Abstract
A polyextremophilic fungal strain, A. flavus QMS-8, previously isolated from the hypersaline soil taken from the rhizospheric region of the neem tree, Azadirachta indica (Meliaceae), was recognized as the proficient strain for decolorization of Acid Orange 52, a toxic dye employed in textile operations. A. flavus QMS-8 immobilized on Luffa cylindrica (Cucurbitaceae) attained 80.6% and consecutively complete dye degradation within a hydraulic retention time of 12–24 h in a stirred tank reactor even at salinities of 1–5% and environmental temperatures ranging in between 34 and 39 °C under optimized settings. The SEM micrographs revealed the attachment of fungal hyphae on L. cylindrica, while enzyme profiling demonstrated the participation of laccase (0.17 IU mL−1), manganese peroxidase (0.16 IU mL−1), and lignin peroxidase (0.06 IU mL−1) in the degradation of Acid Orange 52. The decolorized samples were assessed through a UV-Vis spectrophotometer and HPLC which did not provide any information about the formation of aromatic amines or any other metabolite. The values of biological oxygen demand, chemical oxygen demand, total dissolved solids, and total suspended solids were reduced from 2836, 3000, 3415, and 360 mg L−1 to 615, 1179, 916, and 145 mg L−1, respectively, and aligned closely with the established criteria set forth by the NEQ. Phytotoxicity and cytotoxicity assays of effluent treated in STR demonstrated a significant reduction in the toxic impact of dye on plants and microbial cells, suggesting that the luffa immobilized A. flavus QMS-8 employed in this study holds promise for mitigating the potentially harmful effects associated with Acid Orange 52. These results offer valuable insights into the efficacy and potential environmental benefits of the bioremediation approach for textile effluent investigated in this research.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Prior to the nineteenth century, various natural materials of herbal and animal origins have been applied for dyeing textile materials. In the 1770s, Picric acid (yellow), Murexide (violet), and Pittacal (dark blue) were developed as colorants. In 1856, Mauveine (also known as aniline purple) was invented by William Henry Perkin as the first synthetic dye that subsequently led to the emergence of wide-ranging colorants (Selvaraj et al., 2021).
Amongst various sectors, the textile and clothing sector is the prime consumer of synthetic colorants (Slama et al., 2021). Globally, around 800,000 t of dyes are manufactured yearly, out of which 56% are used in textile processes (Dos Santos et al., 2007; Jamee & Siddique, 2019). Across the board, nearly 10–15% of the synthetic colorants, which accounts for 140,000 t of the dyes are released into the environment through textile effluents due to poor adsorption into the fabric (Rehman et al., 2018). The colorants (even at a value of < 1 ppm) may alter the pH, gas solubility, chemical oxygen demand (COD), clarity, and aesthetic aspects of naturally existing water bodies. Such conditions may also deteriorate the chemical composition and microbial flora of soil, disturbing soil fertility and agricultural yield (Duarte et al., 2013). Reportedly, dye effluents are notable for their intensive biotoxicity to amphibians, microbiota, and other aquatic species (Demirci & Hamamci, 2013). Thus, this may lead to severe pathological complications in humans through the food chain (Bruno et al., 2019).
Of the dyestuffs, azo dyes (holding one or more -N = N- as a chromophore) followed by anthraquinone dyes are the most pivotal class, comprising over 70% production of colored materials (Mishra et al., 2011). Synthetic dyes and their degraded metabolites, such as aromatic amines, have carcinogenic, mutagenic, and cytotoxic effects on human systems. Direct and regular exposure to the dye can lead to an extended range of human health ailments, such as headache, nausea, skin allergies and ulceration, visual impairment, respiratory sensitization, and gastrointestinal irritation. In addition, dyes can also induce growth retardation, development of malignant tumors, congenital malformations, hemorrhages, and cancers (Bruno et al., 2019; Mathur et al., 2006; Moawad et al., 2003; Sponza & Işik, 2002).
Due to the eco-toxic traits of colorants and their by-products, there is a dire need to develop and regularize the methods involved in the removal of color from textile wastewater before their release into water bodies. Industries have adopted an extensive range of physicochemical effluent treatment strategies, such as adsorption, membrane filtration, coagulation/flocculation, and advanced oxidation processes, to remove colorants in water resources. However, these existing methods are less applicable at a commercial scale due to their elevated operational and maintenance costs, less efficiency of the system, intensive infrastructure, generation of secondary pollutants, and other non-adaptable features (Adegoke & Bello, 2015). Moreover, they are insufficient to attain absolute detoxification of dye effluents.
The proficiency of filamentous fungi (molds), in particular the genus Aspergillus as a dye degrader/decolorizer, has also been noted for the remediation of textile wastewater. Fungal mycelia have been recognized as the most practical candidate the for eradication of toxic dyes over other single-celled microbial species due to their inherent adaptability, relative diversification in different environments, oxidative non-stereo-selective exogenous (oxidoreductases), and cell-bound (cytochrome P450 monooxygenases) enzymatic mechanisms to mineralize various dyes (Kaushik & Malik, 2009). In fungal structures, the dissimilation of dyestuff is primarily accomplished by the dissociation of functional groups into colorless amines. Their uninterrupted bioconversion into nontoxic substances under aerophilic settings, through biosorption, biodegradation/enzymatic mineralization, or the combination of both systems, are mechanisms for the bioremediation of textile colorants (Solís et al., 2012).
The application of immobilized mycelial mass over-dispersed free cells is the most suitable method to deal with colored effluent as it allows the reuse of the biomass under harsh environmental perturbations with less or even no risk of clogging in continuous flow systems (Rodríguez Couto, 2009).
However, the inclusion of sodium hydroxide into the dyeing tank causes an increase in the sodium level and the presence of other salts that have been customarily employed in the textile dyeing process for different purposes such as the separation of organic contaminants, preparation of standard solutions of dye as well as to induce precipitation in dyestuff negatively affects dye degradation processes owing to the inability of the fungi to grow in such harsh environmental settings. Therefore, the employment of extremophilic fungi, exclusively with halophilic as well as thermophilic potential is generally viewed as the most appealing approach for the treatment of industrial effluents under growth-limiting environments due to their well-suited enzymes and bioremediation capabilities (Araújo et al., 2020; Csillag et al., 2022; Khalid et al., 2008; Lima et al., 2021).
Hence, the objective of the present study was to develop an efficient system for the removal of hazardous Acid Orange 52 dye in a stirred tank reactor using immobilized cells of an autochthonous polyextremophilic Aspergillus flavus QMS-8, which possesses the remarkable ability to thrive and grow in environments characterized by the presence of multiple extreme stress factors. Multiple stress factors refer to a combination or simultaneous presence of various adverse conditions or environmental pressures that can negatively affect the growth, survival, and normal functioning of microorganisms. These stress factors can include but are not limited to extreme temperatures, high levels of salinity, acidity or alkalinity, high pressure, low nutrient availability, and the presence of toxic compounds.
