics (i.e., sediment particle size and organic matter content; Bergamaschi et al. 1997; Coops and Velde 1996; Davis et al. 2004; Lucas and Greenway 2008), and wave climate (i.e., the frequency and magnitude of waves; Roland and Douglass 2005; NOAA 2015). These characteristics are not only important considerations for marsh establishment, but are also relevant to the capacity of marshes to remove nutrients, especially nitrogen (Fisher and Acreman 2004). Initial planting density may be less important for nitrogen removal as marshes develop (Sparks et al. 2015; Kleinhuizen and Mortazavi 2018), and therefore may present an opportunity for reducing project costs (Sparks et al. 2013). While some studies have attempted to quantify the relative influence of one or more of these factors (e.g., Sparks et al. 2015; Martin et al. 2020), none have investigated how combinations of all of these factors affect nitrogen removal. Further, combinations of these factors frequently occur in nature, as a part of site-specific project design, or could be expected in the future with sea level rise. Therefore, better understanding of the main and interactive effects of sediment type, platform elevation, slope and initial planting density on the nitrogen removal capacity of constructed marshes in wave exposed and protected sites is needed to maximize the cost–benefit of projects.
Sediment characteristics that could influence nitrogen removal include organic matter content (Davis et al. 2004; Howes and Goehringer 1994), surface area and porosity of sediments (Bergamaschi et al. 1997). However, these are often not considered during project design. During microbial metabolism, microbial communities rely heavily on the availability of organic compounds in soils and the exchange of various compounds, including reduced and oxidized forms of nitrogen (i.e., ammonium and nitrate, respectively; Davis et al. 2004). Thus, the limited organic matter associated with sandy sediments may not be sufficient to support removal pathways. The greater pore spaces associated with sandy sediments may also facilitate greater flow of solutes through the rhizosphere, including nitrate. Sediment type is also linked to varying microbial community structure (Yamamoto and Lopez 1985), diversity (Jesus et al. 2009) and productivity (Sinsabaugh and Findlay 1995) which further influences nitrogen removal in marshes (Wetzel 2001). While sediment type in constructed marshes vary by location, sediment amendments that may be required with certain site conditions (e.g., severely eroded sites, higher wave energy) rarely use fine grain sediments. Instead, sediment amendments typically use heavier, sandier sediment as they are less susceptible to erosion from waves and currents (Woodroffe 2002).
Vegetation presence and density also has direct and indirect effects on nitrogen removal. Plant growth can increase with increasing nutrient availability, resulting in the greater abundance of above- and below-ground materials (Fox et al. 2012; Howes et al. 1986; Morris et al. 2013) and the greater incorporation of nutrients into plant tissues (Morris et al. 2013; Silvan et al. 2004; Sparks and Cebrian 2015). The presence of robust vegetation may also slow groundwater flow through the marsh (Sparks et al. 2014), increasing residence time and subsequent removal of nitrogen by marsh vegetation and soil microbes. Plants also indirectly facilitate the removal or transformation of excess nitrogen through their influence on other soil microbial processes (Brix 1997). For example, radial oxygen loss from plant roots (Brix 1997; Tobias and Neubauer 2019) favors the conversion of reduced nitrogen (e.g., ammonium) to oxidized forms (e.g., nitrate) that are more actively removed by plants or by microbes via denitrification. Decaying plant materials also serve as an important carbon source in microbial metabolism that can further facilitate nutrient removal (Howes and Goehringer 1994). However, Sparks et al. (2013, 2015) demonstrated that lower and cost-saving initial planting densities of salt marshes could be as effective at removing nutrients as higher planting densities.
Elevation and slope are also potentially important considerations for constructed wetland projects because of the role each play in soil conditions. Platform elevation is related to plant growth via direct and indirect effects on oxygen availability (e.g., varying hydroperiod; Armstrong 1979; Mendelssohn and McKee 1988). Prolonged inundation at lower platform elevations decreases oxygen availability resulting from plant and microbial metabolism and the slow diffusion of oxygen in water (Armstrong 1979). Hydric soils affect plant nutrient removal in two important ways: by increasing competition for nitrogen with soil microbes (Engelaar et al. 2000) and by the accumulation of phyto-toxins (e.g., sulfides) that limit plant growth (Reddy and DeLaune 2008). Thus, nutrient additions to marshes positioned at lower elevations may reverse the negative effects on plant growth associated with hydric soils (Mendelssohn and McKee 1988) and may result in enhanced nutrient utilization. However, at higher platform elevations, the effects of aerobic soil conditions on nutrient removal capacity are mixed. On the one hand, aerobic conditions at higher platform elevations maximize plant growth (Morris et al. 2002), increase nutrient uptake, and favor oxidized nutrient species (e.g., nitrate) that may be easily exported from the marsh (Kleinhuizen and Mortazavi 2018). On the other hand, inundation under both low and high platform elevations is expected to change as sea level rise continues in the future (IPCC 2019). Likewise, though commonly amended or specified in restoration designs, platform slope is rarely investigated within the context of nutrient removal by plants in coastal wetlands. However, slope could influence the structure and subsequent function of salt marshes (Wigand et al. 2003). Slope has a strong influence on groundwater flow rate which may affect nutrient removal in wetlands (Spieles and Mitsch 1999). Indeed, Sparks et al. (2014) found little nitrate removal in fast flowing sandy soils. Similarly, if gentler slopes reduce water flow, the effect could be an increase in the residence time of nutrients which could allow for further uptake by plants and microbes (Tobias et al. 2001). However, as with platform elevation amendments, it remains unclear if slope amendments improve nutrient removal capacity to levels justifying the increased project costs that would be required.
Finally, the effect of waves on the nutrient removal capacity of constructed wetlands is not fully understood. Wave climate has some influence on the establishment of plants and overall plant growth, such that plant growth is generally maximized in areas experiencing smaller wave heights (Keddy 1985; Roland and Douglass 2005; Silinski et al. 2018). As waves increase in frequency or magnitude, their effects on plant growth and persistence could effectively limit the nutrient removal capacity of constructed marshes. However, some aspects of waves may actually improve soil conditions. For example, wave breaking may increase oxygenation of the rhizosphere (e.g., Hosoi et al. 1977) and encourage plant investment in defenses including greater production of above- and/or below-ground parts for anchoring or energy attenuation (Feagin et al. 2009). These could result in greater nutrient uptake by plants or enhanced oxygenation of the rhizosphere via roots. Alternatively, as wave turbulence increases oxygen availability, it may also facilitate the conversion of nutrients to those more mobile in solution (e.g., NH4 to NO3) that are subsequently removed from the system via uptake by plants and microorganisms, leaching or denitrification.
