Abstract
A green approach for the promotion of the synthesis of Hantzsch products using poly(vinylpyrrolidinium) perchlorate {[PVPH]ClO4} as a new modified polymeric catalyst in the absence of solvent is reported. The title catalyst is easily prepared and characterized using a variety of techniques including infrared spectra, X-ray diffraction, thermal gravimetric analysis, scanning electron microscopy, pH analysis, and Hammett acidity function. Some of the advantages of this novel synthetic method are: easy preparation of the catalyst, short reaction times, high to excellent yields of the products, simple and easy work-up procedure, and reusability of the catalyst.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Polyhydroquinoline derivatives are among the most important classes of organic compounds, which show a wide range of biological and pharmaceutical properties [1–13]. In addition, they can be used as reducing agents for the direct reductive amination of aldehydes and ketones [14]. These significant properties attracted the attention of many organic chemists to the synthesis of these types of compounds. These attempts resulted in the introduction of a variety of reagents for the promotion of the Hantzsch reaction, as a classical method used for this purpose. Some of these reagents are: ceric ammonium nitrate (CAN) [15], Cs2.5H0.5PW12O40 [16], Hf(NPf2)4 [17], Grinding [18], Polymer [19], K7[PW11CoO40] [20], MCM-41 [21], molecular iodine [22], nickel nanoparticle [23], ionic liquids [24], organo-catalyst [25], p-TSA [26], BINOL-phosphoric acid derivatives [27], HClO4·SiO2 [28], HY-zeolite [29], and montmorillonite K-10 [30].
Although, each of these methods have their own advantages, some of them often suffer from one or more disadvantages such as use of stoichiometric amounts of reagents, acidic or basic catalysts, expensive metal precursors, tedious work-up, long reaction times, unsatisfactory product yields, excess of organic solvent, high temperatures, harsh reaction conditions, and difficulty in preparation of the catalyst, which limit the use of these methods. Therefore, it is important to find more convenient methods for the synthesis of polyhydroquinoline derivatives.
Herein and in continuation of our previous report on the preparation and use of the polymer-supported reagents in various organic reactions [31–33] we wish to report the use of poly(vinylpyrrolidone) as a support for HClO4 and the applicability of the prepared reagent in the promotion of the synthesis of the polyhydroquinoline derivatives via the Hantzsch condensation reaction.
Experimental
Chemicals were purchased from Fluka, Merck, and Aldrich chemical companies. Cross-linked poly(vinylpyrrolidone) we purchased it from BASF, the chemical company, Germany. All products were characterized by comparison of their physical constants, IR, and NMR spectroscopy with authentic samples and those reported in the literature.
The FT-IR spectra were run on a VERTEX 70 Bruker company (Germany). Thermogravimetric analyses (TGA) were performed on TG/DTA6300 Sll-Nanotechnology Wide-angle X-ray diffraction (XRD) with measurements performed at room temperature on a Siemens D-500 X-ray diffractometer (Germany), using Ni-filtered Co–Kα radiation (λ = 0.15418 nm). Company (Japan). Samples were heated from 25 to 800 °C at ramp 10 °C min−1 under N2 atmosphere. Scanning election microphotographs (SEM) were obtained on a SEM-Philips XL30. The purity determination of the substrate and reaction monitoring were accompanied by TLC on silica gel polygram SILG/UV 254 plates.
Catalyst preparation
HClO4 (5 mmol, ≅0.43 mL) was added to a suspension of the powdered poly(vinylpyrrolidone) (0.5 g) [crosslinked poly(vinylpyrrolidone) with MW >1,000,000] in 10 mL dry CH2Cl2 over a period of 10 min in an ice bath. After the addition was completed, the mixture was stirred for 2 h and then the mixture was filtered. The solid residue was washed with diethyl ether (10 mL) and dried at 80 °C to afford [PVPH]ClO4 as a white powder (Scheme 1).
