Abstract
Predicted warmer temperatures and more frequent extreme climatic events in the southern Andes may affect the dynamics of the Patagonian forests. These environmental changes may differentially alter the probability of Nothofagus pumilio establishment across its altitudinal range of distribution. We monitored fruit fall, seedling emergence and survival at altitudinal distribution range of N. pumilio forests in Santa Cruz (49°22′ S—72°56′ W), Argentina. Fruit fall, seedling emergence and survival were tested in relation to drought, based on the SPEI (Standardized Precipitation Evapotranspiration Index), interacting with elevation. Fruit fall was consistently higher at low elevation and the abundance of fruits was not affected by drought intensity. Density of new-born seedlings was ~ 9 to 24 times lower at low- than at high-elevation plots in our first observations (2014 and 2016), characterized by warm-dry climatic conditions in spring-early summers. In contrast, seedling abundance was ~ 1.5 times larger at low elevations during relatively cold-wet growing seasons. Survival probability was explained by the interaction between SPEI and elevation. At low elevation and in dry periods, survival probability was lower (CI 54–72%) than in wet periods (CI 68–84%) but at high elevation similar survival was registered even with positive or negative SPEI values. Our results show interacting effects of elevation and drought on tree establishment at the elevation limits, with positive and negative drought effects at high and low elevations, respectively. Predicted increase of extreme drought events during the XXI century could be detrimental for N. pumilio establishment at dry, low-elevation forests.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Understanding the consequences of climate change on mountain’s forests requires a comprehensive view of tree life stages and species-specific processes along their ranges of distribution. At large-spatial scale, cold temperature is usually recognized as the limiting factor modulating plant performance at the upper-elevation limits, whereas at the lower and warmer borders of distribution, water supply, indirectly regulated by temperature, is the main forcing of plant growth (Hampe and Petit 2005). Therefore, the consequences of climate warming on plant vital rates are expected to differ spatially for tree species with wide elevation ranges. Empirical knowledge on the performance of plant reproduction at elevation boundaries could help us to understand vegetation responses to climate changes before the potential expansion or contraction of their range occurs (Harsch et al. 2009; Harsch and Bader 2011; Cagnacci et al. 2020). Demographic information, in particular establishment of new individuals, can provide the basis to predict population performance, as well as changes in distribution and in turnover rates in tree populations. Seedling emergence and survival are the stages in the trees’ life cycle most vulnerable to climate change (Grubb 1977; Peter and Ronald 1996; Hampe and Petit 2005; Engelbrecht et al. 2007; Tercero Bucardo et al. 2007).
Minor variations in environmental conditions at the elevational limits of the forest can induce changes in tree recruitment, but may affect to a lesser degree these demographic processes at intermediate sites with lower environmental stress (Kullman 2007; Kirdyanov et al., 2012). Whilst air temperature decreases along the elevation gradient with indirect effects on environmental humidity, atmospheric pressure gradually decreases and solar radiation increases with elevation (Körner and Paulsen 2004; Körner 2007a). Lower temperatures at higher elevations induce a shortening of the growing season, and in most cases, a reduction in the supply of nutrients from the soil, negatively impacting the growth rates with increasing elevations (Coomes and Allen 2007; Körner 2007b). In addition, trees at high elevation are more exposed to wind and experience longer persistence of snow cover than trees at lower elevation. In contrast, the increment in temperature at the lower forest boundary increases evapotranspiration, which in turn reduces soil water availability for tree recruitment and growth (Sánchez-Salguero et al. 2015). In this context, there is a greater possibility that environmental fluctuations on plant performance will manifest earlier at the extreme forest boundaries (Doak and Morris 2010), justifying the importance of conducting demographic studies in these focal locations.
Nothofagus (southern beech) forests in the Patagonian Andes from South America comprise one of the most singular and large mountain ecosystems in the planet (Donoso 1993). This genus dominates the upper treeline in the highest hostile Andean environment and their species are widely distributed throughout the region, from Central Chile (33.5°S) to Tierra del Fuego (56°S; Veblen et al., 1996). The Nothofagus pumilio ([Poepp. & Endl.] Krasser) forests are important as timber producers, regulators of water runoff and other ecosystem services to populations, like tourism and recreation, on both slopes of the Patagonian Andes (Ramírez et al. 1997; Martínez-Pastur et al. 2000; Bava and Rechene 2004).
