Abstract
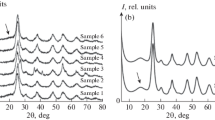
This work examines the specific features of interaction of betulin diphosphate (BDP), exhibiting antitumor and wound-healing properties, with zinc cations during its immobilization on ZnO nanoparticles. The properties of Langmuir and transferred BDP monolayers from an aqueous subphase of zinc sulfate onto the surface of a solid substrate (CaF2, quartz) by the Langmuir—Schaefer method are investigated using IR and UV spectroscopy. It is shown that there is a twofold increase of the molecular area in the immobilized layers, while the compressibility modulus decreases by a factor of 1.5. Zinc oxide nanoparticles with immobilized BDP 10–20 nm in size (surface concentration of BDP is 100 mg g−1) retain the original hexagonal wurtzite structure. The efficiency of betulin diphosphate immobilized on the surface of zinc oxide nanoparticles is demonstrated in in vivo experiments for burn wounds in rats.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
G. A. Tolstikov, O. B. Flekhter, E. E. Shultz, L. A. Baltina, A. G. Tolstikov, Chemistry for Sustainable Development, 2005, 13, 1.
D.-M. Zhang, H.-G. Xu, L. Wang, Y.-J. Li, P.-H. Sun, X.-M. Wu, G.-J. Wang, W.-M. Chen, W.-C. Ye, Med. Res. Rev., 2015, 35, 1127.
M. Chudzik, I. Korzonek-Szlacheta, W. Król, Molecules, 2015, 20, 1610.
A. A. Damle, Y. P. Pawar, A. A. Narkar, Indian J. Exp. Biol., 2013, 51, 485.
A. Spivak, R. Khalitova, D. Nedopekina, L. Dzhemileva, M. Yunusbaeva, V. Odinokov, V. D’yakonov, U. Dzhemilev, Molecules, 2018, 23, 3000.
F. B. Mullauer, L. van Bloois, J. B. Daalhuisen, M. S. Ten Brink, G. Storm, J. P. Medema, R. M. Schiffelers, J. H. Kessler, Anticancer Drugs, 2011, 22, 223.
A. Halder, D. Shukla, S. Das, P. Roy, A. Mukherjee, B. Saha, Cytokine, 2018, 110, 412.
E. A. Lomkova, P. Chytil, O. Janoušková, T. Mueller, H. Lucas, S. K. Filippov, O. Trhlíková, P. A. Aleshunin, Y. A. Skorik, K. Ulbrich, T. Etrych, Biomacromolecules, 2016, 17, 3493.
S. K. Filippov, N. S. Vishnevetskaya, B.-J. Niebuur, E. Koziolová, E. A. Lomkova, P. Chytil, T. Etrych, C. M. Papadakis, Colloid Polym. Sci., 2017, 295, 1313.
O. V. Popova, V. V. Sursyakova, G. V. Burmakina, V. A. Levdansky, A. I. Rubaylo, Dokl. Chem., 2015, 461, 67.
A. Falamaş, S. C. Pînzaru, V. Chiş, C. Dehelean, J. Mol. Struct., 2011, 993, 297.
C. Şoica, C. Dehelean, C. Danciu, H. Wang, G. Wenz, R. Ambrus, F. Bojin, M. Anghel, Int. J. Mol. Sci., 2012, 13, 14992.
H. M. Wang, C. Soica, G. Wenz, Nat. Prod. Commun., 2012, 7, 289.
C. A. Dehelean, C. Soica, C. Peev, S. Ciurlea, S. Feflea, P. Kasa, Farmacia, 2009, 59, 51.
U. Pradere, E. C. Garnier-Amblard, S. J. Coats, F. Amblard, R. F. Schinazi, Chem. Rev., 2014, 114, 9154.
D. V. Ponomaryov, L. R. Grigor’eva, A. V. Nemtarev, O. V. Tsepaeva, V. F. Mironov, I. S. Antipin, Russ. Chem. Bull., 2020, 69, 487.
H. X. Hao, J. K. Wang, Y. L. Wang, J. Chem. Eng. Data, 2004, 49, 1697.
O. Vorobyova, O. Deryabina, D. Malygina, N. Plotnikova, A. Solovyeva, K. Belyaeva, N. Melnikova, Sci. Pharm., 2018, 86, 17.
N. B. Melnikova, D. S. Malygina, I. N. Klabukova, D. V. Belov, V. A. Vasin, P. S. Petrov, A. V. Knyazev, A. V. Markin, Molecules, 2018, 23(5), 1175.
