Abstract
Among medium-sized carnivores, red foxes (Vulpes vulpes) and domestic cats (Felis silvestris catus) are the most abundant species in human-dominated landscapes worldwide. Both are known to be generalist predators that exploit a wide range of prey groups (e.g., mammals, birds, and invertebrates). Identifying red fox and domestic cat predation pressure on shared prey could shed light on their ecological role in shaping wildlife communities in human-dominated landscapes. Here, we assess the seasonal diet of red foxes and domestic cats in terms of composition, breadth, and overlap. Over two years, we collected their scats across three human-dominated study sites: park (n = 220 for foxes and n = 0 for cats), agricultural land (n = 159 for foxes and n = 146 for cats), and managed forest (n = 169 for foxes and n = 47 for cats). We detected similar diet breadth (B) for red foxes and domestic cats (B = 0.32 and B = 0.36, respectively) as well as strong dietary overlap (O = 0.83) between them. Moreover, the diet composition of both predators varied according to the study sites and seasons. Our results confirm the highly flexible trophic behaviour of these carnivores at the study sites, probably as a consequence of prey availability, and also the simultaneity of their predation over the same prey groups. Future studies should simultaneously monitor predator diet as well as predator and prey abundance in human-dominated landscapes to better understand the predatory impact of red foxes and domestic cats.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
By 2050, more than half of the world’s population will live in urbanized areas (United Nations 2019). Consequently, understanding the functioning of these ecosystems is necessary in order to preserve both biodiversity and human life quality. Human-dominated landscapes share a set of general biotic and abiotic characteristics such as the alteration of species richness (McKinney 2008), the variation of microclimatic conditions (Santamouris et al. 2001), the availability of new resources such as anthropogenic food refuse (Fleming and Bateman 2018), and the lack or reduced number of large carnivores (Crooks 2002; Iossa et al. 2010; Bateman and Fleming 2012). In this context, medium-sized carnivores may be “released” in the absence of top-predators (Crooks and Soulé 1999), thus influencing prey populations through top-down processes, as already demonstrated at continental (Ripple et al. 2013) and local scales (Jiménez et al. 2019).
Among medium-sized carnivores, the red fox (Vulpes vulpes) is one of the most widespread species (Schipper et al. 2008), while the domestic cat (Felis silvestris catus) is one of the most popular pets worldwide. Out of 18.83 million pets in France, 11.4 million are cats. Moreover, the generalist trophic behaviour of red foxes and domestic cats makes them successful species in human-dominated landscapes (Bateman and Fleming 2012). Red fox diets in urban areas are mostly characterized by the presence of anthropogenic food refuse (Harris 1981; Doncaster et al. 1990; Contesse et al. 2004; Hegglin et al. 2007; Meckstroth et al. 2007), while in rural areas, they are more diversified with mammals and birds (Goldyn et al. 2003), mammals, invertebrates, and fruits (Ghoshal et al. 2016), or invertebrates and fruits (Dell’Arte et al. 2005) as the principal food categories. Like red foxes, domestic cats living in urban areas consume not only anthropogenic refuse (Eberhard 1954; Jackson 1951) but also mammals and birds (Gordon et al. 2010; Tschanz et al. 2011; Thomas et al. 2012). In rural areas, mammals and birds are among the principal prey categories in domestic cat diet (Kays and Dewan 2004; Brickner-Braun et al. 2007; Flux 2007; Morgan et al. 2009; van Heezik et al. 2010). Little is known, however, about how these predators interact with each other or with their shared prey in human-dominated landscapes. In natural or semi-natural habitats, several studies have assessed the influence of fox presence on cat trophic behaviour (Molsher et al. 2017). Moreover, in other natural habitats there exist some degree of trophic competition between these two predators (Catling 1988; Risbey et al. 1999; Glen et al. 2011).
Simultaneous monitoring of red fox and domestic cat diets is needed in order to determine their degree of trophic overlap, better assess their concomitant potential predation pressure, and clarify their trophic role in shaping prey communities. To date, however, only one study (Meckstroth et al. 2007) has carried out the simultaneous monitoring of red fox and domestic cat diets in a human-dominated landscape, but none has investigated their degree of diet overlap. In the present study, we firstly aimed to describe red fox and domestic cat diets in three human-dominated landscapes (urban park, agricultural land, and managed forest) across seasons and then estimate their diet breadth and overlap. We hypothesised that both predators consumed a wide range of prey (e.g., large diet breadth) and shared most of them (e.g., high degree of diet overlap), will confirm their generalist trophic behaviour mainly driven by temporal and local specificities.
Materials and methods
Study sites
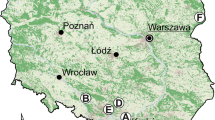
Red fox and domestic cat populations were non-invasively monitored by collecting faecal samples (hereafter, scats) at three suburban study sites (Fig. 1). Sceaux urban park (hereafter, park) (48°46′03.17”N, 2°17′47.48″E), located 20 km south of Paris, covers an area of 1.81 km2 and comprises three main habitat types: open areas (lawns and meadows), wooded areas (composed of 16% Fraxinus sp., 14% Acer platanoïde, 12% Acer pseudoplatanus, and 9% Carpinus), and French formal gardens (predominately ornamental species) (Hauts-de-Seine Conseil Général 2015). The agricultural area of Saclay (hereafter, agricultural land) (48°42′32.18”N, 2°10′33.00″E) is located between the north of the Essonne department and the southeast of the Yvelines department. This fertile agricultural land extending over 27 km2 has a long agricultural tradition with the primary crops of colza, wheat, and barley. Currently, this area is facing urbanisation pressure due to the development of a university and laboratory cluster (Spaak 2013). Rambouillet forest (hereafter, forest) (48°40′29.84”N, 1°48′27.17″E), located in the south of the Yvelines department, is one of the largest forests in the Île-de-France region. This wooded area of 200 km2, mainly composed of oaks and coniferous (68% and 25%, respectively), covers a territory of 29 municipalities (Ministère de l’agriculture et de la pêche 2015).