Seed production and dispersal are prerequisites for successful establishment in the forest. A continuous decrease in seed production with increasing elevation has been reported for Nothofagus species, including N. solandri var. cliffortioides in New Zealand (Wardle 1980; Allen and Platt 1990) and N. pumilio in Southern Patagonia (Barrera et al. 2000; Cuevas 2000, 2002). Fruit fall, seed viability and seedling emergence in N. pumilio decrease with increasing elevation, suggesting that tree establishment at high elevation is regulated by two bottlenecks: seed production and seedling emergence (Cuevas 2000). In addition to seed production and germination, tree establishment could also be limited by seedling survival. The highest mortality rate of trees occurs during the first year of establishment since seedlings are extremely sensitive to variations in environmental factors (Maher and Germino et al. 2006; Soler et al. 2013; Cagnacci et al. 2020). Seedling mortality can be caused by desiccation, overheating, freezing, diseases or herbivory (Stevens and Fox 1991; Piper et al. 2006; Danby and Hik 2007). In addition, mortality can be also induced by competitive interactions between plants (Olofsson 2004). At the low-elevation Nothofagus forests in the ecotone with the Patagonian steppe, climate warming could impose an increased drought stress, limiting the survival of N. pumilio seedlings and trees. Conversely, seedling survival at higher elevations could benefit from the increase in temperature associated with the extension of the growing season and the stress reduction due to heavy snow accumulation (Daniels and Veblen 2004; Piper et al. 2013; Srur et al. 2016, 2018).
Based on the analysis of instrumental and proxy records, temperature in southern Patagonia has increased during the past century with a marked positive trend in the past few decades (Villalba et al. 2003; Falvey and Garreaud 2009; Masiokas et al. 2008, 2009; Lopez et al. 2010; Garreaud et al. 2013). According to Magrin et al. (2014), precipitation has decreased in southern Chile and Argentina since the 1960s concurrent with a warming trend close to 0.1ºC per decade. First, we here describe and analyse the temperature and precipitation variations during 2006–2020 in order to show the recent recurrent droughts in the years of our study. We hypothesize that temperature and precipitation variations differentially modulate tree recruitment at high- versus low-elevation boundaries in the southern Andes. Therefore, in this study we evaluate how seedling emergence and survival at upper and lower elevations vary in relation to climate in southern Patagonia. We postulate that a warmer climate would ameliorate the harsh environmental conditions for tree recruitment imposed by high elevations, but intensified water stress (drought and overheating) for seedlings at lower elevation forests. We test this hypothesis by comparing fruit fall, seedling emergence and survival in N. pumilio forest at low and high elevations over 6 years of changing climate in the southern Andes.
Materials and methods
Study area
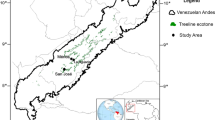
The study area is located in the Río de las Vueltas basin (49° S–72° W), Santa Cruz, Argentina (Fig. 1). All the area is under the influence of strong winds from the west throughout the year. At a regional scale, temperature decreases with elevation, whilst precipitation increases in the east–west direction (Villalba et al. 2003). Meteorological data from Los Huemules meteorological station (49°13′09″ S, − 72°57′38″ W), in the central sector of our study area, indicate a total annual precipitation over 1000 mm and a mean annual temperature of 6 °C.
N. pumilio is the dominant subalpine tree, endemic to the Patagonian Andes of Chile and Argentina (Veblen et al. 1996). The deciduous forests of N. pumilio belong to the SubAntarctic phytogeographical province of the Antarctic Region in southern South America (Cabrera 1976). In the study area, N. pumilio is the only tree species that constitute forest cover on the eastern slopes from ~ 500 to 1100 m asl. These species poor forests mainly contain an herbaceous layer and few shrubs, like Berberis microphylla, Ribes magellanicum, Chilliotrichum difussum, Maytenus magellanica, Gaultheria mucronata and Myoschilos oblongum. Trees grow up to 20 m in height with erect trunks up to 1.5 m in diameter (Roig et al. 1985). Leaves are deciduous, elliptic, 2–4 cm long with regular lobed margins. The fruit is a smallnut (4–7 mm) dispersed by wind in early spring. Seeds do not form a persistent soil bank, they remain dormant during winter and start germination during spring and summer (after October), later they can persist as seedlings for many years until canopy opens (Donoso 1993). N. pumilio may form seedling banks as it was recorded at xeric sites of northern Patagonia (Heinemann et al. 2000; Heinemann 2007).