To evaluate the main and interactive effects of the factors mentioned above on the nitrogen removal capacity of constructed marshes, we manipulated sediment type (i.e., coarse and fine), vegetation density (i.e., 0%, 50%, 100%), marsh platform elevation (high and low) and slope (i.e., steep and gentle) in experimental flumes featuring different treatment combinations at sites with and without wave exposure. We then simulated upland runoff events by pumping a groundwater nutrient solution through the marsh rhizosphere to evaluate the removal capacity of the treatment combinations. Following previous findings investigating some or all of the treatments examined here, we hypothesized that platform elevation and slope would not have a significant effect on nutrient removal and that plots with fine sediments and initially planted at either 50% or 100% density would remove the greatest amount of the groundwater solution as compared to other combinations.
Methods
Study site description
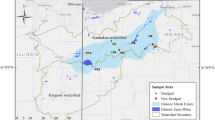
Two sites were selected within Weeks Bay National Estuarine Research Reserve (WBNERR) in Fairhope, Alabama, USA (Fig. 1) to allow for the comparison of protected and exposed shorelines: one along Fish River (i.e., the exposed site) and another within an adjacent, previously dredged canal complex (i.e., the protected site; Fig. 1). Marsh species composition within this mesohaline (salinity ≤ 5) reach of Fish River and its adjacent canals is dominated by the study species, Juncus roemerianus, with sub-dominant species Typha latifolia and Cladium jamaisence also present. Both sites experience a semi-diurnal microtidal regime (tidal range ~ 0.6 m) but differ in their exposure to waves. Boat wake-waves are common at the exposed site whereas boat traffic within the canals at the protected site is limited to only a few slow-moving (i.e., idle speed or trolling) vessels. The exposed and protected sites also differ in shoreline morphology. The shoreline at the exposed site is characterized by intermixed fringing marshes and sandy beaches that slope gently from mean tide level to a depth of 0.25 m over a 3-m distance. In contrast, the shoreline at the protected site features a banked edge covered by various turf grasses (Poaceae spp.) where water depths exceed 1 m in less than 0.3 m.
Map showing the location of the study area (inset), experimental project sites and donor marsh. The study was conducted on and near the Fish River near Mobile Bay, Alabama, USA (inset). Sods used in experiments were collected from a nearby donor marsh and transported for use at the site along the Fish River (exposed site) and within a nearby adjacent canal (protected site)
Experimental design and site construction
The main and interactive effects of varying marsh construction designs on porewater concentrations of oxidized nitrogen species (i.e., NOx) were evaluated at the protected and exposed sites using ANOVAs. At each study site a total of 24 treatment combinations, including two sediment types (i.e., coarse and fine), two platform slopes (i.e., steep and shallow), two platform positions (i.e., steep and shallow) and three initial planting density (i.e., 0%, 50% and 100% cover), were replicated within three blocks (Fig. 2).
Overview of experimental treatment combinations at the exposed (a) and protected (b) sites. At each site, combinations of two sediment types (coarse and fine), two platform slopes (steep and shallow), two platform positions and initial planting density (0%, 50% and 100% cover), were replicated within three blocks. However, while slope treatments were fully randomized at the exposed site, slope treatments were set within two groups (i.e., steep and shallow) at the protected site (b) due to the fixed nature of experimental mesocosms
At the exposed site, the 24 treatment combinations were fully randomized within each of the three experimental blocks (i.e., randomized block design). Treatment combinations were designated in 0.3 m wide × ~ 1.22 m long experimental flumes running perpendicular along a ~ 5 m stretch in each block of the exposed site shoreline. Flumes were separated by ~ 1.27 cm thick × 1.22 m long × 0.6 m tall PVC sheets driven 25 cm (relative to existing grade) into the earth along the ~ 5 m block transect (Fig. 2a). Each flume was fully excavated (i.e., removal of existing sediments) followed immediately by placement of an impervious clay layer at the base of each flume according to the designated slope treatment (0.08 (1:12) for shallow and 0.17 (1:6) for steep). These slopes are characteristic of the range of slopes observed in the field (Temple 2021). Flumes were then filled to 25 cm above the clay layer with sediments and planted according to planting density and platform position treatments. Sediments were purchased locally and included coarse (sand; grain size 0.25–2 mm) and fine (topsoil; grain size ≤ 0.25 mm) sediment types. These commercial sediment types are both typically low in organic matter content (e.g., Martin et al. 2020). Whole sods (i.e., soil and above- and below-ground vegetation components) measuring 30 cm wide × 30 cm long × 30 cm deep at the base were extracted from a nearby donor marsh (Fig. 1) for use in experiments following Sparks et al. (2014, 2015).
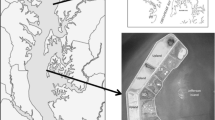
To simulate current and future sea-levels and differentiate platform position, plants and sampling wells were established either at the upper end or lower end of a 60 cm long area in the flumes. Placement of plants for the high platform position treatment (i.e., current sea-level) began 10 cm from the upland edge of flumes, while plants in the lower platform position treatment (i.e., future sea-level) were planted in an area starting 30 cm below the upland edge of flumes (Fig. 2a). After trimming sods to 25 cm depth, initial planting density treatments were established within the flumes by excavating new sediments as follows: 2 whole sods placed back-to-back for 100% initial planting density and 1 whole sod quartered and arranged in a checkerboard pattern for 50% initial planting density; 0% initial planting density plots were left bare (e.g., Sparks et al. 2015). Two porewater wells, each constructed from ~ 3.8 cm diameter × 30 cm tall screened PVC pipe, were then installed in all experimental flumes to 25 cm depth: within and at the end of the planting area (i.e., wells A and B, respectively; Fig. 3). An auger was used to install wells which were then filled with sandy sediment, regardless of experimental sediment treatment. Diffuser plates (Sparks et al. 2015) were also placed to a depth of 20 cm within a 10 cm buffer area above the planting area to facilitate the even distribution of the nutrient solution during experimental runoff simulations (Fig. 3; discussed below).