2 Methodology
The chemicals employed in the experimentation entail Acid Orange 52, also known as methyl orange (Sigma-Aldrich), Sabouraud’s dextrose broth (Oxoid), technical agar (Oxoid), and 0.05% Tween 80 solution was employed as a medium for fungal conidial suspension. Mineral salt media was prepared by dissolving Na2HPO4 ∙ 2H2O (7.8 g), KH2PO4 (6.8 g), MgSO4·7H2O (0.2 g), NaNO3 (0.085 g), Ca(NO3)2∙4H2O (0.050 g), C6H8FeNO7 (0.01 g), and solution trace elements (10 ml) in 1 L of distilled water. Simulated textile effluent (STE) was composed of acetic acid (99.9%): 0.15 ml; (NH4)2Cl: 108.0 mg; KH2PO4: 67.0 mg; NaHCO3: 840.0 mg; MgSO4 ∙ 7H2O: 38.0 mg; CaCl2: 21.0 mg; FeCl3 ∙ 6H2O: 7.0 mg; glucose: 860 mg, per liter of distilled water. All substrates and reagents used in the enzyme assays were purchased from Sigma-Aldrich unless mentioned otherwise. While High-performance liquid chromatography (HPLC) grade reference standards and solvents were utilized to examine the generation of aromatic amines in decolorized samples through HPLC.
2.1 Sampling and Isolation of Indigenous Fungal Flora
The fungal strain was obtained from the rhizosphere of Azadirachta indica (Neem tree) located in the garden area of the Federal Urdu University of Arts, Science, & Technology, Karachi, Pakistan, employing the spread plate method as described by Laraib et al. (2020). The fungal isolates were characterized by verifying the colonial features on Sabouraud dextrose agar (SDA) medium and microscopic traits using Lactophenol cotton blue as mounting fluid and stain.
2.2 Qualitative Screening
Preliminary screening was executed by cutting the fungal disks (1 cm) from the periphery of the colony and inoculating on the center of the SDA medium amended with 0.01% Acid Orange 52 (modified SDA). The plate was kept at 28 °C for 7 days. After the incubation period, the plates were examined for the formation of clear transparent halos around the fungal colony as an indication of dye degradation by fungi.
2.3 Quantitative Screening
The shake flask method was adopted to determine the extent of Acid Orange 52 degradations by fungal isolate. In this procedure, an individual fungal disk (a spherical piece of agar medium embedded with fresh mycelium was inoculated into test flasks containing 20 mL of Sabouraud’s dextrose broth supplemented with 0.01% Acid Orange 52 (modified SDB), and uninoculated flasks served as the control. All flasks were incubated at 28 °C for 3 days on an orbital shaker at 150 rpm. Following that, a 4-mL sample was drained aseptically from each flask, filtered, and then centrifuged at 5000 rpm for 15 min to separate the biomass. The percentage of dye removal of each fungal culture was calculated by measuring the absorbance of the cell-free culture supernatant (CFCS) spectrophotometrically at 505 nm (Gola et al., 2021).
2.4 Molecular Identification
The promising culture, A. flavus QMS-8, was selected for further studies and sent to Beijing Genomics Institute (BGI), China, for DNA extraction and sequencing. The analysis of obtained nucleotide sequences was carried out using the tool BioEdit Sequence Alignment Editor (7.2.5, Ibis Biosciences. USA). The generation of alignments with the existing DNA database was accomplished through BLAST-NCBI (Basic Local Alignment Search Tool of the National Center for Biotechnology). The evolutionary interrelations among a set of species were depicted by constructing a phylogenetic tree employing the neighbor-joining method.
2.5 Cultural Screening for Aflatoxin Production
A cultural technique of ammonium hydroxide vapor-induced color change was adopted to assess the aflatoxigenic potential of one of the promising strains identified as A. flavus QMS-8. A colony pre-grown on a potato dextrose agar (PDA) plate was inverted onto the lid containing 1 to 2 drops of concentrated ammonium hydroxide for a few minutes. The reverse of the colony was then observed for the emergence of a plum-red color as a sign of an aflatoxin-positive strain (Ambachew et al., 2021).
2.6 Preparation of Conidial Suspension
A. flavus QMS-8 was refreshed on SDA plates for 1 week, and conidia were transferred aseptically into flasks containing 50 mL of sterile 0.05% Tween 80 (v/v) solution. The flask was then kept in a shaking incubator (120 rpm) at 28 °C for 30 min. Subsequently, 10 µL from each conidial suspension was taken for conidial count using a hemocytometer. This suspension was immediately employed as inoculum for further experimentation.
2.7 Optimization of Various Nutritional and Physicochemical Factors for the Effective Degradation of Acid Orange 52
A variety of parameters were optimized in minimal salt media (MSM) to augment the decolorization efficiency of A. flavus QMS-8 for Acid Orange 52 by utilizing the one-factor-at-a-time approach. To determine the effect of agitation on the biodegradation efficiency of A. flavus QMS-8, MSM supplemented with 0.01% of Acid Orange 52 dye was used. All test flasks were inoculated with 1% of conidial suspension (v/v), whereas uninoculated flasks served as control. One set of experiments was conducted at room temperature in an orbital shaker at 120 rpm for 120 h, while another set was incubated without shaking. In a similar fashion, the effect of various concentrations of Acid Orange 52 was evaluated by adding different concentrations (from 100 to 1000 ppm) of the dye to MSM (50 mL) under agitation. Likewise, the effect of different carbon sources (glucose, fructose, lactose, maltose, mannitol, and starch) at a concentration of 1% was also scrutinized, maintaining previously known factors, i.e., agitation at 120 rpm and 300 ppm of the dye, unchanged. The influence of various nitrogen sources (peptone, yeast extract, ammonium sulfate, and ammonium nitrate) on fungal dye degradation was also investigated by incorporating 1% of each nitrogen source into the MSM at optimum conditions (agitation at 120 rpm, 300 ppm of the dye, and 1% fructose). To corroborate the optimum pH for the process of dye degradation, the pH of the growth medium was adjusted from 5.0 to 9.0 using 1 M HCl or 1 M NaOH, with optimized parameters such as shaking (120 rpm), Acid Orange 52 (300 ppm), 1% fructose, and 1% ammonium nitrate. Similarly, the effect of temperature on the degradation profile of Acid Orange 52 was observed by incubating the inoculated media at different temperatures (20, 30, and 40 °C) for the desired duration and ideal settings, i.e., agitation at 120 rpm, 300 ppm of the dye, 1% fructose, 1% ammonium nitrate, and pH 5.0 were kept constant. The impact of salinity on the decolorization process was also evaluated by varying the amount of NaCl (1, 5, and 10%) in the growth media supplemented with 300 ppm of the dye and 1% fructose, 1% ammonium nitrate and kept under shaking condition (120 rpm) at pH 5.0 and 30 °C.
In all experiments, 4 mL of the media from each flask was taken regularly after 24 h. The supernatant was prepared and analyzed spectrophotometrically to determine the percent decolorization (%) of the dye.