Data collection
We collected data at 10 N. pumilio monospecific stands at low and high elevation at three sites along Rio Las Vueltas basin. Stands were located in private and national protected areas (Los Glaciares National Park, Estancia Los Huemules, and Estancia Río Toro) (Table 1; Fig. 1). Plots were located in old-growth, multi-aged erect stands without recent evidence of natural or anthropic disturbances (e.g. livestock grazing, fire or logging). None of our plots were located at N. pumilio “krummholz” stands, characterized by shrub-like stunted trees, usually present at stressed treelines or south-facing slopes with relatively short growing seasons due to a long snow cover period (Lara et al. 2005). Plots included 50 alive trees larger than 5 cm diameter at breast height (DBH) (Srur et al. 2008). To account for seedling emergence and seedling survival, inside each plot, 10 subplots (1 × 1 m, Fig. 2a) were established to mark and follow seedling’s fate, biannually between 2012 and 2018. All seedlings were tagged with plastic numbers to follow individual fate. Due to accessibility limitations by a broken bridge given the river flood, seedling survival could not be monitored in Ea. Río Toro for the year 2016. To estimate fruit fall we installed inside each plot 10 seed traps (diameter 25 cm wide and 35 cm in depth, Fig. 2b) and counted the number of fruits collected annually between February 2015 and 2018.
a Diagram of the field design to record N. pumilio demographic changes at the extremes of the elevation range. At high- and low-elevation forest boundaries, 10 subplots (1m2) for monitoring the establishment were located inside a plot with 50 adult trees. Inside each subplot all saplings and seedlings were tagged. Seedling emergence and survival were monitored every 2 years from 2012 to 2018. b Inside each area, 10 seed traps were used to quantify annual fruit fall between 2015 and 2018
Data analysis
Drought characterization
To identify the drought events in our study area, we used a multiscalar drought index based on instrumental climatic data. In the study area, unfortunately there are few meteorological stations and most are recent and often have missing data. To assess the reliability of the data from the meteorological station located in the centre of our study area at Ea. Los Huemules for the other sites we performed correlation analysis with the available data from the other stations in the area. One of the other stations is located in Los Glaciares National Park, which has temperature data from February 2015 to the present, but with missing data for the entire 2016. Unfortunately, this station has not recorded precipitation data. Another station is located at the outlet from Lago Milanesio, within the Ea. Río Toro and has temperature and precipitation records. Another station is located at the outlet from Lago Milanesio, within the Ea. Río Toro, and has temperature and precipitation records only from February 2016 to April 2020. Due to the lack of data for all our study period in those meteorological stations and considering the high correlation of data between all the stations located in the study area (r > 0.5, p < 0.001, see Mat. Supp. S1), we used only the data from the meteorological station at Ea. Los Huemules as it was the most complete and reliable, with records from 2006 to the present.
The Standardized Precipitation Evapotranspiration Index (SPEI) is calculated from the difference between the precipitation and the potential evapotranspiration. This last variable uses the Thornthwaite equation and the mean temperature data for the study area (McKee et al. 1993; Paulo et al. 2012). The SPEI was calculated using the SPEI package version 1.7 (Beguería and Vicente-Serrano 2017) of the free programming language R (R Core Team 2020). To describe trends in regional climate we analysed monthly precipitation and temperature deviations in relation to the period of 2006–2019 and show long-term climate in our study area. In our statistical model for fruit fall we calculated SPEI using climatic data from October to March (6 months), which was the current growing season when fruit were collected. To assess SPEI effect on seedling emergence and survival we used the SPEI calculated from October of two years before to March of the sampling year (18 months). Therefore, we included the growing season from the previous year until the current growing season and so, for example, for the data taken in 2014 the period comprised was from October 2012 to March 2014.
Statistical models
We conducted all analyses with R version 4.0.2 (R Core Team, 2020). To evaluate fruit fall, seedling emergence and survival in relation to elevation and SPEI we applied Generalized Linear Models (GLMMs) using the glmer.nb() and glmer() function of the package lme4 (Bates et al. 2015). Fruit fall, seedling emergence and survival were tested in relation to elevation and SPEI as fixed factors. In all models site was included as random factor in order to control and consider variation of land management and abiotic factors between locations. To test for variation in fruit fall and seedling emergence we assumed a Negative Binomial (log link) distribution of errors to remedy overdispersion (Crawley 2005). To test for variation in survival we assumed a Binomial (logit link) distribution of errors. Interactions between fixed factors were included and tested if required for empirical support in each model.