Cross sectional view of experimental marsh flumes. The simulated groundwater solution (SGW) flowed from subterranean diffusers (hexagonal polygon) above an impermeable layer (i.e., clay layer at protected site (not shown) or PVC flume base) and through the experimental flumes. After SGW simulations, porewater was collected from wells set to 25 cm depth within (A) and after (B) the 60 cm experimental planting area. Plant sods were planted to 25 cm (note: plant cartoon represents above- and below-ground components). Diffusers were set to a depth of 20 cm and installed 10 cm of the upper shoreward boundary of the experimental planting area
Shoreline morphology at the protected site required alternative methods to establish experimental conditions comparable to those at the exposed site. Notably, the abrupt drop in water depth from the edge of protected site shorelines required platform amendments to achieve an elevation (relative to mean high water) similar to that at exposed sites. Therefore, field mesocosms were constructed to house experimental flumes (Fig. 2b). Mesocosms were framed using dimensional lumber and PVC materials (Fig. 2b, Online Resource 1) to inside dimensions representing half of the plots within an exposed site block: ~ 2.5 m wide (parallel to shoreline) × ~ 1.22 m long (perpendicular to shoreline) × 25 cm deep (from the base to top of the mesocosm). Three sides of mesocosm frames were constructed using ~ 5 cm × ~ 30 cm dimensional lumber, while the seaward side of mesocosm boxes was constructed using ~ 5 cm × ~ 5 cm lumber and PVC lattice which was covered in landscaping fabric, so as to facilitate water movement (Online Resource 1). Structural support at the base of mesocosm frames was provided by ~ 5 cm × ~ 10 cm lumber running lengthwise from the landward to shoreward edge of mesocosms. Prior to setting within framed mesocosms, eleven ~ 1.27 cm thick × ~ 1.27 cm deep grooves were cut lengthwise and spaced evenly (~ 30 cm width) along the long end of ~ 2.54 cm thick × ~ 2.5 m wide × ~ 1.22 m PVC sheets that would serve as the impermeable base of mesocosms. After base installation, flume walls, constructed from ~ 1.27 cm thick × ~ 30.27 cm tall × ~ 1.22 m long PVC sheets, were glued using silicone adhesive and set within grooves to create 12 flumes within mesocosms (Online Resource 1). Mesocosms were set on top of concrete cinder blocks in the water near the edge of protected site shorelines so as to facilitate slope and elevation adjustments comparable to those at exposed sites (described above). However, slope could not be manipulated within the mesocosms. Therefore, within each of the three experimental blocks, two mesocosms were constructed. One mesocosm replicated our steep slope from the exposed site, and the other replicated the low slope. This resulted in a split-plot design (i.e., blocks = whole plots, slope treatments/individual mesocosms = split plots). Combinations of sea-level, planting density, and sediment type were then randomly assigned within the mesocosms (i.e., slope split plots) following the methods described for the exposed sites. The exposed and protected sites were constructed in May and September 2016, respectively, and allowed to acclimate over winter 2016 before starting experiments.
Experimental run-off simulations
Experimental run-off simulations were administered over 10-day periods during May and August 2017 (hereafter, “spring” and “summer”) at both the protected and exposed sites to capture any possible changes in nutrient removal that could be attributed to the evolution of growth in J. roemerianus marshes typical in the Northern Gulf of Mexico (nGOM; Eleuterius 1984). A 10-day period was chosen to ensure that plots were saturated by the nutrient plume (i.e., steady state) based on calculations from previous experiments in coarse sediments (e.g., Sparks et al. 2014) and fine sediments (i.e., Sparks et al. 2015). Spring simulations began on May 11th and May 23rd and summer simulations began on August 21st and August 3rd at the protected and exposed sites, respectively.
To simulate run-off events, a gravity-fed continuous drip system was established to direct a simulated groundwater (SGW) solution from upland reservoirs to experimental flumes via installed subsurface diffusers (Online Resource 2; Fig. 3; Sparks et al. 2014). The SGW was mixed onsite using centrifugal pumps in 208 L mixing containers to produce a concentration of 1000 µM KNO3 solution. While this is a high concentration, it was necessary due to high dissolved inorganic nitrogen (DIN) concentrations characteristic of the study area (Novoveská and MacIntyre 2019). These high background DIN concentrations were confirmed in similar previous studies (Martin et al. 2020) and were also observed in some flume porewater samples collected prior to simulations (Online Resources 4 and 5). After mixing, the SGW was pumped from the mixing containers to individual ~ 102 L reservoirs (i.e., one reservoir per experimental flume; Online Resource 2). These reservoirs were connected to subsurface diffusers via 0.95 cm (inside diameter) flexible vinyl tubing and featured an inline valve that allowed drip rate control. The drip rate was set to continuously deliver ~ 34 L/day over the 10-day simulation period (Sparks et al. 2015). During this period, reservoirs were monitored daily and refilled, as necessary.
Porewater sampling and processing
Porewater samples were collected from wells before and immediately following simulated run-off events to assess the effect of treatment combinations on NOx concentrations. Porewater was extracted from wells using sipper tubes (e.g., McKee et al. 1988) and stored in plastic scintillation vials which were frozen until subsequent analysis. Nutrient analyses were performed using a Skalar San + segmented flow autoanalyzer, following standard EPA methods for nutrient analyses, by Technical Support Services at the Dauphin Island Sea Lab, Alabama, U.S.A. (e.g., Sparks et al. 2015; Temple et al. 2019).
Percent cover change
Percent cover of J. roemerianus was expected to increase as was found in similar experiments (Sparks et al. 2014, 2015). However, J. roemerianus is also known to facilitate the growth of other species in similar fringing marshes (Martin et al. 2020). Therefore, the percent cover of each species in experimental flumes was visually estimated before and after Summer 2017. All cover data reflect the relative cover by individual species as they occur naturally in the field since different plant species occur at varying stem densities on a square meter basis (e.g., 300 and 1000 stems m−2 for Spartina spp. and J. roemerianus, respectively).
Statistical analyses
The effects of the varying treatment combinations on porewater NOx concentrations were evaluated using ANOVAs with post hoc tests when appropriate. Four-way ANOVAs were used in initial models but model structure differed between the exposed and protected sites. For the exposed site, slope, sediment type, initial planting density, and platform position were treated as fixed factors while block was treated as a random factor. The model for the protected site featured sediment type, initial planting density, and platform position as fixed factors, and block and slope were treated as random factors. The most offshore porewater wells (B) were lost in one block at the exposed site due to wave action. Therefore, only data from porewater A wells was considered at the exposed site; whereas, data from both wells was considered at the protected site. Data were fourth root transformed as appropriate to meet or approach model assumptions of normal distribution and equal variance. As such, significance is reported at the α = 0.005 level to compensate for mild violations of model assumptions and the high number (24) of treatment levels. Following full model construction, non-significant factors (i.e., p > 0.005; Table 1) were removed and the significance (at the α = 0.05 level) of main and interactive effects of the remaining factors was evaluated in simpler models. Final model selection was made using Akaike information criteria (AIC; Burnham and Anderson 2002). Percent cover change data at both sites were not normally distributed and could not be corrected using transformations and therefore, these responses were compared individually for each initial planting density treatment at each simulation and between exposed and protected sites using Kruskal–Wallis tests. Statistical analyses were performed in R (R core team 2020), using base packages, “lme4” for model construction (Bates et al 2015) and “emmeans” for multiple comparisons (Lenth 2020).