Sampling protocol
One month prior to the start of predator scat sampling, all study sites were cleaned by removing old scats. Scat sampling was carried out four times per year in the middle of each season (October/autumn; January/winter; April/spring; July/summer) over two consecutive years (2014–2016) for each of the three study sites. Carnivora scats are cylindrical (i.e., sausage-shaped) with sub-division tapped at one of the extremities (Chame 2003). These characteristics may therefore promote scat misidentifications among mammalian carnivores of similar body size (Reid 2015; Morin et al. 2016). Red fox and domestic cat scats, however, can be differentiated by their morphometry (Bang and Dahlström 1975) and secondarily by their place of deposition, dietary content, and odour. Red fox scats (8–10 cm length and 2 cm width) are larger than those of domestic cats (6–8 cm length and 1–1.5 cm width) (Chame 2003). Moreover, red fox scats are tubular, long, and twisted with pointed ends, whereas domestic cat scats are smoothed and compacted with well-defined segments and only one tapered extremity (Chame 2003). Red foxes usually deposit scats in prominent positions along tracks to signal their presence to other individuals, whereas domestic cats tend to bury theirs (Seton 1925, Gibbons 2003). Regarding dietary contents, domestic cat scats strictly reflect their carnivorous diet with the additional presence of grass leaves ingested to aid hair elimination and/or Gramineae seeds indirectly ingested with the grass leaves (Chame 2003). Fresh red fox scats present a strong “foxy” smell like middle skunk (Gibbons 2003). Scats were collected by walking along the same paths in each study site and season. They were individually geolocalised, stored in separate plastic bags, and frozen (− 20 °C) until analysis. In this study, we aimed to detect the heterogeneity of predator diet in the main suburban habitats and across seasons. Thus, though informative, within-habitat site replications were not assessed; indeed, this was not possible in the park and agricultural land due to their limited areas. Because of the low number of domestic cat scats collected in the forest and park, we asked volunteer owners to collect the scats of their own vagrant cats during each study period. Four owners from each study site (forest and park) with one to three cats in their house collaborated with us.
Due to the small number of red fox and domestic cat scats containing prey remains in some seasons and years, we decided to pool samples by season (i.e., without considering the year). Only seasons with at least 10 scats with prey remains per predator species were included in the following analyses (Table S1). Despite the collaboration of cat owners, many of the collected scats did not contain any prey remains. As a result, we did not have sufficient numbers of cat scats from the park for all seasons or from the forest in spring to include them in the analyses (Table S1).
Laboratory analyses
Scats were analysed macroscopically and microscopically. Macroscopic analysis was performed by washing the scats individually under a stream of water in a 2 μm sieve. We excluded scats without prey remains (i.e., feathers, hairs, bones, seeds). Prey remains were identified under a binocular to the finest taxonomic level possible using the bone reference collections from the National Museum of Natural History in Paris for mammals and birds, and identification keys for invertebrates and fruits before validating our identifications with specialists of the targeted groups. Indigestible prey remains (e.g., hair, teeth, bones, skin, scales, feathers, exoskeletons, seeds from fleshy fruits) were then classified into 16 main food categories for a more accurate diet comparison: Microtidae (including Myodes glareolus, Microtus agrestis, and Microtus arvalis), Muridae (including Apodemus sylvaticus, Mus musculus, Rattus sp., rattus norvegicus, and Rattus rattus), Soricidae (including Sorex coronatus, Crocidura leucodon, and Crocidura russula), Leporidae (including Oryctolagus cuniculus and Lepus europaeus), small birds (~ size of Passeroidea), large-medium birds (~ size of Corvoidea), Coleoptera, Hymenoptera, Dermaptera, Orthoptera, Heteroptera, Lepidoptera, Arachnida, earthworms, small fruits (Prunus cerasus, P. prunus, and Rubus ulmifolius), and large fruits (Malus sp. and Pyrus sp.). We also reported the frequency of nonorganic (e.g., plastic, foil paper) anthropogenic refuse (hereafter, refuse). Coprophagous, recycling, and ticks were excluded from the invertebrate prey.
Microscopic analysis was carried out by subsampling ~ 2.5 mg of each homogenised predator scat on microscope slides to quantify earthworm consumption based on chaetae counts (Reynolds and Aebischer 1991).
