Abstract
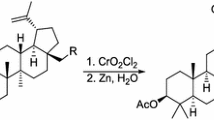
We optimized the process of extraction and purification of betulin from birch bark and modified preparative syntheses of the simplest derivatives of betulin, namely, betulonic acid, betulonic aldehyde, and 3-aminobetulinic acid, as a platform for subsequent modifications.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M. Chudzik, I. Korzonek-Szlacheta, W. Król, Molecules, 2015, 20, 1610; DOI: https://doi.org/10.3390/molecules20011610.
D.-M. Zhang, H.-G. Xu, L. Wang, Y.-J. Li, P.-H. Sun, X.-M. Wu, G.-J. Wang, W.-M. Chen, W.-C. Ye, Med. Res. Rev., 2015, 35, 1127; DOI: https://doi.org/10.1002/med.21353.
G. A. Tolstikov, O. B. Flekhter, E. E. Shults, L. A. Baltina, A. G. Tolstikov, Khimiya v interesakh ustoychivogo razvitiya [Chem. Sustain. Dev.], 2005, 13, 1 (in Russian).
O. A. Vorob’eva, D. S. Malygina, E. V. Grubova, N. B. Mel’nikova, Khimiya rast. syr’ya [Chem. Plant Raw Mater.], 2019, No. 4, 407; DOI: https://doi.org/10.14258/jcprm.2019045419 (in Russian).
P. Yogeeswari, D. Sriram, Curr. Med. Chem., 2005, 12, 657; DOI: https://doi.org/10.2174/0929867053202214.
C.-C. Wen,, Y.-H. Kuo, J.-T. Jan, P.-H. Liang, S.-Y. Wang, H.-G. Liu, C.-K. Lee, X.S.-T. Chang, C.-J. Kuo, S.-S. Lee, C.-C. Hou, P.-W. Hsiao, S.-C. Chien, L.-F. Shyur, N.-S. Yang, J. Med. Chem., 2007, 50, 4087; DOI: https://doi.org/10.1021/jm070295s.
O. B. Flekhter, L. T. Karachurina, L. R. Nigmatullina, T. A. Sapozhnikova, L. A. Baltina, F. S. Zarudii, F. Z. Galin, L. V. Spirikhin, G. A. Tolstikov, O. A. Plyasunova, A. G. Pokrovskii, Russ. J. Bioorg. Chem., 2002, 28, 494.
O. B. Kazakova, G. V. Giniyatullina, G. A. Tolstikov, N. I. Medvedeva, T. M. Utkina, O. L. Kartashova, Russ. J. Bioorgan. Chem., 2010, 36, 383; DOI: https://doi.org/10.1134/S1068162010030155.
G. V. Giniyatyllina, I. E. Smirnova, O. B. Kazakova, N. P. Yavorskaya, I. S. Golubeva, O. S. Zhukova, R. B. Pugacheva, G. N. Apryshko, V. V. Poroikov, Med. Chem. Res., 2015, 24, 3423; DOI: https://doi.org/10.1007/s00044-015-1392-y.
G. V. Giniyatyllina, O. B. Kazakova, I. P. Baikova, E. Y. Yamansarov, I. A. Osterman, E. S. Komarova, D. A. Skvortsov, I. V. Saltikova, A. G. Majouga, Y. A. Ivanenkov, Nat. Prod. Commun., 2019, 1; DOI: https://doi.org/10.1177/1934578X19860670.
N. Melnikova, I. Burlova, T. Kiseleva, I. Klabukova, M. Gulenova, A. Kislitsin, V. Vasin, B. Tanaseichuk, Molecules, 2012, 17, 11849; DOI: https://doi.org/10.3390/molecules171011849.
E. Chrobak, E. Michalik, J. Kusz, M. Nowak, S. Boryczka, Acta Crystallogr., Sect. C: Struct. Chem., 2014, 70, 847; DOI: https://doi.org/10.1107/S2053229614017379.
C.-W. Cui, Y. He, J. Wang, W. Gao, T. Liu, M. Qin, X. Wang, C. Gao, Y. Wang, M.-Y. Liu, Z. Yi, W.-W. Qiu, Eur. J. Med. Chem., 2015, 95, 240; DOI: https://doi.org/10.1016/j.ejmech.2015.03.048.
S. Xiao, Z. Tian, Y. Wang, L. Si, L. Zhang, D. Zhou, Med. Res. Rev., 2018, 38, 951; DOI: https://doi.org/10.1002/med.21484.
K. K. Babievsky, Yu. A. Davidovich, L. V. Snegur, K. A. Kochetkov, Russ. Chem. Bull., 2019, 68, 1944; DOI: https://doi.org/10.1007/s11172-019-2651-8.
A. Ç. Karagöz, M. Leidenberger, F. Hahn, F. Hamoel, O. Friedrich, M. Marschall, B. Kappes, S. B. Tsogoeva, Bioorg. Med. Chem., 2019, 27, 110; DOI: https://doi.org/10.1016/j.bmc.2018.11.018.