Results
The study period was characterized by large climatic variability in terms of monthly temperature and precipitation (Fig. 3). In our study area, after 2012, recurrent positive deviations in temperature and negative precipitation deviations were registered along the growing seasons. Therefore, negative SPEI values were recurrent in our study period from 2012 to 2016 (Fig. 3).
a Monthly variations of precipitation and temperature deviations (in relation to the period of 2006–2020) from Los Huemules meteorological station (49°13′09″ S, − 72°57′38″ W), southern Patagonia, Argentina. To emphasize the long-term climate fluctuations, variations in precipitation (blue) and temperature (red) are shown as 5-month moving averages. b Long-term monthly variations of SPEI (black), dry periods (SPEI < 0) are highlighted in red. The initial experimental setting to monitor seedling emergence and survival was conducted in February 2012 and followed by periodic samplings in February 2014, 2016 and 2018
Fruit fall was consistently more abundant at low- than at high elevations between 2015 and 2018 (Negative binomial GLMM, χ2 = 10.41, p value < 0.05, Mat. Supp. S2) but the magnitude of this difference was variable between years (Fig. 4). There was no interaction between elevation and SPEI to explain fruit fall, neither SPEI affected fruit fall independently. Fruit production showed the lowest record at both elevations in 2016 and the maximum in 2017 (Fig. 4).
In average we estimated a density of 8676 ± 2188 and 6767 ± 2231 new seedlings/ha at low and high elevations, respectively, but the abundance of emerged seedlings showed temporal variation between high and low elevations. Seedling emergence was explained by elevation interacting with SPEI (Negative binomial GLMM, χ2 = 51.88, p value < 0.001, Mat. Supp. S3), reflecting the importance of water deficit for regeneration. Seedling emergence was lower at low-elevation forests in 2014 (low: 0.06 ± 0.40 vs. high: 0.54 ± 1.36) and 2016 (low: 0.01 ± 0.11 vs. high: 0.24 ± 0.65), but higher in 2018 (low: 1.46 ± 3.30 vs. high: 0.95 ± 1.66). Therefore, in drought periods characterized by negative SPEI values (e.g. SPEI = − 0.99), density of emerged seedlings was higher at high elevation but in wet periods (e.g. SPEI = 0.32) the abundance of emerged seedlings was similar at both elevation boundaries (Fig. 5). Seedling survival probability was explained by the interaction between elevation and SPEI index (Binomial GLMM, χ2 = 13.48, p value < 0.05, Mat. Supp. S4). At low elevation and in drought periods, survival probability was lower (CI = 0.54–0.72) than in wet periods (CI = 0.68–0.84). At high elevation similar survival probability was estimated with positive (CI = 0.67–0.82) or negative SPEI values (CI = 0.71–0.85) (Fig. 6).
Density of seedling emergence, mean ± CI 90%, explained by elevation and climate (SPEI index) interaction. In periods of drought (SPEI < 0) seedling emergence was higher at the upper-elevation boundary of N. pumilio forest, but differences in establishment were null in the absence of drought. The climatic anomalies used to show the response are consistent with SPEI -0.99 and 0.32 for our sampling period in 2014 and 2018, respectively, and were obtained predicting a Poisson GLMM
Conclusion and discussion
Our study reports different tree regeneration responses to climate variability at contrasting elevation boundaries of Patagonian mountain forests. Although fruit fall was consistently larger at lower than at higher elevations, seedling establishment along elevation varied over time, particularly in relation to drought conditions. Density of seedling emergence and seedling survival was lower at low elevation in periods with droughts, characterized by a negative SPEI index.
Across our study area, seedling survival probabilities were larger in the upper boundary of elevation during periods of drought. These findings contradict previous studies showing scarce tree recruitment at high-elevation erect N. pumilio forests (Barrera et al. 2000) in Tierra del Fuego (latitude ~ 54°S) or indistinctly along the elevation gradient at 40°S in northern Patagonia (Cagnacci et al. 2020). The difference in the results between these studies and ours could be associated with differences in the climatic conditions prevailing in each study during the monitoring period of establishment. In our region, the biannual periods 2012–2014 and 2014–2016 were characterized by a dry and warm spring–summer climate conditions, whilst abundant rainfall was recorded in the period 2016–2018. Seedling emergence was less abundant at the lower forest boundary during the periods with drought, suggesting that N. pumilio germination in low-elevation forests is limited by warm and dry climatic anomalies. Consistent with our hypothesis, drought conditions favoured establishment at the high elevation but limited it at the lower forest edge. Our observations indicate that establishment patterns are strongly modulated by climatic variability interacting with elevation. Therefore, differences with Barrera et al. 2000 and Cagnacci et al. 2020 could be due to the specific climate conditions during the seedling monitoring, the shorter monitoring time, and the more humid and colder environments at the sampling sites in Tierra del Fuego or dryer climate in Northern Patagonia at 40ºS.