Data analyses
For each scat, we firstly determined the minimum number of consumed prey individuals in each scat (preyMNI) for the main food categories (i.e., excluding refuse). For birds, mammals, and terrestrial arthropods, we counted the number of identical prey fragments contained in each predator scat. For fleshy fruits, preyMNI per predator scat was calculated based on the mean number of seeds (MNS) per fruit collected at the study sites: Rubus ulmifolius (n = 150, MNS = 52.64); Prunus prunus (n = 39, MNS = 1); P. cerasus (n = 30, MNS = 1), Malus sp. (n = 73, MNS = 4.49), and Pyrus sp. (n = 71, MNS = 4.72). For earthworms, we collected the most abundant species (Aporrectodea caliginosa, A. giardi, Allolobophora icterica, and Octolasium cyaneum) from our study sites to measure and calculate their mean fresh weights (g), length (cm), number of segments per individual, and number of chaetae per segment. PreyMNI for earthworms per scat was obtained as follows:
To quantify the ingested prey biomass contained in each scat (preyIBS), we firstly calculated the total ingested biomass (TIB) by multiplying the preyMNI of each prey by its mean mass (MM). The biomass of small mammals (MM = 57.32 g) and birds (MM = 302.54 g) was calculated using weight data extracted from the Amniote trait database (Myhrvold et al. 2015). We estimated invertebrate MM by weighing field-collected individuals from the orders most commonly consumed by the studied predators: Coleoptera (n = 156, MM = 0.29 g), Hymenoptera (n = 26, MM = 0.01 g), Dermaptera (n = 20, MM = 0.03 g), Orthoptera (n = 71, MM = 0.10 g), Heteroptera (n = 76, MM = 0.02 g), and Lepidoptera (n = 92, MM = 0.02 g), as well as the class Arachnida (n = 87, MM = 0.01 g). Fruit MM was obtained by collecting and weighing the following fruits from the study sites: Rubus ulmifolius (n = 150, MM = 2.19 g), Prunus prunus (n = 39, MM = 15.09 g), P. cerasus (n = 30, MM = 3.70 g), Malus sp. (n = 73, mean MM = 90.01 g), and Pyrus sp. (n = 71, MM = 98.55 g). Since red foxes and domestic cats are known to ingest large body mass prey (e.g., Leporidae, large birds) only partially (Artois 1989; Bonnaud et al. 2007), we estimated the ingested proportion of lagomorphs and large-medium birds by calculating the maximum food intake per scat of each predator. To do so, we selected red fox (n = 75) and domestic cat (n = 53) scats containing only small mammals (i.e., prey entirely ingested) and multiplied their preyMNI by their MM to obtain a maximum ingested biomass (MIB in g of fresh biomass) for each predator scat. MIB was 244.09 g and 232.40 g for red fox and domestic cat, respectively. We then calculated the ingested biomass of each prey category per scat (preyIBS) by using one of the two following formulas. If the TIB did not exceed the MIB, we applied the formula:
If the TIB exceeded the MIB due to the consumption of large preyj like lagomorph and large-medium birds, for instance, we applied the following formula:
Where RIBj is the relative ingested biomass of each large prey calculated as follows:
Effects of study sites and seasons on predator diet
We chose multivariate generalised linear models (hereafter, GLMs) to detect factors (i.e., predator, study site, and season) that could affect diet composition, because our dietary data had a strong mean-variance relationship that was taken into account in these models (Wang et al. 2016). Firstly, we fitted one global GLM using the following formula: diet ∼ predator + study site * season. Secondly, we fitted one GLM for each predator species using the following formula: predator diet ∼ study site * season. We then ran univariate analysis of variance for each prey category and adjusted the p value for multiple testing with a step-down resampling procedure (Wang et al. 2016). We used 999 bootstrap iterations to sample multivariate GLM residuals. Models were fitted with the manyglm function from the mvabund package (Wang et al. 2016). Although we only discuss the results from the models with preyIBS as the response variable, as it is the best approximation of the true diet of predators (Klare et al. 2011), we also included the results from the global model for each of the two diet descriptors (preyMNI and preyMNI) as response variables (Table S2) and from the model using preyMNI as the response variable (Table S3).
Predator diet breadths and overlaps
We used preyMNI to calculate the degree of trophic specialisation for each studied predator by estimating their diet breadth (B) and degree of diet overlap (O) using the nichevar and nicheoverlap functions from the indicspecies package, respectively (Caceres and Legendre 2009). Diet breadth ranges from 0 to 1, with values close to 0 for specialised populations and values close to 1 for generalists. Diet overlap ranges from 0 to 1, with values close to 0 for low diet overlap between predator populations and values close to 1 for high diet overlap.
We visually assessed dietary overlap between predators, study sites, and seasons using nonmetric multidimensional scaling (NMDS) plots. This multivariate method is based on a triangular resemblance matrix of Bray-Curtis similarities among all pairs of samples. We used the metaMDS function from the vegan package (Oksanen et al. 2018) to produce NMDS plots. All statistical analyses were carried out using R Studio version 1.1.463. (R Core Team 2019).
Results
In total, we collected 1073 scats, but 332 did not contain any wild prey remains (i.e., only highly digestible organic matter like industrial food and/or refuse), with 55% of them belonging to red foxes (park: n = 94, agricultural land: n = 24, managed forest: n = 65) and 45% to domestic cats (park: n = 85, agricultural land: n = 21, managed forest: n = 43) (Table S1). From the remaining 741 scats containing prey remains, 74% belonged to red foxes (park: n = 220, agricultural land: n = 159, managed forest: n = 169) and 26% to domestic cats (park: n = 0, agricultural land: n = 146, managed forest: n = 47) (Table S1). Out of these 741 scats, 41 scats contained prey remains that were not attributable to the 16 main prey groups defined above (e.g., scats containing unidentifiable parts of invertebrates, seeds, feathers, or hairs). This resulted in the inclusion of 700 scats in preyIBS and preyMNI models, corresponding to 699 degrees of freedom in general models and 521 and 177 degrees of freedom in red fox (n = 522) and domestic cat (n = 178) models, respectively.
We identified a total of 6742 prey items. Based on preyIBS, red fox diet was mainly composed of Rodentia (37%) and large-medium birds (28%), and domestic cat diet of Leporidae (41%) and large-medium birds (27%) (Table 1). Based on the preyMNI, red fox diet was mainly composed of earthworms (53%), and domestic cat diet of earthworms (40%) and Microtidae (21%) (Table 1).
The composition of both predators’ diets (preyIBS) was influenced by predator species, study sites, seasons, and the interaction between study sites and seasons (Table S2a). Predator species affected diet preyIBS in terms of Leporidae, Microtidae, Coleoptera, earthworms, and small fruits. Study site affected both predators’ consumption of Leporidae, Microtidae, Muridae, Soricidae, large-medium birds, Coleoptera, and earthworms. Seasons influenced both predators’ diets in terms of Coleoptera, earthworms, and small fruits. The interaction between study site and season also affected both predators’ diets in terms of Leporidae, large-medium birds, Coleoptera, and small fruits (Table S2a).
Red fox diet
The composition of red fox diet (preyIBS) was influenced by study sites, seasons, and the interaction between study sites and seasons (Table 2a). Study site affected red fox preyIBS in terms of Microtidae, Muridae, large-medium birds, Coleoptera, earthworms, and small fruits. Seasons influenced red fox consumption of Coleoptera, earthworms, and small fruits. The interaction between study site and season also affected red fox preyIBS in terms of large-medium birds, Coleoptera, and small fruits (Table 2a).