Results
Main treatment effects
Main treatment effects varied across porewater wells in flumes and across sites (Table 1). None of the treatments had any detectable effect on porewater NOx at the exposed site (p > 0.005; Table 1; Online Resource 4). Therefore, no other statistical analyses were run for nutrient concentrations at the exposed site. At the protected site, only sediment type and initial planting density had a significant effect on porewater NOx (p < 0.005; Table 1). Neither platform position nor slope had any measurable effects on nutrient concentrations in samples collected after simulations (p > 0.005; Table 1). The significant effects of sediment type and initial planting density are discussed further below.
Effects of sediment type and initial planting density at the protected site
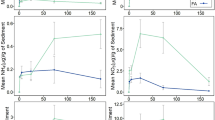
Reduced models consistently confirmed the strong main effects of sediment type and initial planting density (Table 2). Indeed, these main effects had significant effects on porewater NOx collected from both wells after each simulation (p < 0.05; Fig. 4, Table 2). However, the interactive effects of sediment type and initial planting density on porewater NOx were significant only in A wells following the summer simulation (p < 0.05; Table 2).
Mean porewater NOx concentrations (y axis; log scale) collected from A and B wells at the protected site plotted by initial planting density treatment (cover, x axis) and sediment type (bar color). Data show the strong sediment type effect observed in both A and B wells (left and right columns, respectively) following the spring simulation (top row) and summer simulation (bottom row). Lines above bars connect initial planting density treatments that are significantly different. Sediment type is significant in all cases. In general, control (i.e., 0 initial planting density) treatments with coarse sediments had significantly higher NOx concentrations as compared to planting treatments. Error bars indicate ± 1 standard error
Results of pairwise comparisons examining the effects of initial planting density on NOx in A and B wells collected following the spring simulation and those assessed from B wells collected following the summer simulation varied (Table 3). Mean NOx concentration was significantly lower in planted plots (i.e., 50 and 100% initial planting density) as compared to bare plots in spring simulation A wells and summer simulation B wells (p < 0.05; Table 3). Interestingly, while mean B well NOx concentration was significantly lower in plots initially planted at 100% density as compared to bare plots following spring simulation (p < 0.05), NOx concentration in plots initially planted at 50% density was not statistically different from bare plots (p > 0.05; Table 3). However, mean NOx concentration was also not statistically different among planted treatments in B wells following the spring simulation, as was also observed in A and B wells following spring and summer simulations, respectively (p > 0.05; Table 3).
While overall trends in mean NOx concentrations associated with the different treatment combinations assessed following the summer simulation in A wells were straightforward, pairwise comparisons of these combinations varied (Table 4). Overall, NOx concentrations decreased along a gradient of both sediment type and initial planting density (Table 4). The greatest NOx concentrations were observed in bare plots with coarse sediments while the lowest NOx concentrations were found in plots initially planted at 100% density in fine sediments (Table 4). With the exception of the significantly higher NOx concentrations observed in bare plots as compared to those initially planted at 100% density in coarse sediments (p < 0.05), differences among individual treatment combinations broke mostly along the differing sediment types (Table 4). However, even then, NOx concentrations observed in plots initially planted at 100% density in coarse sediments were not statistically different from bare plots and those initially planted at 50 and 100% density in fine sediments (p > 0.05; Table 4).
Plant cover at the exposed and protected sites
Whereas observed cover of J. roemerianus at protected sites prior to SGW simulations remained mostly consistent with initial planting density treatments (Fig. 5a), observed cover changed within bare plots and plots initially planted at 100% planting density at exposed sites (Fig. 5b). The cover of various species (discussed below) increased by 25% in bare plots in the exposed site by the summer simulation (p < 0.05), whereas plots planted at 100% planting density declined in cover of J. roemerianus by nearly 50% (p < 0.05), and plots planted at 50% density did not differ significantly from the initial planting (p > 0.05; Fig. 5b, Online Resource 3). At protected sites, differences in cover between initial planting density treatments were statistically significant only for 100% plots where the cover of J. roemerianus declined by nearly 20% following the summer simulation (p < 0.05; Online Resource 3). Interestingly, observed cover in 50% initial planting density plots did not differ between exposed and protected sites (p > 0.05). In contrast, bare plots at exposed sites featured significantly greater plant cover (i.e., by various species) than the those in the protected sites (p < 0.05) and 100% initial planting density plots at exposed sites had significantly less cover than their protected counterparts (p < 0.05; Online Resource 3). As observed in previous work in the area (Martin et al. 2020), other plant species besides J. roemerianus were found in plots at both sites. Other species found in plots included Eleocharis robbinsii, Typha latifolia, Panicum repens, Sagittaria lancifolia, Panicum virgatum, Spartina patens, Alternanthera philoxeroides, Amaranthus cannabinus, Kosteletzkya virginica, Distichlis spicata and Cladium jamaisence. However, individual cover of other species did not exceed 2% of the vegetated area in any of the plots. Still, combined cover of these species in plots was, on average, greatest at protected sites (Fig. 5a and b).
Observed percent cover (y axis) of the study species (Juncus roemerianus, light gray) and other species (dark gray) by simulation (x axis, bottom) and by initial planting density treatment (x axis, top) at the protected (a) and exposed (b) sites. At the protected site, each planting treatment retained similar observed cover from the start and establishment of the experiment (i.e., simulation 0) throughout spring and summer simulations (i.e., simulations 1 and 2). At the exposed site, only the 50% initial planting density treatment retained similar observed cover from the start and establishment of the experiment (i.e., simulation 0) throughout experimental simulations. The combination of species observed within control and 100% initial planting density treatments increased to 25% cover and declined to 50% cover, respectively
Discussion
This study builds on the work of others investigating the most cost-effective options for fringing marsh restoration in the nGOM (Martin et al. 2020; Sparks et al. 2013, 2014, 2015). Like others, we found that marshes planted initially at 50% and 100% density were statistically similar in terms of nitrogen removal (Table 3; Fig. 4; Sparks et al. 2013, 2015) and that platform position was not a significant factor in nitrogen removal at the protected site (Table 1; Martin et al. 2020). Further, we found a strong effect of sediment type (Table 2, Fig. 4), which agreed with previous experiments conducted individually in fine- and coarse-grained sediments (Sparks et al. 2014, 2015). However, we did not anticipate the results observed at the exposed site where none of the factors had any measurable effect on porewater NOx (Table 1; Online Resource 4).