The concept of increasing reproductive capacity in N. pumilio forests with increasing elevation was early suggested by Rusch (1993), who noted that although shorter growing seasons at high elevation limit tree productivity, recruitment is benefited by more abundant precipitations that mitigate soil water deficit. Consistent with Rush (1993) observations, the main cause of seedling mortality at low-elevation forest appears to be summer desiccation, as typified by the orange coloration of the seedling first leaves or cotyledons observed in the field. Most monitored seedlings in our study were from sexual reproduction since they were initially tagged with the presence of cotyledons. In seed-bearing plants, cotyledons are embryonic leaves, whereas plants from asexual sprouts do not shown them. Our results support the untested hypothesis from Rusch (1993), indicating that larger seedling survival probability could explain higher seedling density at high- than at low-elevation boundary.
Extreme climatic events, rather than the mean climate, seem to be more important for terrestrial ecosystem dynamics (Holmgren et al. 2006; Suarez and Kitzberger 2008; Amoroso et al. 2012). Extreme dry-warm summers between 2012 and 2016 at our study area exacerbated the recorded differences in establishment at both elevation extremes. Cagnacci et al. (2020) showed, with an experimental approach, that seedling mortality was regulated by air temperature during the first year of growth, but by relative air humidity during the second year of their experiments. Therefore, consistent with our results, Cagnacci et al. (2020) showed that establishment is largely dependent on climate conditions and emphasizes the need of long-term monitoring to properly characterize regeneration dynamics. In ecological studies, the use of a stationary temporal assumption could limit the applicability of results beyond the monitoring range. In turn, it has recently been emphasized that climate change is introducing unprecedented non-stationary changes that should be considered for forecasting the dynamics of biological systems (Wolkovich et al. 2014). Our findings highlight the benefits of long-term monitoring over time to understand the responses of forest species to changes in climate along elevational gradients.
Detrimental effects of climate warming for the conservation of plant populations at low elevation, called rear edges, have been reported in Europe (Peñuelas and Boada 2003). Our results support the rear edge hypothesis for forests at the lower elevation boundary in Southern Patagonia, particularly under recent prevalent warm-dry springs and summers. A challenge for future research is to monitor with a life-cycle approach how population dynamics of this Patagonian endemic tree would respond if climatic extremes increase their frequency as predicted for the near future.
Data availability
The data that support the findings of this study are available on request from the corresponding author.
Code availability
The code that supports the findings of this study is available on request from the corresponding author.
References
Allen RB, Platt KH (1990) Annual seedfall variation in Nothofagus solandri (Fagaceae), Canterbury, New Zealand. Oikos 57:199–206
Amoroso MM, Suarez ML, Daniels LD (2012) Nothofagus dombeyi regeneration in declining Austrocedrus chilensis forests: effects of overstory mortality and climatic events. Dendrochronologia 30:105–112. https://doi.org/10.1016/j.dendro.2010.12.005
Barrera MD, Frangi JL, Richter LL, Perdomo MH, Pinedo LB (2000) Structural and functional changes in Nothofagus pumilio forests along an altitudinal gradient in Tierra del Fuego, Argentina. J Veg Sci 11:179–188. https://doi.org/10.2307/3236797
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Sta Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bava JO, Rechene DC (2004) Dinámica de la regeneración de Lenga (Nothofagus pumilio (Poepp. et Endl) Krasser) como base para la aplicación de sistemas silvícolas in Ecología y Manejo de los Bosques de Argentina. In: Arturi MF, Frangi JL, Goya JF (eds) Editorial de la Univ. Nacional de La Plata, Argentina, pp 1–22.