Based on the ingested prey biomass (preyIBS), in the park, red fox diet was mainly composed of large-medium birds (66%), especially in autumn (73%), spring, (99%), and summer (60%) (Table 1). In agricultural land, red fox diet principally comprised large-medium birds (35%), especially in autumn (50%) and summer (37%), followed by Leporidae (28%), mainly in autumn (37%), winter (20%), and summer (30%) (Table 1). In the forest, red fox diet was mainly composed of Leporidae (36%), especially in autumn (98%) and spring (98%), followed by Muridae (29%), principally in winter (53%) (Table 1).
Based on the minimum number of ingested individuals (preyMNI), in the park, red fox diet was mainly composed of earthworms (61%), particularly in autumn (90%), winter (77%), and spring (87%). In agricultural land, red fox diet principally comprised small fruits (38%), especially in summer (55%), followed by earthworms (33%), mostly in autumn (61%), winter (36%), and spring (64%). In the forest, earthworms were the principal food category in red fox diets (56%), especially in winter (79%) and spring (64%) (Table 1).
Domestic cat diet
Domestic cat diet composition was influenced by the seasons as well as the interaction between study site and season. In particular, seasons had an influence on domestic cat preyIBS in terms of earthworm biomass (Table 2b).
Based on the ingested prey biomass (preyIBS), in agricultural land, domestic cat diet was mainly composed of Leporidae (57%), especially in autumn (67%), winter (52%), and spring (77%) (Table 1). In the forest, domestic cat diet principally comprised Muridae (43%), particularly in summer (70%), followed by large-medium birds (18%), especially in summer (64%) (Table 1).
Based on the minimum number of ingested individuals (preyMNI), in agricultural land, domestic cat diet was mainly composed of earthworms (48%), particularly in autumn (60%), winter (64%), and spring (34%), followed by Microtidae (20%), especially in summer (34%) (Table 1). In the forest, domestic cat diet mainly comprised earthworms (22%), particularly in autumn (65%) and winter (47%), followed by Microtidae (22%), especially in summer (52%) (Table 1).
Diet breadth and overlap
According to the results of the diet breadth index, both predators exhibited a narrow diet breadth at all study sites and across seasons (B < 0.40), with the diet breadth of each predator being similar (red fox: B = 0.32, 95% CI [0.29, 0.34]; domestic cat: B = 0.36, 95% CI [0.31, 0.39]) across study sites and seasons (Fig. 2).
Interestingly, in agricultural land, we found a high degree of diet overlap between red fox and domestic cat populations across all seasons (O > 0.75) except summer (O = 0.33, 95% CI [0.20, 0.56]) (Table S4). During this season, dissimilarity between red fox and domestic cat diet (i.e., points located far from each other with little or no overlap) was higher compared to the other seasons (Fig. 3a). By contrast, we detected higher variations in the diet overlap between red fox and domestic cat populations in the forest compared to agricultural land, with the highest value found in winter (O = 0.92 95% CI [0.05, 0.99]) and the lowest in summer (O = 0.25 95% CI [0.04, 0.60]) (Table S4). In winter, red fox and domestic cat diet showed the highest similarity (i.e., most points are spatially close to each other or overlapping), while the lowest diet similarity was observed in summer (Fig. 3b). Due to the limited number of collected cat scats in some habitats and seasons, we did not calculate the diet overlap between red fox and domestic cat populations in the forest in spring or in the park for all seasons.
Nonmetric MultiDimensional Scaling (NMDS) plots, constructed from Bray-Curtis similarity matrices based on preyMNI data, showing diet composition of red foxes and domestic cats across seasons in studied suburban habitats: a) agricultural land and b) forest. Only sample size larger than 10 scats collected per predator, site and season were analyz
Discussion
Results obtained in this study were only based on the analysis of scats containing wild prey remains (i.e., 75% and 56% of red fox and domestic cat scats, respectively). The high proportion of domestic cat scats without any wild prey remains (i.e., containing only highly digestible organic matter like industrial food and/or refuse) is partly due to the high percentage (70%) of scats that were provided by cat owners, meaning that domestic cats were primarily sustained by human-mediated food while having the opportunity to wander and feed on wild prey. Even if the percentage of red fox scats containing only refuse and/or industrial food is lower than that of domestic cat scats, this indicates that red foxes, particularly in suburban areas, are able to feed on human-mediated food. This feeding habit may provide them with some adaptive and/or survival advantages.
Red fox and domestic cat diets
The trophic index based on ingested prey biomass (preyIBS) revealed that large-medium birds and mammals were the prey categories most contributing to red fox and domestic cat food bolus. Invariably, red fox and domestic cat diets were dominated by large prey (i.e., large-medium birds and Leporidae) and/or medium-sized prey (Muridae) across all study sites and seasons. This result suggests that the survival of both predators mainly depends on prey with a large biomass (e.g., medium-large birds, Leporidae), probably because one killed individual represents a large source of metabolizable energy.
The trophic index based on the minimum number of ingested individuals (preyMNI) revealed that earthworms were the most abundantly consumed prey in red fox diet. This result is in good agreement with other studies once refuse is removed from red fox diet (MacDonald 1980; Harris 1981; Doncaster et al. 1990; Reynolds and Aebischer 1991; Saunders et al. 1993; Soulsbury et al. 2008). In domestic cat diet, earthworms along with Microtidae were the most abundantly consumed prey. While the presence of chaetae in predator scats may be increased by phenomena such as soil contamination and secondary predation (i.e., chaetae would have persisted after two digestions), potentially leading to a slight overestimation of earthworm consumption, the high number of consumed earthworms stresses the importance of this prey type for both red foxes and domestic cats, as it may supply these predators with their protein requirements within human-dominated landscapes. Even if striking, the consumption of earthworms (protein-rich prey) by domestic cats is probably opportunistic and focussed on anecic and/or epigeic earthworms (Lee 1985), which are readily available and easy to catch in agricultural and forest soils. This interesting result should be confirmed (i.e., earthworm species identification) in future studies using metabarcoding approaches, for example (Bienert et al. 2012; Boyer et al. 2013; Pansu et al. 2015). In general, the macro- and microscopic diet analyses of predator diets would benefit from metabarcoding approaches to verify prey identification, detect and identify digested soft-bodied prey (e.g., insect larvae, egg content), and confirm predator scat identification (Pompanon et al. 2012; Galan et al. 2018).