2.8 Whole-Cell Immobilization of A. flavus QMS-8 on Carrier Materials
The 25 natural and synthetic carrier materials, chosen due to their stability and cost-effectiveness were jute, polyethylene polymer, rice hull, plastic disks, sand, gravel, natural loofah sponge, corn cob, wood chips, used steel wool, orange peel, banana skin, nylon net, polyurethane foam, plastic net, wheat straw, coconut bagasse, sugarcane bagasse, wheat bran, water beads, walnut shells, scouring pad, sandpaper, pistachio shells, and sunflower seed shells. The supports were washed thoroughly with distilled water. Flasks containing 200 mL of SDB and 15 pieces of carrier material were autoclaved, inoculated with 10 mL of conidial suspension, and kept on a rotary shaker at room temperature for 12–15 days for complete adsorption.
2.9 Configuration of Stirred Tank Reactor for the Removal of Acid Orange 52 by Immobilized A. flavus QMS-8
A bench-top stirred tank bioreactor (STR) comprised of an overhead impeller and a vessel body (2 L volume) was designed and operated in continuous flow mode at room temperature and pH 5.0 under aerobic conditions. STR was fed with a working volume of 1 L of STE containing an optimized concentration of Acid Orange 52 and inoculated separately with different carrier materials with immobilized fungal mycelia. The hydraulic retention time was maintained at 12–120 h. The effluent samples were manually extracted every 12 h, examined for percent of dye degradation, and utilized for physicochemical and toxicity analyses. To investigate the dye adsorption on the carrier materials, similar experiments were also performed using carrier supports without immobilized cells.
2.10 Recycling Potential of Immobilized A. flavus QMS-8
In another experiment using the same procedure, the recycling efficiency of the immobilized A. flavus QMS-8 was evaluated. For this purpose, STR was run and fed with immobilized carrier material yielding maximum decolorization of Acid Orange 52 in STE. Afterward, it was removed from the reactor tank and then reused for the decolorization of fresh STE.
2.11 Decolorization Assay
The extent of dye decolorization was estimated by the method reported by Gola et al. (2021), by analyzing the CFCS spectrophotometrically at the wavelength of 505 nm.
After the incubation period, 4 mL of sample was taken aseptically from each flask, filtered, and centrifuged at 5000 rpm for 15 min to separate biomass. The CFCS was then analyzed spectrophotometrically at 505 nm for Acid Orange 52 (Gola et al., 2021).
The percentage of dye removal was calculated by using the following equation:
where the Initial absorbance is the absorbance taken at 0 h after the inoculation and the Final absorbance is the absorbance taken periodically after every 12 h of inoculation of fungal culture.
Biodegradation of Acid Orange 52 was also validated by scanning the treated (decolorized) and untreated (colored) samples using a UV-Vis spectrophotometer (UV-1800, Shimadzu, Japan) at respective λ maxima and changes in absorbance spectrum were observed.
2.12 Scanning Electron Microscopic Analysis of Immobilized Fungal Mass
The effects of fungal immobilization and biosorption on the surface morphology of the most promising carrier material were explored by scanning electron microscope (SEM) analysis (JEOL, Model number: JSM-6380A, Japan).
2.13 Assessment of Enzyme(s) Involved in the Dye Removal Process
To substantiate and estimate the participation of some major ligninolytic enzymes (lignin peroxidase, manganese peroxidase, and laccase) in the biodegradation of textile dyes, the fungal culture was inoculated in MSM supplemented with 100 ppm of Acid Orange 52 and incubated for 120 h in optimized parameters. Medium from flasks was drawn intermittently every 24 h and processed for the preparation of CFCS, then the activity of each three enzymes was assayed.
2.13.1 Laccase (Lac) Assay
The activity of laccase was detected by the oxidation of 2,2′-azino-bis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) into intense azure-colored cation radicals as specified in the protocol (Wolfenden & Willson, 1982). The reaction mixture contained 100 μL of ABTS, 980 μL of sodium malonate buffer, and 10 μL of CFCS as the laccase source. It was incubated at 37 °C for 5 min, and then OD420 was recorded against the reference blank.
2.13.2 MnP Assay
The manganese peroxidase (MnP) assay was performed according to the method of Glenn and Gold (1985) using 2,6-dimethoxy phenol (DMP) as substrate. The standard reaction mixture for MnP assay was prepared by adding 150 μL of DMP, 600 μL of sodium malonate buffer, 1650 μL of distilled water, 150 μL of MnSO4, 100 μL of H2O2, and 100 μL of CFCS and then incubated for 10 min at 37 °C. The changes in the absorbance of the reaction mixtures were monitored at 270 nm.
2.13.3 LiP Assay
Lignin peroxidase (LiP) activity was determined through the oxidation of veratryl alcohol (3,4-dimethoxybenzylalcohol) to veratryl aldehyde as elaborated (Tien & Kirk, 1984). The LiP assay mixture consisted of tartrate buffer (1 mL), veratryl alcohol (1 mL), H2O2 (500 μL), and 100 μL of CFCS. Afterward, 10-min incubation at 37 °C was carried out and OD310 was noted.
2.14 High-Performance Liquid Chromatography
To examine the generation of aromatic amines as a dye degradation product, decolorized samples were investigated through HPLC (Shimadzu LC-20AT) using six reference standards (p-anisidine, aniline, diphenylamine, pyridine, 3,3-dimethyl benzidine, and o-toluidine). The samples (test and reference standards) were extracted, and the volume of the mixtures was adjusted to 100 mL (with HPLC-grade methanol). Following that, samples were filtered using 0.45 µm membrane filters (Millex-HV). The prepared samples were re-filtered through 0.2 µm membrane filters (Millex-HV) and then injected into the column (LiChorCART 250-4 Cat 1.50982 HPLC Cartridge Merck KGaA 64271 Damstadt, Germany, Li Chrospher 100 RP-8) of the HPLC system.
2.15 Physicochemical Analysis
Treated and untreated STE samples were tested by using standard methods of biological oxygen demand (APHA 5210 B), chemical oxygen demand (APHA 5220 C), total dissolved solids (APHA 2540 C), and total suspended solids (APHA 2450 D) as specified by American Public Health Association, American Water Works Association, Water Environment Federation.
2.16 Evaluation of Phytotoxicity
The biological toxicity of treated (decolorized) and untreated (colored) samples were studied on Triticum aestivum (a monocot species) and Phaseolus mungo (a dicot species) through a seed germination assay. For this purpose, ten uniform-sized seeds of each crop plant for control, treated (decolorized), and untreated (colored) samples were selected and then dipped into sodium hypochlorite solution (1.2%) for 4–5 min for surface sterilization. Next, the seeds were rinsed in sterile distilled water five times, dried with sterile filter paper, and transferred to Petri plates already containing 5 mL of control, treated or untreated STE samples, and covered with filter paper. The plates were incubated under ambient conditions. Each plate was inoculated with 5 mL of the respective samples for seven consecutive days. At the end of the seventh day, the percent of germination (number of germinated seeds), length of plumule (shoot), and radicle (root) were recorded to evaluate the effect of individual treatment on plant growth.