At the protected site, the effects of sediment type and initial planting density on nitrogen removal were largely expected and may be due, in part, to the effects each has on water flow through the marsh and the abundance of organic materials. Flow is often tied to the rate of nitrogen removal in marshes, with slower flows leading to higher removal rates (Sparks et al. 2014; Spieles and Mitsch 1999; Tobias et al. 2001). While we did not measure flow through the flumes in this study (constant flow of introduced solution), we manipulated three factors that can influence flow rates in marshes: vegetation density (i.e., initial planting density), slope, and sediment type (Sparks et al. 2014; Tobias et al. 2001). Of these factors, only initial planting density and sediment type had any observable effect on NOx removal (Table 1) and of the two, sediment type appeared to be the most important (Table 4). Sparks et al. (2014) reported a similar trend among various planting treatments in coarse grain sediments where nutrient removal was similarly low among planting treatments in high flow experimental flumes, even with a modest decline in observed water flow with increasing planting density. These differences stood in stark contrast to previous experiments by the authors showing that planted marshes remove a substantial amount of nutrients in fine and organic soils (Sparks et al. 2013, 2015).
Results from protected site experiments help to explain these differences and lends credence, albeit limited, to the influence of sediment type on the rate of groundwater flow and subsequent biologically mediated nitrogen removal as suggested by Sparks et al. (2014). Indeed, sediment type and initial planting density were consistently important factors in NOx models (Table 2). Planted treatments generally removed more NOx than did bare treatments (Table 3) while fine sediments resulted in lower porewater NOx concentrations (Fig. 4). The interaction between sediment type and initial planting density was only significant in one of the models examined (Table 2) and thus, broad interpretation of these results is limited. However, multiple comparisons analysis did demonstrate the overwhelming influence of sediment type, as treatments with coarse sediments had higher mean NOx concentrations as compared to treatments with fine sediments. Paired with those of Sparks et al. (2013, 2014, 2015), the results observed at the protected site suggests sediment type is a stronger influencer of flow rate, and likely nutrient removal, than the abundance of plant material.
Fine sediments are also typically rich with particulate organic materials, which may be sufficient to facilitate denitrification (Davis et al. 2004; Yamamoto and Lopez 1985).This denitrification can be supplemented further via the production of benthic microorganisms even in the absence of plants (Hamersley and Howes 2003). We found some evidence to support the dominant influence of this pathway in the multiple comparisons analysis. This analysis showed that non-planted plots with fine sediments were statistically similar to plots initially planted at 100% density in coarse sediments (Table 4). High organic matter content may also favor another pathway of effective nitrate removal: dissimilatory nitrate reduction to ammonia (Giblin et al. 2013). Evidence for this pathway would involve an increase in porewater ammonium concentrations following simulations. However, this trend was not observed in the present study. Porewater ammonium concentrations were similar before and after simulations at both exposed and protected sites (Online Resource 5). Further experiments designed explicitly to examine differing flow rate and soil organic matter content in the context of differing sediment types and planting densities are needed to fully understand the relative effects of each on nutrient removal in marshes.
In contrast to the protected site, sediment type and initial planting density treatments were not statistically significant in exposed site NOx models, which is likely due to the indirect effects of waves. The exposed site experienced more wave action than the protected sites and sites from previous experiments (Sparks et al. 2013, 2014, 2015). At this site, waves may have mitigated the significant effects of sediment type and initial planting density via two indirect mechanistic pathways: by controlling biomass production and by limiting soil anoxia (i.e., those conditions ideal for denitrification; Davis et al. 2004).
Waves are known to influence the structure of coastal plant communities (Roland and Douglass 2005; Woodroffe 2002). For example, Roland and Douglass (2005) found marsh coverage and health diminished with the increasing regularity of large waves (i.e., over 30 cm). Cover at our exposed site suggested a similar limiting effect on plant growth. Indeed, cover within plots initially planted at 100% density declined towards 50% density while cover within 50% density plots remained constant and cover within initially bare plots increased (Fig. 5b). This levelling effect on plant growth may help to explain the observed lack of initial planting density treatment effects on porewater NOx concentrations at the exposed site (Table 1). In fact, waves are often linked to various morphological features associated with plant growth that could affect nitrogen removal in marshes. These include stem density, height aboveground (Silinski et al. 2018) and belowground rooting behavior (Balke et al. 2011). Additionally, waves may alter the structure of marsh communities in other important ways that could indirectly affect nitrogen utilization in marshes (e.g., via controls on species distribution or nutrient utilization in plant tissues; Keddy 1985). For example, flexibility of plant shoots is thought to increase the wave tolerance of plant tissues (Rupprecht et al. 2015; Schulze et al. 2019) but is sometimes also linked to the availability and utilization of differing nutrients in plant tissues (e.g., Silinski et al. 2018; Sloey and Hester 2018). While plant responses to many stressors is well documented (Kirwan and Megonigal 2013; Nyman et al. 2006; Temple et al. 2019; Vasquez et al. 2006), plant responses to waves is not and needs further research.
In addition to its influence on plant growth, wave action may have disrupted denitrification at surface level sediments that would typically go anoxic at the exposed site once the marsh was inundated (i.e., mid to high tide). Nitrogen removal in brackish marshes typically follows two major pathways: uptake by plants and microbenthos during biomass production and microbially-mediated denitrification (Davis et al. 2004). Of these two pathways, the latter has often accounted for the greatest pathway of allochthonous nitrogen removal in marshes (VanZomeren et al. 2012). For example, several researchers have estimated that over 60% of nitrogen is removed from marshes via this pathway (VanZomeren et al. 2012; White and Howes 1994). Various studies have demonstrated the factors controlling denitrification in marshes (Hu et al. 2019; Neubauer et al. 2019; Zheng et al. 2016), but in general, the process requires available nitrate and organic carbon sources, the presence of denitrifying microorganisms and anaerobic conditions (Davis et al. 2004). Both exposed and protected sites were presumably established in a way that would satisfy most of these requirements and for at least part of the day (e.g., as influenced by tidal water movement). For example, while not a focus of this study, it is likely that experimental sods used in planting treatments were similar in terms of soil organic matter and microbial community diversity (e.g., including denitrifying microorganisms) at both the protected and exposed sites since they were harvested from the same donor marsh. Additionally, all flumes were supplied a steady supply of available nitrate. However, soil hydroedaphic conditions within the 25 cm depth of flume sediments and sods likely differed between the two sites due to the potential wave-driven reaeration of these sediment layers (e.g., Hosoi et al. 1977) with excessive daytime boating activity. As discussed above, the study area experiences a diurnal microtidal regime. Within this regime, the predominance of daytime high tides shifts semi-annually such that high tides are more frequent during the daytime in summer months and hence, soil anoxia is also most likely during this time. Incidentally, this period (i.e., daytime) also coincides with the greatest frequency of recreational boating traffic (i.e., 8 am to 6 pm; personal observation). As such, wave activity may have reversed the declining hydroedaphic conditions that would be expected with tidal inundation. If so, aerobic soil conditions would persist and thereby limit denitrification (Koch et al. 1992). Thus, we speculate that the convergence of these events nullified the strong sediment type effect on NOx at the exposed site, as was observed in protected site flumes receiving fine sediments even in the absence of vegetation (i.e., control plots with fine sediments; Table 4). Indeed, NOx increased dramatically in both A and B wells following simulations while ammonium concentrations were low and unchanged during the same period (Online Resources 4 and 5). Still, more research is needed to determine the relative influence of waves on various nitrogen removal pathways, including denitrification.