Specific spatiotemporal diet patterns for red foxes and domestic cats
Red fox populations showed study site specificities in terms of the proportion of ingested biomass for certain prey categories (Microtidae, Muridae, large-medium birds, Coleoptera, earthworms, and small fruits) as well as seasonal dietary shifts for some of these resources (Coleoptera, earthworms, and small fruits) (Tables 1 and 2a).
These results are in good agreement with and support the generalist and opportunistic trophic behaviour of red foxes (Sillero et al. 2004). Thanks to this trophic behaviour, red foxes could adapt their diet in contrasting human-dominated landscapes according to the abundance and availability of associated prey communities and fruit resources (e.g., emergence of Coleoptera, fructification of fleshy fruits) (Díaz-Ruiz et al. 2013).
In contrast to red fox diet, domestic cat diet (in terms of ingested prey biomass) was relatively homogeneous across study sites and mainly focussed on mammals (Leporidae and Muridae; Gillies and Fitzgerald 2005 (New Zealand); Kays and Dewan 2004 (US); Krauze-Gryz et al. 2012 (Europe)) and secondarily on large-medium birds, with only the proportion of ingested earthworm biomass changing seasonally (Tables 1 and 2b). This suggests that cats are highly adaptable and efficient hunters, which allows them to survive and reproduce without regard to the type of habitat, thus confirming the opportunistic but strictly carnivore trophic behaviour of domestic cats (Bradshaw et al. 1996; Medina et al. 2011). By consequence, the increased number of vagrant domestic cats due to the higher human population density can lead to negative effects on bird, mammal, and reptile population dynamics in many different types of habitats (Woods et al. 2003; Dauphiné and Cooper 2009; Blancher 2013; Loss et al. 2013; Doherty et al. 2015). As the opportunistic behaviour of these predators likely depends on resource abundance and availability, which are rarely monitored and quantified, this should be investigated in future studies to detect potential patterns of prey preferences.
At all the study sites, both red foxes and domestic cats abundantly consumed earthworms (in preyMNI) in autumn, winter, and spring, probably due to the scarcity of other prey categories during these seasons of relatively low productivity. The use of this alternative high-protein prey may allow red foxes and domestic cats to maintain stable populations all year round within degraded human-dominated landscapes. Interestingly, in agricultural land, domestic cats turned to the consumption of Microtus agrestis individuals as observed in other rural areas in Europe (Krauze-Gryz et al. 2012), while red foxes shifted their diet to temporarily abundant small fruits that are rich in carbohydrates, minerals, and vitamins (e.g., Prunus cerasus, P. prunus, Rubus ulmifolius) (Table 1). Red fox consumption of fruits has already been described in other studies in Europe (Contesse et al. 2004) and Asia (Dell’Arte et al. 2005; Ghoshal et al. 2016). In addition to being an alternative source of nutrient for foxes, these fruits contained seeds that can be efficiently dispersed (Herrera 1989; Matías et al. 2010), leading to the conservation of the vegetation structure of these habitats. Last but not least, the consumption of voles by domestic cats in summer likely corresponds to a peak abundance of this prey, which supports the opportunistic trophic behaviour of cats. As domestic cats are one of the major predators of voles (Lin and Batzli 1995), they could modify the cyclicity of the prey population dynamics (Hansson 1988) and indirectly affect the trophic behaviour of other predators sharing this prey such as red foxes.
Diet breadth and overlap
Although we showed that red foxes and domestic cats are able to exploit a wide spectrum of trophic resources (i.e., mammals, birds, invertebrates, and fruits) within human-dominated landscapes, these predators exhibit quite a narrow diet breadth similarly to those described by other authors in natural habitats (Drygala et al. 2014; Vlasseva et al. 2017; Széles et al. 2018). This result confirms that these predators hunt targeted prey groups with a focus on large birds and mammals.
Because foxes and cats had similarly varied diets across habitat types and seasons, we have confidence in the main result of a high dietary overlap despite the lack of within-habitat site replication.
These are the first results regarding the degree of diet overlap between red foxes and domestic cats within human-dominated landscapes, which are in good agreement with those found in natural habitats (Paltridge 2002; Woinarski et al. 2017). In our study, in summer, red fox and domestic cat diets only marginally overlapped, suggesting that they can hunt a wider prey spectrum to reduce their degree of trophic overlap through niche partitioning. On the contrary, diet overlaps between red foxes and domestic cats were particularly high during the less productive seasons (i.e., autumn, winter, and spring) when they have to share scarcer main and alternative prey (i.e., mammals, birds, and earthworms). This indicates that competition between red foxes and domestic cats may occur, particularly if these predators are in high abundance in city centres (Šálek et al. 2015; Flockhart et al. 2016) and probably more broadly in human-dominated areas. However, competition for food between species is eased by their generalist behaviour. In addition, these high seasonal diet overlaps between red foxes and domestic cats may exacerbate their predation pressure over shared prey populations and can lead to potential negative effects on shared prey population dynamics. To quantify predation impacts on prey population dynamics, future studies should simultaneously monitor predator diets as well as prey and predator availabilities through space and time.