2.17 Microbial Toxicity Assessment with Saccharomyces cerevisiae
The cytotoxic potential of untreated Acid Orange 52 and decolorized STE (treated) samples was determined by employing S. cerevisiae as the test strain (Mendes et al., 2011). For this, the strain was cultivated in MSM. Following the 24-h incubation period, the growth of S. cerevisiae in the medium was assessed by taking the OD640 value = 0.1 as an indication of the mid-exponential growth phase. Then, 1 mL of the pre-grown cell suspension was inoculated separately in flasks containing 50 mL of decolorized or untreated STE samples and incubated at 37 °C for 24 h. The influence of the dye and its degraded metabolites on cell density was evaluated by monitoring the change in OD640 value against a negative control (S. cerevisiae grown up in the absence of treated or untreated dye samples) after 24 h of incubation. The level of toxicity was estimated as the percentage of growth inhibition using the following equation:
where
- ODI640:
-
is the absorbance value taken in the absence of effluent samples (negative control), and
- ODF640:
-
is the absorbance value taken in the presence of effluent samples (Test).
2.18 Statistical Analysis
All experiments were performed in triplicate and the findings were declared as Mean ± standard deviation (Tahir & Yasmin, 2021). Experimental data were analyzed by one-way and two-way analysis of variance (ANOVA) using GraphPad Prism (version 6.0).
3 Results and Discussion
3.1 Isolation and Screening of Fungal Isolate for the Removal of Acid Orange 52
In view of the environmental impacts of synthetic dyes in conjunction with the exploitation of fungal mycelia as dye degrader/decolorizer, an autochthonous filamentous fungus A. flavus QMS-8 was isolated and explored in the present investigations for the bioremediation of hazardous colorant, Acid Orange 52. This reactive dye is affiliated with the mono-azo class, frequently utilized to dye fabrics, and widely known for its biotoxicity and recalcitrant potential (Sonal et al., 2018).
The strain was found positive for dye removal in the qualitative screening as indicated by the emergence of a clear halo around the mycelia grown on dye-supplemented SDA plates. The zone of decolorization may be formed by the action of oxidoreductase enzymes (Husain, 2006). The strain was further evaluated through a quantitative assay using shake flask experiments which revealed that A. flavus QMS-8 eliminated 80% of the dye and was recognized as a proficient degrader of Acid Orange 52; hence, it was selected for further experimentation. Fungi exhibit different phenomena responsible for the removal of dye, including bio-adsorption or biosorption followed by biodegradation. In biosorption, fungi bind to the dye molecules due to the electrostatic attraction between negatively charged molecules of dye and the positively charged cell wall of the fungus (Aksu & Tezer, 2000). However, biodegradation enables fungi to deteriorate the dye by virtue of extracellular and intracellular enzymes (Ngo & Tischler, 2022). Some species belonging to the genus Aspergillus were also reported to produce dye-degrading oxidoreductase enzymes, such as laccase, manganese peroxidases, and lignin peroxidases (Kanayama et al., 2002). In another study, Namnuch et al. (2020) recognized the genus Aspergillus as the most prevalent producer of lignocellulose hydrolytic enzymes in soil.
3.2 Molecular Identification
Identification of A. flavus QMS-8 was evaluated by PCR amplification of the ITS region. The 18S rRNA gene sequence alignment and database comparison of selected strains revealed that A. flavus QMS-8 shared 99.84% homology with A. flavus isolate Type_ E_1 (Fig. 1). The nucleotide sequence was submitted to NCBI Gene Bank with accession number MT430879 for A. flavus QMS-8. Many strains of Aspergillus frequently occurred in dye-contaminated wastewater. The genus Aspergilli was reported to be the most prevalent fungus in textile wastewater and an efficient degrader of various complex dyes (Patel et al., 2022; Sheam et al., 2021).
3.3 Cultural Screening for Aflatoxin Production
Mycotoxigenic strains of A. flavus can liberate aflatoxins as a secondary metabolite, which is a potent hepatocarcinogen, immunosuppressant, and linked to adverse epigenetic effects. Therefore, the strain QMS-8 of A. flavus was also evaluated for the production of aflatoxin by using the simple cultural technique of Ammonium hydroxide vapor-induced color change method. The result revealed that A. flavus QMS-8 is a non-aflatoxigenic strain since no plum red color was detected on the underside of the colony.
3.4 Optimization of Various Nutritional and Physicochemical Factors for the Effective Degradation of Acid Orange 52
The growth and metabolic activities of fungi are greatly influenced by several environmental conditions. The availability of oxygen is the most important factor (Rani et al., 2014), as it supports fungal growth and aids in oxidative reactions for the degradation of azo dye. The strain A. flavus QMS-8 was found capable of exhibiting 64.3, 69.4, 73.2, 76.3, and 77.5% decolorization of Acid Orange 52 at 24, 48, 72, 96, and 120 h, respectively, when incubated with shaking (120 rpm). The removal of the dye considerably decreased by 25–41% when the strain was cultivated in static conditions without shaking (Fig. 2). Aeration increases the dissemination of nutrients and expansion of oxygen in the medium, which ultimately increases the fungal biomass and expression of oxidative enzymes. The decrease in dye removal by fungi under static conditions previously reported as static conditions tends to limit the oxygen supply in the inside layer of fungal mycelia, which results in a low expression of oxidative enzymes (Rani et al., 2014).
The amount of dye present in the medium significantly impacts decolorization as it affects the metabolic activities of fungi. Growth is retarded at high dye concentrations, adversely affecting decolorization. In the present study, the presence of Acid Orange 52 in the range of 100–500 ppm was considered the optimum tolerable concentration for A. flavus QMS-8. However, the highest decolorization was observed at 300 ppm (87.8%) after 120 h, whereas its dye removal ability declined significantly when much higher concentrations (600–1000 ppm) of the dye were incorporated in the media (Fig. 3). Concentrated dye solutions usually have a toxic influence on the decolorization potential of fungi by obstructing the active sites of oxidative enzymes involved in the degradation of dye (Ghodake et al., 2011).
The carbon source has a pronounced effect on fungal growth. Generally, monosaccharides are easily assimilated, and hence rapid growth is observed. Our results suggested that the presence of fructose in the medium triggered the maximum degradation of Acid Orange 52 (59.2%), followed by glucose (55.36%), maltose (50.1%), sucrose (42.43%), and lactose (33%) after 24 h by A. flavus QMS-8 (Fig. 4). The strain achieved maximum degradation in the presence of glucose as it is more easily assimilated than polysaccharides. However, the amount of carbon source should be at the optimal level. In another study, the presence of 5 g L−1 glucose increased the decolorization potential of Phanerochaete chrysosporium, whereas a further increase in glucose concentration decreased the rate of decolorization (Radha et al., 2005). Moreover, polysaccharides including cellulose, starch, and its derivatives negatively affected the rate of dye decolorization by fungi (Vaidyanathan et al., 2011), presumably by decreasing the growth rate.