Other factors may also have played a role in the differences in model results observed at exposed and protected sites. While we assumed comparable salinity between sites, the exposed site may have had more contact with higher salinity waters originating from Weeks and Mobile Bays which could also limit denitrification in marshes. For example, Neubauer et al. (2019) found that marshes more regularly exposed to higher salinities had a lower abundance of denitrifying microorganisms as compared to reference marshes in tidal freshwater systems. This change in microbial community structure resulted in a nearly 70% decline in denitrification in these marshes. Experimental design differences between the sites may have also played a role. For example, mesocosms effectively eliminated groundwater exposure at the protected site which could have further influenced removal processes at the exposed site. Tidal forcing and exchange of materials between ground- and sea waters within the intertidal zone has been demonstrated to favor certain microbial communities and nitrogen utilization pathways (e.g., Liu et al. 2017), for example. A disconnection from groundwater and the lack of potential flushing by waves at the protected site could also lead to higher salinities there that would favor other nitrogen removal pathways as well (Neubauer et al. 2019). The mechanisms driving these processes were beyond the scope of this study but warrant further future research. Finally, flooding at the protected site during the summer simulation may have limited detection of treatment effects since the nutrient concentrations were lower during this period (Fig. 4). Indeed, this simulation occurred when plant productivity and hence, nutrient uptake, in these marshes is greatest (Eleuterius 1984).
As with all experiments, interpretation of results presented here are bound by the scope and design of the experiment. In this study design, the novelty of simultaneous exploration of several potentially important factors to nutrient removal in marshes came with a tradeoff in the replication of experimental combinations. This limitation was compounded by the small spatial extent of the study area. Still, results presented here are generally in line with results from previous similar experiments (Sparks et al. 2013, 2015; Martin et al. 2020), suggesting that the relative effects of sediment type and initial planting density observed in this study would apply more broadly to the oligo- and mesohaline estuaries in the nGOM where J. roemerianus is found. The positioning of porewater wells was also purposefully designed to assess relative treatment affects. As such, results offer little insight into the effects of vegetation buffer width and planting densities therein on nutrient removal in constructed marshes. Future research examining similar topics should focus on these potentially interactive effects in larger-scale experiments over a wider spatial extent.
Conclusion
Shoreline wetland conservation, restoration, and enhancement can be an effective management tool for reducing the magnitude of upland pollutants introduced to our coastal waters. However, there are several site characteristics that must be considered when designing these projects. Our research suggests that sediment type and vegetation density are among the most important considerations for projects targeting nutrient removal. This research further suggests that while accounting for wave climate is standard practice for the purposes of plant establishment, it may also be an important consideration for other project goals such as nutrient removal. In fact, wave action may have significant negative consequences to the objectives of coastal restoration, conservation and enhancement projects.
Availability of data and materials
Upon acceptance, the authors will publish data and associated metadate on the Mississippi State University Institutional Repository (https://ir.library.msstate.edu/).
References
Armstrong W (1979) Aeration in higher plants. Advances in botanical research, vol 7. Academic Press, London, pp 225–332
Balke T, Bouma TJ, Horstman EM, Webb EL, Erftemeijer PL, Herman PM (2011) Windows of opportunity: thresholds to mangrove seedling establishment on tidal flats. Mar Ecol Prog Ser 440:1–9
Barbier EB (2015) Valuing the storm protection service of estuarine and coastal ecosystems. Ecosyst Serv 11:32–38
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67(1):1–48. https://doi.org/10.18637/jss.v067.i01
Bergamaschi BA, Tsamakis E, Keil RG, Eglinton TI, Montluçon DB, Hedges JI (1997) The effect of grain size and surface area on organic matter, lignin and carbohydrate concentration, and molecular compositions in Peru Margin sediments. Geochim Cosmochim Acta 61(6):1247–1260
Bilkovic DM, Mitchell MM, Davis J, Herman J, Andrews E, King A, Mason P, Tahvildari N, Davis J, Dixon RL (2019) Defining boat wake impacts on shoreline stability toward management and policy solutions. Ocean Coast Manag 182:104945
Brix H (1997) Do macrophytes play a role in constructed treatment wetlands? Water Sci Technol 35(5):11–17
Burnham K, Anderson D (2002) Model selection and multi-model inference: a practical information-theoretic approach, 2nd edn. Springer, New York
Coops H, Van der Velde G (1996) Effects of waves on helophyte stands: mechanical characteristics of stems of Phragmites australis and Scirpus lacustris. Aquat Bot 53(3–4):175–185
Davis JL, Nowicki B, Wigand C (2004) Denitrification in fringing salt marshes of Narragansett Bay, Rhode Island, USA. Wetlands 24(4):870–878
Dodds WK (2006) Nutrients and the “dead zone”: the link between nutrient ratios and dissolved oxygen in the northern Gulf of Mexico. Front Ecol Environ 4(4):211–217
Eleuterius LN (1984) Autecology of the black needlerush Juncus roemerianus. Gulf Caribb Res 7(4):339–350
Engelaar WM, Matsumaru T, Yoneyama T (2000) Combined effects of soil waterlogging and compaction on rice (Oryza sativa L.) growth, soil aeration, soil N transformations and 15N discrimination. Biol Fertil Soils 32(6):484–493