References
Artois M (1989) Le renard roux (Vulpes vulpes Linnaeus, 1758) Encyclopédie des carnivores de France
Bang P, Dahlström P (1975) Huellas y Señales de los Animales de Europa, Omega, Barcelona
Bateman PW, Fleming PA (2012) Big city life: carnivores in urban environments. J Zool 287:1–23. https://doi.org/10.1111/j.1469-7998.2011.00887.x
Bienert F, De Danieli S, Miquel C et al (2012) Tracking earthworm communities from soil DNA. Mol Ecol 21:2017–2030. https://doi.org/10.1111/j.1365-294X.2011.05407.x
Blancher P (2013) Estimated number of birds killed by house cats (Felis catus) in Canada. Avian Conserv Ecol 8:3. https://doi.org/10.5751/ACE-00557-080203
Bonnaud E, Bourgeois K, Vidal E et al (2007) Feeding ecology of a feral cat population on a small Mediterranean Island. J Mammal 88:1074–1081. https://doi.org/10.1644/06-MAMM-A-031R2.1
Boyer S, Wratten SD, Holyoake A et al (2013) Using next-generation sequencing to analyse the diet of a highly endangered land snail (Powelliphanta augusta) feeding on endemic earthworms. PLoS One 8:1–8. https://doi.org/10.1371/journal.pone.0075962
Bradshaw JWS, Goodwin D, Legrand-Defrétin V, Nott HMR (1996) Food selection by the domestic cat, an obligate carnivore. Comp Biochem Physiol - A Physiol 114:205–209. https://doi.org/10.1016/0300-9629(95)02133-7
Brickner-Braun I, Geffen E, Yom-Tov Y (2007) The domestic cat as a predator of Israeli wildlife. Isr J Ecol Evol 53:129–142. https://doi.org/10.1560/IJEE.53.2.129
De Caceres M, Legendre P (2009) Associations between species and groups of sites: indices and statistical inference. Ecology 90:3566–3574. https://doi.org/10.1890/08-1823.1
Catling PC (1988) Similarities and contrasts in the diets of foxes, Vulpes vulpes, and cats, Felis catus, relative to fluctuating prey populations and drought. Aust Wildl Res 15:307–317
Chame M (2003) Terrestrial mammal feces a morphometric summary and description. Mem Inst Oswaldo Cruz 98:71–94
Contesse P, Hegglin D, Gloor S et al (2004) The diet of urban foxes (Vulpes vulpes) and the availability of anthropogenic food in the city of Zurich, Switzerland. Mamm Biol 69:81–95. https://doi.org/10.1078/1616-5047-00123
Core Team R (2019) R: a language and environment for statistical computing. In: R Foundation for statistical computing, Vienna, Austria URL https://www.R-project.org/
Crooks K, Soulé M (1999) Mesopredator release and avifaunal extinctions in a fragmented system. Nature 400:563–566. https://doi.org/10.1038/23028
Crooks KR (2002) Relative Sensitivites of mammalian carnivores to habitat fragmentation. Conserv Biol 16:488–502. https://doi.org/10.1046/j.1523-1739.2002.00386.x
Dauphiné N, Cooper RJ (2009) Impacts of free-ranging domestic cats (Felis catus) on birds in the United States: a review of recent research with conservation and management recommendations. Proc fourth Int partners flight Conf tundra to trop 205–219
Dell’Arte GL, Leonardi G, Dell’Arte GL et al (2005) Effects of habitat composition on the use of resources by the red fox in a semi arid environment of North Africa. Acta Oecol 28:77–85. https://doi.org/10.1016/j.actao.2004.12.003
Díaz-Ruiz F, Delibes-Mateos M, García-Moreno JL et al (2013) Biogeographical patterns in the diet of an opportunistic predator: the red fox Vulpes vulpes in the Iberian Peninsula. Mamm Rev 43:59–70. https://doi.org/10.1111/j.1365-2907.2011.00206.x
Doherty TS, Davis RA, van Etten EJB et al (2015) A continental-scale analysis of feral cat diet in Australia. J Biogeogr 42:964–975. https://doi.org/10.1111/jbi.12469
Doncaster CP, Dickman CR, Macdonald DW (1990) Feeding ecology of red foxes in the city of Oxford, England. J Mammal 71:188–194. https://doi.org/10.2307/1382166
Drygala F, Werner U, Zoller H (2014) Diet composition of the invasive raccoon dog (Nyctereutes procyonoides) and the native red fox (Vulpes vulpes) in north-East Germany. Hystrix 24:190–194. https://doi.org/10.4404/hystrix-24.2-8867
Eberhard T (1954) Food habits of Pennsylvania house cats. J Wildl Manag 18:284–286
Fleming PA, Bateman PW (2018) Novel predation opportunities in anthropogenic landscapes. Anim Behav 138:145–155. https://doi.org/10.1016/j.anbehav.2018.02.011
Flockhart DTT, Norris DR, Coe JB (2016) Predicting free-roaming cat population densities in urban areas. Anim Conserv 19:472–483. https://doi.org/10.1111/acv.12264
Flux JEC (2007) Seventeen years of predation by one suburban cat in New Zealand. New Zeal J Zool 34:289–296. https://doi.org/10.1080/03014220709510087
Galan M, Pons J-B, Tournayre O, Pierre É, Leuchtmann M, Pontier D, Charbonnel N (2018) Metabarcoding for the parallel identification of several hundred predators and their preys: application to bat species diet analysis. Mol Ecol Resour 18:474–489. https://doi.org/10.1017/CBO9781107415324.004
Ghoshal A, Bhatnagar YV, Mishra C, Suryawanshi K (2016) Response of the red fox to expansion of human habitation in the trans-Himalayan mountains. Eur J Wildl Res 62:131–136. https://doi.org/10.1007/s10344-015-0967-8
Gibbons DK (2003) Red fox Vulpes vulpes. In: Gibbons DK (ed) Mammal tracks and sign of the northeast. University Press of New England, Hanover and London, pp 60–63
Gillies C, Fitzgerald BM (2005) Order Carnivora: Feral cat. In: King CM (ed) The handbook of New Zealand mammals, 2nd edn. Oxford University Press, Auckland, pp 307–326