In this study, the presence of inorganic nitrogen sources like ammonium nitrate and ammonium sulfate accelerated the rate of decolorization to 70.13 and 60.43%, respectively, after 24 h (Fig. 5). This agrees with Jilani et al. (2011), who reported that the incorporation of ammonium sulfate in the growth medium provoked the regeneration of NADH (electron donor) and therefore enhanced the decolorization of Solar golden yellow R by Pleurotus osttreatus species. The acidic pH of the medium plays a significant role in dye decolorization, whereas alkaline pH decreased the decolorization potential of fungal strains. Furthermore, pH 5.0 induced 86.6% decolorization after 48 h. However, a further increase in pH not only reduced the decolorization potential of A. flavus QMS-8 to 53.7% after 24 h but also disintegrated the fungal mycelia (Fig. 6). It was reported that the pH range from 3 to 6 resulted in effective dye decolorization by fungi (Baldrian, 2004; Mansur et al., 2003). Low pH resulted in an increased number of H+ ions on the fungal cell wall and facilitated the uptake of negatively charged dye molecules (Won et al., 2009).
Temperature plays a crucial role in fungal growth, for the activity of the degradative enzymes and stability of the dye (Bayramoǧlu et al., 2007). The strain maximally decolorized 86.6% of Acid Orange 52 after 72 h at 30 °C followed by decolorization at 40 °C, whereas its decolorization potential was reduced at 20 °C (Fig. 7). Previously, 40 °C was reported as an optimum temperature for azo dye decolorization by Cladosporium cladosporioides (Vijaykumar et al., 2005).
Sodium chloride is widely employed in various dyeing processes; therefore, the ability of fungi to decolorize the dye needs to be investigated in the presence of sodium chloride. A. flavus QMS-8 decolorized 71% of the dye in 1% NaCl concentration (Fig. 8). Further increase in salt concentration (> 5%) resulted in decreased decolorization (56.3%) of Acid Orange 52 because of competition between Na+ ions with the dye molecules for the binding sites. Though a significantly proliferated mycelial mass was observed when the strain was cultivated in the presence of 5 and 10% concentrations of NaCl, owing to the halophilic propensity of the QMS-8, Maurya et al. (2006) reported that uptake of Rhodamine B and Methylene Blue by Phellinus igniarius was diminished with an increase in the ionic strength of the medium (0.00001–0.1 mol L−1).
3.5 Operation of Stirred Tank Reactor for the Removal of Acid Orange 52 by Immobilized A. flavus QMS-8
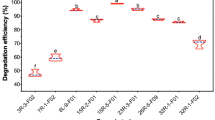
Immobilization of microbial cells for biotechnological and environmental applications offers several advantages including cost reduction and recyclability. The choice of immobilization matrix is of prime importance, based on cost, availability, inert nature, and porosity. In this study, several abundantly available and low-cost matrices were screened for their suitability to support fungal growth for dye decolorization. The data revealed that A. flavus QMS-8 immobilized on a natural luffa sponge in a stirred tank reactor (STR) resulted in 80.6% decolorization at a hydraulic retention time of 12 h. However, A. flavus QMS-8 immobilized over a natural luffa sponge removed 100% of the dye after 60 h (Fig. 9). Natural luffa sponge is a by-product of vegetable fiber of Luffa aegyptiaca or Luffa cylindrica. The matrix has high porosity, good mechanical strength, and low density. The structure of the matrix contains a parallel and antiparallel arrangement of fibers that provides free and open spaces for nutrient exchange. The space is also meant to physically confine fungal hyphae along and provide for the diffusion of oxygen. The candidature of natural luffa sponge for biosorption of nickel by an algal strain has been described previously (Akhtar et al., 2003).
3.6 Recycling Potential of Immobilized A. flavus QMS-8
Moreover, reprocessing proficiency of immobilized A. flavus QMS-8 was estimated seven times, retaining 75.6% of its decolorization activity. Akhtar et al. (2003) reported that a microalgal-luffa sponge immobilized disk can be reused for up to five cycles retaining 92.9% of the initial binding capacity for the biosorption of Ni (II). The experimental findings of this study suggested that the natural luffa sponge possessed all the required characteristics to be utilized as an ideal matrix for immobilization.
3.7 Scanning Electron Microscopic Analysis of Immobilized Fungal Mass
Morphological changes before and after the immobilization of fungal strains were studied by SEM as shown in Fig. 10a, b. SEM images of intact natural luffa sponge showed a number of inner channels, interstitial voids, and rough, hollow, and porous topography (Boudechiche et al., 2016). After the immobilization, the matrix became smooth, tighter, and compact; hence, fungal mycelia covered the superficial surfaces, inner channels, and voids. The inner channels of the luffa sponge effectively allowed the entry of essential elements and respiratory gases through fungal mycelia (Villena et al., 2010).
3.8 Assessment of Enzyme(s) Involved in the Dye Removal Process
In connection with the bioremediation of textile effluents, oxidoreductase enzymes, such as laccase, manganese peroxidase (MnP), and lignin peroxidase (LiP), have been extensively investigated. In the present study, A. flavus QMS-8 was found capable of removing 86.9% of Acid Orange 52 after 96 h and produced 0.17 IU mL−1 of Laccase, 0.16 IU mL−1 of MnP, and 0.06 IU mL−1 of LiP (Fig. 11). Among oxidoreductase enzymes, laccases (particularly derived from mold fungi) are fairly effective in efficiently degrading azo dyes via a highly non-specific free radical catalyzed mechanism rather than inducing direct reduction of azo bonds, thereby not coupled with the generation of hazardous aromatic amines (Kalme et al., 2009). A number of laccases extracted from the Trametes versicolor have also been reported for the decolorization of Indigo Carmine, Acid Red 1, Methyl Orange, and Reactive Blue 4 (Olha et al., 2022). Akin to laccases, fungal ligninolytic peroxidases, i.e., lignin and manganese peroxidases, have also been exploited as the most appropriate scheme owing to their extracellular synthesis under high oxygen tension with inadequate nutrients, no stearic selectivity, and immense oxidation capabilities to degrade miscellaneous azo dyes and other aromatic recalcitrant generated by textile industries.
3.9 Biodegradation Analysis Through UV–Vis Spectroscopy
The removal of the Acid Orange 52 by A. flavus QMS-8 was further assessed by scanning the STE-treated decolorized and untreated (colored) samples using a UV-Vis spectrophotometer and by comparing the changes in the absorbance spectrum of decolorized samples. The characteristic peak 1 at 505 nm depicts λmax for Acid Orange 52 in the STE at 0 h of inoculation with A. flavus QMS-8. It was observed that the specific absorption peak decreased considerably with the incubation time of 12 h on account of the degradation of Acid Orange 52 by A. flavus QMS-8. However, the characteristic peak of Acid Orange 52 disappeared completely after 24 h when A. flavus QMS-8 was cultivated in the dye-containing medium (Fig. 12). Though, the formation of any new peak(s) was not apparent which corresponds to the complete destruction of Acid Orange 52 moieties with no emergence of a new metabolite.
3.10 High-Performance Liquid Chromatography
To examine the generation of aromatic amines as a dye degradation product, decolorized samples were investigated through HPLC. The chromatogram exhibited one major peak at the retention time of ~ 1.762–1.804 min (λmax 254 nm) and ~ 1.778–1.807 min (λmax284 nm), which indicated the absence of any trace of standard (i.e., p-anisidine, aniline, diphenylamine, pyridine, 3,3-dimethyl benzidine, and o-toluidine) found in the decolorized samples (Fig. 13).