Feagin RA, Lozada-Bernard SM, Ravens TM, Möller I, Yeager KM, Baird AH (2009) Does vegetation prevent wave erosion of salt marsh edges? Proc Natl Acad Sci USA 106(25):10109–10113
Fisher J, Acreman MC (2004) Wetland nutrient removal: a review of the evidence. Hydrol Earth Syst Sci 8(4):673–685
Fox L, Valiela I, Kinney EL (2012) Vegetation cover and elevation in long-term experimental nutrient-enrichment plots in Great Sippewissett Salt Marsh, Cape Cod, Massachusetts: implications for eutrophication and sea level rise. Estuar Coast 35(2):445–458
Gedan KB, Kirwan ML, Wolanski E, Barbier EB, Silliman BR (2011) The present and future role of coastal wetland vegetation in protecting shorelines: answering recent challenges to the paradigm. Clim Change 106(1):7–29
Giblin AE, Tobias CR, Song B, Weston N, Banta GT, Rivera-Monroy VH (2013) The importance of dissimilatory nitrate reduction to ammonium (DNRA) in the nitrogen cycle of coastal ecosystems. Oceanography 26(3):124–131
Gittman RK, Peterson CH, Currin CA, Joel Fodrie F, Piehler MF, Bruno JF (2016) Living shorelines can enhance the nursery role of threatened estuarine habitats. Ecol Appl 26(1):249–263
Gupta TR, Foster JH (1973) Valuation of visual-cultural benefits from freshwater wetlands in Massachusetts. J Northeast Econ Counc 2(2):262–273
Hamersley MR, Howes BL (2003) Contribution of denitrification to nitrogen, carbon, and oxygen cycling in tidal creek sediments of a New England salt marsh. Mar Ecol Prog Ser 262:55–69
Hosoi M, Ishida A, Imoto K (1977) Study on Re-Aeration by Waves. Coast Eng Jpn 20(1):121–127
Howes BL, Goehringer DD (1994) Porewater drainage and dissolved organic carbon and nutrient losses through the intertidal creekbanks of a New England salt marsh. Mar Ecol Prog Ser 114(3):289–301
Howes BL, Dacey JWH, Goehringer DD (1986) Factors controlling the growth form of Spartina alterniflora: feedbacks between above-ground production, sediment oxidation, nitrogen and salinity. J Ecol 74:881–898
Hu W, Zhang W, Zhang L, Lin X, Tong C, Lai DY, Chen Y, Zeng C (2019) Short-term changes in simulated inundation frequency differentially affect inorganic nitrogen, nitrification, and denitrification in estuarine marshes. Ecol Ind 107:105571
IPCC (2019) Climate change and land: an IPCC special report on climate change, desertification, land degradation, sustainable land management, food security, and greenhouse gas fluxes in terrestrial ecosystems [P.R. Shukla, J. Skea, E. Calvo Buendia, V. Masson-Delmotte, H.-O. Pörtner, D. C. Roberts, P. Zhai, R. Slade, S. Connors, R. van Diemen, M. Ferrat, E. Haughey, S. Luz, S. Neogi, M. Pathak, J. Petzold, J. Portugal Pereira, P. Vyas, E. Huntley, K. Kissick, M. Belkacemi, J. Malley, (eds.)]. In press
Jesus B, Brotas V, Ribeiro L, Mendes CR, Cartaxana P, Paterson DM (2009) Adaptations of microphytobenthos assemblages to sediment type and tidal position. Cont Shelf Res 29(13):1624–1634
Keddy PA (1985) Wave disturbance on lakeshores and the within-lake distribution of Ontario’s Atlantic coastal plain flora. Can J Bot 63(3):656–660
Kirwan ML, Megonigal JP (2013) Tidal wetland stability in the face of human impacts and sea-level rise. Nature 504(7478):53–60
Kleinhuizen AA, Mortazavi B (2018) Denitrification capacity of a natural and a restored marsh in the Northern Gulf of Mexico. Environ Manag 62(3):584–594
Koch MS, Maltby E, Oliver GA, Bakker SA (1992) Factors controlling denitrification rates of tidal mudflats and fringing salt marshes in south-west England. Estuar Coast Shelf Sci 34(5):471–485
Lenth R (2020) emmeans: estimated marginal means, aka least-squares means. R package version 1.4.6. https://CRAN.R-project.org/package=emmeans
Liu Y, Jiao JJ, Liang W, Luo X (2017) Tidal pumping-induced nutrients dynamics and biogeochemical implications in an intertidal aquifer. J Geophys Res Biogeosci 122(12):3322–3342
Lucas WC, Greenway M (2008) Nutrient retention in vegetated and nonvegetated bioretention mesocosms. J Irrig Drain Eng 134(5):613–623
Martin S, Sparks EL, Constantin AJ, Cebrian J, Cherry JA (2020) Restoring fringing tidal marshes for ecological function and ecosystem resilience to moderate sea-level rise in the Northern Gulf of Mexico. Environ Manag 67(2):384–397
McConchie JA, Toleman IEJ (2003) Boat wakes as a cause of riverbank erosion: a case study from the Waikato River, New Zealand. J Hydrol (New Zealand) 42:163–179
McKee KL, Mendelssohn IA, Hester MW (1988) Reexamination of pore water sulfide concentrations and redox potentials near the aerial roots of Rhizophora mangle and Avicennia germinans. Am J Bot 75(9):1352–1359
Mehvar S, Filatova T, Sarker MH, Dastgheib A, Ranasinghe R (2019) Climate change-driven losses in ecosystem services of coastal wetlands: a case study in the West coast of Bangladesh. Ocean Coast Manag 169:273–283
Mendelssohn IA, McKee KL (1988) Spartina alterniflora die-back in Louisiana: time-course investigation of soil waterlogging effects. J Ecol 76:509–521
Morris JT, Sundareshwar PV, Nietch CT, Kjerfve B, Cahoon DR (2002) Responses of coastal wetlands to rising sea level. Ecology 83(10):2869–2877
Morris JT, Shaffer GP, Nyman JA (2013) Brinson review: perspectives on the influence of nutrients on the sustainability of coastal wetlands. Wetlands 33(6):975–988
Nassauer JI (2004) Monitoring the success of metropolitan wetland restorations: cultural sustainability and ecological function. Wetlands 24(4):756–765
Neubauer SC, Piehler MF, Smyth AR, Franklin RB (2019) Saltwater intrusion modifies microbial community structure and decreases denitrification in tidal freshwater marshes. Ecosystems 22(4):912–928
NOAA (2015) Guidance for considering the use of living shorelines. National Oceanic and Atmospheric Administration, Silver Spring, MD, 36p
Novoveská L, MacIntyre HL (2019) Study of the seasonality and hydrology as drivers of phytoplankton abundance and composition in a shallow estuary, Weeks Bay, Alabama (USA). J Aquac Mar Biol 8(3):69–80
Nyman JA, Walters RJ, Delaune RD, Patrick WH Jr (2006) Marsh vertical accretion via vegetative growth. Estuar Coast Shelf Sci 69(3–4):370–380