Beguería S, Vicente-Serrano S (2017) Calculation of the standardised precipitation evapotranspiration index. SPEI Pack Version 1:7
Cabrera AL (1976) Regiones fitogeográficas argentinas. Encicl Argentina Agric y Jard II:1–85
Cagnacci J, Estravis-Barcala M, Lía MV, Martínez-Meier M, González Polo M, Arana MV (2020) The impact of different natural environments on the regeneration dynamics of two Nothofagus species across elevation in the southern Andes. For Ecol and Manag 464:118034. https://doi.org/10.1016/j.foreco.2020.118034
Coomes DA, Allen RB (2007) Effects of size, competition and altitude on tree growth. J Ecol 95:1084–1097. https://doi.org/10.1111/j.1365-2745.2007.01280.x
Crawley MC (2005) An introduction using R. Wiley, London, p 335
Cuevas JG (2000) Tree recruitment at the Nothofagus pumilio alpine timberline in Tierra del Fuego, Chile. Ecology 88:840–855. https://doi.org/10.1046/j.1365-2745.2000.00497.x
Cuevas JG (2002) Episodic regeneration at the Nothofagus pumilio alpine timberline in Tierra del Fuego, Chile. J Ecol 90:52–60
Danby RK, Hik DS (2007) Responses of white spruce (Picea glauca) to experimental warming at a subarctic alpine treeline. Glob Chang Biol 13:437–451. https://doi.org/10.1111/j.1365-2486.2006.01302.x
Daniels LD, Veblen TT (2004) Spatiotemporal influences of climate on altitudinal treeline in Northern Patagonia. Ecology 85:1284–1296. https://doi.org/10.1890/03-0092
Doak DF, Morris WF (2010) Demographic compensation and tipping points in climate-induced range shifts. Nature 467:959–962
Donoso C (1993) Bosques templados de Chile y Argentina. Editorial Universitaria, Santiago, Chile, Variación, Estructura y Dinámica, p 488
Engelbrecht BM, Comita LS, Condit R, Kursar TA, Tyree MT, Turner BL, Hubbell SP (2007) Drought sensitivity shapes species distribution patterns in tropical forests. Nature 447:80–82
Falvey M, Garreaud RD (2009) Regional cooling in a warming world: recent temperature trends in the southeast Pacific and along the west coast of subtropical South America (1979–2006). J Geophys Res 114:1–16. https://doi.org/10.1029/2008JD010519
Garreaud R, Lopez P, Minvielle M, Rojas M (2013) Large-scale control on the Patagonian climate. J Clim 26:215–230. https://doi.org/10.1175/JCLI-D-12-00001.1
Grubb P (1977) The maintenance of species richness in plant communities: the importance of the regeneration niche. Bio Rev 52:107–145
Hampe A, Petit RJ (2005) Conserving biodiversity under climate change: the rear edge matters. Ecol Lett 8:461–467. https://doi.org/10.1111/j.1461-0248.2005.00739.x
Harsch MA, Bader MY (2011) Treeline form–a potential key to understanding treeline dynamics. Glob Ecol Biogeogr 20:582–596. https://doi.org/10.1111/j.1466-8238.2010.00622.x
Harsch MA, Hulme PE, McGlone MS, Duncan RP (2009) Are treelines advancing? A global meta-analysis of treeline response to climate warming. Ecol Lett 12:1040–1049. https://doi.org/10.1111/j.1461-0248.2009.01355.x
Heinemann K (2007) Aspectos espaciales y temporales del establecimiento de Nothofagus pumilio en claros de bosques maduros en los extremos de un gradiente ambiental en el Noroeste de la Patagonia. PhD Thesis, Universidad Nacional del Comahue, Bariloche, Argentina
Heinemann K, Kitzberger T, Veblen TT (2000) Influences of gap microheterogeneity on the regeneration of Nothofagus pumilio in a xeric old-growth forest of northwestern Patagonia, Argentina. Can J Forest Res 30:25–31
Holmgren M, Stapp P, Dickman CR, Gracia C, Graham S, Gutiérrez JR, Hice C, Jaksic F, Kelt DA, Letnic M, Lima M, López BC, Meserve PL, Milstead WB, Polis GA, Previtali MA, Richter M, Sabaté S, Squeo FA (2006) Extreme climatic events shape arid and semiarid ecosystems. Front Ecol Environ 4:87–95. https://doi.org/10.1890/1540-9295(2006)004[0087:ECESAA]2.0.CO;2
Körner C (2007a) The use of “altitude” in ecological research. Trends Ecol Evol 22:569–574. https://doi.org/10.1016/j.tree.2007.09.006
Körner C (2007b) Climatic treelines: conventions, global patterns, causes (Klimatische Baumgrenzen: Konventionen, globale Muster, Ursachen). Erdkunde 61:316–324
Körner C, Paulsen J (2004) A world-wide study of high altitude treeline temperatures. J Biogeogr 31:713–732. https://doi.org/10.1111/j.1365-2699.2003.01043.x
Kullman L (2007) Tree line population monitoring of Pinus sylvestris in the Swedish Scandes, 1973–2005: implications for tree line theory and climate change ecology. J Ecol 95:41–52
Kirdyanov AV, Hagedorn F, Knorre AA, Fedotova EV, Vaganov EA, Naurzbaev MM, Moiseev PA, Rigling A (2012) 20th century tree-line advance and vegetation changes along an altitudinal transect in the Putorana Mountains, northern Siberia. Boreas 41:56–67
Lara A, Villalba R, Wolodarsky-Franke A, Aravena JC, Luckman BH, Cuq E (2005) Spatial and temporal variation in Nothofagus pumilio growth at tree line along its latitudinal range (35°40"–55° S) in the Chilean Andes. J Biogeogr 32(5):879–893. https://doi.org/10.1111/j.1365-2699.2005.01191.x
Lopez P, Chevallier P, Favier V, Pouyaud B, Ordenes F, Oerlemans J (2010) A regional view of fluctuations in glacier length in southern South America. Global Planet Change 71:85–108
Magrin GO, Marengo JA, Boulanger JP, Buckeridge MS, Castellanos E, Poveda G, Scarano FR, Vicuña S, Alfaro E, Anthelme F (2014) Central and South America. In: Climate change 2014: impacts, adaptation, and vulnerability. Part B: regional aspects. Contribution of working group II to the fifth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge, pp 1499–1566.