N. B. Melnikova, D. S. Malygina, O. N. Solovyeva, O. E. Zhiltsova, V. A. Vasin, P. S. Petrov, I. N. Klabukova, Int. J. Pharm. Pharm. Sci., 2018, 10, 87.
K.-N. Yu, T.-J. Yoon, A. Minai-Tehrani, J.-E. Kim, S. J. Park, M. S. Jeong, S.-W. Ha, J.-K. Lee, J. S. Kim, M.-H. Cho, Toxicol. in Vitro, 2013, 27, 1187.
B. Ahmed, B. Solanki, A. Zaidi, M. S. Khan, J. Musarrat, Toxicol. Res. (Camb.), 2019, 8, 246.
L. Z. Flores-López, H. Espinoza-Gómez, R. J. Somanathan, J. Appl. Toxicol., 2019, 39(1), 16.
C. Danciu, I. Pinzaru, D. Coricovac, F. Andrica, I. Sizemore, C. Dehelean, F. Baderca, V. Lazureanu, C. Soica, M. Mioc, H. Radeke, Eur. J. Pharm. Biopharm., 2019, 134, 1.
M. Mioc, I.Z. Pavel, R. Ghiulai, D. E. Coricovac, C. Farcaş, C. V. Mihali, C. Oprean, V. Serafim, R. A. Popovici, C. A. Dehelean, M. I. Shtilman, A. M. Tsatsakis, C. Şoica, Front. Pharmacol., 2018, 9, 429.
B. Wang, W. Feng, M. Wang, Y. Gu, M. Zhu, H. Ouyang, J. Shi, F. Zhang, Y. Zhao, Z. Chai, H. Wang, J. Wang, J. Nanopart. Res., 2008, 10, 263.
V. Pokharkar, S. Dhar, D. Bhumkar, V. Mali, S. Bodhankar, B. L. Prasad, J. Biomed. Nanotechnol., 2009, 5, 233.
N. Thamer, L. Almashhedy, Int. J. Phytopharm., 2016, 7, 13.
M. Martínez-Carmona, Y. Gun’ko, M. Vallet-Regí, Nano-materials, 2018, 8, 268.
T. Jin, D. Sun, J. Y. Su, H. Zhang, H. J. Sue, J. Food. Sci., 2009, 74, M46.
J.-S. Kim, B.-H. Kang, H.-M. Jeong, S.-W. Kim, B. Xu, Curr. Appl. Phys., 2018, 18, 681.
D. Bera, L. Qian, S. Sabui, S. Santra, P. H. Holloway, Opt. Mater., 2008, 30, 1233.
M. Uchiyama, M. Mihara, Anal. Biochem., 1978, 86, 271.
T. V. Sirota, Biochemistry (Moscow), Suppl. Ser. B: Biomed. Chem., 2011, 5, 253.
H. Aebi, Catalase in Vitro, in Methods in Enzymology, Ed. L. Packer, Academic Press, Cambridge, MA, USA, 1984, Vol. 105, pp. 121.
G. V. Sibgatullina, L. R. Khaertdinova, E. A. Gumerova, A. N. Akulov, Yu. A. Kostyukova, N. A. Nikonorova, N. I. Rumyantseva, Metody opredeleniya redoks-statusa kul’tiviruyemykh kletok rasteniy. Uch.-metod. posob. [Methods for Determining the Redox State of Cultured Plant Cells. Ed. method. manual.], Kazan. (Privolzhskii) fed. un-t, Kazan, 2011, 61 pp. (in Russian).
R. U. Khabriev, Rukovodstvo po eksperimental’nomu (doklinicheskomu) izucheniyu novykh farmakologicheskikh veshchestv [Guidelines for Experimental (Pre-clinical) Study of New Pharmacological Substances], Meditsina, Moscow, 2005, 832 pp. (in Russian).
R. Gennis, Biomembranes: Molecular Structure and Functions, Springer-Verlag, New York, 1989.
M. Broniatowski, M. Flasiński, P. Wydro, Langmuir, 2012, 28, 5201.
D. Henrich, Cryst. Growth Des., 2017, 17, 5764.
M. Broniatowski, J. Colloid Interface Sci., 2012, 381, 116.
Y. Chen, R. Sun, B. Wang, J. Colloid Interface Sci., 2011, 353, 294.
P. Claesson, A. M. Carmona-Ribeiro, K. Kurihara, J. Phys. Chem., 1989, 93, 917.
M. Miller, M. Chu, B. Lin, M. Meron, P. Dutta, Langmuir, 2015, 32, 73.
C. Yi. Kyunghee, Z. Horvolgyi, J. H. Fendler, J. Phys. Chem., 1994, 98, 3872.
G. Brezesinski, D. Vollhardt, Chem. Phys. Chem., 2008, 9, 1670.
E. V. Ermakova, I. N. Meshkov, Y. Y. Enakieva, A. I. Zvyagina, A. A. Ezhov, A. A. Mikhaylov, Y. G. Gorbunova, V. V. Chernyshev, M. A. Kalinina, V. V. Arslanov, Surf. Sci., 2017, 660, 39.