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Rabalais NN, Turner RE, Wiseman WJ Jr (2002) Gulf of Mexico hypoxia, aka “The dead zone.” Annu Rev Ecol Syst 33(1):235–263
Reddy KR, DeLaune RD (2008) Biogeochemistry of wetlands: science and applications. CRC Press, Boca Raton
Roland RM, Douglass SL (2005) Estimating wave tolerance of Spartina alterniflora in coastal Alabama. J Coast Res 213:453–463
Rupprecht F, Möller I, Evans B, Spencer T, Jensen K (2015) Biophysical properties of salt marsh canopies—quantifying plant stem flexibility and above ground biomass. Coast Eng 100:48–57
Schulze D, Rupprecht F, Nolte S, Jensen K (2019) Seasonal and spatial within-marsh differences of biophysical plant properties: implications for wave attenuation capacity of salt marshes. Aquat Sci 81(4):65
Silinski A, Schoutens K, Puijalon S, Schoelynck J, Luyckx D, Troch P, Meire P, Temmerman S (2018) Coping with waves: plasticity in tidal marsh plants as self-adapting coastal ecosystem engineers. Limnol Oceanogr 63(2):799–815
Silvan N, Vasander H, Laine J (2004) Vegetation is the main factor in nutrient retention in a constructed wetland buffer. Plant Soil 258(1):179–187
Sinsabaugh RL, Findlay S (1995) Microbial production, enzyme activity, and carbon turnover in surface sediments of the Hudson River estuary. Microb Ecol 30(2):127–141
Sloey TM, Hester MW (2018) Impact of nitrogen and importance of silicon on mechanical stem strength in Schoenoplectus acutus and Schoenoplectus californicus: applications for restoration. Wetl Ecol Manag 26(3):459–474
Sparks EL, Cebrian J (2015) Effects of fertilization on grasshopper grazing of northern Gulf of Mexico salt marshes. Estuar Coast 38(3):988–999
Sparks EL, Cebrian J, Biber PD, Sheehan KL, Tobias CR (2013) Cost-effectiveness of two small-scale salt marsh restoration designs. Ecol Eng 53:250–256
Sparks EL, Cebrian J, Smith SM (2014) Removal of fast flowing nitrogen from marshes restored in sandy soils. PLoS ONE 9(10):e111456
Sparks EL, Cebrian J, Tobias CR, May CA (2015) Groundwater nitrogen processing in Northern Gulf of Mexico restored marshes. J Environ Manag 150:206–215
Spieles DJ, Mitsch WJ (1999) The effects of season and hydrologic and chemical loading on nitrate retention in constructed wetlands: a comparison of low-and high-nutrient riverine systems. Ecol Eng 14(1–2):77–91
Temple NA (2021) Improving the cost effectiveness of water wave measurements and understanding of its impact on natural and restored marsh communities. Dissertation, Mississippi State University
Temple NA, Grace JB, Cherry JA (2019) Patterns of resource allocation in a coastal marsh plant (Schoenoplectus americanus) along a sediment-addition gradient. Estuar Coast Shelf Sci 228:106337
Tobias C, Neubauer SC (2019) Salt marsh biogeochemistry—an overview. Coastal wetlands. Elsevier, Amsterdam, pp 539–596
Tobias CR, Anderson IC, Canuel EA, Macko SA (2001) Nitrogen cycling through a fringing marsh-aquifer ecotone. Mar Ecol Prog Ser 210:25–39
Van Slobbe E, de Vriend HJ, Aarninkhof S, Lulofs K, de Vries M, Dircke P (2013) Building with Nature: in search of resilient storm surge protection strategies. Nat Hazards 66(3):1461–1480
VanZomeren CM, White JR, DeLaune RD (2012) Fate of nitrate in vegetated brackish coastal marsh. Soil Sci Soc Am J 76(5):1919–1927
Vasquez EA, Glenn EP, Guntenspergen GR, Brown JJ, Nelson SG (2006) Salt tolerance and osmotic adjustment of Spartina alterniflora (Poaceae) and the invasive M haplotype of Phragmites australis (Poaceae) along a salinity gradient. Am J Bot 93(12):1784–1790
Wetzel RG (2001) Limnology: lake and river ecosystems. Gulf Professional Publishing, Oxford
White DS, Howes BL (1994) Long-term 15N-nitrogen retention in the vegetated sediments of a New England salt marsh. Limnol Oceanogr 39(8):1878–1892
Wigand C, McKinney RA, Charpentier MA, Chintala MM, Thursby GB (2003) Relationships of nitrogen loadings, residential development, and physical characteristics with plant structure in New England salt marshes. Estuaries 26(6):1494–1504
Woodroffe CD (2002) Coasts: form, process and evolution. Cambridge University Press, Cambridge
Yamamoto N, Lopez G (1985) Bacterial abundance in relation to surface area and organic content of marine sediments. J Exp Mar Biol Ecol 90(3):209–220
Yozzo D, Titre J, Sexton J (1996) Planning and evaluating restoration of aquatic habitats from an ecological perspective. Institute for Water Resources, US Army Corps of Engineers, Waterways Experiment Station, Vicksburg, MS, IWR Report 96-EL-4
Zedler JB (1996) Tidal wetland restoration: a scientific perspective and southern California focus. California Sea Grant College System, University of California, La Jolla, CA, p 1996
Zheng Y, Hou L, Liu M, Liu Z, Li X, Lin X, Yin G, Gao J, Yu C, Wang R, Jiang X (2016) Tidal pumping facilitates dissimilatory nitrate reduction in intertidal marshes. Sci Rep 6:21338
Acknowledgements
In addition to our funders, the authors would like to thank Gillian Palino, Andrew Lucore, Matthew Virden, Haley Moss, Jamie Amato, Aaron Macy, Ellee Temple, Bryan Miller and Jamie Phillips-Temple for their assistance with experimental set up and data collection. In addition, the authors thank partners at the Weeks Bay NERR and the Management Application Team for their assistance with the project.
Funding
Funding for this project was provided US EPA Wetlands Program (Grant Number: CD-00D40115-0) and NOAA—Mississippi-Alabama Sea Grant Program (GRANT NUMBER: 8200025414).
Author information
Authors and Affiliations
Contributions
ELS and JC designed experiment; NAT, ELS, JC, SEM and DCF implemented experiment and collected data; NAT performed statistical analyses; NAT, ELS, JC, SEM and DCF prepared the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors do not have any conflicts of interest/competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Temple, N.A., Sparks, E.L., Cebrian, J. et al. Nitrogen removal in constructed marshes at sites protected from and exposed to waves. Wetlands Ecol Manage 29, 549–564 (2021). https://doi.org/10.1007/s11273-021-09800-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11273-021-09800-0