Maher EL, Germino MJ (2006) Microsite differentiation among conifer species during seedling establishment at alpine treeline. Ecoscience 13:334–341. https://doi.org/10.2980/i1195-6860-13-3-334.1
Martínez-Pastur G, Cellini JM, Peri PL, Vukasovic RF, Fernández MC (2000) Timber production of Nothofagus pumilio forests by a shelterwood system in Tierra del Fuego (Argentina). For Ecol Manag 134:153–162. https://doi.org/10.1016/S0378-1127(99)00253-4
Masiokas MH, Villalba R, Luckman BH, Lascano ME, Delgado S, Stepanek P (2008) 20th-century glacier recession and regional hydroclimatic changes in northwestern Patagonia. Glob Planet Chang 60:85–100. https://doi.org/10.1016/j.gloplacha.2006.07.031
Masiokas MH, Luckman BH, Villalba R, Delgado S, Skvarca P, Ripalta A (2009) Little ice age fluctuations of small glaciers in the Monte Fitz Roy and Lago del Desierto areas, South Patagonian Andes, Argentina. Palaeogeogr Palaeoclimatol Palaeoecol 281:351–362. https://doi.org/10.1016/j.palaeo.2007.10.031
McKee TB, Doesken NJ, Kleist J (1993) The relationship of drought frequency and duration to the time scales. In Proceedings of the 8th conference on applied climatology; Anaheim, USA. pp 179–183.
Olofsson J (2004) Positive and negative plant–plant interactions in two contrasting arctic-alpine plant communities. Arct Antarct Alp Res 36:464–467
Paulo AA, Rosa RD, Pereira LS (2012) Climate trends and behavior of drought indices based on precipitation and evapotranspiration in Portugal. Nat Hazards Earth Syst Sci 12:1481–1491. https://doi.org/10.5194/nhess-12-1481-2012
Peñuelas J, Boada M (2003) A global change-induced biome shift in the Montseny mountains (NE Spain). Glob Chang Biol 9:131–140. https://doi.org/10.1046/j.1365-2486.2003.00566.x
Peter FK, Ronald R (1996) High temperature and drought stress effects on survival of Pinus ponderosa seedlings. Tree Physiol 16:665–672
Piper FI, Cavieres LA, Reyes-Diaz M, Corcuera LJ (2006) Carbon sink limitation and frost tolerance control performance of the tree Kageneckia angustifolia D. Don (Rosaceae) at the treeline in central Chile. Plant Ecol 185:29–39
Piper FI, Fajardo A, Cavieres LA (2013) Simulated warming does not impair seedling survival and growth of Nothofagus pumilio in the southern Andes. Perspect Plant Ecol Evol Syst 15:97–105. https://doi.org/10.1016/j.ppees.2013.02.003
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Ramírez C, San Martín C, Oyarzún A, Figueroa H (1997) Morpho-ecological study on the South American species of the genus Nothofagus. Plant Ecol 130:101–109
Roig FA, Anchorena J, Dollenz O, Faggi AM, Méndez E (1985) Las comunidades vegetales de la Transecta Botánica de la Patagonia Austral. In: Boelcke O, Moore DM, Roig FA (ed) Transecta Botánica de la Patagonia Austral. Buenos Aires pp. 350–519
Rusch VE (1993) Altitudinal variation in the phenology of Nothofagus pumilio in Argentina. Rev Chil His Nat 66:131–141
Sánchez-Salguero R, Camarero JJ, Hevia A, Madrigal-González J, Linares JC, Ballesteros-Canovas JA, Sánchez-Miranda A, Alfaro-Sánchez R, Sangüesa-Barreda G, Galván JD, Gutiérrez E, Génova M, Rigling A (2015) What drives growth of Scots pine in continental Mediterranean climates: drought, low temperatures or both? Agric Meteorol 206:151–162. https://doi.org/10.1016/j.agrformet.2015.03.004
Soler R, Pastur GM, Peri P, Lencinas MV, Pulido F (2013) Are silvopastoral systems compatible with forest regeneration? An integrative approach in southern Patagonia. Agrofor Syst 87:1213–1227. https://doi.org/10.1007/s10457-013-9631-z
Srur AM, Villalba R, Villagra PE, Hertel D (2008) Influencia de las variaciones climáticas y la concentración de CO2 en el crecimiento y la eficiencia en el uso del agua de Nothofagus pumilio a lo largo de un gradiente altitudinal en la Patagonia. Rev Chil Hist Nat 81:239–256