I. I. Shepeleva, A. V. Shokurov, N. V. Konovalova, V. V. Arslanov, P. A. Panchenko, S. L. Selektor, Russ. Chem. Bull., 2018, 67, 2159.
U. P. Rodrigues-Filho, S. Vaz, Jr., M. P. Felicissimo, M. Scarpellini, D. R. Cardoso, R. C. J. Vinhas, R. Landers, J. F. Schneider, B. R. McGarvey, M. L. Andersen, L. H. Skibsted, J. Inorg. Biochem., 2005, 99, 1973.
N. A. Vorob’yeva, Ph. D. Thesis (Chem.), Lomonosov Moscow State University, Moscow, 2015, 180 pp. (in Russian).
C. Wöll, Prog. Surf. Sci., 2007, 82, 55.
P. Bindu, S. Thomas, J. Theor. Appl. Phys., 2014, 8, 123.
B. Efafi, S. Ghamsari, M. A. Aberoumand, M. H. Majles Ara, H. Hojati Rad, Mater. Lett., 2013, 111, 78.
A. V. Blinov, A. A. Kravtsov, M. A. Yasnaya, A. Yu. Rusanov, E. V. Momot, Mezhvuz. sb. “Fiziko-khimicheskie aspekty izucheniya klasterov, nanostruktur inanomaterialov” [Interuniv. Coll. Vol. “Physicochemical Aspects of the Study of Clusters, Nano-structures, and Nanomaterials”], 2015, 7, 123 pp. (in Russian).
T. Thirugnanam, J. Nanomater., 2013, 6, 1
J. Oliva, L. Diaz-Torres, A. Torres-Castro, P. Salas, L. Perez-Mayen, E. De la Rosa, Opt. Mater. Express, 2015, 5, 1109.
N. A. Salahuddin, M. El-Kemary, E. M. Ibrahim, Nanoscience and Nanotechnology, 2015, 5, 82.
A. S. Lanje, S. K. Sharma, R. S. Ningthoujam, J.-S. Ahn, B. P. Ramchandra, Adv. Powder Technol., 2013, 24, 331.
L. K. Jangir, Y. Kumari, A. Kumar, M. Kumar, K. Awasthi, Mater. Chem. Front., 2017, 1, 1413.
B. Efafi, M. H. A. Majles, S. S. Mousavi, J. Lumin., 2016, 178, 384.
M. Vafee, M. S. Ghamsari, S. Radiman, J. Lumin., 2011, 131, 155.
M. Salavati-Niasari, F. Davar, A. Khansari, J. Alloys Compd., 2011, 509, 61.
Z. Chen, X. X. Li, G. Du, N. Chen, A. Y. M. Suen, J. Lumin., 2011, 131, 2072.
N. Abdulrahman, Z. Nssaif, Tikrit J. Pure Sci., 2016, 21, 49.
H. Hemeg, Int. J. Nanomedicine, 2017, 12, 8211.
M. J. Hajipour, K. M. Fromm, A. A. Ashkarran, D. J. de Aberasturi, I. R. de Larramendi, T. Rojo, V. Serpooshan, W. J. Parak, M. Mahmoudi, Trends Biotechnol., 2012, 30, 499.
C. Han, N. Romero, S. Fischer, J. Dookran, A. Berger, A. Doiron, Nanotechnol. Rev., 2017, 6(5), 383; DOI: https://doi.org/10.1515/ntrev-2016-0054.
Author information
Authors and Affiliations
Corresponding authors
Additional information
This work was financially supported by the Ministry of Higher Education and Science of the Russian Federation within the basic part of the Russian state assignment (Project No. 0729-2020-0039).
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 2, pp. 289–300, February, 2021.
Rights and permissions
About this article
Cite this article
Melnikova, N.B., Malygina, D.S., Vorobyova, O.A. et al. Properties of Langmuir and immobilized layers of betulin diphosphate on aqueous solutions of zinc sulfate and on the surface of zinc oxide nanoparticles. Russ Chem Bull 70, 289–300 (2021). https://doi.org/10.1007/s11172-021-3084-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-021-3084-8