3.11 Physicochemical Analysis
The biochemical oxygen demand (BOD), chemical oxygen demand (COD), total dissolved solids (TDS), and total suspended solids (TSS) of the effluent samples (before and after treatment) were analyzed to evaluate the extent of Acid Orange 52 degradations by A. flavus QMS-8 by quantifying and comparing the organic loads. After employing an STR treatment system at a hydraulic retention time of 24 h, BOD, COD, TDS, and TSS values for Acid Orange 52 reduced from 2836, 3000, 3415, and 360 mg L−1 to 615, 1179, 916, and 145 mg L−1, respectively, and were found to be consistent with the NEQ standards (Mahmood et al., 2019). The national environmental quality (NEQ) standards refer to the specific quality standards and guidelines set by the national regulatory authorities or standardization organizations to regulate the quality of treated effluent released into the environment. These standards outline the permissible limits of various parameters, substances, or contaminants present in the effluent after undergoing treatment processes. By adhering to NEQ standards for effluent treatment, industries and facilities are required to implement appropriate treatment technologies and processes to ensure that the quality of discharged effluent meets the established limits. Compliance with these standards helps prevent pollution, maintains the ecological balance of receiving water bodies, and minimizes potential harm to ecosystems, aquatic life, and public health. It is important to note that NEQ standards for effluent treatment may vary from one country to another (www. https://environment.gov.pk).
Previously, decreased BOD and COD values of real effluent, i.e., 78.1, and 77.6%, respectively, using a fungal consortium comprising Aspergillus flavus and Fusarium oxysporium were reported (Selim et al., 2021). The drastic reduction of BOD, COD, TDS, and TSS values suggested the significance of STR treatment technology for the degradation of azo dyes by A. flavus QMS-8, even though it is imperative to explore the final residues of dye mineralization to make this system an ecologically perfect and economical alternative to conventional treatment methods.
3.12 Evaluation of Phytotoxicity
The use of treated textile wastewater for irrigation directly influences the fertility of the soil. Therefore, it is worthwhile to assess the phytotoxicity of the treated textile effluent. The biological toxicity of untreated (colored) and treated (decolorized) samples was studied on T. aestivum and P. mungo through seed germination assay. Results showed that the untreated sample of Acid Orange 52 drastically affected the germination rate, length of plumule, and radicle of T. aestivum and P. mungo (Table 1). However, decolorized samples of the dye improved the germination rate and the length of plumules and radicles of T. aestivum and P. mungo, compared to results obtained from untreated samples, however, the control plants grown without dye showed better development.
3.13 Microbial Toxicity Assessment with Saccharomyces cerevisiae
Bioassays are essential to evaluate the toxic effects of industrial effluent that might interact with the DNA of aquatic organisms (Schmitt et al., 2005). To evaluate the cytotoxic potential of both the untreated and treated effluent samples, toxicity analysis was conducted employing S. cerevisiae as the test strain. The results showed that the untreated dye sample of Acid Orange 52 caused 86.9% growth inhibition of S. cerevisiae. However, only 27.8% growth inhibition of S. cerevisiae was observed for treated Acid Orange 52 (Fig. 14). Hence, the treated dye effluents were found to be nontoxic in a eukaryotic model, i.e., S. cerevisiae.
4 Conclusions
This research underlined compelling evidence for the effective degradation of Acid Orange 52 by a fungal isolate A. flavus QMS-8 when immobilized on a natural luffa sponge employed under STR conditions. At the hydraulic retention time of 12 h, the strain demonstrated 80.6% decolorization. The observed degradation process was likely mediated by the combined action of three enzymes laccase, LiP, and MnP. Moreover, UV-Vis and HPLC analysis as well as phyto- and microtoxicity assays have validated the outstanding participation of A. flavus QMS-8 for the detoxification of Acid Orange 52 thereby suggesting this system, a potential technology, environmentally sound, manageable, and feasible mode to deal with constrained colored wastewater. Though, there is a need for further research to unlock new insights into the genomics of fungi involved in dye degradation along with integrated DO measurements to fully comprehend the metabolic intricacies and oxygen demands of the fungi in similar cultivation/processes, paving the way for the development of innovative strategies for environmental remediation, biotechnological applications, and sustainable management of dye-contaminated sites.
Data Availability
All data generated or analyzed during this study are included in this published article.
References
Adegoke, K. A., & Bello, O. S. (2015). Dye sequestration using agricultural wastes as adsorbents. Water Resources and Industry, 12, 8–24. https://doi.org/10.1016/j.wri.2015.09.002
Akhtar, N., Iqbal, J., & Iqbal, M. (2003). Microalgal-luffa sponge immobilized disc: A new efficient biosorbent for the removal of Ni(II) from aqueous solution. Letters in Applied Microbiology, 37(2), 149–153. https://doi.org/10.1046/j.1472-765X.2003.01366.x
Aksu, Z., & Tezer, S. (2000). Equilibrium and kinetic modeling of biosorption of Remazol black B by Rhizopusarrhizus in a batch system: Effect of temperature. Process Biochemistry, 36(5), 431–439. https://doi.org/10.1016/S0032-9592(00)00233-8
Ambachew, M., Dawit, K., Migbaru, K., & Pawlos, W. (2021). Magnitude of aflatoxigenic Aspergillus species, level of aflatoxin B1, and associated factors in stored feed at poultry farms in Dire Dawa, Ethiopia. Veterinary Medicine International, 2021. https://doi.org/10.1155/2021/6638083
Araújo, C. A. S., Ferreira, P. C., Pupin, B., Dias, L. P., Avalos, J., Edwards, J., Hallsworth, J. E., & Rangel, D. E. N. (2020). Osmotolerance as a determinant of microbial ecology: A study of phylogenetically diverse fungi. Fungal Biology, 124, 273–288. https://doi.org/10.1016/j.funbio.2019.09.001
Baldrian, P. (2004). Purification and characterization of laccase from the white-rot fungus Daedaleaquercina and decolorization of synthetic dyes by the enzyme. Applied Microbiology and Biotechnology, 63(5), 560–563. https://doi.org/10.1007/s00253-003-1434-0
Bayramoǧlu, G., Yakup, & Arica, M. (2007). Biosorption of benzidine-based textile dyes “Direct Blue 1 and Direct Red 128” using native and heat-treated biomass of TrametesVersicolor. Journal of Hazardous Material, 143, 135–143. https://doi.org/10.1016/j.jhazmat.2006.09.002
Boudechiche, N., Mokaddem, H., Sadaoui, Z., & Trari, M. (2016). Biosorption of cationic dye from aqueous solutions onto lignocellulosic biomass (Luffa cylindrica): Characterization, equilibrium, kinetic and thermodynamic studies. International Journal of Industrial Chemistry, 7(2), 167–180. https://doi.org/10.1007/s40090-015-0066-4
Bruno, L., Cíntia, Z. F. P., João Alencar, P., & Julio, C. P. (2019). Effects of textile dyes on health and the environment and bioremediation potential of living organisms. Biotechnology Research and Innovation, 3(2), 275–290. https://doi.org/10.1016/j.biori.2019.09.001
Csillag, K., Emri, T., Rangel, D. E. N., & Pócsi, I. (2022). pH-dependent effect of Congo Red on the growth of Aspergillus nidulans and Aspergillus niger. Fungal Biology-Uk. https://doi.org/10.1016/j.funbio.2022.05.006