Srur AM, Villalba R, Rodríguez-Catón M, Amoroso MM, Marcotti E (2016) Establishment of Nothofagus pumilio at upper treelines across a precipitation gradient in the northern Patagonian Andes. Arctic, Antarct Alp Res 48:755–766. https://doi.org/10.1657/AAAR0016-015
Srur AM, Villalba R, Rodríguez-Catón M, Amoroso MM, Marcotti E (2018) Climate and Nothofagus pumilio establishment at upper treelines in the Patagonian Andes. Front Earth Sci 6:57
Stevens GC, Fox JF (1991) The causes of treeline. Annu Rev Ecol Syst 22:177–191
Suarez ML, Kitzberger T (2008) Recruitment patterns following a severe drought: long-term compositional shifts in Patagonian forests. Can J for Res 38:3002–3010. https://doi.org/10.1139/X08-149
Tercero Bucardo NO, Kitzberger T, Veblen TT, Raffaele E (2007) A field experiment on climatic and herbivore impacts on post-fire tree regeneration in north-western Patagonia. J Ecol 95:771–779. https://doi.org/10.1111/j.1365-2745.2007.01249.x
Veblen TT, Donoso C, Kitzberger T, Rebertus AJ (1996) Ecology of Southern Chilean and Argentinean forests. In: Veblen TT, Hill RS, Read J (eds) The ecology and biogeography of Nothofagus forests. Yale University Press, Chelsea, Michigan, USA, pp 293–353
Villalba R, Lara A, Boninsegna JA, Masiokas M, Delgado S, Aravena JC, Roig FA, Schmelter A, Wolodarsky A, Ripalta A (2003) Large-scale temperature changes across the southern Andes: 20th-century variations in the context of the past 400 years. Clim Chang 59:177–232. https://doi.org/10.1007/978-94-015-1252-7_10
Wardle P (1980) Ecology and distribution of silver beech (Nothofagus menziesii) in the paringa district, South Westland, New Zealand. N Z J Ecol 3:23–36
Wolkovich EM, Cook BI, McLauchlan KK, Davies TJ (2014) Temporal ecology in the Anthropocene. Ecol Lett 17:1365–1379
Acknowledgements
Our project was partially financed by THEMES-CONICET Project funded by the BNP Paribas Foundation in the frame of its ‘Climate Initiative’ Programme, PIP 2012-2014 and PICT 2018-03691. The authors thank Reinhardt Brand, Mariano Guzmán, Alberto Ripalta, Marcela Tonello, Gonzalo Sottile, Lidia Ferri-Hidalgo, Jorge Gonnet and Milagros Rodriguez-Cantón, who contributed to collect field data. We also give thanks to the administration of Parque Nacional Los Glaciares, Ea. Los Huemules and Ea. Río Toro for helping us with logistic for field sampling and authorization for data collection inside their properties.
Funding
Our project was partially financed by THEMES-CONICET Project funded by the BNP Paribas Foundation in the frame of its ‘Climate Initiative’ Programme, PIP 2012-2014 and PICT 2018-03691 by Agencia Nacional de Promoción de la Investigación, el Desarrollo Tecnológico y la Innovación from Argentina.
Author information
Authors and Affiliations
Contributions
Research idea and design by all authors; data analysis by VA and AS; manuscript writing and editing by all authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Consent to participate
All the authors have approved the contents of this paper and agreed to submission policies.
Consent for publication
All the authors agree to ask consideration for publication in Plant Ecology.
Ethical approval
Not applicable.
Additional information
Communicated by Jaime Moyano.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Aschero, V., Srur, A.M., Guerrido, C. et al. Contrasting climate influences on Nothofagus pumilio establishment along elevational gradients. Plant Ecol 223, 369–380 (2022). https://doi.org/10.1007/s11258-021-01211-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-021-01211-8