Glen AS, Pennay M, Dickman CR et al (2011) Diets of sympatric native and introduced carnivores in the Barrington tops, eastern Australia. Austral Ecol 36:290–296. https://doi.org/10.1111/j.1442-9993.2010.02149.x
Goldyn B, Hromada M, Surmacki A, Tryjanowski P (2003) Habitat use and diet of red fox Vulpes vulpes in an agricultural landscape in Poland. Z Jagdwiss 49:191–200
Gordon JK, Matthaei C, Van Heezik Y (2010) Belled collars reduce catch of domestic cats in New Zealand by half. Wildl Res 37:372–378. https://doi.org/10.1071/WR09127
Hansson L (1988) The domestic cat as a possible modifier of vole dynamics. Mammalia 52:159–164. https://doi.org/10.1515/mamm.1988.52.2.159
Harris S (1981) The food of suburban foxes (Vulpes vulpes), with species reference to London. Mamm Rev 11:151–168
Hauts-de-Seine Conseil Général (2015) Parc de Sceaux: Inventaire pied à pied. Naterre
Hegglin D, Bontadina F, Contesse P et al (2007) Plasticity of predation behaviour as a putative driving force for parasite life-cycle dynamics: the case of urban foxes and Echinococcus multilocularis tapeworm. Funct Ecol 21:552–560. https://doi.org/10.1111/j.1365-2435.2007.01257.x
Herrera CM (1989) Frugivory and seed dispersal by carnivorous mammals, and associated fruit characteristics, in undisturbed Mediterranean habitats. Oikos 55:250. https://doi.org/10.2307/3565429
Iossa G, Soulsbury CD, Baker PJ, Harris S (2010) A taxonomic analysis of urban carnivore ecology. In: Gehrt SD, Riley SPD, Cypher BL (eds) Urban carnivores: ecology, conflict, and conservation. JHU Press, Baltimore, pp 173–184
Jackson WB (1951) Food habits of Baltimore, Maryland, cats in relation to rat populations. J Mammal 82:458–461
Jiménez J, Nuñez-Arjona JC, Mougeot F et al (2019) Restoring apex predators can reduce mesopredator abundances. Biol Conserv 238:108234. https://doi.org/10.1016/j.biocon.2019.108234
Kays RW, Dewan AA (2004) Ecological impact of inside/outside house cats around a suburban nature preserve. Anim Conserv 7:1–11. https://doi.org/10.1017/S1367943004001489
Klare U, Kamler HF, MacDonald DW (2011) A comparison and critique of different scat-analysis. Mamm Rev 41:294–312. https://doi.org/10.1111/j.1365-2907.2011.00183.x
Krauze-Gryz D, Gryz J, Goszczyński J (2012) Predation by domestic cats in rural areas of Central Poland: an assessment based on two methods. J Zool 288:260–266. https://doi.org/10.1111/j.1469-7998.2012.00950.x
Lee KE (1985) Earthworms. Their ecology and relationships with soils and land use. Academic Press, London
Lin YK, Batzli GO (1995) Predation on voles: an experimental approach. J Mammal 76:1003–1012
Loss SR, Will T, Marra PP (2013) The impact of free-ranging domestic cats on wildlife of the United States. Nat Commun 4:1396. https://doi.org/10.1038/ncomms2380
MacDonald DW (1980) The red fox, Vulpes vulpes, as a predator upon earthworms, Lumbricus terrestris. Z Tierpsychol 52:171–200
Matías L, Zamora R, Mendoza I, Hódar JA (2010) Seed dispersal patterns by large Frugivorous mammals in a degraded mosaic landscape. Restor Ecol 18:619–627. https://doi.org/10.1111/j.1526-100X.2008.00475.x
McKinney ML (2008) Effects of urbanization on species richness: a review of plants and animals. Urban Ecosyst 11:161–176. https://doi.org/10.1007/s11252-007-0045-4
Meckstroth AM, Miles AK, Chandra S (2007) Diets of introduced predators using stable isotopes and stomach contents. J Wildl Manag 71:2387. https://doi.org/10.2193/2005-527
Medina FM, Bonnaud E, Vidal E et al (2011) A global review of the impacts of invasive cats on island endangered vertebrates. Glob Chang Biol 17:3403–3510. https://doi.org/10.1111/j.1365-2486.2011.02464.x
Ministére de l’agriculture et de la pêche (2015) Une «forêt de protection» à Rambouillet. Procès Verbal d'état des lieux. Préfecture des Yvelines. Direction départementale de l'equipement et de l'agriculture
Molsher R, Newsome AE, Newsome TM, Dickman CR (2017) Mesopredator management: effects of red fox control on the abundance, diet and use of space by feral cats. PLoS One 12:1–15. https://doi.org/10.1371/journal.pone.0168460
Morgan SA, Hansen CM, Ross JG et al (2009) Urban cat (Felis catus) movement and predation activity associated with a wetland reserve in New Zealand. Wildl Res 36:574–580. https://doi.org/10.1071/WR09023
Morin DJ, Higdon SD, Holub JL et al (2016) Bias in carnivore diet analysis resulting from misclassification of predator scats based on field identification. Wildl Soc Bull 40:669–677. https://doi.org/10.1002/wsb.723
Myhrvold NP, Baldridge E, Chan B et al (2015) An amniote life-history database to perform comparative analyses with birds, mammals, and reptiles. Ecology 96:3109–3000. https://doi.org/10.1890/15-0846R.1
Oksanen AJ, Blanchet FG, Kindt R, et al (2018) Package ‘ vegan ’
Paltridge R (2002) The diets of cats, foxes and dingoes in relation to prey availability in the Tanami Desert, Northern Territory. Wildl Res 29:389–403. https://doi.org/10.1071/WR00010
Pansu J, De Danieli S, Puissant J et al (2015) Landscape-scale distribution patterns of earthworms inferred from soil DNA. Soil Biol Biochem 83:100–105. https://doi.org/10.1016/j.soilbio.2015.01.004