General procedure for the synthesis of the Hantzsch polyhydroquinoline derivatives
A mixture of aldehyde (1 mmol), cyclic diketone (1 mmol), β-ketoester (1 mmol), ammonium acetate (2 mmol), and [PVPH]ClO4 (40 mg, 0.09 mmol) was heated at 100 °C in an oil-bath for the appropriate time (Table 2). After completion of the reaction [monitored by TLC: n-hexane: ethyl acetate (4:1)], the reaction mixture was cooled to room temperature and EtOH (5 mL) was added to it and filtered to separate the catalyst. After evaporation of the solvent from the filtrate, the crude solid product was recrystallized from ethanol to give the pure product. The spectral (IR and 1HNMR) data of a new compound is presented below.
Diethyl 3,3′‐(1,3‐phenylene) bis (2,7,7‐trimethyl‐5‐oxo‐1,4,5, 6,7,8‐hexa hydro quinoline‐3‐carboxylate): Yellow solid; Isolated yield (90 %); Melting point: 323–327 °C; IR (KBr): 3295, 3214, 2959, 1692, 1614, 1489, 1379, 1218 cm−1; 1H NMR (400 MHz, DMSO‐d6): 0.8 (s, 3H, CH3)–0.82 (s, 3H, CH3)–1 (s, 3H, CH3)–1.01 (s, 3H, CH3)–1.07 (t, J = 7.2 Hz, 3H,CH3)–1.17 (t, J = 7.2 Hz, 3H, CH3)–1.92 (d, J = 16 Hz, 1H)–1.98 (d, J = 16 Hz, 1H)–2.14 (d, J = 5.2 Hz, 1H)–2.18 (d, J = 5.2 Hz, 1H)–2.23 (d, J = 16 Hz, 1H)–2.25 (s, 3H, CH3)–2.29 (d, J = 16 Hz, 1H)–2.3 (s, 3H, CH3)–2.39 (d, J = 14.4 Hz, 1H)–2.43 (d, J = 16.8 Hz, 1H)–3.9–4.1 (m, 2xCH2)–4.77 (s, 1H)–4.83 (s, 1H)–7 (s,1H) 6.81–7 (m, 3H, ArH), 9.1 (s, 1H, NH)–9.4 (s, 1H, NH).
Results and discussion
Catalyst characterization
IR analysis
The infrared spectra of PVP and [PVPH]ClO4 are shown in Fig. 1. As can be seen, the FT-IR spectra of PVP are considerably changed after modification as [PVPH]ClO4.
In the FT-IR spectrum of the prepared reagent the characteristic absorption bands at 1090 and 624 cm−1 are assigned to asymmetric and symmetric stretching and bending vibration of ClO4, respectively [31]. Furthermore, in this spectrum, the bands at 1429 (C–N) and 650 (N–C=O) cm−1 disappeared and a moderate absorption at 1650 cm,−1 which can be related to the internal imine groups of the pendant rings of the polymer, appeared [33].
XRD analysis
The X-ray diffraction (XRD) patterns of the PVP and [PVPH]ClO4 samples are shown in Fig. 2. On the basis of this figure it can be concluded that incorporation of HClO4 leads to some changes in the diffractogram of PVP. The PVP diffraction exhibits a diffused background pattern with two diffraction halos appearing around 2θ equal to 18 and 25 indicating that the polymer is amorphous [34]. After modification of PVP by HClO4, the first peak (2θ around 18) disappeared, and the broad peak at 2θ around 25 is slightly reduced. These observations imply that the crystalline size of PVP is decreased after reaction with HClO4 [31, 33].
TGA analysis
The thermal stability of PVP and [PVPH]ClO4 was determined by thermogravimetric analysis (TGA) curves, as shown in Fig. 3. The TGA curve of PVP displayed a weight loss below 120 °C, which corresponded to the loss of the physically adsorbed water and bonded H2O within the gallery of PVP. The large proportion of polymer underwent degradation in the range of 350–470 °C. The TGA analysis of [PVPH]ClO4 shows one-stage decomposition, completely different from PVP. The first weight loss, which appeared at <120 °C, was attributed to the loss of moisture contents. The second weight loss, which started from 250 °C, can be a result of the thermal decomposition of the pendant ClO4 groups [31, 33].