Demirci, O., & Hamamci, D. A. (2013). Antioxidant responses in Phanerochaete chrysosporium exposed to Astrazone Red FBL textile dye. Cell Biochem and Function, 31, 86–90. https://doi.org/10.1002/cbf.2865
Dos Santos, A. B., Cervantes, F. J., & vanLier, J. B. (2007). Review paper on current technologies for decolourisation of textile wastewaters: Perspectives for anaerobic biotechnology. Bioresource Technology, 98(12), 2369–2385. https://doi.org/10.1016/j.biortech.2006.11.013
Duarte, F., Morais, V., Maldonado-Hódar, F. J., & Madeira, L. M. (2013). Treatment of textile effluents by the heterogeneous Fenton process in a continuous packed-bed reactor using Fe/activated carbon as catalyst. Chemical Engineering Journal, 232, 34–41. https://doi.org/10.1016/j.cej.2013.07.061
Ghodake, G., Jadhav, U., Tamboli, D., Kagalkar, A., & Govindwar, S. (2011). Decolorization of textile dyes and degradation of mono-azo dye amaranth by Acinetobactercalcoaceticus NCIM 2890. Indian Journal of Microbiology, 51(4), 501–508. https://doi.org/10.1007/s12088-011-0131-4
Glenn, J. K., & Gold, M. H. (1985). Purification and characterization of an extracellular Mn(II)-dependent peroxidase from the lignin-degrading basidiomycete, Phanerochaetechrysosporium. Archives of Biochemistry and Biophysics, 242(2), 329–341. https://doi.org/10.1016/0003-9861(85)90217-6
Gola, D., Tyagi, P. K., Arya, A., Gupta, D., Raghav, J., Kaushik, A., Agarwal, M., Chauhan, N., & Srivastava, S. K. (2021). Antimicrobial and dye degradation application of fungi-assisted silver nanoparticles and utilization of fungal retentate biomass for dye removal. Water Environment Research. https://doi.org/10.1002/wer.1629
Husain, Q. (2006). Potential applications of the oxidoreductive enzymes in the decolorization and detoxification of textile and other synthetic dyes from polluted water: A review. Critical Reviews in Biotechnology, 26(4), 201–221. https://doi.org/10.1080/07388550600969936
Jamee, R., & Siddique, R. (2019). Biodegradation of synthetic dyes of textile effluent by microorganisms: An environmentally and economically sustainable approach. European Journal of Microbiology and Immunology, 1–5. https://doi.org/10.1556/1886.2019.00018
Kalme, S., Jadhav, S., Jadhav, M., & Govindwar, S. (2009). Textile dye degrading laccase from Pseudomonas desmolyticum NCIM 2112. Enzyme and Microbial Technology, 44(2), 65–71. https://doi.org/10.1016/j.enzmictec.2008.10.005
Kanayama, N., Tohru, S., & Keiichi, K. (2002). Purification and characterization of an alkaline manganese peroxidase from Aspergillus terreus LD-1. Journal of Bioscience and Bioengineering, 93(4), 405–410. https://doi.org/10.1016/S1389-1723(02)80075-5
Kaushik, P., & Malik, A. (2009). Fungal dye decolourization: Recent advances and future potential. Environment International, 35(1), 127–141.
Khalid, A., Arshad, M., & Crowley, D. E. (2008). Accelerated decolorization of structurally different azo dyes by newly isolated bacterial strains. Applied Microbiology and Biotechnology, 78, 361–369.
Laraib, Q., Shafique, M., Jabeen, N., Naz, S. A., Nawaz, H. R., Solangi, B., Zubair, A., & Sohail, M. (2020). Luffa cylindrica Immobilized with Aspergillus terreus QMS-1: An Efficient and Cost-Effective Strategy for the Removal of Congo Red using Stirred Tank Reactor. Polish Journal of Microbiology, 69(2), 193–203.
Lima, D. M. C. G., Costa, T. P. C., Emri, T., Pócsi, I., Pupin, B., & Rangel, D. E. N. (2021). Fungal tolerance to Congo red, a cell wall integrity stress, as a promising indicator of ecological niche. Fungal Biology-Uk, 125, 646–657. https://doi.org/10.1016/j.funbio.2021.03.007
Mahmood, Q., Shaheen, S., Bilal, M., Tariq, M., Zeb, B. S., & ZahidUllah, Ali A. (2019). Chemical pollutants from an industrial estate in Pakistan: a threat to environmental sustainability. Applied Water Science, 9, 47. https://doi.org/10.1007/s13201-019-0920-1
Mansur, M., Arias, M. E., Copa-Patiño, J. L., Flärdh, M., & González, A. E. (2003). The white-rot fungus Pleurotusostreatus secretes laccase isozymes with different substrate specificities. Mycologia, 95(6), 1013–1020. https://doi.org/10.1080/15572536.2004.11833017
Mathur, N., Bhatnagar, P., & Bakre, P. (2006). Assessing mutagenicity of textile dyes from pali (Rajasthan) using ames bioassay. Applied Ecology &. Environmental Research, 4(1), 111–118. https://doi.org/10.15666/aeer/0401_111118
Maurya, N. S., Mittal, A. K., Cornel, P., & Rother, E. (2006). Biosorption of dyes using dead macro fungi: Effect of dye structure, ionic strength and pH. Bioresource Technology, 97(3), 512–521. https://doi.org/10.1016/j.biortech.2005.02.045
Mendes, S., Farinha, A., Ramos, C. G., Leitão, J. H., Viegas, C. A., & Martins, L. O. (2011). Synergistic action of azoreductase and laccase leads to maximal decolourization and detoxification of model dye-containing wastewaters. Bioresource Technology, 102(21), 9852–9859. https://doi.org/10.1016/j.biortech.2011.07.108
Mishra, A., Kumar, S., & Pandey, K. (2011). A. Laccase production and simultaneous decolorization of synthetic dyes in unique inexpensive medium by new isolates of white rot fungus. International Biodeterioration and Biodegradation, 65(3), 487–493. https://doi.org/10.1016/j.ibiod.2011.01.011
Moawad, H., Abd El-Rahim, W. M., & Khalafallah, M. (2003). Evaluation of biotoxicity of textile dyes using two bioassays. Journal of Basic Microbiology, 43(3), 218–229. https://doi.org/10.1002/jobm.200390025
Namnuch, N., Thammasittirong, A., & Thammasittirong, S. N. (2020). Lignocellulose hydrolytic enzymes production by Aspergillus flavus KUB2 using submerged fermentation of sugarcane bagasse waste. Mycology, 12(2), 119–127. https://doi.org/10.1080/21501203.2020.1806938
Ngo, A. C. R., & Tischler, D. (2022). Microbial degradation of azo dyes: Approaches and prospects for a hazard-free conversion by microorganisms. International Journal of Environmental Research and Public Health, 19(8), 4740. https://doi.org/10.3390/ijerph19084740
Olha, O. Z., Olga, V. K., Mykhailo, O. K., Victor, M. A., & Alexander, N. S. (2022). Aliphatic oxime 3-(hydroxyimino)pentan-2,4-dione as new potential mediator for laccase-catalyzed decolorization of dyes. Journal of Environmental Chemical Engineering, 10(4), 108149. https://doi.org/10.1016/j.jece.2022.108149
Patel, H., Yadav, V. K., Yadav, K. K., Choudhary, N., Kalasariya, H., Alam, M. M., Gacem, A., Amanullah, M., Ibrahium, H. A., Park, J. W., Park, S., & Jeon, B. H. (2022). A recent and systemic approach towards microbial biodegradation of dyes from textile industries. Water, 14, 3163. https://doi.org/10.3390/w14193163
Radha, K. V., Regupathi, I., Arunagiri, A., & Murugesan, T. (2005). Decolorization studies of synthetic dyes using Phanerochaetechrysosporium and their kinetics. Process Biochemistry, 40(10), 3337–3345. https://doi.org/10.1016/j.procbio.2005.03.033
Rani, B., Kumar, V., Singh, J., Bisht, S., Teotia, P., Sharma, S., & Kela, R. (2014). Bioremediation of dyes by fungi isolated from contaminated dye effluent sites for bio-usability. Brazilian Journal of Microbiology, 45(3), 1055–1063. https://doi.org/10.1590/s1517-83822014000300039
Rehman, K., Shahzad, T., Sahar, A., Hussain, S., Mahmood, F., & Siddique, M. H. (2018). Effect of Reactive Black 5 azo dye on soil processes related to C and N cycling. Peer J, 6, 4802. https://doi.org/10.7717/peerj.4802
Rodríguez Couto, S. (2009). Dye removal by immobilised fungi. Biotechnology Advances, 27(3), 227–235.