Pompanon F, Deagle BE, Symondson WOC et al (2012) Who is eating what: diet assessment using next generation sequencing. Mol Ecol 21:1931–1950. https://doi.org/10.1111/j.1365-294X.2011.05403.x
Reid REB (2015) A morphometric modeling approach to distinguishing among bobcat, coyote and gray fox scats. Wildlife Biol 21:254–262. https://doi.org/10.2981/wlb.00105
Reynolds JC, Aebischer NJ (1991) Comparison and quantification of carnivore diet by fecal analysis - a critique, with recommendations, based on a study of the fox Vulpes-Vulpes. Mamm Rev 21:97–122. https://doi.org/10.1111/j.1365-2907.1991.tb00113.x
Ripple WJ, Wirsing AJ, Wilmers CC, Letnic M (2013) Widespread mesopredator effects after wolf extirpation. Biol Conserv 160:70–79. https://doi.org/10.1016/j.biocon.2012.12.033
Risbey DA, Calver MC, Short J (1999) The impact of cats and foxes on the small vertebrate fauna of Heirisson prong, Western Australia. I. Exploring potential impact using diet analysis. Wildl Res 26:621–630
Šálek M, Drahníková L, Tkadlec E (2015) Changes in home range sizes and population densities of carnivore species along the natural to urban habitat gradient. Mamm Rev 45:1–14. https://doi.org/10.1111/mam.12027
Santamouris M, Papanikolaou N, Livada I et al (2001) On the impact of urban climate on the energy consumption of buildings. Sol Energy 70:201–216. https://doi.org/10.1016/S0038-092X(00)00095-5
Saunders G, White PCL, Harris S, Rayner JMV (1993) Urban foxes (Vulpes vulpes): food acquisition, time and energy budgeting of a generalized predator. Symp zool Soc L 65:215–234
Schipper J, Chanson JS, Chiozza F, et al (2008) The status of the world’s land and marine mammals: diversity, threat, and knowledge. Science (80- ) 322:225–230. doi: https://doi.org/10.1126/science.1165115
Seton ET (1925) On the study of scatology. J Mammal 36:47–49. https://doi.org/10.1644/870.1.Key
Sillero C, Macdonald D, Hoffmann M (2004) Canids : foxes, wolves, jackals and dogs. Status asurvey and Conservation Action Plan
Soulsbury CD, Iossa G, Baker PJ, Harris S (2008) Environmental variation at the onset of independent foraging affects full-grown body mass in the red fox. 2411–2418. doi: https://doi.org/10.1098/rspb.2008.0705
Spaak D (2013) Le plateau de Saclay: Une terre pionnière pour la protection foncière en attente d’un projet pour ses espaces ouverts et agricoles. Terre et Cité
Széles GL, Purger JJ, Molnár T, Lanszki J (2018) Comparative analysis of the diet of feral and house cats and wildcat in Europe. Mammal Res 63:43–53. https://doi.org/10.1007/s13364-017-0341-1
Thomas RL, Fellowes MDE, Baker PJ (2012) Spatio-temporal variation in predation by urban domestic cats ( Felis catus ) and the acceptability of possible management actions in the UK. 7:20–23. doi: https://doi.org/10.1371/journal.pone.0049369
Tschanz B, Hegglin D, Gloor S, Bontadina F (2011) Hunters and non-hunters: skewed predation rate by domestic cats in a rural village. Eur J Wildl Res 57:597–602. https://doi.org/10.1007/s10344-010-0470-1
United Naions, Department of Economic and Social Affairs PD (2019) World Urbanization Prospects: The 2018 Revision (ST/ESA/SER.A/420)
van Heezik Y, Smyth A, Adams A, Gordon J (2010) Do domestic cats impose an unsustainable harvest on urban bird populations? Biol Conserv 143:121–130. https://doi.org/10.1016/j.biocon.2009.09.013
Vlasseva A, Chassovnikarova T, Atanassov N (2017) Autumn-winter diet and food niche overlap between red fox (Vulpes vulpes L., 1758) and golden jackal (Canis aureus L., 1758) in two regions in Bulgaria. Acta Zool Bulg 69:217–220
Wang Y, Naumann U, Eddelbuettel D, et al (2016) Statistical methods for analysing multivariate abundance data. Package ‘mvabund’. R Proj. Stat. Comput. Vienna, Austria
Woinarski JCZ, South SL, Drummond P et al (2017) The diet of the feral cat (Felis catus), red fox (Vulpes vulpes) and dog (Canis familiaris) over a three-year period at Witchelina reserve, in arid South Australia. Aust Mammal 40:204–213. https://doi.org/10.1071/AM17033
Woods M, Mcdonald RA, Phen STE et al (2003) Predation of wildlife by domestic cats Felis catus in Great Britain. Mamm Rev 33:174–188. https://doi.org/10.1046/j.1365-2907.2003.00017.x
Acknowledgements
This work was supported by a Region Ile-de-France PhD scholarship for IC and by the Chair “Modélisation Mathématique et Biodiversité” of VEOLIA-Ecole Polytechnique-MnHn-FX and the Labex BASC through flagship projects for DZL. Raquel Monclús kindly reviewed this manuscript. We also thank all the students who participated in the field and laboratory work.
Author information
Authors and Affiliations
Contributions
IC collected and analysed the data and led the writing of the manuscript. DZL contributed to performing the diet descriptors. EB conceived the ideas and designed the methodology. All authors contributed to the writing and gave their final approval for publication.
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 58 kb)
Rights and permissions
About this article
Cite this article
Castañeda, I., Zarzoso-Lacoste, D. & Bonnaud, E. Feeding behaviour of red fox and domestic cat populations in suburban areas in the south of Paris. Urban Ecosyst 23, 731–743 (2020). https://doi.org/10.1007/s11252-020-00948-w
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-020-00948-w