SEM analysis
The samples of PVP and [PVPH]ClO4 were also analyzed by scanning electron microscopy (SEM) with various magnifications for determining the particle shape, surface morphology and size distribution, as represented in Fig. 4. These images show that with chemical modification the primary morphology of PVP was completely changed, and the particles were aggregated. It should be noted that after the reaction of PVP with HClO4 the polymer became swollen, which increased the surface area of the catalyst and finally its catalytic activity.
pH analysis
To 25 mL of an aqueous solution of NaCl (1 M) with a primary pH 5.65, [PVPH]ClO4 (0.5 g) was added and the resulting mixture was stirred for 2 h at room temperature after which the pH of the solution decreased to 1.34. This is equal to a loading of 2.28 mmol H+/g of the catalyst [33].
Hammett acidity
The Hammett acidity method is an effective way to strengthen the acidity of an acid in organic solvents, using the UV–Vis technique [33]. The Hammett function is defined as:
The pK (I)aq is the pK a value of aqueous solution of the indicator and [IH+]s and [I]s are the molar concentrations of protonated and unprotonated forms of the indicator in the solvent. According to Lambert–Beer’s Law, the value of [I]s/[IH+]s can be determined and calculated through the UV–Visible spectrum. For this purpose, 4-nitroaniline (pK(I)aq = 0.99) as the basic indicator and CCl4 as the solvent were chosen. As can be seen in Fig. 5, the maximal absorbance of the unprotonated form of the indicator was observed at 330 nm in CCl4. When [PVPH]ClO4 as a catalyst was added to the indicator solution, the absorbance of the unprotonated form of the indicator decreased, which indicated that the indicator was partially in the form of [IH+]. The obtained results are listed in Table 1.
Catalytic activity
On the basis of the above mentioned studies, we anticipated that [PVPH]ClO4 could be used as an efficient catalyst for the promotion of the reactions, which need the use of an acidic catalyst to speed-up. In order to confirm this decision, we started to study the applicability of this polymeric catalyst in the acceleration of the Hantzsch reaction.
At first step, optimization of the reaction conditions was done by studying the reaction of 4-chlorobenzaldehyde with ethylacetoacetate, 5,5-dimethyl-1,3-cyclohexanedione (dimedone), and ammonium acetate in the presence of various amounts of [PVPH]ClO4 in the absence of solvent and also in different solvents such as dichloromethane, acetonitrile, and ethanol under thermal conditions (Table 2). In this study different ratios of substrates were also examined. The optimum amounts were found to be 1:1:1:2:0.09 for 4-chlorobenzaldehyde, ethylacetoacetate, dimedone, ammonium acetate, and the catalyst, respectively (Scheme 2).
After optimization of the reaction conditions and in order to show the general applicability of the method, different types of aldehydes were subjected to the same reaction under the determined conditions. The obtained results showed that in all cases the corresponding polyhydroquinolines were obtained in good to excellent yields in appropriate times (Table 3, entries 1–12). We have also found that the same results were obtained when methylacetoacetate is used in place of ethylacetoacetate (Table 3, entries 17–20). It can be easily seen that in all cases, regardless of the substituent, the reaction gave the products in good to high yields during relatively short reaction times.
To investigate the versatility of the selected method, the reaction of 1,3-cyclohexanedione with various aldehydes was also carried out in the presence of this reagent. Short reaction times and high yields of the products showed that this substrate also reacted well under the selected conditions (Table 3, entries 13–16).
The reaction of dimedone with p-phthaldialdehyde and/or m-phthaldialdehyde in the presence of [PVPH]ClO4 (0.18 mmol) was also successful and the desired bis-products were obtained after short reaction times in high yields (Table 3, entries 21 and 22).
A plausible mechanism of the reaction is shown in Scheme 3. On the basis of this mechanism, the main role of the catalysts is the activation of carbonyl group for nucleophilic attack. This proposed mechanism is found in the literature [36, 37].
Table 4, compares the efficiency of [PVPH]ClO4 with other catalysts in the synthesis of the polyhydroquinoline derivative of 2-chlorobenzaldehyde. In this Table the following points should be considered: (a) Although the same reaction can be carried out in the absence of catalyst, in this situation the reaction time is very long (Table 4, entry 1), and (b) Although the same reaction can be carried out using lower amounts of some of the reported catalysts, the main drawback is that their preparation is difficult and time consuming (Table 4, entries 7, 8), and/or starting material is expensive (Table 4, entries 3–5, 7, 8). On the other hand, easy preparation of the catalyst, use of inexpensive supporter polymer, and lower amounts of [PVPH]ClO4 are the most important advantages of the present method.