Schmitt, M., Gellert, G., & Lichtenberg-Fraté, H. (2005). The toxic potential of an industrial effluent determined with the Saccharomyces cerevisiae-based assay. Water Research, 39(14), 3211–3218. https://doi.org/10.1016/j.watres.2005.05.034
Selim, M. T., Salem, S. S., Mohamed, A. A., El-Gamal, M. S., Awad, M. F., & Fouda, A. (2021). Biological treatment of real textile effluent using Aspergillus flavus and Fusarium oxysporium and their consortium along with the evaluation of their phytotoxicity. Journal of Fungi (basel), 7(3), 193. https://doi.org/10.3390/jof7030193.PMID:33803129;PMCID:PMC8001397
Selvaraj, V., SwarnaKarthika, T., Mansiya, C., & Alagar, M. (2021). An over review on recently developed techniques, mechanisms and intermediate involved in the advanced azo dye degradation for industrial applications. Journal of Molecular Structure, 1224, 129–195. https://doi.org/10.1016/j.molstruc.2020.129195
Sheam, M. M., Biswas, S. K., Ahmed, K. R., Syed, S. B., Hossain, M. S., Khan, M. S. A., Hasan, M. R., Zohra, F. T., & Rahman, M. M. (2021). Mycoremediation of reactive red HE7B dye by Aspergillus salinarus isolated from textile effluents. Current Research in Microbial Sciences, 2, 100056. https://doi.org/10.1016/j.crmicr.2021.100056
Slama, H. B., Chenari Bouket, A., Pourhassan, Z., Alenezi, F. N., Silini, A., Cherif-Silini, H., Oszako, T., Luptakova, L., Golińska, P., & Belbahri, L. (2021). Diversity of synthetic dyes from textile industries, discharge impacts and treatment methods. Applied Sciences, 11, 6255. https://doi.org/10.3390/app11146255
Solís, M., Solís, A., Pérez, H. I., Manjarrez, N., & Flores, M. (2012). Microbial decolouration of azo dyes: A review. Process Biochemistry, 47(12), 1723–1748. https://doi.org/10.1016/j.procbio.2012.08.014
Sonal, S., Singh, A., & Mishra, B. K. (2018). Decolorization of reactive dye Remazol Brilliant Blue R by zirconium oxychloride as a novel coagulant: optimization through response surface methodology. Water Science and Technology, 78(2), 379–389.
Sponza, D. T., & Işik, M. (2002). Decolorization and azo dye degradation by anaerobic/aerobic sequential process. Enzyme & Microbial Technology, 31(1–2), 102–110. https://doi.org/10.1016/S0141-0229(02)00081-9
Tahir, U., & Yasmin, A. (2021). Decolorization and discovery of metabolic pathway for the degradation of Mordant Black 11 dye by Klebsiella sp. MB398. Brazilian Journal of Microbiology, 52(2), 761–771. https://doi.org/10.1007/s42770-021-00470-x
Tien, M., & Kirk, T. K. (1984). Lignin-degrading enzyme from Phanerochaetechrysosporium: Purification, characterization, and catalytic properties of a unique H2O2-requiring oxygenase. Proceedings of the National Academy of Sciences, 81(8), 2280–2284. https://doi.org/10.1073/pnas.81.8.2280
Vaidyanathan, V. K., Selvaraj, D. K., Premkumar, P., & Subramanian, S. (2011). Screening and induction of laccase activity in fungal species and its application in dye decolorization. African Journal of Microbiology Research, 5(11), 1261–1267. https://doi.org/10.5897/ajmr10.894
Vijaykumar, M. H., Veeranagouda, Y., Neelakanteshwar, K., & Karegoudar, T. B. (2005). Decolorization of 1:2 metal complex dye Acid blue 193 by a newly isolated fungus, Cladosporium cladosporioides. World Journal of Microbiology and Biotechnology, 22(2), 157–162. https://doi.org/10.1007/s11274-005-9013-4
Villena, G. K., Fujikawa, T., Tsuyumu, S., & Gutiérrez-Correa, M. (2010). Structural analysis of biofilms and pellets of Aspergillus niger by confocal laser scanning microscopy and cryo scanning electron microscopy. Bioresource Technology, 101(6), 1920–1926. https://doi.org/10.1016/j.biortech.2009.10.036
Wolfenden, B. S., & Willson, R. (1982). Radical-cations as reference chromogens in kinetic studies of ono-electron transfer reactions: Pulse radiolysis studies of 2, 2′-azinobis-(3-ethylbenzthiazoline-6-sulphonate. Journal of the Chemical Society, Perkin Transactions, 1(2), 805–812.
Won, S. W., Vijayaraghavan, K., Mao, J., Kim, S., & Yun, Y. S. (2009). Reinforcement of carboxyl groups in the surface of Corynebacteriumglutamicum biomass for effective removal of basic dyes. Bioresource Technology, 100(24), 6301–6306. https://doi.org/10.1016/j.biortech.2009.07.063
Acknowledgements
The authors wish to thank all who assisted in conducting this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Laraib, Q., Shafique, M., Versiani, M.A. et al. Enhanced Degradation of Acid Orange 52 Using a Polyextremophilic Ascomycete in Stirred Tank Bioreactor. Water Air Soil Pollut 234, 509 (2023). https://doi.org/10.1007/s11270-023-06508-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-023-06508-0