M. S. Denisov, Yu. A. Beloglazova, Biomed. Chem.: Res. Methods, 2020, 3, No. 2, Art. ID e00127; DOI: https://doi.org/10.18097/BMCRM00127.
V. A. Glushkov, D. A. Shemyakina, N. K. Zhukova, L. V. Pavlogradskaya, M. V. Dmitriev, D. V. Eroshenko, A. R. Galeev, I. G. Mokrushin, Russ. J. Org. Chem., 2019, 55, 1690; DOI: https://doi.org/10.1134/S1070428019110083.
S. A. Kuznetsova, G. P. Skvortsova, Y. N. Maliar, E. S. Skurydina, O. F. Veselova, Russ. J. Bioorg. Chem., 2014, 40, 742; DOI: https://doi.org/10.1134/S1068162014070073.
Pat. RF 2270202 C1 (in Russian).
E. N. Koptelova, N. A. Kutakova, S. I. Tret’yakov, Khimiya rast. syr’ya [Chem. Plant Raw Mater.], 2013, No. 4, 159; DOI: https://doi.org/10.14258/jcprm.1304159 (in Russian).
V. V. Zhuk, V. A. Yanovsky, O. B. Sambueva, A. A. Bakibaev, Khimiya rast. syr’ya [Chem. Plant Raw Mater.], 2014, No. 3, 247; DOI: https://doi.org/10.14258/jcprm.1403247 (in Russian).
B. N. Kuznetsov, Khimiya rast. syr’ya [Chem. Plant Raw Mater.], 1998, No. 1, 5 (in Russian).
N. Miura, Y. Matsumoto, S. Miyairi, S. Nishiyama, A. Naganuma, Mol. Pharmacol., 1999, 56, 1324.
J. Xu, Z. Li, J. Luo, F. Yang, T. Liu, M. Liu, W.-W. Qiu, J. Tang, J. Med. Chem., 2012, 55, 3122.
L. B. Shon, A. P. Kaplun, A. A. Shpilevsky, Yu. E. Andiya-Pravdivy, S. G. Alekseeva, V. B. Grigor’ev, V. I. Shvets, Bioorg. Khim. [Russ. J. Bioorg. Chem.], 1998, 24, 787 (in Russian).
T. S. Khlebnicova, Y. A. Piven, A. V. Baranovsky, F. A. Lakhvich, S. V. Shishkina, D. Zicāne, Z. Tetere, I. Rāviņa, V. Kumpiņš, I. Rijkure, I. Mieriņa, U. Peipiņš, M. Turks, Steroids, 2017, 117, 77; DOI: https://doi.org/10.1016/j.steroids.2016.08.002.
T. S. Khlebnicova, Y. A. Piven, F. A. Lakhvich, I. V. Sorokina, T. S. Frolova, D. S. Baev, T. G. Tolstikova, Anti-Inflammatory Anti-Allergy Agents Med. Chem., 2020, 19, 254; DOI: https://doi.org/10.2174/1871523018666190426152049.
N. B. Melnikova, D. S. Malygina, O. A. Vorobyeva, A. G. Solovyeva, K. L. Belyaeva, D. V. Orekhov, A. V. Knyazev, Russ. Chem. Bull., 2021, 70, 289; DOI: https://doi.org/10.1007/s11172-021-3084-8.
D. V. Ponomaryov, L. R. Grigor’eva, A. V. Nemtarev, O. V. Tsepaeva, V. F. Mironov, I. S. Antipin, Russ. Chem. Bull., 2020, 69, 487; DOI: https://doi.org/10.1007/s11172-020-2787-6.
M. S. Denisov, V. A. Glushkov, Russ. Chem. Bull., 2020, 69, 2013; DOI: https://doi.org/10.1007/s11172-020-2993-2.
O. V. Tsepaeva, A. V. Nemtarev, L. R. Grigor’eva, V. F. Mironov, Russ. Chem. Bull., 2021, 70, 179; DOI: https://doi.org/10.1007/s11172-021-3074-x.
J. L. C. Sousa, C. S. R. Freire, A. J. D. Silvestre, A. M. S. Silva, Molecules, 2019, 24, 355; DOI: https://doi.org/10.3390/molecules24020355.
D. S. H. L. Kim, J. M. Pezzuto, E. Pisha, Bioorg. Med. Chem. Lett., 1998, 8, 1707; DOI: https://doi.org/10.1016/S0960-894X(98)00295-9.
Funding
This work was financially supported by the Ministry of Higher Education and Science of the Russian Federation (Russian state Assignment No. 075-00697-22-00) using the equipment of the Center for Molecular Structure Studies at the A. N. Nesmeyanov Institute of Organoelement Compounds of the Russian Academy of Sciences. M. Kaverin is grateful to Dr. Sci. (Chem.) K. K. Babievsky for helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 10, pp. 2236–2240, October, 2022.
No human or animal subjects were used in this research.
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Kaverin, M.V., Morozova, P.A. & Snegur, L.V. Betulin, betulonic acid, 3-aminobetulinic acid. Improved extraction and preparative syntheses of derivatives. Russ Chem Bull 71, 2236–2240 (2022). https://doi.org/10.1007/s11172-022-3651-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-022-3651-7