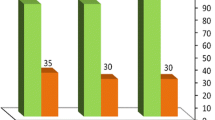
The reusability of the catalyst was checked in the reaction of 4-hydroxybenzaldehyde (Table 3, entry 5) under the optimized reaction conditions. After the separation of the product, the catalyst was washed with EtOH, dried, and reused for the same reaction. This process was carried out over five runs and all reactions led to the desired products with high efficiency (Fig. 6). It should be noted that the pH analysis of the recovered catalyst showed nearly the same loading of H+ as the freshly prepared catalyst. These results suggest that changing the catalyst and/or leaching of the acid species does not occur during the course of the reaction.
Reusability of [PVPH]ClO4 (Table 3, entry 5)
Conclusion
In this paper, [PVPH]ClO4 is prepared using commercially available starting materials and characterized with a variety of techniques. In continuation, this modified polymeric catalyst is used for the synthesis of polyhydroquinoline derivatives in the absence of solvent. Handling of the catalyst is easy, and this method is safer than the other ways, which, in them, the mineral acids like H2SO4 or HClO4, are used as the catalysts. [PVPH]ClO4 is very stable in the presence of moisture compared with some of the other forms of the modified PVP (e.g., [PVP–SO3H]Cl) and even after several days it is not destroyed and its color is not changed at all. Also, this catalyst could be successfully recovered and recycled at least for five runs without significant loss in its activity.
References
T. Godfraid, R. Miller, M. Wibo, Pharmocol. Rev. 38, 321 (1986)
A. Sausins, G. Duburs, Heterocycles 27, 279 (1988)
P.P. Mager, R.A. Coburn, A.J. Solo, D.J. Triggle, H. Rothe, Drug Des. Discov. 28, 273 (1992)
R. Mannhold, B. Jablonka, W. Voigt, K. Schonafinger, K. Schraven, Eur. J. Med. Chem. 27, 229 (1992)
F. Bossert, H. Meyer, E. Wehinger, Angew. Chem. Int. Ed. Engl. 20, 762 (1981)
H. Nakayama, Y. Kasoaka, Heterocycles 42, 901 (1996)
V. Klusa, Drugs Future 20, 135 (1995)
R.G. Bretzel, C.C. Bollen, E. Maester, K.F. Federlin, Am. J. Kidney Dis. 21, 54 (1993)
R.G. Bretzel, C.C. Bollen, E. Maester, K.F. Federlin, Drugs Future 17, 465 (1992)
R. Boer, V. Gekeler, Drugs Future 20, 499 (1995)
Y.L. Chen, K.C. Fang, J.Y. Sheu, S.L. Hsu, C.C. Tzeng, J. Med. Chem. 44, 2374 (2001)
G. Roma, M.D. Braccio, G. Grossi, M. Chia, Eur. J. Med. Chem. 35, 1021 (2000)
M.P. Maguire, K.R. Sheets, K. McVety, A.P. Spada, A. Zilberstein, J. Med. Chem. 37, 2129 (1994)
T. Itoh, K. Nagata, A. Kurihara, M. Miyazaki, A. Ohsawa, Tetrahedron Lett. 43, 3105 (2002)
Sh Ko, Ch-F. Yao, Tetrahedron 62, 7293 (2006)
H. Khabazzadeh, E. Tavakilinejad Kermani, D. Afzali, A. Amiri, A. Jalaladini, Arabian J. Chem. 5, 167 (2012)
M. Hong, C. Cai, W.-B. Yi, J. Fluor Chem. 131, 111 (2010)
S. Kumar, P. Sharma, K.K. Kapoor, M.S. Hundal, Tetrahedron 64, 536 (2008)
A. Dondoni, A. Massi, E. Minghini, V. Bertolasi, Tetrahedron 60, 2311 (2004)
M.M. Heravi, K. Bakhtiari, N.M. Javadi, F.F. Bamoharram, M. Saeedi, H.A. Oskooie, J. Mol. Catal. A Chem. 264, 50 (2007)
L. Nagarapu, M. Dharani Kumari, N.V. Kumari, S. Kantevari, Catal. Commun. 78, 1871 (2007)
S. Ko, M.N.V. Sastry, C. Lin, C.F. Yao, Tetrahedron Lett. 46, 5771 (2005)
S.B. Sapkal, K.F. Shelke, B.B. Shingate, M.S. Shingare, Tetrahedron Lett. 50, 1754 (2009)
S.J. Ji, Z.Q. Jiang, J. Lu, T.P. Loh, Synlett 5, 831 (2004)
A. Kumar, R.A. Maurya, Tetrahedron 63, 1946 (2007)
A. Kumar, R.A. Maurya, Synlett 6, 883 (2008)
C.G. Evans, J.E. Gestwicki, Org. Lett. 11, 2957 (2009)
M. Maheswara, V. Siddaiah, G.L.V. Damu, C.V. Rao, ARKIVOC ii, 201 (2006)
B. Das, B. Ravikanth, R. Ramu, B. Vittal Rao, Chem. Pharm. Bull. 54, 1044 (2006)
G. Song, B. Wang, X. Wu, Y. Kang, L. Yang, Synth. Commun. 35, 2875 (2005)
N.G. Khaligh, F. Shirini, Ultrason. Sonochem. 20, 26 (2013)
F. Shirini, O.G. Jolodar, J. Mol. Catal. A Chem. 356, 61 (2012)
F. Shirini, P.N. Moghadam, S. Moayedi, M. Seddighi, RSC Adv. 4, 38581 (2014)
X.Y. Li, X. Wang, D.G. Yu, S. Ye, Q.K. Kuang, Q.W. Yi, X.Z. Yao, J. Nanomater. 2012, 7 (2012)
A. Zare, F. Abi, A.R. Moosavi-Zare, M.H. Beyzavi, M.A. Zolfigol, J. Mol. Liq. 178, 113 (2013)
A. Khazaei, M.A. Zolfigol, A.R. Moosavi-Zare, J. Afsar, A. Zare, V. Khakyzadeh, M.H. Beyzavi, Chin. J. Catal. 34, 1936 (2013)
M. Tajbakhsh, H. Alinezhad, M. Norouzi, S. Baghery, M. Akbari, J. Mol. Liq. 177, 44 (2013)
S. Rostamnia, F. Pourhassan, Chin. Chem. Lett. 24, 401 (2013)
M. Nasr-Esfahani, S.J. Hoseini, M. Montazerozohori, R. Mehrabi, H. Nasrabadi, J. Mol. Catal. A Chem. 382, 99 (2014)
R. Surasani, D. Kalita, A.V.D. Rao, K. Yarbagi, K.B. Chandrasekhar, J. Fluorine Chem. 135, 91 (2012)
B.P. Bandgar, P.E. More, V.T. Kamble, J.V. Totre, ARKIVOC xv, 1 (2008)
B. Janardhan, B. Rajitha, P.A. Crooks, J. Saudi. Chem. Soc. (2014). doi:10.1016/j.jscs.2014.01.009
Z. Zarnegar, J. Safari, Z. Mansouri-Kafroudi, Catal. Commun. (2014). doi:10.1016/j.catcom.2014.10.023
A. Maleki, M. Kamalzare, M. Aghaei, J. Nanostruct. Chem. (2014). doi:10.1007/s40097-014-0140-z
S. Igder, A.R. Kiasat, M.R. Shushizadeh, Res. Chem. Intermed. (2014). doi:10.1007/s11164-014-1808-1
Acknowledgments
We are thankful to the University of Guilan Research Councils for the partial support of this work.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Abedini, M., Shirini, F. & Mousapour, M. Poly(vinylpyrrolidinium) perchlorate as a new and efficient catalyst for the promotion of the synthesis of polyhydroquinoline derivatives via Hantzsch condensation. Res Chem Intermed 42, 2303–2315 (2016). https://doi.org/10.1007/s11164-015-2150-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-015-2150-y